14 - Kidney Disease and Hypertension in Pregnancy
Editors: Schrier, Robert W.
Title: Manual of Nephrology, 6th Edition
Copyright 2005 Lippincott Williams & Wilkins
> Table of Contents > 14 - The Patient with Kidney Disease and Hypertension in Pregnancy
function show_scrollbar() {}
14
The Patient with Kidney Disease and Hypertension in Pregnancy
Phyllis August
Marshall D. Lindheimer
In most instances, pregnancy in women with renal disorders is successful, provided kidney function is well preserved and hypertension absent.
The kidney and blood pressure in normal pregnancy. The anatomy and function of the kidneys and lower urinary tract are altered during gestation. Physiologic alterations in volume homeostasis and blood pressure (BP) control also occur, and recognizing this is a prerequisite for the appropriate interpretation of data from pregnant patients with renal disease or hypertension (Table 14-1).
Table 14-1. Renal Changes in Normal Pregnancy
Alteration Manifestation Clinical relevance Increased renal size Renal length approximately 1 cm greater on radiographs Postpartum decreases in size should not be mistaken for parenchymal loss Dilation of pelves, calyces, and ureters Resembles hydronephrosis on renal ultrasound or intravenous pyelography (more marked on right) Not to be mistaken for obstructive uropathy; elective evaluation should be deferred to the twelfth postpartum week; upper urinary tract infections are more virulent; retained urine leads to collection errors Increased renal hemodynamics Glomerular filtration rate and renal plasma flow increase 35% to 50% Serum creatinine and urea nitrogen values decrease during normal gestations; >0.8 mg/dL creatinine already suspect; protein, amino acid, and glucose urinary excretion all increase Changes in acid-base metabolism Renal bicarbonate threshold decreases Serum bicarbonate is 4 to 5 mol/L lower in normal gestation Renal water handling Osmoregulation altered Serum osmolality decreases 10 mOsm/L (serum sodium decreases 5 mEq/L) during normal gestation Osmotic thresholds for thirst and AVP decrease; the metabolic clearance of AVP increases markedly; high levels of vasopressinase circulating Increased metabolism of AVP may cause transient diabetes insipidus in pregnancy AVP, arginine vasopressin. Anatomic and functional changes in urinary tract. Kidney length increases approximately 1 cm during normal gestation. The major anatomic alterations of the urinary tract during pregnancy, however, are seen in the collecting system, where calyces, renal pelves, and ureters dilatate, often giving the erroneous impression of obstructive uropathy. The dilation is accompanied by hypertrophy of ureteral smooth muscle and hyperplasia of its connective tissue, but whether bladder reflux is more common in gravidas is unclear. The cause of the ureteral dilation is disputed. Some researchers favor hormonal mechanisms, whereas other researchers believe that it is obstructive in origin. Clearly, as pregnancy progresses, assumption of a supine or upright posture may cause ureteral obstruction when the enlarged uterus entraps the ureters at the pelvic brim (Fig. 14-1). These morphologic changes have considerable clinical relevance. Stasis in the dilated urinary tract may contribute to the propensity of pregnant women with asymptomatic bacteriuria to develop frank pyelonephritis. The widened ureters contain substantial volumes of urine, which may lead to collection errors in tests that require timed urine volumes. These errors may be avoided by the following simple protocol. Gravidas to be tested should receive a water load and remain in bed positioned in lateral recumbency for 1 hour before the start of the collection. This procedure minimizes inaccuracies by standardizing the procedures and by producing a modest water diuresis, so that residual urine is diluted and of recent origin. Acceptable norms of kidney size should be increased by 1 cm if estimated during pregnancy or the immediate puerperium, and reductions of renal length noted several months postpartum need not be attributed to renal disease. Rarely, ureteral dilation is of sufficient magnitude to cause a distention syndrome (characterized by abdominal pain, and on occasion small increments in serum creatinine levels presenting in late gestation; these resolve with the placement of ureteral stents). Also, because dilation of the ureters may persist until the twelfth postpartum week, elective ultrasonographic or radiologic examination of the urinary tract should be deferred, if possible, until after this time.
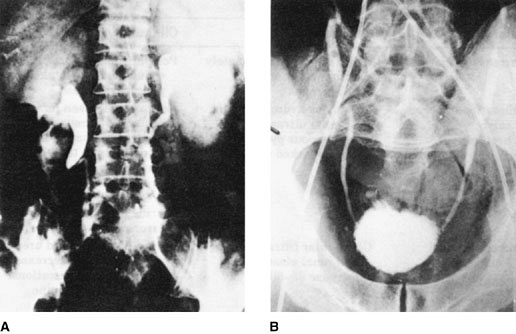
Figure 14-1. Intravenous pyelogram. A: Ureteral dilation of pregnancy. The right ureter is sharply cut off at the pelvic brim where it crosses the iliac artery (the iliac sign). B: Relationship between the ureters and iliac arteries can be demonstrated in postmortem studies. Note the iliac sign at the pelvic brim on the right. (From Dure-Smith P. Pregnancy dilation of the urinary tract. Radiology 1970;96:545. Reprinted with permission.)
Renal hemodynamics. The changes in renal hemodynamics in gestation are the most striking and clinically significant of all the urinary tract alterations of pregnancy.
Glomerular filtration rate (GFR) and renal plasma flow (RPF) increase to levels 30% and 50% above nongravid values during pregnancy. Increments in GFR that are already present during the early days after conception reach a maximum during the first trimester. The basis for the increase in GFR and RPF is unknown. Animal studies suggest that renal vasodilation (mediated by nitric oxide) leading to increased glomerular
P.215
plasma flow is a contributing, but not the sole, factor. RPF is greatest at midgestation, declining somewhat in the third trimester. Although increments in GFR measured by the infusion of inulin appear to be sustained until term, 24-hour creatinine clearance declines during the last 4 weeks of pregnancy, accompanied by increases in serum creatinine levels of 15% to 20%.The increase in GFR has important clinical implications. Because creatinine production is unchanged during pregnancy, increments in its clearance result in decreased serum levels. Using the Hare method, one group of investigators observed that true serum creatinine, which averaged 0.67 mg per dL in nongravid women, decreased to 0.46 mg per dL during gestation [to convert to SI units ( mol per L), multiply serum creatinine (mg per dL) by 88.4]. In studies that also measured creatinine chromogen (which yielded results resembling those reported in most clinical laboratories), values were 0.83 mg per dL in nonpregnant women and decreased to 0.74, 0.58, and 0.53 mg per dL in the first, second, and third trimester of pregnancy, respectively. Thus, values considered normal in nongravid women may reflect decreased renal function during pregnancy.
