132 - Transhiatal Esophagectomy Without Thoracotomy
Editors: Shields, Thomas W.; LoCicero, Joseph; Ponn, Ronald B.; Rusch, Valerie W.
Title: General Thoracic Surgery, 6th Edition
Copyright 2005 Lippincott Williams & Wilkins
> Table of Contents > Volume II > The Mediastinum > Section XXV - Anatomy > Chapter 155 - The Thymus
Chapter 155
The Thymus
Thomas W. Shields
EMBRYOLOGY
The thymus in humans primarily arises from the third pharyngeal pouches (thymus III). Thymic tissue may also arise from the fourth pharyngeal pouches (thymus IV), but in humans, these latter primordia may be altogether absent or rudimentary and give rise only to vestigial tissue masses. Patton (1968) noted that thymic IV tissue, when present, usually becomes associated with the thyroid and may ultimately become embedded within its substance.
According to Patton (1968) the thymic primordia appear late in the sixth week of gestation as ventral outgrowths of the third pharyngeal pouches. It is believed that both the ventral pharyngeal pouch III endoderm and the ectoderm of the floor of the branchial furrow from fusion of the second, third, and fourth branchial clefts contribute to the primordial cell mass. Norris (1938) and others postulated that some of the epithelial components of the thymus arise from the ectodermal cells of the aforementioned branchial clefts. von Gaudecker's (1986a) studies support this concept, and he believes the ectodermal cells from the cervical vesicle primarily of the third cleft actively proliferate, migrate, and completely surround the endodermal thymic primordium from the third pharyngeal pouch. Ectodermal cells are thus thought to be mainly the origin of the epithelial cells of the cortex and endodermal cells the source of the medullary epithelial cells, but this is far from settled. Kornstein (1995), Norris (1938), and Weller (1933) each studied the same collection of embryos and reached opposite conclusions. Also, immunohistochemical studies have shown that human thymic epithelial cells at 7 weeks' development all express common antigens. Thus, Kornstein (1995) notes that it is possible that thymic epithelial cells derive from a common stem cell. Divergence occurs later in differentiation of epithelial subtypes, as suggested by the immunohistochemical differences in newborn and adult thymic cells in the subcapsular, cortical, and medullary areas.
Morphologic and immunohistochemical studies done by McFarland (1984), Haynes (1984), De Maagd (1985), and von Gaudecker (1986b) and their associates demonstrated that detectable immunohistochemical differences (expression of distinctive surface antigens) exist between the epithelial cells of the thymic cortex and the thymic medulla. Hirokawa and colleagues (1988) also reported that the localization of various thymic epithelial cell markers in the newborn human thymus reflects possible different phenotypic origins of epithelial cells in the cortex and medulla (Table 155-1). Marino and M ller-Hermelink (1985) and M ller-Hermelink and colleagues (1986) used these immunohistochemical and morphologic differences of the cortical and medullary epithelial cells to classify epithelial thymic tumors (thymomas) (see Chapter 173). Hirokawa and associates (1988) confirmed these differences and noted that most thymic tumors (thymomas) are of cortical epithelial cell origin.
The primordia of the thymus elongate rapidly in the seventh week but retain their connection with the third pouch and remain associated with parathyroid tissue III (Fig. 155-1). The right and left thymic tissue masses move toward the midline just caudal to the thyroid primordium and by the eighth week make contact with each other but do not fuse.
P.2348
They remain separate but connected lobes. The gland migrates caudad and slides under the sternum in front of the great vessels into the mediastinum to lie in contact with the superior portion of the ventral aspect of the pericardium (Fig. 155-2).
Table 155-1. Localization of Various Markers in Epithelial Cells of Newborn Human Thymus | ||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||
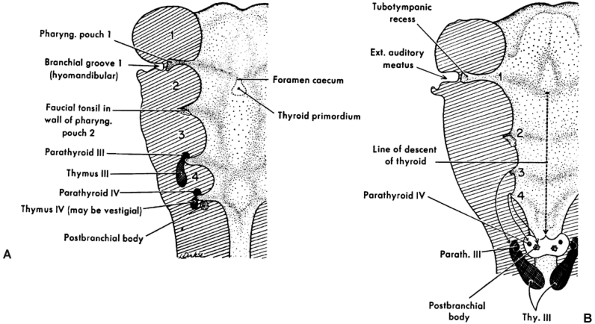 |
Fig. 155-1. Diagrams showing the origin of the pharyngeal derivatives. A. The primary relations of the primordia to the pharyngeal pouches. B. Course of migration of some primordia from their place of origin. Ext., external; Parath., parathyroid; Pharyng., pharyngeal; Thy., thymus. From Patton BM: Human Embryology. 3rd Ed. New York: McGraw-Hill, 1968. With permission. |
GROSS ANATOMY
In the newborn, according to the studies of Boyd (1932) and Kendall and associates (1980), the gland reaches a mean weight of 15 g in the early neonatal period. Steinmann (1986),
P.2349
however, reported that the weight in grams in children from newborn to the age of 1 was 27.3 g with a standard deviation of 16.4 g. The gland reaches its largest relative size in childhood, although it continues to grow until puberty to a mean weight of 30 to 40 g. The recorded weight is less in Steinmann's (1986) studies. A gradual process of involution then occurs throughout adulthood, and the gland is reduced in weight to between 5 and 25 g. A study by Steinmann (1986), as noted, found the mean weight of the thymus in the various age groups to be generally less than the aforementioned mean weights. In all the age groups except for individuals older than 85 years, the mean thymic weight varied between 20 and 28 g, with an average standard deviation of 12.5. In very old individuals, mean weight varies from 18 g in individuals aged 85 to 90 years to 12 g in those 91 to 107 years of age, with a mean standard deviation of 5.4 and 6.9, respectively. Figure 155-3 shows the variations in size of the gland on computed tomography (CT) in an adolescent boy and an adult woman.
