120 - Secondary Tumors of the Lung
Editors: Shields, Thomas W.; LoCicero, Joseph; Ponn, Ronald B.; Rusch, Valerie W.
Title: General Thoracic Surgery, 6th Edition
Copyright 2005 Lippincott Williams & Wilkins
> Table of Contents > Volume II > The Esophagus > Section XXII - Trauma to the Esophagus > Chapter 138 - Esophageal Trauma
function show_scrollbar() {}
Chapter 138
Esophageal Trauma
Thomas W. Shields
The esophagus is subject to multiple types of trauma that lead to perforation of the organ with consequential egress of air into the surrounding tissue spaces as well as soilage by oral pharyngeal secretions and their contained pathogenic aerobic and anaerobic microorganisms, and often concomitant soilage by refluxing gastric juice. The perforation may be free or contained, or may fistulize into an adjacent structure. At times, the injury may be confined within the wall of the esophagus, with resultant destruction of the mucosal layer and subsequent stricture of the esophagus, although such injury may be severe enough to result in total necrosis of the entire esophageal wall. The injury may occur in the cervical, thoracic, or abdominal portions of the esophagus. This discussion is organized by the anatomic site because the signs and symptoms, the radiographic features, the diagnosis, and the treatment are essentially governed by the site of the injury. The etiology, of course, is most important and is discussed as is appropriate for a given location.
INJURIES OF THE CERVICAL ESOPHAGUS
Most injuries to the cervical portion of the esophagus are the result of iatrogenic misadventures, for example, endoscopic procedures and attempted endotracheal tube placement, as noted by O'Neill and co-workers (1984), and dilation (bougienage) of proximal strictures. Perforation may occur from an impacted swallowed foreign body (most commonly pork or chicken bones, although swallowed fish bones also may lodge in the cervical portion of the esophagus, but these are more likely to remain in the hypopharynx). External trauma due to gunshot or stab wounds is seen less commonly, but one must be suspicious of such a possibility in penetrating wounds of the neck or even with blunt trauma. Injury to the anterior wall of the cervical esophagus may occur during an emergent tracheostomy or the placement of stents in that area. Finally, of course, chemical burns (e.g., lye ingestion) may occur, especially in children (Table 138-1).
Symptoms and Signs of Cervical Esophageal Injury
Partial disruption of the cervical esophagus with mucosal tear may occur more frequently with endoluminal injuries than recognized, but it may be serious if an intramural abscess develops. A perforation of all layers of the esophagus, however, is more common. Usually, the endoscopist may suspect injury because of unusual difficulty or the occurrence of bleeding during the procedure. With a complete perforation of the wall, air and secretions are forced out of the esophagus on swallowing. Such contamination and air may extend into the other adjacent fascial compartments of the neck and may descend into the mediastinum.
Clinically, within several hours of the injury, the patient complains of pain and stiffness in the neck, dysphagia, and, at times, respiratory distress. On examination, the patient is febrile and has varying degrees of dysphonia (usually a nasal twang to the voice) and cervical tenderness. Crepitation caused by subcutaneous air may be found on palpation of the neck.
Radiographic examination of the neck may reveal air in the fascial planes, widening of the retroesophageal space, and obliteration of the normal cervical vertebral curvature. Massive pneumomediastinum may occur (Fig. 138-1). In some neglected patients, a retroesophageal abscess with an air-fluid level may be present.
In most patients with instrumental perforation of the cervical esophagus, as a rule, neither radiographic examination of the esophagus with the use of a contrast medium nor esophagoscopy is indicated. In fact, such examinations may be misleading because they frequently fail to identify the site of perforation; therefore, they are not recommended.
Management of Perforating Esophageal Injuries
The management of these injuries is adequate drainage of the visceral compartment of the neck and prevention of continued contamination of the area. These goals are best achieved by an anterior cervical mediastinotomy, repair of
P.2102
the laceration, and drainage of the area. The technique of cervical mediastinotomy is essentially the exposure of the cervical esophagus as described in Chapter 129. Repair of the perforation is best performed with interrupted, nonabsorbable, fine sutures. At times, owing to either the degree of inflammation or the duration of time between the injury and its recognition (about 12 hours or more), or both, no attempt at direct repair of the laceration is indicated. In such a situation, one must rely on adequate drainage, cessation of swallowing, and aspiration of oropharyngeal secretions. Appropriate antibiotics to control the microorganisms in the oropharyngeal secretions are indicated in all patients.
Table 138-1. Injuries of the Cervical Esophagus | |
|---|---|
|
Occasionally, when the injury is recognized immediately and is thought to be minor, or when there is a question about the presence of a perforation through all layers of the esophagus, expectant treatment may be used. This therapy consists of cessation of swallowing, aspiration of oropharyngeal secretions, rest, and antibiotics. With such a regimen, a successful response may be obtained; however, acute cervical mediastinal suppuration may occur; thus, the patient must be observed carefully. Berry and Ochsner (1973) reported that two thirds of their patients managed expectantly required subsequent drainage.
The morbidity of instrumental injury to the cervical esophagus is minimal when recognized early and when treated appropriately. Death is rare and, when it does occur, is related to spread of the infection into the mediastinum and failure to recognize the underlying problem.
Extraluminal Injuries of the Cervical Esophagus
Sheely and colleagues (1975) noted that the cervical esophagus is injured in only about 0.5% of penetrating neck injuries.
Although the cervical esophagus is in a protected location, Yap and associates (1984) pointed out that it is more vulnerable to external penetrating injuries than the thoracic esophagus. Gunshot wounds are the most common cause of injury, but injury from stab wounds also occurs (Fig. 138-2).
The diagnosis of penetrating injury is greatly facilitated by awareness of its possible existence. These wounds frequently are associated with an injury to the trachea, the large vessels of the neck, the spinal cord, or a combination of these. Occasionally, an isolated injury of the esophagus may occur, particularly with stab wounds by a thin-bladed instrument.
Clinically, dysphagia, spitting of blood, or both may be present and should increase suspicion of possible esophageal injury. Subcutaneous emphysema from an isolated injury of the esophagus occurs only in about one third of such instances. When crepitus is palpable, a combined injury of the trachea and esophagus must be considered. Retropharyngeal air, subcutaneous air, or both may be seen in radiographs of the neck. Occasionally, radiographs of the chest reveal a pneumomediastinum, but this finding almost always indicates concomitant airway injury.
An esophagogram, although not recommended, may be obtained to confirm the suspicion of esophageal injury, but this examination, of necessity obtained under emergency conditions, may fail to disclose the injury. The same is true for esophagoscopy, and this latter examination usually is not indicated.
Penetrating injuries of the neck should be exposed through an incision along the anterior border of the sternocleidomastoid muscle. The esophagus can be mobilized and repaired from either side of the neck; therefore, the side selected depends primarily on the site of the penetration and the possibility of associated injury to one or more of the vascular structures. Occasionally, both sides of the neck must be explored through separate incisions. The injury of the esophagus is repaired in two layers using interrupted nonabsorbable sutures. Adequate drainage of the neck is essential. If the upper mediastinum is contaminated, this area should be drained as well. Drains should be left in place until oral feedings have been reinstituted (5 to 7 days) and there is no evidence of an esophagocutaneous fistula. Aspiration of the oropharyngeal secretions, as well as appropriate antibiotics, should be used in all these injuries. If the trachea and esophagus were injured simultaneously, Feliciano and associates (1985) recommend rotation of a flap of viable muscle between the repair of the esophagus and that of the trachea.
The morbidity and mortality rates related to esophageal injury should be minimal when the problem is recognized promptly. Major complications and death are more often related to associated injuries, especially those of the cervical spinal cord.
The management of impacted foreign bodies, as well as chemical injuries, is discussed with those injuries of the thoracic esophagus.
INJURIES TO THE THORACIC ESOPHAGUS
Intraluminal Penetrating Trauma
As with the cervical esophagus, penetrating trauma may be classified as arising either internally (intraluminal) or externally (extraluminal). Intraluminal penetrating injuries are conveniently separated into four categories:(a)instrumental,
P.2103
P.2104
(b) foreign body injuries, (c) noninstrumental (barotrauma), and (d) other rare causes (Table 138-2).
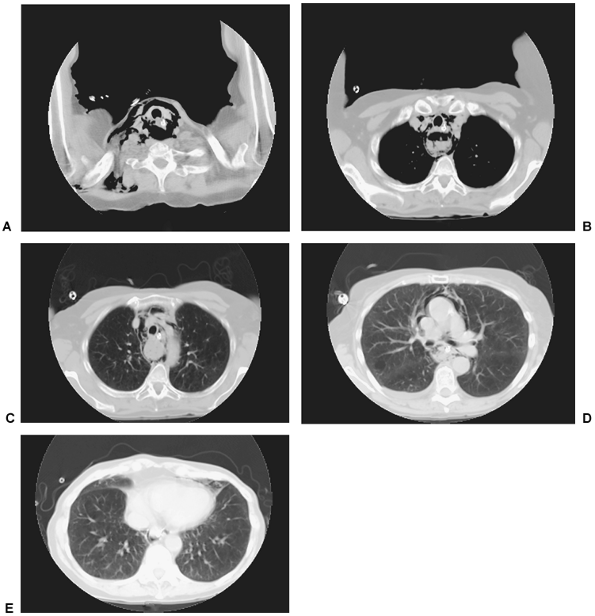 |
Fig. 138-1. Computed tomography scans of the mediastinum of a 72-year-old woman who had a distal carcinoma of the esophagus. A small cervical perforation on the right side occurred during a diagnostic flexible esophagoscopy but was treated conservatively at an outside hospital. A. Massive collection of air seen in the fascial spaces of the neck. B, C. Air extending down into the mediastinum surrounding the trachea and esophagus. A posterior inflammatory mass is seen behind the trachea. D. Air around the superior vena cava and ascending aorta just below the level of the carina. E. Air anterior to the heart. Courtesy of Geoffrey M. Graeber, West Virginia University School of Medicine, Morgantown, West Virginia. |
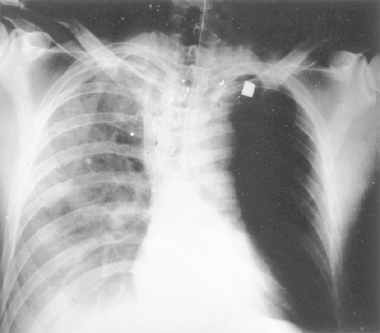 |
Fig. 138-2. Gunshot wound of the neck with perforation of the wall of the cervical segment of the esophagus. |
Instrumental Injuries
Instrumental injuries may occur during diagnostic or therapeutic procedures involving the esophagus. The incidence is low in both flexible and rigid esophagoscopy. In the latter, Wychulis and associates (1969) reported the incidence to be about 0.4% in routine diagnostic examination. Meyers and Ghahremani (1975) noted, however, that the actual number of perforations might be increasing because of more frequent use of the flexible esophagoscope.
The more common sites of perforation are at two of the normal anatomic sites of narrowing of the thoracic portion of the esophagus: the distal end as it reaches the diaphragm to join the stomach, and the area of narrowing at the level of the aortic arch and left main stem bronchus. When a pathologic process is present, examination with biopsy or therapeutic dilation may lead to rupture at the involved site. An incidence of 4.9% following pneumatic dilation for achalasia was reported by Bennett and Hendrix (1970), but it was only 1.4% in the series reported by Ferguson (1991). However, such injuries constitute up to 20% of esophageal perforations in many series, as exemplified by the experience of Iannettoni and associates (1997) at the University of Michigan. Rarely, rupture may occur with improper placement and inflation of the gastric balloon of a Sengstaken-Blakemore tube to control bleeding esophageal varices (Fig. 138-3). Perforation during the placement of an indwelling tube for palliation of a malignant stricture is not an uncommon event. Shemesh and Bat (1986) reported that endoscopic injection sclerotherapy for esophageal varices has resulted in esophageal perforations. Edling and Bacon (1991) have reported that small pleural effusions complicate this procedure about 50% of the time. If the effusion persists or occupies more than 25% of the hemithorax, Light (2000) advises that a thoracentesis be done to rule out an empyema or an esophageal perforation (the latter diagnosis is suggested by a high pleural fluid amylase level). When either is present, a contrast swallow should be done. Perforation also may occur after sclerotherapy of bleeding from a Mallory Weiss laceration in the gastric cardia. As noted subsequently, such an injury ended up as gangrene and perforation of the esophagus in a patient reported by Salo and associates (2000). However, perforations after endoscopic management of bleeding from a Mallory Weiss laceration are usually not followed by such a serious complication, and the perforation can be managed by one of the conventional surgical interventions.
