12 - Positron Emission Tomography Imaging of Thoracic Neoplasms
Editors: Shields, Thomas W.; LoCicero, Joseph; Ponn, Ronald B.; Rusch, Valerie W.
Title: General Thoracic Surgery, 6th Edition
Copyright 2005 Lippincott Williams & Wilkins
> Table of Contents > Volume I - The Lung, Pleura, Diaphragm, and Chest Wall > Section IV - Diagnostic Procedures > Chapter 16 - Bronchoscopic Evaluation of the Lungs and Tracheobronchial Tree
Chapter 16
Bronchoscopic Evaluation of the Lungs and Tracheobronchial Tree
William H. Warren
Penfield L. Faber
Visual inspection of the airways was once an art performed using rigid instrumentation in the hands of a handful of highly skilled surgeons. This procedure was almost always performed in the operating theater under general anesthesia. In the early 1970s, fiberoptic bronchoscopy was introduced, revolutionizing examination of the airways. Over the ensuing years, flexible bronchoscopy has almost completely replaced the rigid bronchoscope as the diagnostic instrument of choice. Flexible bronchoscopy is now performed by pulmonologists, anesthesiologists, otolaryngologists, critical care specialists, and thoracic surgeons in a variety of clinical settings, including the hospital ward, intensive care unit, and operating room. The well-trained thoracic surgeon must have a clear understanding of all aspects of both flexible and rigid bronchoscopy, including indications, anesthetic techniques, instrument options, and the management of complications.
RIGID BRONCHOSCOPY
The indications for rigid bronchoscopy include examination and management of obstructing airway lesions, retrieval of foreign bodies, suctioning of inspissated secretions, and obtaining tissue biopsy specimens of endobronchial lesions deeper and more generous than those provided by the flexible bronchoscope. A rigid bronchoscope should always be available when tracheal lesions are examined. Bleeding from biopsy sites or edema from tissue trauma can obstruct a compromised lumen, which can be forcibly dilated with a rigid instrument, and ventilation can be restored through the instrument.
The rigid bronchoscopes most commonly used in adults have an internal diameter of 6, 7, or 8 mm and are 40 cm in length (Fig. 16-1). Traditional models provide only a tunnel view, but modern rigid bronchoscopes also can be used with telescopic lenses. Illumination is supplied from a halogen light source. A fiberoptic cable is attached to a light carrier that passes down the side wall of the bronchoscope, through the telescope, or both. By using the telescope, generous biopsy specimens can be taken without compromising visualization. Both models have a ventilating side port permitting assisted ventilation (Fig. 16-2).
Rigid bronchoscopy almost always is performed under general anesthesia with all of its attendant risks. Despite the clearer view provided with telescopes, sampling the upper lobe or distal airway cannot be easily accomplished through the rigid bronchoscope. The advantages and disadvantages of rigid bronchoscopy are listed in Table 16-1.
Rigid bronchoscopy is generally performed under general anesthesia, using the ventilation port on the side of the bronchoscope and capping the end of the scope with an eyepiece to convert the procedure to a nearly closed system. If no telescope is being used, the glass eyepiece must be removed for biopsy and aspiration (Fig. 16-3). If a telescope is used, plastic caps seal the scope. Some loss of tidal volume around the bronchoscope is inevitable, but this can be minimized by packing the hypopharynx with saline-soaked gauze. The anesthesiologist must monitor the adequacy of the ventilation continuously; increased minute ventilation and tidal volumes may be required.
To pass the bronchoscope, the patient is placed in a supine position with an assistant positioning the head so that the neck is slightly flexed and the chin extended. The endoscopist elevates the epiglottis with the tip of the bronchoscope and passes the lubricated instrument through the glottis, into the upper trachea. Telescopes provide a magnified field of vision. Examination of the trachea is performed as the bronchoscope passes through the glottis and advances down to the carina. The carina is assessed for sharpness and mobility during ventilation. Splaying or fixation suggests the presence of a subcarinal mass. Biopsy of the bronchial mucosa on the carina is performed if mucosal or submucosal changes are observed. Thirty- and 90-degree angle-viewing telescopes allow the endoscopist to view the upper lobe bronchi. Alternatively, a flexible bronchoscope
P.285
can be passed through an appropriately sized rigid instrument to view upper lobe orifices and all segmental bronchi (Fig. 16-4).
 |
Fig. 16-1. Standard 8-mm rigid bronchoscope with light carrier, rigid biopsy forceps, and glass eyepiece. |
The risks of rigid bronchoscopy include injury to the gums and tooth dislodgment, hypoventilation, airway bleeding, and direct injury to the larynx or rupture of the tracheobronchial tree. With experience, these complications can be minimized.
The rigid bronchoscope must be sterilized after each use. The scope and ancillary equipment must be mechanically cleaned of all blood and mucous debris before they are sent for sterilization. All glass and metal components should be autoclaved, and the plastic caps should be wrapped separately and gas sterilized.
 |
Fig. 16-2. A. Jackson ventilating rigid bronchoscope with eyepiece in place. The patient is ventilated through the side port. B. Dumon-Harrell rigid bronchoscope. The surgeon looks down the Hopkins rod and can simultaneously suction and obtain a biopsy through the various side ports. C. Dumon-Harrell rigid bronchoscope. The telescope, suction catheter, and semirigid biopsy forceps are simultaneously passed down the rigid bronchoscope. A neodymium:yttrium- aluminum-garnet (Nd:YAG) laser fiber can also be passed without compromising ventilation. |
Table 16-1. Rigid Bronchoscopy | |
|---|---|
|
FLEXIBLE BRONCHOSCOPY
Ideally, flexible bronchoscopy should be performed in a dedicated area of the hospital or outpatient facility. Because
P.286
intravenous sedation is routinely used, recovery areas are also needed. In the best clinical scenario, such suites would allow clinicians to perform procedures with fluoroscopic or CT guidance and with the ability to administer general anesthesia. The examination room must be large enough to store all equipment, supplies, and accessories. It is ideal to have a storage area adjacent to the endoscopic suite. Special holding racks and storage cases should be routinely used to minimize the risk for scope breakage.
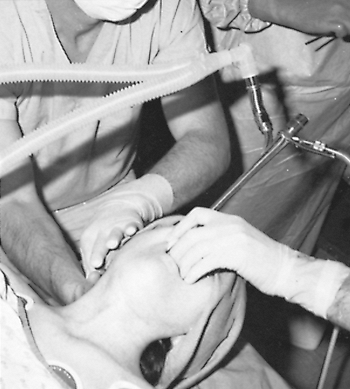 |
Fig. 16-3. Rigid ventilating bronchoscopy with biopsy performed using general anesthesia and open tube technique. |
A portable cart supplied with a light source, bronchoscope, and accessories is a convenient way to perform bedside bronchoscopy. It should be returned to the storage area for proper cleaning and restocking.
Flexible bronchoscopy has many advantages over rigid bronchoscopy, as listed in Table 16-2. Diagnostic material from peripheral as well as central lesions can be performed with minimal risk. The examination is usually performed under topical anesthesia and intravenous sedation, avoiding the risks of general anesthesia. Retained secretions can be aspirated at the bedside in the ward or in the intensive care unit. Patients receiving ventilator support can be examined without compromising the airway using a side-arm adaptor. The scope can be passed through narrowed and distorted airways or beyond an obstructing lesion. Flexible bronchoscopy is particularly valuable in assessing lobar and segmental airways to the upper lobes. Still or videophotography is easily performed to document findings (Fig. 16-5).
Table 16-2. Flexible Fiberoptic Bronchoscopy | |
|---|---|
|
 |
Fig. 16-4. Flexible bronchoscope passed through a rigid bronchoscope to examine upper lobe orifices and airways beyond the tip of the rigid bronchoscope. |
A new and persistent cough or a change in the cough pattern of a smoker warrants a bronchoscopic examination. Furthermore, a wheeze, particularly one that is unilateral and fails to clear with coughing, should be investigated. In so doing, lesions (especially those in the trachea and major airways) can be diagnosed, even in the absence of radiologic findings.