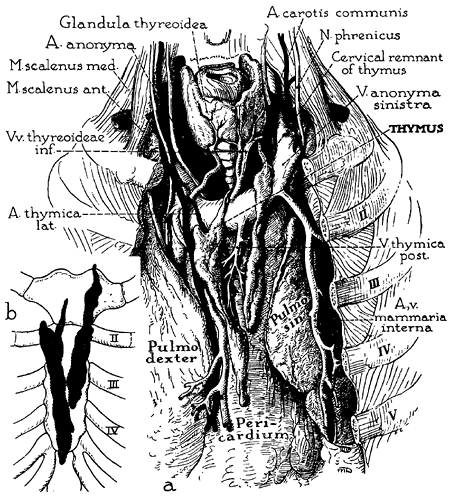 |
Fig. 155-2. Anatomy of thymus in the adult, emphasizing the form, visceral relations, and blood supply. Detailed drawing (a): In this specimen, as is typical in the adult, the thymus is elongated and finger shaped, consists of two parts, and has the gross appearance of paired fatty lobes. The thymic arteries usually derive from the adjacent internal mammary arteries; at least some of the veins terminate in the left innominate vein (thymic bodies are retracted to expose the chief veins). The lobes occupy the sulcus between the anterior borders of the lungs and may extend downward into the cardiac notch. Schematic drawing (b): The sternocostal relations of the thymus in the same specimen. From Bell RH, et al: Form, size, blood supply and relations of the adult thymus. Q Bull Northwestern U Med School 28: 156, 1954. With permission. |
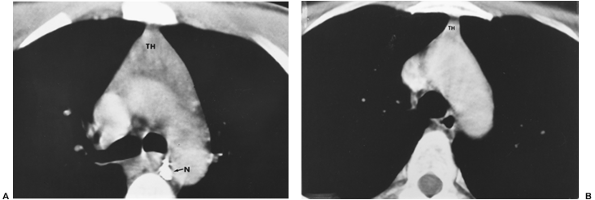 |
Fig. 155-3. A. CT image of a normal thymus (TH) in a 17-year-old boy. A calcified subcarinal lymph node (N) is demonstrated. B. Normal residual thymic tissue (TH) in a 36-year-old woman is demonstrated. |
The thymus, although classically described as having a right and left lobe, may actually be a composite of three or more lobular structures. Nonetheless, the gland tends to maintain its original paired character. The lobules are generally asymmetric (Fig. 155-4). Grossly, the gland has a roughly H-shaped configuration, with extension of the upper poles of either side into the base of the neck, and with a greater or lesser attachment to the thyroid gland by the thyrothymic ligament. The lower poles of each side extend down over the pericardium.
In addition to the variations in size and configuration of the thymic gland, numerous collections of varying amounts of identifiable thymic tissue, both gross and microscopic, may be found as additional mediastinal lobes and islets of tissue outside the capsule of the gland extending from the neck to the diaphragm. In a small series of patients, Masaoka and colleagues (1975) identified a 72% incidence of microscopic collections of thymic tissue in the mediastinal
P.2350
fatty tissues of the anterior compartment outside the thymic capsule. Fukai and colleagues (1991) described a 51.8% incidence of this phenomenon in a series of 27 subjects. Thymic tissue was found in anterior mediastinal fat in 44.4% of specimens and in 7.4% of retrocarinal fat; none was found in preaortic fatty tissues. Ashour (1995), in surgical dissection in 38 myasthenic patients, reported an incidence of 39.5% of ectopic thymic tissue foci. Nineteen sites were located in 15 patients: 12 (63%) were in the neck, and 4 patients (21%) had thymic tissue in fat of the cardiophrenic angle. The variations in location of thymic tissue outside the gland were also well documented by the studies of Jaretzki and Wolff (1988) and Jaretzki (1997). The cervical and mediastinal variations in the location of these thymic tissue foci are schematically illustrated in Fig. 155-5.
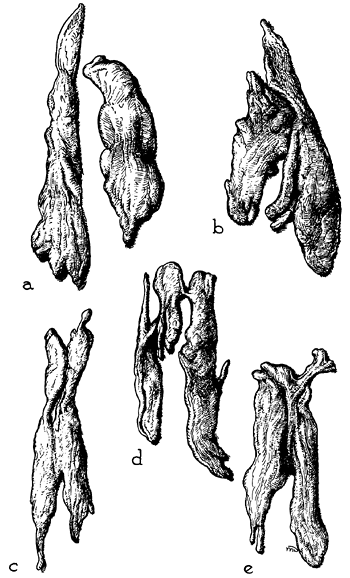 |
Fig. 155-4. Selected examples of adult thymus show variations in form and size (excised glands from laboratory specimens, one-fifth natural size). Usually, the lobes differ in size and shape. They may be wholly separate (a), fused (c) to (e), or merely contiguous (b). In most cases, they come to a tapering extremity cranially, where they lie near the thyroid gland in the neck. From Anson BJ: An Atlas of Human Anatomy. Philadelphia: W.B. Saunders, 1963. With permission. |
Thymic tissue may be found in the neck in about 36% of people, outside the normal cervical extensions of the thymic lobes. Most, if not all, such tissue likely represents arrested descent of thymus III tissues. In the mediastinum, almost all individuals have some thymic tissue beyond the confines of the classic mediastinal lobes. Unencapsulated thymic tissue can be found in the region of the phrenic nerves, behind the innominate vein, in the aortopulmonary window, in the aortocaval groove, and in the anterior and cardiophrenic fatty tissues. The presence of thymic tissue in these locations readily explains the occasional occurrence of epithelial thymic tumors (see Chapter 173) and true thymic cysts (see Chapter 197) in these areas. The significance of these variations in location in the management of myasthenia gravis is discussed in Chapters 179 and Chapters 180.