P.216
For example, in gravid women, concentrations of serum creatinine exceeding 0.8 mg per dL or of serum urea nitrogen that are greater than 13 mg per dL suggest the need for additional evaluation of renal function.Other consequences of the increased renal hemodynamics. Increased GFR and RPF also alter urinary solute content. For example, excretion of glucose, most amino acids, and several water-soluble vitamins increases, and these increments in the nutrient content of urine may be a factor in the enhanced susceptibility of gravidas to urinary tract infections (UTIs). Urinary protein excretion also increases during gestation, but the fate of albumin excretion is more complex and disputed.
Acid-base regulation in pregnancy. Renal acid-base regulation is altered during gestation. The bicarbonate threshold decreases, and early-morning urines are often more alkaline than those in the nongravid state. In addition, plasma bicarbonate concentrations decrease approximately 4 mol per L, averaging 22 mol per L. This change most likely represents a compensatory renal response to hypocapnia, because pregnant women hyperventilate, and their Pco2 averages only 30 mm Hg. The mild alkalosis (arterial pH averages 7.44) found in pregnancy is in accord with this view. Because steady-state Pco2 and HCO3 levels are already diminished, pregnant women are, in theory, at a disadvantage when threatened by sudden metabolic acidosis (e.g., lactic acidosis in preeclampsia, diabetic ketoacidosis, or acute renal failure); however, they respond with appropriate increments in urinary titratable acid and ammonia after an acid load, and proton regeneration is already evident at blood pH levels higher than those in similarly tested nonpregnant women. Finally, when managing gravidas with pulmonary disorders, it should be noted that a Pco2 of 40 mm Hg, normal in nonpregnant women, signifies considerable carbon dioxide retention in pregnancy.
Water excretion. After conception, a rapid decrease in plasma osmolality levels of 5 to 10 mOsm per kg below that of nongravid subjects occurs. If this decrease occurred in a nonpregnant woman, she would cease secreting antidiuretic hormone and enter a state of water diuresis; however, gravidas maintain this new osmolality, diluting and concentrating urine appropriately when the woman is subjected to water loading or dehydration. This suggests a resetting of the osmoreceptor system, and, indeed, clinical studies demonstrate that the osmotic thresholds for both thirst and arginine vasopressin (AVP) release are decreased in pregnant women. Furthermore, the plasma of pregnant women contains large quantities of a placental enzyme (vasopressinase) capable of destroying substantial quantities of AVP in vitro; moreover, the in vivo production and metabolic clearance of antidiuretic hormone are increased fourfold after midgestation.
The changes in osmoregulation and AVP metabolism may be responsible for two unusual syndromes of transient diabetes insipidus that complicate pregnancy. One, in which polyuria is responsive to both AVP and deamino-8-d-arginine vasopressin (dDAVP), probably occurs in women with unapparent partial central diabetes insipidus whose disease is brought to the fore by the increment in hormonal disposal rates during late gestation. The other disorder, in which the marked polyuria continues despite large doses of AVP, is responsive to dDAVP, an analogue resistant to inactivation by vasopressinase. These gravidas may have excessively high circulating levels of the aminopeptidase enzyme due to increased activation.
Volume regulation. Most healthy women gain approximately 12.5 kg during the first pregnancy and 1 kg less during subsequent pregnancies. Generations of physicians have considered these averages as upper limits of permissible weight gains, forgetting that a mean has plus and minus deviations. As a result, many gravidas were scolded for excessive weight gain, and their salt, calories, or both were restricted. Most of the increment is fluid, with total body water increasing 6 to 8 L, 4 to 6 L of which is extracellular. Plasma volume increases 50% during gestation, the largest rate of increment occurring during midpregnancy, whereas increments in the interstitial space are greatest in the third trimester. A gradual cumulative retention of approximately 900 mEq of sodium occurs in pregnancy; this is distributed between the products of conception and the maternal extracellular space. These alterations in maternal intravascular and interstitial compartments produce apparent hypervolemia, yet the gravida's volume receptors sense these changes as normal. Thus, when salt restriction or diuretic therapy limits this physiologic expansion, maternal responses resemble those in salt-depleted nonpregnant women. This is one compelling reason for the reluctance to recommend sodium restriction or diuretics during pregnancy. Pregnant women are now advised to salt their food to taste, and some researchers believe that a liberal sodium intake is beneficial during gestation. Another physiologic adaptation that appears to influence sodium balance during pregnancy is the marked stimulation of the renin-angiotensin-aldosterone system. Aldosterone levels are markedly increased during pregnancy, despite normal blood pressure and normal potassium balance. It is likely that the increased aldosterone secretion is a compensatory mechanism to counteract the increase in sodium excretion that would be expected as a result of the large increase in GFR and renal plasma flow. Arterial vasodilation that causes relative arterial underfilling, as occurs in pregnancy, is known to stimulate the renin-angiotensin-aldosterone system. Moreover, increases in aldosterone balance the natriuretic effects of the large increases in progesterone during pregnancy.
Blood pressure regulation. Mean BP starts to decrease early in gestation, with diastolic levels in midpregnancy averaging 10 mm Hg less than measurements postpartum. In later pregnancy, BP increases, gradually approaching nonpregnancy values near term. Because cardiac output rises quickly in the first trimester and remains relatively constant thereafter,
P.218
the decrease in pressure is due to a marked decrement in peripheral vascular resistance. The slow rise toward nonpregnant levels after a midtrimester nadir is interesting, because it demonstrates that increasing vasoconstrictor tone is a feature of late gestation in normal women as well as in women in whom preeclampsia is developing. The cause of the decrease in peripheral resistance during pregnancy is obscure. Studies of arterial compliance in pregnancy demonstrate early rises, perhaps due to alterations in vessel ground substance. Elevations of plasma estrogen and progesterone to concentrations that may relax smooth muscle occur, and increments in vasodilating prostaglandins may also be present during gestation. Hormonally mediated increases in endothelial nitric oxide production may also contribute to the vasodilation in pregnancy. Despite lower BP, the levels of all components of the renin-angiotensin system are increased during pregnancy. Exaggerated hypotensive responses to converting enzyme inhibition in normal gravidas suggest that the increased renin-angiotensin system in pregnancy is a normal physiologic response to decreased BP and increased sodium excretion.Lack of awareness of the fluctuation in BP during normal gestation may lead to diagnostic errors. For example, women with mild essential hypertension often experience a decrease in BP during early pregnancy, and BP may even approach normal levels. They may be then erroneously labeled preeclamptic in the last trimester, when frankly elevated pressures occur.
Mineral metabolism. Serum calcium levels decrease in pregnancy, in conjunction with a decrement in circulating albumin concentrations. Ionized calcium levels, however, remain in the normal nonpregnant range. Striking changes relating to calcium regulatory hormones also occur during normal pregnancy. Production of 1,25, dihydroxy-vitamin D3 increases as early as the first trimester, reaching circulating levels that are approximately two times nonpregnant values. Gastrointestinal absorption of calcium increases, resulting in an absorptive hypercalciuria, with 24-hour urine excretion often exceeding 300 mg per day (in appropriately nourished individuals). Intact parathyroid hormone levels are lower during normal pregnancy.
