III - Musculoskeletal System
Editors: Mills, Stacey E.
Title: Histology for Pathologists, 3rd Edition
Copyright 2007 Lippincott Williams & Wilkins
> Table of Contents > III - Musculoskeletal System > 6 - Myofibroblast
function show_scrollbar() {}
6
Myofibroblast
Walter Sch rch
Thomas A. Seemayer
Boris Hinz
Giulio Gabbiani
Discovery of the Myofibroblast
The myofibroblast was discovered in 1971 in electron micrographs from contracting (healing) experimental granulation tissue (1). Soon thereafter, its biochemical, pharmacologic, and immunohistochemical features were delineated (2,3,4,5). Since these early days, the list of pathologic conditions in which this cell has been identified has grown considerably (6,7,8). Looking back, it is somewhat surprising that such a pivotal element of diverse fundamental processes had not been defined earlier (9).
The road to discovery stems from interest in the process of wound healing as traced from the time of fossils to the ancient world (10). Indeed, the fate of civilizations rested on the ability of people to recover from wounds inflicted through battle or disease. Nearly a century ago, Carrel and Hartmann hypothesized that contractile forces were present in granulating wounds (11). For years it was believed, even taught, that collagen was the element essential for wound contraction. Dogma changed (slowly) with two reports in the mid-1950s. In one, experiments established that wound contraction was normal in guinea pigs rendered scorbutic (12). In the other, fibroblasts, under appropriate conditions, could be induced to contract in vitro (13). These findings cast doubt on the contractility of collagen and suggested that cells were central to tissue contraction.
In 1969, Majno and colleagues performed seminal experiments that established that histamine caused postcapillary venular interendothelial gaps that brought about vascular leakage (14). In electron micrographs, such endothelial cells were shrunken, distorted, and with notched nuclei. On this basis, they reasoned that gap formation might be produced by active endothelial contraction (14). This suggestion, made before the establishment of the concept that nonmuscle cells contain contractile proteins, was not easily accepted; in turn, it stimulated work based on the possibility that endothelial and other mesenchymal cells could exert contractile activities.
A few years later, the ultrastructural observation was made that the cytoplasm of granulation tissue fibroblasts was loaded with bundles and aggregates of microfilaments (1), a feature typical of smooth muscle cells. On this basis, the possibility that these modified fibroblastic cells were responsible for granulation tissue contraction was relatively
P.124
easy to suggest (1); however, acceptance took some time (15). Further experiments, employing pharmacologic agents known to effect cellular contraction/relaxation, established that granulating wounds indeed contained contractile cells, and the term myofibroblast was proposed (2). Subsequently, myofibroblasts were found to be capable of being decorated by human smooth muscle antibodies (4); these were then shown to be specifically directed against actin (5). Shortly thereafter, myofibroblasts were identified within nodules of Dupuytren's disease (16) and in human granulation tissue (17) and shown to transmit their contractile forces from cell to cell through intermediate (adherens) junctions and from cell to stroma by means of microtendons, the whole being synchronized by intercellular gap junctions (18). The microtendon, an apparatus connecting myofibroblasts to the surrounding extracellular matrix, was named fibronexus (19).
In the late 1970s, Tremblay (20) described the presence of myofibroblasts in the stroma of invasive mammary carcinomas. The neoplasms in which these myofibroblasts had been noted were firm and retracted, unassociated with an inflammatory infiltrate. Because myofibroblasts are normally not present in mammary stroma, it was suggested that they contributed to the retraction phenomena and desmoplasia, which characterized these neoplasms (20). It was then reasoned that such contractile cells might be contained in diverse carcinomas characterized by retraction and desmoplasia. Accordingly, a series of invasive and metastatic carcinomas was examined ultrastructurally. Myofibroblasts were present in the stroma of each tumor and were particularly numerous in those that were hard, sclerotic, and retracted (21). Within several years, the spatial distribution of such cells within invasive and metastatic carcinomas was described (22,23), and it was proposed that similarities between the process of wound healing and the stromal response to neoplastic invasion might exist (22).
In the following years detailed studies of intermediate filament proteins and actin isoforms of myofibroblasts in various settings and conditions were performed; this led to the finding that myofibroblasts from diverse pathologic settings were heterogeneous in their content of intermediate filaments and actin isoforms (24,25). The presence of -smooth muscle actin, the actin isoform characteristic of vascular smooth muscle cells, was suggested as the marker of the myofibroblastic phenotype (25). In these works, it was shown that there is a correlation between the phenotypic modulation of myofibroblasts and the clinical behavior of lesions containing these cells. In particular, it was shown that myofibroblasts in granulation tissue of normally healing wounds express -smooth muscle actin only temporarily (25), whereas myofibroblasts with a smooth muscle phenotype persist in hypertrophic scars, fibrocontractive diseases, quasi-neoplastic proliferative conditions, and within the stroma of certain neoplasms (24,26).
More recently, it was shown that transforming growth factor 1 (TGF- 1) is the most important stimulator of myofibroblastic differentiation (27,28), as well as of collagen production by this cell (29). For TGF- 1 to be active, the ED-A splice variant of cellular fibronectin must be present in the extracellular matrix (30). This provided the first hint of the mechanisms controlling the modulation of the myofibroblastic phenotype. It was also shown that myofibroblasts undergo apoptosis during the transition between granulation tissue and scar tissue (31). The possibility that myofibroblasts could arise from such specialized mesenchymal cells as hepatic perisinusoidal stellate cells (32), mesangial (33) and renal tubular cells (34), and mesothelial cells (35) was demonstrated. Quite unexpectedly, the hematogenous origin of myofibroblasts in several human and experimental pathologic settings was recently established (36,37).
In 2005, some 34 years after the initial discovery and four decades after the quest began, the myofibroblast is recognized as a central element in normal and abnormal wound healing, in diverse reactive proliferative conditions, and within the stroma of certain invasive and metastatic neoplasms.
The sections that follow more fully characterize the myofibroblast, describe the settings in which it is found, and relate recent studies that provide further insight into the biology of this unique cell.
Characterization of the Myofibroblast
Ultrastructural
As initially described in granulation tissue and nodules of Dupuytren's disease (1,16), myofibroblasts share morphologic features in common with fibroblasts and smooth muscle cells.
Fibroblasts of adult animals and humans display a slender fusiform and smooth, contoured nucleus, a well-developed Golgi area, numerous and often dilated cisternae of rough endoplasmic reticulum, scattered mitochondria, and small numbers of microfilaments, the latter sometimes arranged in discrete bundles beneath the plasma membrane. Cell contours are generally smooth or display a few short cytoplasmic extensions. Plasmalemmal attachment plaques, dense patches or dense bands (38,39), basal lamina, pinocytotic vesicles, intercellular junctions, and cell-to-stroma attachment sites are absent.
Smooth muscle cells are enveloped by a continuous basal lamina. Their plasma membrane is studded with plasmalemmal attachment plaques or so-called membrane-associated dense bodies, dense plaques, dense patches or dense bands (39), and numerous pinocytotic vesicles. Intercellular gap junctions and adherens junctions are present (40).
P.125
The cytoplasm is laden with bundles of microfilaments, usually disposed parallel to the long axis of the cell, among which numerous dense bodies are interspersed. The material of the dense bodies appears similar to the one forming the dense bands, which are attached to the cell membrane in certain vascular smooth muscle cells. Some dense bodies are in continuity with dense bands. Dense bodies and dense bands probably correspond to Z-lines of striated muscle fibers. In both structures, -actinin has been demonstrated by immunohistochemical techniques (41,42). Force transmission from the contractile apparatus to the cell membrane in smooth muscle cells occurs via the insertion of bundles of actin filaments into the dense bands (38). Transmission of the contractile force occurs also across cell membranes of smooth muscle cells and from cell membranes to the stroma. Although it seems clear that the traction generated by the myofilaments is transmitted to the dense bands, the exact mechanism of the transmission of the traction across the cell membrane is not fully understood. The fibronectin receptor as a transmembrane receptor glycoprotein complex (43,44,45,46) has extracellular binding sites for fibronectin (47), suggesting specific interactions between cytoplasmic actin filaments and extracellular fibronectin fibers across the plasma membrane at cell-to-matrix attachment sites. A close association between bundles of cytoplasmic actin filaments (stress fibers) and bundles of extracellular fibronectin fibrils has been observed in transformed fibroblasts and in myofibroblasts of granulation tissue in vivo; this structure, designated fibronexus, is specialized for enhanced cell-to-matrix connections (48,49). Contractile forces from cell to cell are transmitted through adherens or intermediate junctions, which are symmetrical structures formed by two complementary dense bands that match each other in adjacent smooth muscle cells (38). Their nuclei are elongated with blunt ends and are deformed by shallow invaginations. In contrast to fibroblasts, the Golgi area and the rough endoplasmic reticulum are poorly developed.
Myofibroblasts (Figure 6.1 A E) disclose irregular, often stellate, cellular outlines with numerous and long cytoplasmic extensions and are connected by intermediate or adherens junctions (Figure 6.1D) (50) and by gap junctions (Figure 6.1E), the latter considered as low-resistance pathways for intercellular communications (18). In addition, myofibroblasts are partly enveloped by a basal lamina and display plasmalemmal attachment plaques, dense patches or dense bands, and pinocytotic vesicles (Figure 6.1C). They are also connected by microtendons to the extracellular matrix by cell-to-stroma attachment sites through the fibronexus, a transmembrane complex of intracellular microfilament bundles in apparent continuity with extracellular fibronectin fibers (Figure 6.1B) (19,49). At the surface of myofibroblasts, three types of fibronexus are observed: (a) plaquelike; (b) tracklike; and (c) tandem associations (49). These cell-to-stroma attachment sites are well-developed and numerous in myofibroblasts compared with their attenuated appearance in smooth muscle cells. Myofibroblasts contain numerous bundles of cytoplasmic microfilaments (stress fibers), usually arranged parallel to the long axis of the cell and among which are interspersed numerous dense bodies (Figure 6.1A). As in vascular smooth muscle cells, these structures may be in continuity with dense bands or plasmalemmal attachment plaques. Rough endoplasmic reticulum and Golgi area are well developed. The nucleus displays deep indentations (Figure 6.1A), an ultrastructural feature that has been correlated with cellular contraction in several systems (51,52,53,54). Several nuclear bodies are usually present, and nucleoli are conspicuous. Myofibroblasts generally are surrounded by substantial amounts of extracellular matrix.
A precise definition of the myofibroblast is an issue of major importance for the surgical pathologist. In our opinion, a myofibroblast can only be defined by ultrastructure, since immunohistochemical studies reveal that myofibroblasts have a heterogeneous and complex pattern of protein expression. The three essential ultrastructural elements that define a myofibroblast are: (a) stress fibers (i.e., bundles of micro- (myo-) filaments with interspersed dense bodies running parallel to the long axis of the cell, commonly located beneath the cell membrane); (b) well-developed cell-to-stroma attachment sites (fibronexus); and (c) intercellular intermediate and gap junctions (55,56). This definition has now been accepted by several major textbooks (57,58,59) in regard to the issue of myofibroblasts in tumor pathology. We do not deny that the light and immunohistochemical microscopic differences between smooth muscle cells and myofibroblasts may be subtle, especially when considering degrees of differentiation of smooth muscle and myofibroblastic proliferations. Nonetheless, the myofibroblast is defined as a highly differentiated cell by ultrastructure alone.
Histologic
Although morphologically defined with the electron microscope, myofibroblasts disclose several typical histologic traits that permit their presumptive recognition in routine paraffin or (even better) in plastic sections in settings in which they previously were identified by ultrastructure. The cells are usually large, spindle-shaped, and often stellate (spiderlike) with several long cytoplasmic extensions, and they possess distinct acidophilic to amphophilic and fibrillar cytoplasm with cablelike condensations (stress fibers) running through the subplasmalemmal cytoplasm parallel to the long axis. The nuclei often are indented or reveal strangulations of nuclear segments, a feature thought to reflect cellular contraction, and contain finely granular, regularly dispersed chromatin and conspicuous nucleoli (Figure 6.2). Well-differentiated myofibroblasts with the previously mentioned traits are observed in poorly-collagenized and edematous areas of various settings in which they were
P.126
originally described; for example, in granulation tissue, in zones of early invasive carcinomas, in invasive and metastatic carcinomas characterized by retraction and desmoplasia, and in several other proliferative conditions. In heavily collagenized zones, myofibroblasts are difficult to recognize with the light microscope since they correspond ultrastructurally to poorly-developed myofibroblasts or fibroblasts.
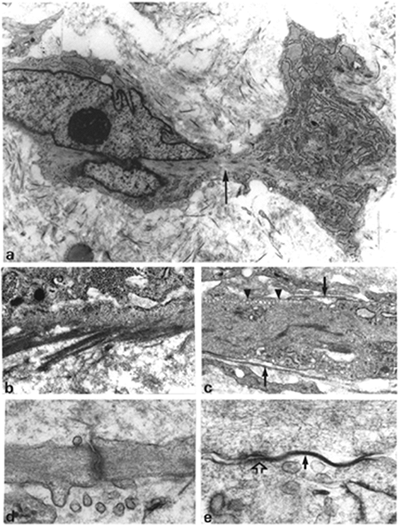 |
Figure 6.1 Ultrastructural characterization of the myofibroblast. A. Typical myofibroblast with irregular shape and cytoplasmic extensions, well-developed rough endoplasmic reticulum, and bundle of cytoplasmic microfilaments (arrow) with numerous dense bodies running through the cytoplasm (stress fibers) giving rise to strangulation of a nuclear segment. (Source: Sch rch W, Seemayer T, Lagac R, Gabbiani G. The intermediate filament cytoskeleton of myofibroblasts. Virchows Arch A. 1984; 403:323 336. ) B. Microtendons in apparent continuity with bundles of cytoplasmic microfilaments (cell-to-stroma attachment sites; i.e., fibronexus). C. Cytoplasm of myofibroblast demonstrating basal lamina (arrows), pinocytotic vesicles (arrowheads), and plasmalemmal attachment plaques. D. Intermediate or adherens junction between two cytoplasmic extensions of myofibroblasts. E. Gap junction (arrow), followed by intermediate junction (open arrow) joining two myofibroblasts. (Source:Sch rch W, Skalli O, Gabbiani G. Cellular biology of Dupuytren's disease. In McFarlane RM, McGrouther DA, Flint MH, eds. Biology and Treatment. Edinburgh: Churchill Livingstone: 1990:31 47. ) (Uranyl acetate and lead citrate; A, 9900; B, 25,000; C, 18,200; D, 39,000; E, 78,000.) |
It is possible that in the near future myofibroblasts might be clearly recognized and defined by immunohistochemical examination of the complex stress fiber associated cell-to-matrix junctions, using multiple labeling techniques and employing confocal laser microscopy on paraffin sections (for illustrations see Figure 6.3 and 6.4).
Immunohistochemical
For a better understanding and appreciation of the various cytoskeletal phenotypes of myofibroblasts, a detailed description of cytoskeletal proteins of muscular tissues, particularly smooth muscle cells, is presented.
Specific cytoskeletal proteins have been defined during the differentiation of muscular tissues (60,61,62,63). These proteins have served as reliable markers of cellular adaptation to physiologic and pathologic conditions (64).
Desmin is a muscle differentiation marker that appears early in embryogenesis (65,66). This intermediate filament, however, does not permit one to distinguish between different muscle types (67). Moreover, desmin is present in
P.127
stromal cells of several organs, which traditionally were considered fibroblastic in nature (68). When smooth muscle cells are cultured, desmin disappears (69). Smooth muscle myosin is a precise marker of smooth muscle differentiation. This contractile protein, however, disappears rapidly from smooth muscle cells in several conditions in vivo and also early in culture (69,70). These findings suggest that smooth muscle myosin is a more reliable marker of smooth muscle differentiation than smooth muscle origin. Vascular smooth muscle cells are heterogeneous with respect to intermediate filament proteins. Most contain vimentin as their sole detectable intermediate filament; a lesser proportion also expresses desmin (71,72,73,74,75). Parenchymal smooth muscle cells of the respiratory, gastrointestinal, and genitourinary tracts represent a homogeneous population in which desmin is almost the exclusive intermediate filament protein (76,77,78).
 |
Figure 6.2 Histological aspect of myofibroblasts from the exudativo-productive layer of human granulation tissue, approximately 15 days old. Myofibroblasts disclose stellate, spiderlike shapes with long cytoplasmic extensions and distinct fibrillar cytoplasm with cablelike (arrows) subplasmalemmal condensations (stress fibers) (hematoxylin-phloxine-saffron). |
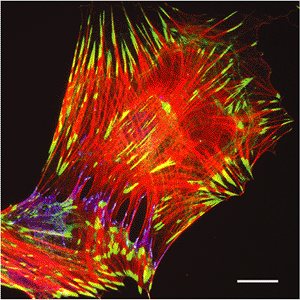 |
Figure 6.3 Stress fiber associated cell-to-matrix and cell-to-cell junctions in cultured myofibroblasts. Rat lung myofibroblasts were cultured on planar glass substrates and immunostained for -smooth muscle actin (red) as a component of contractile stress fibers, paxillin (green) as a component of cell-matrix focal adhesions, and -catenin (blue) as a marker for cell-cell adherens junctions. Myofibroblasts form large, supermature focal adhesions with the extracellular matrix and adherens junctions with adjacent cells; both structures are located at the terminal portion of -smooth muscle actin positive stress fibers. (Bar, 20 m.) |
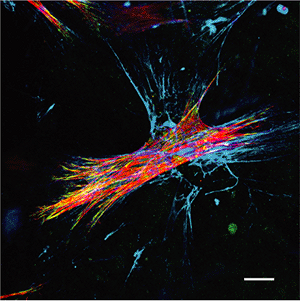 |
Figure 6.4 Fibronexus organization in myofibroblasts placed in three-dimensional collagen gels. Rat lung myofibroblasts were cultured in restrained collagen gels and immunostained for -smooth muscle actin (red) in stress fibers, vinculin (green) in cell-matrix adhesions, and ED-A fibronectin (blue) in the extracellular matrix. The image has been reconstructed from the overlay of 10 optical sections of 0.2 m acquired with a laser scanning confocal microscope; yellow color indicates colocalization of -smooth muscle actin and vinculin; white shows colocalization of both proteins with ED-A fibronectin. Note that extracellular fibronectin fibrils are co-orientated with intracellular stress fibers; they also penetrate in the surrounding extracellular matrix in the continuation of stress fibers. This organization corresponds to the fibronexus originally described by means of electron microscopy. (Bar, 20 m.) |
With regard to actin expression, at least six isoforms are defined in mammals (79,80,81): two nonmuscle actins ( and ), two smooth muscle actins ( and ) and two sarcomeric actins ( -cardiac and -skeletal). The emergence of distinct muscle and cytoplasmic actin isoforms is phylogenetically ancestral, dating before chordates (82,83). The nonmuscle actins, the so-called cytoplasmic actins, are considered the archetypes (80) because of their presence in all nonmuscle cells, including eukaryotic unicellular organisms. According to amino acid patterns, -skeletal actin represents the most
P.128
differentiated isoform (80). At the protein level, -skeletal actin is most closely related to -cardiac actin, whereas -smooth-muscle actin is more closely related to the cytoplasmic actins. Some years ago it was suggested that both -cardiac and -smooth muscle actin represent embryonic or fetal actin isoforms, as they are expressed during skeletal myogenesis (60). Two-dimensional gel electrophoresis resolves only three isoforms: and (nonmuscle and muscle actins) and the -actins (smooth, striated skeletal, and striated cardiac). The biochemical identification of the six actin isoforms requires chemical analysis of the amino-terminal tryptic peptide in cellular extracts. The six actin isoforms may also be determined by RNA extraction and Northern blot hybridization using specific probes (84) and, more recently, with specific antibodies for the six actin isoforms (85). Vascular smooth muscle cells are characterized by a predominance of the -smooth muscle actin isoform. In contrast, parenchymal smooth muscle cells contain large amounts of the -smooth muscle actin isoform (79,80,81,86). The pattern of -, - and -actin isoform expression varies in smooth muscle tissues of adult mammals (86). This pattern varies also during nonneoplastic pathologic conditions, such as atheromatosis (87,88), but changes only slightly in uterine leiomyomas, compared with normal myometrium (86). During the early months of life, 50% of cells in the aortic media lack -smooth muscle actin, whereas -smooth muscle actin negative cells constitute less than 1% in the adult. These findings demonstrate that, at least in arteries, differentiation of smooth muscle cells is completed after birth (67). These observations collectively suggest that the pattern of -actin isoform expression and, particularly, the expression of -smooth muscle actin in vascular smooth muscle cells are related to the degree of smooth muscle differentiation.
Pericytes resemble vascular smooth muscle cells (89). In a meticulously executed treatise published in 1923, Zimmermann showed that pre- and postcapillary pericytes are connected to vascular smooth muscle cells (90). In 1991, an elegant study showed that pre- and postcapillary pericytes indeed expressed -smooth muscle actin, whereas the midcapillary pericytes fail to express this actin isoform (91). Pericytes were also shown to resemble vascular smooth muscle cells by their intermediate filament expression. Both cell types express vimentin or vimentin and desmin (92). In addition, the intermediate filament composition of pericytes discloses species and tissue differences similar to those observed in vascular smooth muscle cells (71,72,73,74).
Myofibroblasts in normal tissue, granulation tissue, and pathologic tissues disclose five cytoskeletal phenotypes: phenotype V, represented by cells expressing only vimentin; phenotype VA, represented by cells expressing vimentin and -smooth muscle actin; phenotype VAD, represented by cells expressing vimentin, -smooth muscle actin, and desmin; phenotype VD, represented by cells expressing vimentin and desmin; and phenotype VA (D) M, representing myofibroblasts expressing vimentin, -smooth muscle actin, and smooth muscle myosin heavy chains, with and without desmin. The five phenotypes are readily defined in frozen and paraffin sections using immunohistochemistry, employing single, double, or triple staining techniques. Myofibroblasts of the various immunophenotypes may also express the -and -cytoplasmic actins, although it is more common that they express -smooth muscle actin. This has led to the misconception that, for a cell to be classified as a myofibroblast, it must express -smooth muscle actin. This is not true, as cells other than myofibroblasts express - smooth muscle actin. There are situations in which cells have the ultrastructural characteristics of myofibroblasts (e.g., stress fibers) but do not express -smooth muscle actin; for example, interstitial cells in alveolar septa and the early phase of granulation tissue (93,94). (For illustration see below.) Considering these data, it is apparent that the distinction between smooth muscle cells and myofibroblasts remains complex at the immunohistochemical level and that no single immunophenotype is distinctive for myofibroblasts.
Biochemical
Myofibroblasts possess not only contractile forces, but also synthetic properties. Four major groups of macromolecules comprise the extracellular matrix: (a) collagens; (b) glycoproteins (e.g., fibronectins, laminins, tenascin); (c) proteoglycans (e.g., aggrecan, synchrons, perlecan, decorin); and, (d) elastins with their associated proteins (95). Myofibroblasts possess synthetic properties for several extracellular matrix components: collagens type I, III, IV and V (29,96,97,98), glycoproteins (99), and proteoglycans such as fibronectin, laminin, and tenascin (100). In addition, liver myofibroblasts in the murine schistosomiasis model secrete lysyl oxidase, an enzyme that initiates the first step in the cross-linking of collagen and elastin, a crucial function for the stabilization of the extracellular matrix (101,102).
