10 - Peripheral Nervous System
Editors: Mills, Stacey E.
Title: Histology for Pathologists, 3rd Edition
Copyright 2007 Lippincott Williams & Wilkins
> Table of Contents > V - Head and Neck > 14 - The Ear and Temporal Bone
function show_scrollbar() {}
14
The Ear and Temporal Bone
Bruce M. Wenig
Leslie Michaels
Introduction
The ear can be considered as three distinct regions, or compartments, to include the external ear, the middle ear and temporal bone, and the inner ear (Figure 14.1).
The external ear consists of the auricle (pinna), external auditory canal (or meatus), and the tympanic membrane at the medial end of the auditory canal.
The middle ear cavity includes the ossicles, auditory (eustachian or pharyngotympanic) tube connecting the middle ear space to the nasopharynx, and expansion of the middle ear cavity in the form of air cells in the temporal bone.
The inner ear is embedded in the petrous portion of the temporal bone and consists of a membranous (otic) labyrinth that lies within a dense bone referred to as the otic capsule, which is excavated to form the osseous (periotic) labyrinth (1).
The inner ear is the sense organ for both hearing and balance. The external and middle ears are the sound-conducting apparatus for the auditory part of the inner ear.
External Ear
Embryology
The external ear develops from the first brachial groove. The external auricle (pinna) forms from the fusion of the auricular hillocks or tubercles (a group of mesenchymal tissue swellings from the first and second branchial arches) that lie around the external portion of the first branchial groove (2). The external auditory canal is considered a normal remnant of the first branchial groove. The tympanic membrane forms from the first and second branchial pouches and the first branchial groove (2). The ectoderm of the first
P.372
branchial groove gives rise to the epithelium on the external side; the endoderm from the first branchial pouch gives rise to the epithelium on the internal side; and the mesoderm of the first and second branchial pouches gives rise to the connective tissue lying between the external and internal epithelia (Figure 14.2) (2).
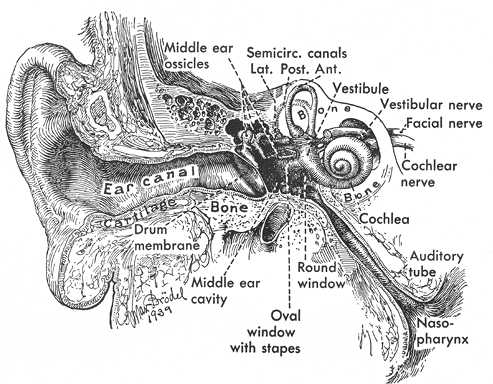 |
Figure 14.1 Modified coronal section through the ear and temporal bone depicting the anatomic compartments of the ear: the external, middle, and inner ears (Reprinted with permission from: Hollinshead WH. The ear. In: Hollinshead WH, ed. Anatomy for Surgeons. 3rd ed. Philadelphia: Harper and Row; 1982:159 221 ). |
Anatomy
The anatomy of the external ear is seen in Figure 14.3. The outer portion of the external ear includes the auricle leading into the external auditory canal. The skeleton of the auricle consists of a single plate of elastic cartilage conforming to the shape of the ear. The lobule is the only part of the auricle that is devoid of skeletal support. The cartilage of the auricle is continuous with that of the external auditory canal.
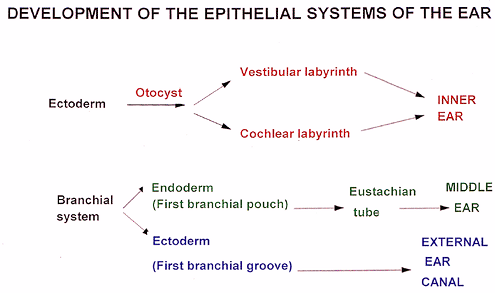 |
Figure 14.2 Diagrammatic representation of the embryology of the epithelia of the inner, middle, and external ears. |
The auricle is attached to the bony skull by three ligaments: anterior, superior, and posterior (1). The anterior ligament attaches the helix and the tragus to the zygomatic process. The superior ligament attaches the spine of the helix to the superior margin of the bony external meatus. The posterior ligament attaches the medial surface (eminence) of the concha to the mastoid process. The auricle is anchored through its continuity with the cartilage of the meatus and through the skin and extrinsic muscles.
The extrinsic muscles of the ear include the anterior, superior, and posterior auricular muscles. These muscles are usually functionless but may be subject to voluntary control (as in ear wiggling ). There are also small intrinsic muscles in connection with the cartilage of the external ear, but they are of no apparent importance. The extrinsic and intrinsic muscles of the ear are innervated by the facial nerve.
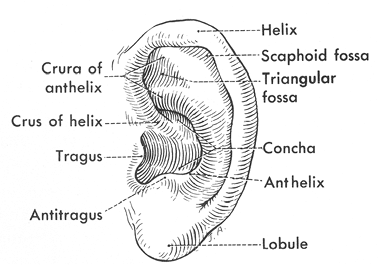 |
Figure 14.3 Anatomy of the external ear. |
P.373
The external auditory canal or meatus extends from the concha to its medial limit, which is the external aspect of the tympanic membrane. The lateral portion of its wall consists of cartilage and connective tissue (1); the medial portion of its wall consists of bone. The cartilaginous part of the external auditory canal constitutes slightly less than half its total length. Inconstant fissures, referred to as the fissures of Santorini, occur in the cartilage; these fissures may transmit infection from the canal to the parotid gland and superficial mastoid regions, or vice versa. The bony part of the canal is formed by both the tympanic part and the petrous part of the temporal bone. The anterior, inferior, and lower posterior parts of the bony wall are formed by the C-shaped part of the temporal bone developed from the annulus tympanicus of the fetus. However, the annulus is incomplete in the posterosuperior part of the wall, and this part of the wall in adults is formed by the squamous and petrous parts of the temporal bone. In adults, the anterior and inferior walls of the cartilaginous canal are closely related to the parotid gland. The anterior wall of the bony canal is closely related to the mandibular condyle, the posterior wall to the mastoid air cells, and the medial portion of the superior wall to the epitympanic recess.
The tympanic membrane (eardrum) is situated obliquely at the end of the external auditory canal, sloping medially both from above downward and from behind forward. The tympanic membrane is a fibrous sheet interposed between the external auditory canal and the middle ear cavity (Figure 14.4). The connective tissue interposed between these two layers consists of radiating fibers that are attached to the manubrium of the malleus and are reinforced peripherally by circular fibers. The latter are thickened at the margin of the tympanic membrane to form a fibrocartilaginous ring (annulus fibrocartilagineus membranae tympani), attaching the tympanic membrane to the tympanic sulcus of the temporal bone. In the upper portion of the tympanic membrane, there is a limited area where the connective tissue fibers are lacking; this area is referred to as the pars flaccida, or Schrapnell's membrane. In this area, the tympanic portion of the temporal bone is deficient; this gap is referred to as the tympanic incisure, or the notch of Rivinus. The tympanic membrane attaches to the temporal bone. The remainder of the tympanic membrane, in which there are intact connective tissue fibers, is referred to as the pars tensa.
The outer aspect of the tympanic membrane is concave. The center of the concavity is referred to as the umbo, which is the strong point of attachment of the manubrium of the malleus to the tympanic membrane. In the anteriosuperior portion of the tympanic membrane, the lateral process of the malleus is attached; from this point of attachment, the anterior and posterior mallear folds pass to the cartilaginous annulus and separate the pars flaccida from the pars tensa. In otoscopic examinations of the tympanic membrane, the bright area of light reflection present downward and forward from the umbo is referred to as the cone of light.
Histology
Histologically, the auricle is essentially a cutaneous structure composed of keratinizing, stratified squamous epithelium with associated cutaneous adnexal structures that include hair follicles, sebaceous glands, and eccrine sweat glands (Figure 14.5). In addition to the hair follicles and sebaceous glands, the outer third of the external auditory canal is noteworthy for the presence of modified apocrine glands (called ceruminal glands) that replace the eccrine glands seen in the auricular dermis (Figure 14.6). Ceruminal glands produce cerumen and are arranged in clusters
P.374
composed of cuboidal cells with eosinophilic cytoplasm, often containing a granular, golden-yellow pigment. These cells have secretory droplets along their luminal border. Peripheral to the secretory cells are flattened myoepithelial cells. The ducts of the ceruminal glands terminate in the hair follicle or on the skin. The ducts of ceruminal glands lack apocrine or myoepithelial cells. In the inner portion of the external auditory canal, ceruminal glands and the other adnexal structures are absent.
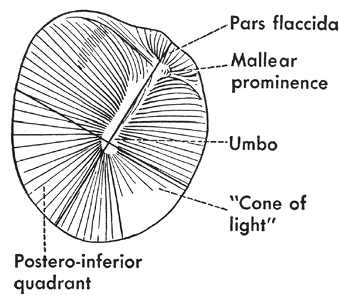 |
Figure 14.4 External (lateral) view of the tympanic membrane. |
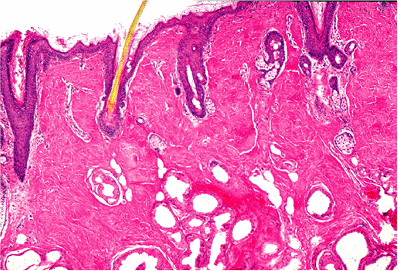 |
Figure 14.5 The auricle is a cutaneous structure histologically composed of keratinizing stratified squamous epithelium with associated cutaneous adnexal structures that include hair follicles, sebaceous glands, and eccrine sweat glands. |
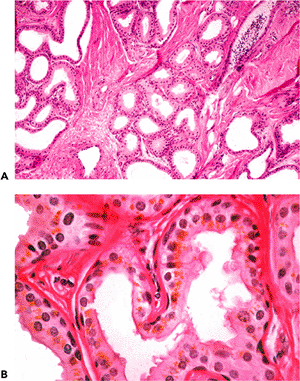 |
Figure 14.6 In addition to the hair follicles and sebaceous glands, the outer third of the external auditory canal is noteworthy for the presence of modified apocrine glands called ceruminal glands that replace the eccrine glands seen in the auricular dermis. A. Ceruminal glands are submucosal in location and are arranged in clusters or lobules. B. Ceruminal glands are composed of two cell layers, including the inner or secretory cells (containing intracytoplasmic cerumen and appearing as granular, golden-yellow pigmentation) and flattened-appearing myoepithelial cells located peripheral to the secretory cells. Focally, the secretory cells show holocrine (decapitation) type secretion. |
The subcutaneous tissue is composed of fibroconnective tissue, fat, and elastic-type fibrocartilage, which gives the auricle its structural support (Figure 14.7). The ear lobe is devoid of cartilage and is replaced by a pad of adipose tissue. The perichondrium is composed of loose vascular connective tissue.
Similar to the auricle, the external auditory canal is lined by keratinizing squamous epithelium that extends to include the entire canal and covers the external aspect of the tympanic membrane. The tympanic membrane has a central bilaminated zone, including lateral radially arranged
P.375
and medial circularly arranged collagenous fibers (Figure 14.8). The inner two-thirds of the external auditory canal contain bone rather than cartilage. Because adnexal structures are absent, there is relatively close apposition of the epithelium to the subjacent bone.
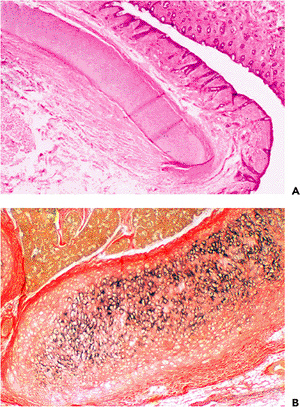 |
Figure 14.7 A. The cartilage of the external ear is elastic. B. Elastic stains show the abundant amount of elastic fibers (black staining) in the auricular cartilage. |
 |
Figure 14.8 Section of pars tensa of tympanic membrane. The following layers may be distinguished from left to right: middle ear epithelium, lamina propria, circular arrangement of collagenous fibers (i.e., at right angles to former layer), radial arrangement of collagenous fibers, lamina propria, and stratified squamous epithelium. |
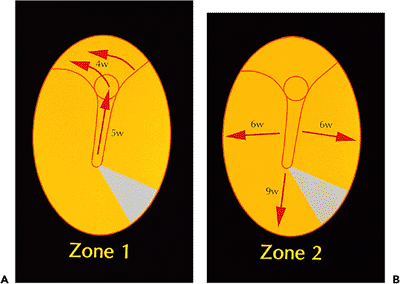 |
Figure 14.9 Summary of pathways of migration on tympanic membrane as determined by serial photography of dye markings. The tympanic membrane and adjacent deep external canal epithelium are depicted as being viewed en face. Two discrete pathways are present: A. passing upward along a tongue of epithelium and over the handle of the malleus to join epithelium, moving in a posterosuperior direction over the pars flaccida region (Zone 1), and B. a radial pathway moving centrifugally from the pars flaccida and handle of malleus regions to the periphery (Zone 2). The times given for each region are the weeks required for dye to be completely cleared from that region. |
Auditory Epithelial Migration
Auditory epithelial migration represents the mechanism by which keratin is removed from the tympanic membrane. Without such a self-cleaning process, the keratin squames normally produced by the stratified squamous epithelium of the tympanic membrane would continuously build up and interfere with the conduction of sound via the tympanic membrane. The entire epithelium, including keratin, moves from the tympanic membrane onto the deep external auditory canal. From the deep external auditory canal, the epithelium moves laterally to the junction of the deep (osseous) canal and the cartilaginous canal, where it is desquamated (3,4,5).