P.217
Clinical evaluation of renal function in pregnancy
Examination of the urine. The association of proteinuria with eclampsia was first noted in the 1840s, and the science of prenatal care advanced dramatically when physicians began to systematically examine the urine of gravidas, primarily for albuminuria. In certain instances, latent renal disease is first uncovered by the detection of excessive protein excretion or microscopic hematuria during a routine prenatal evaluation.
Healthy nonpregnant women excrete considerably less than 100 mg of protein in the urine daily, but due to the relative imprecision and variability of testing methods used in hospital laboratories, proteinuria is not considered abnormal until it exceeds 150 mg per day. During pregnancy, protein excretion increases, and excretion up to 300 mg per day may still be normal. On occasion, a healthy gravida can excrete more than that amount. The problem is compounded by the fact that approximately 5% of healthy adolescents and young adults have postural proteinuria, which may become apparent only during pregnancy. In addition, postural proteinuria may increase near term, when gravidas tend to assume a more lordotic posture, which augments protein excretion. Another cause of increased proteinuria in pregnancy may be the compression of the renal veins by the enlarged uterus, especially when gravidas lie supine. Thus, when the gravida is tested for postural proteinuria (see Chapter 8), she should be positioned in lateral recumbency.
Few attempts have been made to quantitate the urine sediment in pregnancy. The excretion of both red and white blood cells may increase during normal gestation, and one to two red blood cells per high-powered field is acceptable in a urinalysis.
Renal function tests. The clearance of endogenous creatinine, the most satisfactory approximation of GFR in nongravid subjects, is equally useful for assessing renal function in gravidas. Gravidas, as well as nonpregnant women, show little variation (approximately 10% per day) in urinary creatinine excretion and, presumably, in creatinine production, which in a given woman is similar both during and after gestation. The lower limit of normal creatinine clearance during gestation should be 30% greater than the average of 110 to 115 mL per minute for nongravid women.
Acid excretion and urinary concentration and dilution are similar in gravid and nonpregnant women. Thus, tests such as ammonium loading (rarely indicated in gestation) give values similar to those in nongravid women. When examining urinary diluting ability, the clinician should be aware that supine posture can interfere with this test. Therefore, studies to detect minimal urinary osmolal concentrations should be performed with the patient lying on her side. However, although lateral recumbency is the required position for prenatal measurement of most renal function parameters, this posture interferes with tests of concentration. For example, a urine osmolality that was 800 mOsm per kg after overnight dehydration may decrease to 400 mOsm per kg within 1 hour through fluid mobilization from the extremities during bed rest, thus resulting in volume-induced inhibition of AVP secretion, a mild osmotic diuresis, or both. These observations demonstrate the importance of upright posture, such as quiet sitting, when maximum urinary concentration is measured in pregnancy.
Role of renal biopsy in pregnancy. Percutaneous renal biopsy is seldom performed during gestation. In fact, pregnancy was once considered a relative contraindication to the procedure because of early reports of excessive bleeding and other complications in gravid women. This view, however, stemmed from the period when many biopsies were performed in hypertensive patients at a time predating the understanding of the coagulation abnormalities that may occur in preeclamptic women. It is now evident that if the renal biopsy is performed in women with well-controlled BP and normal coagulation indices, morbidity is similar to that of nonpregnant patients. Our opinion is that renal biopsy should be considered only when renal function suddenly deteriorates remote from term and no obvious cause is present. This is because certain forms of rapidly progressive glomerulonephritis, when diagnosed early, may respond to aggressive treatment such as steroid pulses and, perhaps, plasma exchange. Another situation in which biopsy is recommended is symptomatic nephrotic syndrome. Although some might consider a therapeutic trial of steroids in such cases, we prefer to determine beforehand whether the lesion is likely to respond to steroids, because pregnancy is itself a hypercoagulable state prone to worsening by such treatment. On the other hand, proteinuria alone in a normotensive woman with well-preserved renal function who has neither marked hypoalbuminemia nor intolerable edema would lead us to examine the patient at more frequent intervals and defer the renal biopsy to the postpartum period. This is because the consensus among most investigators is that prognosis is determined primarily by the level of renal function and the presence or absence of hypertension rather than by the type of renal lesion (see section III.D). We take a similar position in the management of pregnancies with asymptomatic microscopic hematuria alone, when neither stone nor tumor is suggested by ultrasonography. Finally, renal biopsies should not be performed after gestational week 30, because at this stage the fetus will probably be delivered in any case, and the decision usually has to be made quickly and independent of biopsy results.
P.219
Renal disease in pregnancy
Asymptomatic bacteriuria. UTI is the most common renal problem occurring in pregnancy. The urine of gravidas supports bacterial growth better than that of nonpregnant women because of its increased nutrient content. This, as well as ureteral dilation, stasis, and occasional obstruction,
P.220
would be expected to increase the susceptibility of pregnant women to UTI. Surprisingly, this is not the case, and, with the exception of certain high-risk groups (diabetic patients and gravidas with sickle cell trait), the prevalence of asymptomatic bacteriuria during gestation varies between 4% and 7%, a value similar to that in sexually active nonpregnant women. The natural history of asymptomatic UTIs is, however, quite different in pregnancy.Although in the nonpregnant state asymptomatic bacteriuria is quite benign, progression to overt cystitis or pyelonephritis occurs in up to 40% of affected gravidas. Therefore, screening all pregnant women for the presence of asymptomatic bacteriuria and treating those with positive urine cultures is important.
Method of urine collection. Pregnant women contaminate midstream urine specimens more frequently. The incidence can be reduced by the use of multiple vulval washings combined with carefully supervised collection procedures. In some women, suprapubic aspiration is required to differentiate contamination from true infection. Pregnancy is not a contraindication to this procedure.
If the urine is sterile at the beginning of pregnancy, it usually remains so until term. Still, a small number (1% to 2%) of gravidas whose original urine cultures are negative subsequently have bacteriuria. Abnormal urinalysis and the presence of dysuria do not differentiate between contamination and true infection. For example, dysuria occurs in 30% of gravidas whose urines are sterile, and the urine may be infected and still contain fewer than two leukocytes per high-power field.
Method of treatment. The optimum way to manage asymptomatic UTI in pregnancy has not been precisely defined. In the earlier literature, some authors recommended continuous antibiotic treatment from the time the bacteriuria was detected until delivery. This was based on the belief that the relapse rate was high, and that most bacteriuric women have renal parenchymal involvement as opposed to bladder infection. However, it is now apparent that one-half of these infections involve only the bladder, and the majority of these patients are cured by standard short-course (or even single-dose) therapy. More than 90% of the uropathogens involved are aerobic gram-negative rods, usually Escherichia coli, and we recommend a 4 7-day course of the antibiotic to which the cultured organism is sensitive, preferably a short-acting sulfa drug, nitrofurantoin, amoxicillin, or a cephalosporin. This approach, when combined with surveillance for recurrent bacteriuria, has been shown to be quite effective.