Concerning collagen synthesis in granulating wounds, the collagen initially produced is type III. This form of collagen imparts a measure of plasticity to the wound in the early phase of healing. When granulation tissue is resorbed following wound closure, myofibroblasts disappear through the process of apoptosis (see below) and the more rigid type I collagen is biochemically identified (97,98). In similar fashion, the proliferative cellular phase of palmar fibromatosis and the young edematous mesenchyme of areas corresponding to early stromal invasion of breast carcinomas, both rich in myofibroblasts, contain increased amounts of type III collagen (26,103,104). Increased amounts of type V collagen are biochemically identified in desmoplastic human breast carcinomas, apparently also produced by myofibroblasts (98).
P.129
Pharmacologic
Strips of granulation tissue exposed in vitro to a variety of pharmacological agents contract and relax in a manner analogous to smooth muscle. Prostaglandin F1, bradykinin, serotonin, endothelin-1, histamine, angiotensin, norepinephrine, epinephrine and vasopressin initiate contraction. The intensity of the response depends on the origin, age, and initial degree of contraction of the granulation tissue tested. Prostaglandins E1 and E2 and papaverine induce relaxation in tissues in a contracted state (2,3,17). Cytocholasin B abolishes the contraction of granulation tissue, probably as the result of microfilament disruption (105). Trocinate ( -diethylaminoethylphenylthioacetate), another inhibitor of smooth muscle contraction, has been reported to decrease contraction when applied topically on rabbit wounds (106).
Strips of cirrhotic liver, when exposed to smooth muscle stimulating agents, contract significantly when compared with strips of normal liver (107). Pronounced myofibroblastic interstitial fibrosis is also produced in lungs of bleomycin-injected rats. When strips of these fibrotic lungs are exposed to acetylcholine, epinephrine, and a K+-depolarizing solution, the force developed is approximately twice that of normal lung tissue strips (108). The relative reactivity to various stimulating agents of myofibroblasts from diverse sources varies; thus, acetylcholine causes contraction of strips of fibrotic lungs but not of granulation tissue from a skin wound or a granuloma pouch, and serotonin induces retraction of the granuloma pouch but not strips from a skin wound (3). In addition to this heterogeneity in the pharmacological reactivity of granulation tissue strips from various sources, there are also differences between the response of strips of granulation tissue and strips of smooth muscle; the former reach their peak contraction slower but maintain it longer than the latter.
Whereas the various enumerated agents were shown to reveal their activity on granulation tissue in vitro, the exact mechanism leading to myofibroblast contraction in vivo remains to be elucidated. In this context, it is noteworthy that when hepatic stellate cells are subjected to in vivo ischemia reperfusion injury, they exhibit a de novo temporary increase of -smooth muscle actin expression. A similar phenomenon takes place during the initial phases of liver transplantation in humans (109). Likely, this represents a response to ischemic injury.
Endothelin-1 was originally isolated from the conditioned medium of cultured porcine endothelial cells and was shown to be the most potent vasopressor substance yet characterized (110). Endothelin-1 may be an endogenous modulator of myofibroblast-mediated contraction because it causes reversible and concentration-dependent contraction of granuloma pouch granulation tissue, the 21-day granulation tissue being the most responsive. This response can be inhibited by calcium antagonists (111,112). The vasopressor effect of endothelin-1 possibly is controlled and mediated through the action of cytokines, among others, TGF- (113,114), which, in turn, is able to induce -smooth muscle actin expression in fibroblasts and myofibroblasts. More recently it has been shown that the intracellular administration of the N-terminal peptide of -smooth muscle actin, the actin isoform responsible for myofibroblast contraction (see below), decreases force generation by myofibroblasts in vitro and inhibits wound contraction in vivo (115); this may represent a useful tool for the control of tissue retraction and remodeling during several pathological situations.
In Vitro Culture Studies
When myofibroblasts from various sources (granulating wounds, Dupuytren's disease, and invasive breast cancer) are cultured, they maintain to a certain extent their unique resemblance to fibroblasts and smooth muscle cells. Cultured fibroblasts may express different phenotypic features, and a spectrum of differentiation steps has been described (116). In particular, primary passaged fibroblastic cells in culture express -smooth muscle actin (68,79,117). Cytoskeletal proteins such as desmin and smooth muscle myosin heavy chains are also variably expressed by cultured fibroblasts derived from different organs or pathologic tissues, but expression is generally low and absent in several populations (118). Myofibroblasts cultured from skin wound granulation tissue maintain some biologic features different from those of dermal fibroblasts (119). If the growth rate and the actin concentration of cultured fibroblasts from normal dermis and myofibroblasts of human granulation tissue are compared, myofibroblasts grow slower than fibroblasts (120) and contain a significantly higher concentration of actin (121). Wound-healing fibroblasts were shown to develop greater contractile properties than dermal fibroblasts (122). Similarly, fibroblastic cells cultured from Dupuytren's nodules maintain biologic features different from those of normal dermis or fascial fibroblasts yet are similar to those of neoplastic or embryonic fibroblasts (123). However, it is important to state that the percentage of cells exhibiting myofibroblast features in culture does not necessarily reflect their proportion in the tissue of origin. When fibroblast cultures from various species, including man, were established using cloning and subcloning techniques, a certain percentage of cells was positive for -smooth muscle actin (118). This concept of fibroblast heterogeneity is now well-established in vitro (124) and in vivo [see review by Schmitt-Gr ff et al. (8)]. Alpha-smooth muscle actin is expressed by fibroblasts cultured from the lens of the eye, mammary gland, perisinusoidal cells of the liver, and glomerular mesangial cells, sites where stromal cells normally expressing this protein do not occur (125,126,127,128) but which may give rise to reactive stromal cells expressing
P.130
-smooth muscle actin in pathologic conditions. It is likely that -smooth muscle actin expression in cultured fibroblasts stems from the culture conditions imposed upon the fibroblasts. Fibroblastic modulation to a myofibroblast phenotype in vitro may thus reflect a cellular response to their altered biochemical and mechanical environment, somewhat analogous to that which occurs in wounds.
Myofibroblast Development: A Two-Stage Model
Fibroblasts cultured on planar culture dishes rapidly attach to the wall of the container and move across its surface; adherence and mobility are attributed to the de novo development of a system of microfilament bundles called stress fibers (129). These may measure up to 2 m in diameter and connect to the extracellular matrix at sites of focal adhesions (130) and to adjacent cells at sites of adherens junctions (50) (Figure 6.3). Initially, stress fibers are mainly composed of cytoplasmic actins, as shown by immunofluorescence and immunoelectron microscopy with specific antibodies (131,132,133). Several studies have shown that stress fibers also contain actin-associated proteins such as myosin, tropomyosin, -actinin, and filamin (7). The formation of stress fibers and cell-matrix adhesions is in sharp contrast to the cortical arrangement of actin in fibroblasts in most normal connective tissues and represents the first step in the development of the myofibroblast phenotype. The term protomyofibroblast was recently proposed for fibroblasts with contractile stress fibers that do not (yet) express -smooth muscle actin (134).
Development of the protomyofibroblast is predominantly controlled by the mechanical properties of the extracellular matrix [for reviews see (134,135)] and requires the permanent feedback between intracellular and extracellular tension. Stress fiber formation in fibroblasts on rigid glass or plastic surfaces is increased after contractile activity stimulation and is lost upon application of inhibitors of contraction (136). In contrast, fibroblasts grown on compliant substrates fail to develop protomyofibroblastic features even in the presence of contraction agonists (137). When embedded in a three-dimensional gel of newly polymerized collagen, fibroblasts acquire a dendritic morphology similar to that in normal dermis (138); they extend and retract long processes, possibly to explore and organize the new environment (139). In free-floating collagen gels the forces developed by such processes lead to collagen gel condensation (139). However, since collagen fibers are free to move, overall matrix stress does not develop, and fibroblasts maintain their dendritic appearance (139). In contrast, collagen reorganization in restrained gels produces matrix stiffening and increases global stress, inducing alignment of fibroblasts along the lines of tension and the formation of stress fibers (134,139) (Figure 6.4). Similarly, after stress-release of restrained collagen gels, protomyofibroblasts rapidly loose stress fibers and matrix contacts (140). Importantly, fibroblasts are capable of adjusting their contractile activity according to the external load; controlled stress-release of collagen gels in a culture increases fibroblast contractile activity, whereas gel stretching leads to fibroblast relaxation, a phenomenon that has been termed tensional homeostasis (141).
A similar mechanism of protomyofibroblast development appears to apply in vivo. In most normal connective tissues, fibroblasts are protected from external tensile stress by their surrounding matrix (134). This situation changes dramatically with an altered microenvironment, such as when a dermal wound is provisionally filled with a clot of fibrin and blood platelets, which, in conjunction with white blood cells, release a variety of cytokines (142,143). These changes stimulate fibroblasts to migrate into the wound bed, where they proliferate and initiate restoration of the dermis by secreting and organizing the dermal matrix. The increasing number of migrating fibroblasts enhances matrix rigidity by applying tractional forces to the newly formed granulation tissue, ultimately leading to the development of stress fibers.
A number of recent studies have demonstrated that mechanical stress is a prerequisite for the second step of myofibroblast development, signaled by the expression of -smooth muscle actin in stress fibers (134,68). In vitro, the level of -smooth muscle actin expression increases with increasing matrix rigidity, as demonstrated by growing differentiated myofibroblasts in collagen substrates or on polyacrylamide elastomers exhibiting different degrees of stiffness (144,145). In vivo, application of mechanical stress to granulation tissue fibroblasts by splinting the wound edges with a plastic frame accelerates expression of -smooth muscle actin compared to normally healing wounds; stress release by removal of the frame rapidly leads to the disassembly of stress fibers and loss of -smooth muscle actin expression (146).
In addition to mechanical stress, transformation of the protomyofibroblast into the differentiated myofibroblast requires the concerted action of cytokines and specific components of the extracellular matrix. It is increasingly accepted that TGF- 1 is the major growth factor inducing myofibroblast differentiation (27,147,148) from fibroblastic cells (147,149) and mesangial cells (33,150). More recently, thrombin (151) and endothelin-1 (152) have been shown to induce myofibroblast differentiation, the latter either directly or in coculture with epidermal cells (153). It has been demonstrated in experimental animals that the subcutaneous administration of granulocyte macrophage colony stimulating factor (GM-CSF) promotes the development of granulation tissue rich in -smooth muscle actin positive myofibroblasts (154,155); this action, however, is indirect and could be mediated by TGF- . TGF- mediated expression of -smooth muscle actin depends
P.131
upon the presence of the fibronectin splice variant ED-A fibronectin (30) in the extracellular matrix (Figure 6.4). This clearly demonstrates the complex interplay of diffusible and immobilized factors in promoting the development of differentiated myofibroblasts. Other cytokines and/or growth factors have been shown to facilitate or inhibit myofibroblast development and proliferation. Nerve growth factor (156) facilitates the process, however, it is not known whether this action is independent of TGF- 1; interferon- inhibits the process (118).
Mechanisms of Force Generation and Transmission
Several observations suggest that stress fibers are the force-generating elements in wound contraction, since they contract upon addition of adenosine triphosphate to glycerinated fibroblasts (13,157,158). As well, microinjection experiments revealed that stress fibers are functionally analogous to skeletal muscle fibers (158,159). Several models have been developed to study the contractile activity of fibroblastic cells in vitro. Using two-dimensional deformable silicone substrates, fibroblasts have been shown to produce long-lasting wrinkles of the substrate, suggesting the production of continuous isometric tension (160). This technique has been greatly improved over the past years by employing different elastic polymers and mathematical models to calculate cell-generated forces from substrate distortions [for a review see (161)]. In a more tissuelike approach to assess the dynamics of wound contraction, fibroblasts are cultured in collagen or fibrin matrices that are either free-floating and retracting over days or mechanically restrained for days and then stress released, leading to contraction within minutes [for review see (139)]. Importantly, stress fibers and matrix adhesion (i.e., the myofibroblastic phenotype) only develop in mechanically restrained gels. Hence, the choice of the appropriate collagen model facilitates study of the traction forces of migrating fibroblasts or stress fiber-mediated contraction.
Similar to what occurs in smooth muscle, stress fiber contraction may be regulated by elevated levels of intracellular Ca2+, leading to activation of myosin light chain kinase and phosphorylation of the myosin light chain. However, experimental and clinical observations show that granulation tissue retraction, in contrast to rapid and reversible contraction of smooth muscle, is the result of a continuous isometric force exerted on the surrounding connective tissue. This retraction is then stabilized by deposition of newly synthesized matrix components and thus becomes irreversible (134). In the last few years, the work of several laboratories has suggested that the isometric tension produced by stress fibers is regulated by Rho/Rho-kinase, which in its active form leads to long lasting tensile activity by the inhibition of myosin phosphatase (162). Phosphatase inhibitors stimulate myofibroblast contraction in vitro in the absence of any other contraction agonist (163). In contrast, increasing intracellular Ca2+ with ionophore has no contractile effect, indicating that activation of myosin light chain kinase alone is not sufficient to promote myofibroblast tension development (164). More recently it has been reported that thrombin activation of human lung myofibroblast tension development is mediated by protein kinase C and RhoA and depends on the activation of Ca2+-mediated and Rho-kinase signaling pathways (151).
During the past several years, it has become evident that the expression of -smooth muscle actin in stress fibers is instrumental in force generation by myofibroblasts. Compared with -smooth muscle actin negative fibroblasts, myofibroblasts develop higher contractile force as demonstrated using deformable silicone substrates (164,165) and contracting collagen gels (165,166). Stable transfection with -smooth muscle actin confers upon fibroblasts a higher contractile activity compared with transfection with cytoplasmic or sarcomeric actin isoforms; this effect is exerted in the absence of any change in the expression of other contractile proteins, such as smooth muscle or nonmuscle myosin (165). The mechanism by which -smooth muscle actin promotes myofibroblast-enhanced contractile activity has not been defined; however, it is inhibited in vitro and in vivo and by the intracellular delivery of the -smooth muscle actin specific N-terminal sequence AcEEED (115).
The force generated by stress fibers is transmitted to the extracellular matrix at sites of cell-matrix adhesions (130). In vivo, myofibroblasts form a specialized adhesion complex, the fibronexus (49), which is characterized by a firm co-alignment of intracellular actin fibers with extracellular fibronectin fibrils (Figure 6.1B); these in turn are connected to collagen in the wound matrix (19). In vitro, differentiated myofibroblasts communicate with the extracellular matrix through specialized supermature focal adhesions (145,167), which have a diameter of 6 to 30 m and strongly express the cytoplasmic proteins vinculin, paxillin, and tensin and the transmembrane integrins v 3 and 5 1 (145,167). This is in contrast to smaller focal adhesions (FAs) (2 6 m) of -smooth muscle actin-negative fibroblasts that do not exhibit significant levels of tensin and 5 1 integrin or to fibrillar adhesions that are generally negative for vinculin, paxillin and v 3 integrin (130,168). Focal adhesion supermaturation depends on the high contractile activity developed by -SMA containing stress fibers (145), analogous to the maturation of classical FAs from nascent focal complexes in response to up-regulated cell contractile activity (136). It has been proposed that supermature focal adhesions are particularly efficient in promoting tissue contraction (134,135) by providing high adhesion to the substrate (145) and by immobilizing the cells in the wound bed (169).
P.132
Tissue Distribution of Myofibroblasts
Normal Tissues
Myofibroblasts were described in normal human and animal tissues on the basis of ultrastructural and/or immunohistochemical evidence of smooth muscle differentiation. The normal settings in which myofibroblasts were observed include the external theca of the rat ovarian follicle (170); developing human palatal mucosa (171); rat, rabbit, and human intestinal mucosa (172,173,174); rat and mouse adrenal capsule (175), human, lamb, and monkey pulmonary alveolar septa (176); rat testicular stroma (68); rat testicular capsule (177); human theca externa of the ovary (178); Wharton's jelly of human umbilical cord (179); bovine endometrial caruncle (180); and periodontal ligament of the mouse (181) and rat (182), where they facilitate tooth eruption. Stromal cells with myoid features were also identified in rat and human lymph nodes and in the human spleen (183). Another group of stromal cells with myoid features include hepatic perisinusoidal cells (184), those in the human uterine submucosa (185) and human bone marrow (186), glomerular mesangial cells of mouse, rat, and human (187), and, possibly, pre-and postcapillary pericytes (89,91).
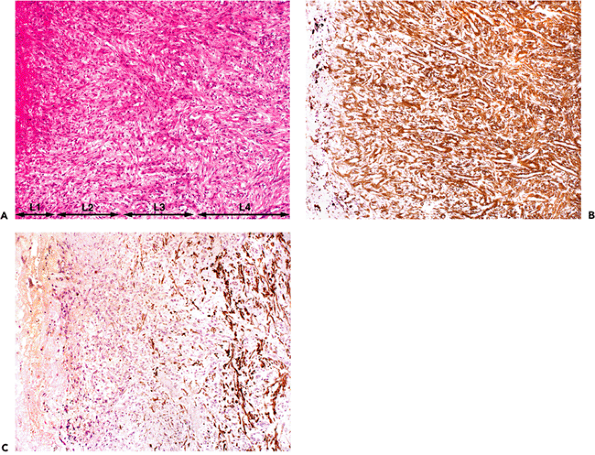 |
Figure 6.5 Human chronic granulation tissue from pleural empyema. A. The four layers (L1 L4) are clearly discernible: L1, alterative; L2, exudative; L3, exudativo-productive; L4, cicatrizing (hematoxylin-phloxine-saffron). B. Most myofibroblasts from the exudative and exudativo-productive layer (L2 andL3) reveal intense immunostaining for -smooth muscle actin (phenotype VA) (avidin-biotin-complex-peroxidase). Note that myofibroblastic cells change their orientation within the different layers. Within the exudative layer, their long axis is perpendicular to the surface, whereas in the exudativo-productive layer their long axis is often oblique to the surface. Within the cicatrizing layer, myofibroblastic cells are oriented parallel to the surface, which indicates that the modulation of cellular orientation serves to transmit contractile forces to effect wound closure. C. In the cicatrizing layer (L4), numerous myofibroblasts express desmin (phenotype VAD). (B and C are step-sections.) |
Immunohistochemical studies disclosed heterogeneous cytoskeletal phenotypes among all of these stromal cells (myofibroblasts) in terms of intermediate filament protein, smooth muscle actin, and smooth muscle myosin expression; these include V, VD, VA (D) M, VA, and VAD phenotypes (188,189). This cytoskeletal heterogeneity could reflect different functional needs since all of these stromal cells seem to participate in visceral contraction or extracellular matrix remodeling, a view supported by the observation that stromal cells with myoid features are generally present in organs requiring contraction or high degrees of remodeling (190). Another recently advanced interpretation proposes that most spindle cells in normal tissues cited as being myofibroblasts might be closer to pericytes,
P.133
smooth muscle cells (191), or stromal cells with myoid features of variable degrees that correspond to functional demands. As shall be seen in the following sections, myofibroblasts and/or stromal cells with myoid features are not stable in terms of cytoskeletal phenotypes. In normal, abnormal, and pathologic conditions, the phenotype V may change into phenotype VA, VAD, VD, and, eventually, into VA (D) M, but terminal smooth muscle differentiation (smooth muscle metaplasia) is never attained. Thus, the myofibroblast remains an enigmatic cell, one that appears and disappears after completion of its functions or, exceptionally, one that may persist in certain pathologic conditions.
Granulation Tissue
Granulation tissue (Figures 6.5,6.6,6.7) consists of a bed of fibroblastic cells separated by a collagenous matrix containing capillary buds, fibrin, and inflammatory cells. According to the relative predominance of each constituent, four layers are classically distinguished: (a) alterative; (b) exudative; (c) exudativo-productive; and (d) cicatrizing (Figure 6.6A). Granulation tissue fibroblasts characteristically disclose ultrastructural features of myofibroblasts. They are most numerous and best developed within the exudativo-productive layer and become progressively replaced toward the deepest cicatrizing layer by fibroblasts. The orientation of the myofibroblasts varies in the different layers of granulation tissue. In the exudative layer, the long axis is perpendicular to the surface, whereas in the exudativo-productive and cicatrizing layers, the long axis is parallel to the surface (Figure 6.6A). These data suggest that the spatial orientation of myofibroblasts in granulating wounds varies, possibly to maximize the transmission of contractile forces and thereby effect wound closure. When the collagenous matrix is analyzed, type III collagen predominates. When granulation tissue is resorbed following wound closure, myofibroblasts disappear (25,97,192) and the more rigid type I collagen is identified (96).
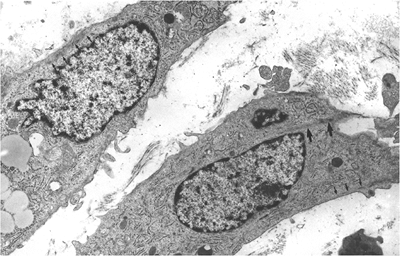 |
Figure 6.6 Human granulation tissue, approximately 5 days old, composed of phenotype V myofibroblasts. These cells disclose subplasmalemmal bundles of microfilaments with few dense bodies (small arrows) and also intracytoplasmic bundles of microfilaments with dense bodies (large arrows) corresponding to stress fibers in formation (uranyl acetate and lead citrate, 12,500). |
Analysis of cytoskeletal proteins by immunohistochemical methods reveals that myofibroblasts from normal healing wounds never express desmin or smooth muscle myosin heavy chains during the process of wound closure in the experimental animal (25). Smooth muscle differentiation in early granulation tissue is absent and myofibroblasts are often poorly-developed and correspond to V cells (Figure 6.5). Smooth muscle differentiation of myofibroblasts, however, becomes temporarily apparent because myofibroblasts express -smooth muscle actin (VA cells) (Figure 6.7A and B) in increasing amounts from the eighth to the fifteenth day; this protein is located within bundles of microfilaments (stress fibers), as illustrated by immunoelectron microscopic techniques (Figure 6.7C and D). This actin isoform disappears progressively from myofibroblasts and is not detectable after the thirtieth day by immunohistochemical and immunoelectron microscopic methods. These results clearly indicate that granulation tissue myofibroblasts temporarily acquire a VA phenotype. This is valid when wound repair is accomplished by primary intention. When repair is accomplished by secondary intention in chronic granulation tissue (e.g., chronic gastric ulcer, cutaneous ulceration, or pleural empyema), myofibroblasts of the VAD phenotype may be detectable (Figure 6.6B and C).