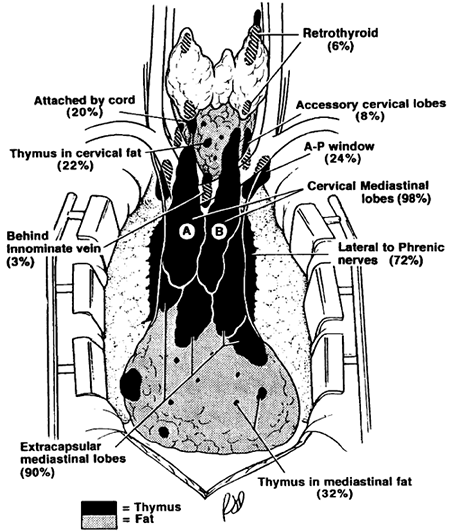 |
Fig. 155-5. Composite anatomy of thymus. The illustration is based on surgical-anatomic studies of 50 transcervical-transsternal maximal thymectomies for myasthenia gravis. Thymic tissue was found outside the classic cervical mediastinal lobes (A and B) in 32% of specimens in the neck and in 98% of specimens in the mediastinum. From Jaretzki A III: Thymectomy for myasthenia gravis: analysis of the controversies regarding technique and results. Neurology 48(Suppl 5):S52, 1997. With permission. |
In the adult, the upper portions of the gland normally lie on the anterior surface of the left innominate vein. On occasion, one or both lobes lie behind this vein instead of in front of it. Jaretzki and Wolff (1988) found both lobes posterior to the vein in 1 of 50 patients (2%) and one lobe in this position in 2 of 50 patients (4%). Other anomalies in position include partial or complete failure of descent of one or both thymic lobes and the presence of thymic tissue at the root of either lung or even within the pulmonary parenchyma. The inferior limits of the adult thymus in relation to the sternocostal levels have been recorded by Bell and colleagues (1954) (Fig. 155-6), and as noted by Jaretzki and Wolff (1988), thymic tissue may be located as low as the cardiophrenic fat pads.
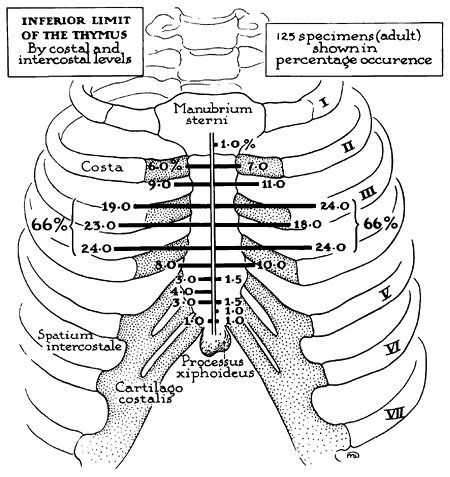 |
Fig. 155-6. Inferior limit of the adult thymus in relation to sternocostal levels. The site of the inferior termination of the thymus is subject to striking variation. However, there is a zone of concentration, or preponderance: In two-thirds of the specimens, this zone lies between the third and fourth sternocostal articulations. From Bell RH, et al: Form, size, blood supply and relations of the adult thymus. Q Bull Northwestern U Med School 28: 156, 1954. With permission. |
P.2351
The arterial blood supply of the thymus (see Fig. 155-2) is mainly from branches of the internal mammary arteries, but the gland may also receive small branches from the inferior thyroid arteries and the pericardiophrenic arteries. Venous drainage may be partially through small veins accompanying these arterial branches. The main drainage, however, is through a centrally located venous trunk on the posterior aspect of the gland that drains into the anterior aspect of the left innominate vein as a single vessel; occasionally, a branch may enter the superior vena cava.
No afferent lymph channels enter the gland, although efferent channels have been identified. These are believed to drain only the capsule and fibrous septa of the gland. These efferent channels terminate at the anterior mediastinal, pulmonary hilar, and internal mammary lymph nodes. Both sympathetic and parasympathetic nerve fibers enter the gland.
HISTOLOGIC FEATURES
Each thymic lobe is covered by a fibrous capsule that extends into the parenchyma as fibrous connective tissue septa, which divide the gland into various-sized lobules ranging from 0.5 to 2.0 mm in size. Each lobule is composed of a cortex and medulla. The medullary areas extend from one lobule into adjacent ones (Fig. 155-7).