Auditory epithelial migration occurs in two separate and discrete pathways (Figure 14.9) (6). In one pathway, the epithelium moves upward over the handle of the malleus and then posterosuperiorly across the pars flaccida, moving laterally over the deep canal (Figure 14.10). The other pathway is radially moving away from the handle of the malleus and pars flaccida to the periphery of the tympanic membrane and then to the deep canal (Figure 14.11). Michaels and Soucek (3,4,5) have extensively evaluated the process of auditory epithelial migration and correlate the pathways to the development of the epithelia of the tympanic membrane and deep external canal in the embryo and fetus (6). The process of auditory epithelial migration has been felt to represent a possible pathogenesis for the development of cholesteatoma
P.376
(see below). However, there is no definitive evidence linking auditory epithelial migration to the development of cholesteatoma.
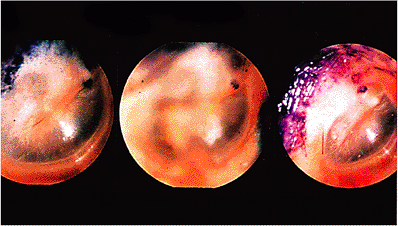 |
Figure 14.10 Pathway of auditory epithelial migration as shown by movement of blue dye daubed on tympanic membrane. Dye is seen on the day it was daubed, just anterior and inferior to the lateral process of the malleus. In the next photograph, taken 9 days later, it has moved posteriorly and superiorly to lie over that structure. Thirteen days later, in the third Photograph, it has crossed the pars flaccida region, moving in the same direction toward the external canal. |
Middle Ear
Embryology
The middle ear space develops from invagination of the first branchial pouch (pharyngotympanic tube) from the primitive pharynx. The auditory tube and tympanic cavity develop from the endoderm of the first branchial pouch; the malleus and incus develop from the mesoderm of the first branchial arch (Meckel's cartilage), while the stapes develops from the mesoderm of the second branchial arch (Reichert's cartilage) (Figure 14.2) (2).
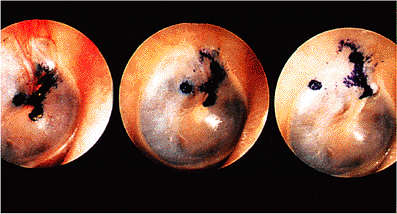 |
Figure 14.11 In this daubed tympanic membrane, an irregular array of dye is seen on the handle of malleus region on the sixth day after its deposition. By the fifteenth day, in the second photograph, a round dot that was just posterior to the handle of the malleus has separated and is commencing to travel backward; the main mass of dye is moving discretely upward along the handle of the malleus. This process has advanced on the twenty-seventh day in the third photograph, the posterior dye having reached the back edge of the tympanic membrane and the large mass now situated across the pars flaccida at an angle that has now changed to a posterosuperior one. |
Anatomy
The middle ear, or tympanic cavity, lies within the temporal bone between the tympanic membrane and the squamous portions of the temporal bone laterally and the petrous portion of the temporal bone surrounding the inner ear medially. The anatomic limits of the tympanic cavity include (1,7,8):
lateral or internal aspect made up by the tympanic membrane and squamous portion of the temporal bone
medial aspect bordered by the petrous portion of the temporal bone
superior (roof) delimited by the tegmen tympani (a thin plate of bone that separates the middle ear space from the cranial cavity)
inferior (floor) aspect bordered by a thin plate of bone separating the tympanic cavity from the superior bulb of the internal jugular vein
anterior aspect delimited by a thin plate of bone separating the tympanic cavity from the carotid canal (that houses the internal carotid artery)
posterior aspect delimited by the petrous portion of the temporal bone, containing the mastoid antrum and mastoid air cells
The tympanic cavity communicates anteriorly with the nasopharynx by way of the auditory tube, and it communicates posteriorly with the mastoid air cells by way of the aditus and mastoid antrum.
The contents of the tympanic cavity include the ossicles (malleus, incus, and stapes), ligaments of the ossicles, tendons of the ossicular muscles, auditory tube, tympanic cavity proper, epitympanic recess, mastoid cavity, and chorda tympani of the facial nerve (cranial nerve VII). The middle and external ears function as conduits for sound conduction for the auditory part of the internal ear.
Lateral Wall
The tympanic cavity extends above the level of the tympanic membrane as the epitympanic recess (attic). In this area lie the head of the malleus and the body of the short process of the incus. The epitympanic recess projects laterally above the external acoustic meatus; it is this portion of the tympanic cavity that has a part of the squamous portion of the temporal bone as its lateral wall.
Roof
The roof of the tympanic cavity is the tegmen tympani, a thin plate of bone separating the middle ear cavity from the
P.377
cranial cavity. In children, the unossified petrosquamous suture of the tegmen tympani may allow the direct passage of infection from the middle ear to the meninges of the middle cranial fossa (1). In adults, especially in the setting of a long-standing history of chronic otitis media, compromise of the tegmen tympani may result in acquired encephalocele, in which glial-type tissue is present within the middle ear cavity. In adults, veins from the middle ear perforate the petrosquamous suture to end in the petrosquamous and the superior petrosal sinuses and may potentially transmit infection directly to the cranial venous sinuses (1).
Floor
The floor of the tympanic cavity is usually a thin plate of bone separating the cavity from the internal jugular vein. In the presence of a large superior bulb of the internal jugular vein, it may bulge into the middle ear and may present dehiscences (9).
Posterior Wall
The posterior wall of the tympanic cavity opens through the narrow aditus ad antrum in the wider mastoid (tympanic) antrum. Below the aditus is a relatively thin bone separating the tympanic cavity from the antrum, and it is from this posterior wall that the pyramidal eminence projects, with an aperture at its apex from which the tendon of the stapedius muscle is transmitted. Above and behind the pyramidal eminence, the facial nerve curves downward to change its course from horizontal to vertical. The chorda tympani, arising from the facial nerve, then enters the tympanic cavity through the canaliculus of the chorda in the posterior wall.
Anterior Wall
The lower part of the anterior wall is part of the petrous apex. This area consists of a thin plate of bone (which may be incomplete or may contain air cells) that separates the cavity from the carotid canal in which the internal carotid artery is located. The upper part of the anterior wall is deficient since the canal containing the tensor tympani muscle opens in this location, and immediately below this area is the tympanic orifice of the auditory tube.
Medial Wall
The medial wall of the tympanic cavity is the petrous portion of the temporal bone surrounding the internal ear and separating the middle ear cavity from the inner ear cavity.
Several markings of importance are found on its surface, including the broad prominence produced by the anterior end of the lateral semicircular canal and the prominence of the facial (fallopian) canal produced by the horizontal portion of the facial nerve in its course between the inner and middle ears. The cochleariform process transmits the tendon of the tensor tympani muscle. Its apex is the landmark for the position of the turn (geniculum, or external genu) between the anterolaterally and posteriorly directed horizontal portions of the facial nerve. Immediately below the facial canal is the fossula fenestrae vestibuli (the stapes niche); it contains the oval window, which is closed by the base of the stapes. Below the oval window is the promontory formed by the basal turn of the cochlea. The tympanic nerve plexus lies on the promontory. Below the back part of the promontory, the cochlear fossula (the round window niche) leads to the round window or fenestrae cochlea. Behind the promontory is a depression referred to as the sinus tympani, a site that may harbor infections and may transmit infections to the ampullary end of the posterior canal and posterior end of the lateral canal if the infection is deeply situated (1).
Middle Ear Ossicles and Muscles
The middle ear bones, or ossicles, include the malleus, incus, and stapes (Figure 14.12). The parts of the malleus include a head, upper and lower manubrium (handle), lateral process, and anterior process. The malleus is closely attached to the tympanic membrane by its manubrium (handle) and its lateral process, while its head projects above the epitympanic recess to articulate with the body of the incus. The anterior (long) process (processus gracilis) of the malleus extends obliquely downward from the neck toward the tympanosquamous fissure. In infants, the anterior process may reach the tympanosquamous fissure; but, in adults, the distal part is transformed to connective tissue, forming the anterior ligament of the malleus (1). The malleus is also attached to the tympanic wall by superior and lateral mallear ligaments. The lateral ligament attaches the neck of the malleus to the margin of the tympanic notch.
The parts of the incus include its body and the long and short processes. The body of the incus is fitted against the head of the malleus and lies in the epitympanic recess. The short process (crus) rests in a depression referred to as the fossa of the incus, which is situated in the posterior wall of the tympanic cavity below the aditus ad antrum. The long process (crus) of the incus descends parallel and slightly posteromedial to the manubrium of the malleus but at its lower end turns medial to articulate with the stapes (1). The incus is held in place by a posterior ligament that attaches to its short process and by a superior ligament to attach to the body. The knoblike expansion of the long crus of the incus (at the incudomallear joint) is referred to as the lenticular process.
The stapes is formed by its two crura, a head that lies at the junction of the crura and a footplate that lies on the oval window (Figure 14.13). The head of the stapes articulates with the incus. From its articulation with the incus, the
P.378
stapes passes almost horizontally to the oval (vestibular) window. The footplate of the stapes is attached to the oval window by the annular ligament. The latter, a ring of elastic fibers, allows movement of the stapes but seals any potential space between its footplate and the edges of the oval window.
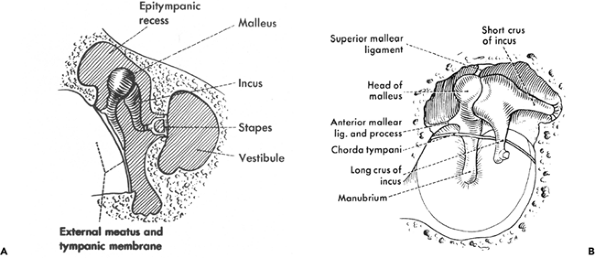 |
Figure 14.12 Diagrammatic depiction of the anatomy of the middle ear ossicles: A. frontal aspect (rotated through approximately 90 ), and B. medial aspect. (Reprinted with permission from Hollinshead WH. The ear. In: Hollinshead WH, ed. Anatomy for Surgeons. 3rd ed. Philadelphia: Harper & Row; 1982:159 122. |
The incudomallear and incudostapedial joints are synovial (diarthroidal) (see below). In addition to their ligaments, the stapes and manubrium of the malleus have muscles attached to them. The stapedius muscle diminishes the excursion of the base of the stapes by its reflex contraction. Important functions ascribed to the stapedius muscle are to protect the inner ear from excessive sound and to improve discrimination for higher frequencies in speech. The stapedius muscle is innervated by a branch of the facial nerve.
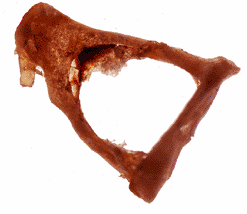 |
Figure 14.13 Intact resected stapes, showing (from left to right) its head, two crura, and footplate. |
The tensor tympani muscle draws the manubrium medially, thereby tightening the tympanic membrane. The tensor tympani muscle is felt to primarily protect against excessive noise but also functions, in conjunction with the tensor veli palitini muscle, to respond to swallowing and electric stimulation from the tongue. The action of these two muscles pumps air from the tympanic cavity into the auditory tube, forcing air into the nasopharynx and helping to open the isthmus (10). The tensor tympani muscle is innervated by a branch of the mandibular nerve.
Auditory (Eustachian or Pharyngotympanic) Tube
The auditory tube extends from its tympanic ostium high on the anterior wall of the tympanic cavity to a nasopharyngeal ostium situated posterior to the inferior nasal concha (1). The tube is not straight but slightly S-shaped. In the adult, the tympanic ostium is approximately 2 to 2.5 cm higher than is the nasopharyngeal end; the tube runs downward, medially, and anteriorly to the nasopharynx. The length of the tube in adults varies from 31 to 38 mm (11). In infants, the tube is shorter, relatively wider, and more horizontal in its course and, therefore, an easier pathway for infections ascending from the nasopharynx to the tympanic cavity.
The tube can be divided into an osseous portion and cartilaginous portion. The osseous portion or canal has a bony wall and is the lateral, or tympanic, third of the tube. The anteromedial two-thirds has a cartilaginous and connective tissue wall and is referred to as the cartilaginous portion of
P.379
the tube. The cartilaginous and osseous tubes meet at an obtuse angle.
Histology
Tympanic Cavity Proper
Histologically, the epithelial lining of the tympanic cavity is a single layer of respiratory epithelium of flattened to cuboidal epithelium (Figure 14.14). Under normal conditions, there are no glandular elements within the middle ear; the presence of glandular epithelium in the middle ear is abnormal (see below under Selective Abnormalities). Further, stratified squamous epithelium is not present in the tympanic cavity under normal conditions nor does squamous metaplasia occur in the middle ear (5). Ciliated pseudostratified columnar epithelium may be found in limited patches among the flattened or cuboidal epithelium.