The study of the ontogenesis of wound healing reveals that many species possess the unique ability to heal wounds without scarring (193,194,195,196,197,198). Estes et al. (199),
P.134
examining fetal wounds in the fetal lamb, recently showed that there are differences between early and late gestational wound healing. In the lamb, term gestation is approximately 145 days. Early gestational wounds (75 days) healed without scarring by repair of the epidermis, reconstitution of epidermal appendages, and remodeling of the dermal collagenous network. In contrast, late gestational wounds (100 and 120 days) healed with scarring through formation of granulation tissue containing myofibroblasts that were mostly derived from local resident fibroblasts. The situation appears to be different in healing corneal wounds, in which corneal keratocytes transform into repair corneal fibroblasts or myofibroblasts (200,201,202).
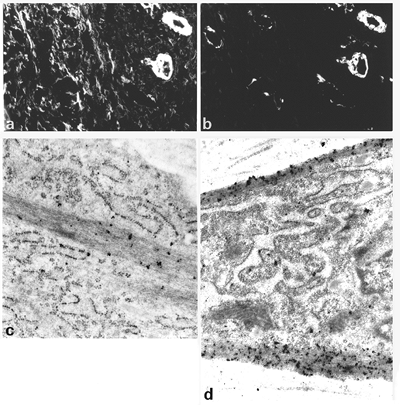 |
Figure 6.7 Experimental granulation tissue from the rat, 15 days old. A. Double immunofluorescent staining for -smooth muscle actin (A). B. Double immunofluorescent staining for desmin. Myofibroblastic cells reveal intense staining for -smooth muscle actin (A) but are negative for desmin (B). Vascular smooth muscle cells are positive both for smooth muscle actin and desmin. C. Immunoelectron microscopic localization of -smooth muscle actin within intracytoplasmic bundles of microfilaments, that is, stress fibers. D. Immunoelectron microscopic localization of -smooth muscle actin in subplasmalemmal bundles of microfilaments. (C, 31,000; D, 28,400.) |
Pathologic Tissues
Upon analysis of the many pathologic conditions in which myofibroblasts have been described, three fundamental processes emerge: (a) responses to injury and repair phenomena or situations related to inflammation and tissue remodeling; (b) quasineoplastic proliferative conditions; and (c) the stromal response to neoplasia (6,203). This
P.135
concept, enunciated some 25 years ago, appears valid to this day (6).
Responses to Injury and Repair Phenomena
Responses to injury and repair phenomena comprise human and experimental cirrhosis (204,205,206), tenosynovitis (207), radiation-induced pseudosarcoma of skin (208), burn contracture (209), ischemic contractures of intrinsic muscles of the hand (210), renal interstitial fibrosis during obstructive nephropathy (211), pulmonary sarcoidosis (212), giant cell granuloma of jaws (213), schistosomal liver fibrosis (214), regenerating tendon (215), fibrous capsule around silicon mammary implants (216,217), nodular hyperplasia of the liver (218), ganglia of soft tissue (219), hypertrophic scars (220), cataract (221), bleomycin-induced pulmonary interstitial fibrosis in the rat (222), fibrous heart plaque in the carcinoid syndrome (223), atherosclerotic lesions in humans and experimental animals (224,225,226,227), localized and systemic scleroderma (228), and experimental hydronephrosis (229). When cytoskeletal proteins of these conditions are analyzed most reactive cells correspond to the VA, some to the VAD, and few to the VD and VA (D) M phenotype (188,192).
A recent report proposed that a reactive myofibroblastic proliferation with increased deposition/formation of extracellular matrix might be responsible for the progressive and irreversible obstruction of airways in chronic bronchial asthma (230). In asthma, it appears that epithelial-mesenchymal interactions may play an important role in its pathogenesis. Epithelial injury and subepithelial collagen deposition are characteristic of asthma. It was proposed that epithelial cell proliferation increases after airway injury in asthmatics, that epithelial cells stimulate lung myofibroblast collagen production, and that both processes are modulated by allergen-recruited inflammatory cells, proinflammatory cytokines, growth factors, and mediator-generating enzymes. Beneath the damaged bronchial epithelium, there is an increase in the number of subepithelial myofibroblasts that deposit interstitial collagens, causing thickening and increased density of the subepithelial basement membrane (231,232,233).
Focal segmental glomerular hyalinosis/sclerosis (FSGS) is another state which might belong to the group of responses to injury and repair phenomena (Figure 6.8A I). The condition is associated with significant proteinuria and hypertension; many patients develop chronic renal failure, requiring dialysis and eventual renal transplantation. In FSGS, mesangial cells, which normally express only cytoplasmic actins and therefore correspond to myofibroblasts with a V phenotype, may gradually acquire a VA phenotype, expressing -smooth muscle actin as revealed by immunohistochemical techniques (Figure 6.8D). Moreover, they develop stress fibers as observed by ultrastructural examination (Figure 6.8I). These findings are similar to those in experimental immune complex nephritis in which mesangial expression of smooth muscle actin correlates with mesangial cell proliferation (33). In FSGS, mesangial cells expressing -smooth muscle actin become progressively apparent in the early stage of the disease (podocytosis; Figure 6.8C and D) (234). Their numbers increase as the lesion progresses to the hyalinosis stage (Figure 6.8E and F). At the stage of sclerosis, the number of mesangial cells immunostained for -smooth muscle actin is reduced, somewhat analogous to that which occurs over time in wound healing (Figure 6.8G and H).
In abnormally healing wounds (hypertrophic scars and keloids), one observes several important differences. Hypertrophic scars always exhibit nodular structures in which fibroblastic cells, small vessels, and fine randomly organized collagen fibers are present. Within these nodules, numerous myofibroblasts of the VA phenotype and, in lesser numbers, myofibroblasts of the VAD phenotype are identified (Figure 6.9A H). Exceptionally, myofibroblasts of the VA (D) M phenotype are observed. Keloids contain large thick bands of closely packed cell fibers and rare nodular structures, the latter containing few or no VA cells (235,236). VAD cells are not observed within classical keloids (231), however VA and VAD cells are frequently observed in relatively small keloids of Caucasians (237).
Quasi-neoplastic Proliferative Conditions
This group embodies the poorly-understood but very important and frequent soft tissue proliferations included under the broad heading of fibromatoses, as well as many other soft tissue proliferations (often mimicking sarcomas) that share a predominant myofibroblastic composition and a variable proliferative potential yet do not disseminate or metastasize (6,203).
Myofibroblasts constitute the principal cellular components of superficial and deep musculoaponeurotic fibromatoses (238). Superficial (fascial) fibromatoses include palmar fibromatosis [Dupuytren's disease (16,103,239,240,241)], plantar fibromatosis [Ledderhose's disease (16)], penile fibromatosis [Peyronie's disease (240)], and knuckle pads (243). Deep musculoaponeurotic fibromatoses comprise extra-abdominal, abdominal and intra-abdominal variants, collectively named desmoid tumors (238). To this group belong the infantile fibromatoses (244). Other soft tissue proliferations predominantly composed of myofibroblasts are nodular fasciitis (245), proliferative fasciitis (246), proliferative myositis (247), giant fibroma of oral mucosa (248), dermatofibroma (249), elastofibroma (250), plasma cell granuloma of the lung (251), digital fibroma of infancy (252), and juvenile nasopharyngeal angiofibroma (253). Myofibroblasts are also present in cardiac myxomas (254) and in uterine plexiform tumors (255).
P.136
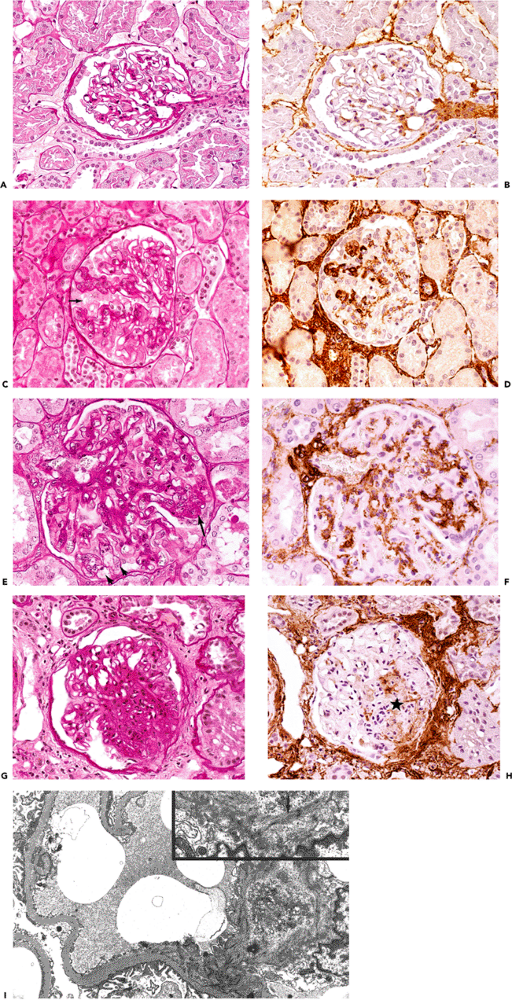 |
Figure 6.8 Evolution of focal segmental glomerular hyalinosis/sclerosis using step-sections stained with PAS and -smooth muscle actin. A. and B. Normal glomerulus; B is without significant immunostaining of mesangial cells for -smooth muscle actin. C. and D. Early stage of focal segmental hyalinosis characterized by vacuolar degeneration of podocytes with hyalin PAS-positive droplets and increased mesangial matrix in two glomerular segments; that is, podocytosis (arrow). Mesangial cells of the two glomerular segments disclose significant immunostaining for -smooth muscle actin (D). E. and F. Typical segmental lesion with hyaline endomembranous PAS-positive deposit (arrow) and foam cells (arrowheads). Mesangial cells disclose significant immunostaining for -smooth muscle actin. G. and H. Segmental sclerosis with decreased immunostaining for -smooth muscle actin (asterisk). I. Mesangial cell disclosing stress fibers composed of microfilaments with dense bodies (arrow), enhanced in inset (uranyl acetate and lead citrate, 8000; inset 15,150). |
P.137
Dupuytren's Disease
Among quasineoplastic proliferations, Dupuytren's fibromatosis has been studied extensively by morphologic, immunohistochemical, and biochemical techniques (24,26,256,257). Cytoskeletal proteins have been widely used as markers of differentiation for neoplastic and quasineoplastic proliferations and as markers of adaptation to physiologic situations, particularly for muscular and related soft tissue proliferations (24,26,64).
According to Luck (258), the nodules of Dupuytren's disease are assigned to three different phases, depending on the histologic pattern: (a) proliferative phase; (b) involutional phase; and (c) residual phase (Figure 6.10A D). Patients with Dupuytren's disease often present multiple nodules showing considerable variation in their histologic appearance. The classification is, therefore, based on the predominant histologic pattern (105,259).
Sections from proliferative phase nodules feature high cellular density, decreasing from the center to the periphery (Figure 6.10A). They are well-vascularized and display a poorly-collagenized appearance. Ultrastructurally, they are composed of myofibroblasts with numerous and long cytoplasmic extensions, joined by numerous gap and adherens junctions (Figure 6.11A and inset). Their plasma membrane displays focal deposition of basal lamina, plasmalemmal attachment plaques, and pinocytotic vesicles, as well as cell-to-stroma attachment sites in the form of fibronexus (49). The cytoplasm features a well-developed rough endoplasmic reticulum and Golgi apparatus and numerous stress fibers, the latter usually oriented parallel to the long axis of the cell (Figure 6.11A). The nucleus is typically indented and often contains one or several nuclear bodies. The extracellular matrix is composed of a few mature collagen fibers (64 nm periodicity) admixed
P.138
P.139
with indistinct granular and basal lamina-like material (Figure 6.11A).
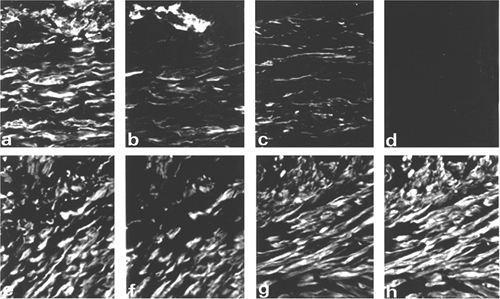 |
Figure 6.9 Double immunofluorescent staining of two hypertrophic scars (A D and E H) with antibodies to vimentin (A and E), -smooth muscle actin (B and F), -smooth muscle actin (C and G), and desmin (D and H). One hypertrophic scar (A D) contains V and VA cells, and the other, from a site of smallpox vaccination (E H), contains mainly VAD cells. Note that most small blood vessels are positive for vimentin and -smooth muscle actin. (Source: Sappino AP, Sch rch W, Gabbiani G. Differentiation repertoire of fibroblastic cells: expression of cytoskeletal proteins as marker of phenotypic modulations. Lab Invest 1990;63:144 161. ) |
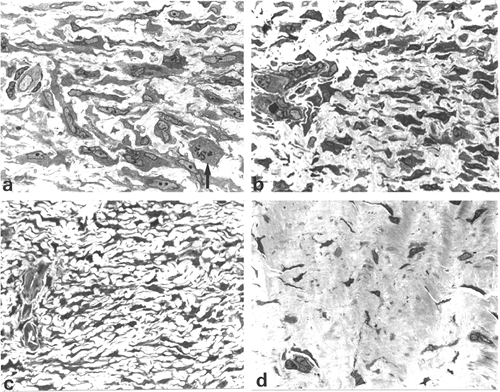 |
Figure 6.10 Dupuytren's disease: semithin sections. A. Proliferative phase nodule illustrating large elongated cells with numerous cytoplasmic extensions and indented nuclei, some in cell division (arrow). B. and C. Involutional phase nodule composed of aligned spindle cells that display fewer, shorter, and smaller cytoplasmic extensions than in A. D. Residual phase nodule showing slender spindle cells in a poorly-vascularized and densely collagenous matrix. (Toluidine blue-stain.) (Source: Sch rch W, Skalli O, Gabbiani G. Cellular biology of Dupuytren's disease. In: McFarlane RM, McGrouther DA, Flint MH, eds. Dupuytren's Disease: Biology and Treatment. London: Churchill Livingstone; 1990:31 47. ) |
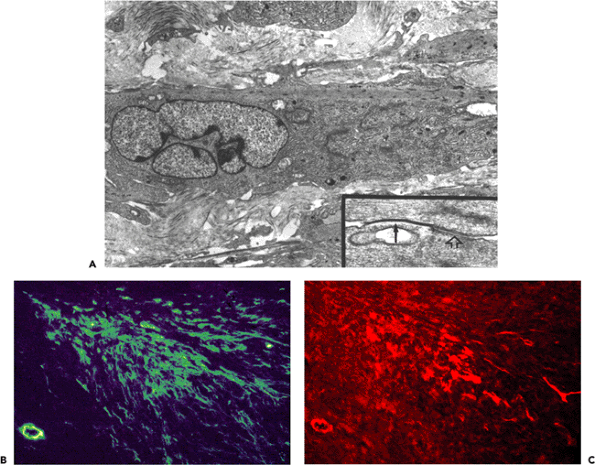 |
Figure 6.11 Dupuytren's disease: proliferative phase nodule. A. Transmission electron micrograph of proliferative phase nodule. Note large typical myofibroblast with cytoplasmic extensions, well-developed rough endoplasmic reticulum and Golgi areas, and prominent cytoplasmic bundle of microfilaments with numerous dense bodies oriented parallel to the long axis of the cell. The nucleus is indented. The extracellular matrix contains few mature collagen fibers. Inset: Gap junction between two myofibroblasts (arrow) followed by an intermediate junction (open arrow). (Uranyl acetate and lead citrate: 7500; inset, 72,000.) B. and C. Double immunofluorescent staining for -smooth muscle actin (B) and desmin (C). The majority of the proliferating cells comprising the nodule correspond to VA cells, while lesser numbers of cells express VAD and V phenotypes. |
Involutional phase nodules also feature high cellularity, but the cells are smaller than those of the proliferative phase and tend to be aligned in the same direction (Figure 6.10B C). Ultrastructurally, these nodules are composed of myofibroblasts that are also connected by gap and adherens junctions. These intercellular junctions, however, seem to be less numerous than in proliferative phase nodules. The most striking difference with proliferative phase nodules is the increased amount of collagen that envelopes myofibroblasts. By immunoelectron microscopy, -smooth muscle actin is localized within bundles of microfilaments of myofibroblasts of the proliferative and involutional phase nodules.
Residual phase nodules are hypocellular and the slender and aligned cells are surrounded by thick bands of collagen, giving them a tendonlike appearance (Figure 6.10D). By ultrastructure, these nodules are composed of mature fibroblasts (Figure 6.12A), some containing discrete subplasmalemmal bundles of microfilaments without dense bodies. Occasional poorly-developed adherens-type junctions (Figure 6.12A and inset) connect the fibroblasts, but
P.140
gap junctions are no longer observed. The slender fibroblasts show smooth, contoured nuclei and are embedded in a dense collagenous matrix formed by thick bands of tightly packed collagen fibers. In conclusion, significant ultrastructural differences exist between proliferative, involutional, and residual phase nodules in Dupuytren's disease in relation to the cells, intercellular junctions, and composition of the extracellular matrix.
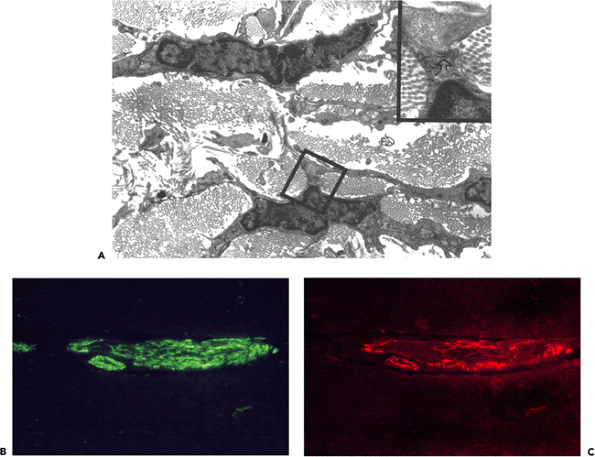 |
Figure 6.12 Dupuytren's disease: residual phase nodule. A. Transmission electron micrograph illustrating slender fibroblasts with smooth contoured nuclei embedded in a dense collagenous matrix and joined by poorly-differentiated junction (open arrow, inset) (uranyl acetate and lead citrate, 12,150; inset, 40,500). B. and C. Double immunofluorescent staining for -smooth muscle actin (B) and desmin (C). Cells comprising the residual phase nodule correspond to V cells. A few isolated cells express only VAD or VA phenotypes. |
When the collagenous matrix of Dupuytren's disease is analyzed by immunohistochemical techniques, proliferative phase nodules reveal a predominance of type III collagen, whereas in the residual fibroblastic phase, type I collagen predominates (103). Differences between proliferative and residual phase nodules are also defined in the vascularization. In proliferative phase nodules, capillaries are numerous and feature, ultrastructurally, large and prominent pericytes that display distinct smooth muscle differentiation; whereas in residual phase nodules, capillaries are few in number and are surrounded by small and inconspicuous pericytes that are devoid of a well-developed microfilamentous apparatus (26). Analogous to wound healing, the cicatrizing process within proliferative and involutional phase nodules is centripetal, being completed within residual phase nodules.
When immunohistochemical techniques are employed to study the cellular phases of Dupuytren's disease the
P.141
following results are obtained. Cells of the proliferative phase nodules always express vimentin, which is associated in approximately 80% of the cells with -smooth muscle actin (68) and in about 20 to 40% with desmin when double-labeling immunofluorescence techniques are performed (Figure 6.11B C). Rarely, isolated cells positive for vimentin, -smooth muscle actin, and smooth muscle myosin heavy chains with or without desmin are present [VA (D) M phenotype] (189). In involutional phase nodules, desmin-positive cells are less numerous or even absent, whereas -smooth muscle actin-positive cells are still present, albeit in lesser numbers. In residual phase nodules few or no -smooth muscle actin positive cells persist, and the remaining slender cells express solely vimentin (Figure 6.12B C). Accordingly, cells comprising the nodules of Dupuytren's disease express different cytoskeletal phenotypes: (a) phenotype V; (b) phenotype VAD; (c) phenotype VA; and (d) phenotype VD. In most proliferative phase nodules of Dupuytren's disease and also in the cellular areas of musculoaponeurotic fibromatoses, the number of VA cells considerably exceeds the number of VAD and VD cells (24). At the heavily collagenized interphase of involutional and residual nodules, the number of VAD and VD cells decreases progressively and is replaced by an almost pure population of V cells (24,26,257). Despite their heterogeneity in intermediate filament proteins and actin isoforms, myofibroblasts from Dupuytren's disease (69,256) express usually only nonmuscle myosins. Exceptionally, isolated cells expressing smooth muscle myosin heavy chains [VA (D) M phenotypes] are observed (189). In these tissues, the extracellular matrix around myofibroblasts is strongly stained with antibodies to fibronectin but not to laminin (256,260).
Other Quasi-Neoplastic Proliferative Conditions
A heterogeneous cytoskeletal composition is also observed in myofibroblasts of dermatofibromas, which reveal at least three cytoskeletal phenotypes: VA, VAD, and V cells, with a predominance of VA cells in cellular dermatofibromas and an almost exclusive composition of V cells in fibrous dermatofibromas (261). Whether myofibroblasts of the VA (D) M phenotype exist has not yet been determined. By ultrastructure, cellular dermatofibromas are composed of well-developed myofibroblasts, joined by gap and intermediate junctions, and admixed with variable numbers of fibroblasts and macrophages. Fibrous dermatofibromas, in contrast, are composed almost exclusively of fibroblasts and feature only small numbers of poorly developed myofibroblasts.
This heterogeneous cellular and cytoskeletal phenotypic composition of dermatofibromas permits one to distinguish them from dermatofibrosarcoma protuberans, which represents a pure fibroblastic neoplasm both at the ultrastructural level and with regard to the cytoskeletal immunophenotype (261). Furthermore, the heterogeneous cytoskeletal composition of dermatofibroma identifies this lesion definitively as a quasineoplastic reactive and proliferative condition, whereas dermatofibrosarcoma protuberans represents a fibroblastic neoplasm (261).
Nodular and proliferative fasciitis are predominantly composed of myofibroblasts with similar cytoskeletal phenotypes of VA and rare VAD cells, the latter being more prominent in the proliferative variant (Figure 6.13A C).
Infantile myofibromatosis reveals a predominance of VA cells with limited numbers of VAD cells. By ultrastructure, in contrast to other fibromatoses, smooth muscle differentiation appears to be more prominent than in conventional fibromatoses, although typical myofibroblasts are numerous, a feature that justifies the term infantile myofibromatosis. Furthermore, massive apoptosis has been documented in infantile myofibromatosis and is proposed as a putative mechanism of regression of this proliferative myofibroblastic lesion (262).
Stromal Response to Neoplasia
Legions of medical students have been taught that many invasive and metastatic carcinomas are characterized by hard consistency and retraction and are often fixed to adjacent tissues. Typical examples are invasive ductal mammary carcinomas, associated with skin and or nipple retraction (Figure 6.14A), annular stenosing colon carcinomas (Figure 6.14B), gastric linitis plastica, the so-called frozen pelvis in advanced gynecological carcinomas, the woody hard nodule of invasive prostatic carcinoma, and metastatic carcinoma in matted lymph nodes fixed to surrounding tissues and the overlying skin. The hard consistency and the retraction phenomena are due to the desmoplastic stromal reaction and contracting myofibroblasts.