Thymic Epithelial Cells
Marchevsky and Kaneko (1984) described the cortex as composed of densely packed lymphocytes (thymocytes), admixed with epithelial and mesenchymal cells. According to von Gaudecker (1986a), two types of epithelial cells are found predominantly in the cortex. One type lines the organ and all perivascular spaces, forming a flattened cytoplasmic lamella. The second type is found predominantly in the outer cortical areas. These cells have euchromatic nuclei and electron-lucent cytoplasm, which frequently encircles lymphatic cells. Bearman and associates (1978), among others, have demonstrated vesicles, tonofilaments, well-developed desmosomes, and secretory granules in both variations of these cortical epithelial cells. In the inner cortex, single, lightly stained epithelial cells may be noted. This cell type is also recognized scattered within the medulla. In the medullary portions of the gland, the epithelial cells vary in appearance with either spindled or round nuclei; the cytoplasm is often sparce. Large epithelial cells with abundant tonofilaments with well-developed rough endoplasmic reticulum and numerous cytoplasmic vesicles are also present. These cells often are arranged in round, keratinized structures known as Hassall's corpuscles (Fig. 155-8). These complex tubular structures are composed of clumps of mature medullary epithelial cells forming concentric layers. Varying degrees of central keratinization or calcification, or both, may be present. Occasionally, these structures may become cystic.
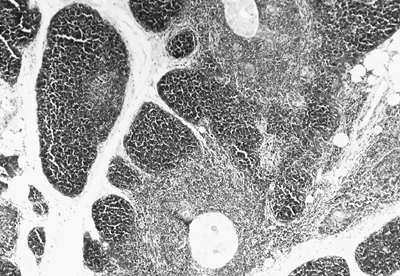 |
Fig. 155-7. Photomicrograph of thymic lobule. Cortex and medullary area separated by septal connective tissue are well seen. |
The medullary cells appear more electron dense than the peripheral (cortical) epithelial cells. However, similar darkly stained epithelial cells may also be seen in the inner cortex. Thus, a true distinct difference is not complete between the epithelial cells of the cortex and medulla. Nonetheless, in reality, the cell types in the two areas are
P.2352
heterogeneous, as noted by their antigenic determinants found in other various cells of the thymus.
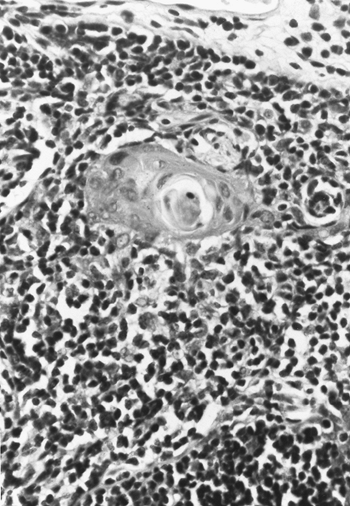 |
Fig. 155-8. Photomicrograph of a typical Hassall's corpuscle. |
Of interest, von Gaudecker (1986a) has theorized that the larger cortical cells with euchromatic round or ovoid nuclei and blunt cytoplasmic protrusions, as well as Hassall's corpuscles, derive from the ectodermal component of the thymic anlagen. Other epithelial cells, found mainly in the medulla but also in the intercortical areas, have a more electron-dense appearance with long slender processes and heterochromatic nuclei. These cells are believed to be descendants of the endoderm. M ller-Hermelink and associates (1986) agree with the concept of heterogeneity of normal thymic epithelial cells, as do Hirokawa and associates (1988). In support of von Gaudecker's interpretation, these authors note that enzyme histochemical studies indicate a functional heterogeneity among thymic epithelial cells, showing different immunohistochemical phenotypes in the subcortical, inner cortex, and medullary areas, including those of the Hassall's corpuscles. Subcapsular epithelial cells and medullary epithelial cells have similar, but not identical, antigen expression. Both have secretory function, and various thymic hormones have been identified immunohistochemically within cytoplasm of these cells. The inner cortical cells have a different antigenicity and are nonsecretory. Hassall's corpuscles are not believed to be secretory, although Savino and colleagues (1984) localized the presence of thymic hormones in these structures. The antigenic characteristic is that Hassall's corpuscles react strongly to high-molecular-weight keratin, which is a feature of mature epithelial cells. So-called nurse cells are a subset of epithelial cells primarily located in the cortex. A nurse cell may contain up to 200 lymphocytes in its cytoplasm. It is thought that the nurse cells provide a specialized microenvironment for T cell development.
Suster and Rosai (1990) summarized the antigenicity of the four cell types: (a) inner cortical epithelium reacts strongly with TE-3, a murine monoclonal antibody raised against human thymic stroma; (b) subcapsular cortical epithelial cells and medullary epithelial cells react with TE-4 monoclonal antibodies and also react with A2B5, a monoclonal antibody directed against a complex neuronal ganglioside [another marker for both these cell types is that detected with anti-p19, an antibody that defines the structural core protein of the human T cell lymphoma virus (HTLV)]; and (c) inner cortical and subcapsular cortical cells express HLA-DR, but this expression is absent in medullary epihelial cells and Hassall's corpuscles (Table 155-2). Other substances are also expressed by these various cells but are not germane to the present discussion.