Auditory Tube
The lining of the auditory tube is a low ciliated epithelium for much of its length except as it approaches its nasopharyngeal end, where it becomes ciliated pseudostratified columnar epithelium containing goblet cells. In its cartilaginous portion, it also contains seromucinous glands (Figure 14.15). The auditory tubes contain a lymphoid component, particularly in children, that is referred to as Gerlach's tubal tonsil (Figure 14.16). Reactive hyperplasia of this lymphoid component, particularly in children, may close off the auditory tube, providing a desirable milieu for otitis media. The mucosa of the osseous portion of the auditory tube is separated from the carotid canal by a thin plate of bone measuring 1 mm in thickness (5). Dehiscence of the carotid canal is fairly frequent (12). Squamous carcinoma of the middle ear or auditory tube, a rare occurrence, may easily penetrate this area and gain access to the carotid artery, with the potential for widespread dissemination (13). The cartilage of the nasopharyngeal portion of the auditory tube is hyalin type.
 |
Figure 14.14 The epithelial lining of the tympanic cavity is a single layer of epithelium (cuboidal to respiratory). Under normal conditions, glands are not identified within the tympanic cavity. |
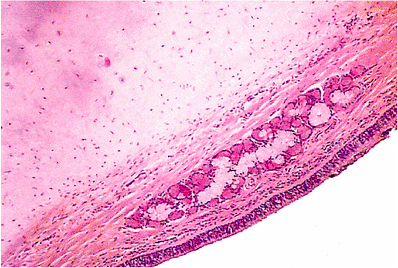 |
Figure 14.15 Cartilaginous portion of auditory tube with seromucinous glands. |
Mastoid Air Cells
The mastoid air cells represent a network of intercommunicating spaces that emanate from the tympanic cavity (5). Each air cell is lined by flattened to cuboidal epithelium, which rests on periosteum that covers a thin frame of lamellar bone (Figure 14.17).
Pneumatization of the Temporal Bone
In the newborn, the rudimentary mastoid bone contains a single air space, the antrum, surrounded by diploic bone
P.380
containing hematopoietic elements (14). As the mastoid process develops, the marrow spaces hollows out. The mesenchymal component occupying the space is resorbed and the developing air-containing cells become lined by the advancing endodermal epithelium. The mastoid process is constantly pneumatized in adults, although not in the infant. The cells grow out from the antrum, as well as from each other, forming complex interlocking chains of thin-walled cavities opening into each other. The antrum apparently always has air cells; the mastoid process is usually one of several types, including pneumatized (containing air cells), diploic (containing marrow), mixed (containing air cells and marrow), or sclerotic. Approximately 80% of mastoid are well pneumatized by the age of 3 or 4 years; but, in approximately 20% of people, normal pneumatization fails to occur (1,15).
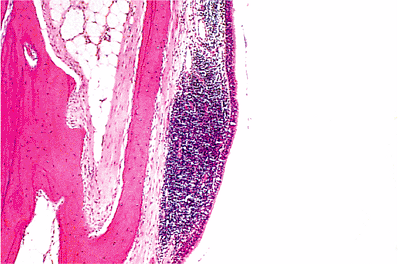 |
Figure 14.16 Mucosa of auditory tube. The lining is of ciliated columnar epithelium. In the lamina propria beneath, there are numerous lymphocytes that are probably the result of inflammation. |
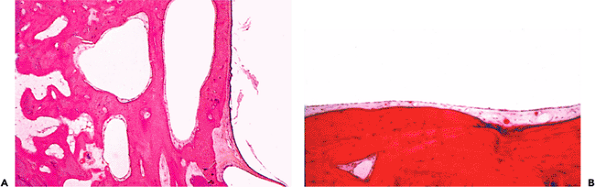 |
Figure 14.17 A. Mastoid air cells (center), tympanic membrane (lower right), and squamous epithelium of the osseous portion of the external canal (right). Note the thin covering of skin over the external ear canal and the proximity of bone to it. B. Higher magnification shows the very thin epithelial layer of the mastoid air cells resting on perisoteum covering lamellar bone. |
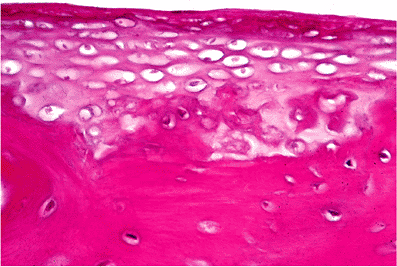 |
Figure 14.18 Stapes footplate showing persistence of cartilage. |
Middle Ear Ossicles
The middle ear ossicles develop from cartilage with a single center of ossification for bone; there is no epiphyseal ossification. The persistence of cartilage in each of the ossicles (Figure 14.18) and the bifurcation of the stapes to form the crura with the obturator foramen between them distinguishes the middle ear ossicles from other long bones (5).
The head of the stapes is formed of endochondral bone with a cartilaginous cap at the incudostapedial joint. The crura of the stapes are formed of periosteal bone only. From the middle ear aspect of the stapes footplate to its vestibular surface, the histologic findings include the flattened to cuboidal epithelium of the tympanic cavity, a thin layer of bone, cartilage, and a single flattened (perilymphatic) epithelial cell layer (Figure 14.19).
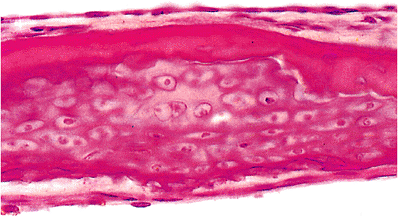 |
Figure 14.19 Stapes footplate. Beneath the cubical epithelium of the middle ear (above), there is a thin layer of bone. Below this, the footplate consists of cartilage, and there is a basal flattened layer of cells comprising the lining of the vestibule. |
P.381
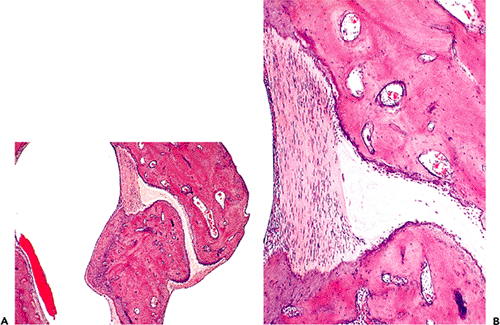 |
Figure 14.20 A. Incudomalleal joint. Note the joint capsule at each end of the joint. The joint space is occupied by the fibrocartilage of the articular disk. B. Higher power view of the incudomalleal joint; note one end of joint capsule and articular disk. |
The malleus and incus have an outer covering of periosteal bone layer and an inner core of endochondral bone with well-formed haversian systems. The manubrium (handle) of the malleus is predominantly covered by retained cartilage rather than periosteal bone. The entire inner core of the manubrium, as well as the rest of the malleus, is composed of endochondral bone. The anterior process is formed in membrane early in fetal life and merges with the malleus after its formation (5). At its superior aspect, the manubrium is separated from the tympanic membrane by a ligament covered by the middle ear epithelium. The short process of the incus shows a tip of unossified cartilage.
Middle Ear Joints
Both the incudomalleal and incudostapedial joints are diarthrodial. Middle ear epithelium is present on the outer surface of the joint capsule, and synovial membrane is present on its inner surface. The joint capsule is comprised of fibrous tissue with a high elastic fiber content (5). The articular disk, representing the space in between the articular ends, is comprised predominantly of fibrocartilage (Figure 14.20). The articular processes of both the malleus and the incus are covered by cartilage.
The annular ligment binds the cartilaginous edge of the stapes footplate to the cartilaginous rim of the vestibular window (stapediovestibular joint) (Figure 14.21) and is composed of fibrous tissue, with elastic fibers being prominent near the ligament surfaces (16). Cartilage also covers the articular surfaces of the stapediovestibular joints.
The fissula ante fenestrum is the canal linking the middle ear with the vestibule. It lies in the bone just anterior to the stapediovestibular joint and develops as a slit filled with fibrous tissue that is often with associated cartilage (Figure 14.22).
Middle Ear Muscles
The muscles of the middle ear, including the tensor tympani and stapedius muscles, are composed of a central tendon formed by elastic tissue with muscle fibers radiating from it (Figure 14.23). This configuration has been described as feather-shaped. The tensor tympani muscle has a prominent mature adipose tissue component (Figure 14.24) that is believed to function as insulation for the cochlea against electric effects from its contraction (5).
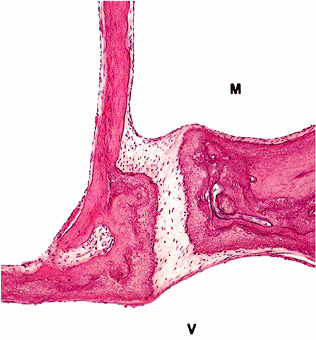 |
Figure 14.21 Stapediovestibular joint, part of footplate of stapes, adjacent bony labyrinthine wall, and crus of stapes. The footplate shows a lamina of cartilage on its vestibular surface, which is continuous with the cartilage of the stapediovestibular joint. (M, middle ear cavity; V, cavity of vestibule) |
P.382
Inner Ear
Embryology
The first division of the ear to develop is the inner ear, which appears toward the end of the first month of gestation (2,17). The membranous labyrinth, including the utricle, saccule, semicircular ducts, cochlear duct, and endolymphatic sac, arises from the placodal thickening of the ectoderm to become a closed otic vesicle (otocyst). The otic vesicle forms from the invagination of the surface ectoderm, located on either side of the neural plate, into the mesenchyme. This invagination eventually loses its connection with the surface ectoderm. The membranous labyrinth, which is essentially tubular and saccular is filled with fluid, the endolymph or endolymphatic fluid. The early development of the membranous labyrinth takes place in mesenchyme and subsequently in the cartilage destined to form the petrous portion of the temporal bone (1). The membranous labyrinth lies in cavities excavated from
P.383
this mesenchyme or cartilage. The space lying between the inner surface of the bony wall and the outer surface of the membranous labyrinth is the perilymphatic space. The perilymphatic space develops around the membranous labyrinth by fusion of mesenchymal spaces to form larger ones surrounding the membranous portion. The bony labyrinth, including the vestibule, semicircular canals and cochlea arises from the mesenchyme around the otic vesicle (2,17,18).
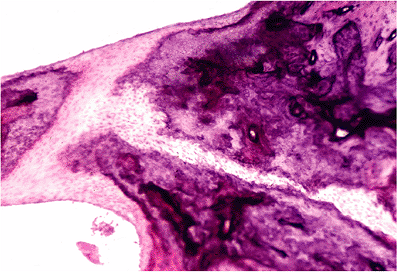 |
Figure 14.22 Fissula ante fenestrum is the canal within bone that links the middle ear with the vestibule and develops as a slit filled with fibrous tissue. |
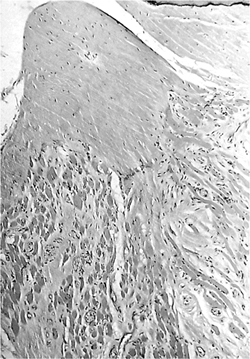 |
Figure 14.23 Stapedius muscle and tendon. The skeletal muscle fibers and fibrous bands between them radiate to a tendon. |
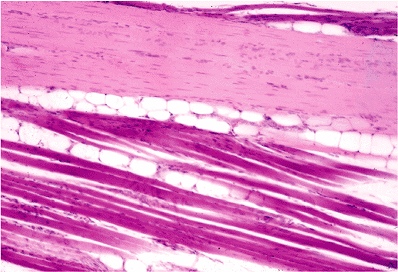 |
Figure 14.24 Tensor tympani muscle showing the presence of a mature adipose tissue component. |
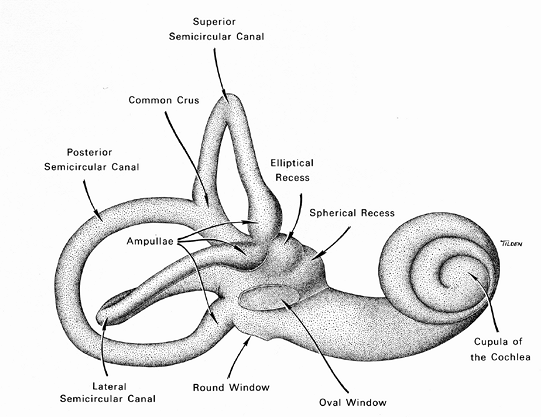 |
Figure 14.25 Schematic depicting the osseous labyrinth. |
Anatomy
The internal (inner) ear, or labyrinth, is embedded within the petrous portion of the temporal bone and comprises the medial portion of the temporal bone adjacent to the cranial cavity. The inner ear contains the membranous labyrinth, which is surrounded by an osseous layer or bony shell termed the osseous (bony) labyrinth (Figure 14.25). The membranous labyrinth contains the cochlea, which is the organ of hearing, and the vestibular system, which is the system of balance (equilibrium).
Osseous Labyrinth (Otic Capsule)
The osseous labyrinth consists of the vestibule and cochlear capsule. The central portion of the osseous labyrinth cavity is the vestibule, a large ovoid perilymphatic space approximately 4 mm in diameter and containing both the saccule and utricle of the membranous labyrinth. In the floor of the bony vestibule are seen the elliptical recess for the anterior end of the utricle and, anterior and lateral to this, the spherical recess for the saccule. In the lateral wall of the vestibule is the oval window, in which the base of the stapes is situated. Through the stapes, the perilymph of the vestibule receives vibrations from the tympanic membrane and ossicular chain (set up by sound waves reaching the tympanic cavity). Along the medial wall and floor of the vestibule, where it abuts the lateral end of the internal acoustic meatus, are small openings for the entrance of the nerve branches to the vestibular portion of the ear (1).
The bony cochlea, a part of the otic capsule, is a hollowed spiral about two- to three-fourths turns, diminishing from a relatively broad base to a pointed cupula, or apex. It is so named because of its resemblance to a snail shell. The base of the cochlea lies against the anteromedial surface of the vestibule and next to the anterior surface of the lateral (blind) end of the internal auditory canal. A central core of bone called the modiolus runs forward from the cochlea but does not reach the cupula. It is around this central core that the spiral channels of the cochlea (perilymphatic and endolymphatic) are arranged. A layer of bone arranged in a spiral fashion unites the modiolus to the peripheral wall of the bony cochlea and separates successive spiral cavities from each other (1). The modiolus is hollow to accommodate
P.384
the cochlear nerve. The base of the modiolus lies against the lateral end of the internal auditory canal, to which the cochlear nerve runs.