Myofibroblasts are particularly numerous within the stroma of desmoplastic and retracted primary invasive and metastatic carcinomas (6,20,21,22,23,263), and the retraction associated with such carcinomas is attributed to the contractile forces generated by stromal myofibroblasts. Myofibroblasts are usually not observed in the stroma contiguous to in situ carcinomas (Figures 6.15A B, 6.16A B) (6,23), suggesting that invasion beyond the basal lamina is required to evoke a myofibroblastic stromal reaction. On occasion, stromal cells expressing -smooth muscle actin are observed around in situ ductal breast carcinoma (personal observation, Figure 6.15C D). By ultrastructure, the periductal stromal cells expressing -smooth muscle actin may disclose the typical morphologic features of myofibroblasts with well-developed stress fibers and fibronexus. This early myofibroblastic stromal reaction around ducts of in situ carcinomas is not uniform in a given case but may change from one duct to another (Figure 6.17A B). Myofibroblasts have also been described in squamous intra-epithelial lesions of the uterine cervix, close to the basal lamina, in increasing numbers and intensity of staining
P.142
P.143
from low-grade to high-grade variants, using immunohistochemical techniques (264).
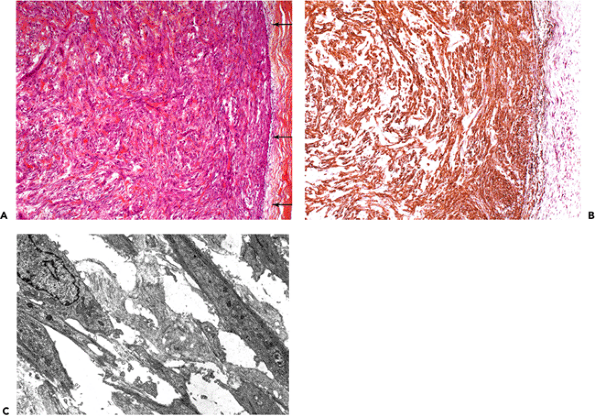 |
Figure 6.13 Nodular fasciitis of the forearm. A. Histology illustrating highly vascularized spindle cell proliferation, derived from the subcutaneous fascia (arrows) (hematoxylin-phloxine-saffron). B. The majority of the spindle cells and vascular smooth muscle cells express -smooth muscle actin (VA cells). C. By ultrastructure, most of the stromal cells within the nodule correspond to typical myofibroblasts. (Uranyl acetate and lead citrate, 6900.) |
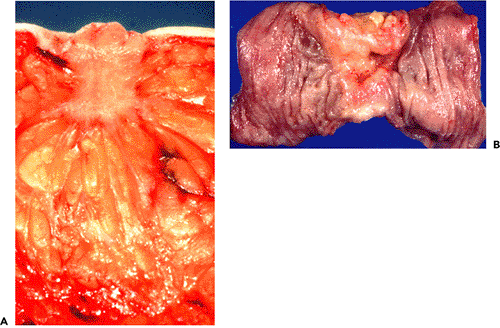 |
Figure 6.14 Gross appearance of infiltrating ductal carcinoma of the breast and of infiltrating colon carcinoma. A. Note irregular stellate shape of the carcinoma and retraction of the cut surface and the nipple. B. The colon carcinoma features annular stenosis. The carcinoma invaded the pericolic fibroadipose tissue. |
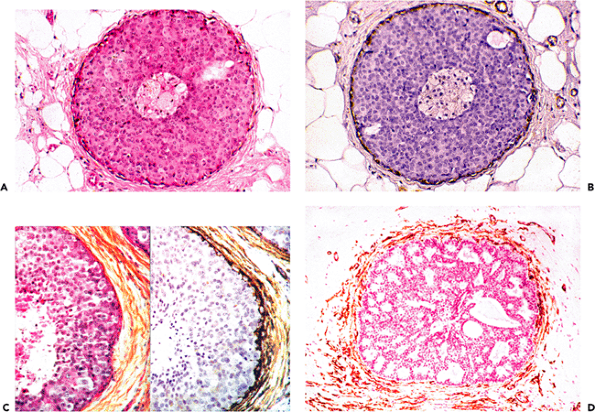 |
Figure 6.15 In situ breast carcinoma. A. and B. Step-sections of in situ cribriform carcinoma, disclosing continuous layer of myoepithelial cells as revealed by immunostaining for -smooth muscle actin. Periductal stromal cells lack immunostaining for -smooth muscle actin (B). C. Step-sections of comedocarcinoma: Numerous periductal stromal cells disclose significant staining for -smooth muscle actin. D. In situ cribriform carcinoma with numerous periductal stromal cells stained for -smooth muscle actin. (A and C left part: hematoxylin-phloxine-saffron.) |
Stromal cells with myofibroblastic features are notably absent or equivocally present within carcinomas lacking significant retraction and desmoplasia (Figure 6.18A C) (23).
Myofibroblasts are not uniformly distributed within desmoplastic carcinomas. When their spatial relation to other components of breast carcinomas is analyzed, they are most numerous within the young mesenchymal stroma, areas corresponding to early stromal invasion, or, more consistently, in the peripheral invasive cellular front of mammary carcinomas (Figures 6.19A F, 6.20A) (23). In the central sclerotic area of such neoplasms, myofibroblasts are poorly-developed or absent; this possibly is a reflection of apoptosis (Figures 6.19G H, 6.20B) (23). Similarly, myofibroblasts are numerous in the cellular, edematous, and poorly collagenized stroma of other invasive and metastatic carcinomas (23).
Three types of myofibroblastic stromal reactions are observed within infiltrating ductal mammary carcinomas: (a) precocious (Figure 6.19A B), myofibroblasts precede the carcinoma cells by some distance into adjacent tissue; (b) synchronous (Figure 6.19C D), myofibroblasts appear spatially among the carcinoma cells; and (c) late (Figure 6.19E F), myofibroblasts are identified central to the peripheral invasive cellular front of the carcinoma cells (23). These three types of myofibroblastic stromal reactions are observed in different areas of the invading front of most infiltrating ductal carcinomas of the breast, the synchronous stromal reaction being usually predominant (23). When the collagenous matrix is analyzed, increased amounts of type III collagen are present within the young mesenchyme, areas with numerous myofibroblasts. In contrast, type I collagen is most prominent within the central sclerotic zone of breast carcinomas (106), areas in which myofibroblasts are replaced by fibroblasts (Figure 6.19G H) (22,23).
P.144
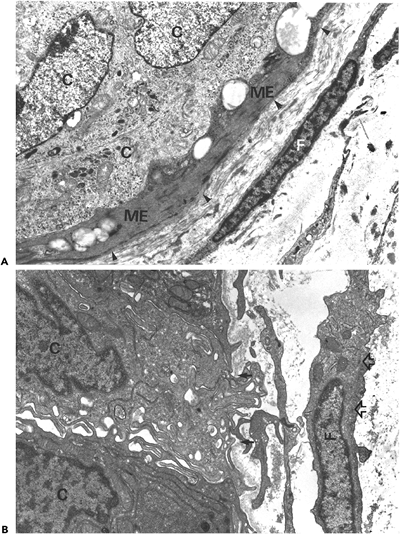 |
Figure 6.16 Ultrastructure of in situ ductal carcinoma of the breast. A. A continuous layer of myoepithelial cells (ME) and a continuous basal lamina (arrowheads) separate the carcinoma cells (C) from the surrounding stroma. The stromal fibroblast (F) discloses smooth cellular and nuclear contours; the cytoplasm is scant and devoid of bundles of microfilaments. B. Ultramicroinvasive ductal carcinoma. A carcinoma cell (C) protrudes with a cytoplasmic extension into the periductal stroma through a gap within the basal lamina (arrows). The periductal fibroblast reveals abundant cytoplasm and discloses aggregates of microfilaments with attenuated dense bodies (open arrows). (Uranyl acetate and lead citrate, A, 11,250; B, 13,500). (Source: Sch rch W, Lagac R, Seemayer TA. Myofibroblastic stromal reactions in retracted scirrhous carcinomas of the breast. Surg Gynec Oncol 1982;154:351 358. ) |
P.145
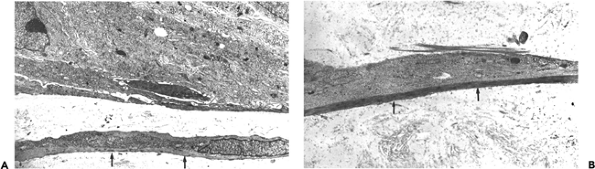 |
Figure 6.17 A. and B. Ultrastructure of in situ ductal carcinoma of the breast. In situ carcinoma with periductal myofibroblasts with well-developed stress fibers (arrows) (uranyl acetate and lead citrate; A, 7875; B, 12,500). |
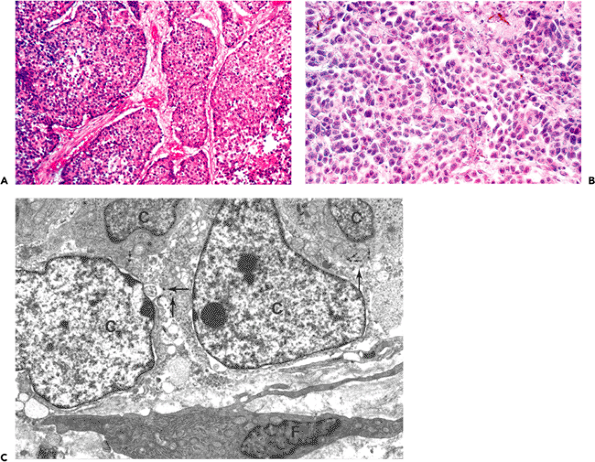 |
Figure 6.18 Oat-cell carcinoma of the lung. A. Histological aspect illustrating clusters of small neoplastic cells separated by small connective tissue septa (hematoxylin-phloxine-saffron). B. Stromal cells reveal no significant staining for -smooth muscle actin (avidin-biotin-complex-peroxidase). C. Transmission electron micrograph illustrating neoplastic cells (C) with scattered electron-dense neurosecretory-type granules (arrows) in close proximity to a fibroblast (F) with a smooth, contoured nucleus devoid of microfilaments (uranyl acetate and lead citrate, 10,300). |
P.146
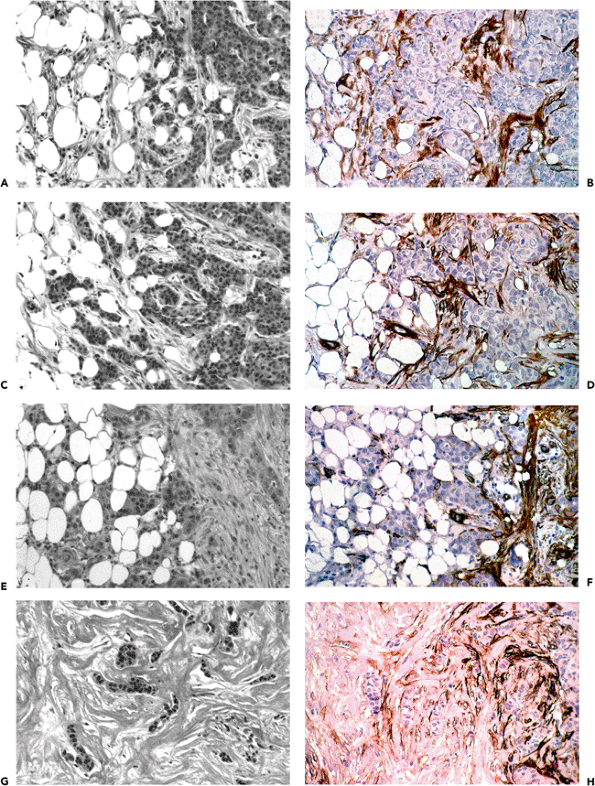 |
Figure 6.19 Ductal-infiltrating carcinoma of the breast with stromal desmoplasia, step-sections stained with hematoxylin-phloxine-saffron and with antibodies to -smooth muscle actin. A. and B. Precocious stromal reaction; stromal cells precede carcinoma cells by some distance into the adjacent fatty tissue. The majority of these stromal cells express -smooth muscle actin (B). C. and D. Synchronous stromal reaction; stromal cells are distributed amongst the carcinoma cells. Most of these stromal cells express -smooth muscle actin (D). E. and F. Late stromal reaction; stromal cells appear central to the peripheral invasive front of carcinoma cells and express -smooth muscle actin (F). G. and H. Central sclerotic area of ductal-infiltrating carcinoma. Clusters of carcinoma cells are surrounded by thick bands of collagen (G). At the border of the invasive cellular front of the carcinoma, a decrease of the immunostaining of the stromal cells toward the central area (left to right side) is observed. (A, C, E and G, hematoxylin-phloxine-saffron; B, D, F, and H, avidin-biotin-complex-peroxidase.) |
P.147
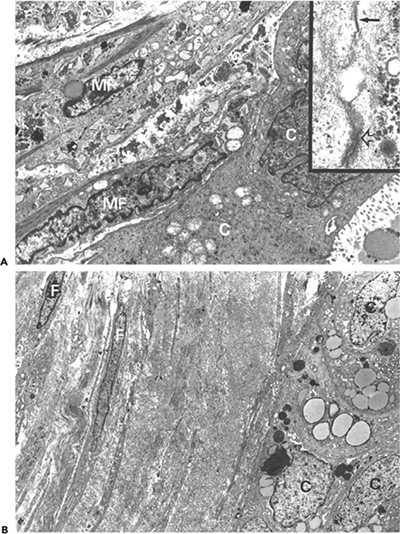 |
Figure 6.20 Ultrastructural aspect of ductal-infiltrating carcinoma of the breast. A. Peripheral invasive cellular front revealing numerous typical stromal myofibroblasts (MF) with notched nuclei and bundles of cytoplasmic microfilaments with dense bodies around neoplastic cells (C) adjacent to an acinus, which is in the lower right corner. Inset: Stromal myofibroblasts are joined by gap (arrow) and intermediate junctions (open arrow). (Source: Sch rch W, Seemayer TA, Lagac R, Gabbiani G. The intermediate filament cytoskeleton of myofibroblasts. Virchows Arch A 1984;403:323 336. ) B. Central sclerotic area illustrating stromal cells (F) with smooth contoured nuclei devoid of abundant cytoplasmic microfilaments and separated by thick bands of mature collagen around clusters of carcinoma cells(C). (Source:Sch rch W, Seemayer TA, Lagac R. Stromal myofibroblasts in primary invasive and metastatic carcinomas. Virchows Arch A 1981;391:125 139. ) (Uranyl acetate and lead citrate, A, 8400; inset, 60,000; B, 5000.) |
P.148
Many pulmonary carcinomas, especially peripheral adenocarcinomas, are associated with some degree of scarring and are often associated with pleural retraction. If this process is pronounced, the term scar carcinoma is applied to these neoplasms. In 1962, Carroll (265) reported that the presence of elastic fibers and anthracotic pigment in scars suggested that they had been present prior to the development of the neoplasm. The more recent literature suggests that scarring represents a desmoplastic stromal reaction in response to neoplastic invasion rather than a preexistent condition. In favor of this latter interpretation is the presence of increased amounts of type III collagen within
P.149
pulmonary carcinomas with marked scarring (266), as is seen in early invasive zones of mammary carcinoma (104). In addition, the majority of stromal cells in scar carcinomas of the lung reveal ultrastructural features of myofibroblasts (267), suggesting that pulmonary carcinomas with scarring are neoplasms with a desmoplastic stromal reaction, analogous to many invasive and metastatic carcinomas elsewhere.
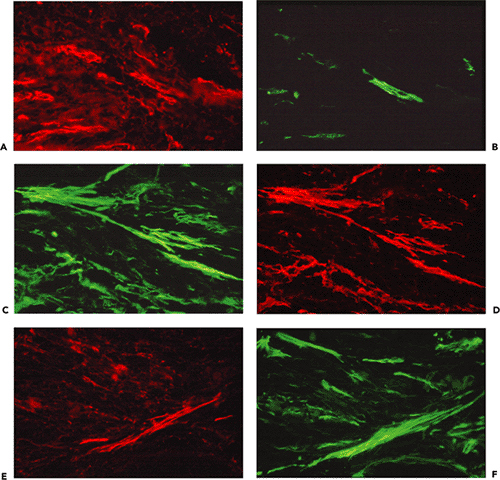 |
Figure 6.21 Step-sections of peripheral invasive cellular front of ductal breast carcinoma revealing V, VA, VAD, and VA (D) M myofibroblasts. A. and B. Double immunofluorescent staining for -smooth muscle actin (A) and desmin (B). Stromal cells expressing -smooth muscle actin (VA cells) are more numerous than those expressing -smooth muscle actin and desmin (VAD cells). C. and D. Double immunofluorescent staining for -smooth muscle actin (C) and smooth muscle myosin heavy chain (D). Stromal cells expressing -smooth muscle actin (VA cells) are slightly more numerous than those expressing smooth muscle myosin heavy chain [VA (D) M cells]. E. and F. Double immunofluorescent staining for desmin (E) and myosin heavy chain (F). Stromal cells expressing smooth muscle myosin heavy chain (VM cells) are far more numerous than those that express desmin [VA (D) M cells]. In conclusion, the peripheral invasive front of ductal breast carcinomas contains predominantly VA cells, followed by VA (D) M cells. |
Analysis of cytoskeletal proteins, including intermediate filaments, actin isoforms, and smooth muscle myosin heavy chains reveals phenotypic heterogeneity of stromal cells in invasive and metastatic carcinomas. Areas with numerous myofibroblasts, corresponding to early stromal invasion of breast carcinomas, contain a predominance of VA cells admixed with variable numbers of VAD, VA (D) M, and V cells (Figure 6.21A F), suggesting that certain stromal cells undergo a form of cytodifferentiation not too dissimilar from smooth muscle metaplasia (VA (D) M cells). In contrast, sclerotic areas disclose numerous V cells with occasional VA cells. No VAD and VA (D) M cells are observed (results not shown).
Myofibroblasts have also been described in sarcomas where they generally constitute a small fraction of the cell population (268,269,270). They were identified in all cases of malignant fibrous histiocytomas and well-differentiated sclerosing liposarcomas (270). Though most numerous in areas of desmoplasia, in no instance did myofibroblasts constitute the dominant cellular constituent of either neoplasm (Figure 6.22) (270). Myofibroblasts have been identified with lesser frequency and in smaller numbers in fibrosarcoma, synovial sarcoma, malignant hemangiopericytoma, and neuroblastoma. No myofibroblasts were observed in a wide assortment of diverse sarcomas in which desmoplasia was not a feature.
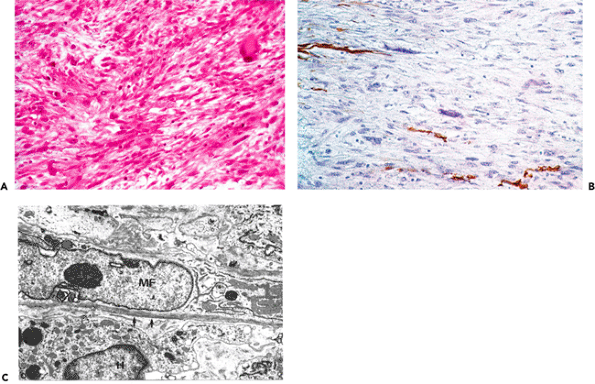 |
Figure 6.22 Malignant fibrous histiocytoma (storiform-pleomorphic type). A. Histological aspect illustrating spindle cell tumor with storiform pattern and isolated pleomorphic cells (hematoxylin-phloxine-saffron). B. Few spindle cells disclose immunostaining for -smooth muscle actin (avidin-biotin-complex-peroxidase). C. Transmission electron micrograph illustrating a typical myofibroblast (MF) with cytoplasmic bundle of microfilaments; the cell is partly enveloped by a basal lamina (arrows) (uranyl acetate and lead citrate, 25,000). |
Myofibroblasts have been identified in nodular sclerosing Hodgkin's disease at the nodule-stromal interphase, which is usually heavily collagenized (271). These areas contain numerous VA and V cells with very occasional VAD cells (Figure 6.23). Whether this contributes to the relatively favorable prognosis of this variant of Hodgkin's disease is an open question.
Neoplasms of Myofibroblasts: Benign and Malignant
Finally, several reports describe myofibroblastic neoplasms. In our opinion, neoplastic transformation of the myofibroblast, in the extreme is possible (272) but certainly remains an uncommon event. The plethora of articles related to this matter, particularly in the mid- and late-1990s, stems from the criteria employed to define this cell. The
P.150
P.151
myofibroblast is presently defined solely at the ultrastructural level; its definition at the light microscopic and immunohistochemical levels is less precise and, on occasion, imprecise. A significant number of reports describe myofibroblastic neoplasms; some were considered as sarcomas (273,274,275,276,277,278,279), and many more were described as benign myofibroblastomas or tumors of similar character with an assortment of designations (280,281,282,283,284,285,286,287,288,289,290,291).
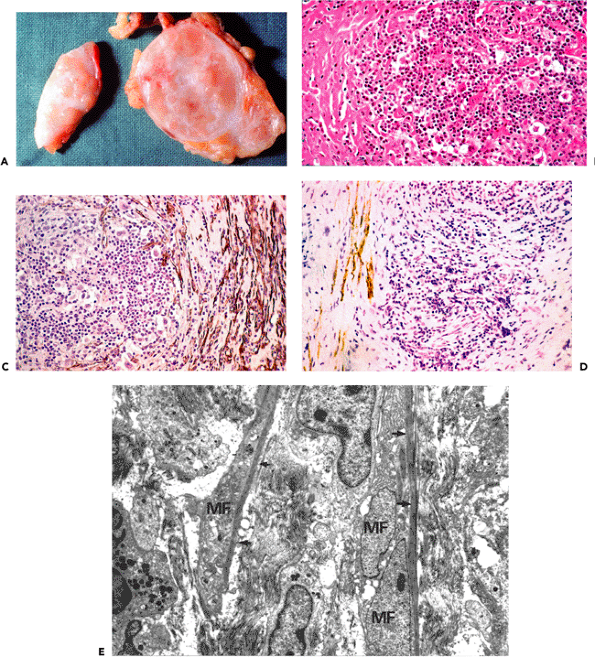 |
Figure 6.23 Nodular sclerosing Hodgkin's disease. A. Gross aspect of cut surface of lymph node demonstrating nodules surrounded by thick connective tissue septa (lymph node cirrhosis). (Courtesy of Dr. Roger Gareau, Department of Pathology, H tel-Dieu Hospital, University of Montreal, Montreal, Quebec, Canada.) B. Histologic aspect illustrating nodules of atypical lymphoid nodules with some lacunar cells. The nodules are enveloped by dense collagenous containing numerous spindle cells (hematoxylin-phloxine-saffron). C. Several spindle cells around the nodule express -smooth muscle actin (VA cells). D. Few spindle cells express desmin (VAD cells). All internodular stromal cells express antivimentin (results not shown). E. Transmission electron micrograph from internodular stroma illustrating numerous typical myofibroblasts (MF) with bundles of cytoplasmic microfilaments and dense bodies (arrows). (Uranyl acetate and lead citrate, 5000.) |
The benign myofibroblastic proliferations are generally well-circumscribed lesions, contrary to the poorly circumscribed and often infiltrating quality of reactive and quasineoplastic proliferative conditions; for example, fibromatoses, nodular and proliferative fasciitis, and proliferative myositis, lesions replete with myofibroblasts. Although thought to be composed of myofibroblasts, most of the benign myofibroblastomas were not evaluated ultrastructurally; in the few cases for which this technique was employed, typical myofibroblasts were not found. For similar reasons one might cast a jaundiced eye on the presence of myofibroblasts in mammary myofibroblastomas (289,290), the palisaded myofibroblastoma, the intranodal hemorrhagic spindle-cell tumors with amianthoid fibers of lymph nodes (288,291), soft tissue myofibroblastomas (284), angiomyofibroblastomas of the vulva (281,282), angiomyofibroblastoma-like tumors of the male genital tract (285), meningeal myofibroblastomas (287), and pulmonary myofibroblastic tumors (280). Immunohistochemically, the proliferating cells composing the so-called myofibroblastomas and related neoplasms disclose heterogeneous cytoskeletal phenotypes, such as positive reaction for -smooth muscle actin and absence of reactivity for desmin in the palisaded myofibroblastoma (291) and intranodal hemorrhagic spindle-cell tumors of lymph nodes (288) and staining for desmin associated with a negative reaction for -smooth muscle actin in angiomyofibroblastoma of the vulva (281). The so-called myofibroblastomas and all other related neoplasms most likely represent myogenic stromal tumors, a designation proposed by B gin (292), possibly derived from myogenic stromal cells that have variable degrees of smooth muscle differentiation (rather than myofibroblastic neoplasms) because myofibroblasts, using strict ultrastructural criteria, either were not identified or were rare (Figure 6.24).