Thymic Lymphocytes
Lymphocytes (thymocytes), however, dominate the histologic picture of the gland. Embryologically, by the tenth week, prothymocytes (small lymphoid cells) originating in the fetal liver and bone marrow enter the thymus, According to Haynes and Hale (1998), those from the fetal liver begin
P.2353
to migrate by the seventh week of gestation and those from the bone marrow by the twenty-second week onward. Initially, the cells migrate to the outer cortex and possibly the corticomedullary junction, according to Boyd and Hugo (1991). At the time of entrance into the thymus, the prothymocytes (T lymphocyte precursors) are immature, triple-negative T lymphocytes (CD3+, CD4+, CD8+). The CD number refers to a cluster of differentiation of all surface markers that are negative in immature cells but become positive (immunocompetent) to various monoclonal antiboides during the process of thymic lymphocyte maturation. During the process of maturation that takes place in the thymic epithelial space, the thymocytes become initially CD3+ CD4+CD8+; these T lymphocytes are found mostly in the cortex. As maturation continues and becomes completed, the T lymphocytes have become either CD3+CD4+ or CD3+CD8+. The process of maturation to the mature T lymphocytes of either type is highly complex and is regulated by cytokines, cell adhesion molecules, thymic polypeptide hormones, and cell surface self-recognition antigens especially related to each individual human leukocyte type, as pointed out by Lele and associates (2001) in their review of the thymus in infancy and childhood. The process of maturation is to ensure self tolerance and to permit the recognition and elimination of foreign antigen that enters the body. The process of maturation also, as noted by Res and Spits (1999), is essential for the selection of T lymphocytes that do not recognize self antigens. The CD4+ T lymphocytes become helper cells and are the more common of the two. These cells also promote antibody formation by B lymphocytes. The CD8+ T lymphocytes become suppressor, cytotoxic killer cells. At maturity, the thymocytes located in the subcapsular region of the gland constitute 0.5% to 5% of the total, the cortically located thymocytes 60% to 80%, and the medullary located thymocytes 15% to 20%. Most of the thymic lymphocytes die within the thymic cortex by the process of programmed cell death (apoptosis), and only about 10% of the mature thymocytes reach the bloodstream.
Table 155-2. Epithelial Cells of the Thymus: Antigenic Characteristics | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
After the first year of life, the thymic mass decreases at a steady rate (about 3% per year) until middle age when, according to Steinmann (1986), it is reduced to 1% per year. With aging, the involuting thymus shows a decrease in lymphocytes, Hassall's corpuscles, and other normal elements. Lipid-laden macrophages and adipose cells increase in number, but normal thymic tissue, although atrophic, persists throughout life. The epithelial stromal tissue, particularly the cortex, is replaced by fat, as observed by George and Ritter (1996). The T cell compartment shows a reduction of CD4+ cells, and the aged thymus is impaired in its ability to repopulate the T cells when repletion is required.
Other Cell Types
Suster and Rosai (1990) have enumerated the additional cell types found in the normal thymus (Table 155-3). B lymphocytes may be found grouped as lymphoid follicles or distributed as individual cells. Some authors have suggested that a significant proportion of non-Hodgkin's lymphomas of the B cell type located in the anterior mediastinum may arise from the B cell population within the thymus.
Table 155-3. Additional Cell Types Found in the Normal Thymus | |
|---|---|
|
Neuroendocrine cells are few, but their presence has been postulated as the probable reservoir for the development of carcinoid tumors and other neuroendocrine neoplasms, including calcitonin-positive medullary carcinomas.
Myoid cells are readily demonstrated in the gland. Drenckhahn and co-workers (1979) postulated a potential role of the myoid cells in the pathogenesis of myasthenia gravis. The myoid cells are characterized by an acidophilic cytoplasm that contains cross striations. The aforementioned authors reported that these striations react with antisera to actin and myosin. These myoid cells also react to troponin and to acetylcholine receptor. Rare thymic neoplasms thought to be derived from the myoid cells have been described by Henry (1972) [an unusual thymic tumor with a striated muscle (myoid) component] and by Murakami and colleagues (1984) (a thymic tumor with massive proliferation of myoid cells).
Parker and associates (1999) have recorded the identification of nevus cell aggregates in the thymus. These cells reacted positively to S100 protein but showed no reactivity to keratins, HMB-45, or p53. A low reactivity to Ki67 was observed in some of these aggregates of nevus cells. The significance of the presence of these cells is obscure.
Germ cells are thought not to be present in the normal thymus. Neither Suster nor Rosai (1990) believe that there is any direct evidence that they have actually been demonstrated in a normal thymus.
Occasionally, ectopic parathyroid tissue may be embedded in or adjacent to thymic tissue in the mediastinum (see Chapter 156).
THYMIC FUNCTION
The thymus is essential for the development of cellular immunity. The origin of thymic lymphocytes is, as noted, extrathymic from the bone marrow in the embryo. These
P.2354
prothymocyte lymphocytes enter the thymus, proliferate, gain their cellular immunocompetency, and mature into T lymphocytes, the majority of which are inducer cells (C3+C4+) and a minority of which are suppressor, cytotoxic cells (C3+C8+). Humoral immunity, mediated through the elaboration of immunoglobulins by B lymphocytes, originates in humans by some mammalian equivalent of the avian bursa of Fabricius. Although the origin of these cells is thymus independent, an interaction between T cells and B cells aids in the production of immunoglobulins.