The vestibular aqueduct extends through the otic capsule from the vestibule to the posterior cranial fossa, transmitting the endolymphatic duct. The terminal end of the vestibular aqueduct is the endolymphatic sac, a dilated area that ends blindly outside the dura (1). The cochlear duct opens at one end into the lower end of the scala tympani and at the other end into the subarachnoid cavity (1). The issue as to whether the cochlear duct represents an open channel between the subarachnoid space and the perilymphatic space at the lower end of the scala tympani remains controversial (1). A possible role ascribed to the cochlear duct is to serve as part of the pressure-adjusting mechanism of the perilymph in conjunction with the round window (19,20,21).
Membranous Labyrinth
The membranous (otic) labyrinth is the spiral-appearing structure that resembles the shell of a snail. The principle components of the membranous labyrinth are the cochlear duct, utricle, saccule, ductus reuniens, semicircular canals with their ampullae, and the endolymphatic sac and duct.
 |
Figure 14.26 Schematic illustration of the membranous labyrinth, containing the cochlea (organ of hearing) and the vestibular (system of balance) systems. The relationship between the endolymph, containing scala media (cochlear duct), and the perilymph, containing scala vestibuli and scala tympani, is shown. (Reprinted with permission from: Nager GT. Anatomy of the membranous cochlea and vestibular labyrinth. In: Nager GT, ed. Pathology of the Ear and Temporal Bone. Baltimore: Williams & Wilkins; 1993:3 48. ) |
Cochlear Duct
The membranous cochlea, or cochlear duct, is a cone-shaped spirally oriented membranous tube between the osseous spiral lamina and the outer osseous wall of the cochlea, to which it is attached (7). The cochlear duct, also referred to as the scala media, lies between the scala vestibuli and the scala tympani (Figure 14.26). These three compartments are fluid-filled. The cochlear duct, as well as the entire membranous labyrinth, contains endolymph. The scala vestibuli and the scala tympani contain perilymph. The cerebrospinal fluid communicates directly with the perilymphatic space through the cochlear aqueduct (perilymphatic duct) (Figure 14.27).
The cochlear duct contains the sensory end organ of hearing known as the spiral organ of Corti. The organ of Corti rests on the basilar membrane, which separates the
P.385
cochlear duct from the scala tympani (Figure 14.26). Together, the organ of Corti and the basilar membrane form the spiral membrane that is the floor or tympanic wall of the cochlear duct. The spiral ligament is a thickened, modified portion of periosteum of the bony cochlea, forming the outer curved wall of the cochlea duct and adjacent parts of the scalae. The scala tympani lies below the basilar membrane, while the scala vestibuli lies above the cochlear duct and is separated from it by Reissner's membrane. Reissner's membrane forms the roof of the cochlear duct. The scala tympani and scala vestibuli communicate with each other only at the apex, known as the helicotrema. The scala vestibuli winds toward the apex of the cochlea at the helicotrema, becoming the scala tympani which, in turn, coils back toward the round window (Figure 14.28). The scala vestibuli and the scala tympani communicate with the middle ear via the oval window and round window, respectively. The scala tympani ends blindly at the round window membrane, but the scala vestibuli opens up at that level into the perilymphatic space of the vestibulum. The cochlear duct connects with the vestibular system via ductus reuniens located at the saccule. In this way, the three semicircular canals that comprise the vestibular system are filled with endolymph.
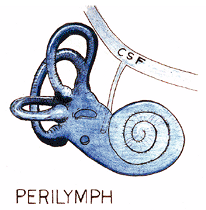 |
Figure 14.27 Schematic depicting the direct communication between the perilymphatic space and cerebrospinal fluid (CSF) through the cochlear aqueduct (perilymphatic duct). |
Utricle
The utricle is an elongated to oval-shaped portion of the membranous labyrinth, lying superior to the saccule in the medial wall of the vestibule (Figure 14.29). It is larger in diameter than the semicircular ducts and receives both ends of each semicircular duct (for a total of five, since the anterior and posterior ducts share a common opening). The macule of the utricle, located on the inferior surface of the utricle (utricular recess), is a sensory end organ. The utriculosaccular duct usually arises from the utricle; it communicates with the endolymphatic duct and connects the utricle to the saccule. As previously indicated, the three semicircular canals communicate with the utricle via openings formed by the union of the nondilated or nonampullary ends of the superior and posterior canals, termed the commun crus.
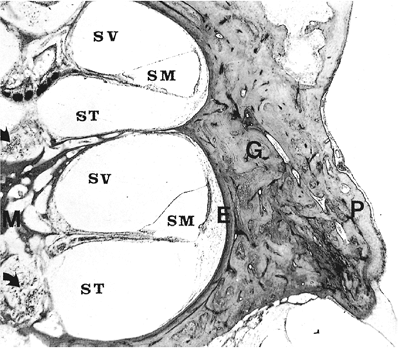 |
Figure 14.28 Cochlea, bony cochlea, and modiolus. (Arrows, spiral ganglion cells of basal and middle coils in modiolus; E, endosteal layer of bone; G, endochondral layer containing globuli interossei; M, modiolus; P, periosteal layer; SM, scala media; ST, scala tympani; SV, scala vestibuli) |
Saccule
The saccule is located anteromedial to the upper (anterior) end of the utricle (Figure 14.29) and tends to be more round than the utricle. The saccule and utricle are continuous via the utriculosaccular duct and with the cochlear duct by the ductus reuniens (also referred to as the canalis reuniens of Hensen) (1). The macule of the saccule contains
P.386
the sensory nerve endings of this portion of the inner ear and is an oval thickening on the lateral wall.
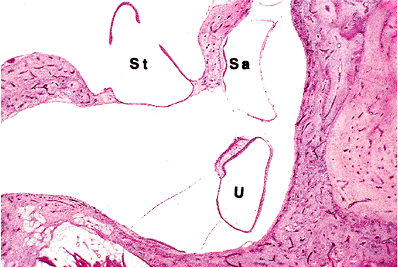 |
Figure 14.29 Ventricle of cat showing utricle (U) and saccule (Sa). (St, stapes) |
Semicircular Canals
The semicircular ducts include the anterior or superior duct, the posterior duct, and the lateral ducts. The end of each semicircular duct is expanded to form the ampulla. The anterior duct is directed anterolaterally, the posterior duct is directed posterolaterally, and the lateral ducts form a laterally directed angle of approximately 90 between themselves. The osseous canals follow a similar direction. The three semicircular canals communicate with the utricle via openings formed by the union of the nondilated or nonampullary ends of the superior and posterior canals (the common crus). From the common crus, the anterior duct curves upward, while the posterior duct curves backward and then downward. The other (membranous) ampullary ends of the semicircular canals contain the sensory endings of the ducts. At the ampullary ends, the anterior and posterior ducts empty into the utricle. The lateral semicircular duct lies in an approximate horizontal plane; both of its ends also connect to the utricle, with the anterior end being the ampulla.
Endolymphatic Duct and Sac
The endolymphatic duct traverses the medial portion of the petrous pyramid in its own bony canal, the vestibular aqueduct (7). The endolymphatic duct can be divided into segments. The first segment is the dilated portion referred to as the sinus, representing the common channel into which the utricular and saccular ducts open. The next segment of the duct is narrow and referred to as the isthmus. After the isthmus, the duct widens again to become the endolymphatic sac. The majority of the endolymphatic sac is located within the funnel-shaped cranial aperture of the cochlear duct, lying within a duplication of the posterior fossa dura and partially covered medially by a thin bony shelf referred to as the operculum. The endolymphatic sac ends in a terminal dilatation or fovea of the sac. Two portions of the endolymphatic sac are recognized: a proximal rugose portion with an irregular lumen caused by numerous folds of the epithelial lining and a distal portion with a smooth epithelial lining.
The membranous vestibular system contains the receptor organs for sense of motion and position. The neural structures of the inner ear, including the vestibulocochlear nerve and the facial nerve, enter the inner ear through the internal auditory canal.
Inner Ear Innervation
The nerve to the inner ear is cranial nerve VIII, variably referred to as the acoustic, auditory, or vestibulocochlear nerve. This nerve functionally consists of vestibular and cochlear divisions. In the internal auditory canal, these two parts are closely associated; but, at the lateral end of the canal, the nerve trunk divides into three parts, including two vestibular and one cochlear.
The vestibular nerve arises from the bipolar cells of the superior and inferior division of the (afferent) vestibular, or Scarpa's, ganglion located at the lateral end of the internal auditory canal. Peripherally, the vestibular nerve divides into two main divisions, the superior and inferior divisions. The superior part of the ganglion gives nerves to the ampullae of the lateral and anterior (superior) canals and to the saccular and utricle maculae. The inferior part of the ganglion gives rise to a posterior ampullary nerve and a nerve to the saccule. The inferior part also gives rise to a branch to the cochlear division.
The cells of origin for the cochlear nerve form the spiral ganglion, which represents the first of four neurons between the auditory end organ and the auditory cortex. The spiral ganglion is located in coils of the modiolus at the base of attachment of the osseous spiral lamina (see Figure 14.28). The osseous spiral lamina is a thin trabecula of bone surrounding afferent nerve fibers that run from the organ of Corti through the habenula perforata to the acoustic nerve and efferent fibers to the outer hair cells that arise from the olivocochlear system of Rasmussen. The efferent fibers make their exit from the brain with the vestibular part of the nerve and join the cochlear branch of cranial nerve VIII via the vestibulocochlear communicating branch, or nerve of Oort through the modiolus (1). Prior to reaching the modiolus, the nerve fibers are unmyelinated but are myelinated upon reaching the cochlear modiolus. The central fibers, or axons, of these bipolar neurons unite to form nerve bundles and pass from the cochlear modiolus through nerve channels in the osseous spiral foraminous tract into the internal auditory canal, where they form the cochlear nerve.
Within the internal auditory canal, the vestibulocochlear nerve is usually connected to the facial nerve. Together, the three nerves enter the posterior cranial fossa, transverse the cerebellopontine angle, and enter the brainstem at the posterior lower lateral aspect of the pons. The central auditory pathways consist of three additional neurons that form numerous connections with nuclei throughout the central nervous system as part of a complex auditory reflex system reaching the auditory cortex in the anterior transverse gyrus of the superior temporal lobe (7,8).
The facial nerve enters the temporal bone through the internal auditory meatus within the petrous portion of the temporal bone in company with (lying above) the vestibulocochlear nerve and the internal auditory artery. The facial nerve then passes Bill's bar, which represents a pointed bony projection separating the facial nerve from the superior division of the vestibular nerve. At the outer end of the canal, the facial nerve pierces arachnoid and dura to enter its own bony canal, the facial canal (fallopian
P.387
canal, or aqueduct of Fallopius). This canal continues for a short distance, and the facial nerve comes to lie just above the cochlea, where it bears the geniculate ganglion. The greater petrosal nerve comes off the geniculate ganglion and passes anteriorly and medially to enter the middle cranial fossa. Immediately beyond the geniculate ganglion, the facial nerve turns sharply (external genu; geniculum) laterally and posteriorly. As it runs backward in the bone of the lateral wall of the vestibule (which is the medial wall of the tympanic cavity), the facial nerve inclines downward and laterally where the bone surrounding it forms a bulge or projection, referred to as the prominence of the facial canal. This bulge or prominence is a normal finding, and it may be large enough to cover the oval window and base of the stapes. The facial nerve then makes a broad curve downward to run almost vertically through the mastoid process to the stylomastoid foramen, where the facial canal ends and the nerve exits from the skull.
Shortly before leaving the stylomastoid foramen, the facial nerve gives off the chorda tympani, which is composed of sensory and preganglionic motor fibers. Slightly above the stylomastoid foramen, the chorda tympani leaves the facial trunk and takes a recurrent course upward and forward in its canaliculus ( iter chordae posterius ) to enter the tympanic cavity through its posterior wall. Within the tympanic cavity, it passes between the malleus and incus and leaves the tympanic membrane through a canal in the pterygotympanic fissure ( iter chordae anterius ), where it joins the lingual nerve to be distributed to the anterior two-thirds of the tongue (taste buds) and to the submandibular ganglion, through which postganglionic fibers reach the submandibular and sublingual salivary glands.
Histology
Osseous Labyrinth
The cavity of osseous labyrinth (otic capsule) surrounds and replicates the outline of the membranous labyrinth lying within it. The osseous labyrinth is extremely dense and includes three layers: an outer periosteal layer; a middle layer in which there is persistence of much of the calcified cartilaginous matrix referred to as globuli interossei or globuli ossei (Figure 14.30); and an inner layer abutting the membranous labyrinth and lined by a thin layer of internal periosteum (also referred to as endosteum) (5). The density of the osseous labyrinth is necessary to insulate and safeguard the delicate vibrations of the fluids contained within it and is necessary in maintaining the integrity and functions of hearing and balance (5). As noted by Michaels (5) the bone of the adult osseous labyrinth is neither lamellar nor woven bone but somewhere in between ; in contrast to other adult bone, the osseous labyrinth lacks the normal developmental process of removal and replacement of calcified cartilaginous matrix and of primitive bone (5).