Table 138-2. Intraluminal Penetrating Injuries of the Thoracic Esophagus | |
|---|---|
|
Perforation also has occurred after transesophageal echocardiography during cardiac operations. This latter event is more common than heretofore thought. Daniel and co workers (1991) recorded esophageal perforation as one of the
P.2105
complications of transesophageal echocardiography and noted that the incidence was 0.18% but that it was associated with a low mortality rate of 0.0098%. A fatal outcome is mainly due to failure to identify initially the occurrence of the injury. The cause of the injury is not known, but it has been suggested to result from the development of high intraluminal pressure at the site of the probe in an area where the esophagus has been fixed to the surrounding tissues by previous infection, irradiation, or by the presence of chronic ischemia due to local vascular insufficiency. Urbanowicz and colleagues (1990) have shown experimentally that high pressure in the probe did not occur in the normal esophagus in either humans or canines. The same authors reported that tissue injury also was not found to occur because of prolonged heating through the ultrasound energy. Since the early report by Daniel and colleagues (1991) of this injury, numerous reports have been recorded in the literature, such as those by Kharasch and Sivarajan (1996), as well as by Massey (2000) and Brinkman (2001) and their associates, among others, including two cases in Port and associates' (2003) recent series. The important factor in all the aforementioned injuries is to consider the possibility of its occurrence when clinical signs or symptoms of abnormal pleural contamination by air or salivary contents occur when none should be expected.
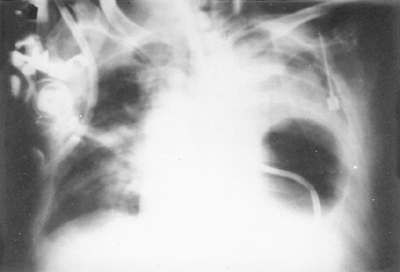 |
Fig. 138-3. Perforation of the thoracic esophagus by the gastric balloon of a Sengstaken-Blakemore tube. |
The clinical features and management of perforation from any of the aforementioned sources depend on the location and extent of the injury. The time interval between injury and its recognition is an important factor in regards to the extent of the inflammatory process, but at present, it is not regarded as a major factor in determining the appropriate therapeutic management nor of a successful outcome of repair of the injury.
Clinical Presentation of Thoracic Perforation
Although perforation of the wall of the thoracic esophagus may lead initially only to contamination of the visceral compartment of the mediastinum, most often, the mediastinal pleural layer is perforated as well, and this perforation leads to prompt contamination of the affected pleural space. The left pleural space is usually involved when the injury is in the most distal portion of the esophagus, the right when the perforation is more proximal in the esophagus. Because of the necrotizing inflammatory process that accompanies these injuries, the infection may spread and involve other structures in or adjacent to the mediastinum; the pericardium or even the central nervous system (CNS) may become involved. The occurrence of CNS involvement is quite rare and is seen most often with a perforation located in the proximal portion of the thoracic esophagus. Frequently, a localized mediastinal abscess is present that has fistulized into the esophageal subarachnoid space, with subsequent infection and air in the thecal space that may migrate into the meninges of the cranium (Fig. 138-4). Computed tomographic (CT) and magnetic resonance image (MRI) examinations demonstrate varying amounts of air in the subarachnoid space. Irritation or infection of the meninges is manifest by a panoply of symptoms that occur with spinal and cranial meningitis. Esophageal-subarachnoid fistulas have been described by Kotler (1975), Cornwell (1986), Ring (1995), Boulis (1999), and Jurani (2002) and their colleagues. The initial esophageal perforation in the aforementioned reports was the result of: (a) dilation of lye strictures, (b) rupture of a carcinoma of the esophagus, (c) blunt trauma, (d) placement of an esophageal stent for a benign stricture, and (e) a spontaneous perforation, respectively.
 |
Fig. 138-4. Magnetic resonance imaging sagittal view. T1-weighted image demonstrating air in the thecal sac (arrowheads) and paraesophageal abscess (arrow). From Jurani CC, Early GL, Roberts SR: Spontaneous esophageal perforation presenting as meningitis. Ann Thorac Surg 73:1294, 2002. With permission. |
Injuries to the thoracic esophagus are associated with pain, fever, dysphagia, and, frequently, respiratory distress. The pain may be thoracic, precordial, or even epigastric. Radiation of the pain may occur to the intrascapular region. With contamination of the pleural space, the patient may experience severe unilateral pleural pain that is aggravated by breathing as well as pain in the ipsilateral shoulder area. Fever is present, and tachycardia frequently is disproportionate to the degree of temperature elevation. Dysphagia is present and may be localized by the patient to the vicinity of perforation. The degree of respiratory distress varies with the severity of the pleural contamination, the amount of hydropneumothorax, and, at times, the presence of airway compression. Excessive thirst may be present.
Physical examination may reveal the patient to be toxic, with grunting, guarded respirations. Subcutaneous air may
P.2106
be palpated in the neck, and signs suggestive of a hydropneumothorax may be elicited.
Diagnostic Procedures
Radiographic examination of the chest may reveal a widened mediastinal shadow, mediastinal air, and varying amounts of air or fluid, or both, in either pleural space (Fig. 138-5). Pneumopericardium rarely may be observed; even more so is the demonstration of air in the spinal cord. The radiologic examination may be normal in 12% to 33% of the cases as reported by Wychulis (1969) and Han (1985) and their associates. Evaluation of the lumen of the esophagus and demonstration of the site of the leak may be accomplished by fluoroscopy and ingestion of a suitable contrast medium. A water-soluble contrast medium can be used, but better results are obtained with water suspended barium sulfate. CT may be helpful when the aforementioned diagnostic studies have been negative. In addition, Jones and Ginsberg (1992) recommend the use of flexible esophagoscopy, as suggested by Moghissi and Pender (1988), as an adjunct diagnostic method to localize accurately and directly visualize the site of perforation.
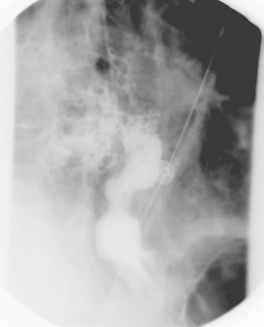 |
Fig. 138-5. Radiograph of the chest that shows extravasation of contrast material into the right pleural space from perforation of the distal end of the esophagus following dilation of a stricture caused by gastroesophageal reflux disease in a 62-year-old man. Courtesy of Geoffrey M. Graeber, West Virginia University School of Medicine, Morgantown, West Virginia. |
Management of Thoracic Esophageal Perforations
As Mayer and associates (1977) emphasized, the management of these injuries is based on four principles: (a) elimination of the source of soilage, (b) provision of adequate drainage, (c) augmentation of host defenses by antibiotics, and (d) maintenance of adequate nutrition. The techniques to achieve these four desired goals vary with the initial status of the esophagus (normal or diseased, benign or malignant); the extent of contamination of the mediastinum or the pleural space, or both; and the underlying nutritional status of the patient. The time of recognition and institution of therapy after the occurrence of injury is of less importance than formerly believed.
At present, most surgeons recommend immediate surgical intervention except under unusual circumstances when a nonoperative strategy may be used. Such is the case of a chronic, contained perforation, as described by Cameron (1979) and Port (2003) and their associates, in which the perforation is contained within the mediastinum and contrast studies show that the contrast material drains readily back into the esophageal lumen and there are minimal or no clinical signs of sepsis evident. Nasogastric suction is initiated, and appropriate antibiotics are given as well. Parenteral nutrition or feeding jejunostomy is established in most of these cases. Sawyer and colleagues (1995) managed 22% of 31 acute perforations in this manner. Their criteria for selecting such an approach were: (a) a recent perforation (within 24 hours), (b) no food intake after the episode of perforation, (c) the perforation not proximal to a high-grade stenosis, (d) minor symptoms without clinical signs of sepsis or hemodynamic compromise, (e) a contained perforation within the mediastinum, and (f) contrast studies showing good, prompt drainage from a small perforation into the esophageal lumen. Strict adherence to these criteria must be followed if one is to use this conservative approach. Port and colleagues (2003) carried this out in 5 of 26 perforations (19%), although they did not confine its use only to the early recognized perforations.
The aforementioned approach is the exception rather than the rule. Multiple surgical options are available (Table 138-3), but in most cases, primary closure, usually with buttressing of the suture line, is recommended, as reported by Ohri (1993), Whyte (1995), Wright (1995), Wang (1996), and Port (2003) and their co workers, among others, regardless of the amount of time that has elapsed since the occurrence of the esophageal perforation. The actual technique of closure with and without the use of a buttress of tissue to cover the closure varies from group to group. The techniques used by the University of Michigan and the Massachusetts General Hospital staffs are seen in Figures 138-6 and 138-7. Correction of any distal obstructing process (functional or organic) is essential for success, as is adequate mediastinal and pleural drainage.
When the perforation has occurred after a pneumatic dilation for achalasia, the usual recommendation is to mobilize the lower esophagus and to perform a modified Heller
P.2107
esophagomyotomy (see Chapter 130) opposite the site of the perforation. If the integrity of the diaphragmatic hiatus has been compromised, an antireflux procedure should be done. Recently, Urbani and Mathisen (2000) have advocated that in place of mobilization of the esophagus and the performance of a Heller procedure, the esophageal wound is d brided, and the entire length of the mucosal tear is exposed and securely closed. An intercostal muscle flap is then sutured (4 0 silk sutures using a horizontal mattress technique) to the edges of the ruptured esophageal muscle to ensure a tight closure (Fig. 138-8). The approximation of the muscle flap to the ruptured edges of the esophageal muscle maintains the myotomy created by the balloon dilation. Drainage of the thorax is established, and a draining gastrostomy and feeding jejunostomy are performed. As per usual, antibiotics as well as nutritional support are indicated. At the time of their report, the patients so treated have had excellent results.
Table 138-3. Operative Management of Esophageal Perforations | |
|---|---|
|
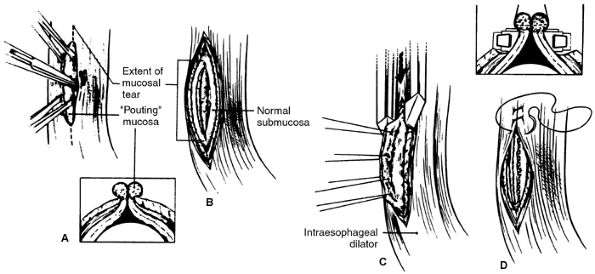 |
Fig. 138-6. Primary closure of an esophageal perforation. A. Mucosa pouting through muscular defect (insert) is grasped with Allis clamps in preparation for exposure of entire tear. Right-angle clamp is used to separate muscularis from underlying submucosa, and vertical esophagomyotomy (dotted line) is made to expose entire limits of tear. B. These maneuvers expose circumferential rim of normal submucosa that can be closed. C. Stay sutures, placed into inflamed pouting mucosa, are used to elevate normal submucosa into jaws of GIA stapler (insert), which is placed below inflamed, edematous mucosal edges (dotted line). D. Repair is then completed by closing muscularis with running suture. From Whyte RI, Iannettoni MD, Orringer MB: Intrathoracic esophageal perforation: the merit of primary repair. J Thorac Cardiovasc Surg 109:140, 1995. With permission from the Society of Thoracic Surgeons. |
A number of surgeons, including Vogel (1991) and Bardaxoglou (1994) and their colleagues, as well as Tasdemir (1996), have reported the use of fibrin glue to obtain or to support a primary repair of a perforated esophagus. It has been used to seal the suture line, to obtain better adherence of a transposed muscle flap, and by Bardaxoglou and colleagues (1994) to seal an absorbable mesh polyglactin mesh (over the primary repair). These authors reported four favorable results in five patients. The use of fibrin glue may become a valuable adjunct in the surgical repair of esophageal perforations, but additional studies must be obtained before general acceptance.
Recently, Nana and associates (2003) have successfully treated an esophageal perforation due to transesophageal echocardiography by the placement of a coated esophageal stent (Ultraflex, 2.2 cm diameter and 13 cm long, Boston Scientific, Natick, MA) and pleural drainage. The technique of using a stent for the treatment of a perforation after echocardiography was initially described by Zalunardo and collaborators (2002). Additional experience will be needed to establish the ultimate value of this approach for control of an instrumental injury to the esophagus.