The proper function of the immune system depends on the normal development of cellular and humoral components. The thymus-dependent system consists principally of the thymus and a circulating pool of small T lymphocytes. These lymphocytes act as recognition agents capable of discerning self-antigens from nonself-antigens and producing cell-mediated immune reactions, as are seen in delayed hypersensitivity and allograft reactions. The bursa-dependent system consists of the bursa (or its mammalian equivalent, probably Peyer's patches of small bowel), lymphoid follicles, and plasma cells. This system is responsible for the production of immunoglobulins (IgA, IgG, and IgM) and specific antibodies. Both systems direct the differentiation of lymphoid precursor or stem cells into immunologically competent cells capable of reacting to antigens. Any disturbance in the development of either system can be expected to lead to one of the various immunologic deficiency syndromes. Significantly, some of these syndromes have been associated with various neoplasms of the thymus as well as with congenital thymic hypoplasias and agenesis. Much of the understanding of these conditions derives from studies of thymus-related immune deficiency syndromes of infancy, such as that by Good and Gabrielsen (1964). Knowledge gained from experiments in animals is beginning to lead to therapeutic applications. Indeed, the thymus has been implicated in certain immunologic deficiency states as well as in certain hematologic, endocrinologic, autoimmune, collagen, and neuromuscular defects, as well as in neoplasia. The mechanisms, nature, and extent of thymic involvement in these conditions remain obscure.
Of particular interest to surgeons are the immunologic consequences of thymectomy because extirpation is the only commonly used thymic operation. Although thymectomy in certain species, particularly neonatal thymectomy, is often associated with significant alterations in immunity, this relationship has not been observed in either adult or pediatric patients. Although lymphocyte count and tests of immunologic capacity may be diminished by thymectomy in humans, as Adner and associates (1966) reported, no particular clinical problems have developed to correlate with these laboratory observations. With the exception of the management of thymic neoplasms and the empiric management of myasthenia gravis, no clear indications for thymectomy exist in clinical practice at present.
THYMIC HYPERPLASIA
Changes in the lymphatic content and structure of the thymus are seen to accompany many autoimmune states. Levine and Rosai (1978) suggested that thymic hyperplasia should be separated into two categories based on gross and histologic criteria. The first is lymphoid or follicular hyperplasia, characterized by the presence of lymphoid follicles with activated germinal centers (Fig. 155-9). The second is true hyperplasia, characterized by an increase in the size and weight of the thymus gland, which retains a normal or nearly normal morphology for the patient's age and is not accompanied by lymphoid follicles with activated germinal centers.
Lymphoid or Follicular Hyperplasia
The thymus may be of normal or abnormal size and weight. Lymphoid or follicular hyperplasia is frequently associated with autoimmune diseases. It is estimated that 60%
P.2355
to 90% of myasthenic patients who do not have a thymoma have histologically demonstrable follicular hyperplasia of the thymus gland.
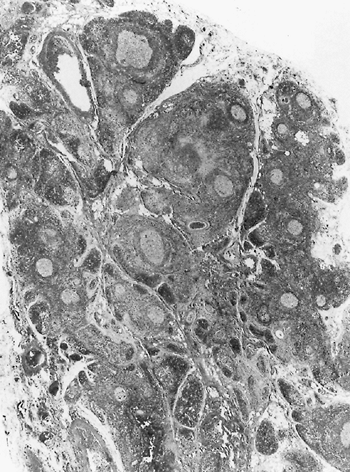 |
Fig. 155-9. Photomicrograph of follicular hyperplasia of the thymus gland in a patient with myasthenia gravis. Note prominent activated germinal centers. From Cove H: The mediastinum. In: Coulson SF (ed): Surgical Pathology. 2nd Ed. Philadelphia: Lippincott, 1988. With permission. |
True Thymic Hyperplasia
Two types of true thymic hyperplasia are seen: (a) idiopathic true hyperplasia with marked to massive enlargement of the gland with no apparent cause, and (b) rebound thymic hyperplasia in children after treatment of various tumors. The second type occurs particularly after chemotherapy and is occasionally associated with other systemic disease states, especially Graves' disease, or after a period of physical stress (e.g., burns, operations, and other similar events).
Thymic Rebound Hyperplasia
Thymic rebound enlargement has been observed after the treatment of Hodgkin's disease by Shin and Ho (1983), and by Tartas and associates (1985), among others. Cohen and colleagues (1980) noted rebound hyperplasia after the treatment of other tumors in children. It has occurred after burn injuries, as reported by Lee (1979) and Gelfand (1972) and their co-workers. Yulish and Owens (1980) also reported its occurrence during the treatment of primary hypothyroidism in a child.
Thymic hyperplasia is a relatively common feature of Graves' disease and has been reported frequently by numerous investigators, such as Judd and Bueso-Ramos (1990) and Bergman and associates (1982). Balcom and associates (1985) have described rebound hyperplasia in a newborn with Beckwith-Wiedemann syndrome (exophthalmos, macroglossia, and gigantism, often associated with neonatal hypoglycemia). Nomori and co-workers (1990) reported true thymic hyperplasia in an adolescent with non-Hodgkin's lymphoma. It remains unclear whether this relationship was one of cause and effect or only coincidental.
The importance of recognizing the rebound nature of thymic hyperplasia is that once the diagnosis is established, no specific treatment is necessary. This is in marked contrast to the management of idiopathic true (massive) thymic hyperplasia, which, except in rare incidences, requires surgical excision.