Importance of postpartum evaluation. Asymptomatic UTI has been linked to premature labor, hypertension, and anemia during gestation, but these assertions have not been proved. On the other hand, an increased incidence of occult urinary tract pathology is present in these gravidas. Therefore, women with bacteriuria during pregnancy may benefit from evaluation of their urinary tract postpartum, especially those in whom the infection is resistant to therapy.
Symptomatic bacteriuria. The clinical approach to symptomatic UTI during gestation differs from that for asymptomatic bacteriuria.
Acute pyelonephritis. Pyelonephritis was a cause of maternal death in the preantibiotic era, and 3% of pregnant patients in a more recently reported series developed septic shock. At one time, symptomatic UTIs complicated almost 2% of all gestations, but prenatal screening combined with rapid treatment of asymptomatic bacteriuria has reduced this incidence to approximately 0.5%. The bacteriology of these infections resembles that in asymptomatic patients (predominantly E. coli), and most cases present after midpregnancy. The clinical presentation of pyelonephritis in pregnancy can be dramatic. As noted before, the disease caused maternal deaths in the preantibiotic era, and upper UTIs in gravidas are associated with exaggerated effects of endotoxemia,
P.221
including shock, respiratory distress syndrome, marked renal dysfunction, and hematologic and liver abnormalities. Symptomatic UTIs have also been implicated in the etiology of intrauterine growth retardation, prematurity, congenital anomalies, and fetal demise. Thus, although treatment of cystitis may take place in an outpatient setting, treatment of pyelonephritis should be aggressive and is best performed in the hospital.Most patients with pyelonephritis respond quickly, with defervescence within 48 to 72 hours. However, in contrast to patients with cystitis or asymptomatic bacteriuria, these women are likely to experience relapse. Thus, gravidas with acute pyelonephritis should receive the therapeutic dose of antibiotics for 2 to 3 weeks. Subsequently, we recommend continuous suppressive therapy during the remainder of pregnancy and for the first 2 weeks of the puerperium, because up to 60% of the patients will have a recurrence of pyelonephritis if left untreated. An alternative approach, frequent surveillance for recurrent infection with prompt treatment when significant bacteriuria is identified, has been claimed to be as effective as suppressive therapy.
Perirenal abscess and renal abscess formation or carbuncle, although infrequent complications of gestation, should be considered in the differential diagnosis of postpartum fever. It is important to recognize that a high incidence of positive urine cultures occurs in the postpartum period perhaps 17% to 20% in the first few days after delivery, decreasing to 4% after the third postpartum day. These cases, which resolve spontaneously, may reflect a temporary breakdown in the normal host antibacterial mechanisms in the immediate postpartum period rather than true infection.
Antibiotic use in pregnancy. The first-choice antibiotic for symptomatic infections changes from decade to decade because of the rapid emergence of resistant strains, thus resulting in the use of drugs that have not yet withstood the test of time for safety in pregnancy. We continue to recommend starting treatment with cephalosporins, because a significant percentage of community-acquired E. coli infections are resistant to ampicillin. For routine cystitis, nitrofurantoin is often effective, and it is acceptable during pregnancy.
The physician should also be aware of problems specific to the use of antibiotics in obstetrics and anticipate the potential fetal toxicity of agents that cross the placental barrier. (Information concerning drug safety during pregnancy is listed in the Physicians' Desk Reference, which is updated annually.) In brief, sulfa drugs should not be used near term, because they may precipitate kernicterus in the newborn. The anti folic acid activity of trimethoprim has been associated with anomalies such as cleft palate in animals, and this combination drug should also be avoided, at least before midpregnancy.
Aminoglycosides should also be prescribed sparingly, because streptomycin has been associated with fetal ototoxicity and nephrotoxicity. Fluoroquinolones cross the placenta and should be avoided if possible. Tetracyclines are contraindicated because they deposit in fetal bones and teeth and may cause severe reactions in the mother, including hepatic failure.
Acute renal failure (ARF)
Incidence. Before 1970, the incidence of ARF in pregnancy severe enough to require dialytic therapy was estimated at between 1 in 2,000 and 1 in 5,000 gestations, and it represented a considerable proportion of cases reported in large series. Since then, the number of patients with ARF from obstetric causes has declined markedly, and the incidence is now estimated to be less than 1 in 20,000 pregnancies. This trend, attributed to the liberalization of abortion laws and improvement of prenatal care, has not been shared by the poorer and less industrialized nations, in
P.222
which such patients compose up to 25% of referrals to dialysis centers and in which renal failure in pregnancy continues to be an important cause of maternal and fetal mortality.The frequency distribution of ARF during gestation was bimodal, with one peak early in pregnancy (12 to 18 weeks) comprising most of the cases associated with septic abortion, and a second peak between gestational week 35 and the puerperium, primarily due to preeclampsia and bleeding complications, especially placental abruption.
Causes. ARF in pregnancy can be induced by any of the disorders leading to renal failure in the general population, such as acute tubular necrosis (ATN). Early in pregnancy, the most common problems are prerenal disease due to hyperemesis gravidarum, and ATN resulting from a septic abortion. Several different uncommon disorders can lead to ARF later in pregnancy. Mild to moderately severe preeclampsia is not usually associated with renal failure, because renal function is generally maintained in the normal or near-normal range for a nonpregnant woman. A variant of preeclampsia, the HELLP syndrome (hemolysis, elevated liver enzymes, and low platelet count; see section VI.B) may be associated with significant renal dysfunction, especially if not treated promptly.
Thrombotic microangiopathy. An important and difficult differential diagnosis is that of ARF in late pregnancy in association with microangiopathic hemolytic anemia and thrombocytopenia. Pregnancy is considered to be a risk factor for thrombotic thrombocytopenia purpura/hemolytic uremic syndrome (TTP/HUS). However, whether the pathogenesis of these disorders in pregnancy is similar to that in nonpregnant individuals is unclear. TTP/HUS is rare in pregnancy, and must be distinguished from the HELLP variant of preeclampsia, a much more common condition. The distinction of these syndromes is important for therapeutic and prognostic reasons, but considerable overlap exists in their clinical and laboratory features. Features that may be helpful in making the diagnosis include timing of onset and the pattern of laboratory abnormalities which in TTP may include decreased levels of a von Willebrand cleaving protease. Preeclampsia typically develops in the third trimester, with only a few cases developing in the postpartum period, usually within a few days of delivery. TTP usually occurs antepartum, with many cases developing in the second trimester, as well as the third. HUS is usually a postpartum disease. Symptoms may begin antepartum, but most cases are diagnosed postpartum.