Sarcomas composed entirely or partially of cells that disclose some degree of morphologic or immunohistochemical features of myofibroblasts but lack the typical ultrastructural traits of myofibroblasts could well belong to the group of myogenic sarcomas (63). One has to remember that for a cell to be classified as a myofibroblast it need not express -smooth muscle actin (134). In fact, whether or not a cell expresses -smooth muscle actin has no bearing on whether or not it is a myofibroblast. In our opinion, while myofibroblastic sarcomas may exist, they are rare and their identification requires electron microscopy (57,58). Finally, to conclude this controversial issue, it might be well to cite Juan Rosai (59):
Cells with myofibroblastic (myoid) features can be found in a large number of benign and malignant soft tissue lesions, which means that we are in danger of creating a waste-basket category, just as large if not larger than that of malignant fibrous histiocytoma. Therefore, if there is to be a category of myofibroblastic tumors, it would be wise to reserve it for lesions that are composed almost entirely of cells having the hybrid features of myofibroblasts and which do not fit the criteria of already established entities.
Now, a few words concerning inflammatory myofibroblastic tumor (IMT). In the 2002 World Health Organization (WHO) classification of soft tissue tumors, IMT is presented as a clinical/pathologic entity, albeit one that is genetically heterogeneous. Its synonyms are numerous: plasma cell granuloma, plasma cell pseudotumor, inflammatory myofibrohistiocytic proliferation, omental mesenteric myxoid hamartoma, inflammatory pseudotumor, and inflammatory fibrosarcoma. The entity appears to have emerged in a manner reminiscent of malignant fibrous histiocytoma (293). That this tumor discloses predominant myofibroblastic differentiation is questionable. Classically, these tumors present in the lung, mesentery, and omentum of children or adolescents and may be accompanied by fever, weight loss, fatigue, anemia, thrombocytosis, polyclonal hyperglobulinemia, and an elevated erythrocyte sedimentation rate (294). Histologic sections reveal a polymorphous mixture of cells, including spindle cells, plasma cells, lymphocytes, eosinophils, and occasional ganglion-like cells. The spindle cells in 50% of the tumors express cytoplasmic ALK protein, and this correlates with rearrangments of the ALK receptor tyrosine kinase gene at 2p23, as detected by fluorescent in situ hybridization (FISH). This genetic aberration is most commonly seen in pediatric IMT tumors but is not specific for the condition. In addition to anaplastic lymphoma kinase (ALK) expression, p80 is consistently expressed in IMT (295). Most of the tumors are biologically benign; however, up to 25% of the nonpulmonary tumors may recur and, in rare instances, the tumor may metastasize.
Thus, in our opinion, most of the described myofibroblastic sarcomas described are not unequivocally composed of myofibroblasts. That said, we have to admit that a few conditions (particularly malignant neoplasms) composed of spindle cells disclosing ultrastructural features of myofibroblasts have been described, but they are rare (57,296). As for IMT, whether or not this is a specific entity in which the myofibroblast is the principal cell is an open question.
Multiple Origins of the Myofibroblast
Considering the many conditions in which myofibroblasts occur, their heterogeneous cytoskeletal composition, and
P.152
the various functions attributed to them, it seems difficult at first glance to assume a common origin for these cells. In 1867, Cohnheim (297) proposed the vascular theory, which states that leukocytes are transformed into fibroblasts during the process of wound healing. Several subsequent studies, however, provided evidence that granulation tissue fibroblasts arise rather from local connective tissue cells (298,299,300,301). As we shall see below, circulating leukocytes and resident tissue fibroblasts are now recognized as precursors to the myofibroblasts found in granulation tissue.
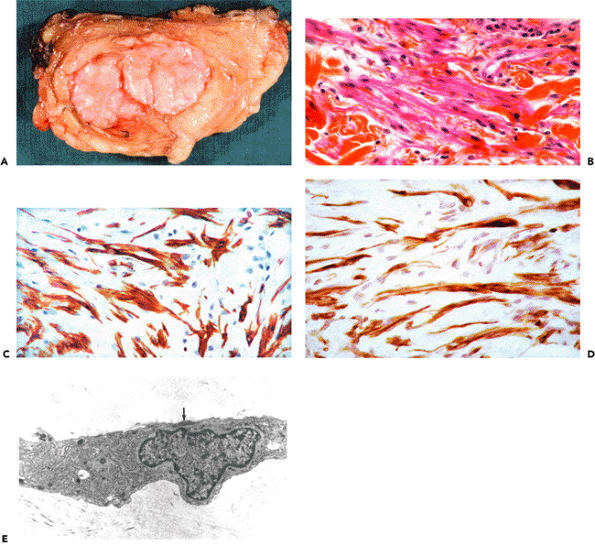 |
Figure 6.24 Myofibroblastoma of a male breast. A. Gross appearance disclosing well-circumscribed bilobar tumor. B. Histologic aspect illustrating spindle cells with acidophilic cytoplasm and bland nuclei (hematoxylin-phloxine-saffron). C. and D. Intense immunostaining for -smooth muscle actin and desmin. E. Ultrastructural aspect disclosing discrete smooth muscle differentiation. Short bundle of microfilaments and segment of basal lamina (arrow). (Uranyl acetate and lead citrate, 17,750.) |
Amongst connective tissue cells that could transform into myofibroblasts, any mesenchymal cell is a potential candidate: foremost is the fibroblast, followed by the pericyte and the smooth muscle cell (302). With the accumulated knowledge of cytoskeletal proteins and actin isoforms in these three cell types, both in vivo and in vitro, all of these cells could be considered as possible progenitors of myofibroblasts.
Granulation tissue myofibroblasts are principally derived from local fibroblasts (25,260,303). Within experimental and human granulation tissues, myofibroblasts temporarily express a marker of smooth muscle differentiation, -smooth muscle actin, which disappears after wound closure (25). This suggests that differentiation of myofibroblasts toward smooth muscle cells is only partial, at least during normal wound healing, because myofibroblasts
P.153
in this condition never express desmin or smooth muscle myosin heavy chain isoforms.
Recently, the cytoskeletal features of myofibroblasts during wound healing, Dupuytren's disease, and the stroma of mammary carcinomas were investigated. In these three conditions, myofibroblasts disclosed a progressive differentiation toward the smooth muscle phenotype (189). Whereas myofibroblasts during wound healing express only -smooth muscle actin, myofibroblasts in Dupuytren's disease express smooth muscle myosin heavy chains, at least in some cases. An important proportion of myofibroblasts within the stroma of all cases of mammary carcinomas express, in addition to -smooth muscle actin, desmin and smooth muscle myosin heavy chain isoforms. This suggests that fibroblastic cells are capable of proceeding well along the lines of smooth muscle cell differentiation. However myofibroblasts have not been shown to express smoothelin (304,305,306), a terminal smooth muscle cell differentiation marker, in any of the pathologic states examined (305). Hence, smoothelin expression may be used as a discrimination marker between the two cells.
Ultrastructural data provide evidence that during pathologic or culture conditions, fibroblasts and smooth muscle cells acquire morphologic features resembling myofibroblasts (87,307,308,309,310,311), suggesting that both cell types might be progenitors of myofibroblasts. Indeed, an extensive study on the modulation of mesenchymal cells within the mammary gland stroma when placed in culture in a microenvironment mimicking conditions observed in vivo indicates that although most myofibroblasts are derived from fibroblasts, a certain proportion are derived from vascular smooth muscle cells and a lesser proportion from pericytes (312). With the caveat that it is difficult to extrapolate in vitro data to in vivo situations, this work supports the concept of a heterogeneous origin of myofibroblastic cells.
A vascular origin of the myofibroblast was also proposed on the basis of morphologic observations. It was suggested that desmin-positive cells migrate from the wall of vessels to the tissue (257). A possible source of myofibroblasts expressing vimentin and desmin also are the stromal cells of various organs positive for desmin but negative for -smooth muscle actin (67,183,184,313). The possibility that myofibroblasts arise from specialized mesenchymal cells of certain organs has found a convincing confirmation in recent years. An abundant clinical and experimental literature has shown that, during the onset of experimental and human hepatic fibrosis and cirrhosis, perisinusoidal stellate cells of the liver are the most likely source of myofibroblastic cells (32,314,315,316). The conditions facilitating the modulation of perisinusoidal stellate cells into myofibroblasts have been studied, and extracellular matrix components and cytokines have been suggested as possible initiators (317,318,319,320,149). Similarly, glomerular mesangial cells have been shown to acquire myofibroblastic features, including the expression of -smooth muscle actin and collagen, in several experimental and human pathologic situations (33,229,321,322). Lung septal fibroblasts, which normally possess contractile features without expressing -smooth muscle actin (323), can be induced to express this protein and collagen type I mRNA upon pathologic stimuli, such as bleomycin treatment (324,29).
Recently, advances have been made demonstrating that myofibroblasts can originate from circulating precursors and also be the product of epithelial-mesenchymal transitions. Buccala et al. (36) have identified a leukocyte subpopulation, named fibrocyte, with fibroblast-like properties. Peripheral blood fibrocytes can rapidly enter the site of injury at the same time as circulating inflammatory cells. It has been suggested that circulating fibrocytes may represent an important source of myofibroblasts during healing of extensive burn wounds, where it may be difficult for fibroblasts to migrate from the wound edge (37). This study has also shown that fibrocyte development is systematically elevated in burn patients. Furthermore, TGF- 1, which is elevated in the serum of burn patients (325), stimulates the modulation of peripheral blood mononuclear cells into collagen-producing cells, underlying the well-known role of this cytokine in the differentiation of the myofibroblast (326,27). It has been also shown that bone marrow derived myofibroblasts contribute to the stroma reaction, at least in experimental situations (325). Another location in which fibrocytes contribute to myofibroblast population is the bronchial submucosa during the development of asthma (327). Epithelial-mesenchymal transition plays an important role in myofibroblast accumulation taking place in kidney interstitial fibrosis, the source of myofibroblasts being tubular epithelial cells, (34) and in dialysis-induced peritoneal fibrosis, the source of myofibroblasts being mesothelial cells (35).
Thus, it appears that several cells, including fibroblasts, vascular smooth muscle cells, pericytes, perisinusoidal stellate cells in the liver, renal tubular epithelial cells, mesangial cells, bloodborne cells (fibrocytes), and mesothelial cells, can modulate (upon appropriate stimulation) into a myofibroblastic phenotype. It should be stressed, however, that the major source of myofibroblasts in whatever setting they appear is the resident fibroblast.
Mechanisms of Myofibroblast Regression
Granulation tissue formation involves the replication and migration of fibroblasts from normal tissues to the area of inflammation and the modulation of at least a proportion of them to the myofibroblastic phenotype. Angiogenesis takes place in a coordinated way, and granulation tissue acquires its typical features. When the wound closes, a gradual evolution toward scar tissue takes place that involves
P.154
the disappearance of vascular cells and myofibroblasts with a proportional increase of extracellular matrix components. This phenomenon, which ends with the establishment of a scar, is more or less rapid according to the species, the location of granulation tissue, and the type of inflammation (7). When granulation tissue cells are not eliminated, there is the development of pathologic scarring (i.e., hypertrophic scars and keloids), which are distinct clinical and pathologic conditions (232), both characterized by a relative high degree of cellularity.
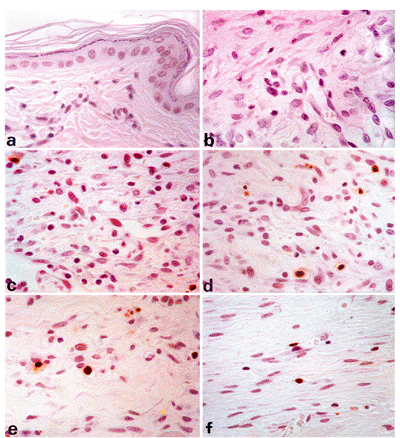 |
Figure 6.25 Identification of apoptotic cells in rat tissues by in situ end labeling of fragmented DNA. A. Normal rat skin, no apoptotic cells are detected. B. Twelve-day-old wound tissue. C. Sixteen-day-old wound tissue. D. Twenty-day-old wound tissue. E. Twenty-five-day-old wound tissue. F. Thirty-day-old wound tissue. At 12 days, when -smooth muscle actin expression is maximal, there is no nuclear staining for apoptotic cells; after this, the number of labeled cells increases, with a maximum at 20 days (D). Thereafter (E and F), the number of labeled cells decreases. (A F, 1000.) |
Recently, using several morphologic and biochemical techniques, it has been shown that the reduction in cell number (myofibroblasts and vascular cells) observed during the transition between granulation tissue and scar formation is achieved to a great extent through apoptosis (Figure 6.25) (31); whether the lack of apoptosis plays a role in the establishment of hypertrophic scar and keloid remains to be explored. It appears that apoptosis of granulation tissue cells takes place essentially after wound closure and affects myofibroblasts and vascular cells over a
P.155
period of time, rather than occurring as a single and massive wave of cellular apoptosis. This observation is in line with the gradual resorption of granulation tissue after wound closure and with the observation that dead cells are digested by macrophages and surrounding cells. It appears that granulation tissue cell apoptosis can be accelerated significantly by the application of a viable cutaneous flap (328). This observation underlines the importance of cell communication between normal connective tissue and granulation tissue. These reports suggest that, at least during normal wound healing, the process of myofibroblast differentiation generally ends with cell death; thus, myofibroblasts can be considered terminally differentiated cells.
The question that remains to be answered is the stimulus that leads to apoptosis during wound healing. The loss of mechanical stress seems to be an important signal for differentiated myofibroblasts to de-differentiate and/or disappear. At the end of normal wound healing the extracellular matrix is reconstituted and assumes the mechanical load, thereby releasing embedded myofibroblasts from stress. Myofibroblast apoptosis has been induced by the stress-release of wound granulation tissue after removal of a flap coverage with splinting characteristics in vivo (329) and by the relaxation of attached collagen gels (330,331) in vitro. Fibroblasts in mechanically unrestrained floating versus anchored collagen matrices also show differences in cell proliferation and DNA synthesis. After contraction of floating collagen matrices there is a marked decline in DNA synthesis; the cell cycle becomes arrested and cell regression begins. In contrast, fibroblastic cells in anchored matrices continue to proliferate and to synthesize DNA.
Recently gene products regulating cell death have been identified (332,333,334,335,336,337). In fibroblasts, the c-myc protein (338) and interleukin-1 converting enzyme, the mammalian homologue of the Caenorhabditis elegans gene ced-3 (339), have been shown to induce apoptosis. In turn, it has been shown that the bcl-2 protein is capable of blocking apoptosis (340); however, fibroblasts lack bcl-2 expression, as assessed by antibody staining. A possible mechanism for apoptosis induction could be via the direct action and/or withdrawal of cytokines or growth factors (341,342,343). Several factors have been shown to increase the rate of wound healing, including platelet-derived growth factor (PDGF) (344), TGF- (344,345,346), and tumor necrosis factor (TNF) (347). These factors may be present in the normal healing wound, released by platelets and inflammatory cells (348,349). It is probable that, as the wound heals and resolves, there is a decrease in the level of these factors. A possible explanation for the death of at least a subpopulation of myofibroblasts and vascular cells could be that they are growth-factor dependent. Alternatively, factors selectively causing the death of myofibroblasts and vascular cells might be liberated after epithelialization has been completed. Additional work is necessary to identify these hypothetical factors, but it appears that apoptosis is the mechanism through which vascular and myofibroblastic cells are gradually eliminated from normally healing granulation tissue.
Concluding Remarks
For this third edition, every section has been updated. In addition, several new topics have been added to reflect recent developments; additional photographs have been submitted; the reference list has been expanded.
Since the 1971 discovery of the myofibroblast in granulating wounds, one cannot help but be fascinated with the subject as the body of knowledge related to this pivotal cell expands, largely through the contributions of cellular and molecular biology.
It would appear that following induction of a large skin wound, resting fibroblasts are triggered through the effects of mechanical forces and possibly yet unknown cytokines released at the wound site to assume a protomyofibroblastic phenotype characterized by the presence of stress fibers that contain cytoplasmic actin isoforms. These cells continue to modulate and eventually assume a myofibroblastic phenotype characterized by -smooth muscle actin incorporation into stress fibers and the formation of specialized supermature focal adhesions. This process is regulated by TGF- 1 and ED-A cellular fibronectin and results in connective tissue remodeling with an increase of extracellular matrix synthesis, collagen type III in particular, and tissue retraction (i.e., wound contraction). Recent studies strongly indicate that the forces generated by myofibroblast stress fibers produce isometric tension; this is different from the reversible contraction taking place in smooth muscle cells and involves the Rho/Rho-kinase pathway, as well as regulated activity of myosin phosphatase.
As wound healing approachs completion, genes that encode for apoptotic proteins are expressed to initiate myofibroblastic cell death; the formerly cellular wound is then converted into a poorly cellular scar. Commensurate with this, there is a shift from collagen type III to collagen type I gene expression and synthesis, resulting in the deposition of type I collagen that provides strength to the developing scar. Furthermore, cytokines that stimulate extracellular matrix synthesis early on are repressed once wound closure is completed and a functional basement membrane has been synthesized; this suggests the existence of a feedback loop (350). It is likely that deviations from this finely orchestrated process contribute to the development of hypertrophic scars and keloids.
Regarding the diverse assortment of quasineoplastic myofibroblastic proliferative processes, the cellular/molecular mechanisms central to their pathogenesis remain essentially unexplored.
P.156
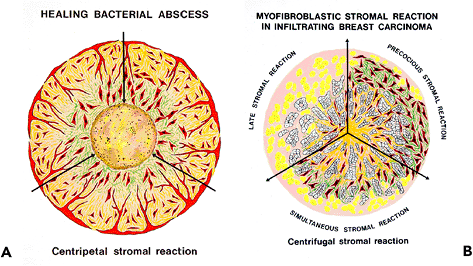 |
Figure 6.26 Schematic illustration of the stromal reaction. A. In a healing bacterial abscess, the cicatrizing layer is at the periphery, and the two layers containing myofibroblasts (exudativo-productive and exudative, respectively) are developing toward the center. B. In infiltrating ductal breast carcinoma, the cicatrizing area is in the center and myofibroblasts are disposed variably in the peripheral invasive cellular front of the carcinoma; precocious (preceding the invasive carcinoma cells), simultaneous (amongst the invasive carcinoma cells), and late (following the invasive carcinoma cells). In normal wound healing the stromal reaction is centripetal; whereas in invasive ductal breast carcinomas, the stromal reaction is centrifugal, indicating that cancers are wounds that do not heal. |
Turning to the myofibroblastic response associated with diverse invasive and metastatic carcinomas, it was originally proposed that this represented an expression of host response to the cancer. The hypothesis appears valid today, although one could posit whether this is beneficial since many of these cancers, despite the attending desmoplasia, continue to exact lives. Yet, death, in these settings, stems largely from the ability of the neoplastic cell to enter vascular channels and disseminate. It remains possible that these myofibroblasts, while affecting contraction and elaborating collagens and other extracellular matrix components, also release enzymes that permit tissue and vascular invasion. Recently, the mechanisms regulating the cross talk between tumor cells and stroma myofibroblasts have started to be clarified (351). Thus, it appears that the concomitant production of growth factors and/or cytokines (such as TGF- , hepatocyte growth factor, or stromal cell derived factor-1) and synthesis of extracellular matrix components (such as tenascin) by stromal myofibroblasts stimulates the invasive activity of malignant epithelial cells (352,353,354). If one considers that during development connective tissue remodeling plays an important role in epithelial morphogenesis (353), it is possible to conceive that cross talk between stroma and epithelium regulates both physiologic and pathologic epithelial organization. Clearly, future studies of human cancer should focus not only on the neoplastic cell, but also on the regulation of extracellular matrix synthesis and the cell-to-extracellular matrix interactions of tumors (i.e., the stroma).
Twenty-four years ago (22), we proposed that similarities might exist between the process of wound healing and the stromal response to neoplastic invasion. This assumption may also be extended to quasineoplastic proliferative conditions (e.g., Dupuytren's disease). During normal wound healing and within nodules of Dupuytren's disease and possibly other quasineoplastic proliferations, the myofibroblastic/fibroblastic reaction appears to be centripetal (Figure 6.26A), whereas within neoplastic invasion this reaction is centrifugal (Figure 6.26B), indicating that cancers are wounds that do not heal (355). The underlying cellular/molecular mechanisms explaining these fundamental differences, including the presence, delay, or absence of apoptosis, remain to be explored.
Finally, we conclude, as in the 1997 edition, with a most intriguing report uncovered in a literature search of TGF- . It would appear that fetal skin wounds in a murine model heal without scarring; such wounds, apart from that contained in platelets, are devoid of TGF- (356). Once again, one is reminded of the lessons to be learned by study of the events of early life.
Acknowledgments
This work was supported in part by the Cancer Research Society Inc., Montreal, Canada, the Swiss National Science Foundation (Grant No. #31-61.336.00 to GG and 3100A0-102150/1 to BH), and the Macdonald Stewart Foundation, Montreal, whose benefactors, Mrs. Liliane Stewart and the late David M. Stewart, have generously supported the Department of Pathology of the H tel-Dieu Hospital of Montreal over many years. We thank Mr. Som Chatterjee and Ms. Myrielle Vermette for skillful technical assistance and Mr. Jean-Jacques Dufour for the photographic work.