Sputum can be obtained in a sterile fashion to assess for opportunistic infections, especially in the immunocompromised patient. However, if the patient is suspected of having tuberculosis, induced sputum specimens (using inhaled saline) should be examined before contaminating the scope and the health care workers. Endoscopic lobar and segmental saline lavage have proven therapeutic benefit in postoperative patients unable to clear their secretions. Retained thick and tenacious mucous secretions are easily suctioned at the bedside with minimal patient discomfort.
Although the most common cause of hemoptysis is chronic bronchitis, carcinoma is common in patients with abnormal chest radiographs. Even if the chest radiograph is normal, according to Jackson (1985) and Poe (1988) and their associates, malignancy should be considered seriously if the patient is 40 years of age or older, has a significant smoking history, or has episodes of hemoptysis lasting for more than 1 week in duration. In addition to lung cancer, hemoptysis associated with a wheeze or atelectasis may be caused by a bronchial carcinoid or an inflammatory bronchial stricture.
An abnormal chest radiograph suggesting carcinoma warrants a careful evaluation of the entire tracheobronchial tree. Clinical judgment should always be used in deciding when
P.287
to use bronchoscopy, but the physician should err on the side of performing an endoscopic evaluation to rule out a neoplasm. An obstructing carcinoma may be the underlying cause of an unresolving pneumonia; an upper lobar infiltrate should be viewed with particular suspicion. Bronchoscopy also may be used to assess for metastatic tumors.
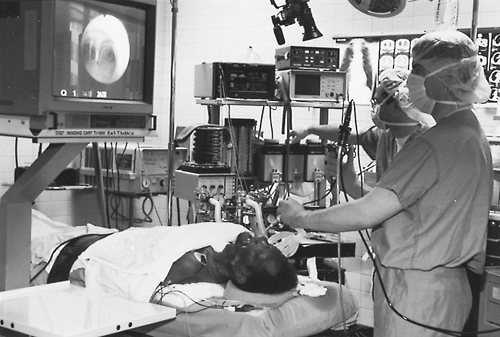 |
Fig. 16-5. Flexible bronchoscopy using a video-assisted technique. |
Bronchoscopy is indicated in the diagnosis and management of lung abscess. The passage of brushes and biopsy forceps into the abscess cavity can promote bronchial drainage. Furthermore, occasionally an obstructing neoplasm or foreign body is discovered.
The therapeutic value of bronchoscopy to remove aspirated gastric contents remains in question. However, a rapid and efficient bronchoscopy examination can support the diagnosis and may have therapeutic benefit, particularly if particulate matter is retrieved.
After inhalation of smoke or caustic fumes, bronchoscopy is a safe and expeditious way of assessing damage to the tracheobronchial mucosa. In these compromised patients, the airways frequently need repeated bronchoscopy to d'bride necrotic mucosa and inspissated secretions. The inflammatory response to this injury may progress for days, leading to delayed airway obstruction.
Patients with cystic fibrosis may also, on occasion, require aspiration of persistently thick and tenacious secretions. However, routine bronchoscopy, once a mainstay of their care, has been largely supplanted by the routine use of vigorous physiotherapy and mucolytic agents.
The role of bronchoscopic examination of a patient with a fever of unknown origin or a pulmonary infiltrate after lung transplantation is widely accepted, as summarized by Higenbottam and colleagues (1987). However, the need for routine surveillance biopsies in lung transplant recipients is still controversial. According to Trulock (1995), routine surveillance bronchoscopies with random biopsies should be performed 2 to 3 weeks, 6 to 8 weeks, 9 to 12 weeks, 6 months, and 1 year after transplantation and annually thereafter. The most frequent unsuspected findings have been acute rejection and silent cytomegalovirus pneumonia. Early detection and treatment of these entities are thought to decrease the risk for chronic rejection according to Steinhoff and associates (1991). Others, such as Stillwell and colleagues (1995), maintain that, in asymptomatic patients, early stages of acute rejection or viral infection pose little risk for leading to more serious conditions.
Another indication for bronchoscopic assessment of the tracheobronchial tree is the suspicion of airway trauma. Wheezing, hemoptysis, and the presence of subcutaneous or mediastinal emphysema are the classic findings, but injury of the airway is often overshadowed by concomitant injuries such as aortic rupture or myocardial contusion. Even when suspected, the endoscopic findings of a torn bronchus can be remarkably subtle. Early diagnosis and repair of these injuries are of the utmost importance.
Transbronchoscopic bronchial biopsies, cytologic brushings, transbronchial needle aspiration (TBNA), and bronchoalveolar lavage (BAL) are readily performed, and the endoscopist should be familiar with indications, techniques, and complications of each. Airway bleeding can be the most devastating complication. Even severely ill and debilitated patients can undergo bronchoscopy safely if it is performed in an appropriate facility by an experienced endoscopist.
Currently available standard flexible bronchoscopes range from 6.2 mm in outer diameter (3.2-mm working channel to aspirate inspissated secretions and blood clots) to 2.7 mm in outer diameter (1.2-mm working channel) for pediatric patients (Table 16-3). One of the most often used adult bronchoscopes has an external diameter of 5.9 mm and a 2. 2-mm working channel, allowing clear visualization and specimen sampling of the entire tracheobronchial tree down to the fourth- or fifth-order bronchi (Fig. 16-6). A forward field of
P.288
view is 120 degrees; the angle of deflection is 180 degrees upward and 130 degrees downward. A narrower instrument with an external diameter of 4.9 mm and an instrument channel of 2.2 mm easily passes through a bronchus narrowed by stricture or tumor, and its 180-degree upward deflection facilitates examination of the often difficult to reach apical subsegment.
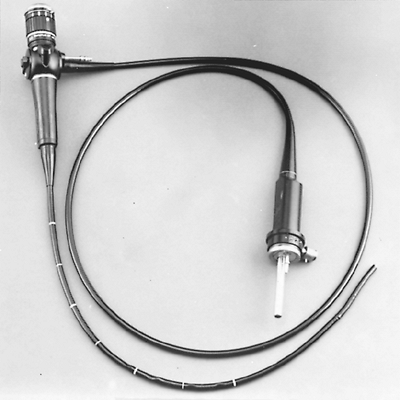 |
Fig. 16-6. Flexible fiberoptic bronchoscope (Olympus BF-10, Melville, NY). |
Table 16-3A. Standard Viewing Fiberoptic Bronchoscope Specifications | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Table 16-3B. Videofiberoptic Bronchoscopes | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Videobronchoscopes provide a significantly larger image with higher resolution than is seen in standard flexible bronchoscopes. However, these images can only be viewed with a videoprocessor and monitor. For these superior images, however, these bronchoscopes have a larger outer diameter (Table 16-3). These scopes are fully immersible, and most can be autoclaved. The active endoscopist should have a variety of flexible bronchoscopes available for diagnostic and therapeutic versatility (Fig. 16-7).
The Examination
Anesthetic Considerations
Intravenous access is routinely available, and supplemental oxygen is provided usually through nasal cannulae as recommended by Prakash and Stubbs (1991). Pulse oximetry, cuff blood pressure, and electrocardiographic monitoring are routinely performed. Sedation should be administered to provide patient comfort and cooperation. In several studies, however, 50% of the life-threatening complications from bronchoscopy stemmed from hypoxemia, hypercapnia, and respiratory depression secondary to oversedation. Therefore, the agents and their dosages must be individualized and may not be necessary in every case.
Preoperative opiates are sometimes given for their analgesic and antitussive properties. Meperidine has an elimination half-life of 3.2 hours, but clearance is decreased in patients with renal, hepatic, or both renal and hepatic failure. It, like all opiates, can cause respiratory depression and hypotension. Naloxone, a specific opioid antagonist, must be available whenever opioids are administered.