Preeclampsia is much more common than TTP/HUS, and it is usually preceded by hypertension and proteinuria. Renal failure is unusual even with severe cases, unless significant bleeding or hemodynamic instability, or marked disseminated intravascular coagulation (DIC) occurs. In some cases, preeclampsia develops in the immediate postpartum period, and when thrombocytopenia is severe, it may be indistinguishable from HUS. However, preeclampsia spontaneously recovers, whereas HUS only infrequently improves.
In contrast to TTP/HUS, preeclampsia may be associated with mild DIC and prolongation of prothrombin and partial thromboplastin time. Another laboratory feature of preeclampsia/HELLP syndrome that is not usually associated with TTP/HUS is marked elevations in liver enzymes. The presence of fever is more consistent with a diagnosis of TTP than preeclampsia or HUS. The main distinctive features of HUS are its tendency to occur in the postpartum period and the severity of the associated renal failure. Treatment of preeclampsia/HELLP syndrome is delivery and supportive care. More aggressive treatment is rarely indicated. Treatment of TTP/HUS includes plasma infusion or exchange and other modalities used in nonpregnant patients with
P.223
these disorders, although clinical trials of these modalities in pregnancy have not been performed.Bilateral renal cortical necrosis may be induced by abruptio placenta or other clinical events complicated by severe obstetric hemorrhage (e.g., uterine rupture). Both primary DIC and severe renal ischemia have been proposed as the initiating events. Affected patients typically present with oliguria or anuria, hematuria, and flank pain. Ultrasonography or computed tomographic (CT) scanning may demonstrate hypoechoic or hypodense areas in the renal cortex. Most patients require dialysis, but 20% to 40% have partial recovery of renal function.
Acute pyelonephritis. Some pregnant women may develop ARF in association with pyelonephritis.
Acute fatty liver of pregnancy (fatty infiltration of hepatocytes without inflammation or necrosis) is a rare complication of pregnancy that is associated with significant azotemia. Women with this disorder often complain of anorexia and occasionally of abdominal pain in the third trimester. Clinical features suggesting preeclampsia, including hypertension and proteinuria, are not uncommon. Laboratory tests reveal elevations in liver enzymes, hypoglycemia, hypofibrinogenemia, and prolonged partial thromboplastin time. Delivery is indicated, and most patients improve shortly afterwards.
Urinary tract obstruction. Pregnancy is associated with dilation of the collecting system, which is not usually associated with renal dysfunction. Rarely, complications such as large uterine fibroids, which may enlarge in the setting of pregnancy, can lead to obstructive uropathy. Rarely, acute urinary tract obstruction in pregnancy is induced by a kidney stone. Diagnosis can usually be made by ultrasonography. Often the stones pass spontaneously, but occasionally cystoscopy is necessary for insertion of a stent to remove a fragment of stone and relieve obstruction, particularly if there is sepsis or a solitary kidney.
The management of ARF occurring in gestation or immediately postpartum is similar to that in nongravid subjects (see Chapter 11), but several points peculiar to pregnancy deserve emphasis. Because uterine hemorrhage near term may be concealed and blood loss underestimated, any overt blood loss should be replaced early. Gravidas should be slightly overtransfused to forestall the development of acute tubular or cortical necrosis. Both peritoneal dialysis and hemodialysis have been successfully used in patients with obstetric-related acute renal failure. Neither pelvic peritonitis nor the enlarged uterus is a contraindication to the former method. In fact, this form of treatment is more gradual than hemodialysis and thus less likely to precipitate labor. Because urea, creatinine, and other metabolites that accumulate in uremia traverse the placenta, dialysis should be undertaken early, with the aim of maintaining the blood urea nitrogen at approximately 50 mg per dL. In essence, the advantages of early dialysis in nongravid patients are even more important for the pregnant patient, making arguments for prophylactic dialysis quite compelling. Excessive fluid removal should be avoided, because it may contribute to hemodynamic compromise, reduction of uteroplacental perfusion, and premature labor. Some obstetricians and perinatologists recommend continuous fetal monitoring during hemodialysis treatments, starting at midpregnancy. Finally, the physician should be aware of potential dehydration in the neonate, because the newborn usually undergoes a brisk urea-induced diuresis.
Pregnancy in women with preexisting renal disease. The current approach to management of pregnancy in women with chronic renal disease (CRD) is primarily based on retrospective studies, and more definitive views must await prospective data from large series in which clinical, pathologic, and
P.224
functional observations are correlated. Nevertheless, several generalizations can be made and some guidelines presented regarding gestation in women with chronic kidney dysfunction (Table 14-2).Table 14-2. Summary of Pregnancy in Women with Preexisting Renal Diseasea
Disease Comments Chronic glomerulonephritis and focal glomerular sclerosis (FGS) Increased incidence of high blood pressure occurs late in gestation, but usually no adverse effect results if renal function is preserved and hypertension is absent before gestation. Some disagree, believing that coagulation changes in pregnancy exacerbate disease, especially immunoglobulin A nephropathy, membranoproliferative glomerulonephritis, and FGS. Systemic lupus erythematosus (SLE) Controversial: prognosis is most favorable if disease is in remission 6 months or more before conception. Periarteritis nodosa and scleroderma Both fetal and maternal prognosis is poor. Therapeutic abortion should be considered. Diabetic nephropathy No adverse effect on the renal lesion. Increased frequency of infections. High incidence of heavy proteinuria and hypertension near term. Chronic pyelonephritis (infectious tubulointersti- tial disease) Bacteriuria in pregnancy may lead to exacerbation. Polycystic kidneys Few problems when function is preserved and hypertension is absent. However, the incidence of preeclampsia is increased. Urolithiasis Ureteral dilation and stasis do not seem to affect natural history, but infections can be more frequent. Stents have been successfully placed during gestation. Reflux nephropathy Prognosis disputed. We believe such pregnancies have a good outcome, although urinary infection may be more common. Previous urologic surgery Urinary tract infection is common with urinary diversion, and renal function may undergo reversible decrease. Cesarean section might be necessary to avoid disruption of the continence mechanism if artificial sphincters or neourethras have been constructed. After nephrectomy, solitary pelvic kidneys Pregnancy is well tolerated. Might be associated with other malformations of the urogenital tract. Dystocia occurs rarely with a pelvic kidney. aGeneralizations are for women with only mild renal dysfunction (serum creatinine level <1.5 mg/ dL) and without hypertension at conception. Prognosis. Counseling and managing women with CRD is based on the following general approach: Fertility and ability to sustain an uncomplicated pregnancy relate to the degree of functional impairment, and whether hypertension is present, and not to the underlying disorder.