P.157
References
1. Gabbiani G, Ryan GB, Majno G. Presence of modified fibroblasts in granulation tissue and their possible role in wound contraction. Experientia 1971;27:549 550.
2. Majno G, Gabbiani G, Hirschel BJ, Ryan GB, Statkov PR. Contraction of granulation tissue in vitro: similarity to smooth muscle. Science 1971;173:548 550.
3. Gabbiani G, Hirschel BJ, Ryan GB, Statkov PR, Majno G. Granulation tissue as a contractile organ. A study of structure and function. J Exp Med 1972;135:719 734.
4. Hirschel BJ, Gabbiani G, Ryan GB, Majno G. Fibroblasts of granulation tissue: immunofluorescent staining with antismooth muscle serum. Proc Soc Exp Biol Med 1971;138:466 469.
5. Gabbiani G, Ryan GB, Lamelin JP, et al. Human smooth muscle autoantibody. Its identification as antiactin antibody and a study of its binding to nonmuscular cells. Am J Pathol 1973;72:473 488.
6. Seemayer TA, Lagac R, Sch rch W, Thelmo WL. The myofibroblast: biologic pathologic, and theoretical considerations. Pathol Annu 1980;15(pt 1):443 470.
7. Skalli O, Gabbiani G. The biology of the myofibroblast relationship to wound contraction and fibrocontractive diseases. In: Clark RAF, Henson PM, eds. The Molecular and Cellular Biology of Wound Repair. New York: Plenum Press; 1988:373 402.
8. Schmitt-Gr ff A, Desmouli re A, Gabbiani G. Heterogeneity of myofibroblast phenotypic features: an example of fibroblastic cell plasticity. Virchows Arch 1994;425:3 24.
9. Majno G. The story of the myofibroblasts. Am J Surg Pathol 1979;3:535 542.
10. Majno G. The Healing Hand: Man and Wound in the Ancient World. Cambridge: Harvard University Press, 1975.
11. Carrel A, Hartmann A. Cicatrization of wounds. I. The relation between the size of the wound and the rate of its cicatrization. J Exp Med 1916;24:429 450.
12. Abercrombie M, Flint MH, James DW. Wound contraction in relation to collagen formation in scorbutic guinea pigs. J Embryol Exp Morphol 1956;4:167 175.
13. Hoffmann-Berling H. Adenosintriphosphat als betriebsstoff von zellbewegungen. Biochim Biophys Acta 1954;14:182 194.
14. Majno G, Shea SM, Leventhal M. Endothelial contraction induced by histamine-type mediators: an electron microscopic study. J Cell Biol 1969;42(3):647 672.
15. Gabbiani G. Curr Contents: Citation Classics 1988;31:16.
16. Gabbiani G, Majno G. Dupuytren's contracture: fibroblast contraction? An ultrastructural study. Am J Pathol 1972;66:131 146.
17. Ryan GB, Cliff WJ, Gabbiani G, et al. Myofibroblasts in human granulation tissue. Hum Pathol 1974;5:55 67.
18. Gabbiani G, Chaponnier C, H ttner I. Cytoplasmic filaments and gap junctions in epithelial cells and myofibroblasts during wound healing. J Cell Biol 1978;76:561 568.
19. Singer II, Kawka DW, Kazazis DM, Clark RA. In vivo co-distribution of fibronectin and actin fibers in granulation tissue: immunofluorescence and electron microscope studies of the fibronexus at the myofibroblast surface. J Cell Biol 1984;98:2091 2106.
20. Tremblay G. Stromal aspects of breast carcinoma. Exp Mol Pathol 1979;31:248 260.
21. Seemayer TA, Lagac R, Sch rch W, Tremblay G. Myofibroblasts in the stroma of invasive and metastatic carcinoma: a possible host response to neoplasia. Am J Surg Pathol 1979;3:525 533.
22. Sch rch W, Seemayer TA, Lagac R. Stromal myofibroblasts in primary invasive and metastatic carcinomas. A combined immunological, light and electron microscopic study. Virchows Arch A Pathol Anat Histopathol 1981;391:125 139.
23. Sch rch W, Lagac R, Seemayer TA. Myofibroblastic stromal reaction in retracted scirrhous carcinoma of the breast. Surg Gynecol Obstet 1982;154:351 358.
24. Skalli O, Sch rch W, Seemayer TA, et al. Myofibroblasts from diverse pathologic settings are heterogeneous in their content of actin isoforms and intermediate filament proteins. Lab Invest 1989;60:275 285.
25. Darby I, Skalli O, Gabbiani G. Alpha-smooth muscle actin is transiently expressed by myofibroblasts during experimental wound healing. Lab Invest 1990;63:21 29.
26. Sch rch W, Skalli O, Gabbiani G. Cellular biology of Dupuytren's disease. In: McFarlane RM, McGrouther DA, Flint MH, eds. Dupuytren's Disease. London: Churchill Livingstone; 1990.
27. Desmouli re A, Geinoz A, Gabbiani F, Gabbiani G. Transforming growth factor-beta 1 induces alpha-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol 1993;122:103 111.
28. R nnov-Jessen L, Petersen OW. Induction of alpha-smooth muscle actin by transforming growth factor-beta1 in quiescent human breast gland fibroblasts. Lab Invest 1993;68:696 707.
29. Zhang K, Rekhter MD, Gordon D, Phan SH. Myofibroblasts and their role in lung collagen gene expression during pulmonary fibrosis. A combined immunohistochemical and in situ hybridization study. Am J Pathol 1994;145:114 125.
30. Serini G, Bochaton-Piallat ML, Ropraz P, et al. The fibronectin domain ED-A is crucial for myofibroblastic phenotype induction by transforming growth factor-beta1. J Cell Biol 1998;142:873 881.
31. Desmouli re A, Redard M, Darby I, Gabbiani G. Apoptosis mediates the decrease in cellularity during the transition between granulation tissue and scar. Am J Pathol 1995;146:56 66.
32. Friedman SL. The cellular basis of hepatic fibrosis. Mechanisms and treatment strategies. N Engl J Med 1993;328:1828 1835.
33. Johnson RJ, Iida H, Alpers CE, et al. Expression of smooth muscle cell phenotype by rat mesangial cells in immune complex nephritis. Alpha-smooth muscle actin is a marker of mesangial cell proliferation. J Clin Invest 1991;87:847 858.
34. Kalluri R, Neilson EG. Epithelial-mesenchymal transition and its implications for fibrosis. J Clin Invest 2003;112:1776 1784.
35. Yanez-Mo M, Lara-Pezzi E, Selgas R, et al. Peritoneal dialysis and epithelial-to-mesenchymal transition of mesothelial cells. N Engl J Med 2003;348:403 413.
36. Bucala R, Spiegel LA, Chesney J, Hogan M, Cerami A. Circulating fibrocytes define a new leukocyte subpopulation that mediates tissue repair. Mol Med 1994;1:71 81.
37. Yang L, Scott PG, Giuffre J, Shankowsky HA, Ghahary A, Tredget EE. Peripheral blood fibrocytes from burn patients: identification and quantification of fibrocytes in adherent cells cultured from peripheral blood mononuclear cells. Lab Invest 2002;82:1183 1192.
38. Gabella G. Structural apparatus for force transmission in smooth muscles. Physiol Rev 1984;64:455 477.
39. Somlyo AV. Ultrastructure of vascular smooth muscle. In: Bohr DF, Somlyo AP, Sparks HV, eds. The Handbook of Physiology: The Cardiovascular System, Vol II: Vascular Smooth Muscle. Bethesda, Md: American Physiological Society, 1980;33 67.
40. H ttner I, Kocher O, Gabbiani G. Endothelial and smooth muscle cells. In: Camilleri JP, Berry CL, Fiessinger JN, Bariety J, eds. Diseases of the Arterial Wall. New York: Springer-Verlag; 1989:3 41.
41. Schollmeyer JE, Goll DE, Robson RM, Stromer MH. Localization of alpha actinin and tropomyosin in different muscles [abstract]. J Cell Biol 1973;59:306.
42. Schollmeyer JE, Furcht LT, Goll DE, Robson RM, Stromer MH. Localization of contractile proteins in smooth muscle cells and in normal and transformed fibroblasts. In: Goldman R, Pollard T, Rosenbaum J, eds. Cell Motility. Vol A. Cold Spring Harbor, NY: Cold Spring Harbor Laboratory; 1976:361 388.
43. Brown PJ, Juliano RL. Expression and function of a putative cell surface receptor for fibronectin in hamster and human cell lines. J Cell Biol 1986;103:1595 1603.
44. Hasegawa T, Hasegawa E, Chen WT, Yamada KM. Characterization of a membrane-associated glycoprotein complex implicated in cell adhesion to fibronectin. J Cell Biochem 1985;28:307 318.
45. Knudsen KA, Horwitz AF, Buck CA. A monoclonal antibody identifies a glycoprotein complex involved in cell-substratum adhesion. Exp Cell Res 1985;157:218 226.
46. Rogalski AA, Singer SJ. An integral glycoprotein associated with the membrane attachment sites of actin microfilaments. J Cell Biol 1985;101:785 801.
P.158
47. Horwitz A, Duggan K, Buck C, Beckerle MC, Burridge K. Interaction of plasma membrane fibronectin receptor with talin: a transmembrane linkage. Nature 1986;320;531 533.
48. Hynes RO, Yamada KM. Fibronectins: multifunctional modular glycoproteins. J Cell Biol 1982;95(pt 1):369 377.
49. Singer II. The fibronexus: a transmembrane association of fibronectin-containing fibers and bundles of 5nm microfilaments in hamster and human fibroblasts. Cell 1979;16:675 685.
50. Hinz B, Gabbiani G. Cell-matrix and cell-cell contacts of myofibroblasts: role in connective tissue remodeling. Thromb Haemost 2003;90:993 1002.
51. Bloom S, Cancilla PA. Conformational changes in myocardial nuclei of rats. Circ Res 1969;24:189 196.
52. Franke WW, Schinko W. Nuclear shape in muscle cells. J Cell Biol 1969;42:326 331.
53. Lane BP. Alterations in the cytologic detail of intestinal smooth muscle cells in various stages of contraction. J Cell Biol 1965;27:199 213.
54. Majno G, Shea SM, Leventhal M. Endothelial contraction induced by histamine-type mediators. An electron microscopic study. J Cell Biol 1969;42:647 672.
55. Sch rch W, Seemayer TA, Gabbiani G. The myofibroblast: a quarter century after its discovery. Am J Surg Pathol 1998;22:141 147.
56. Eyden B. The fibronexus in reactive and tumoral myofibroblasts: further characterization by electron microscopy. Histol Histopathol 2001;16:57 70.
57. Weiss SW, Goldblum JR, eds. Enzinger and Weiss's Soft Tissue Tumors. 4th ed. St. Louis: Mosby; 2001:416.
58. Erlandson RA. In: Weidner N, Cote RJ, Suster S, Weiss LM, eds. Modern Surgical Pathology. Role of Electron Microscopy in Modern Diagnostic Surgical Pathology. Philadelphia: WB Saunders; 2003:83.
59. Rosai J. Rosai and Ackerman's Surgical Pathology. Mosby; 2004:2254.
60. Caplan AI, Fiszman MY, Eppenberger HM. Molecular and cell isoforms during development. Science 1983;221:921 927.
61. Sch rch W, Skalli O, Seemayer TA, Gabbiani G. Intermediate filament proteins and actin isoforms as markers for soft tissue tumor differentiation and origin. I. Smooth muscle tumors. Am J Pathol 1987;128:91 103.
62. Sch rch W, Skalli O, Lagac R, Seemayer TA, Gabbiani G. Intermediate filament proteins and actin isoforms as markers for soft-tissue tumor differentiation and origin. III. Hemangiopericytomas and glomus tumors. Am J Pathol 1990;136:771 786.
63. Sch rch W, B gin LR, Seemayer TA, et al. Pleomorphic soft tissue myogenic sarcomas of adulthood. A reappraisal in the mid-1990s. Am J Surg Pathol 1996;20:131 147.
64. Rungger-Br ndle E, Gabbiani G. The role of cytoskeletal and cytocontractile elements in pathologic processes. Am J Pathol 1983;110:361 392.
65. Baba F, Skalli O, Sch rch W, Seemayer TA, Gabbiani G. Chemically induced rhabdomyosarcomas in rats. Ultrastructural, immunohistochemical, biochemical features and expression of alpha-actin isoforms. Virchows Arch B Cell Pathol Incl Mol Pathol 1988;55:263 277.
66. Baba F, Musevi-Aghdam J, Sch rch W, Royal A, Gabbiani G. Coexpression of alpha-sarcomeric actin, alpha-smooth muscle actin and desmin during myogenesis in rat and mouse embryos. I. Skeletal muscle. Differentiation 1990;44:132 142.
67. Skalli O, Bloom WS, Ropraz P, Azzarone B, Gabbiani G. Cytoskeletal remodeling of rat aortic smooth muscle cells in vitro: relationships to culture conditions and analogies to in vivo situations. J Submicrosc Cytol 1986;18:481 493.
68. Skalli O, Ropraz P, Trzeciak A, Benzonana G, Gillessen DG, Gabbiani G. A monoclonal antibody against alpha-smooth muscle actin: a new probe for smooth muscle differentiation. J Cell Biol 1986;103(pt 2):2787 2796.
69. Benzonana G, Skalli O, Gabbiani G. Correlation between the distribution of smooth muscle or non muscle myosins and -smooth muscle actin in normal and pathological soft tissues. Cell Motil Cytoskeleton 1988;11:260 274.
70. Larson DM, Fujiwara K, Alexander RW, Gimbrone MA Jr. Myosin in cultured vascular smooth muscle cells: immunofluorescence and immunochemical studies of alterations in antigenic expression. J Cell Biol 1984;99:1582 1589.
71. Schmid E, Osborn M, Rungger-Br ndle E, Gabbiani G, Weber K, Franke WW. Distribution of vimentin and desmin filaments in smooth muscle tissue of mammalian and avian aorta. Exp Cell Res 1982;137:329 340.
72. Gabbiani G, Schmid E, Winter S, et al. Vascular smooth muscle cells differ from other smooth muscle cells: predominance of vimentin filaments and a specific -type actin. Proc Natl Acad Sci U S A 1981;78:298 302.
73. Frank ED, Warren L. Aortic smooth muscle cells contain vimentin instead of desmin. Proc Natl Acad Sci U S A 1981;78:3020 3024.
74. Osborn M, Caselitz J, Puschel K, Weber K. Intermediate filament expression in human vascular smooth muscle and in arteriosclerotic plaques. Virchows Arch A Pathol Anat Histopathol 1987;411:449 458.
75. Travo P, Weber K, Osborn M. Co-existence of vimentin and desmin type intermediate filaments in a subpopulation of adult rat vascular smooth muscle cells growing in primary culture. Exp Cell Res 1982;139:87 94.
76. Schmid E, Tapscott S, Bennett GS, et al. Differential location of different types of intermediate-sized filaments in various tissues of the chicken embryo. Differentiation 1979;15:27 40.
77. Lazarides E. Intermediate filaments as mechanical integrators of cellular space. Nature 1980;283:249 256.
78. Franke WW, Schmid E, Freudenstein C, et al. Intermediate-sized filaments of the prekeratin type in myoepithelial cells. J Cell Biol 1980;84:633 654.
79. Vandekerckhove J, Weber K. At least six different actins are expressed in a higher mammal: an analysis based on the amino acid sequence of the amino-terminal tryptic peptide. J Mol Biol 1978;126:783 802.
80. Vandekerckhove J, Weber K. The complete amino acid sequence of actins from bovine aorta, bovine heart, bovine fast skeletal muscle, and rabbit slow skeletal muscle. A protein-chemical analysis of muscle actin differentiation. Differentiation 1979;14:123 133.
81. Vandekerckhove J, Weber K. Actin typing on total cellular extracts: a highly sensitive protein-chemical procedure able to distinguish different actins. Eur J Biochem 1981;113:595 603.
82. Hennessey ES, Drummond DR, Sparrow JC. Molecular genetics of actin function. Biochem J 1993;291(pt 3):657 671.
83. Miwa T, Manabe Y, Kurokawa K, et al. Structure, chromosome location, and expression of the human smooth muscle (enteric type) gamma-actin gene: evolution of six human actin genes. Mol Cell Biol 1991;11:3296 3306.
84. Sch rch W, Bochaton-Piallat ML, Geinoz A, et al. All histological types of primary human rhabdomyosarcoma express alpha-cardiac and not alpha-skeletal actin messenger RNA. Am J Pathol 1994;144:836 846.
85. Chaponnier C, Gabbiani G. Pathological situations characterized by altered actin isoform expression. J Pathol 2004;204:386 395.
86. Skalli O, Vandekerckhove J, Gabbiani G. Actin-isoform pattern as a marker of normal or pathological smooth-muscle and fibroblastic tissues. Differentiation 1987;33:232 238.
87. Kocher O, Skalli O, Bloom WS, Gabbiani G. Cytoskeleton of rat aortic smooth muscle cells. Normal conditions and experimental intimal thickening. Lab Invest 1984;50:645 652.
88. Gabbiani G, Kocher O, Bloom WS, Vandekerckhove J, Weber K. Actin expression in smooth muscle cells of rat aortic intimal thickening, human atheromatous plaque, and cultured rat aortic media. J Clin Invest 1984;73:148 152.
89. Skalli O, Pelte MF, Peclet MC, et al. Alpha-smooth muscle actin, a differentiation marker of smooth muscle cells is present in microfilamentous bundles of pericytes. J Histochem Cytochem 1989;37:315 321.
90. Zimmermann KW. Der feine Bau der Blutkapillaren. Z Anat Entwicklungsgesch 1923;68:29 109.
91. Nehls V, Drenckhahn D. Heterogeneity of microvascular pericytes for smooth muscle type alpha-actin. J Cell Biol 1991;113:147 154.
92. Fujimoto T, Singer SJ. Immunocytochemical studies of desmin and vimentin in pericapillary cells of chicken. J Histochem Cytochem 1987;35:1105 1115.
93. Hinz B, Mastrangelo D, Iselin CE, Chaponnier C, Gabbiani G. Mechanical tension controls granulation tissue contractile activity and myofibroblast differentiation. Am J Pathol 2001;159:1009 1020.
P.159
94. Kapanci Y, Ribaux C, Chaponnier C, Gabbiani G. Cytoskeletal features of alveolar myofibroblasts and pericytes in normal human and rat lung. J Histochem Cytochem 1992;40:1955 1963.
95. Loreal O, Clement B, Deugnier Y. Hepatic fibrogenesis. Rev Prat 1997;47(5):482 6. French.
96. Gabbiani G, LeLous M, Bailey AJ, Bazin S, Delaunay A. Collagen and myofibroblasts of granulation tissue. A chemical, ultrastuctural and immunologic study. Virchows Arch B Cell Pathol 1976;21:133 145.
97. Rudolph R, Guber S, Suzuki M, Woodward M. The life cycle of the myofibroblast. Surg Gynecol Obstet 1977;145:389 394.
98. Barsky SH, Rao CN, Grotendorst GR, Liotta LA. Increased content of type V collagen in desmoplasia of human breast carcinoma. Am J Pathol 1982:108:276 283.
99. Gressner AM, Bachem MG. Cellular sources of noncollagenous matrix proteins: role of fat-storing cells in fibrogenesis. Semin Liver Dis 1990;10:30 46.
100. Berndt A, Kosmehl H, Katenkamp D, Tauchmann V. The appearance of the myofibroblastic phenotype in Dupuytren's disease is associated with a fibronectin, laminin, collagen type IV and tenascin extracellular matrix. Pathobiology 1994;62:55 58.
101. Sommer P, Gleyzal C, Raccurt M, et al. Transient expression of lysyl oxidase by liver myofibroblasts in murine schistosomiasis. Lab Invest 1993;69:460 470.
102. Jourdan-Le Saux C, Gleyzal C, Garnier JM, Peraldi M, Sommer P, Grimaud JA. Lysyl oxidase cDNA of myofibroblast from mouse fibrotic liver. Biochem Biophys Res Commun 1994;199:587 592.
103. Meister P, Gokel JM, Remberger K. Palmar fibromatosis- Dupuytren's contracture. A comparison of light electron and immunofluorescence microscopic findings. Pathol Res Pract 1979;164:402 412.
104. Lagac R, Grimaud JA, Sch rch W, Seemayer TA. Myofibroblastic stromal reaction in carcinoma of the breast: variations of collagenous matrix and structural glycoproteins. Virchows Arch A Pathol Anat Histopathol 1985;408:49 59.
105. Wessells NK, Spooner BS, Ash JF, et al. Microfilaments in cellular and developmental processes. Science 1971;171:135 143.
106. Madden JW, Morton D Jr, Peacock EE Jr. Contraction of experimental wounds. I. Inhibiting wound contraction by using a topical smooth muscle antagonist. Surgery 1974;76:8 15.
107. Irl C, Kocher O, Gabbiani G. Contractility of myofibroblasts during experimental liver cirrhosis. J Submicrosc Cytol Pathol 1980;12:209 217.
108. Evans JN, Kelley J, Low RB, Adler KB. Increased contractility of isolated lung parenchyma in an animal model of pulmonary fibrosis induced by bleomycin. Am Rev Respir Dis 1982;125:89 94.
109. Rubbia-Brandt L, Mentha G, Desmouli re A, et al. Hepatic stellate cells reversibly express alpha-smooth muscle actin during acute hepatic ischemia. Transplant Proc 1997;29:2390 2395.
110. Yanagisawa M, Kurihara H, Kimura S, et al. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 1988;332:411 415.
111. Appleton I, Tomlinson A, Chandler CL, Willoughby DA. Effect of endothelin-1 on croton oil-induced granulation tissue in the rat. A pharmacologic and immunohistochemical study. Lab Invest 1992;67:703 710.
112. Thiemermann C, Corder R. Is endothelin-1 the regulator of myofibroblast contraction during wound healing? Lab Invest 1992;67:677 679.
113. Kurihara H, Yoshizumi M, Sugiyama T, et al. Transforming growth factor-beta stimulates the expression of endothelin mRNA by vascular endothelial cells. Biochem Biophys Res Commun 1989;159:1435 1440.
114. Hahn AW, Resink TJ, Kern F, B hler FR. Effects of endothelin-1 on vascular smooth muscle cell phenotypic differentiation. J Cardiovasc Pharmacol 1992;20(suppl 12):S33 S36.
115. Hinz B, Gabbiani G, Chaponnier C. The NH2-terminal peptide of alpha-smooth muscle actin inhibits force generation by the myofibroblast in vitro and in vivo. J Cell Biol 2002;157:657 663.
116. Bayreuther K, Rodemann HP, Hommel R, Dittmann K, Albiez M, Francz PI. Human skin fibroblasts in vitro differentiate along a terminal cell lineage. Proc Natl Acad Sci U S A 1988;85:5112 5116.
117. Leavitt J, Gunning P, Kedes L, Jariwalla R. Smooth muscle alpha-actin is a transformation-sensitive marker for mouse NIH 3T3 and rat-2 cells. Nature 1985;316:840 842.