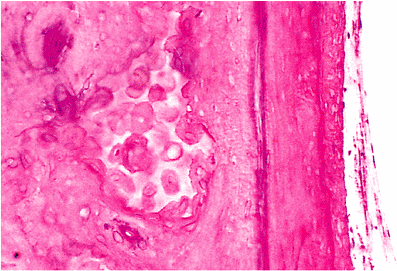 |
Figure 14.30 Globuli ossei (left) and endosteum (right) of cochlea. |
Membranous Labyrinth
The membranous labyrinth consists of epithelium-lined channels surrounded by connective tissue. The three basic divisions of the membranous labyrinth (including the semicircular canals, the utricle and saccule, and the cochlear duct) have similar structure, consisting of a specialized thickened epithelium surrounded by and attached to a fibrogelatinous membrane. The specialized epithelium consists of supporting cells and neuroepithelium or hair cells. The neuroepithelium has processes ( hairs ) that project from the free edge of the cells.
Cochlea
The organ of Corti consists of neurotransmitting hair cells that rest on the basilar membrane and is arranged in a spiral like the duct itself (Figure 14.31). The organ of Corti consists of supporting cells and hair cells. The supporting, or pillar, cells are of several different types. Among the more important supporting cells are the phalangeal cells, which are arranged in two groups: an inner or single row of cells and an outer row of cells (cells of Deiters) formed from three to five rows of cells, depending on the level of the cochlea, with more rows of cells toward the apex and fewer rows of cells toward the base.
The inner row of phalangeal cells is associated with a single layer of hair cells; the outer row of phalangeal cells alternate with rows of hair cells. The phalangeal cells get their name from the shape of the stiff processes that project
P.388
from the cells contributing to the reticular membrane that covers the free surface of the organ (1). The hair cells have numerous (40 to 100 per cell) hairs projecting from the reticular (cuticular) surface. The outer hair cells are more sensitive, are short and wedge-shaped between the apices of the phalangeal cells in order to reach the basilar membrane, and are believed to be responsible for the cochlear microphonics (1). The inner hair cells are long, are less susceptible to damage than the outer hair cells, and believed to be less sensitive to sound.
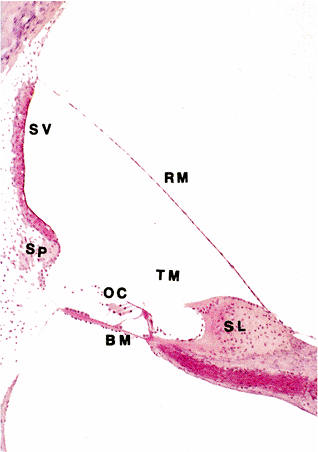 |
Figure 14.31 Scala media of cat. (BM, basilar membrane; OC, organ of Corti; RM, Reissner's membrane; SL, spiral limbus; SP, spiral prominence; SV, stria vascularis; TM, tectorial membrane) |
Intercellular spaces among the cells of the organ of Corti are apparently filled with intercellular substance. The largest of these spaces runs the entire length of the organ of Corti between inner and outer rows of phalangeal and hair cells and is referred to as the tunnel, or canal, of Corti (Figure 14.32). The tunnel of Corti is bounded by special supporting cells, the inner and outer pillars (Corti's rods) (Figure 14.32). The tunnel and pillars together form Corti's arch.
The basilar membrane is fibrous tissue that supports the organ of Corti. The basilar membrane has the tectorial membrane attached to it. The basilar membrane has fibers that pass from the bony spiral lamina to the spiral crest of the spiral ligament. The basilar membrane increases in size from the base to the apex of the cochlea and is felt to have resonator action with deformation of the membrane by sound, beginning at its lower end and traveling toward the helicotrema. The tectorial membrane is a gelatinous structure with numerous fine fibers. Like the basilar membrane, the tectorial membrane increases in size from the base to the apex of the cochlea and is believed to have a vibratory effect on the hair cells.
Together, the organ of Corti and the basilar membrane form the spiral membrane, which is the floor or tympanic wall of the cochlear duct. The spiral ligament is a thickened, modified portion of periosteum of the osseous cochlea, which forms the outer or curved wall of the cochlear duct and adjacent parts of the scalae. The vestibular, or Reissner's, membrane is thin and consists of two layers of cells: an inner cell layer of ectodermal origin (consisting of epithelial-like clusters) and an outer layer of mesodermal origin (consisting of large, flat, and elongated cells) (5). This membrane forms the roof of the cochlear duct. In M ni re's disease (see below), the vestibular membrane bulges toward the scala vestibuli. In the outer (vertical) wall of the cochlear duct is the stria vascularis, which is supplied by 30 to 35 small arteries that originate from the modiolar region of the scala vestibuli and pass outward to the lateral wall of the osseous labyrinth (Figure 14.31) (22). It is believed to be the source of the endolymph (23). The tissue spaces of the spiral ligament serve as a site of absorption (24). The stria vascularis is altered in ototoxic conditions, as may occur secondary to use of cisplatin, diuretic agents, and other drugs (see below under Presbycusis and Other Hearing Loss) (5).
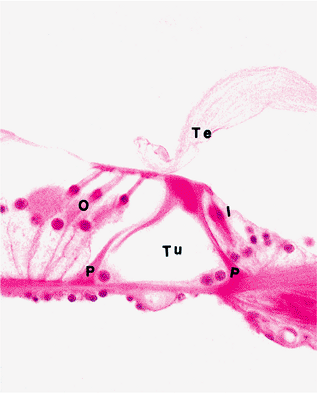 |
Figure 14.32 Higher power view of the organ of Corti (see Figure 14.31). (I, inner hair cells; O, outer hair cells; P, pillar cells (walls of tunnel); Te, tectorial membrane; Tu, tunnel of Corti) |
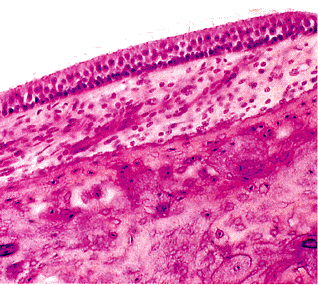 |
Figure 14.33 Higher power of a part of Figure 14.29, showing macula of the saccule. |
P.389
Semicircular Canals, Utricle, and Saccule
The end of each semicircular duct is expanded to form an ampulla. The sensory endings in the ampullae of the ducts are the cristae. Each crista consists of thickened epithelium; above each crista rests a gelatinous formation of viscous protein polysaccharide called the cupola. The hairs of the neuroepitheial hair cells project into the base of the cupola. As a result of the gelatinous nature of the cupola, it may be bent by the pressure of the endolymph, which apparently stimulates the hair cells and, therefore, the nerve endings of the cristae.
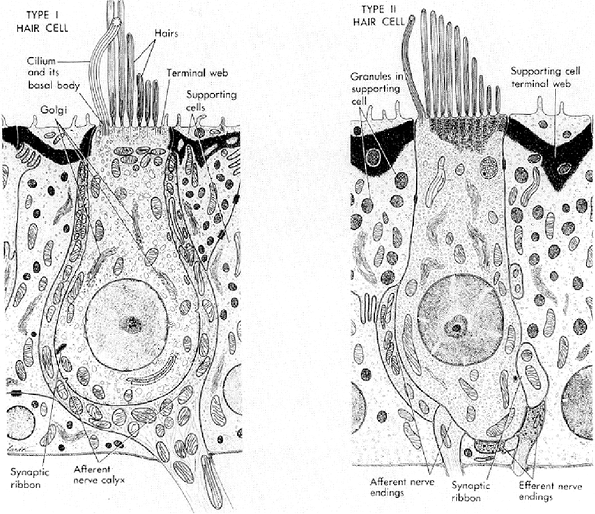 |
Figure 14.34 Schematic representation of the ultrastructure of vestibular hair cells showing the principal features of type I and type II hair cells and their supporting cells (Reprinted with permission from: Nager GT. Anatomy of the membranous cochlea and vestibular labyrinth. In: Nager GT, ed. Pathology of the Ear and Temporal Bone. Baltimore: Williams & Wilkins; 1993:3 48 ). |
The utricle and saccule, representing the two main membranous structures of the vestibule, are lined by a sensory epithelium known as the macula (Figure 14.33). The maculae are identical to one another in structure and are similar to the cristae of the semicircular canals. By transmission electron microscopy, these sensory cells are of two types: type I cells are flask-shaped with a swollen basal portion; type II cell is cylindrical. Type I cells are attached to fibers of the sensory nerves by a wide chalicelike terminal, and the terminal of type II cells is connected by buttonlike attachments of the nerve (Figure 14.34) (5). The sensory epithelium consists of hair cells, which in turn have stiff, immotile projections embedded in the gelatinous otolithic membrane. Also embedded in the otolithic membrane are crystalline bodies, referred to as otoliths, that contain calcium carbonate and a protein suspended in a jellylike polysaccharide. It is only in the presence of otoliths that the maculae differ from the other sensory areas of the ear.
 |
Figure 14.35 Schematic illustration of the cranial and spinal nerves showing that most of the nerves have glia extending for only a fraction of a millimeter beyond their external origins with the exceptions of the optic nerve, which is really a tract of brain (given the presence of neuroglia throughout its length), and the vestibulocochlear nerve (cranial nerve VIII). The vestibulocochlear nerve typically has glia extending from 6 to 8 mm along its course. (From: Tarlov IM. Structure of the nerve root. II. Differentiation of sensory from motor roots: observations on identification of function in roots of mixed cranial nerves. Arch Neurol Psychiatr 1937;37:1338 1355 ). |
P.390
Nerves and Paraganglia
Most cranial and spinal nerves have glia extending only a fraction of a millimeter beyond their external origins (25,26). The optic nerve contains neuroglia throughout its length and, thereby, really is a tract of brain rather than a true nerve. The exception to the other cranial nerves is the eighth (vestibulocochlear) nerve, which typically has glia extending from 6 to 8 mm along its course (Figure 14.35). This distribution of glia along cranial nerve VIII helps explain the greater occurrence of glial tumors on this nerve as compared to the other cranial nerves (25). The vestibular and cochlear divisions are fused near the entrance to the internal auditory meatus; at this location, the nerve changes in appearance from pale staining proximally to dark staining distally. This change in appearance is the result of the abrupt transition of the coverings of the nerve fibers from the pale staining oligodendroglia to the darker staining Schwann cells (Figure 14.36). This glial-Schwann sheath junction of the cranial nerve VIII is referred to as the Obersteiner-Redlich zone. Acoustic neuromas (also referred to as vestibular neuromas) may arise anywhere between this junction and the cribrosa area at the fundus of the canal (27).
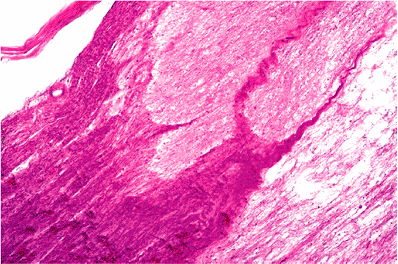 |
Figure 14.36 The vestibular and cochlear divisions of the vestibulocochlear nerve are fused near the entrance to the internal auditory meatus. At this location is the glial-Schwann sheath junction, also referred to as the Obersteiner-Redlich zone, where the nerve changes in appearance from pale staining proximally to dark staining distally as a result of the abrupt transition of the coverings of the nerve fibers from the pale staining oligodendroglia to the darker staining Schwann cells. |
Paraganglia similar in structure to the carotid body are identified in the ear and may give rise to jugulotympanic paragangliomas. The majority of paraganglia are found in relation to the jugular bulb, and a minority are found under the mucosa of the medial side of the middle ear promontory (Figure 14.37).
Endolymphatic Sac and Duct
The lining epithelium of the endolymphatic duct is low cuboidal (Figure 14.38), and the epithelium of the endolymphatic sac is taller and has a papillary appearance (Figure 14.39). An aggressive neoplasm, termed the endolymphatic sac papillary tumor, is presumed to originate from the endolymphatic sac epithelium (28). This tumor, initially considered to represent a low-grade malignancy (i.e., adenocarcinoma), is potentially a locally destructive but not metastatic tumor characterized by variably appearing epithelium, including nondescript low-cuboidal to
P.391
papillary and glandular appearing neoplasm (28,29). Patients with this tumor often describe symptoms similar to those occurring in M ni re's disease, including vertigo and spinning of the room. This tumor has been found to be associated with von Hippel-Lindau (VHL) syndrome, including the identification of the VHL gene (30,31).
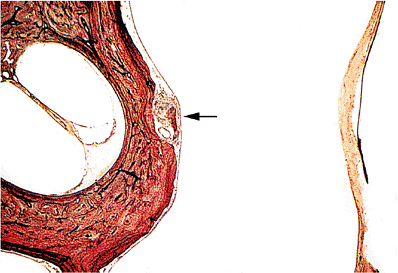 |
Figure 14.37 Normal tympanic paraganglion (arrow) under mucosa of medial side of middle ear over promontory. The tympanic membrane is on the right (Gomori's reticulin stain). |
Composition and Circulation of the Perilymph and Endolymph
Perilymph, which is partly a filtration of cerebrospinal fluid (CSF) and partly a filtration from blood vessels of the ear, has a similar chemical composition as CSF, resembling extracellular fluid with low potassium and high sodium concentrations. The similarities of perilymph and CSF support the concept that perilymph is derived from CSF. The anatomic basis for this concept is based on the consideration that the cochlear aqueduct (perilymphatic duct) opens into both the subarachnoid and perilymphatic spaces (1). An increase in CSF pressure results in flow into the labyrinth.