Intravenous benzodiazepines are often administered, providing anxiolysis and antegrade amnesia. Midazolam has become the preferred agent according to a bronchoscopy survey reported by Colt and colleagues (2000). It is water soluble, with rapid onset, short duration of action, and an elimination half-life of only 2 hours in normal subjects. Renal failure does not alter the distribution, elimination, or clearance, but liver disease is associated with prolonged sedation. The recommended dose is 0.07 mg/kg. Diazepam is not water soluble and can cause significant, if only transient, phlebitis. Its elimination half-life is 24 to 57 hours in normal patients. Both agents should be used with caution, especially in elderly patients and those with limited pulmonary reserve. Respiratory depression is a major side effect of both benzodiazepines; the degree of respiratory depression is similar with both drugs. Flumazenil is a specific benzodiazepine antagonist and should be available any time these agents are used. Because its elimination half-life is only 1 hour, repeat doses or a continuous infusion may be needed. Matot and Kramer (1999) reported that alfentanil-propofol is a reasonable alternative to meperidine-midazolam, particularly in patients with known or at risk for coronary artery disease.
 |
Fig. 16-7. Flexible fiberoptic bronchoscopes viewed on end. From left to right: 4.9-mm bronchoscope with 2.2-mm channel; 5.7-mm bronchoscope with 2.2-mm channel; 5.8-mm bronchoscope with 2.8-mm channel; and 6.3-mm bronchoscope with 3.2-mm channel. |
Table 16-4. Indications for Bronchoscopy | |
|---|---|
|
P.289
Some physicians continue to premedicate patients with an antisialagogue, such as atropine or glycopyrrolate, to reduce secretions and inhibit vasovagal responses, as documented by Williams and colleagues (1998). These medications also make topical anesthesia more effective. However, according to Hasanoglu and co-workers (2001), tachycardias and episopes of hypotension were more commonly observed when atropine was administered. According to the survey reported by Colt and colleagues (2000), only 62% of respondents routinely use atropine as a premedication. Far fewer used glycopyrrolate.
Topical anesthesia is preferred for fiberoptic bronchoscopy, but general anesthesia may be considered, particularly for prolonged examinations required to identify carcinoma in situ in a patient with normal chest radiography findings. It is occasionally requested by extremely anxious patients.
The most common agents for topical anesthesia are lidocaine (1% and 2%) and tetracaine (0.5%, 1.0%, and 2.0%). Complications from topical anesthesia usually result from the administration of excessive amounts, as documented by Credle (1974), Suratt (1976), and Pereira (1978) and their associates. If carefully measured amounts are given and the endoscopist is always aware of the total milligram dosage instilled, reactions are minimized.
Lidocaine is a safe agent with a recommended dose of 0.2 to 0.3 mL of 1% lidocaine per kilogram, but larger amounts have been given without serious side effects. It is important to remember that the extent of absorption and bioavailability depends on tissue vascularity and the technique of application as well as the total dose administered, according to Mainland and associates (2001). Lidocaine should always be titrated slowly, and patients must be monitored for mental status changes. The first sign of toxicity is usually central nervous system excitation or seizure before cardiovascular collapse. The duration of action is short.
Tetracaine is another effective topical anesthetic, but side effects frequently occur when a dose of 80 mg is exceeded. The duration of action is prolonged, and the first sign of toxicity may be sudden cardiovascular collapse.
Combination of benzocaine-tetracaine is a popular topical anesthetic agent with rapid onset of action useful in rapid intubation. It is often administered in a propellant spray with a tolerance dose delivered in only 2 seconds. Dosing is therefore difficult to regulate, and this agent should be avoided when performing flexible bronchoscopy. Complications of all topical anesthetic agents include methemoglobinemia, whereby patients become cyanotic owing to the oxidation of iron from its normal reduced state, leaving it unavailable for oxygen binding and transport. Elderly, infant, and anemic patients are particularly susceptible, as documented by Karim and coinvestigators (2001). Intravenous methylene blue (1 mg/kg) is the recommended initial definitive treatment. Rarely, exchange transfusion or hemodialysis is necessary.
Several satisfactory methods are available to administer topical anesthesia. Using the nasotracheal route, the nasopharynx is anesthetized initially using an atomized topical agent, and the flexible bronchoscope is then passed through the nares to a level just proximal to the false cords. With the larynx in clear view, additional topical anesthetic is administered directly onto the vocal cords and into the trachea. The bronchoscope is then passed through the glottis, and additional topical anesthesia is instilled down the tracheobronchial tree.
A second method of delivering topical anesthesia consists of initial spraying of the hypopharynx with 1% or 2% lidocaine using an atomizer. Five milliliters of 4% lidocaine are then injected transtracheally through the cricothyroid membrane using a short 21-gauge needle to minimize the risk for lacerating the posterior wall of the trachea. Slight bleeding often occurs with this technique, so it should be avoided when a patient is being examined for hemoptysis of unknown origin. Care is taken to confirm the position of the needle in the tracheal lumen by aspirating air before injecting. It is possible to inject directly into the false cords and precipitating laryngospasm. Anesthesia of the larynx is achieved as the patient coughs out the medication. Supplemental 2% lidocaine is then instilled into the tracheobronchial tree while advancing the flexible bronchoscope. Because topical anesthetic agents inhibit bacterial growth, care should be taken to minimize the amount aspirated into collection traps for microbiological studies.
P.290
Flexible bronchoscopy is accomplished easily using general anesthesia. In the adult, a swivel adaptor is attached to the endotracheal tube, and ventilation is maintained through the side arm of the adaptor. The bronchoscope is passed through a tight-fitting plastic diaphragm on the adaptor. As large an endotracheal tube as possible should be selected to minimize peak airway pressure and allow for adequate ventilation.
The flexible fiberoptic bronchoscope can be inserted either through the nose or the mouth into the hypopharynx. Although the nasal route may be more comportable for the patient and avoids possibility of the patient chewing the instrument, this option does not permit easy withdrawal and reinsertion for cleaning of the lens and clearing the channel of thick mucus. In addition, biopsy and brushing cytology specimens must be withdrawn through the entire length of the channel, leading potentially to a decreased yield of diagnostic material.
An alternative technique involves passing an uncuffed 8.0-mm endotracheal tube (overtube) through the orotracheal route (Fig. 16-8). The patient breathes around and through this overtube. Airway access is provided without repeated trauma to the larynx and subglottic region. The overtube allows rapid withdrawal and reinsertion of the bronchoscope. Brushing and biopsy specimens are retrieved, leaving the brush or biopsy forceps beyond the tip of the bronchoscope, avoiding loss of the specimen in the working channel. In addition, airway suctioning using a large-caliber catheter can be performed through the overtube in the event of significant airway bleeding. Airflow resistance, however, has been shown to increase significantly when a 5.8-mm bronchoscope is passed through an endotracheal tube smaller than an 8.5-mm outer diameter. The use of a smaller endotracheal tube could lead to hypercapnia and respiratory distress.
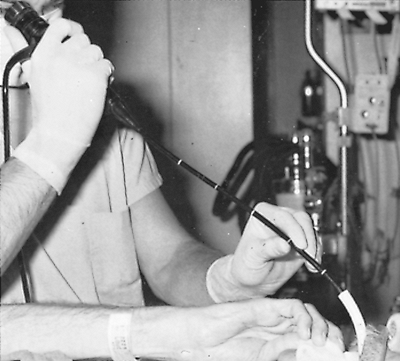 |
Fig. 16-8. Flexible fiberoptic bronchoscope is inserted through an oral endotracheal tube. |
Supplemental oxygen, oximetry, and electrocardiogram monitoring are important because arterial desaturation and increases in the heart rate and cardiac index are very frequently observed during the procedure, according to Markou and collegues (1999).
The Endoscopic Examination
The first phase of a diagnostic bronchoscopy is visualization of the larynx and the vocal cords. Unsuspected leukoplakia, carcinoma in situ, and invasive carcinoma may be found. Vocal cord mobility must be assessed. Recurrent laryngeal palsy secondary to carcinoma of the lung generally is considered a sign of inoperability.
All lobar and segmental bronchi must be examined carefully and systematically because a second lesion not visible on the chest radiograph is occasionally identified. The tracheobronchial tree remote from the area in question is assessed first. This way, not only will a complete examination be performed, but also, if samples from a different site are taken, they are less likely to be subject to cross-contamination.
After this has been performed, the bronchus leading to the known area of disease is examined. The character of the secretions and the bronchial mucosa can be clues to the nature of the underlying pathologic condition. Subtle mucosal abnormalities associated with carcinoma include mucosal thickening, irregular bronchial folds or corrugation, and increased submucosal vascularity. These findings may be associated with a loss of definition of the cartilaginous rings or circular folds, bronchial stenosis, or extrinsic bronchial distortion. The extent and the exact location of these endoscopic findings must be carefully observed when anticipating surgical intervention.