Idiopathic True (Massive) Thymic Hyperplasia
True massive thymic hyperplasia is rare: Fewer than 50 cases have been recorded, and some of these may have been of the rebound hyperplasia type. Reviews of the literature by Ricci (1989), Linegar (1993), and Takagi (1995) and their associates, as well as by Kornstein (1995), have one or more cases not listed in an error of omission or listed as an inappropriate inclusion in their tabulations. Thus, the exact number of cases reported is impossible to determine. Nonetheless, features of this condition may be described. The major feature is that there is no discernible cause; most cases occur between the ages of 1 and 15 years. The next common age group is 1 year of age or younger. Rarely does idiopathic massive hyperplasia occur after age 15 years; the oldest patient was a 27-year-old woman reported by Takagi and colleagues (1995). It occurs twice as often in boys as in girls. About 85% of patients are symptomatic. Cough, shortness of breath, respiratory distress, and pulmonary infection are the more common symptoms. Chest pain does occur but is unusual. Lymphocytosis in the peripheral blood is seen in about one fourth of patients. The radiographic findings are of a large anterior mass that extends into both hemithoraces, generally to a greater extent into one than into the other (Fig. 155-10). Biopsy only or diagnostic fine-needle aspiration may be done, as reported by Riazmontazer and Bedayat (1993), but almost all patients are managed by surgical excision of the massive, benign thymic gland. The surgical approach may be accomplished
P.2356
through a median sternotomy, a clamshell incision, or even a unilateral posterolateral thoracotomy, as suggested by Linegar and colleagues (1993). The operation is curative, and no subsequent difficulty has been recorded.
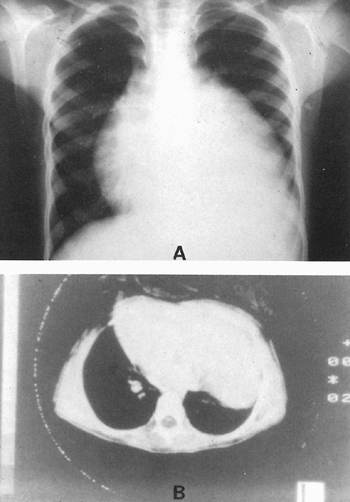 |
Fig. 155-10. True thymic hyperplasia. A. Posteroanterior radiograph of a massive enlargement of the thymus obscuring the cardiac silhouette on the left in a 12-year-old boy presenting with mild dyspnea. B. Computed tomographic scan shows the presence of a large parenchymatous density adjacent to the heart, which has shifted to the right. From Ricci CM, et al: True thymic hyperplasia: a clinicopathological study. Ann Thorac Surg 47:741, 1989. With permission. |
REFERENCES
Adner MM, et al: Immunologic studies of thymectomized and nonthymectomized patients with myasthenia gravis. Ann N Y Acad Sci 135:536, 1966.
Anson BJ: An Atlas of Human Anatomy. Philadelphia: W.B. Saunders, 1963.
Ashour M: Prevalence of ectopic thymic tissue in myasthenia gravis and its clinical significance. J Thorac Cardiovasc Surg 109:632, 1995.
Balcom RJ, et al: Massive thymic hyperplasia in an infant with Beckwith-Wiedemann syndrome. Arch Pathol Lab Med 109:153, 1985.
Bearman RM, Levine GD, Bensch KG: The ultrastructure of the normal human thymus. A study of 36 cases. Anat Rec 190:755, 1978.
Bell RH, et al: Form, size, blood supply and relations of the adult thymus. Bull Northwestern Univ Med School 28:156, 1954.
Bergman TA, Mariash CN, Oppenheimer JH: Anterior mediastinal mass in a patient with Graves' disease. J Clin Endocrinol Metab 55:587, 1982.
Boyd E: The weight of the thymus gland in health and in disease. Am J Dis Child 43:1162, 1932.
Boyd RI, Hugo P: Towards an integrated view of thymopoiesis. Immunol Today 12:71, 1991.
Cohen M, et al: Thymic rebound after treatment of childhood tumors. AJR Am J Roentgenol 135:151, 1980.
Cove H: The mediastinum. In: Coulson SF (ed): Surgical Pathology. 2nd Ed. Philadelphia: Lippincott, 1988.
De Maagd RA, et al: The human thymus microenvironment: heterogeneity detected by monoclonal anti-epithelial cell antibodies. Immunology 54:745, 1985.
Drenckhahn D, et al: Myosin and actin containing cells in the human postnatal thymus. Ultrastructural and immunohistochemical findings in normal thymus and in myasthenia gravis. Virchows Arch [B] 32:33, 1979.
Fukai I, et al: Distribution of thymic tissue in the mediastinal adipose tissue. J Thorac Cardiovasc Surg 101:1099, 1991.
Gelfand DW, et al: Thymic hyperplasia in children recovering from thermal burns. J Trauma 12:813, 1972.
George AJT, Ritter MA: Thymic involution with ageing: obsolescence or good housekeeping? Immunol Today 17:267, 1996.
Good RA, Gabrielsen AE (eds): The Thymus in Immunobiology: Structure, Function, and Role in Disease. New York: Harper & Row, Hoeber Medical Division, 1964.
Haynes BF, Hale LP: The human thymus. A chimeric organ comprised of central and peripheral lymphoid components. Immunol Res 18:175, 1998.
Haynes BF, et al: Phenotypic characterization and ontogeny of mesodermal derived and endocrine epithelial components of the human thymic microenvironment. J Exp Med 159:1149, 1984.