118. Desmouli re A, Rubbia-Brandt L, Abdiu A, Walz T, Macieira-Coelho A, Gabbiani G. Alpha-smooth muscle actin is expressed in a subpopulation of cultured and cloned fibroblasts and is modulated by gamma-interferon. Exp Cell Res 1992;201:64 73.
119. Vande Berg JS, Rudolph R, Woodward M. Growth dynamics of cultured myofibroblasts from human breast cancer and nonmalignant contracting tissues. Plast Reconstr Surg 1984;73:605 618.
120. Vande Berg JS, Rudolph R, Woodward M. Comparative growth dynamics and morphology between cultured myofibroblasts from granulating wounds and dermal fibroblasts. Am J Pathol 1984;114:187 200.
121. Vande Berg JS, Rudolph R, Poolman WL, Disharoon DR. Comparative growth dynamics and actin concentration between cultured human myofibroblasts from granulating wounds and dermal fibroblasts from normal skin. Lab Invest 1989;61:532 538.
122. Germain L, Jean A, Auger FA, Garrel DR. Human wound healing fibroblasts have greater contractile properties than dermal fibroblasts. J Surg Res 1994;57:268 273.
123. Azzarone B, Failly-Crepin C, Daya-Grosjean L, Chaponnier C, Gabbiani G. Abnormal behavior of cultured fibroblasts from nodule and nonaffected aponeurosis of Dupuytren's disease. J Cell Physiol 1983;117:353 361.
124. Dugina V, Alexandrova A, Chaponnier C, Vasiliev J, Gabbiani G. Rat fibroblasts cultured from various organs exhibit differences in alpha-smooth muscle actin expression, cytoskeletal pattern, and adhesive structure organization. Exp Cell Res 1998;238:481 490.
125. Schmitt-Gr ff A, Pau H, Spahr R, Piper, HM, Skalli O, Gabbiani G. Appearance of alpha-smooth muscle actin in human eye lens cells of anterior capsular cataract and in cultured bovine lens-forming cells. Differentiation 1990;43:115 122.
126. R nnov-Jessen L, van Deurs B, Celis JE, Petersen OW. Smooth muscle differentiation in cultured human breast gland stromal cells. Lab Invest 1990;63:532 543.
127. Rockey DC, Friedman SL. Cytoskeleton of liver perisinusoidal cells (lipocytes) in normal and pathological conditions. Cell Motil Cytoskeleton 1992;22:227 234.
128. Elger M, Drenckhahn D, Nobiling R, Mundel P, Kriz W. Cultured rat mesangial cells contain smooth muscle alpha-actin not found in vivo. Am J Pathol 1993;142:497 509.
129. Buckley IK, Porter KR. Cytoplasmic fibrils in living cultured cells. A light and electron microscope study. Protoplasma 1967;64:349 380.
130. Geiger B, Bershadsky A, Pankov R, Yamada KM. Transmembrane crosstalk between the extracellular matrix-cytoskeleton crosstalk. Nat Rev Mol Cell Biol 2001;2:793 805.
131. Goldman RD. The use of heavy meromyosin binding as an ultrastructural cytochemical method for localizing and determining the possible functions of actin-like microfilaments in nonmuscle cells. J Histochem Cytochem 1975;23:529 542.
132. Goldman RD, Lazarides E, Pollack R, Weber K. The distribution of actin in non-muscle cells. The use of actin antibody in the localization of actin within the microfilament bundles of mouse 3T3 cells. Exp Cell Res 1975;90:333 344.
133. Willingham MC, Yamada SS, Davies PJ, Rutherford AV, Gallo MG, Pastan I. Intracellular localization of actin in cultured fibroblasts by electron microscopic immunochemistry. J Histochem Cytochem 1981;29:17 37.
134. Tomasek JJ, Gabbiani G, Hinz B, Chaponnier C, Brown RA. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol 2002;3:349 363.
135. Hinz B, Gabbiani G. Mechanisms of force generation and transmission by myofibroblasts. Curr Opin Biotechnol 2003;14:538 546.
136. Chrzanowska-Wodnicka M, Burridge K. Rho-stimulated contractility drives the formation of stress fibers and focal adhesions. J Cell Biol 1996;133:1403 1415.
137. Pelham RJ Jr, Wang Y. Cell locomotion and focal adhesions are regulated by substrate flexibility. Proc Natl Acad Sci U S A 1997;94:13661 13665.
138. Silver FH, Siperko LM, Seehra GP. Mechanobiology of force transduction in dermal tissue. Skin Res Technol 2003;9:3 23.
P.160
139. Grinnell F. Fibroblast biology in three-dimensional collagen matrices. Trends Cell Biol 2003;13:264 269.
140. Mochitate K, Pawelek P, Grinnell F. Stress relaxation of contracted collagen gels: disruption of actin filament bundles, release of cell surface fibronectin, and down-regulation of DNA and protein synthesis. Exp Cell Res 1991;193:198 207.
141. Brown RA, Prajapati R, McGrouther DA, Yannas IV, Eastwood M. Tensional homeostasis in dermal fibroblasts: mechanical responses to mechanical loading in three-dimensional substrates. J Cell Physiol 1998;175:323 332.
142. Martin P. Wound healing: aiming for perfect skin regeneration. Science 1997;276:75 81.
143. Werner S, Grose R. Regulation of wound healing by growth factors and cytokines. Physiol Rev 2003;83:835 870.
144. Arora PD, Narani N, McCulloch CA. The compliance of collagen gels regulates transforming growth factor-beta induction of alpha-smooth muscle actin in fibroblasts. Am J Pathol 1999;154:871 882.
145. Hinz B, Dugina V, Ballestrem C, Wehrle-Haller B, Chaponnier C. Alpha-smooth muscle actin is crucial for focal adhesion maturation in myofibroblasts. Mol Biol Cell 2003;14:2508 2519.
146. Hinz B, Mastrangelo D, Iselin CE, Chaponnier C, Gabbiani G. Mechanical tension controls granulation tissue contractile activity and myofibroblast differentiation. Am J Pathol 2001;159:1009 1020.
147. Roberts AB, Sporn MB, Assoian RK, et al. Transforming growth factor type beta: rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc Natl Acad Sci U S A 1986;83:4167 4171.
148. Ignotz RA, Massagu J. Transforming growth factor-beta stimulates the expression of fibronectin and collagen and their incorporation into the extracellular matrix. J Biol Chem 1986;261:4337 4345.
149. Friedman SL, Yamasaki G, Wong L. Modulation of transforming growth factor beta receptors of rat lipocytes during the hepatic wound healing response. Enhanced binding and reduced gene expression accompany cellular activation in culture and in vivo. J Biol Chem 1994;269:10551 10558.
150. Border WA, Ruoslahti E. Transfoming growth factor-beta in disease: the dark side of tissue repair. J Clin Invest 1992;90:1 7.
151. Bogatkevich GS, Tourkina E, Abrams CS, Harley RA, Silver RM, Ludwicka-Bradley A. Contractile activity and smooth muscle alpha-actin organization in thrombin-induced human lung myofibroblasts. Am J Physiol Lung Cell Mol Physiol 2003;285: L334 L343.
152. Shi-Wen X, Chen Y, Denton CP, et al. Endothelin-1 promotes myofibroblast induction through the ETA receptor via a rac/phosphoinositide 3-kinase/Akt-dependent pathway and is essential for the enhanced contractile phenotype of fibrotic fibroblasts. Mol Biol Cell 2004;15:2707 2719.
153. Shephard P, Hinz B, Smola-Hess S, Meister JJ, Krieg T, Smola H. Dissecting the roles of endothelin, TGF-beta and GM-CSF on myofibroblast differentiation by keratinocytes. Thromb Haemost 2004;92:262 274.
154. Vyalov S, Desmouli re A, Gabbiani G. GM-CSF-induced granulation tissue formation: relationships between macrophage and myofibroblast accumulation. Virchows Archiv B Cell Pathol Incl Mol Pathol 1993;63:231 239.
155. Rubbia-Brandt L, Sappino AP, Gabbiani G. Locally applied GM-CSF induces the accumulation of alpha-smooth muscle actin containing myofibroblasts. Virchows Archiv B Cell Pathol Incl Mol Pathol 1991;60:73 82.
156. Micera A, Vigneti E, Pickholtz D, et al. Nerve growth factor displays stimulatory effects on human skin and lung fibroblasts, demonstrating a direct role for this factor in tissue repair. Proc Natl Acad Sci U S A 2001;98:6162 6167.
157. Isenberg G, Rathke PC, H lsmann N, Franke WW, Wohlfarth-Bottermann KE. Cytoplasmic actomyosin fibrils in tissue culture cells: direct proof of contractility by visualization of ATP-induced contraction in fibrils isolated by laser micro-beam dissection. Cell Tissue Res 1976;166:427 443.
158. Kreis TE, Birchmeier W. Stress fiber sarcomeres of fibroblasts are contractile. Cell 1980;22(pt 2):555 561.
159. Burridge K. Are stress fibres contractile? Nature 1981;294:691 692.
160. Harris AK, Stopak D, Wild P. Fibroblast traction as a mechanism for collagen morphogenesis. Nature 1981;290:249 251.
161. Beningo KA, Wang YL. Flexible substrata for the detection of cellular traction forces. Trends Cell Biol 2002;12:79 84.
162. Katoh K, Kano Y, Amano M, Onishi H, Kaibuchi K, Fujiwara K. Rho-kinase: mediated contraction of isolated stress fibers. J Cell Biol 2001;153:569 584.
163. Parizi M, Howard EW, Tomasek JJ. Regulation of LPA-promoted myofibroblast contraction: role of Rho, myosin light chain kinase, and myosin light chain phosphatase. Exp Cell Res 2000;254:210 220.
164. Wrobel LK, Fray TR, Molloy JE, Adams JJ, Armitage MP, Sparrow JC. Contractility of single human dermal myofibroblasts and fibroblasts. Cell Motil Cytoskeleton 2002;52:82 90.
165. Hinz B, Celetta G, Tomasek JJ, Gabbiani G, Chaponnier C. Alpha-smooth muscle actin expression upregulates fibroblast contractile activity. Mol Biol Cell 2001;12:2730 2741.
166. Arora PD, McCulloch CA. Dependence of collagen remodelling on alpha-smooth muscle actin expression by fibroblasts. J Cell Physiol 1994;159:161 175.
167. Dugina V, Fontao L, Chaponnier C, Vasiliev J, Gabbiani G. Focal adhesion features during myofibroblastic differentiation are controlled by intracellular and extracellular factors. J Cell Sci 2001;114(pt 18):3285 3296.
168. Cukierman E, Pankov R, Yamada KM. Cell interactions with three-dimensional matrices. Curr Opin Cell Biol 2002;14:633 639.
169. R nnov-Jessen L, Petersen OW. A function for filamentous alpha-smooth muscle actin: retardation of motility in fibroblasts. J Cell Biol 1996;134:67 80.
170. O'Shea JD. An ultrastructural study of smooth muscle-like cells in the theca externa of ovarian follicles of the rat. Anat Rec 1970;167:127 131.
171. Boya J, Carbonell AL, Martinez A. Myofibroblasts in human palatal mucosa. Acta Anat (Basel) 1988;131:161 165.
172. G ldner FH, Wolff JR, Keyserling DG. Fibroblasts as a part of the contractile system in duodenal villi of rat. Z Zellforsch Mikrosk Anat 1972;135:349 360.
173. Kaye GI, Lane N, Pascal RR. Colonic pericryptal fibroblast sheath: replication, migration, and cytodifferentiation of a mesenchymal cell system in adult tissue. II. Fine structural aspects of normal rabbit and human colon. Gastroenterology 1968;54:852 865.
174. Sappino AP, Dietrich PY, Skalli O, Widgren S, Gabbiani G. Colonic pericryptal fibroblasts. Differentiation pattern in embryogenesis and phenotypic modulation in epithelial proliferative lesions. Virchows Arch A Pathol Anat Histopathol 1989;415:551 557.
175. Bressler RS. Myoid cells in the capsule of the adrenal gland and in monolayers derived from cultured adrenal capsules. Anat Rec 1973;177:525 531.
176. Kapanci Y, Assimacopoulos A, Irl C, Zwahlen A, Gabbiani G. Contractile interstitial cells in pulmonary alveolar septa: a possible regulator of ventilation-perfusion ratio? Ultrastructural, immunofluorescence, and in vitro studies. J Cell Biol 1974;60:375 392.
177. Gorgas K, B ck P. Myofibroblasts in the rat testicular capsule. Cell Tissue Res 1974;154:533 541.
178. Czernobilsky B, Shezen E, Lifschitz-Mercer B, et al. Alpha smooth muscle actin (alpha-SM actin) in normal human ovaries, in ovarian stromal hyperplasia and in ovarian neoplasms. Virchows Arch B Cell Pathol Incl Mol Pathol 1989;57:55 61.
179. Parry EW. Some electron microscope observations on the mesenchymal structures of full-term umbilical cord. J Anat 1970;107(pt 3):505 518.
180. Tabone E, Andujar MB, DeBarros SS, Dos Santos MN, Barros CL, Graca DL. Myofibroblast-like cells in non-pathological bovine endometrial caruncle. Cell Biol Int Rep 1983;7:395 400.
181. Beertsen W, Everts V, van den Hooff A. Fine structure of fibroblasts in the periodontal ligament of the rat incisor and their possible role in tooth eruption. Arch Oral Biol 1974;19:1087 1098.
P.161
182. Beertsen W. Migration of fibroblasts in the periodontal ligament of the mouse incisor as revealed by autoradiography. Arch Oral Biol 1975;20:659 666.
183. Toccanier-Pelte MF, Skalli O, Kapanci Y, Gabbiani G. Characterization of stromal cells with myoid features in lymph nodes and spleen in normal and pathologic conditions. Am J Pathol 1987;129:109 118.
184. Yokoi Y, Namihisa T, Kuroda H, et al. Immunocytochemical detection of desmin in fat-storing cells (Ito cells). Hepatology 1984;4:709 714.
185. Glasser SR, Julian J. Intermediate filament protein as a marker of uterine stromal cell decidualization. Biol Reprod 1986;35:463 474.
186. Charbord P, Lerat H, Newton I, et al. The cytoskeleton of stromal cells from human bone marrow cultures resembles that of cultured smooth muscle cells. Exp Hematol 1990;18:276 282.
187. Becker CG. Demonstration of actomyosin in mesangial cells of the renal glomerulus. Am J Pathol 1972;66:97 110.
188. Desmouli re A, Gabbiani G. Modulation of fibroblastic cytoskeletal features during pathological situations: the role of extracellular matrix and cytokines. Cell Motil Cytoskeleton 1994;29:195 203.
189. Chiavegato A, Bochaton-Piallat ML, D'Amore E, Sartore S, Gabbiani G. Expression of myosin heavy chain isoforms in mammary epithelial cells and in myofibroblasts from different fibrotic settings during neoplasia. Virchows Arch 1995;426:77 86.
190. Sappino AP, Sch rch W, Gabbiani G. Differentiation repertoire of fibroblastic cells: expression of cytoskeletal proteins as marker of phenotypic modulations. Lab Invest 1990;63:144 161.
191. Eyden BP, Ponting J, Davies H, Bartley C, Torgersen E. Defining the myofibroblast: normal tissues, with special reference to the stromal cells of Wharton's jelly in human umbilical cord. J Submicrosc Cytol Pathol 1994;26:347 355.
192. Desmouli re A, Gabbiani G. The role of the myofibroblast in wound healing and fibrocontractive diseases. In: Clark RAF, ed. The Molecular and Cellular Biology of Wound Repair. 2nd ed. New York: Plenum Press; 1996:391 423.
193. Burrington JD. Wound healing in the fetal lamb. J Pediatr Surg 1971;6:523 528.
194. Goss AN. Intra-uterine healing of fetal rat oral mucosal, skin and cartilage wounds. J Oral Pathol 1977;6:35 43.
195. Adzick NS, Harrison MR, Glick PL, et al. Comparison of fetal, newborn, and adult wound healing by histologic, enzyme-histochemical, and hydroxyproline determinations. J Pediatr Surg 1985;20:315 319.
196. Rowsell AR. The intra-uterine healing of foetal muscle wounds: experimental study in the rat. Br J Plast Surg 1984;37:635 642.
197. Krummel TM, Nelson JM, Diegelmann RF, et al. Fetal response to injury in the rabbit. J Pediatr Surg 1987;22:640 644.
198. Adzick NS, Longaker MT. Scarless fetal healing: therapeutic implications. Ann Surg 1992;215:3 7.
199. Estes JM, Vande Berg JS, Adzick NS, MacGillivray TE, Desmouli re A, Gabbiani G. Phenotypic and functional features of myofibroblasts in sheep fetal wounds. Differentiation 1994;56:173 181.
200. Chakravarti S, Wu F, Vij N, Roberts L, Joyce S. Microarray studies reveal macrophage-like function of stromal keratocytes in the cornea. Invest Ophthalmol Vis Sci 2004;45:3475 3484.
201. Pei Y, Sherry DM, McDermott AM. Thy-1 distinguishes human corneal fibroblasts and myofibroblasts from keratocytes. Exp Eye Res 2004;79:705 712.
202. Anderson S, DiCesare L, Tan I, Leung T, SundarRaj N. Rho-mediated assembly of stress fibers is differentially regulated in corneal fibroblasts and myofibroblasts. Exp Cell Res 2004;298:574 583.
203. Seemayer TA, Sch rch W, Lagac R. Myofibroblasts in human pathology. Hum Pathol 1981;12:491 492.
204. Bhathal PS. Presence of modified fibroblasts in cirrhotic livers in man. Pathology 1972;4:139 144.
205. Cassiman D, Libbrecht L, Desmet V, Denef C, Roskams T. Hepatic stellate cell/myofibroblast subpopulations in fibrotic human and rat livers. J Hepatol 2002;36:200 209.
206. Rudolph R, McClure WJ, Woodward M. Contractile fibroblasts in chronic alcoholic cirrhosis. Gastroenterology 1979;76:704 709.
207. Madden JW. On the contractile fibroblast. Plast Recontstr Surg 1973;52:291 292.
208. Woyke S, Domagala W, Olszewski W, Korabiec M. Pseudosarcoma of the skin. An electron microscopic study and comparison with fine structure of the spindle-cell variant of squamous carcinoma. Cancer 1974;33:970 980.
209. Larson DL, Abston S, Willis B, et al. Contracture and scar formation in the burn patient. Clin Plast Surg 1974;1:653 656.
210. Madden JW, Carlson EC, Hines J. Presence of modified fibroblasts in ischemic contracture of the intrinsic musculature of the hand. Surg Gynecol Obstet 1975;140:509 516.
211. Nagle RB, Kneiser MR, Bulger RE, Benditt EP. Induction of smooth muscle characteristics in renal interstitial fibroblasts during obstructive nephropathy. Lab Invest 1973;29:422 427.
212. Judd PA, Finnegan P, Curran RC. Pulmonary sarcoidosis: a clinico pathological study. J Pathol 1975;115:191 198.
213. El-Labban NG, Lee KW. Myofibroblasts in central giant cell granuloma of the jaws: an ultrastructural study. Histopathology 1983;7:907 918.
214. Grimaud JA, Borojevic R. Myofibroblasts in hepatic schistosomal fibrosis. Experientia 1977;33:890 892.
215. Postacchini F, Natali PG, Accinni L, Ippolito E, de Martino C. Contractile filaments in cells of regenerating tendon. Experientia 1977;33:957 959.
216. Rudolph R, Woodward M. Spatial orientation of microtubules in contractile fibroblasts in vivo. Anat Rec 1978;191:169 181.
217. Zimman OA, Robles JM, Lee JC. The fibrous capsule around mammary implants: an investigation. Aesthetic Plast Surg 1978;2:217 234.
218. Callea F, Mebis J, Desmet VJ. Myofibroblasts in focal nodular hyperplasia of the liver. Virchows Arch A Pathol Anat Histol 1982;396:155 166.
219. Ghadially FN, Mehta PN. Multifunctional mesenchymal cells resembling smooth muscle cells in ganglia of the wrist. Ann Rheum Dis 1971;30:31 42.
220. Baur PS, Larson DL, Stacey TR. The observation of myofibroblasts in hypertrophic scars. Surg Gynecol Obstet 1975;141:22 26.
221. Novotny GE, Pau H. Myofibroblast-like cells in human anterior capsular cataract. Virchows Arch A Pathol Anat Histopathol 1984;404:393 401.
222. Woodcock-Mitchell J, Adler KB, Low RB. Immunohistochemical identification of cell types in normal and in bleomycin-induced fibrotic rat lung. Cellular origin of interstitial cells. Am Rev Respir Dis 1984;130:910 916.
223. Lagac R, Delage C, Boutet M. Light and electron microscopic study of cellular proliferation in carcinoid heart disease. Recent Adv Stud Cardiac Struct Metab 1975;10:605 616.
224. Thomas WA, Jones R, Scott RF, Morrison E, Goodale MF, Imai H. Production of early atherosclerotic lesions in rats characterized by proliferation of modified smooth muscle cells. Exp Mol Pathol 1963;52(suppl 1):40 61.
225. Flora G, Dahl E, Nelson E. Electron microscopic observations on human intracranial arteries. Changes seen with aging and atherosclerosis. Arch Neurol 1967;17:162 173.
226. Wissler RW. The arterial medial cell, smooth muscle, or multifunctional mesenchyme? Circulation 1967;36:1 4.
227. Gabbiani G, Badonnel MC. Contractile apparatus in aortic endothelium of hypertensive rat. Recent Adv Stud Cardiac Struct Metab 1975;10:591 601.
228. Sappino AP, Masouy I, Saurat JH, Gabbiani G. Smooth muscle differentiation in scleroderma fibroblastic cells. Am J Pathol 1990;137:585 591.
229. Diamond JR, van Goor H, Ding G, Engelmyer E. Myofibroblasts in experimental hydronephrosis. Am J Pathol 1995;146:121 129.
230. Gabbrielli S, Di Lollo S, Stanflin N, Romagnoli P. Myofibroblast and elastic and collagen fiber hyperplasia in the bronchial mucosa: a possible basis for the progressive irreversibility of airway obstruction in chronic asthma. Pathologica 1994;86:157 160.
231. Gizycki MJ, Adelroth E, Rogers AV, O'Byrne PM, Jeffery PK. Myofibroblast involvement in the allergen-induced late response in mild atopic asthma. Am J Respir Cell Mol Biol 1997;16:664 673.
P.162
232. Holgate ST, Davies DE, Lackie PM, Wilson SJ, Puddicombe SM, Lordan JL. Epithelial-mesenchymal interactions in the pathogenesis of asthma. J Alllergy Clin Immunol 2000;105(pt 1):193 204.
233. Hastie AT, Kraft WK, Nyce KB, et al. Asthmatic epithelial cell proliferation and stimulation of collagen production: human asthmatic epithelial cells stimulate collagen type III production by human lung myofibroblasts after segmental allergen challenge. Am J Respir Crit Care Med 2002;165:266 272.