Bronchial Biopsy and Brushing
When mucosal changes of malignancy are visualized, transbronchial brushing and biopsy should be performed, which results in a combined diagnostic accuracy higher than 90%. Biopsies should be attempted, even when the lesion is suspected to be a bronchial carcinoid, because lung-sparing resections can be performed with minimal resection margins with this pathologic entity. Small fragments of tissue are obtained with 1.5-mm biopsy forceps, and multiple biopsies are often necessary to provide diagnostic material. The differential diagnosis of this entity includes granulation tissue, polypoid squamous carcinoma, and an endobronchial papilloma. Bleeding, if it occurs, can be controlled with topical 1/100,000 epinephrine or neodymium:yttrium-aluminum-garnet (Nd:YAG) laser photoablation.
Biopsy specimens of segmental lesions beyond the field of vision are often obtained under fluoroscopic or, more recently, computed tomography (CT) control. Biopsy through the flexible bronchoscope requires persistence and practice, especially when the lesion is in the apical segments of the
P.291
upper lobes. The accuracy of establishing the diagnosis of carcinoma increases with the number and types of specimens obtained. Many lesions have large necrotic areas, which do not provide diagnostic material if a biopsy is performed. After each biopsy, the forceps is placed in saline. Concentrated formalin solution is added at the conclusion of the procedure to give a final dilution of 10%.
Bronchial brushings are generally performed after the biopsy tissue has been obtained. Several models are available. A 7-mm brush is our recommendation for routine use because more cellular material is obtained (Fig. 16-9); 1.7-mm brushes have the advantage that they can be ensheathed and withdrawn through the working channel of the bronchoscope, but the yield of material is smaller, as documented by Elkus (1994) and Lundgren (1983) and their associates. In a recent comparative study reported by Sato and colleagues (2002), the efficacy of cell collection is determined by the diameter, not the length, of the bristles.
The bronchial brush may be inserted into narrowed segmental bronchi to provide cytologically diagnostic material. The brush is passed vigorously over the surface of the lesion and is then promptly stroked onto the surface of the glass slide, which is immersed immediately in 95% ethanol (Fig. 16-10). Improved results are achieved if four separate brush specimens are obtained and sent to the laboratory.
Endobronchial mucosal changes may not be detected in peripheral masses. Under these circumstances, fluoroscopic guidance is invaluable. Ideally, the lesion is seen to move when it is sampled. When the lesion is beyond the field of vision, the diagnostic yield by biopsy alone is 46%; however, the yield increases to 60% when combined with bronchial brushings.
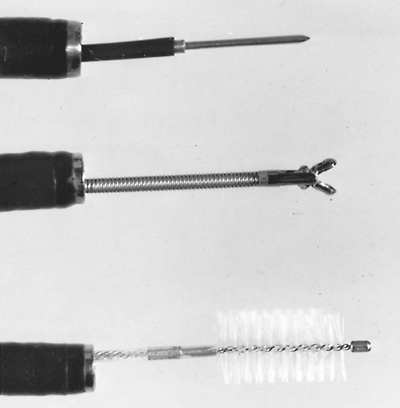 |
Fig. 16-9. Flexible fiberoptic bronchoscope with (from top to bottom) a 21-gauge transbronchial needle, a flexible cup biopsy forceps, and a 7-mm nylon brush. |
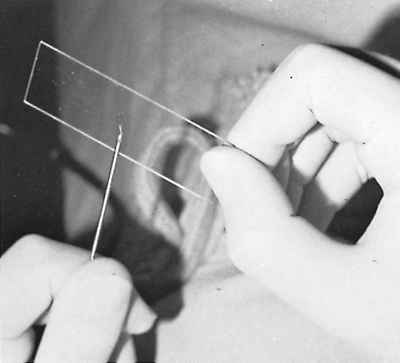 |
Fig. 16-10. Bronchial brush is rapidly smeared onto a glass slide, which is immediately immersed in 90% ethanol to fix the material for cytologic examination. |
Transbronchial Biopsy and Brushing
Transbronchial brushings and biopsies also may be used to assess pulmonary infiltrates. The diagnoses of sarcoidosis, tuberculosis, bronchioloalveolar carcinoma, and pulmonary alveolar proteinosis can all be established on the basis of bronchoscopic material. The flexible bronchoscope is initially wedged in the segmental bronchus, and the biopsy forceps is advanced to the periphery of the diseased region. The forceps is then drawn back 3 to 4 mm, and the jaws are opened and advanced slightly to obtain the sample of the lung. Placement of the biopsy forceps near, but not at, the lung surface minimizes the risk for pneumothorax. The bronchoscope is left wedged in the segmental bronchus in the event of bleeding to allow irrigation with 1/100,000 epinephrine. Five to seven transbronchial biopsy specimens have been found to provide the optimal diagnostic yield. These samples should all be taken from the same lung to avoid the complication of bilateral pneumothorax and to help to localize the bleeding site in the event of significant hemoptysis. Coagulation studies and a platelet count should be obtained preoperatively to identify patients at high risk for bleeding. Uremic patients are also known to be at high risk for significant hemoptysis, and precautions must be taken.
Transbronchial Needle Aspiration
Since the mid-1980s, the technique of TBNA, championed by Wang (1995), has found increasing favor in sampling subcarinal and paratracheal lymph nodes and for examining widened spurs endoscopically.
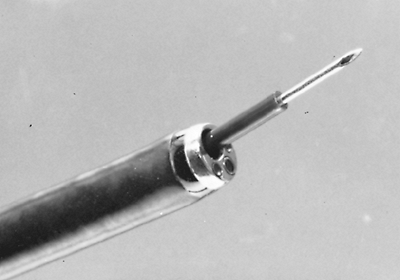 |
Fig. 16-11. Transbronchial needle for aspiration of mediastinal and hilar lymph nodes and masses. |
P.292
Cytologic and histologic specimens are obtained by passing an ensheathed 21-gauge needle down the working channel of the bronchoscope (Fig. 16-11). Failure to withdraw the needle into the protective outer sheath has led to serious and costly repairs of flexible bronchoscopes, as documented by Mehta and co-workers (1990). Once the sheath is advanced beyond the tip of the scope, the 1.5-cm needle is advanced beyond the sheath, through the wall of the trachea or bronchus, and into the mediastinal mass or suspicious lymph nodes (Fig. 16-12). Obviously, in chosing the location to be sampled, knowledge of the location of the major vessels adjacent to the airway is essential. The needle can be reinserted into the region in question several times, applying gentle suction each time the needle is advanced; suction must be avoided once the needle is completely withdrawn. Five milliliters of saline are then flushed through the needle to obtain an optimal cytologic sample, which is immediately centrifuged; the resulting pellet is resuspended in 1 mL of saline. The supernatant cell suspension is fixed in 95% ethanol and prepared with Papanicolaou's stain. Tissue fragments are fixed in Bouin's solution and submitted for a cell block.
Transbronchial needle aspiration should be performed before brushing or lavaging to avoid contaminating the airway, which could lead to an increased false-positive rate. There is increased diagnostic accuracy when TBNA is performed with visible evidence of submucosal or peribronchial tumor. Transbronchial needle aspiration has at least a 15% false-negative rate when assessing mediastinal nodes. This number has been much higher in the hands of most less experienced bronchoscopists.
Preoperative CT or magnetic resonance imaging is imperative to define precisely the lesion in question as well as to assess the proximity of major vessels. Recently, the technique of endobronchonial ultrasonography (EUS) has been used to assess and localize paratracheal and peribronchial pathology. Using this technique, a 2-mm diameter, 95-cm long catheter containing a single ultrasound transducer is located within an inflatable balloon. The catheter is passed down the working channel of the bronchoscope, and the balloon is inflated with sterile water. With the airway temporarily occluded, the motor of the ultrasound unit continuously rotates the transducer in the center of the balloon to obtain a real-time cross-sectional image of the airway. This technique produces a cross-sectional image of the peribronchial structures approximately 2 cm in depth (Fig. 16-13). The normal tracheobronchial wall is seen as a five-layered structure. This technique can reliably identify the depth of tumor invasion in the tracheobronchial wall and direct tumor invasion of contiguous mediastinal structures. However, using the criteria of size, echo density, homogeneity in structure, or demarcation of the nodal tissue from surrounding tissue, EUS cannot distinguish metastatic nodal disease from anthrasilicotic nodes. Therefore, although ultrasound can be used as an imaging technique, it has limited diagnostic value. After selection and evaluation of the biopsy site, the ultrasound catheter is withdrawn, and the needle apparatus is inserted. Unlike the gastrointestinal tract, at this time, it is not possible to perform TBNA simultaneously with real-time EUS imaging. Also, some of these balloons contain natural latex and must not be used in latex-sensitive patients. Experience with this technique in the United States is limited, but preliminary results are encouraging, as reported by Schuder (1991) and Goldberg (1994) and their associates and by Naidich and Harkin (1994). Silvestri and associates (1996) have used simultaneous
P.293
esophageal ultrasound to visualize the placement of the transbronchial needle into mediastinal nodes. Some prefer to perform TBNA under CT real-time imaging, but this is cumbersome. Its ultimate value to these various techniques will depend on cost-effectiveness in directing TBNA and thereby increasing the yield.