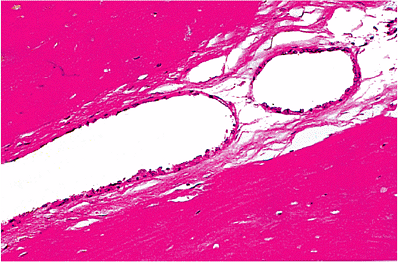 |
Figure 14.38 Endolymphatic duct within the vestibular aqueduct. The duct is lined by low cuboidal epithelium. |
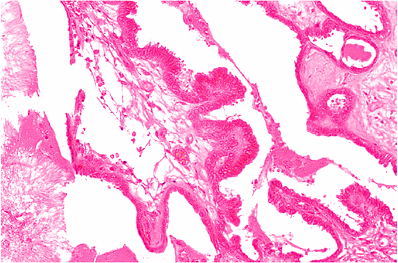 |
Figure 14.39 Endolymphatic sac, which is lined by tall columnar epithelium arranged on papillae. |
The perilymphatic spaces of each osseous semicircular canal are continuous on both ends with the perilymphatic space of the vestibule, and this space is continuous with the scala vestibuli, which is continuous with the scala tympani at the helicotrema. All perilymphatic spaces open widely into each other. Due to areas of discontinuity or deficiency in the compact bone of the petrous portion of the temporal bone, foci of communication may exist between the perilymphatic space and other cavities. Such areas of potential communication include the middle and inner ear via the round and oval windows. In addition, the vestibular and cochlear aqueducts and the foramina for the nerves and blood vessels of the inner ear serve as potential channels between the inner ear and the cranial cavity.
Endolymph is an intracellular-like fluid containing high potassium and low sodium concentrations. Endolymph contains more than 30 times as much potassium as does perilymph or CSF but about one-tenth as much sodium (32). Endolymph has a low protein content; its protein is entirely globulin instead of an admixture of globulin and albumin (1). It has a viscosity similar to the vitreous of the eye because of its high mucopolysaccharide content. The electrolyte concentration of the endolymph is critical for normal functioning of the sensory organs. It is generally believed that the main sources of endolymph are the stria vascularis, the epithelium of the ampullae of the semicircular ducts, and the epithelium of the maculae of the utricle and saccule.
Endolymph circulates through the cochlear duct (scala media) downward to the base of the cochlea, through the ductus reuniens into the saccule, and then into the endolymphatic sac and duct, where it is reabsorbed. The cochlear duct communicates with the vestibular endolymph-containing sacs through two canals so that the endolymphatic system is, like the perilymphatic system, a continuous one.
P.392
Conduction of Sound
Conduction of sound occurs via air and via bone. The pinna and external auditory canal conduct sound waves in air to the tympanic membrane. Conduction of sound by air is less efficient as compared with the ossicular route. The ossicular chain, including the malleus, incus, and stapes, enhances the sound energy transmission by conveying vibrations from the tympanic membrane to the footplate of the stapes and hence through the oval window of the vestibule to the perilymph. From the vestibular perilymph, vibrations derived from sound waves pass directly to the perilymphatic spaces of the cochlea, first via the scala vestibuli (upper compartment) ascending from the oval window, and then to the scala tympani (lower compartment) descending to the round window. The walls of the scala media (i.e., the cochlear duct, which contains endolymph) receive waves of vibrations from the adjacent perilymph (containing the scala vestibuli and scala tympani). Through the endolymph, the waves of vibrations affect the sensory cells of the organ of Corti, the sensory organ of sound reception located in the scala media, from which it passes to the cochlear nerve with transmission via central pathways to the cerebral cortex.
Selected Abnormalities and Pathology
External Ear
Abnormalities of the external ear include those associated with first and second branchial arch syndromes. First and second branchial arch syndromes include otologic and nonotologic abnormalities. The otologic manifestations or abnormalities include malformed or absent external ears, atretic external auditory canal, and impaired hearing. The nonotologic abnormalities include asymmetric facies, abnormalities of the temporomandibular joint, neuromuscular abnormalities, and associated abnormalities of the cardiovascular, renal, and central nervous systems. Goldenhar's syndrome, also known as oculoauriculovertebral dysplasia, is a first and second branchial arch syndrome characterized by ear tags, preauricular pits and fissures, epidermoids, lipodermoids, and vertebral column abnormalities (33).
Ear abnormalities can also be seen in association with other abnormalities, including Down's syndrome (Figure 14.40). Cryptotia is a rare anomaly in which the superior portion of the auricle is buried in the scalp (Figure 14.41). Microtia represents gross hypoplasia of the pinna with a blind or absent external auditory canal. Microtia is typically bilateral although the degree of hypoplasia may differ on the two sides.
Accessory tragi (also referred to as accessory or supernumerary ears, accessory auricle, or polyotia) appear at birth and may be solitary or multiple, unilateral or bilateral, sessile or pedunculated, and soft or cartilaginous skin-covered nodules or papules. They are located on the skin surface, often anterior to the auricle, and may be mistaken clinically for a papilloma. Histologically, accessory tragi recapitulate the normal external auricle and include skin, cutaneous adnexal structures, and a central core of cartilage. Accessory tragi are thought to be related to second branchial arch anomalies. They may occur independently of other congenital anomalies but also may occur in association with cleft palate or lip, mandibular hypoplasia, or other anomalies such as Goldenhar's syndrome (oculoauriculovertebral dysplasia) (33).
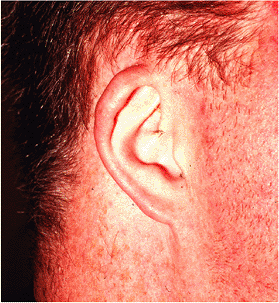 |
Figure 14.40 Individual with Down's syndrome. In comparison to non-Down's syndrome individuals, patients with Down's syndrome have external ears that are smaller, low set, and have an incompletely developed superior helix. |
In adults, diagonal earlobe crease has been associated with coronary artery disease and has been referred to as Frank's sign (34). The crease runs diagonally backward and downward across the lateral surface of the earlobe from the external meatus (Figure 14.42). Depending on the extent and depth of the crease, three grades have been assigned, with grade 1 being the least obvious appearing crease, grade 2 including a superficial crease across 100% of the earlobe or a deep crease across 50% of the earlobe, and grade 3 represented by a deep crease along 100% of the earlobe. Bilateral grades 2 and 3 creases are associated with a significantly higher risk of death from atherosclerosis and myocardial infarction (35).
Middle Ear
Otitis Media
Otitis media is either an acute or chronic infectious disease of the middle ear space. Otitis media is predominantly, but not exclusively, a childhood disease. The most common
P.393
microorganisms implicated in causing disease of the middle ear are Streptococcus pneumoniae and Haemophilus influenzae (36). Otoscopic examination reveals a hyperemic, opaque, bulging tympanic membrane with limited mobility; purulent otorrhea may be present. Bilateral involvement is not uncommon. The middle ear infection is felt to result from infection via the auditory tube at the time of or following a pharyngitis (bacterial or viral).
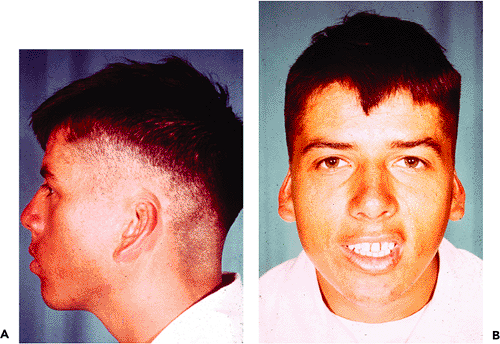 |
Figure 14.41 A. and B. Individual with cryptotia, a rare anomaly in which the superior portion of the auricle is buried in the scalp. |
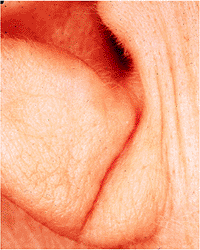 |
Figure 14.42 Individual with a grade 3 earlobe crease represented by a deep crease along 100% of the earlobe. This person had a similar earlobe crease of the opposite ear. Bilateral grades 2 and 3 creases are associated with a significantly higher risk of death from atherosclerosis and myocardial infarction. |
In general, otitis media is managed medically. However, at times tissue is removed for histopathologic examination. The pathologic alterations are generally straightforward, but secondary changes such as glandular metaplasia of the surface epithelium (the result of chronic infection) may occur that might be confused with a true gland-forming neoplasm.
The histologic changes in chronic otitis media include a variable amount of chronic inflammatory cells, consisting of lymphocytes, histiocytes, plasma cells, and eosinophils. Multinucleated giant cells and foamy histiocytes may be present. The middle ear low cuboidal epithelium may or may not be seen. However, glandular metaplasia, a response of the middle ear epithelium to the infectious process, may be present (Figure 14.43). The glands tend to be more common in nonsuppurative otitis media than in suppurative otitis media. The metaplastic glands are unevenly distributed in the tissue specimens, are variably shaped, and are separated by abundant stromal tissue. The glands are lined by a columnar to cuboidal epithelium, with or without cilia or goblet cell metaplasia. Glandular secretions may or may not be present so that the glands may appear empty or contain varying
P.394
secretions, including thin (serous) or thick (mucoid) fluid content. The identification of cilia is confirmatory of middle ear glandular metaplasia and is a feature is not found in association with middle ear adenomas (36). Further, the haphazard arrangement of the glands in the background of changes of chronic otitis media should allow for differentiating metaplastic from neoplastic glands. Acute inflammatory cells may be superimposed by chronic otitis media.
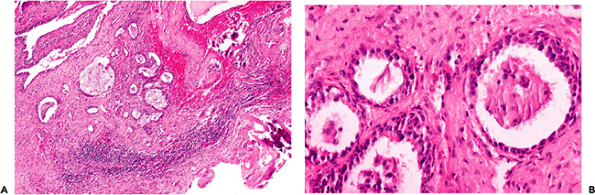 |
Figure 14.43 Under normal conditions, glands are not identified in the middle ear space; however, glandular metaplasia can be found in the setting of otitis media. A. Otitis media, showing chronic inflammation, fibrosis, glandular metaplasia, and foci of calcifications (lower right); residual normal cuboidal epithelium of the middle ear is seen in the upper left. B. Higher magnification showing glandular metaplasia is the setting of chronic otitis media. |
In addition to the inflammatory cell infiltrate and glandular metaplasia, other histopathologic findings can be seen in association with chronic otitis media (or represent sequelae of chronic otitis media) and include fibrosis, granulation tissue, tympanosclerosis, cholesterol granulomas, and reactive bone formation. Due to the presence of scar tissue, the middle ear ossicles may be destroyed (partial or total) or may become immobilized. Perforation of the tympanic membrane pars tensa may occur with resulting ingrowth of squamous epithelium, potentially leading to the development of cholesteatoma (see below).
Tympanosclerosis represents dystrophic mineralization (calcification or ossification) of the tympanic membrane or middle ear that is associated with recurrent episodes of otitis media (37). The incidence of tympanosclerosis in otitis media varies from 3 to 33% (37). Tympanosclerosis of the tympanic membrane can be seen in children following myringotomy and tube insertion. In this setting, the tympanosclerotic foci may or may not be permanent. Tympanosclerosis of the middle ear typically affects older patients, represents irreversible accumulation of mineralized material, and is associated with conductive hearing loss (38,39).
On gross examination, tympanosclerotic foci may be localized or diffuse and appear as white nodules or plaques (Figure 14.44). Histologically, dense clumps of mineralized, calcified, or ossified material or debris can be seen within the stromal tissues or in the middle (connective tissue) aspect of the tympanic membrane (Figure 14.45). Tympanosclerosis may cause scarring and ossicular fixation.
Cholesterol granuloma is a histologic designation describing the presence of a foreign body granulomatous response to cholesterol crystals derived from the rupture of red blood cells with breakdown of the lipid layer of the erythrocyte cell membrane. Cholesterol granulomas arise in the middle ear and mastoid in any condition in which there is hemorrhage combined with interference in drainage and ventilation of the middle ear space (40). Cholesterol granuloma of the middle ear may present as idiopathic hemotympanum; patients may also complain of hearing loss and tinnitus. The majority of cholesterol granulomas in the middle ear and temporal bone have an indolent biologic behavior and cause no significant bone resorption (40).
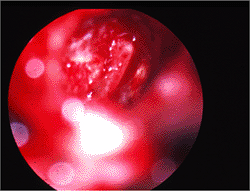 |
Figure 14.44 Tympanic membrane in tympanosclerosis showing calcified plaque on the tympanic membrane. |
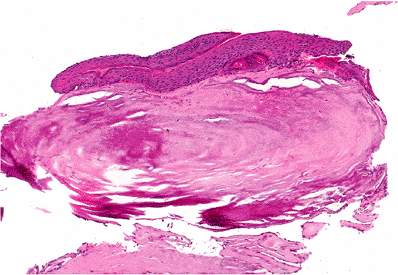 |
Figure 14.45 Tympanosclerosis. The tympanic membrane is thickened and calcified and is covered on its external (external auditory canal) aspect by keratinizing squamous epithelium (top) and on its internal (tympanic cavity) aspect by cuboidal epithelium (bottom right). |
In contrast to cholesterol granulomas of the middle ear and temporal bone, cholesterol granulomas of the petrous apex may behave aggressively, producing a tumorlike mass with expansion of the cyst and erosion/destruction of adjacent structures. Depending on the direction of expansion, apical cholesterol granulomas may invade into the cochlea, cerebellopontine angle, jugular foramen, cranial nerves V to XI, brain stem, and cerebellum, producing life-threatening symptoms (41). Involvement of the petrous apex is more likely to be associated with sensorineural hearing loss; additional signs and symptoms may include headaches, cranial nerve deficits, and bone erosion with involvement of the posterior or middle cranial fossa (41,42). On axial computed tomography, apical cholesterol granulomas appear as round to ovoid to irregular-shaped cysts with smooth margins and evidence of bone remodeling.