Degree of impairment. Patients are arbitrarily considered in three categories: preserved or mildly impaired renal function (serum creatinine less than or at 1.4 mg per dL), moderate renal insufficiency (creatinine 1.5 to 3.0 mg per dL), and severe renal insufficiency (creatinine higher than or equal to 3 mg per dL). In Table 14-3 are summarized the
P.225
maternal and fetal prognosis in each category, and Table 14-4 depicts the improvement in pregnancy outcomes that has occurred since the 1950s, reflecting advances in prenatal and neonatal care. We generally counsel avoidance of conception in women who have moderate or severe renal dysfunction, because up to 40% of pregnancies in the former category are complicated by either hectic hypertension or sudden declines in GFR, which may not reverse after delivery. An even higher incidence of serious maternal problems occurs when renal insufficiency is severe. This is especially true for women receiving dialytic therapy, in whom fewer than 50% of the gestations succeed, and problems of extreme prematurity plague many of those that do. Notably, although we base prognosis primarily on the degree of functional impairment, the underlying disease may also play a role. Thus, all authorities recommend against pregnancy in women with scleroderma and periarteritis nodosa, and some have reservations concerning patients who have lupus erythematosus, immunoglobulin A nephropathy, or focal glomerular sclerosis.Table 14-3. Pregnancy and Renal Disease: Functional Renal Status and Prospectsa
Category Mild Moderate Severe Prospects Cr <1.5 mg/dL Cr 1.5 3.0 mg/dL Cr >3.0 mg/dL Pregnancy complications 25% 47% 86% Successful obstetric outcome 96% (85%) 90% (59%) 47% (8%) Long-term sequelae <3% (9%) 25% (71%) 53% (92%) Cr, serum creatinine. aEstimates are based on 1,862 women with 2,799 pregnancies (1973 1992) and do not include collagen diseases. Numbers in parentheses refer to prospects when complication(s) develop before 28 weeks' gestation. (From Davison JM, Lindheimer MD. Renal disorders. In: Creasy RK, Resnick RK, eds. Maternal fetal medicine, 3rd ed. Philadelphia: WB Saunders, 1994. Reprinted with permission.) Table 14-4. Renal Disease and Pregnancy: Improvements in Perinatal Mortality Over Four Decadesa
Morbidity and mortality (%) Renal disease Pregnancy outcome 1950s 1960s 1970s 1980s Mild Preterm delivery 8 10 19 25 (Cr <1.5 mg/dL) Perinatal mortality 18 15 7 <5 Moderate Preterm delivery 15 21 40 52 (Cr 1.5 3.0 mg/dL) Perinatal mortality 58 45 23 10 Severe Preterm delivery 100 100 100 100 (Cr >3.0 mg/dL) Perinatal mortality 100 91 58 53 Cr, serum creatinine. aEstimates are based on 2,952 women with 4,011 pregnancies (1954 1992) and do not include cases of lupus erythematosus. (From Davison JM, Lindheimer MD. Renal disorders. In: Creasy RK, Resnick RK, eds. Maternal fetal medicine, 3rd ed. Philadelphia: WB Saunders, 1994. Modified with permission.) Level of blood pressure (BP). The BP level at time of gestation is an important prognostic index. In the absence of hypertension, the natural history of most established renal parenchymal disease is unaffected by gestation (although preeclampsia may occur more readily).
P.226
In contrast, when renal disease and hypertension coexist, the gestation is more likely to be complicated, either by severe increments in BP or by additional reductions in renal function. We permit women with well-controlled BP and only mild renal dysfunction to conceive or continue gestation, but these gravidas must be seen frequently and should understand that their gestation must be terminated if renal function deteriorates or if their BP becomes difficult to control.Proteinuria. Urinary protein excretion, which increases in normal pregnancy, may increase markedly in pregnant women with underlying parenchymal renal disease. In one large series, one-third of the patients with preexisting renal disease developed nephrotic-range proteinuria during gestation. These increments do not necessarily reflect worsening of the underlying kidney disease.
Renal hemodynamics. Gravidas with kidney disorders who have only minimal renal dysfunction usually experience increments in GFR during gestation, even though levels do not reach those seen in normal pregnant women. Thus, a decrement in serum creatinine level early in pregnancy is a good prognostic sign. If serum creatinine levels before conception exceed 1.4 mg per dL, decrements during gestation are less common, and, as noted, the prognosis of such pregnancies is more guarded. Finally, Table 14-5 summarizes fetal and maternal outcomes of 906 pregnancies in women with various primary glomerular disorders. Of importance, the data are mainly retrospective, reflecting an important need for prospective observational studies.
Table 14-5. Maternal and Fetal Outcomes (%) in 558 Women with Primary Glomerulopathies
Renal decrease Renal lesion Pregnancies Spontaneous abortion Perinatal loss Preterm delivery Reversible function Progressive hypertension Reversible Permanent Focal glomerulosclerosis 85 3 23 32 13 5 32 10 Membranous glomerulopathy 110 12 4 35 3 2 22 3 Membranoproliferative 165 17 8 19 6 3 20 12 Immunoglobulin A nephropathy 268 5 15 21 12 2 25 12 Mesangial proliferative 278 5 12 9 2 3 36 7 Mean 906 8 13 19 8 3 27 9 (From Imbasciati E, Ponticelli C. Pregnancy and renal disease: predictors for fetal and maternal outcome [editorial]. Am J Nephrol 1991;11:353 362. Modified with permission.)
Glomerulonephritis. Absence of gravidas in large epidemiologic surveys of poststreptococcal glomerulonephritis is remarkable and has led to speculations that pregnancy protects women from this disease. However, this form of immune complex nephritis does occur rarely in gestation, in which it may mimic preeclampsia. Its prognosis is favorable, because in those instances in which the occurrence of acute poststreptococcal glomerulonephritis during gestation was properly documented, renal function recovered rapidly and the pregnancy usually had a successful outcome.
The prognosis of chronic glomerulonephritis during pregnancy is difficult to evaluate because most reported cases are poorly documented, especially the prepregnancy level of renal function and BP. Still, it appears that if proteinuria or abnormal urinary sediment is the sole manifestation of the disease, pregnancy proceeds normally. Some authorities disagree, claiming that women with membranoproliferative glomerulonephritis (MPGN) and immunoglobulin A nephropathy are more prone to exacerbations during pregnancy, a view we do not share. Although C3 nephritic factor may pass from mother to fetus, the neonate appears unaffected, and maternal complement levels may actually rise during gestation.
Hereditary nephritis is an uncommon disorder that may first be manifested during pregnancy, when women with this disease develop frank nephrotic syndrome. A variety of hereditary nephritis accompanied by platelet abnormalities has been described. Pregnancy in these women has been successful from a renal standpoint, but their gestations have been complicated by bleeding problems.