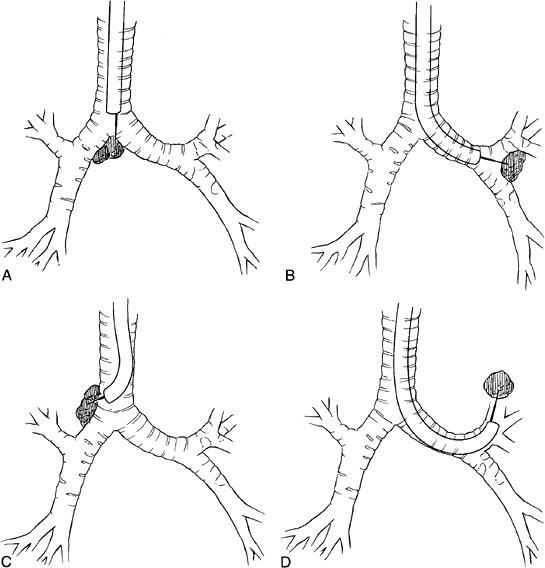 |
Fig. 16-12. Transbronchial needle aspirate of (A) subcarinal lymph nodes, (B) a mass at the bifurcation of the left main-stem bronchus, (C) a nodal mass at the right tracheobronchial angle, and (D) a central mass in the left upper lobe. |
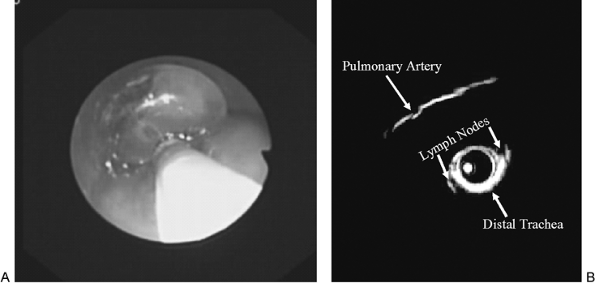 |
Fig. 16-13. (A) Photograph demonstrating bronchoscopic placement in the distal trachea of the ultrasound catheter through the biopsy channel. The balloon is inflated to make contact with the entire surface of the trachea. Since the entire tracheal lumen is occluded, ventilation is held until the exam can be completed. (B) Photograph taken from an ultrasound examination of the distal trachea demonstrating the right main pulmonary artery and both right and left paratracheal lymph nodes. |
Obviously, one of the main intraoperative complications of TBNA is airway bleeding. Mediastinal infection is a rare complication of TBNA of the peribronchial tissues.
Shure and Fedullo (1983) reported use of TBNA to establish the diagnosis of peripheral primary lung carcinoma presenting as a peripheral nodule or mass. Here, again, EUS has been used to localize the placement of the needle. In this case, a balloonless ultrasound probe is wedged into the segmental bronchus, placing the ultrasound transducer in direct contact with the bronchial wall.
BRONCHOALVEOLAR LAVAGE
BAL is a useful technique to recover material from the terminal bronchiole and alveolar sacs. BAL is performed by wedging the tip of the flexible bronchoscope into a subsegmental bronchus, irrigating and aspirating the segment with 20- to 50-mL aliquots of sterile saline. A total volume of 100 to 300 mL of saline is instilled, and 40% to 60% of this volume is recovered as a specimen, depending in part on the patience of the bronchoscopist. In patients who have loss of elastic recoil, recovery of fluid is less, according to Helmers and Hunninghake (1989), because the subsegmental bronchiolar walls collapse when suction is applied. In addition to microbiological studies, lavage specimens have been used to diagnose malignancies and to obtain inflammatory cells and pneumocytes for research studies as discussed by Kvale (1996).
BAL is a useful technique for obtaining microbiological specimens, especially in immunosuppressed patients. Fungal, bacterial, and viral culture specimens are easily acquired. In patients with acquired immunodeficiency syndrome, the diagnosis of Pneumocystis carinii pneumonia also can be made with a diagnostic yield exceeding 85%, according to Pisani and Wright (1992).
Cytologic specimens also can be obtained to establish the diagnosis in a variety of noninfectious, diffuse interstitial pulmonary diseases, including lipoid pneumonitis, histiocytosis, and berylliosis, all of which are uncommon entities, as documented by Daniele and associates (1985). BAL is of more limited value in the diagnosis of fibrosing alveolitis, sarcoidosis, and hypersensitivity pneumonitis, as noted by Stoller and colleagues (1987). Levy and associates (1988) reported that BAL has been used to establish the diagnosis of peripheral primary lung carcinoma. Rennard (1990) and Pirozynski (1992) also noted this fact, but BAL has not been widely advocated as a step in the diagnosis of cancer in North America, and it should not be considered a routine diagnostic procedure.
Potential complications of BAL include bronchospasm, hypoxia, fever, and transient decline in pulmonary function. Patients must therefore be observed carefully after the procedure. The safety of BAL in patients with symptomatic asthma or with a forced expiratory volume in 1 second of less than 60% of predicted value has not been established,
P.294
according to a National Heart, Lung, and Blood Institute workshop report (1985).
Complications
Despite the relatively low risk, the benefits of performing bronchoscopy must be weighed against the potential for complication in each patient. Credle and colleagues (1974) reported a complication rate of 0.08% and a mortality rate of 0.01% in more than 24,000 flexible bronchoscopies. Premedication and topical anesthesia were responsible for 11 of the 22 major complications. More dilute solutions of the topical anesthetic agent provide a wider margin of safety. Intravenous diazepam counteracts the systemic effects of excessive amounts of lidocaine and should be readily available. Elderly and debilitated patients should receive minimal premedication, and topical anesthesia must be administered in carefully measured amounts. Respiratory depression is a frequent complication of intravenous sedation in this patient population.
Careful evaluation and preparation of the patient, as well as adequate facilities to monitor the patient, are required to minimize complications. Pulse oximetry, continuous electrocardiography monitoring, and intermittent cuff blood pressure readings are all important parameters. Supplemental oxygen is supplied to minimize the risk for hypoxemia. Respiratory depression has been documented in the recovery period. According to Peacock and associates (1990), topical anesthesia may be responsible for prolonged periods of respiratory depression. Belen (1981) and Matsushima (1984) and their associates recommend that patients should not undergo pulmonary function testing for at least 8 hours after bronchoscopy. General anesthesia may be indicated if the patient has a history of intolerance to topical anesthetic agents or of difficult endoscopic examinations under local anesthesia.
Massive bleeding is a recognized complication of bronchoscopy. Bleeding disorders must be corrected by anticoagulant reversal or by the infusion of platelets either during or immediately before the procedure. However, acccording to Herth and colleagues (2002), aspirin does not increase the risk for bleeding after transbronchial biopsies. No absolute contraindications exist for flexible bronchoscopy. A brushing or biopsy should not be done unless the prothrombin time is higher than 40% of normal and the platelet count is greater than 50,000/mL. Patients with uremia or pulmonary hypertension also bleed easily, and brushings and biopsies in these patients should be avoided. Topical epinephrine solution of 1/100,000 can be instilled into the segmental bronchus before brushing to minimize bleeding or to control established bleeding. In the event of endobronchial hemorrhage, the scope should be wedged in the segmental bronchus to tamponade the lumen, thereby allowing blood in the distal airway to clot.