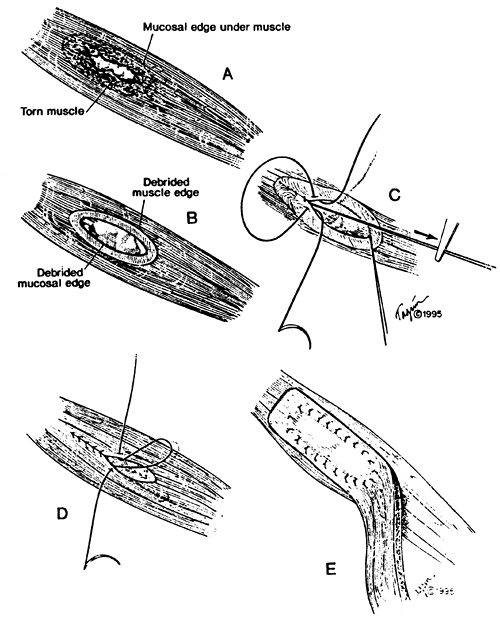 |
Fig. 138-7. Technique of reinforced primary repair of esophageal perforation. A. Ragged partially necrotic muscle at the border of the tear hides the full extent of the mucosal injury. B. Necrotic muscle has been d brided back to expose the mucosal defect fully, and the mucosal edge is trimmed to healthy tissue. C. Inventing interrupted fine 4-0 sutures are placed and tied so that the knots remain inside. Inward traction on the previously placed suture facilitates proper mucosal inversion. Suturing is begun in each corner and finished in the middle. D. A second layer of interrupted fine 4-0 sutures is placed if possible in the muscle layer to cover the mucosal repair. E. A previously harvested intercostal muscle flap with an intact vascular bundle is swung on its posteriorly based pedicle to cover and buttress the repair. This is sutured over the closure with interrupted mattress sutures in a circumferential fashion to make a third watertight layer. All sutures are placed first before trying to facilitate an exact repair. From Wright CD, et al: Reinforced primary repair of thoracic esophageal perforation. Ann Thorac Surg 60:245, 1995. With permission from the Society of Thoracic Surgeons. |
P.2108
When the esophageal injury has occurred in or above an obstructing lesion, direct repair of the esophageal injury is contraindicated unless the obstructing lesion can be corrected at the same time. If not, then continued soilage must be prevented by diversion of oropharyngeal and gastric secretions from the area of esophageal injury in concert with adequate drainage of the mediastinal and pleural space.
The prevention of continued soilage is a perplexing problem. Numerous methods have been proposed, from the simple use of multiple indwelling nasogastric tubes with or without a decompressing gastrostomy to complex surgical procedures to close the perforation or to isolate the thoracic esophagus or even primary esophagectomy. Inherent in all suggested treatments, as noted, is the adequate drainage of the pleural and mediastinal spaces.
The major problem generally is the reluctance of the surgeon to adopt an aggressive policy in the management of these injuries, and frequently it is a story of too little, too late. Most often, once the patient's condition is stabilized by fluid replacement and other necessary resuscitative measures, the patient should undergo exploratory thoracotomy and the appropriate procedure carried out.
One option is an esophageal exclusion and diversion. Many techniques have been suggested, but one of the more suitable ones is a modified esophageal exclusion initially suggested by Urschel and associates (1974), in which pharyngoesophagogastric continuity can be restored without major surgical intervention. Urschel's technique, as described in the fourth edition of this text (1994) has been superseded by the use of absorbable surgical staples to seal off the esophagus. Chang and associates (1992) diverted the esophagus with a T tube placed in the cervical esophagus and then placed a suture line of absorbable staples distal and proximal to the site of perforation. The T tube is removed in 4 weeks, and normal swallowing returns rapidly. Bardini and colleagues (1992) reported a similar technique; the esophagus is stapled in the neck and in the abdominal portion with Polysorb 55 absorbable sutures (U.S. Surgical, Norwalk, CT), drainage is either through the neck or abdomen depending on the level of the perforation, the pleural cavity is drained only if necessary, a feeding jejunostomy is put in place, and the upper cervical esophagus is decompressed by a soft nasogastric tube. Paramesh and colleagues (1995) reported the use of nonabsorbable staples to achieve the esophageal exclusion (Ethicon TPH 60 stapler) and recorded spontaneous recanalization of the esophagus in 2 weeks without the occurrence of any evidence of leakage from the original perforated site. Triggiani and Belsey (1977), however, as well as DeMeester (1986), do not hesitate to exclude the esophagus completely, with subsequent restoration of pharyngogastric continuity with an interposed segment of bowel or stomach at a later date. When excluding the esophagus, the upper segment should be left as long as possible. The excess length of viable proximal esophagus is brought out through the cervical mediastinotomy incision and placed subcutaneously beneath the skin of the anterior chest wall. The esophageal stoma is placed at an appropriate site on the chest wall. An adequate length of proximal esophagus is thus preserved for subsequent reconstruction, rather than having only a short segment of cervical esophagus when the esophagus is divided at a higher level and brought out as a cervical esophageal stoma at the site of the cervical incision. However, these aforementioned procedures have fallen out of favor and only infrequently are done, particularly because there has been a high mortality rate in patients so treated.
Another technique for the management of a patient with a perforation that is difficult to close (a friable esophageal perforation or one in which the diagnosis has been markedly delayed) is the use of a T tube to establish a controlled fistula
P.2109
for drainage of the perforation (Fig. 138-9). First described by Abbott and associates in 1970, this technique periodically has been resurrected and was reported by Bufkin and co-workers (1996) with successful results. Although not recommended as a routine measure, it may be considered as an acceptable alternative in a poor risk patient.
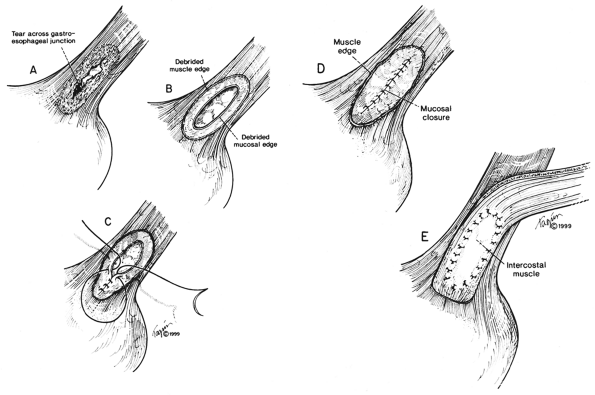 |
Fig. 138-8. A. Esophageal perforation usually crosses the gastroesophageal junction. B. Any devitalized tissue must be d brided and extent of mucosal tear accurately identified. C. The mucosa is carefully closed with interrupted 4-0 silk sutures with knots on the inside to invert the mucosa. D. The mucosal closure should be checked for any leaks. E. The pedicled intercostal muscle is carefully sewn to the muscular edge of the defect with interrupted 4-0 silk mattress sutures. From Urbani M, Mathisen DJ: Repair of esophageal perforation after treatment for achalasia. Ann Thorac Surg 69:1609, 2000. With permission. |
In patients with carcinoma or other serious accompanying esophageal disease (megaesophagus, severe reflux with major stricture, severe lye burn, or late lye stricture), esophagectomy may be considered as the treatment of choice, as Mayer and colleagues (1977) suggested; reconstitution of esophagogastric continuity may be done at the same time or at a later date.
Occasionally, a patient with irradiated, nonresectable carcinomas suffers a perforation during an attempted peroral dilation. This injury may result in a chronic, contained perforation, as Cameron and associates (1979) described, or as a free perforation. I have conservatively managed several such patients successfully, and Wesdorp and colleagues (1984) treat all such perforations of esophageal tumors in this conservative manner and report a success rate of more than 90%. Other investigators, such as Sandrasagra and associates (1978), have recommended the insertion of an esophageal tube as a stent in such situations. However, when there is a free perforation of a malignant lesion or an extensive benign stricture or other serious esophageal disease that cannot be readily repaired, or such a repair is inadvisable, esophageal resection has become an acceptable option. Orringer and Stirling (1990) and Orringer and associates (1993) have carried this out on a number of occasions (more than 24 patients) with success. A standard or a transhiatal resection may be done, and as noted previously, reconstruction may be carried out at the same time or may be delayed. Salo and associates (1993) have managed delayed-diagnosis perforations with evident mediastinal sepsis by primary esophagectomy with delayed reconstruction. Altorjay and colleagues (1998) carried out esophagectomy in 27 patients as the therapy for a perforated esophagus. A major stricture was present in 19, and cancer or achalasia in 2 each. Eight of the esophagectomies were transhiatal, and 19 were transthoracic. Primary reconstruction was done in 25 patients, and in 14 of these, the transposed organ was placed in the bed of the excised esophagus. The mortality rate was only 3.7%, with postoperative complications in 41%. A satisfactory result was obtained in most patients.
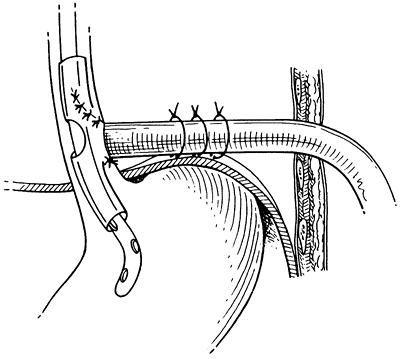 |
Fig. 138-9. T tube placed in the esophagus, secured to the diaphragm away from the aorta and brought out through the chest wall. From Bufkin BL, Miller JI Jr, Mansour KA: Esophageal perforation: emphasis on management. Ann Thorac Surg 61:1447, 1996. With permission.2 |
P.2110
Except for very rare circumstances, the consensus is that an expectant approach of simple pleural drainage in the management of esophageal perforation is less than ideal and is followed by frequent failure. Santos and Frater (1986), however, reported success with drainage only when it was accompanied by copious irrigation of the esophagus and pleural space by peroral intake of large volumes of fluid. I have had one success with such management through a serendipitous chain of events. Antibiotic coverage should be guided by culture of the drainage. Nutritional support may require intravenous hyperalimentation or a feeding jejunostomy. Management of each patient must be individualized, but the aforementioned initial four goals of successful treatment remain unchanged.
Minimally Invasive Techniques in Esophageal Perforations1
As expertise is gained in video assisted esophageal surgery, minimally invasive surgical approaches to esophageal perforations and leaks have been developed and have become the preferred approach in many situations. Nguyen and co-workers (2001) have summarized the fundamental goals in the minimally invasive approach to esophageal perforations: (a) identification of the esophageal perforation, (b) d bridement of necrotic debris, (c) control of the leak (primary closure or T-tube placement), and (d) wide drainage of the mediastinum.
Transcervical drainage is a highly effective means of controlling hypopharyngeal and proximal esophageal perforations, usually occurring secondary to trauma (gunshot wounds, difficult intubations). This approach can allow adequate exposure to control and repair perforations or leaks extending into the upper mediastinum down to the level of the carina. Mid distal esophageal perforations typically require transthoracic drainage. Some authors advocate the routine use of thoracotomy to provide maximal exposure in order to carry out a thorough d bridement, repair, or drainage. Increasingly, thoracoscopy is supplanting thoracotomy when conditions are right, as reviewed by Chung and Ritchie (2000). Scott and Rosin (1995) were the first to report a minimally invasive repair of esophageal rupture secondary to Boerhaave's syndrome. The usual thoracoscopic approach employs three to four trocars positioned conventionally through a right chest approach (see Fig. 136-3 in Chapter 136). A left video assisted thoracic surgery (VATS), as described by Ikeda and co-workers (2001), or transabdominal approach, as advocated by Landen and El Nakadi (2002), can be employed in distal esophageal perforations, or when the leak is demonstrated by esophagram to extend into the left chest. The initial step is retraction of the lung and evacuation of fibrinous debris and purulent exudates. Intraoperative endoscopy is performed to assist in identifying the site of perforation. The suspected region can be submerged under irrigation during endoscopic insufflation to pinpoint the precise location of perforation. Once identified, the devitalized margins of the perforation are d brided, and the decision is made whether to attempt a primary closure, depending on the degree of surrounding tissue injury as well as the clinical condition of the patient. If the defect is small (<1 cm) and surrounded by viable tissue, a primary closure can be performed with interrupted sutures. In the case of larger injuries or perforations surrounded by severely inflamed tissue, wide drainage is performed with placement of a T tube to control the leak. Wide defects may allow direct placement of a T tube through the perforation site. For smaller breaches, a loop wire can be inserted transthoracically through the perforation defect. Endoscopy is performed to retrieve the loop wire, which is then brought back through the mouth. The end of a T tube is sutured to the loop wire, and the wire and T tube are pulled transorally down the esophagus and out through the chest, with the T tube tip positioned intraluminally at the perforation site. Final positioning of the T tube can be confirmed by endoscopy. Jackson Pratt drains, as well as a No. 28F to 32F chest tube, are positioned strategically to provide wide drainage of the mediastinum and chest.