The histology of cholesterol granulomas is the same irrespective of location and includes the presence of irregular-shaped clear-appearing spaces surrounded by histiocytes and/or multinucleated giant cells (foreign body granuloma) (Figure 14.46). Cholesterol granulomas are not related to cholesteatomas but may occur in association with or independent of a cholesteatoma.
Cholesteatoma (Keratoma)
Cholesteatoma is a pseudoneoplastic lesion of the middle ear characterized by the presence of stratified squamous epithelium that forms a saclike accumulation of keratin within the middle ear space (akin to an epidermal inclusion cyst). Despite their invasive growth, cholesteatomas are not considered to be true neoplasms. The term cholesteatoma is a misnomer in that it is not a neoplasm nor does it contain cholesterol. Perhaps the designation of keratoma would be more accurate, but the term cholesteatoma is entrenched in the literature. In the middle ear and inner ear, cholesteatomas take three forms: acquired cholesteatoma, congenital cholesteatoma, and cholesteatoma of the petrous apex. Depending on the site of origin in the tympanic membrane, each of these cholesteatomas may be subdivided into pars flaccida (Schrapnell's membrane) and pars tensa cholesteatomas.
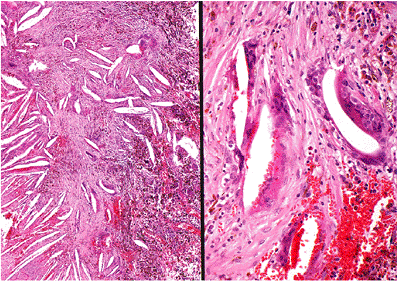 |
Figure 14.46 Cholesterol granuloma appears as empty, irregularly shaped clefts or spaces surrounded by histiocytes and multinucleated giant cells. Fresh hemorrhage and hemosiderin pigment are readily apparent. |
Acquired Cholesteatoma
Acquired cholesteatoma is the most common type of cholesteatoma. It tends to be more common in men than in women and occur in older children and young adults. Acquired cholesteatoma is derived from entry of external ear canal epidermis into the middle ear. The latter may occur in one of several ways: via perforation of the tympanic membrane, following localized retraction of the tympanic membrane with epithelial invagination or ingrowth of a band of stratified squamous epithelium into the middle ear; via entrapment of squamous epithelium following surgery and/or trauma; or via squamous metaplasia of the middle ear mucosa (43). A decrease in middle ear pressure can induce retraction of certain regions of the tympanic membrane in the pars flaccida, pars tensa, or both (43). Retraction pockets are felt to represent the precursors for the development of cholesteatoma (Figure 14.47) (44,45). Dysfunction of the auditory tube leading to chronic (recurrent) otitis media is felt to represent a causative factor (43).
The upper posterior part of the middle ear space is the most common site of acquired cholesteatoma. Initially,
P.395
cholesteatomas may remain clinically silent until extensive invasion of the middle ear space and mastoid occurs. Symptoms include hearing loss, malodorous discharge, and pain and may be associated with a polyp arising in the attic of the middle ear or perforation of the tympanic membrane. Otoscopic examination may reveal the presence of white debris within the middle ear, which is considered diagnostic.
 |
Figure 14.47 Section of malleus from an adult ear with a retraction pocket of the tympanic membrane at autopsy. There is a thin layer of stratified squamous epithelium between the bone and middle ear epithelium. This was found on serial section to be an ingrowth of the stratified squamous epithelium from the outer epithelial covering of the retraction pocket. |
Congenital Cholesteatoma
Congenital cholesteatoma is a cholesteatoma of the middle ear that exists in the presence of an intact tympanic membrane, presumably occurring in the absence of chronic otitis media that may result in perforation or retraction of the tympanic membrane. Congenital cholesteatomas are found in infants and young children. Small colonies of epidermoid cells, referred to as epidermoid formations, are found on the lateral anterior superior surface of the middle ear in temporal bones after 15 weeks gestation (43). During the first postpartum year, the epidermoid colonies disappear; however, if the epidermoid cells do not disappear but continue to grow, they will become a congenital cholesteatoma. The latter have also been referred to as epidermoid cysts (46). In the majority of cases, congenital cholesteatomas are found in the anterosuperior part of the middle ear. Early lesions show no symptoms and are discovered by otoscopic examination. In later lesions, the signs and symptoms may be the same as acquired cholesteatoma.
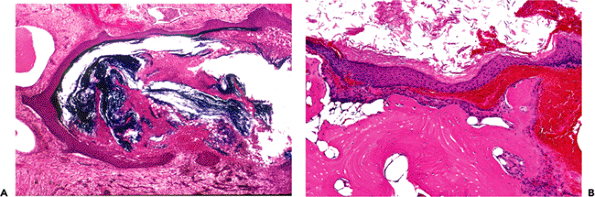 |
Figure 14.48 Cholesteatoma of the middle ear. A. The histologic diagnosis of cholesteatoma is based on the presence of finding keratinizing squamous epithelium within the middle ear space. B. Osseous involvement. |
Cholesteatoma of the Petrous Apex
Cholesteatoma of the petrous apex is an epidermoid cyst of this location and bears no relation to cholesteatoma of the middle ear. It is likely of congenital origin, but no cell rests have been discovered that may explain the origin of these lesions. Symptoms usually relate to involvement of the cranial nerves VII and VIII in the cerebellopontine angle (46).
Pathology
Cholesteatomas appear as cystic, white to pearly appearing masses of varying size that contain creamy or waxy granular material. The histologic diagnosis of cholesteatoma is made in the presence of a stratified keratinizing squamous epithelium, subepithelial fibroconnective or granulation tissue, and keratin debris (Figure 14.48). The essential diagnostic feature is the keratinizing squamous epithelium, and the presence of keratin debris alone is not diagnostic of a cholesteatoma. The keratinizing squamous epithelium is
P.396
cytologically bland and shows cellular maturation without evidence of dysplasia. In spite of its benign histology, cholesteatomas are invasive and have widespread destructive capabilities.
The destructive properties of cholesteatomas result from a combination of interrelated reasons, including mass effect with pressure erosion of surrounding structures from the cholesteatoma, the production of collagenase (which has osteodestructive capabilities by its resorption of bony structures), and bone resorption (20). Collagenase is produced by both the squamous epithelial and the fibrous tissue components of the cholesteatoma. This local aggressive behavior is the result of the continuing accumulation of the cholesteatomatous material with progressive erosion of surrounding structures. Depending on the location and extent of the cholesteatoma, erosion may include the lateral wall of the attic, the middle ear ossicles, the tegmental bone over the attic and antrum, and the mastoid cortex (43). Less frequent progression includes erosion of the lateral sinus and jugular bulb, the vestibular and cochlear capsules, the facial canal, the dura of the middle and posterior cranial fossa, the semicircular canals, and the facial nerve (43). Sequelae of such erosions may include semicircular canal fistulas, exposed tympanic facial nerve, or brain herniation through the tegmen.
The histologic diagnosis of cholesteatomas is relatively straightforward in the presence of keratinizing squamous epithelium. In contrast to cholesteatomas, squamous cell carcinoma shows dysplastic or overtly malignant cytologic features with a prominent desmoplastic stromal response to its infiltrative growth. Cholesteatomas do not transform into squamous cell carcinomas. In an attempt to determine whether cholesteatomas were low-grade squamous carcinomas, Desloge et al. (47) performed DNA analysis on human cholesteatomas to determine whether ploidy abnormalities were present. In ten cases with interpretable data, nine were euploid and one was aneuploid. These authors concluded that, due to a lack of overt genetic instability, cholesteatomas could not be considered to be malignant neoplasms. Cholesterol granuloma is not synonymous with cholesteatoma. These entities are distinctly different pathologic entities and should not be confused with one another.
Otosclerosis
Otosclerosis is a disorder of the bony labyrinth and stapedial footplate that exclusively occurs in humans and is of unknown etiology. Otosclerosis means hardening of the ear and is derived from Greek (ous, ear; skleros, hard; osis, condition); osseous ankylosis (from the Greek ankoulon, to stiffen); chronic metaplastic ostitis; progressive otospongiosis. Otosclerosis primarily causes conductive hearing loss that usually begins in the second and third decades of life and is slowly progressive. The extent of the hearing loss directly correlates with the degree of stapedial footplate fixation. It is not uncommon for patients with otosclerosis also to have vestibular disturbances (48,49). Otosclerosis usually involves both ears; however, unilateral disease can occur in up to 15% of cases (50).
Although many theories regarding the etiology of otosclerosis appear in the literature, there is no consensus. Hereditary factors are often cited as among the causes. Surgical management of the conductive hearing loss caused by stapes fixation (stapedectomy) is the treatment of choice, with replacement of the fixed stapes by a prosthesis. The resected bone may include the entire stapes (with the footplate) or only the superstructure that includes the head and crura (without the footplate).
Histologically, the initial alterations include resorption of bone around blood vessels. The cellular fibrovascular tissue replaces the resorbed bone, resulting in softening of the bone (otospongiosis). Immature bone is laid down with continuous active resorption and remodeling. The new bone is rich in ground substance and deficient in collagen; but, over time, more mature bone with increased collagen and less ground substance is produced, resulting in densely sclerotic bone. This process most often begins from the adjacent temporal bone (anterior to the oval window) and eventually involves the footplate of the stapes moving across the annulus fibrosus or stapediovestibular joint (Figure 14.49). Stapedial involvement causes fixation of the stapes, with inability to transmit sound waves resulting in conductive hearing loss. While the otosclerotic changes can be seen in the resected stapedial footplate, even when the footplate is removed intact it may be free of otosclerotic changes as fixation results via pressure on the nonotosclerotic footplate from swelling of the otosclerotic process in the adjacent temporal bone (7).
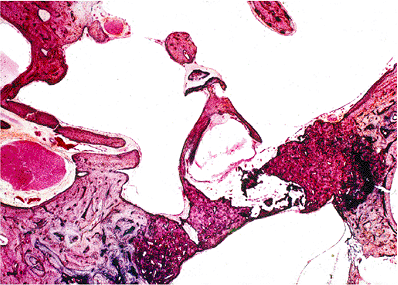 |
Figure 14.49 Otosclerosis of the temporal bone (anterior to the oval window) involving the footplate of the stapes moving across the annulus fibrosus or stapediovestibular joint. Stapedial involvement causes fixation of the stapes, with inability to transmit sound waves resulting in conductive hearing loss. |
 |
Figure 14.50 Causes of conductive hearing loss. |
P.397
Inner Ear
Presbycusis and Other Hearing Loss
Hearing loss may include conductive hearing loss and sensorineural hearing loss. There are many causes for conductive and sensorineural hearing loss, respectively (Figures 14.50,14.51). Hearing loss that occurs with increasing age is referred to as presbycusis. There is still controversy as to the underlying electrophysiologic and histopathologic alterations associated with the development of presbycusis. Degenerative alterations within various microanatomic structures of the cochlea, including the hair cells, spiral ganglion cells, stria vascularis, and basilar membrane, have been invoked as the cause of presbycusis (51). Alternatively, damage to the outer hair cells alone has been invoked as the cause of presbycusis (52,53). As Soucek and colleagues have shown in their studies of aged ears (52,53), the histopathologic changes include scanty to absent outer hair cells of the third row (Figure 14.52), with complete loss of both inner and outer hair cells of all rows at the extreme lower end of the basal coil. In contrast to these aforementioned sites, Soucek and colleagues found that the inner hair cells sustained minimal loss, the first row of outer hair cells had
P.398
greater loss, and the second row more loss but not to the extent seen in the third row of outer hair cells or at the extreme lower end of the basal coil (54,55). In addition, these authors also found the presence of enormously lengthened and thickened sterocilia and giant stereocilia, which they felt contributing to the development of presbycusis.
 |
Figure 14.51 Cause of sensorineural hearing loss. |
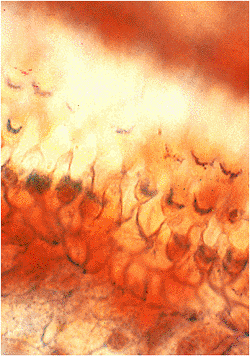 |
Figure 14.52 Surface preparation of outer hair cells from basal coil of cochlea of an elderly man. There are many gaps among the hair cells of the first two rows. (P, pillar cells) (Osmic acid, Alcian blue, and phloxine eosin, oil immersion.) |
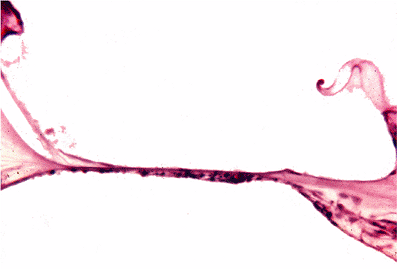 |
Figure 14.53 Viral labyrinthitis- (end stage) related hearing loss with total degeneration of the organ of Corti. |
Degenerative alterations within various microanatomic structures of the membranous labyrinth resulting in sensorineural hearing loss may occur secondary to infectious disease (Figure 14.53), metabolic abnormalities (Figure 14.54), trauma (Figure 14.55), sensory presbycusis (Figure 14.56), and use of certain medications, including cisplatin, diuretic agents, and other drugs (Figures 14.57,14.58).