A pneumothorax can occur in up to 3% of patients undergoing a transbronchial lung biopsy. When lung biopsies are performed for diffuse lung disease, it is important to perform this procedure under fluoroscopic control to avoid perforation of the lung. Patients may complain of sharp chest pain if the parietal pleura is irritated.
Bronchospasm is a potential complication in patients with known asthma, but it also may occur in patients with severe chronic obstructive lung disease. Severe asthmatics should be premedicated with corticosteroids and bronchodilators. Laryngospasm is the direct consequence of inadequate topical anesthesia; it can be avoided if the topical agent is placed precisely onto the vocal cords and into the tracheobronchial tree.
Patients with hepatitis, human immunodeficiency virus, or suspected active tuberculosis can undergo bronchoscopy if special care is taken by all personnel in the handling of the specimens and if all instruments used are appropriately sterilized. Infections transmitted after properly cleaning instruments are uncommon. However, tuberbulosis and gram-negative organisms have been transmitted by inadequately cleaned flexible bronchoscopes.
Sepsis immediately following bronchoscopy is uncommon, but fever is seen occasionally. Well-designed prospective studies have documented an exceedingly low risk for bacteremia during and following bronchoscopy. Nevertheless, BAL in patients with pneumonia can cause a sepsislike clinical picture, according to Pugin and Suter (1992). Therefore, considering the risks, Dajani and associates (1990) reported that the American Heart Association recommended that patients with underlying valvular heart disease should receive prophylactic antibiotics before bronchoscopy to minimize the risk for bacterial endocarditis. This recommendation holds true for patients with joint replacements.
Bronchoscopy should not be performed in a patient with bilateral vocal cord paralysis. The passage of the bronchoscope through the glottis can lead to edema, causing life-threatening airway obstruction and necessitating emergent intubation or tracheostomy. Patients with a tracheal obstruction should be examined cautiously, and biopsy or dilation of the tracheal lesion should be avoided if the airway is severely compromised, unless one is prepared to proceed directly to rigid bronchoscopy or definitive tracheal surgery.
Cleaning of Flexible Bronchoscopes
The care and cleaning of fiberoptic flexible bronchoscopes has become an issue of some concern. A variety of gram-negative and fungal organisms and Mycobacterium tuberculosis have been recovered from inadequately cleaned bronchoscopes, and some, including tuberculosis, have been allegedly transmitted to patients through a contaminated bronchoscope, causing clinical infection as reported by Sammartino (1982), Gubler (1992), Nicolle (1992), Frazer (1992), and Agerton (1997) and their co-workers, as well as by Prakash (1993). In some cases, the bronchoscope did not undergo adequate mechanical cleaning
P.295
before being disinfected. Vigorous manual cleaning of bronchoscopes, including running brushes down the instrument channel is essential because Nicholson and associates (1995) have shown that without proper prior manual cleaning, even immersion for 60 minutes in 2% glutaraldehyde will not eliminate M. tuberculosis from the bronchoscope. Wheeler and colleagues (1989) identified a problem with inadequate cleaning of the suction valve. In other cases, the automated disinfecting machines were faulty according to Gubler (1992) and Frazer (1992) and their co-workers. Guidelines for cleaning fiberoptic bronchoscopes have been well established and reported by Martin and Reichelderfer (1994), but clinical practices vary widely among institutions.
SPECIAL CONSIDERATIONS
Foreign Body Retrieval
The suspicion of foreign body aspiration is an indication for bronchoscopy both to establish the diagnosis and attempt removal. When using flexible bronchoscopy, however, the endoscopist must be absolutely certain that a more complicated problem does not result from either losing the foreign body, impacting it distally in the tracheobronchial tree, or causing airway bleeding from manipulation. In this regard, the rigid bronchoscope remains the instrument of choice, allowing good exposure and airway control, especially in removing foreign bodies from the airways of infants and children, according to Weissberg and Schwartz (1987) as well as Pasaoglu and colleagues (1991). Grasping forceps, snares, baskets, and balloon-tipped catheters have all been used to extract foreign bodies using flexible bronchoscopy, as summarized by Lan and co-workers (1989) and Mehta and Rafanan (2001). Although foreign bodies in adult patients can be retrieved with various types of snares passed down the working channel of the fiberoptic bronchoscope, most foreign bodies can be removed easily and quickly with a rigid bronchoscope, especially in the pediatric population.
Inglis and Wagner (1992) reported the advantages of combining flexible and rigid bronchoscopy to improve the detection and retrieval rate of foreign bodies, especially when fragments are lodged in the distal airway or in the upper lobe bronchi (Fig. 16-14). Clearly, endoscopists engaged in the art of foreign body retrieval must be facile with both rigid and flexible bronchoscopy, according to Kelly and Marsh (1996).
Autofluorescence Bronchoscopy
In some centers, sputum cytology is used to screen patients at high-risk for malignancy. Others believe that surveillance bronchoscopy should be performed in these patients because it is much more successful in establishing the diagnosis and can localize the pathology. Occasionally, patients with suspicious or positive cytologic findings are identified in the presence of a normal chest radiograph. In such cases, the mouth, pharynx, larynx, and entire tracheobronchial tree must be examined carefully to identify the site of the early carcinoma.
 |
Fig. 16-14. Flexible bronchoscope with a foreign body basket retrieving a tooth, and a rigid bronchoscope with foreign body forceps grasping a peanut. |
Autofluorescence bronchoscopy has been a particularly valuable aid in identifying the site and extent of disease. Using a helium-cadmium laser for illumination, in vivo spectroscopy with an optical multichannel analyzer is performed during the bronchoscopic examination. Areas of severe dysplasia and carcinoma can be easily recognized by their decrease in autofluorescence intensity, whereas normal tissues autofluoresce predominantly in the short (green) wavelengths of the visible spectrum, as documented by Lam and associates (1994). Preferential horizontal diffusion of longer wavelength fluorescent light from adjacent normal submucosa causes the premalignant and malignant epithelium to appear red (Fig. 16-15; see Color Fig. 16-15).
Several fluorescence bronchoscopy systems are currently commercially available. The ultimate value of fluorescence bronchoscopy depends on the cost of the equipment, ease of use, and future of screening bronchoscopy for detection of early lung cancer, including in patients who have undergone a previous curative surgical procedure, according to Weigel and coinvestigators (2001).
Massive Hemoptysis
Massive hemoptysis (600 mL in 24 hours) should be assessed with a rigid bronchoscope urgently, but under optimal conditions. Airway control with rapid and repeated
P.296
suctioning is readily accomplished, and a major bronchus can be packed with an epinephrine-soaked pledget or inflation with a Fogarty balloon. Although massive hemoptysis can be assessed initially using a flexible bronchoscope through a cuffed endotracheal tube, clots are not easily removed, and they often obscure visualization. The site of massive hemoptysis must be localized, as described by Saw and co-workers (1976) to prepare for possible surgical excision, Nd:YAG laser photoablation, endobronchial tamponade, or bronchial artery embolization.
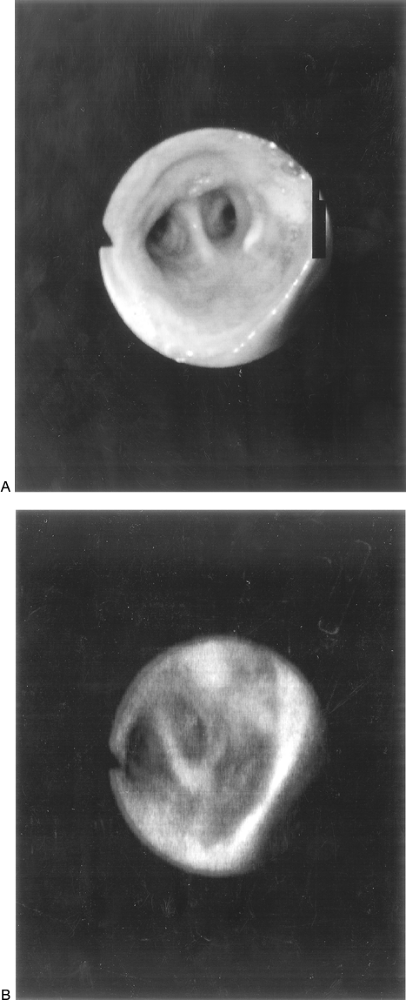 |
Fig. 16-15. A. Photograph of a normal-appearing bronchial mucosa using a white light source. B. Photograph of the same bronchial mucosa using a helium-cadmium laser light source to induce autofluorescence. (See Color Fig. 16-15.) |
When using YAG laser photoablation, rigid bronchoscopes permit photoablation and rapid d bridement of an obstructing or a bleeding lesion while simultaneously maintaining control of the airway and providing a suction channel for evacuation of clots and secretions. The rigid bronchoscope is necessary for the placement of silicone endobronchial stents. However, self-expanding metal bronchial stents can be deployed through a large endotracheal tube.