Special circumstances encountered during esophageal perforation provide opportunities for creative and effective surgical approaches to this serious problem. Perforations involving an esophageal diverticulum can be managed by minimally invasive diverticulectomy and drainage, as reported by Tinoco and colleagues (1999). Perforation of the
P.2111
distal esophagus after esophageal dilation for achalasia can occur with a frequency of 0% to 15%. The location of the perforation is typically the left posterior esophagus. Full-thickness perforations tend to begin within a centimeter of the squamocolumnar junction and extend proximally from a few millimeters to as much as 10 cm. These perforations can usually be repaired using a thoracoscopic or transabdominal laparoscopic technique with suture closure of the perforation, contralateral Heller myotomy, and Toupet posterior fundoplication, as summarized by Nathanson (1993) and colleagues and Bell (1997). A contralateral myotomy is performed extending 5 cm along the length of the esophagus and extending 1 cm onto the surface of the stomach, with care taken to spare the vagus nerves. A posterior fundoplication is then performed in the manner of Toupet (1963), suturing the edges of the myotomy to the edges of the plicated stomach over a length of 4 cm. This technique has the advantage of covering the closed esophageal perforation with a gastric serosal patch, while at the same time treating the underlying motility disorder. A closed suction drain is placed into the mediastinum. Early recognition and prompt surgical intervention allow primary closure in most circumstances without the need for muscular flaps or esophageal exclusion. Although the most common surgical approach is transthoracic, the excellent visualization of the distal 6 to 7 cm of thoracic esophagus is amenable to a transabdominal laparoscopic approach, as advocated by Swanstrom and Pennings (1995). Definitive treatment of the underlying condition (achalasia) is permitted by performance of a myotomy and fundoplication, thus diminishing the risk for postmyotomy reflux. As summarized by Laisaar (1998), a transthoracic approach should probably be used if the tear extends more than 5 cm proximally, or if there is gross extravasation of contrast into the left pleural space. Adequate visualization of the entire circumference of the esophagus is possible by gentle rotation; thus, the location of the tear on the circumference of the esophagus is not a significant issue in deciding on this approach.
Advantages of a minimally invasive approach include an excellent, magnified view of the entire thoracic cavity as well as the ability to achieve adequate d bridement and drainage of the mediastinum and pleura, as summarized by Roberts (1997) and co-workers. Either primary closure or an esophageal T tube with drainage can be used to control the esophageal leak. If any difficulties arise, it is always possible to convert to open thoracostomy. By avoiding thoracotomy, however, the patient experiences less postoperative pain, a decrease in wound-related complications, and a faster postoperative recovery.
Mortality and Morbidity
The morbidity and mortality rates vary with instrumental injury of the esophagus. Historically, Jones and Ginsberg (1992) noted a mortality rate of 19% in patients with instrumental perforation. It is difficult to determine the mortality rate after treatment of iatrogenic perforations because almost all reports combine the mortality rate of all perforations treated at any given institution. Nonetheless, in 11 reports in the literature beginning in 1995 through early 2003, 327 patients with esophageal perforations were reported, and an overall mortality rate of 11.9% was recorded. The mortality data for each report are recorded in Table 138-4.
Early morbidity in most series is mainly due to a leak from the suture line [20% in Iannettoni and colleagues' (1997) series]. In Wright and associates' (1995) series, the rate was 11% in patients without preoperative infection and 70% in those that had such an infection. In almost all, the leak was managed successfully by simple closed tube thoracotomy to establish satisfactory drainage. Persistent empyema of the affected pleural space may occur infrequently.
Late dysphagia, according to Saabye and colleagues (1988), is rare in patients in whom the esophagus was normal at the time of perforation; however, in patients with stricture, almost one half required subsequent dilations or even an esophagectomy in one patient. In Sawyer and associates' series (1995), almost two thirds of the patients complained of long term dysphagia, and 21% of all patients required subsequent dilations. In the Michigan University series reported by Iannettoni and co workers (1997), 43% of the patients who had undergone primary repair required postoperative dilations, whereas 52% of those who had undergone resection did so. The poorest long term results occurred in patients who initially had long standing gastroesophageal reflux with stricture.
Foreign Body Injuries
In review of the literature, Barber and associates (1984) mentioned a variety of objects and materials that were inappropriately ingested, especially by children, by mentally disturbed or deranged persons, and occasionally by individuals with upper and lower dentures. Many such objects pass into the stomach; others lodge in the esophagus and require removal by endoscopic manipulation. In some instances,
P.2112
sharp or jagged foreign bodies lacerate the wall partially or completely. Most commonly, such laceration occurs in the cervical esophagus, but any point of normal narrowing in the thoracic esophagus or at a diseased area may be the site of perforation. The more common offenders are: (a) bones, especially chicken or pork bones (fish bones more commonly lodge in the hypopharynx although many will pass into the thoracic esophagus); (b) bits of shellfish shells; (c) partial dentures; (d) plastic eating utensils (frequently in the mentally disturbed); and (e) metal objects such as open safety pins or metal beverage can openers. Perforation of the wall may occur spontaneously or during the extraction of the foreign body. Although a patient may become acutely ill from perforation by such objects, an indolent course with late abscess formation or pyopneumothorax is common (Fig. 138-10). These complications tend to occur, especially in a child or a mentally troubled individual who does not recognize the ingestion of the foreign body.
Table 138-4. Mortality associated with Perforation of the Thoracic Esophagus | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
The diagnosis of foreign body perforation may be suggested by the history and physical examination and a high index of suspicion in the various patient groups. Radiographic examinations may reveal the foreign body, but frequently they are negative, owing to the low density of many of the foreign bodies. The use of soft tissue techniques, as noted by Love and Berkow (1978), permits visualization of about 75% of ingested bones, and further improvement may be noted with the use of xeroradiography. The other radiographic features depend on the degree and the extent of the inflammatory process external to the esophageal wall. When an unexplained hydropneumothorax is evident, thoracentesis confirms the pyogenic nature of the process.
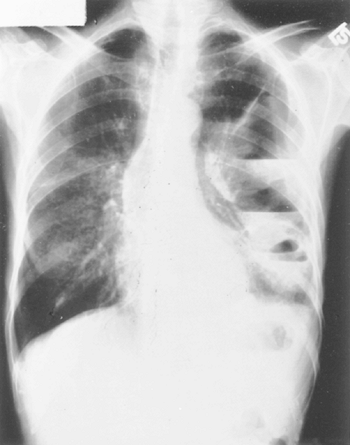 |
Fig. 138-10. Loculated pyopneumothorax following erosion of a nonopaque foreign body through the wall of the distal end of the thoracic esophagus. |
Endoscopy with removal of the foreign body is indicated, along with appropriate drainage of the involved area. Esophagotomy is necessary for removal of any foreign body that cannot be removed by endoscopic manipulation. In the neck, this procedure is accomplished through a cervical mediastinotomy, whereas in the chest, either a posterior mediastinotomy or a thoracotomy approach is indicated, depending on the extent of the disease process. It would appear that a thoracoscopic approach for the removal of an impacted foreign body could be done without difficulty. The morbidity and mortality of foreign body perforation vary with the time of recognition and the appropriateness of the therapy. Bladergroen and associates (1986) reported a 94% survival rate in this group of patients.
Barotrauma (Noninstrumental Injuries)
Rapid increase of the intraluminal pressure within the esophagus may result in partial or complete rupture of the esophageal wall. This rupture is usually the result of internal changes in pressure accompanying vomiting, but it may occur with defecation, convulsions, lifting, and even labor of childbirth. It may also be a remote effect of blunt abdominal trauma and deceleration injuries. Rarely, it may be the result of the exposure to compressed air. Twelve accidental pneumatic ruptures of the esophagus were reported by Gelfand and associates (1977). Letquart and colleagues (1981) have also reviewed this subject. In 12 cases, 3 deaths occurred in 5 nonsurgically treated patients, and only 1 death occurred in the 7 patients who underwent operation. Common sources of the barotrauma were accidents involving compressed air hoses and tanks or explosion of an inflated tire when a young child bit a bulge on an inner tube that protruded through a defect in the wall of a tire. Conlan and associates (1984) reported several cases that occurred in children who had been playing with carbonated beverages.
Boerhaave's Syndrome (Spontaneous Rupture)
Hermann Boerhaave described the first case of rupture of the esophagus in 1724. This was discovered in the autopsy of Baron von Wassenaer, Grand Admiral of the Dutch Fleet, who died after stuffing himself with food and drink and inducing vomiting by taking an ipecaclike drug as an aftermath of his overindulgence. Nesbitt and Walsh (1998) have noted that the details of Boerhaave's findings have been recorded in the report of Debers and Mitchell published in 1955. According to Barrett (1947), Meyer reported the antemortem diagnosis of Boerhaave's syndrome in 1852 but successful treatment was not published until the mid-1940s by Barrett (1947), Frink (1947), and Olsen and Clagett (1947).
P.2113
The etiology of Boerhaave's syndrome is thought to be a rapid increase of the intraluminal pressure of the esophagus that may occur with the act of vomiting [high pressure forced through a patent lower esophageal sphincter against a closed upper esophageal sphincter (the cricopharyngeus muscle)]. Hardy and Wallace (1977) noted that a force of 0.5 to 1.5 kg (in V/sec2) of pressure was necessary to rupture the normal esophagus but that less pressure is needed when esophageal disease is present. Most often, the rupture is located in the distal portion of the intrathoracic esophagus as it lies anterior and to the left of the aorta (the terminal 6 to 8 cm). The injury extends through all layers of the posterolateral wall on the left. It has been suggested by some that this is the result of the distribution of the smooth muscle at this location. In this region, the longitudinal fibers taper out as they pass onto the stomach wall, resulting in a weakened area at this site. However, Hagen and DeMeester (2000) have suggested that the rupture occurs here because of the lack of support from adjacent structures, the esophagus being covered on its left lateral wall only by the parietal pleura of the chest wall.
On occasion, a partial disruption occurs. Rarely, extensive dissection of the air within the intramural layers of the esophagus occurs, as described by Borrie and Sheat (1970) and by Kelley (1972) and Berliner (1982) and their associates. A partial laceration of the wall may extend into the proximal stomach and cause major upper gastrointestinal bleeding, the Mallory Weiss syndrome [Mallory and Weiss (1929)]. In this situation, the problem is control of the bleeding. If bleeding does not stop with expectant, supportive management or endoscopic sclerosing injection, surgical intervention is required. This operative procedure is best accomplished by an abdominal approach, gastrotomy, and suture ligation of the bleeding point.
Clinical Features of Boerhaave's Syndrome and Other Spontaneous Ruptures of the Esophagus.
Massive bleeding with complete rupture of the esophageal wall is rare. Varying amounts of bleeding were recorded in 55% of the patients seen by Abbott and colleagues (1970), but this high incidence has not been confirmed by other authors. Classically, patients with so called spontaneous rupture of the esophagus present with severe chest pain and dyspnea after an episode of vomiting. Shoulder pain may be present, but some patients complain only of abdominal pain that can be located more specifically in the epigastrium. Marked thirst is occasionally observed. In many patients (e.g., the unconscious or unreliable alcoholic patient), the history is unobtainable, and the diagnosis must be established by the physical examination, radiographic findings, and at times, thoracentesis. Unfortunately, a major delay in time of diagnosis relative to the occurrence of the event is common in many cases of spontaneous rupture.
The physical findings vary with the duration of the disease and the involvement of either pleural space. Subcutaneous emphysema is present in most patients, and cyanosis may be observed. The abdominal findings may vary from epigastric tenderness to upper abdominal distention. Bowel sounds frequently are decreased to absent.
Diagnosis of Boerhaave's Syndrome.
Radiographic examination of the chest and abdomen reveals, in varying degrees, mediastinal emphysema, pleural effusion, hydropneumothorax, and rarely pneumoperitoneum. Early, and particularly helpful in patients with abdominal complaints, a patchy, irregular density may be visible behind the left cardiac silhouette. This feature has been termed the V sign by Naclerio (1957). CT with contrast media opacification of the esophagus is helpful in localizing the injury and is especially helpful when the clinical features are obscure (Fig. 138-11).
When patients have a hydropneumothorax and the history and other features do not suggest the underlying cause, a thoracentesis may be diagnostic. In adults, when thoracentesis reveals a pyopneumothorax, the diagnosis of a perforating injury of the esophagus must be considered the most likely possibility until proved otherwise.
Treatment of Boerhaave's Syndrome or Spontaneous Rupture.