Henry K: An unusual thymic tumour with a striated muscle (myoid) component (with a brief review of the literature on myoid cells). Br J Dis Chest 66:291, 1972.
Hirokawa K, et al: Immunohistochemical studies in human thymomas. Localization of thymosin and various cell markers. Virchows Arch [B] 55: 371, 1988.
Jaretzki A III: Thymectomy for myasthenia gravis: analysis of the controversies regarding technique and results. Neurology 48(Suppl 5):S52, 1997.
Jaretzki A III, Wolff M: Maximal thymectomy for myasthenia gravis. Surgical anatomy and operative technique. J Thorac Cardiovasc Surg 96:711, 1988.
Judd R, Bueso-Ramos C: Combined true thymic hyperplasia and lymphoid hyperplasia in Graves' disease. Pediatr Pathol 10:829, 1990.
Kendall MD, Johnson HRM, Singh J: The weight of the human thyroid gland at necropsy. J Anat 131:483, 1980.
Kornstein MJ: Pathology of the Thymus and Mediastinum. Philadelphia: WB Saunders, 1995, pp. 14, 42.
Lee Y, Moallem S, Clauss RH: Massive hyperplastic thymus in a 22-month-old infant. Ann Thorac Surg 27:356, 1979.
Lele SM, Lele MS, Anderson VM: The thymus in infancy and childhood. Embryologic, anatomic, and pathologic considerations. Chest Surg Clin N Am 11:233, 2001.
Levine GD, Rosai J: Thymic hyperplasia and neoplasia: a review of current concepts. Hum Pathol 9:495, 1978.
Linegar AG, et al: Massive thymic hyperplasia. Ann Thorac Surg 55:1197, 1993.
Marchevsky AM, Kaneko M: Surgical Pathology of the Mediastinum. New York: Raven Press, 1984, p. 29.
Marchevsky AM, Kaneko M: Surgical Pathology of the Mediastinum. 2nd Ed. New York: Raven, 1992.
Marino M, M ller-Hermelink HK: Thymoma and thymic carcinoma. Relation of thymic epithelial cells to the cortical and medullary differentiation of the thymus. Virchows Arch [A] 407:119, 1985.
Masaoka A, Nagaoka Y, Kotake Y: Distribution of thymic tissue at the anterior mediastinum. J Thorac Cardiovasc Surg 70:747, 1975.
McFarland E, Scearce RM, Haynes BF: The human thymic microenvironment: cortical thymic epithelium is an antigenically distinct region of the thymic microenvironment. J Immunol 133:1241, 1984.
M ller-Hermelink HK, Marino M, Palestio G: Pathology of thymic epithelial tumors. Curr Top Pathol 75:206, 1986.
Murakami S, et al: A thymic tumor with massive proliferation of myoid cells. Acta Pathol Jpn 34:1375, 1984.
Nomori H, et al: A case of massive true thymic hyperplasia with non-Hodgkin's lymphoma. Chest 98:1304, 1990.
Norris EH: The morphogenesis and histogenesis of the thymus gland in man: in which the origin of the Hassall's corpuscles of the human thymus is discovered. Contrib Embryol 27:191, 1938.
Parker JR, Rao JY, Ord ez NG: Benign nevus cell aggregates in the thymus: a case report. Mod Pathol 12:329, 1999.
Patton BM: Human Embryology. 3rd Ed. New York: McGraw-Hill, 1968, p. 438.
Res P, Spits H: Developmental stages in the human thymus. Semin Immunol 11:39, 1999.
Riazmontazer N, Bedayat G: Aspiration cytology of an enlarged thymus presenting as a mediastinal mass. A case report. Acta Cytol 37:427, 1993.
Ricci C, et al: True thymic hyperplasia: a clinicopathological study. Ann Thorac Surg 47:741, 1989.
Savino W, et al: Thymic hormone-containing cells. 5. Immunohistological detection of metallo-H-thionein within the cells bearing thymulin (a zinc-containing hormone) in human and mouse thymuses. J Histochem Cytochem 32:942, 1984.
Shin MS, Ho KJ: Diffuse thymic hyperplasia following chemotherapy for nodular sclerosing Hodgkin's disease. An immunologic rebound phenomenon? Cancer 51:30, 1983.
Steinmann GG: Changes in the human thymus during aging. Curr Top Pathol 75:43, 1986.
Suster S, Rosai J: Histology of the normal thymus. Am J Surg Pathol 14:284, 1990.
Takagi M, et al: Massive thymic hyperplasia associated with dyspnea: a case report. J Jpn Assoc Chest Surg 10:620, 1995.
Tartas NE, et al: Diffuse thymic enlargement in Hodgkin's lymphoma. JAMA 254:406, 1985.
von Gaudecker B: The development of the human thymus microenvironment. Curr Top Pathol 75:1, 1986a.
von Gaudecker B, et al: Immunohistochemical characterization of the thymic microenvironment: light microscopic and ultrastructural immunocytochemical study. Cell Tissue Res 244:403, 1986b.
Weller GI: Development of the thyroid, parathyroid and thymus glands in man. Contrib Embryol 141:95, 1933.
Yulish BS, Owens RP: Thymic enlargement in a child during therapy for primary hypothyroidism. AJR Am J Roentgenol 135:157, 1980.
EAN: 2147483647
Pages: 203