INTERVENTIONAL BRONCHOSCOPY
The past two decades have seen the advent of many endobronchial interventional procedures. These include the development endobronchial electrocaudery, Nd:YAG laser photoablation, cryoablation, photodynamic therapy (using chemical tissue sensitizers, such as porphyrin compounds), the placement of endobronchial stents, and the placement of radioactive brachytherapy sources in the form of seeds or catheters. These topics are discussed elsewhere (see Chapter 108).
PEDIATRIC BRONCHOSCOPY
Bronchoscopic examination of infants and small children requires expertise and familiarity with all available instrumentation, as emphasized by Wood (1996). Examination of the infant airway using smaller Storz rigid instruments with viewing telescopes is usually performed using general anesthesia (Fig. 16-16). A 3.0- or 3.5-mm sheath permits passage of the 2.7-mm optical telescope that provides a good view of the infant's bronchi. A small suction catheter can be passed down the barrel. Secretions are removed readily for microbiological studies. Small biopsy and foreign body forceps can be manipulated through this small channel with the viewing telescope in place. Muntz and associates (1992) reported their experience performing transbronchial lung biopsies in the pediatric population through a rigid bronchoscope.
Flexible fiberoptic bronchoscopy has become a practical tool in pediatrics with the development of a bronchoscope with an external diameter of 3.5 mm and a channel of 1.2 mm. It has found favor in clearing secretions as well as aiding in localizing and retrieving foreign bodies. Examination of infants may be performed using sedation and topical anesthesia, provided the procedure is brief. Airway stenosis and obstruction are contraindications to flexible instrumentation.
Complications related to pediatric bronchoscopy can be life threatening. A small bronchus may be perforated with resultant pneumothorax or pneumomediastinum. Laryngospasm, subglottic edema, and bronchospasm may all result from manipulation and may compromise the airway.
P.297
Stridorous breathing after the procedure may be an indication to humidify supplemental oxygen and the administer systemic corticosteroids.
 |
Fig. 16-16. Infant rigid bronchoscope (Storz), 3.5-mm diameter, with fiberoptic lighting. A. Components (from top to bottom): forward-viewing endoscopic telescope (Hopkins); bronchoscope with detachable window plus side channels for connection to anesthesia equipment, suctioning, and insertion of proximal light; proximal prismatic light carrier; and fiberoptic lighting cable. B. The bronchoscope after assembly. |
REFERENCES
Agerton T, et al: Transmission of a highly drug-resistant strain (strain W1) of mycobacterium tuberculosis. Community outbreak and nosocomial transmission via a contaminated bronchoscope. JAMA 278:1073, 1997.
Belen J, et al: Modification of the effect of fiberoptic bronchoscopy on pulmonary mechanics. Chest 79:516, 1981.
Colt HG, Prakash UBS, Offord KP: Bronchoscopy in North America. Survey by the American Association for Bronchology, 1999. J Bronchol 7: 8, 2000
Credle WF Jr, Smiddy JF, Elliot RC: Complications of fiberoptic bronchoscopy. Am Rev Respir Dis 109:67, 1974.
Dajani AS, et al: Prevention of bacterial endocarditis: recommendations by the American Heart Association. JAMA 264:2919, 1990.
Daniele RP, et al: Bronchoalveolar lavage: role in the pathogenesis, diagnosis and management of interstitial lung disease. Ann Intern Med 102: 93, 1985.
Elkus RL, et al: A comparison of withdrawn and nonwithdrawn brushes in the diagnosis of lung cancer. J Bronchol 1:269, 1994.
Frazer VJ, et al: Contamination of flexible fiberoptic bronchoscopes with Mycobacterium chelonae linked to an automated bronchoscope disinfecting machine. Am Rev Respir Dis. 145:853, 1992.
Goldberg BB, et al: US-assisted bronchoscopy with use of miniature transducer-containing catheters. Radiology 190:233, 1994.
Gubler JGH, Salfinger M, von Graevenitz A: Pseudoepidemic of nontuberculous mycobacteria due to a contaminated bronchoscope cleaning machine: report of an outbreak and review of the literature. Chest 101: 1245, 1992.
Hasanoglu HC, et al: Flexible bronchoscopy. Is atropine necessary for premedication? J Bronchol 8:5, 2001.
Herth FJF, Becker HD, Ernst A: Aspirin does not increase bleeding complications after transbronchial biopsy. Chest 122:1461, 2002.
Helmers RA, Hunninghake GW: Bronchoalveolar lavage. In Wang KP, ed. Biopsy Techniques in Pulmonary Disorders. New York: Raven Press, 1989.
Higenbottam T, et al: The diagnosis of lung rejection and opportunistic infection by transbronchial lung biopsy. Transplant Proc 19:3777, 1987.
Inglis AF Jr, Wagner DV: Lower complication rates associated with bronchial foreign bodies over the last 20 years. Ann Otol Rhinol Laryngol 101:61, 1992.
Jackson CV, Savage PJ, Quinn DL: Role of fiberoptic bronchoscopy in patients with hemoptysis and a normal chest roentgenogram. Chest 87: 142, 1985.
Karim A, et al: Methemoglobinemia complicating topical lidocaine used during endoscopic procedure. Am J Med 111:150, 2001.
Kelly SM, Marsh BR: Airway foreign bodies. Chest Surg Clin N Am 6: 253, 1996.
Kvale PA: Bronchoscopic biopsies and bronchoalveolar lavage. Chest Surg Clin N Am 6:205, 1996.
Lam A, et al: Early localization of bronchoscopic carcinoma. Diagn Ther Endosc 1:75, 1994.
Lan RS, et al: Use of fiberoptic bronchoscopy to retrieve bronchial foreign bodies in adults. Am Rev Respir Dis 140:1734, 1989.
Levy H, Horak DA, Lewis MI: The value of bronchial washings and bronchoalveolar lavage in the diagnosis of lymphangitic carcinomatosis. Chest 94:1028, 1988.
Lundgren R, Bergman F, Angstrom T: Comparison of transbronchial fine needle aspiration biopsy, aspiration of bronchial secretion, bronchial washing, brush biopsy and forceps biopsy in the diagnosis of lung cancer. Eur J Respir Dis 64:378, 1983.
Mainland P, et al: Absorption of lidocaine during aspiration anesthesia of the airway. J Clin Anesth 13:440, 2001.
Markou NK, et al: Fluctuations in gas exchange and cardiovascular parameters during flexible bronchoscopy. J Bronchol 6:241, 1999.
Martin MA, Reichelderfer M: APIC guidelines for infection prevention and control in flexible endoscopy. Association for Professionals in Infection Control and Epidemiology, Inc. 1991, 1992, and 1993. APIC Guidelines Committee. Am J Infect Control 22:19, 1994.
Matot I, Kramer MR: Sedation in outpatient flexible bronchoscopy. Alfentanil-propofol versus meperidine-midazolam. J Bronchol 6:74, 1999.
Matsushima Y, et al: Alterations in pulmonary mechanics and gas exchange during routine fiberoptic bronchoscopy. Chest 86:184, 1984.
Mehta AC, Rafanan AL: Extraction of airway foreign body in adults. J Bronchol 8:123, 2001.
Mehta AC, et al: The high price of bronchoscopy. Maintenance and repair of the flexible fiberoptic bronchoscope. Chest 98:448, 1990.
Muntz H, Wallace M, Lusk RP: Pediatric transbronchial lung biopsy. Ann Otol Rhinol Laryngol 101:135, 1992.
Naidich D, Harkin TJ: US-assisted bronchoscopy: is seeing believing? Radiology 190:18, 1994.