Once the diagnosis is established, a multifaceted therapeutic approach is indicated to achieve the four aforementioned goals in the management of the esophageal injuries. Direct surgical repair, when there has been either an early diagnosis or a late one, with buttressing of the closure is the procedure of choice, as described previously, and leads to the least morbidity and mortality. In some recent cases of Boerhaave's rupture of the esophagus, as noted earlier in this chapter, operative repair has been carried out by a thoracoscopic approach. Scott and Rosin (1995) reported the first case, but the patient developed a postoperative leak; however, a second case, reported by Ikeda and colleagues (2001), suffered no postoperative complications. The latter authors believed that a more secure and easily accomplished closure was obtained by the use of the Endo-stitch loading unit (U.S. Surgical, Norwalk, CT). Landon and El Nakadi (2002) also have used a minimally invasive technique in three cases of Boerhaave's syndrome through an abdominal (laparoscopic) approach. In patients whose injury is of long duration, however, complications are frequent, and a protracted course may be expected. Most complications (development of an empyema, persistent fistulas, and late abscess formation) are related to leakage from the site of repair. Infrequently, functional and, at times, actual surgical isolation of the esophagus from the gastrointestinal tract or T-tube drainage, as well as adequate pleural drainage, may be indicated. These latter two steps may be required when no direct surgical repair of the rupture site is feasible. An esophagectomy is rarely indicated in these cases. A nasogastric tube in the esophagus for aspiration of oral secretions and gastrostomy to prevent reflux into the esophagus are indicated. Antibiotics and hyperalimentation are essential adjuncts in the management of these patients.
Mortality in patients with a Boerhaave's rupture depends on the underlying physical status of the patient and the aggressiveness
P.2114
and appropriate individualization of therapy. Abbott and colleagues (1970) reported a mortality rate of 51.4% in 35 treated patients; all 12 conservatively treated patients died. Keighley and associates (1972) reported only an 8% mortality rate in a smaller number of patients; the norm is probably somewhere in between. Jones and Ginsberg (1992) reported a mortality rate of 39% in their collective review. However, these authors did note that in two series [i.e., those of Graeber and colleagues (1987) and Triggiani and Belsey (1977)], there was no significant difference in the mortality following spontaneous and iatrogenic perforations. In Lawrence and co-workers' (1999) series of 21 patients with a Boerhaave's rupture, 17 patients had a primary closure, 2 were treated conservatively, and 2 had a thoracoscopic debridement. Three patients died, a mortality rate of 14%.
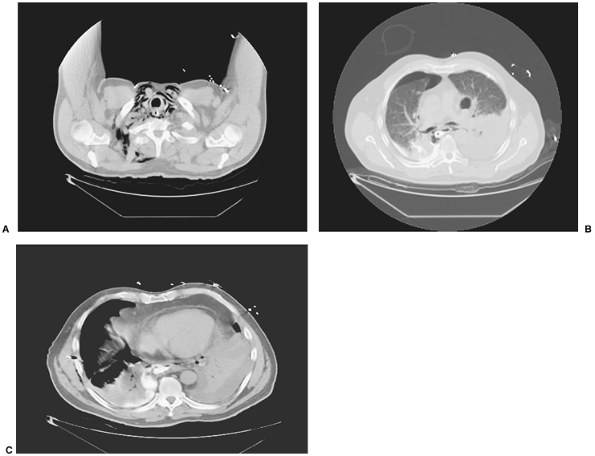 |
Fig. 138-11. Three selected levels of a CT scan with contrast swallow in a 57-year-old man who had a postemetic perforation of the esophagus (Boerhaave's syndrome). The patient had presented in an atypical fashion because the perforation was into the right pleural space. A. Scan of cervical area with collection of air in right fascial spaces. Contrast material seen in the esophageal lumen. B. Bilateral pyopneumothoraces with contrast material in the esophagus and right pleural space. C. Site of perforation of esophagus with egress of contrast material into the right pleural space. Courtesy of Geoffrey M. Graeber, West Virginia University School of Medicine, Morgantown, West Virginia. |
Unusual Injuries of the Esophagus
Rupture of a Barrett's Ulcer
The rupture of a Barrett's ulcer of the esophagus is an unusual event. Guillem and associates (2000) described three cases and retrospectively reviewed 31 previous reports. The sites of perforation are listed in Table 138-5. The presentation and clinical findings were heterogeneous and were basically determined by the site of the perforation. Only
P.2115
14.3% of the patients were known to have Barrett's esophagitis. Most of the patients were men, and in those who history was available, the symptoms in the order of decreasing frequency were: (a) hematemesis (39%), (b) fever (35%), (c) abdominal pain (30), (d) thoracic pain (30%), (e) melena (26%), and (f) nausea and vomiting (26%). The diagnosis was established preoperatively in 61%, at operation in 13%, and at autopsy in 26% of 23 patients. Nonsurgical management was successful in 2 of 9 patients and with various surgical procedures in 10 of 20 patients in the 31 cases from the literature, including three cases of the authors. Of the authors' three cases, two of the three patients survived. Although survival followed drainage alone, antireflux procedures alone, and primary closure, the preferred surgical procedure is an early esophagectomy with either an immediate or a delayed reconstruction; the latter may be the better procedure. Guillem and colleagues (2000) stressed the seriousness of the presence of an aortoesophageal fistula and suggested the value of esophageal exclusion diversion in these cases. Again, either an immediate or a delayed substernal gastric interposition can be done.
Table 138-5. Sites of Perforation in Barrett's Ulcera | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||
With nonsurgical management (11 patients), only 2 (22%) of 9 followed patients survived. With surgical intervention of various types (20 patients), 10 (77%) of 13 patients with known follow-up survived. The overall mortality rate was 45% for 31 of the 34 cases presented in the review.
Esophageal Perforation Associated with Gas Gangrene
Salo and co workers (2000) reported the occurrence of perforation of the esophagus associated with gas gangrene of the esophagus, mediastinum, and adjacent structures rapidly following the endoscopic ethanol injection of a bleeding Mallory-Weiss tear. Whether the esophageal rupture came first or followed the gangrene of the esophagus is conjecture. Nonetheless, emergency esophagectomy and local d bridement were carried out. The local infection by the Clostridium perfringens organs continued unabated. Multiple broad-spectrum antibiotics (imipenem, vancomycin, fluconazole, and metronidazole, as well as ceftriaxone irrigations) and subsequent seven hyperbaric oxygen therapy sessions finally controlled the infection. Multiple surgical procedures were required, and gastrointestinal continuity was reestablished by a colon interposition with eventual full recovery. The authors could find only one other example of the association of gas gangrene and perforation of the esophagus. This occurred in the cervical esophagus after the endoscopic removal of a chicken bone reported by Monies-Chass and colleagues (1975). Recovery occurred with drainage, antibiotic, and hyperbaric oxygen treatment.
Rupture and Necrotizing Esophagitis in Immunocompromised Patients
Inflammatory diseases of the esophagus are predominately found in severely immunocompromised patients as discussed in Chapter 140. A number of these patients develop a necrotizing esophagitis, sustain an esophageal perforation, or develop a tracheoesophageal or bronchoesophageal fistula. The underlying immunosuppression results from chemotherapy (usually for a lymphoma), from human immunodeficiency virus (HIV) infection, acquired immunodeficiency syndrome (AIDS), or in a patient who has been on an immunosuppressive regimen for bone marrow or organ transplantation. The offending organisms may be drug-resistant bacteria, fungal organisms, viral, or mycobacterial (both typical or environmental in type) organisms.
Gaissert and associates (2003) managed four patients in a 7 year period and identified 21 additional patients with the aforementioned complications of severe esophageal infections. Perforation occurred in 11, tracheoesophageal or bronchoesophageal fistula in 8, and esophageal necrosis in 6 patients. Thirteen patients were treated conservatively, and only 1 patient was known to have survived (the underlying disease process was severe diabetes mellitus) and had been reported by Kim and co workers (2000). Twelve patients were managed surgically, primarily by drainage, diversion, or both (5 patients with 2 deaths and 1 not reported), esophageal stent and drainage (1 patient with survival), and esophagectomy (6 patients with 1 death). Overall, a 72.7% survival rate was noted in the surgically managed patients, with the result not reported in 1 patient. This is in contrast to a survival rate of only 10% with the results not reported in 3 of the 13 patients managed conservatively. The successful esophagectomies were done by Sehhat (1976) and Gaissert (1999, 2003) and their associates. The gastrointestinal continuity was reestablished either at the time of the esophagectomy or at a later date.
Tuberculous (TB) infection of the esophagus is rare and is usually the result of extension from the lung or involved mediastinal nodes. In the immunocompromised patient, however, the infection may be primary in nature. Ulceration, fistulization, or even perforation may occur. According to Grubbs and co workers (2001), perforation per se is rare, and they could only find four such cases in the literature
P.2116
that had been reported by Williford (1983), Adkins (1990), Schroder (1996), and Ghandour (1997) and their associates. An adequate course of antituberculosis medication and subsequent surgical closure are indicated, although, in Grubbs and colleagues' (2001) case, the perforation healed without surgical intervention.
Esophageal Perforation Associated with Zollinger-Ellison Syndrome
Ng and colleagues (2001) recorded a nonbarometric spontaneous perforation of the distal portion of the thoracic esophagus. Other than the presence of a Zollinger-Ellison syndrome, no other etiology could be discerned.
Extraluminal Penetrating Esophageal Injuries
Extraluminal penetrating injuries of the esophagus may occur following gunshot and stab wounds of the chest and upper abdomen, thermal wounds, surgical trauma, blunt trauma, and migration of a foreign body into the esophageal lumen (Table 138-6).
Gunshot and Stab Wounds of the Chest and Upper Abdomen
External penetrating injuries of the esophagus are seldom encountered. As reported by Conn and associates (1963), these injuries represent less than 1% of the intrathoracic injuries caused by penetrating trauma and, as noted by Sheely and colleagues (1975). Penetrating injuries of the thoracic esophagus may result from gunshot wounds in either a civilian or military setting, or by explosive fragments usually encountered in military operations. The central anatomic location of the esophagus; the protection afforded by the vertebral bodies, heart, and aorta; and the relatively small, compact size of the esophagus all diminish its susceptibility to injury.
The diagnosis of penetrating injuries of the thoracic esophagus is difficult. Most often, these injuries occur as part of a complex injury involving at the least the thoracic wall and lung; however, one or more of the following structures may be involved: the heart, great vessels, diaphragm, or one or more intraabdominal structures.
Table 138-6. Injuries of the Esophagus by External Causes | |
|---|---|
|
Although a variety of symptoms and findings are present in penetrating injuries of the esophagus, most of these may just as easily be attributed to concomitant involvement of other thoracic structures. Dysphagia strongly suggests the possibility of esophageal injury, but it often is not present. Pneumomediastinum is frequently present when the esophagus is injured, but it is caused more commonly by tracheal or bronchial injury.
The path of bullets through the body frequently may be predicted fairly accurately by observing the wound entrance and exit or by noting the wound entrance and determining the location of the bullet within the body by radiographic studies. The path of the missile may be erratic, however, so that any combination of injuries must be considered in each individual patient. The anatomic structures involved may be influenced by the position of the victim at the time of injury, and as noted, the path of the missile may be somewhat zigzag or the missile may have ricocheted off bony structures.
Careful review of the radiographs of the chest frequently reveals small fragments of the bullet outlining the tract of injury. When these observations indicate the possibility of esophageal injury, exploration should be considered. An esophagogram may be attempted to demonstrate the location of the injury, but this study may appear normal and should not cloud the clinical impression, especially when done with Gastrografin; a water suspension of barium sulfate is a better contrast material in these situations. Esophagoscopy is not particularly helpful. Although these examinations may further reinforce the clinical suspicion of esophageal injury, it is still easy to miss the perforation despite such diligent, time consuming examinations.
The treatment of penetrating injuries is based on early recognition, primary repair, and adequate drainage, as noted in the management of intraluminal injuries. The associated injuries to other vital structures in the thorax lead to early exploration in many of these patients. The esophageal injury usually is discovered by systematic exploration of the path of the missile. The repair of the esophagus is part of a complicated surgical procedure requiring attention to other injured structures. Most of the esophageal injuries are through-and-through perforations, in which there should always be an even number of rents in the esophagus. Occasionally, a lateral tear occurs, resulting in one large laceration. As noted by Symbas and colleagues (1972), extensive d bridement may be necessary because of diffuse hemorrhage, acute coagulation necrosis, or acute inflammation in the adjacent wall of the esophagus.
Pass and colleagues (1987) recommend that when early diagnosis and exploration occur, the esophageal defect should be repaired primarily. At times, only a minimum of d bridement is necessary, and the edges of the wound may be approximated by interrupted sutures of nonabsorbable material placed into the full thickness of the esophageal wall. This closure may be reinforced with a second muscular
P.2117
layer suture line. The adjacent mediastinal pleura is widely incised to ensure adequate drainage into the pleural space, which in turn is drained in the appropriate manner.