Collagen vascular disease
Lupus nephritis. The effect of gestation in women with lupus erythematosus who have renal involvement is difficult to evaluate, in part because of the unpredictable course of the disease regardless of pregnancy. Activity of the disease in the 6 months before conception is often a useful prognostic guide (the longer the remission, the better the outlook). Although most pregnancies, in the presence of preserved function, proceed uneventfully or are accompanied by only transient functional declines; in approximately 10%, gestation appears to cause permanent renal damage and to accelerate the renal disease. Also, placental transmission of maternal autoantibodies is associated with an increased frequency of
P.227
spontaneous abortion in these women, and certain anticytoplasmic antibodies (especially SS-A/Ro) cause a neonatal lupus syndrome characterized by congenital heart block, transient cutaneous lesions, or both. Women with systemic lupus erythematosus (SLE) have a high incidence of detectable levels of antiphospholipid antibodies (anticardiolipin antibodies, lupus anticoagulant). High titers of these antibodies are associated with several complications of pregnancy, including spontaneous fetal loss, hypertensive syndromes indistinguishable from preeclampsia, and thrombotic events including deep vein thrombosis, pulmonary embolus, myocardial infarction, and strokes. Also, pregnant women with circulating antiphospholipid antibodies can manifest a rare form of rapid renal failure postpartum, associated with glomerular thrombi. Thus, women with SLE should be screened for antiphospholipid antibodies early in gestation. The therapeutic approach when gravidas manifest antiphospholipid antibodies is disputed, and many would not treat asymptomatic patients who manifest low titers. However, when titers are elevated [more than 40 GPL (immunoglobulin G antiphospholipid level)], most authorities prescribe aspirin (80 to 325 mg per day). Heparin in combination with aspirin is recommended for patients with a history of thrombotic events and may also be advisable when titers are higher than 80 GPL.A flare of lupus nephritis may be difficult to distinguish from preeclampsia when a woman with a history of lupus develops worsening renal function, proteinuria, and hypertension. Elevation in liver enzymes and new onset severe hypertension is more consistent with preeclampsia. Hypocomplementemia, and severe nephritic syndrome without hypertension is more consistent with lupus nephritis. Often, a flare of nephritis in the third trimester appears to trigger superimposed preeclampsia, and improvement in BP and proteinuria occurs only after delivery. However, in the presence of abnormal serologic testing, it is often reasonable to treat worsening proteinuria and azotemia with increased prednisone in the hope that it will improve, particularly if the fetus is immature. However, close maternal and fetal surveillance is of utmost importance, and delivery should be considered in the setting of obvious signs of HELLP syndrome, accelerating hypertension and/or azotemia, and other signs of worsening maternal condition.
Previously, patients with lupus nephropathy were believed to be prone to relapse in the immediate puerperium, and some physicians still start or increase steroid treatment during and after delivery. Such views of stormy puerperium are now disputed, and most authorities institute or change therapy only if signs of increased or de novo disease activity appear.
Pregnancy in patients with periarteritis nodosa and scleroderma with renal involvement appears to be disastrous, possibly due to the associated hypertension, which frequently becomes malignant in nature. Earlier case reports document poor fetal prognosis and maternal deaths particularly in the era before angiotensin converting enzyme (ACE) inhibitors. Although such a poor prognosis partly reflects the selectivity of a handful of case reports, prudence dictates that pregnancies of gravidas with these diseases be terminated at an early stage until more is known about their natural history during pregnancy.
Diabetes mellitus is among the more common medical disorders encountered in the prenatal clinic. Many of the patients are juvenile (type I, insulin-dependent) diabetics, who probably harbor early microscopical changes in their kidneys. Nevertheless, most gestations in diabetic patients with normal renal function succeed, especially if blood glucose levels are maintained close to the normal range during the prenatal period. These diabetic women, however, have an increased prevalence of bacteriuria and an increased susceptibility to symptomatic infection during pregnancy. With these
P.228
exceptions, such women rarely have complications, although they seem to have a higher incidence of preeclampsia.The effects of gestation in diabetic patients with overt nephropathy are similar to those in women with other forms of renal parenchymal disease. Patients with the mildest functional impairment have little trouble (although more than 50% experience third-trimester hypertension and increased proteinuria, the latter sometimes massive), whereas the presence of hypertension before conception imparts a poorer prognosis. Older reports of frequent deteriorations reflect experience from centers in which the gravidas were managed with stringent salt restriction and prophylactic diuretics. Without such restrictions, women with biopsy-proven diabetic nephropathy actually display increases in GFR during gestation.
Nephrotic syndrome. The most common cause of nephrotic-range proteinuria (more than 3.5 g per day) in late pregnancy is preeclampsia, a diagnosis that may be missed when diastolic pressures are between 85 and 95 mm Hg. The fetal prognosis in preeclampsia with heavy proteinuria is poorer than in other preeclamptic states, but maternal prognosis is similar. Most of the usual causes of nephrotic syndrome, including membranous nephropathy, proliferative or membranoproliferative glomerulonephritis, lipoid nephrosis (nil disease), diabetic nephropathy, amyloidosis, and focal segmental glomerulosclerosis have been described in gravidas. Many of these conditions do not respond to corticosteroids, and some may be aggravated by them, underscoring the importance of establishing a tissue diagnosis before starting therapy.
One should not confuse physiologic changes during gestation with the exacerbation of a disease causing the nephrotic syndrome; many women with a variety of non-nephrotic renal disorders develop heavy proteinuria when pregnant. Such increments in urinary protein may relate to the increased renal hemodynamics, alterations in the glomerular barrier, and possibly a rise in renal vein pressure. Other alterations in pregnancy that simulate symptoms accompanying nephrotic syndrome include decrements in serum albumin (approximately 0.5 to 1.0 g per dL), increments in the levels of cholesterol and other circulating lipids, and edema, which can occur at one time or another in up to 80% of normal gestations. Accordingly, an entity once termed cyclic nephrotic syndrome of pregnancy reflects instances of quiescent mild renal disease, which becomes more apparent during gestation.
Management of nephrotic pregnant patients includes dietary replenishment of urine protein losses (in rare instances, gestation has been sustained until an appropriate delivery date using albumin infusions). Although restriction of dietary protein has been recommended in certain forms of nephrotic syndrome in nonpregnant patients, and has led to decrements in the excretion of and increments in levels of circulating albumin, such therapy should not be attempted in gravid women, because it could be deleterious to fetal development. Diuretics should be avoided, if possible, because these women are already oligemic, and further intravascular volume depletion might impair uteroplacental perfusion. Also, because BP normally declines during pregnancy, diuretics might provoke more ready circulatory collapse or thromboembolic episodes. Exceptions to this, however, are certain nephrotics who also manifest salt-sensitive hypertension (mainly women with diabetic nephropathy). In such cases, modest salt restriction, combined with the judicious use of diuretics, may avoid termination in the early third trimester because of poorly controlled BP.