234. Bari ty J. La biopsie r nale. In: Droz D, Lanz B, eds. La biopsie r nale dans la hyalinose segmentaire et focale des glom rules. Paris, France; Publisher: Editions INSERM. 1996:109 134.
235. Sch rch W, Seemayer TA, Gabbiani G. Myofibroblasts. In: Sternberg SS, ed. Histology for Pathologists. New York: Raven Press; 1992:109 144.
236. Ehrlich HP, Desmouli re A, Diegelmann RF, et al. Morphological and immunochemical differences between keloid and hypertrophic scar. Am J Pathol 1994;145:105 113.
237. Santucci M, Borgognoni L, Reali UM, Gabbiani G. Keliods and hypertrophic scars of Caucasians show distinctive morphologic and immunophenotypic profiles. Virchows Arch 2001;438:457 463.
238. Enzinger FM, Weiss SW. Fibromatoses. In: Enzinger FM, Weiss SW, eds. Soft Tissue Tumors. 3rd ed. St Louis: CV Mosby; 1994:201 229.
239. Chiu HF, McFarlane RM. Pathogenesis of Dupuytren's contracture: a correlative clinical-pathological study. J Hand Surg (Am)1978;3:1 10.
240. Navas-Palacios JJ. The fibromatoses. An ultrastructural study of 31 cases. Pathol Res Pract 1983;176:158 175.
241. Ushijima M, Tsuneyoshi M, Enjoji M. Dupuytren type fibromatoses. A clinicopathologic study of 62 cases. Acta Pathol Jpn 1984;34:991 1001.
242. Ariyan S, Enriquez R, Krizek TJ. Wound contraction and fibrocontractive disorders. Arch Surg 1978;113:1034 1046.
243. Allen PW. The fibromatoses: a clinicopathologic classification based on 140 cases. Am J Surg Pathol 1977;1:255 270.
244. Chung EB, Enzinger FM. Infantile myofibromatosis. Cancer 1981;48:1807 1818.
245. Wirman JA. Nodular fasciitis, a lesion of myofibroblasts: an ultrastructural study. Cancer 1976;38:2378 2389.
246. Chung EB, Enzinger FM. Proliferative fasciitis. Cancer 1975;36:1450 1458.
247. Povysil C, Matejovsky Z. Ultrastructural evidence of myofibroblasts in pseudomalignant myositis ossificans. Virchows Arch A Pathol Anat Histol 1979;381:189 203.
248. Weathers DR, Campbell WG. Ultrastructure of the giant-cell fibroma of the oral mucosa. Oral Surg Oral Med Oral Pathol 1974;38:550 561.
249. Stiller D, Katenkamp D. Cellular features in desmoid fibromatosis and well-differentiated fibrosarcomas: an electron microscopic study. Virchows Arch A Pathol Anat Histol 1975;369:155 164.
250. Ramos CV, Gillespie W, Narconis RJ. Elastofibroma. A pseudotumor of myofibroblasts. Arch Pathol Lab Med 1978;102:538 540.
251. Buell R, Wang NS, Seemayer TA, Ahmed MN. Endobronchial plasma cell granuloma (xanathomatous pseudotumor): a light and electron microscopic study. Hum Pathol 1976;7:411 426.
252. Bhawan J, Bacchetta C, Joris I, Majno G. A myofibroblastic tumor. Infantile digital fibroma (recurrent digital fibrous tumor of childhood). Am J Pathol 1979;94:19 36.
253. Taxy JB. Juvenile nasopharyngeal angiofibroma: an ultrastructural study. Cancer 1977;39:1044 1054.
254. Ferrans VJ, Roberts WC. Structural features of cardiac myxomas. Histology, histochemistry, and electron microscopy. Hum Pathol 1973;4:111 146.
255. Fisher ER, Paulson JD, Gregorio RM. The myofibroblastic nature of the uterine plexiform tumor. Arch Pathol Lab Med 1978;102:477 480.
256. Tomasek JJ, Schultz RJ, Episalla CW, Newman SA. The cytoskeleton and extracellular matrix of the Dupuytren's disease myofibroblast : an immunofluorescence study of a nonmuscle cell type. J Hand Surg (Am) 1986;11:365 371.
257. Shum DT, McFarlane RM. Histogenesis of Dupuytren's disease: an immunohistochemical study of 30 cases. J Hand Surg (Am) 1988;13:61 67.
258. Luck JV. Dupuyten's contracture: a new concept of the pathogenesis correlated with surgical management. J Bone Joint Surg Am 1959;41-A:635 664.
259. Iwasaki H, M ller H, Stutte HJ, Brennscheidt U. Palmar fibromatosis (Dupuytren's contracture). Ultrastructural and enzyme histochemical studies of 43 cases. Virchows Arch A Pathol Anat Histopathol 1984;405:41 53.
260. Eddy RJ, Petro JA, Tomasek JJ. Evidence for the nonmuscle nature of the myofibroblast of granulation tissue and hypertrophic scar. An immunofluorescence study. Am J Pathol 1988;130:252 260.
261. Matte C, Cadotte M, Sch rch W. Intermediate filament proteins and actin isoforms of dermatofibrosarcoma protuberans and dermatofibroma. Lab Invest 1990;62(1):64A(abst. 373).
262. Fukasawa Y, Ishikura H, Takada A, et al. Massive apoptosis in infantile myofibromatosis. A putative mechanism of tumor regression. Am J Pathol 1994;144:480 485.
263. Ohtani H, Sasano N. Myofibroblasts and myoepithelial cells in human breast carcinoma. An ultrastructural study. Virchows Arch A Pathol Anat Histol 1980;385:247 261.
264. Cintorino M, Bellizzi de Marco E, Leoncini P, et al. Expression of -smooth-muscle actin in stromal cells of the uterine cervix during epithelial neoplastic changes. Int J Cancer 1991;47:843 846.
265. Carroll R. The influence of lung scars on primary lung cancer. J Pathol Bacteriol 1962;83:293 297.
266. Madri JA, Carter D. Scar cancers of the lung: origin and significance. Hum Pathol 1984;15:625 631.
267. Barsky SH, Huang SJ, Bhuta S. The extracellular matrix of pulmonary scar carcinomas is suggestive of a desmoplastic origin. Am J Pathol 1986;124:412 419.
268. Gabbiani G, Kaye GI, Lattes R, Majno G. Synovial sarcoma. Electron microscopic study of a typical case. Cancer 1971;28:1031 1039.
269. Gabbiani G, Fu YS, Kaye GI, Lattes R, Majno G. Epithelioid sarcoma. A light and electron microscopic study suggesting a synovial origin. Cancer 1972;30:486 499.
270. Lagac R, Sch rch W, Seemayer TA. Myofibroblasts in soft tissue sarcomas. Virchows Arch A Pathol Anat Histol 1980;389:1 11.
271. Seemayer TA, Lagac R, Sch rch W. On the pathogenesis of sclerosis and nodularity in nodular sclerosing Hodgkin's disease. Virchows Arch A Pathol Anat Histol 1980;385:283 291.
272. Taccagni G, Rovere E, Masullo M, Christensen L, Eyden B. Myofibrosarcoma of the breast: review of the literature on myofibroblastic tumors and criteria for defining myofibroblastic differentiation. Am J Surg Pathol 1997;21:489 496.
273. Churg AM, Kahn LB. Myofibroblasts and related cells in malignant fibrous and fibrohistiocytic tumors. Hum Pathol 1977;8:205 218.
274. D'Andiran G, Gabbiani G. A metastasizing sarcoma of the pleura composed of myofibroblasts. In: Fenoglio CM, Woolf CM, eds. Progress in Surgical Pathology. New York: Masson Publishing; 1980:31 40.
275. Eyden BP, Christensen L, Tagore V, Harris M. Myofibrosarcoma of subcutaneous soft tissue of the cheek. J Submicrosc Cytol Pathol 1992;24:307 313.
276. Eyden BP, Ponting J, Davies H, Bartley C, Torgersen E. Defining the myofibroblast: normal tissues, with special reference to the stromal cells of Wharton's jelly in human umbilical cord. J Submicrosc Cytol Pathol 1994;26:347 355.
277. Mentzel T, Dry S, Katenkamp D, Fletcher CD. Low-grade myofibroblastic sarcoma: analysis of 18 cases in the spectrum of myofibroblastic tumors. Am J Surg Pathol 1998;22:1228 1238.
278. Eyden BP, Banerjee SS, Harris M, Mene A. A study of spindle cell sarcomas showing myofibroblastic differentiation. Ultrastruct Pathol 1991;15:367 378.
279. Fisher C. Myofibroblastic malignancies. Adv Anat Pathol 2004;11:190 201.
280. Alobeid B, Beneck D, Sreekantaiah C, Abbi RK, Slim MS. Congenital pulmonary myofibroblastic tumor: a case report with cytogenetic analysis and review of the literature. Am J Surg Pathol 1997;21:610 614.
P.163
281. Fletcher CD, Tsang WY, Fisher C, Lee KC, Chan JK. Angiomyofibroblastoma of the vulva. A benign neoplasm distinct from aggressive angiomyxoma. Am J Surg Pathol 1992;16:373 382.
282. Fukunaga M, Nomura K, Matsumoto K, Doi K, Endo Y, Ushigome S. Vulval angiomyofibroblastoma. Clinicopathologic analysis of six cases. Am J Clin Pathol 1997;107:45 51.
283. Ghadially FN, McNaughton JD, Lalonde JM. Myofibroblastoma: a tumor of myofibroblasts. J Submicrosc Cytol 1983;15:1055 1063.
284. Herrera GA, Johnson WW, Lockard VG, Walker BL. Soft tissue myofibroblastomas. Mod Pathol 1991;4:571 577.
285. Laskin WB, Fetsch JF, Mostofi FK. Angiomyofibroblastomalike tumor of the male genital tract: analysis of 11 cases with comparison to female angiomyofibroblastoma and spindle cell lipoma. Am J Surg Pathol 1998;22:6 16.
286. Ockner DM, Sayadi H, Swanson PE, Ritter JH, Wick MR. Genital angiomyofibroblastoma. Comparison with aggressive angiomyxoma and other myxoid neoplasms of skin and soft tissue. Am J Clin Pathol 1997;107:36 44.
287. Prayson RA, Estes ML, McMahon JT, Kalfas I, Sebek BA. Meningeal myofibroblastoma. Am J Surg Pathol 1993;17:931 936.
288. Suster S, Rosai J. Intranodal hemorrhagic spindle-cell tumor with amianthoid fibers. Report of six cases of a distinctive mesenchymal neoplasm of the inguinal region that simulates Kaposi's sarcoma. Am J Surg Pathol 1989;13:347 357.
289. Thomas TM, Myint A, Mak CK, Chan JK. Mammary myofibroblastoma with leiomyomatous differentiation. Am J Clin Pathol 1997;107:52 55.
290. Wargotz ES, Weiss SW, Norris HJ. Myofibroblastoma of the breast. Sixteen cases of a distinctive benign mesenchymal tumor. Am J Surg Pathol 1987;11:493 502.
291. Weiss SW, Gnepp DR, Bratthauer GL. Palisaded myofibroblastoma. A benign mesenchymal tumor of lymph node. Am J Surg Pathol 1989;13:341 346.
292. B gin LR. Myogenic stromal tumor of the male breast (so-called myofibroblastoma). Ultrastruct Pathol 1991;15:613 622.
293. Fletcher CD, Unni KK, Mertens F, eds. Pathology and Genetics of Tumors of Soft Tissue and Bone. World Health Organization Classification of Tumors. Lyon, France: IARC; 2002:91 93.
294. O'Brien JE, Stout AP. Malignant fibrous xanthomas. Cancer 1964;17:1445 1455.
295. Cessna MH, Zhou H, Sanger WG, et al. Expression of ALK1 and p80 in inflammatory myofibroblastic tumor and its mesenchymal mimics: a study of 135 cases. Mod Pathol 2002;15:931 938.
296. Eyden B. Electron microscopy in the study of myofibroblastic lesions. Semin Diagn Pathol 2003;20:13 24.
297. Cohnheim J. Ueber Entz ndung und Eiterung. Virchows Arch Path Anat 1867;40:1 79.
298. Arey LB. Wound healing. Physiol Rev 1936;16:327 406.
299. Grillo HC. Derivation of fibroblasts in healing wound. Arch Surg 1964;88:218 224.
300. Allg wer M. The Cellular Basis of Wound Repair. Springfield, IL: Charles C Thomas, 1956.
301. Ross R, Everett NB, Tyler R. Wound healing and collagen formation. VI. The origin of the wound fibroblast studied in parabiosis. J Cell Biol 1970;44:645 654.
302. Crocker DJ, Murad TM, Geer JC. Role of the pericyte in wound healing. An ultrastructural study. Exp Mol Pathol 1970;13:51 65.
303. Oda D, Gown AM, Vande Berg JS, Stern R. The fibroblast-like nature of myofibroblasts. Exp Mol Pathol 1988;49:316 329.
304. van der Loop FT, Gabbiani G, Kohnen G, Ramaekers FC, van Eys GJ. Differentiation of smooth muscle cells in human blood vessels as defined by smoothelin, a novel marker for the contractile phenotype. Arterioscler Thromb Vasc Biol 1997;17:665 671.
305. Christen T, Verin V, Bochaton-Piallat M, et al. Mechanisms of neointima formation and remodeling in the porcine coronary artery. Circulation 2001;103:882 888.
306. van der Loop FT, Schaart G, Timmer ED, Ramaekers FC, van Eys GJ. Smoothelin, a novel cytoskeletal protein specific for smooth muscle cells. J Cell Biol 1996;134:401 411.
307. Chamley JH, Campbell GR, McConnell JD, Gr schel-Stewart U. Comparison of vascular smooth muscle cells from adult human, monkey and rabbit in primary culture and subculture. Cell Tissue Res 1977;177:503 522.
308. Moss NS, Benditt EP. Spontaneous and experimentally induced arterial lesions. I. An ultrastructural survey of the normal chicken aorta. Lab Invest 1970;22:166 183.
309. Mosse PR, Campbell GR, Wang ZL, Campbell JH. Smooth muscle phenotypic expression in human carotid arteries. I. Comparison of cells from diffuse intimal thickenings adjacent to atheromatous plaques with those of the media. Lab Invest 1985;53:556 562.
310. Olivetti G, Anversa P, Melissari M, Loud AV. Morphometric study of early postnatal development of the thoracic aorta in the rat. Circ Res 1980;47:417 424.
311. Poole JC, Cromwell SB, Benditt EP. Behavior of smooth muscle cells and formation of extracellular structures in the reaction of arterial walls to injury. Am J Pathol 1971;62:391 414.
312. R nnov-Jessen L, Petersen OW, Koteliansky VE, Bissell MJ. The origin of the myofibroblasts in breast cancer. Recapitulation of tumor environment in culture unravels diversity and implicates converted fibroblasts and recruited smooth muscle cells. J Clin Invest 1995;95:859 873.
313. Franke WW, Moll R. Cytoskeletal components of lymphoid organs. I. Synthesis of cytokeratins 8 and 18 and desmin in subpopulations of extrafollicular reticulum cells of human lymph nodes, tonsils and spleen. Differentiation 1987;36:145 163.
314. Ramadori G, Veit T, Schwogler S, et al. Expression of the gene of the -smooth muscle-actin isoform in rat liver and in rat fat-storing (ITO) cells. Virchows Arch B Cell Pathol Incl Mol Pathol 1990;59:349 357.
315. Schmitt-Gr ff A, Kr ger S, Bochard F, Gabbiani G, Denk H. Modulation of alpha smooth muscle actin and desmin expression in perisinusoidal cells of normal and diseased human livers. Am J Pathol 1991;138:1233 1242.
316. Blazejewski S, Preaux AM, Mallat A, et al. Human myofibroblastlike cells obtained by outgrowth are representative of the fibrogenic cells in the liver. Hepatology 1995;22:788 797.
317. Milani S, Herbst H, Schuppan D, Riecken EO, Stein H. Cellular localization of laminin gene transcripts in normal and fibrotic human liver. Am J Pathol 1989;134:1175 1182.
318. Milani S, Herbst H, Schuppan D, Surrenti C, Riecken EO, Stein H. Cellular localization of type I, III, and IV procollagen gene transcripts in normal and fibrotic human liver. Am J Pathol 1990;137:59 70.
319. Takahara T, Nakayama Y, Itoh H, et al. Extracellular matrix formation in piecemeal necrosis: immunoelectron microscopic study. Liver 1992;12:368 380.
320. Nagy P, Schaff Z, Lapis K. Immunohistochemical detection of transforming growth factor- 1 in fibrotic liver diseases. Hepatology 1991;14:269 273.
321. Goumenos DS, Brown CB, Shortland J, El Nahas AM. Myofibroblasts, predictors of progression of mesangial IgA nephropathy? Nephrol Dial Transplant 1994;9:1418 1425.
322. Boukhalfa G, Desmouli re A, Rondeau E, Gabbiani G, Sraer JD. Relationship between -smooth muscle actin expression and fibrotic changes in human kidney. Exp Nephrol 1996;4:241 247.
323. Kapanci Y, Ribaux C, Chaponnier C, Gabbiani G. Cytoskeletal features of alveolar myofibroblasts and pericytes in normal human and rat lung. J Histochem Cytochem 1992;40:1955 1963.
324. Vyalov SL, Gabbiani G, Kapanci Y. Rat alveolar myofibroblasts acquire -smooth muscle actin expression during bleomycin-induced pulmonary fibrosis. Am J Pathol 1993;143:1754 1765.
325. Tredget EE, Shankowsky HA, Pannu R, et al. Transforming growth factor-beta in thermally injured patients with hypertrophic scars: effects of interferon alpha-2b. Plast Reconstr Surg 1998;102:1317 1330.
326. Ishii G, Sangai T, Oda T, et al. Bone-marrow-derived myofibroblasts contribute to the cancer-induced stromal reaction. Biochem Biophys Res Commun 2003;309:232 240.
P.164
327. Schmidt M, Sun G, Stacey MA, Mori L, Mattoli S. Identification of circulating fibrocytes as precursors of bronchial myofibroblasts in asthma. J Immunol 2003;171:380 389.
328. Garbin S, Pittet B, Montandon D, Gabbiani G, Desmouli re A. Covering by a flap induces apoptosis of granulation tissue myofibroblasts and vascular cells. Wound Repair Regen 1996;4:244 251.
329. Carlson MA, Longaker MT, Thompson JS. Granulation tissue regression induced by musculocutaneous advancement flap coverage. Surgery 2002;131:332 337.
330. Grinnell F, Zhu M, Carlson MA, Abrams JM. Release of mechanical tension triggers apoptosis of human fibroblasts in a model of regressing granulation tissue. Exp Cell Res 1999;248:608 619.
331. Fluck J, Querfeld C, Cremer A, Niland S, Krieg T, Sollberg S. Normal human primary fibroblasts undergo apoptosis in three-dimensional contractile collagen gels. J Invest Dermatol 1998;110:153 157.
332. Evans VG. Multiple pathways to apoptosis. Cell Biol Int 1993;17:461 476.
333. Lee S, Christakos S, Small MB. Apoptosis and signal transduction: clues to a molecular mechanism. Curr Opin Cell Biol 1993;5:286 291.
334. Schwartzman RA, Cidlowski JA. Apoptosis: the biochemistry and molecular biology of programmed cell death. Endocrinol Rev 1993;14:133 151.
335. White E. Death-defying acts: a meeting review on apoptosis. Genes Dev 1993;7:2277 2284.
336. Williams GT, Smith CA. Molecular regulation of apoptosis: genetic controls on cell death. Cell 1993;74:777 779.
337. Martin SJ, Green DR, Cotter TG. Dicing with death: dissecting the components of the apoptosis machinery. Trends Biochem Sci 1994;19:26 30.
338. Evan GI, Wyllie AH, Gilbert CS, et al. Induction of apoptosis in fibroblasts by c-myc protein. Cell 1992;69:119 128.
339. Miura M, Zhu H, Rotello R, Hartwieg EA, Yuan J. Induction of apoptosis in fibroblasts by IL-1 -converting enzyme, a mammalian homolog of the C. elegans cell death gene ced-3. Cell 1993;75:653 660.
340. Reed JC. Bcl-2 and the regulation of programmed cell death. J Cell Biol 1994;124:1 6.
341. Laster SM, Wood JG, Gooding LR. Tumor necrosis factor can induce both apoptic and necrotic forms of cell lysis. J Immunol 1988;141;2629 2634.
342. Robaye B, Mosselmans R, Fiers W, Dumont JE, Galand P. Tumor necrosis factor induces apoptosis (programmed cell death) in normal endothelial cells in vitro. Am J Pathol 1991;138:447 453.
343. Moulton BC. Transforming growth factor- stimulates endometrial stromal apoptosis in vitro. Endocrinology 1994;134:1055 1060.
344. Pierce GF, Mustoe TA, Senior RM, et al. In vivo incisional wound healing augmented by platelet-derived growth factor and recombinant c-sis gene homodimeric proteins. J Exp Med 1988;167:974 987.
345. Mustoe TA, Pierce GF, Thomason A, Gramates P, Sporn MB, Deuel TF. Accelerated healing of incisional wounds in rats induced by transforming growth factor- . Science 1987;237:1333 1336.
346. Beck LS, DeGuzman L, Lee WP, Xu Y, Siegel MW, Amento EP. One systemic administration of transforming growth factor- 1 reverses age- or glucocorticoid-impaired wound healing. J Clin Invest 1993;92:2841 2849.
347. Schultz GS, White M, Mitchell R, et al. Epithelial wound healing enhanced by transforming growth factor- and vaccinia growth factor. Science 1987;235:350 352.
348. Mooney DP, O'Reilly M, Gamelli RL. Tumor necrosis factor and wound healing. Ann Surg 1990;211:124 129.
349. Martin P, Hopkinson-Woolley J, McCluskey J. Growth factors and cutaneous wound repair. Prog Growth Factor Res 1992;4:25 44.
350. Streuli CH, Schmidhauser C, Kobrin M, Bissell MJ, Derynck R. Extracellular matrix regulates expression of the TGF 1 gene. J Cell Biol 1993;120:253 260.
351. Micke P, Ostman A. Tumour-stroma interaction: cancer-associated fibroblasts as novel targets in anti-cancer therapy? Lung Cancer 2004;45(suppl 2):S163 S175.
352. De Wever O, Nguyen QD, Van Hoorde L, et al. Tenascin-C and SF/HGF produced myofibroblasts in vitro provide convergent pro-invasive signals to human colon cancer cells through RhoA and Rac. Faseb J 2004;18:1016 1018.
353. Doljanski F. The sculpturing role of fibroblast-like cells in morphogenesis. Perspect Biol Med 2004;47:339 356.
354. Orimo A, Gupta PB, Sgroi DC, et al. Stromal fibroblasts in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 2005;121:335 348.
355. Dvorak HF. Tumors: wounds that do not heal. Similarities between tumor stroma generation and wound healing. N Engl J Med 1986;315:1650 1659.
356. Whitby DJ, Ferguson MW. Immunohistochemical localization of growth factors in fetal wound healing. Dev Biol 1991;147:207 215.