P.298
National Heart, Lung, and Blood Institute Workshop Summaries. Summary and recommendations of a workshop on the investigative use of fiberoptic bronchoscopy and bronchoalveolar lavage in asthmatics. Am Rev Respir Dis 132:180, 1985.
Nicolle LE, et al: Pseudo-outbreak of blastomycosis associated with contaminated bronchoscopes. Infect Control Hosp Epidemiol 13:324, 1992.
Nicholson G, et al: The efficacy of the disinfection of bronchoscopes contaminated in vitro with Mycobacterium tuberculosis and Mycobacterium avium-intracellulare in sputum: a comparison of Sactimed-I- Sinald and glutaradlehyde. J Hosp Infect 29:257, 1995.
Pasaoglu I, et al: Bronchoscopic removal of foreign bodies in children: retrospective analysis of 822 cases. Thorac Cardiovasc Surg 39:95, 1991.
Peacock AJ, Benson-Mitchell R, Godfrey R: Effect of fibreoptic bronchoscopy on pulmonary function. Thorax 45:38, 1990.
Pereira W, Kovnat DM, Snider GL: A prospective cooperative study of complications following flexible fiberoptic bronchology. Chest 73:813, 1978.
Pirozynski M: Bronchoalveolar lavage in the diagnosis of peripheral, primary lung cancer. Chest 102:372, 1992.
Pisani RJ, Wright AJ: Clinical utility of bronchoalveolar lavage in immunocompromised hosts. Mayo Clin Proc 76:221, 1992.
Poe RH, et al: Utility of fiberoptic bronchoscopy in patients with hemoptysis and a nonlocalizing chest roentgenogram. Chest 93:70, 1988.
Prakash UBS: Does the bronchoscope propagate infection? Chest 104: 552, 1993.
Prakash UBS, Stubbs SE: The bronchoscopy survey. Some reflections. Chest 100:1660, 1991.
Pugin J, Suter PM: Diagnostic bronchoalveolar lavage in patients with pneumonia produces sepsis-like systemic effects. Intensive Care Med 18:6, 1992.
Rennard SI: Future directions for bronchoalveolar lavage. Lung 168:1050, 1990.
Sammartino MT, Israel RH, Magnussen CR: Pseudomonas aeruginosa contamination of fibreoptic bronchoscopes. J Hosp Infect 3:65, 1982.
Sato M, et al: The efficacy of cell collection devices with a cytologic brush depends on the diameter of the bristles, not the on the diameter of the brush. J Bronchol 9:177, 2002.
Saw EC, et al: Flexible fiberoptic bronchoscopy and endobronchial tamponade in the management of massive hemoptysis. Chest 70:589, 1976.
Schuder G, et al: Endoscopic ultrasonography of the mediastinum in the diagnosis of bronchial carcinoma. Thorac Cardiovasc Surg 39:299, 1991.
Shure D, Fedullo PF: Transbronchial needle aspiration of peripheral masses. Am Rev Respir Dis 128:1090, 1983.
Silvestri GA, et al: Endoscopic ultrasound with fine-needle aspiration in the diagnosis and staging of lung cancer. Ann Thorac Surg 61:1441, 1996.
Steinhoff G, et al: Early diagnosis and effective treatment of pulmonary CMV infection after lung transplantation. J Heart Lung Transplant 10:9, 1991.
Stillwell PC, Mehta AC, Anoliga AC: Is surveillance bronchoscopy indicated in lung transplant patients? Consurveillance. J Bronchol 2:74, 1995.
Stoller JK, Rankin JA, Reynolds HY: The impact of bronchoalveolar lavage analysis on clinicians' diagnostic reasoning about interstitial lung disease. Chest 92:839, 1987.
Suratt PM, Smiddy JF, Gruber B: Deaths and complications associated with fiberoptic bronchoscopy. Chest 69:747, 1976.
Trulock EP: Is surveillance bronchoscopy indicated in lung transplant recipients? Pro surveillance. J Bronchol 2:69, 1995.
Wang KP: Transbronchial needle aspiration and percutaneous needle aspiration for staging and diagnosis of lung cancer. Clin Chest Med 16:535, 1995.
Weigel TL, et al: Postoperative fluorescence bronchoscopic surveillance in non-small cell lung cancer patients. Ann Thorac Surg 71:967, 2001.
Weissberg D, Schwartz I: Foreign bodies in the tracheobronchial tree. Chest 91:730, 1987.
Wheeler PW, Lancaster D, Kaiser AB: Bronchopulmonary cross-colonization and infection related to mycobacterial contamination of suction valves of bronchoscopes. J Infect Dis 159:954, 1989.
Williams T, Brooks T, Ward C: The role of atropine premedication in fiberoptic bronchoscopy using intravenous midazolam sedation. Chest 113:1394, 1998.
Wood RE: Pediatric bronchoscopy. Chest Surg Clin N Am 6:237, 1996.
Reading References
Albertini RE, Harrell JH, Moser KM: Management of arterial hypoxemia induced by fiberoptic bronchoscopy. Chest 67:134,1975.
Brodsky JB: Anesthestic considerations for bronchoscopic procedures in patients with central-airway obstruction. J Bronchol 8:36, 2001.
Dasgupta AD, Mehta AC: Transbronchial needle aspiration: an underused diagnostic technique. Clin Chest Med 20:39, 1999.
Fulkerson WJ: Fiberoptic bronchoscopy. N Engl J Med 311:511, 1984.
Harrow EM, Wang KP: The staging of lung cancer by bronchoscopic transbronchial needle aspiration. Chest Surg Clin North Am 6:223, 1996.
Haussinger K, et al: Autofluorescence bronchoscopy: the D-light system. Prog Respir Res 30:243, 2000.
Kvale PA: Flexible bronchoscopy with brush and forceps biopsy. In: Wang KP, ed. Biopsy Techniques in Pulmonary Disorders. New York: Raven Press, 1989.
Lam S, et al: Detection of dysplasia and carcinoma in situ with a lung imaging fluorescence endoscopic device. J Thorac Cardiovasc Surg 106:1035, 1993.
Lam S, et al: Localization of bronchial intraepithelial neoplastic lesions by fluorescence bronchoscopy. Chest 3:696, 1998.
Leonhard M: New incoherent autofluorescence/fluorescence system for early detection of lung cancer. Diagn Ther Endosc 5:71, 1999.
Lindholm C-E, et al: Cardiorespiratory effects of flexible fiberoptic bronchoscopy in critically ill patients. Chest 74:362, 1978.
Miller MB, Kvale PA: Diagnostic bronchoscopy. Chest Surg Clin North Am 2:599, 1992.
Milman N, et al: Pulse oximetry during fibreoptic bronchoscopy in local anaesthesia: frequency of hypoxaemia and effect of oxygen supplimentation. Respiration 61:342, 1994.
Prakash UBS, Offord KP, Stubbs SE: Bronchoscopy in North America: the ACCP survey. Chest 100:1668, 1991.
Radke JR, et al: Diagnostic accuracy in peripheral lung lesions. Factors predicting success with flexible fiberoptic bronchoscopy. Chest 76:176, 1979.
Rubins JB, Bofenkamp C: Routine culture for tuberculosis during bronchoscopy for cancer is not warranted. J Bronchol 6:236, 1999.
Susanto I: Managing a patient with hemoptysis. J. Bronchol 9:40, 2002.
Takemoto Y, et al: Ultrasound-guided flexible bronchoscopy for the diagnosis of tumor invasion to the bronchial wall and mediastinum. J Bronchol 7:127, 2000.
Venmans BJW, et al: Clinically relevant information obtained by performing autofluorescence bronchoscopy. J Bronchol 7:118, 2000.
Weinstein HJ, Bone RC, Ruth WE: Contamination of fiberoptic bronchoscope with Proteus species. Am Rev Resp Dis 116: 541, 1997.
EAN: 2147483647
Pages: 203
- Enterprise Application Integration: New Solutions for a Solved Problem or a Challenging Research Field?
- Intrinsic and Contextual Data Quality: The Effect of Media and Personal Involvement
- A Hybrid Clustering Technique to Improve Patient Data Quality
- Relevance and Micro-Relevance for the Professional as Determinants of IT-Diffusion and IT-Use in Healthcare
- Development of Interactive Web Sites to Enhance Police/Community Relations