When operative exploration has been delayed because injury of the esophagus was unsuspected or when the esophageal wall has been extensively damaged, serious complications frequently ensue. Primary repair is still desirable whenever possible, although other options may be necessary. With late repairs, fistulization may be encountered more frequently. To achieve a satisfactory outcome, the surgeon's ingenuity and resourcefulness are severely tested. Richardson and Tobin (1994) and Richardson and colleagues (1996) have suggested that when significant esophageal tissue has been destroyed and primary closure is impossible without a stricture of the lumen, a muscle flap should be used to close the defect or defects. The latissimus dorsi is the muscle of choice. When this is not available, these authors have used a flap from the rhomboid muscle brought into the chest by excising a portion of the fourth rib. With distal wounds, a diaphragmatic flap has been used successfully on a number of occasions. A posteriorly based diaphragmatic flap is elevated, and double armed sutures are placed inside the defect and sutured directly onto the diaphragmatic flap to close the esophageal defect. The diaphragm is then closed in two layers. After closure of the esophageal wounds, the area is widely opened, making sure the area of the repaired esophagus is well drained. This muscle flap technique of esophageal closure can be used with early as well as with late identified injuries.
The morbidity and mortality rates of thoracic injuries are increased by the presence of an esophageal injury. However, it is difficult, if not impossible, to quantitate the exact role of the esophageal injury. In the review by Jones and Ginsberg (1992), the mortality rate attributed to traumatic esophageal injury was recorded to be only 9%.
Thermal Injuries
Thermal injury to the esophagus with subsequent perforation was first reported by Mohr and colleagues (2001), but the manuscript was not published until 2002. The thermal injury occurred during intraoperative radiofrequency ablation of atrial fibrillation (IRAAF). The perforation occurred late in the postoperative period (postoperative day 10) and created a fistula between the esophagus and left atrium with the occurrence of air embolisms to the CNS; surgical intervention was successful. Gillinov and associates (2001) presented a similar esophageal perforation and erosion into the left atrium after an IRAAF but with a fatal outcome. Subsequently, Doll and co workers (2003) described four similar perforations [including the original patient described by Mohr and colleagues (2001)] in 387 IRAAF procedures done by their group (an incidence of 1%). One patient died of massive air embolism; the other patients survived after multiple surgical procedures. Doll and co-workers (2003) had information (personal communications) of the occurrence of four similar events after IRAAF that occurred in Germany; however, there was only one survivor. The salient features are the development of neurologic symptoms late in the postoperative period (10 to 12 days) after IRAAF. Successful treatment is prompt diagnosis followed by closure of the atrial defect and esophagectomy. Gastrointestinal continuity may be restored either at the time of the esophagectomy or at a later date. Patwardhan and associates (2002) recommend bipolar radiofrequency coagulation rather than unipolar to reduce the risk for this complication.
Operative Trauma
Exclusive of direct esophageal procedures, operative injury to the esophagus occurs infrequently. Laceration of the cervical esophagus during thyroidectomy or laryngectomy has been reported. The thoracic esophagus may be injured during mediastinoscopy, vagotomy, hiatal hernia repair, a gastroesophageal antireflux procedure, or the removal of an adherent mediastinal tumor or cyst in the visceral compartment. Esophageal injury also may occur during a pneumonectomy, most often for inflammatory disease, as Takaro and colleagues (1960) noted; Shama and Odell (1985) reported an incidence of 0.5% in a series of 869 pneumonectomies for inflammatory disease. However, it also may occur during a resection for carcinoma of the lung, as Benjamin (1969) and Massard (1994) and their colleagues have reported.
When injury to the esophagus is recognized during the aforementioned procedures, direct primary repair is almost always successful. When the injury is unrecognized, all the complications common to any esophageal rupture may occur. Accordingly, the treatment must be directed to alleviate the resulting problems on an individual basis.
Blunt Trauma
Blunt trauma to the esophagus is infrequent but may be sustained during deceleration injuries to the chest, such as hitting the steering wheel during high speed collisions. The esophageal wall may be ruptured simultaneously with the adjacent membranous tracheal wall, or its blood supply may be compromised so that necrosis and subsequent perforation into the trachea occur. This injury may occur when the esophagus and trachea are compressed between the sternum and thoracic vertebral bodies. Hughes and Fox (1954) estimated that about one third of acquired benign tracheoesophageal fistulas have this mechanism of development; the remainder are caused by an inflammatory process. Infrequently, an acute rupture results from a rapid increase in intraluminal pressure. Tomaszek and Rosner (1984) and Pollock and associates (1981) mentioned esophageal rupture occurring with fractures of the cervical spine. Rarely, a necrotizing injury of the esophageal wall occurs if the esophagus is torn away from its blood supply by severe blunt injury. Necrosis of a greater or lesser length of the esophagus may result from such an injury.
P.2118
In the compressive injury of the esophagus and trachea, despite the usual presence of subcutaneous air, the frequent association of multiple system trauma delays the recognition of the esophagotracheal injury. The presence of the fistula is evidenced, usually sometime after the third day, by spasms of coughing on eating or drinking. Aspiration of oropharyngeal secretions into the lungs occurs, with the development of pulmonary infection. Once the fistula is suspected, the diagnosis should be confirmed by endoscopic and radiographic studies. When the condition of the patient is stabilized, direct repair of the fistula is indicated. Chapman and Braun (1970) and Beal and associates (1988) recommended that the repair be done as soon as possible, preferably through a right thoracotomy. Division of the azygos vein is done to permit wide exposure of the esophagus and trachea. Dissection, isolation, and division of the fistula is carried out without compromising the lumen of either structure. The fistulous openings of each organ are closed with interrupted, fine, absorbable sutures, or with fine sutures of Vicryl. A flap of adjacent tissue, usually pleura or, when this is unavailable, a vascularized pedicled flap of intercostal muscle, should be interposed between the two closures. A tracheostomy, if not already present, has been suggested to protect the tracheal suture line.
When a free perforation occurs from blunt trauma, the injury should be managed as described in the discussion of intraluminal perforation.
When extensive necrosis occurs with dissolution of the esophagus, emergency esophagectomy or surgical exclusion of the esophagus from the gastrointestinal tract with appropriate mediastinal and pleural drainage may be lifesaving for the patient. Subsequent reconstitution of gastrointestinal tract continuity may be accomplished later by a gastric or a colon interposition.
Migration of a Foreign Body into the Esophagus
A rare cause of esophageal injury is the occurrence of migration and erosion of an Angelchik prosthesis into the esophageal lumen, requiring removal and repair of the esophageal injury, as reported by Albin and associates (1985). This prosthesis, introduced by Angelchik and Cohen in 1979, had a period of brief popularity in the treatment of gastroesophageal reflux disease (GERD) but is no longer used because of its many complications, as noted by Beauchamp and associates (2001). Lucite plombage spheres also have eroded into the esophagus, as reported by Horowitz and colleagues (1992).
THE ABDOMINAL ESOPHAGUS
Abdominal Esophageal Perforations
Instrumental injury to the abdominal portion of the esophagus is infrequent, as Walsh (1979) noted. With the injury in this location, contamination of the peritoneal space may occur, with the development of signs and symptoms of an acute abdominal catastrophe. One must remember, however, that perforation of the distal thoracic esophagus may mimic such an event. Radiographic examination of the chest and abdomen, as well as of the esophagus, with contrast material should resolve the actual site of the perforation. At times, the injury may be confined to the retroperitoneal space, and a more indolent course may be observed. The aforementioned therapeutic goals enumerated for the management of injuries located in the thoracic esophagus pertain in the abdominal area as well. The surgical approach for drainage and possible repair in this situation is transabdominal rather than transthoracic.
Surgical Injuries
The abdominal portion of the esophagus is subject to the many injuries discussed in the section on injury to the thoracic portion of the esophagus. In particular, it is in danger of operative injury during both open and thoracoscopic antireflux procedures. In their report of esophageal injury in children, Engum and colleagues (1996) reported that 7 of the 25 injuries occurred in the abdominal portion (5 during antireflux procedures and 2 as a result of endoscopic injuries). The only death in the 25 children occurred in one of the patients in the abdominal group (a mortality rate of 14.2%), whereas no deaths occurred in the thoracic group. In adults, the actual incidence of injury is impossible to document. Nonetheless, both endoluminal and surgical injuries do occur, the latter especially during the performance of an antireflux procedure, either with an open or laparoscopic repair, so that vigilance to detect such injuries is mandatory during these procedures, as discussed by Krasna in Chapter 143 of this text.
DRUG INDUCED ESOPHAGEAL INJURIES
In recent years, an increasing number of esophageal injuries attributed to retention of medication in the esophagus have been reported. Factors such as preexisting esophageal pathologic conditions, ingestion of tablets or capsules unaccompanied by water, or recumbence at the time of or shortly after the ingestion of medication may predispose to retention of medication within the esophagus.
Many medications are inert in respect to their effect on the esophageal mucosa, but a large number of medications, if retained in contact with the esophageal mucosa, may result in mild esophagitis or actual ulceration and, in rare instance, perforation of the esophagus. Although the list of drugs reported to have caused injury is large, Oakes and Sherck (1987) stress that the most commonly reported drugs associated with these injuries are tetracycline, emperonium, and potassium chloride.
Diagnosis is usually established by esophagoscopy or barium swallow in a patient with recent onset of retrosternal
P.2119
pain, odynophagia, or dysphagia. Because of the frequently superficial and subtle changes in many of these injuries, esophagoscopy appears to be the preferred diagnostic approach.
When recognized early, treatment consists of withdrawal of the medication by the oral route, substitution of a liquid form if available, and supportive treatment. In most instances, this plan allows for complete healing of the esophagus. As with any injury of the esophagus, patients must be observed for stricture formation. If this occurs, esophageal dilations provide adequate treatment. In those unusual circumstances in which perforation occurs, appropriate surgical intervention is required.
CHEMICAL TRAUMA
Chemical burns of the esophagus result from the ingestion of caustic substances, either a strong acid or alkali. The latter is the more common offender because it is found in many household cleaning agents, usually as sodium or potassium hydroxide. Most of the injuries occur from accidental ingestion by young children, usually under 5 years of age. Occasionally, adults may ingest one of these agents, usually in an attempted suicide. Pathologically, the site of caustic injury of the esophagus may be located almost equally in any one of its anatomic subdivisions or may be widespread throughout. Normally, the greater period of contact is in the lower esophagus; hence, more extensive injury usually occurs in this area.
The burn injury to the esophagus has been divided into three phases. Inflammation, edema, and necrosis occur during the initial few days after injury. Sloughing of esophageal tissue with mucosal ulceration and the appearance of soft, red, moist granulation tissue occur in the second phase. The esophageal wall is weakest during this period, which may last 3 to 4 weeks. In the third phase, cicatrization and stricture formation may progress for many weeks as the destroyed esophageal submucosa and muscularis are replaced with scar tissue.
The signs and symptoms of the initial injury are related to the strength and amount of the substance swallowed. Increased salivation and dysphagia are common. Burns of the mouth and pharynx are easily observed. Respiratory distress occurs when the burn extends into the epiglottis or larynx. In severe instances, shock and life threatening respiratory distress may occur within an hour of injury. When the offending agent happens to be in a very dilute solution, symptoms may be minimal, and the individual may have no evidence of oral burns.
The injury to the esophagus occurs almost simultaneously with the contact of the caustic agent with the esophageal mucosa. Treatment, therefore, is designed to minimize the extent of scarring and subsequent stricture formation. Early esophagoscopy, within the first 2 days, is recommended. The esophagoscope is passed until the first area of burn is observed. No attempt is made to pass beyond this area if severe damage is found. However, if only mild erythema is noted, completion of upper endoscopy should be performed. Webb and associates (1970) outlined useful criteria for making an estimate of the severity of the burn, and these criteria may be correlated roughly with the incidence of subsequent stricture formation.
Further examination of the esophagus usually is not necessary for 3 to 4 weeks. At this time, the indicated examination is a radiographic evaluation of the esophagus by a barium swallow. The number and length of any strictures usually are well delineated by this study. Interval radiographic studies then may be used to evaluate the progression of healing or stricture formation and the effects of treatment.
Borja (1969) and Haller (1971) and their associates suggested a 3 to 6 week course of corticosteroids and antibiotics in the acute phase of the injury in an attempt to diminish the frequency and the degree of stricture formation. Additional reports by Moore (1986), as well as by Moazam (1987) and Rubin (1989) and their colleagues supported the use of steroids; however, Ferguson and co workers (1989) concluded from their observations that the use of steroids had no beneficial effect. Subsequently, controlled studies (i.e., steroids versus no steroids) by Anderson and associates (1990) as well as by Ulman and Mutaf (1998) showed no significant differences in healing rates and stricture formation in either group. These authors concluded that the use of steroids in acute caustic injuries of the esophagus was not indicated. Despite these reports, Lang and Behrens (1993) and Howell and colleagues (1992) continued to support the use of steroids in patients with severe grade II and grade III burns of the esophagus, although they did concur that steroid use was unnecessary and not indicated in grade I or mild grade II injuries. Boukthir and coinvestigators (2004) have reported that high doses of steroids seem to improve the prognosis of severe grade II injuries and may prevent stricture formation. The role of steroids in the management of grade III, deep, and/or circumferential burns, remains unsettled. From the data in most reports, regardless of the use of steroids, strictures of varying degrees occur in almost all patients with such severe injuries. Subsequent bougienage is recommended in those patients with a definite stricture of the esophagus. This procedure is started during the early phase of cicatrix formation, which begins the third or fourth week after injury. Danger of instrumental perforation, however, is present, and a string or wire guide is essential to avoid this risk. Bougienage is repeated at varying intervals dictated in part by the initial severity of the burn and serial radiographic studies. The most important factor, however, is the ability of the patient to swallow and to maintain nutrition. Recurrent or increasing dysphagia indicates the need for additional mechanical dilation.