M ni re's Disease
M ni re's disease is an idiopathic disorder of the inner ear associated with a symptom complex of spontaneous,
P.399
episodic attacks of vertigo, sensorineural hearing loss, tinnitus, and a sensation of aural fullness. The onset of vertigo is frequently sudden, reaching maximum intensity within a few minutes, lasting from an hour or more, and either subsiding completely or continuing as a sensation of unsteadiness for hours to days.
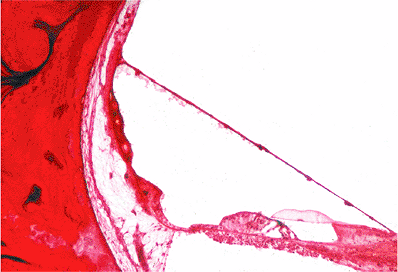 |
Figure 14.54 Metabolic-related hearing loss in a patient with diabetes is characterized by the presence of hyalinized vasculature structures in the stria vascularis. |
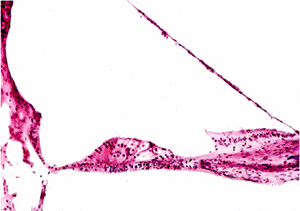 |
Figure 14.55 Posttraumatic hearing loss with focal avulsion of the organ of Corti. |
 |
Figure 14.56 Sensory presbycusis characterized by the loss of hair cells in the organ of Corti. |
 |
Figure 14.57 Strial presbycusis with atrophy; this is secondary to ototoxic effect caused by cisplatin therapy. |
 |
Figure 14.58 Kanamycin ototoxic effects on the organ of Corti, including loss of hair cells. |
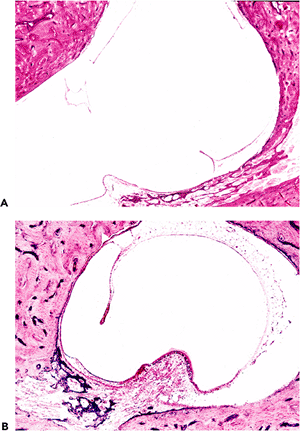 |
Figure 14.59 M ni re's disease showing dilatation (A) and rupture of the membranous labyrinth (B). |
The pathogenesis of M ni re's disease is distortion of the membranous labyrinth, defined as changes in the microanatomy of the membranous labyrinth as a consequence of the over-accumulation of endolymph (endolymphatic hydrops) and at the expense of the perilymphatic space (54,55). Endolymph, which is produced by the stria vascularis in the cochlea and by cells in the vestibular labyrinth, circulates in a radial and longitudinal fashion. In patients with M ni re's disease, it is believed that there is inadequate absorption of endolymph by the endolymphatic sac (55).
In the early stages of the disease, endolymphatic hydrops primarily involves the cochlear duct and saccule, but in the later stages the entire endolymphatic system is involved. Alterations of the membranous labyrinth include dilatation, outpouching, rupture, and collapse (Figure 14.59). Fistulae (unhealed ruptures) may occur. Severe cytoarchitectural and atrophic changes may occur in the sense organs with loss of neurons in the cochlea.
Temporal Bone Dissection
For a detailed discussion of proper postmortem removal, sectioning, and processing for microscopic evaluation of the temporal bone, the reader is referred to other texts (7,56).
References
1. Hollinshead WH. The ear. In: Hollinshead WH, ed. Anatomy for Surgeons. 3rd ed. Philadelphia: Harper & Row; 1982:159 221.
2. Moore KL. The ear. In: Moore KL, ed. The Developing Human: Clinically Oriented Embryology. 4th ed. Philadelphia: WB Saunders; 1988:412 420.
3. Michaels L, Soucek S. Development of the stratified squamous epithelium of the human tympanic membrane and external canal: the origin of auditory epithelial migration. Am J Anat 1989;184:334 344.
4. Michaels L, Soucek S. Stratified squamous epithelium in relation to the tympanic membrane: its development and kinetics. Int J Pediatr Otorhinolaryngol 1991;22:135 49.
5. Michaels L. The ear. In: Sternberg SS, ed. Histology for Pathologists. 2nd ed. Philadelphia: Lippincott-Raven; 1997:337 366.
6. Michaels L, Soucek S. Auditory epithelial migration on the human tympanic membrane: II. The existence of two discrete migratory pathways and their embryologic correlates. Am J Anat 1990;189:189 200.
7. Nager GT. Anatomy of the membranous cochlea and vestibular labyrinth. In: Nager GT, ed. Pathology of the Ear and Temporal Bone. Baltimore: Williams & Wilkins; 1993:3 48.
8. Schuknecht HF. Anatomy. In: Schuknecht HF, ed. Pathology of the Ear. 2nd ed. Philadelphia: Lea & Febiger; 1993:31 74.
P.400
9. Maybaum JL, Goldman JL. Primary jugular bulb thrombosis. A study of twenty cases. Arch Otolaryngol 1933;17:70 84.
10. Kamerer DB. Electromyographic correlation of tensor tympani and tensor veli palatini muscles in man. Laryngoscope 1978;88:651 662.
11. Graves GO, Edwards LF. The Eustachian tube: a review of its descriptive, microscopic, topographic and clinical anatomy. Arch Otolaryngol 1944;39:359 397.
12. Moreano EH, Paparella MM, Zelterman D, Goycoolea MV. Prevalence of carotid canal dehiscence in the human middle ear: a report of 1000 temporal bones. Laryngoscope 1994;104(pt 1):612 618.
13. Michaels L, Wells M. Squamous cell carcinoma of the middle ear. Clin Otolaryngol Allied Sci 1980;5:235 248.
14. Nager GT. Pneumatization of the temporal bone. In: Nager GT, ed. Pathology of the Ear and Temporal Bone. Baltimore: Williams & Wilkins; 1993:53 62.
15. Tremble GE. Pneumatization of the temporal bone. Arch Otolaryngol 1934;19:172 182.
16. Davies DV. A note on the articulations of the auditory ossicles and related structures. J Laryngol Otol 1948;62:533 536.
17. Dayal VS, Farkashidy J, Kokshanian A. Embryology of the ear. Can J Otolaryngol 1973;2:136 142.
18. Lysakowski A, McCrea RA, Tomlinson RD. Anatomy of vestibular end organs and neural pathways. In: Cummings CW, Frederickson JM, Harker LA, Krause CJ, Richardson MA, Schuller DE, eds. Otolaryngology: Head and Neck Surgery. 3rd ed. St. Louis: Mosby; 1998:2561 2583.
19. Kobrak HG. Influence of the middle ear on labyrinthine pressure. Arch Otolaryngol 1935;21:547 .
20. Lindsey JR, Schuknecht HF, Neff WD, Kimura RS. Obliteration of the endolymphatic sac and the cochlear aqueduct. Ann Otol Rhinol Laryngol 1952;61:697 716.
21. Tonndorf J, Tabor JR. Closure of the cochlear windows: its effect upon air- and bone-conduction. Ann Otol Rhinol Laryngol 1962;71:5 29.
22. Belemer JJ. The vessels of the stria vascularis: with special reference to their functions. Arch Otolaryngol 1936;23:93 97.
23. Guild SR. The circulation of the endolymph. Am J Anat 1927;39:57 81.
24. Altmann F, Waltner JG. New investigations on the physiology of the labyrinthine fluids. Laryngoscope 1950;60:727 739.
25. Tarlov IM. Structure of the nerve root. II. Differentiation of sensory from motor roots: observations on identification of function in roots of mixed cranial nerves. Arch Neurol Psychiatr 1937;37:1338 1355.
26. Hollinshead WH. The cranium. In: Hollinshead WH, ed. Anatomy for Surgeons. 3rd ed. Philadelphia: Harper & Row; 1982:26 27.
27. Hyams VJ, Batsakis JG, Michaels L. Acoustic neuroma. In: Hartmann WH, Sobin LH, eds. Atlas of Tumor Pathology. Tumors of the Upper Respiratory Tract and Ear, Second Series, Fascicle 25. Washington, DC: Armed Forces Institute of Pathology; 1988:323 326.
28. Heffner DK. Low-grade adenocarcinoma of probable endolymphatic sac origin. A clinicopathologic study of 20 cases. Cancer 1989;64:2292 2302.
29. Wenig BM, Heffner DK. Endolymphatic sac tumors: fact or fiction? Adv Anat Pathol 1996;3:378 387.
30. Megerian CA, McKenna MJ, Nuss RC, et al. Endolymphatic sac tumors: histopathologic confirmation, clinical characterization, and implication in von Hippel-Lindau disease. Laryngoscope 1995;105(pt 1):801 808.
31. Sgambati MT, Stolle C, Choyke PL, et al. Mosaicism in von Hippel-Lindau disease: lessons from kindreds with germline mutations identified in offspring with mosaic parents. Am J Hum Genet 2000;66:84 91.
32. Smith CA, Lowry OH, Wu ML. The electrolytes of the labyrinthine fluids. Laryngoscope 1954;64:141 153.
33. Schuknecht HF. Developmental defects. In: Schuknecht HF, ed. Pathology of the Ear. 2nd ed. Philadelphia: Lea & Febiger; 1993:115 189.
34. Frank ST. Aural sign of coronary heart disease. N Engl J Med 1973;289:327 328.
35. Patel V, Champ C, Andrews PS, Gostelow BE, Gunasekara NP, Davidson AR. Diagonal earlobe creases and atheromatous disease: a postmortem study. Jr Coll Physicians Lond 1992;26:274 277.
36. Wenig BM. The ear. In: Mills SE, Carter D, Greenson JK, Oberman HA, Reuter VE, Stoler MH, eds. Sternberg's Diagnostic Surgical Pathology. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2004:1033 1072.
37. Bhaya MH, Schachern PA, Morizono T, Paparella MM. Pathogenesis of tympanosclerosis. Otolaryngol Head Neck Surg 1993;109(pt 1):413 420.
38. Gibb AG, Pang YT. Current considerations in the etiology and diagnosis of tympanosclerosis. Eur Arch Otorhinolaryngol 1994;251:439 451.
39. Nager GT, Vanderveen TS. Cholesterol granuloma involving the temporal bone. Ann Otol Rhinol Laryngol 1976;85(pt 1):204 209.
40. Nager GT. Cholesterol granulomas. In: Nager GT, ed. Pathology of the Ear and Temporal Bone. Baltimore: Williams & Wilkins; 1994:914 939.
41. Thedinger BA, Nadol JB Jr, Montgomery WW, Thedinger BS, Greenberg JJ. Radiographic diagnosis, surgical treatment, and long-term follow-up of cholesterol granulomas of the petrous apex. Laryngoscope 1989;99:896 907.
42. Nager GT. Cholesteatomas of the middle ear. In: Nager GT, ed. Pathology of the Ear and Temporal Bone. Baltimore: Williams & Wilkins; 1994:298 350.
43. Michaels L. The biology of cholesteatoma. Otolaryngol Clin North Am 1989;22:869 881.
44. Wells MD, Michaels L. Role of retraction pockets in cholesteatoma formation. Clin Otolaryngol Allied Sci 1983;8:39 45.
45. Schuknecht HF. Cholesteatoma. In: Schuknecht HF, ed. Pathology of the Ear. 2nd ed. Philadelphia: Lea & Febiger; 1993:204 206.
46. de Souza CE, Sperling NM, da Costa SS, Yoon TH, Abdel Hamid M, de Souza RA. Congenital cholesteatomas of the cerebellopontine angle. Am J Otol 1989;10:358 363.
47. Desloge RB, Carew JF, Finstad CL, et al. DNA analysis of human cholesteatomas. Am J Otol 1997;18:155 159.
48. Cody DT, Baker HL Jr. Otosclerosis: vestibular symptoms and sensorineural hearing loss. Ann Otol Rhinol Laryngol 1978;87(pt 1):778 796.
49. Morales-Garcia C. Cochleo-vestibular involvement in otosclerosis. Acta Otolaryngol 1972;73:484 492.
50. Schuknecht HF. Otosclerosis. In: Schuknecht HF, ed. Pathology of the Ear. 2nd ed. Philadelphia: Lea & Febiger; 1993:365 379.
51. Schuknecht HF. Disorders of aging. In: Schuknecht HF, ed. Pathology of the Ear. 2nd ed. Philadelphia: Lea & Febiger; 1993:415 446.
52. Soucek S, Michaels L, Frohlich A. Pathological changes in the organ of Corti in presbyacusis as revealed in microslicing and staining. Acta Otolaryngol Suppl 1987;436:93 102.
53. Soucek S, Michaels L, Frohlich A. Evidence for hair cell degeneration as the primary lesion in hearing loss of the elderly. J Otolaryngol 1986;15:175 183.
54. Paparella MM. The cause (multifactorial inheritance) and pathogenesis (endolymphatic malabsorption) of Meniere's disease and its symptoms (mechanical and chemical). Acta Otolaryngol 1985;99:445 451.
55. Klis SF, Buijs J, Smoorenburg GF. Quantification of the relation between electrophysiologic and morphologic changes in experimental endolymphatic hydrops. Ann Otol Rhinol Laryngol 1990;99(pt 1):566 570.
56. Schuknecht HF. Histologic techniques. In: Schuknecht HF, ed. Pathology of the Ear. 2nd ed. Philadelphia: Lea & Febiger; 1993:7 12.