Surgical intervention is seldom necessary in the acute phase of caustic injuries to the esophagus unless there is extension of the caustic injury through the entire wall of the esophagus. It is sometimes difficult to determine whether this has occurred, and in such a situation, a CT scan of the esophagus often permits this distinction. A 24 hour observation during which corticosteroids and antibiotics are administered is reasonable in unclear circumstances.
Table 138-7. Endoscopic Grading of Corrosive Burns of Esophagus and Stomacha | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||
P.2120
Gago and colleagues (1972), however, reported emergency resection of a necrotic esophagus within hours of severe caustic injury in an adult and a delayed resection in an infant. Burrington and Raffensperger (1978) reported several acute tracheoesophageal fistulas resulting from caustic ingestion in small children. In these unusually severe injuries, tracheostomy, cervical esophagostomy, and gastrostomy with isolation (blind ending) of the thoracic esophagus are required. Several months later, it is then necessary to create a substitute esophagus using a reversed gastric tube or colon interposition.
Estrera and associates (1986) recommended an aggressive approach for second or third degree burns of the esophagus (Table 138-7) rather than the aforementioned standard treatment of antibiotics, and subsequent dilation as required. In patients with second degree burns, they suggest intraesophageal stenting through a celiotomy approach, and the stent is left in place for at least 21 days. Antibiotics and steroids also are given. In patients with third degree burns without full thickness involvement, a stent is likewise used, but in those with extensive full thickness necrosis, urgent radical total esophagogastrectomy is carried out along with a temporary cervical esophagostomy and jejunostomy. Reconstruction is performed at a later date. In such patients with third degree burns treated with a delayed resection, the mortality rate was 100%; with the aggressive urgent approach, the mortality rate was 0% in their report. When an esophagectomy is indicated, Hwang and colleagues (1987) suggested that it is best done by the transhiatal route. Engum and associates (1996) reported one esophagectomy for an acute lye injury in their series of 24 esophageal injuries in children.
Surgical intervention may also be considered in long-standing lye strictures when it becomes difficult or impossible to maintain an adequate lumen despite repeated dilation. In these situations, it becomes necessary to create a substitute esophagus using either the stomach, a reversed gastric tube, or a colon interposition, the latter described in Chapter 127. An increased risk for the development of carcinoma in residual, strictured esophagus has been suggested and the remaining esophagus should be excised.
REFERENCES
Abbott OA, et al: Atraumatic so-called spontaneous' rupture of the esophagus. A review of 47 personal cases with comments on a new method of surgical therapy. J Thorac Cardiovasc Surg 59:67, 1970.
Adkins MS, Raccuia JS, Acinapura AJ: Esophageal perforation in a patient with acquired immunodeficiency syndrome. Ann Thorac Surg 50:299, 1990.
Albin J, et al: Intrathoracic esophageal perforation with the Angelchik antireflux prosthesis: report of a new complication. Gastrointest Radiol 10:330, 1985.
Altorjay A, et al: The role of esophagectomy in the management of esophageal perforations. Ann Thorac Surg 65:1433, 1998.
Anderson KD, Rouse TM, Randolph JG: A controlled trial of corticosteroids in children with corrosive injury of the esophagus. N Engl J Med 323:637, 1990.
Angelchik JP, Cohen R: A new surgical procedure for the treatment of gastroesophageal reflux and hiatal hernia. Surg Gynecol Obstet 148:246, 1979.
Barber GB, et al: Esophageal foreign body perforation: report of an unusual case and review of the literature. Am J Gastroenterol 79:509, 1984.
Bardaxoglou E, et al: Oesophageal perforation: primary suture repair reinforced with absorbable mesh and fibrin glue. Br J Surg 81:399, 1994.
Bardini R, et al: Temporary double exclusion of the perforated esophagus using absorbable staples. Ann Thorac Surg 54:1165, 1992.
Barrett NR: Report of a case of spontaneous perforation of the oesophagus successfully treated by operation. Br J Surg 35:216, 1947.
Beal SL, Pottmeyer EW, Spisso JM: Esophageal perforation following external blunt trauma. J Trauma 28:1425, 1988.
Beauchamp G, Ouellette D, Dupere S: Open antireflux repairs for simple reflux esophagitis: short- and long-term results. Chest Surg Clin N Am 11:555, 2001.
Bell RCW: Laparoscopic closure of esophageal perforation following pneumatic dilatation for achalasia: report of two cases. Surg Endosc 11: 476, 1997.
Benjamin I, Olsen AM, Ellis FH Jr: Esophagopleural fistula. A rare postpneumonectomy complication. Ann Thorac Surg 7:139, 1969.
Bennett JR, Hendrix TR: Treatment of achalasia with pneumatic dilatation. Mod Treat 7:1217, 1970.
Berliner L, Redmond P, Pachter HL: Spontaneous intramural perforation of the esophagus: case report and review of the literature. Am J Gastroenterol 77:355, 1982.
Berry BE, Ochsner JL: Perforation of the esophagus. A 30 year review. J Thorac Cardiovasc Surg 65:1, 1973.
Bladergroen MR, Lowe JE, Postlethwait RW: Diagnosis and recommended management of esophageal perforation and rupture. Ann Thorac Surg 42:235, 1986.
Borja AR, et al: Lye injuries of esophagus. Analysis of ninety cases of lye ingestion. J Thorac Cardiovasc Surg 57:533, 1969.
Borrie J, Sheat J: Spontaneous intramural oesophageal perforation. Thorax 25:294, 1970.
P.2121
Boukthir S, et al: High doses of steroids in the management of caustic esophageal burns in children [article in French]. Arch Pediatr 11:13, 2004.
Boulis NM, et al: Epidural abscess: a delayed complication of esophageal stenting for benign stricture. Ann Thorac Surg 68:568, 1999.
Brinkman WT, et al: Transesophageal echocardiography: not an innocuous procedure. Ann Thorac Surg 72:1725, 2001.
Bufkin BL, Miller JI Jr, Mansour KA: Esophageal perforation: emphasis on management. Ann Thorac Surg 61:1447, 1996.
Burrington JD, Raffensperger JG: Surgical management of tracheoesophageal fistula complicating caustic ingestion. Surgery 84:329, 1978.
Cameron JL, et al: Selective nonoperative management of contained intrathoracic esophageal disruptions. Ann Thorac Surg 27:404, 1979.
Chang C-H, et al: One-stage operation for treatment of delayed diagnosis of esophageal perforation. Ann Thorac Surg 53:617, 1992.
Chapman ND, Braun RA: The management of traumatic tracheo-esophageal fistula caused by blunt chest trauma. Arch Surg 100:681, 1970.
Chung DA, Ritchie AJ: Videothoracoscopic drainage of mediastinal abscess: an alternative to thoracotomy. Ann Thorac Surg 69:1573, 2000.
Conlan AA, et al: Pharyngoesophageal barotrauma in children: a report of six cases. J Thorac Cardiovasc Surg 88:452, 1984.
Conn JH, et al: Thoracic trauma: analysis of 1022 cases. J Trauma 3:22, 1963.
Cornwell J, Walden C, Ghahremani GG: CT demonstration of fistula between esophageal carcinoma and spinal canal. J Comput Assist Tomogr 10:871, 1986.
Daniel WG, et al: Safety of transesophageal echocardiography. A multicenter survey of 10,419 examinations. Circulation 83:817, 1991.
Debers VI, Mitchell RE Jr: Herman Boerhaave's attocis, nec descripti pruis morbi histora. Bull Med Libr Assoc 43:217, 1955.
DeMeester TR: Perforation of the esophagus [Editorial]. Ann Thorac Surg 42:231, 1986.
Doll N, et al: Esophageal perforation during left atrial radiofrequency ablation: is the risk too high? J Thorac Cardiovasc Surg 125:836, 2003.
Edling JE, Bacon BR: Pleuropulmonary complications of endoscopic variceal sclerotherapy. Chest 99:1252, 1991.
Engum SA, et al: Improved survival in children with esophageal perforation. Arch Surg 131:604, 1996.
Estrera A, et al: Corrosive burns of the esophagus and stomach: a recommendation for an aggressive surgical approach. Ann Thorac Surg 41: 276, 1986.
Feliciano DV, et al: Combined tracheoesophageal injuries. Am J Surg 150:710, 1985.
Ferguson MK: Achalasia: current evaluation and therapy. Ann Thorac Surg 52:336, 1991.
Ferguson MK, et al: Early evaluation and therapy for caustic esophageal injury. Am J Surg 157:116, 1989.
Frink NW: Spontaneous rupture of the esophagus report of a case with recovery. J Thorac Surg 16:291, 1947.
Gago O, et al: Aggressive surgical treatment for caustic injury of the esophagus and stomach. Ann Thorac Surg 13:243, 1972.
Gaissert HA, et al: Surgical management of necrotizing Candida esophagitis. Ann Thorac Surg 67:231, 1999.
Gaissert HA, et al: Infectious necrotizing esophagitis: outcome after medical and surgical intervention. Ann Thorac Surg 75:342, 2003.
Gelfand ET, Fisk RL, Callaghan JC: Accidental pneumatic rupture of the esophagus. J Thorac Cardiovasc Surg 74:142, 1977.
Gillinov AM, Petersson G, Rice TW: Esophageal injury during radiofrequency ablation for atrial fibrillation. J Thorac Cardiovasc Surg 123: 1239, 2001.
Ghandour Z, Al Karawi MA, Muhamed AE: Spontaneous esophageal perforation: unusual presentation of tuberculosis. Endoscopy 29:143, 1997.
Graeber GM, et al: A comparison of patients with endoscopic esophageal perforation and patients with Boerhaave's syndrome. Chest 92:995, 1987.
Grubbs BC, et al: Distal esophageal perforation caused by tuberculosis. J Thorac Cardiovasc Surg 121:1003, 2001.
Guillem PG, et al: Perforation of Barrett's ulcer: a challenge in esophageal surgery. Ann Thorac Surg 69:1707, 2000.
Hagen JA, DeMeester TR: Anatomy of the esophagus. In Shields TW, LoCicero J III, Ponn RB (eds): General Thoracic Surgery. 5th Ed. Philadelphia: Lippincott Williams & Wilkins, 2000, p. 1595.
Haller JA Jr, et al: Pathophysiology and management of acute corrosive burns of the esophagus: results of treatment in 285 children. J Pediatr Surg 6:578, 1971.
Han SY, et al: Perforation of the esophagus: correlation of site and cause with plain film findings. AJR Am J Roentgenol 145:537, 1985.
Hardy JD, Wallace WH: Spontaneous rupture of the esophagus. In Hardy JD (ed): Rhoads Textbook of Surgery, Principles and Practice. Philadelphia: JB Lippincott, 1977.
Horowitz MD, et al: Late complications of plombage. Ann Thorac Surg 53:803, 1992.
Howell JM, et al: Steroids for the treatment of corrosive esophageal injury: a statistical analysis of past studies. Am J Emerg Med 10:421, 1992.
Hughes FA, Fox JR: Acquired non-malignant esophagotracheobronchial fistula. J Thorac Surg 27:384, 1954.
Hwang TL, Shen-Chen SM, Chen MF: Nonthoracotomy esophagectomy for corrosive esophagitis with gastric perforation. Surg Gynecol Obstet 164:537, 1987.
Iannettoni MD, et al: Functional outcome after surgical treatment of esophageal perforation. Ann Thorac Surg 64:1606, 1997.
Ikeda Y, et al: Thoracoscopic repair of a spontaneous rupture of the esophagus with the endoscopic suturing device. J Thorac Cardiovasc Surg 121:178, 2001.
Jones WG II, Ginsberg RJ: Esophageal perforation: a continuing challenge. Ann Thorac Surg 53:534, 1992.
Jurani CC, Early GL, Roberts SR: Spontaneous esophageal perforation presenting as meningitis. Ann Thorac Surg 73:1294, 2002.
Keighley MR, et al: Morbidity and mortality of oesophageal perforation. Thorax 27:353, 1972.
Kelley DL, Nengebauer MK, Fosburg RG: Spontaneous intramural esophageal perforation. J Thorac Cardiovasc Surg 63:504, 1972.
Kharasch ED, Sivarajan M, et al: Gastroesophageal perforation after intraoperative transesophageal echocardiography. Anesthesiology 85: 426, 1996.
Kim BW, et al: Esophagomediastinal fistula and esophageal stricture as a complication of esophageal candidiasis: a case report. Gastrointest Endosc 52:772, 2000.
Kotler R, Schild JA, Holinger PH: Delayed C.N.S. complications. Laryngoscope 85:1379, 1975.
Laisaar T: Video-assisted thoracoscopic surgery in the management of acute purulent mediastinitis and pleural empyema. Thorac Cardiovasc Surg 46:51, 1998.
Landen S, El Nakadi I: Minimally-invasive approach to Boerhaave's syndrome: a pilot study of three cases. Surg Endosc 16:1354, 2002.
Lang T, Behrens R: Caustic burns of the upper gastrointestinal tract. Diagnosis and therapy. [Article in German]. Monatsschr Kinderheilkd 141: 434, 1993.
Lawrence DR, et al: Primary esophageal repair for Boerhaave's syndrome. Ann Thorac Surg 67:818, 1999.
Letquart JP, et al: Rupture barotraumatique de l'esophage. J Chir (Paris) 118:483, 1981.
Light RW: Physiology of pleural fluid production and benign pleural effusion. In Shields TW, LoCicero J III, Ponn RB (eds): General Thoracic Surgery. 5th Ed. Philadelphia: Lippincott Williams & Wilkins, 2000, p. 687.
Love L, Berkow AE: Trauma to the esophagus. Gastrointest Radiol 2:305, 1978.
Mallory GK, Weiss S: Hemorrhage from lacerations of the cardiac orifice of the stomach due to vomiting. Am J Med Sci 178:506, 1929.
Massard G, et al: Esophagopleural fistula: an early and long-term complication after pneumonectomy. Ann Thorac Surg 58:1437, 1994.
Massey SR, et al: Oesophageal perforation following perioperative transoesophageal echocardiography. Br J Anaesth 84:643, 2000.
Mayer Jr JE, Murray CA, Varco RL: The treatment of esophageal perforation with delayed recognition and continuing sepsis. Ann Thorac Surg 23:586, 1977.
Meyers MA, Ghahremani GG: Complications of fiberoptic endoscopy. I. Esophagoscopy and gastroscopy. Radiology 115:293, 1975.
Moazam F, et al: Caustic ingestion and its sequelae in children. South Med J 80:187, 1987.
Moghissi K, Pender D: Instrumental perforations of the oesophagus and their management. Thorax 43:642, 1988.
Mohr FW, et al: Curative treatment of atrial fibrillation: acute and midterm results of intraoperative radiofrequency ablation of atrial fibrillation in 150 patients. Presented at the 81st Annual Meeting of the American Association for Thoracic Surgery, May 7, 2001.
Mohr FW, et al: Curative treatment of atrial fibrillation with intraoperative radiofrequency ablation: short-term and midterm results. J Thorac Cardiovasc Surg 123:919, 2002.
Monies-Chass I, Joachims HZ, Altman MM: Hyperbaric oxygen in anaerobic infection of the mediastinum. J Laryngol Otol 89:1147, 1975.
P.2122
Naclerio EA: The V-sign' in the diagnosis of spontaneous rupture of the esophagus. An early roentgen clue. Am J Surg 93:291, 1957.
Nana AM, et al: Esophageal perforation by echoprobe during cardiac surgery: treatment by endoscopic stenting. Ann Thorac Surg 75:1955, 2003.
Nathanson LK, et al: Videothoracoscopic primary repair of early distal oesophageal perforation. Aust N Z J Surg 63:399, 1993.
Nesbitt JC, Walsh GL: Surgical management of esophageal perforation. In Franco KL, Putnam JB Jr (eds): Advanced Therapy in Thoracic Surgery. Hamilton, Ontario: BC Decker Inc, 1998, p. 500.
Ng T, Maziak DE, Shamji FM: Esophageal perforation: a rare complication of Zollinger-Ellison syndrome. Ann Thorac Surg 72:592, 2001.
Nguyen NT, et al: Thoracoscopic management of postoperative esophageal leak. J Thorac Cardiovasc Surg 121:391, 2001.
Oakes DE Sherck JP: Drug-induced esophageal injuries. In DeMeester TR, Matthews HR (eds): Benign Esophageal Disease. St. Louis: CV Mosby, 1987, p. 269.
Ohri SK, et al: Primary repair of iatrogenic thoracic esophageal perforation and Boerhaave's syndrome. Ann Thorac Surg 55:603, 1993.
Olsen AM, Clagett OT: Spontaneous rupture of the esophagus. Postgrad Med 2:417, 1947.
O'Neill JE, Griffin JP, Cottrell JE: Pharyngeal and esophageal perforation following endotracheal intubation. Anesthesiology 60:487, 1984.
Orringer MB, Marshall B, Stirling MC: Transhiatal esophagectomy for benign and malignant disease. J Thorac Cardiovasc Surg 105:265, 1993.
Orringer MB, Stirling MC: Esophagectomy for esophageal disruption. Ann Thorac Surg 49:35, 1990.
Paramesh V, Rumisek JD, Chang FC: Spontaneous recanalization of the esophagus after exclusion using nonabsorbable staples. Ann Thorac Surg 59:1214, 1995.
Pass LJ, et al: Management of esophageal gunshot wounds. Ann Thorac Surg 44:253, 1987.
Patwardhan AM, Lad VS, Pai V: Esophageal injury during radiofrequency ablation for atrial fibrillation: inherent safety of radiofrequency bipolar coagulation. J Thorac Cardiovasc Surg 124:642, 2002.
Pollock RA, et al: Esophageal and hypopharyngeal injuries in patients with cervical spine trauma. Ann Otol Rhinol Laryngol 90:323, 1981.
Port JL, et al: Thoracic esophageal perforations: a decade of experience. Ann Thorac Surg 75:1071, 2003.
Reeder LB, DeFilippi VJ, Ferguson MK: Current results of therapy for esophageal perforation. Am J Surg 169:615, 1995.
Richardson JD, Tobin GR: Closure of esophageal defects with muscle flaps. Arch Surg 129:541, 1994.
Richardson JD, et al: Complex and challenging problems in trauma. Complex thoracic injuries. Surg Clin North Am 76:725, 1996.
Ring D, et al: Vertebral osteomyelitis after blunt traumatic esophageal rupture. Spine 20:98, 1995.
Roberts JR, et al: Thoracoscopic management of descending necrotizing mediastinitis. Chest 112:850, 1997.
Rubin MM, Jui V, Cozzi GM: Treatment of caustic ingestion. J Oral Maxillofac Surg 47:286, 1989.
Saabye J, Nielsen HO, Andersen K: Long-term observation following perforation and rupture of the esophagus. Scand J Thorac Cardiovasc Surg 22:79, 1988.
Salo JA, et al: Management of delayed esophageal perforation with mediastinal sepsis. Esophagectomy or primary repair? J Thorac Cardiovasc Surg 106:1088, 1993.
Salo JA, et al: Successful treatment of mediastinal gas gangrene due to esophageal perforation. Ann Thorac Surg 70:2143, 2000.
Sandrasagra FA, English TA, Milstein BB: Esophageal intubation in the management of perforated esophagus with stricture. Ann Thorac Surg 25:399, 1978.
Santos GH, Frater RW: Transesophageal irrigation for the treatment of mediastinitis produced by esophageal rupture. J Thorac Cardiovasc Surg 91:57, 1986.
Sawyer R, Phillips C, Vakil N: Short- and long-term outcome of esophageal perforation. Gastrointest Endosc 41:130, 1995.
Schroder J, et al: Sepsis syndrome induced by tuberculous perforation of the esophagus. Infection 24:162, 1996.
Scott HJ, Rosin RD: Thoracoscopic repair of a transmural rupture of the oesophagus. J R Soc Med 88:414P, 1995.
Sehhat S, et al: Oesophageal moniliasis causing fistula formation and lung abscess. Thorax 31:361, 1976.
Shama DM, Odell JA: Esophagopleural fistula after pneumonectomy for inflammatory disease. J Thorac Cardiovasc Surg 89:77, 1985.
Sheely CH II, et al: Penetrating wounds of the cervical esophagus. Am J Surg 130:707, 1975.
Shemesh E, Bat L: Esophageal perforation after fiberoptic endoscopic injection sclerotherapy for esophageal varices. Arch Surg 121:243, 1986.
Swanstrom LL, Pennings J: Laparoscopic esophagomyotomy for achalasia. Surg Endosc 9:286, 1995.
Symbas PN, et al: Penetrating wounds of the esophagus. Ann Thorac Surg 13:552, 1972.
Takaro T, Walkup HE, Okano T: Esophagopleural fistula as a complication of thoracic surgery: a collective review. J Thorac Cardiovasc Surg 40: 179, 1960.
Tasdemir O: Beneficial effects of fibrin glue on esophageal perforation. Ann Thorac Surg 61:1589, 1996.
Tinoco RC, Tinoco AC, El-Kadre L: Perforated epiphrenic diverticulum treated by video laparoscopy. Surg Endosc 13:270, 1999.
Tomaszek DE, Rosner MJ: Occult esophageal perforation associated with cervical spine fracture. Neurosurgery 14:492, 1984.
Toupet A: Technique d'oesophago-gastroplastie avec phreno-gastropexie appliquee dans la cure radicale des hernies hiatales et comme complement de l'operation d Heller dans les cardiospasmes. Mem Acad Chir 89:394, 1963.
Triggiani E, Belsey R: Oesophageal trauma: incidence, diagnosis, and management. Thorax 32:241, 1977.
Ulman I, Mutaf O: A critique of systemic steroids in the management of caustic esophageal burns in children. Eur J Pediatr Surg 8:71, 1998.
Urbani M, Mathisen DJ: Repair of esophageal perforation after treatment for achalasia. Ann Thorac Surg 69:1609, 2000.
Urbanowicz JH, et al: Transesophageal echocardiography and its potential for esophageal damage. Anesthesiology 72:40, 1990.
Urschel HC Jr, et al: Improved management of esophageal perforation: exclusion and diversion in continuity. Ann Surg 179:587, 1974.
Vogel S, et al: Treatment of iatrogenic esophageal perforation. Zentrabl Chir 116:751, 1991.
Walsh P: Rupture of the abdominal oesophagus. Br J Surg 66:601, 1979.
Wang N, et al: Delayed primary repair of intrathoracic esophageal perforation: is it safe? J Thorac Cardiovasc Surg 111:114, 1996.
Webb WR, et al: An evaluation of steroids and antibiotics in caustic burns of the esophagus. Ann Thorac Surg 9:95, 1970.
Wesdorp IC, et al: Treatment of instrumental oesophageal perforation. Gut 25:398, 1984.
Whyte RI, Iannettoni MD, Orringer MB: Intrathoracic esophageal perforation: the merit of primary repair. J Thorac Cardiovasc Surg 109:140, 1995.
Williford ME, et al: Esophageal tuberculosis: findings on barium swallow and computed tomography. Gastrointest Radiol 8:119, 1983.
Wright CD, et al: Reinforced primary repair of thoracic esophageal perforation. Ann Thorac Surg 60:245, 1995.
Wychulis AR, Fontana RS, Payne WS: Instrumental perforations of the esophagus. Dis Chest 55:184, 1969.
Yap RG, et al: Traumatic esophageal injuries: 12-year experience at Henry Ford Hospital. J Trauma 24:623, 1984.
Zalunardo MP, et al: Late oesophageal perforation after intraoperative transoesophageal echocardiography. Br J Anaesth 88:595, 2002.
Reading References
Brewer L, et al: Options in the management of perforation of the esophagus. Am J Surg 152:62, 1986.
Cohn H, Hubbard A, Patton G: Management of esophageal injuries. Ann Thorac Surg 48:309, 1989.
Grillo HG, Wilkins EW Jr: Esophageal repair following late diagnosis of intrathoracic perforations. Ann Thorac Surg 29:387, 1975.
Michel L, Grillo H, Malt RA: Esophageal perforation. Ann Thorac Surg 33:203, 1982.
Richardson J, et al: Unifying concepts in treatment of esophageal leaks. Am J Surg 149:157, 1985.
Symbas PN, Hatcher CR, Vlasis SE: Esophageal gunshot injuries. Ann Surg 191:703, 1980.
1This section supplied by M.J. Schuchert and J.D. Luketich, University of Pittsburgh School of Medicine, Pittsburg, PA.
EAN: 2147483647
Pages: 203