Prognosis in most nephrotic gravidas with preserved function is good, but some claim that fetal outcome is poorer if full-blown nephrotic syndrome is already present very early in gestation. In addition, hypoalbuminemia may be associated with small-for-date newborns. Focal segmental glomerulosclerosis is a disease of varied (and often unknown) etiology whose natural history during gestation remains disputed. Some claim pregnancy leads to
P.229
irreversible functional loss and hypertension-sustained postpartum; others find the natural history of this entity in pregnancy similar to that of most other disorders.Tubulointerstitial disease
Chronic pyelonephritis. Dilation and stasis in the urinary tract make chronic pyelonephritis in gravidas more prone to exacerbation. These women should have a high fluid intake and should be told to rest frequently on their sides. The prognosis of pregnant women with noninfectious interstitial nephritis seems similar to that of pregnant women with glomerular disease. Renal function deterioration may occur rapidly in this group of patients when they are inadvertently salt restricted during gestation.
Reflux nephropathy. Some authors believe that pregnancy adversely influences the course of reflux nephropathy. Others note that patients who do poorly have preexisting hypertension and moderate functional insufficiency. Obviously, the propensity for UTI in these patients demands close scrutiny during pregnancy, including frequent urine cultures and prompt treatment when signs of UTI appear.
Adult dominant polycystic kidney disease may remain undetected in gestation. Careful questioning of gravidas for a family history of renal problems and judicious use of ultrasonography may lead to its earlier detection. Patients with minimal functional impairment have few complications, but a greater propensity toward preeclampsia exists. They are also prone to UTIs, and it may therefore be prudent to culture their urines more frequently. Hypertension usually accompanies or antedates the onset of functional deterioration, and pregnancy in such gravidas is more hazardous.
Some women with autosomal dominant polycystic kidney disease manifest cysts in their livers that may enlarge with repeated pregnancy as well as with oral contraceptive use. A high incidence of cerebral aneurysms also occurs in certain affected families. When aware of such family clustering, usually identified by a history of subarachnoid hemorrhages among relatives, the patient should undergo screening using magnetic resonance angiograph (MRA). If an aneurysm is detected, neurosurgical consultation should be obtained, and the obstetrician may wish to avoid natural labor. All these patients should undergo genetic counseling before pregnancy to ensure they are aware that 50% of their offspring are at risk. Finally, predicting the fetal outcome using molecular probes on cells cultured from the amniotic fluid is possible.
Solitary and pelvic kidneys. Women with solitary kidneys appear to tolerate gestation well. However, if the nephrectomy was performed for nephrolithiasis or chronic pyelonephritis, the remaining kidney is often infected. Patients with these conditions must be carefully scrutinized by frequent examination and culture of the urine throughout pregnancy and in the puerperium.
Pelvic kidneys are apparently associated with decreased fetal survival, often due to the presence of other malformations of the urogenital tract of the mother. In addition, dystocia may occur when the kidney is in the true pelvis.
Urolithiasis and hematuria. The prevalence of urolithiasis in gestation varies between 0.03% and 0.35% in the Western hemisphere. Many of the stones contain calcium, and some are infective in origin. The older literature tended to stress the dramatic complications that occur when calculi cause obstructive uropathy and infection supervenes, but more recent reports, including one survey of 148 gestations in 78 nonselected stone formers, suggest that pregnancy has little influence on the course of stone disease (although women with renal calculi may have an increased incidence of spontaneous abortions). It should be noted that most of the reported series focus on women whose calculi are mainly of the noninfective variety, and
P.230
little is known of the natural history of the more serious infected struvite stones during gestation. In any event, UTI in the presence of nephrolithiasis requires prompt and prolonged treatment (3 to 5 weeks), followed by suppressive therapy through the immediate puerperium, because the calculus may represent a nidus of infection resistant to sterilization.Experience with cystinuria in pregnancy is limited, but most women with this disease also do well in gestation. d-Penicillamine as used in these patients appears to have no apparent adverse effects on mother or fetus.
Renal calculi are among the most common causes of abdominal pain (of nonobstetric origin) requiring hospitalization during gestation, and, when complications suggest the need for surgical intervention, pregnancy should not be a deterrent to x-ray examination. If the stone obstructs the ureter, intervention with either uteteral stenting, percutaneous nephrostomy, or rarely, surgery, is indicated. Spontaneous gross or microscopical hematuria occasionally complicates an otherwise uneventful gestation. The differential diagnosis includes all causes of hematuria in nongravid patients (see Chapter 8), but frequently no etiology is demonstrable, and the bleeding subsides postpartum. It has been suggested that these events are due to the rupture of small veins around the dilatated renal pelvis. Hematuria may or may not occur in subsequent gestations. In any event, investigation of the hematuria can often be deferred until after delivery, and noninvasive techniques such as ultrasound and magnetic resonance imaging are helpful in arriving at such decisions.
Renal transplantation
Fetal and maternal complications. Several thousand pregnancies have been reported in allograft recipients. As expected, prognosis is better when the transplanted kidney comes from a living donor. Most (more than 90%) gestations allowed to proceed beyond the first trimester succeed, but both maternal and fetal complications, due in part to the immunosuppressive therapy, can be anticipated; these include steroidinduced hyperglycemia, severe hypertension, UTI, septicemia, ectopic pregnancy, and uterine rupture. Fetal problems such as intrauterine growth retardation, congenital anomalies, prematurity, hypoadrenalism, hepatic insufficiency, thrombocytopenia, and serious infection have been reported.
Suggested criteria for pregnancy. The following relative criteria for transplant recipients wishing conception are suggested:
Good health and stable renal function for 1 2 years after transplantation
Stature compatible with good obstetric outcome
Absent or at most minimal proteinuria
No or easily managed hypertension
No evidence of pelvicalyceal distention on an excretory urogram performed before attempting pregnancy
Serum creatinine of 2 mg per dL or less, preferably less than 1.4 mg per dL
Drug therapy: prednisone, 15 mg per day or less; azathioprine, 2 mg per kg per day or less; cyclosporine, less than 5 mg per kg per day
Several thousand woman have gone through pregnancy after renal transplantation, most of whom have received prednisone. Prednisone crosses the placenta in small amounts, and is not believed to adversely affect the fetus, although adrenal insufficiency and thymnic hypoplasia have been described. The well known impact of glucocorticoids on maternal BP and blood sugar are important issues during pregnancy. High doses of prednisone may be associated with premature rupture of membranes. Azathioprine has also been used extensively in pregnancy. Inactive metabolites appear in fetal blood, and high doses may lead to myelosuppression of the fetus. The National Transplantation Pregnancy Registry has reported on the experience with cyclosporine in pregnancy. This drug is associated with slightly lower birthweights, increased maternal hypertension and an increased frequency of preeclampsia, although
P.231
the data have been collected retrospectively without a control population. Cyclosporine levels may decrease during pregnancy. No information on whether or not drug dosage should be increased is available. If renal function is normal, and the patient is more than 2 years post transplantation, it is probably not necessary to increase the cyclosporine dosage to maintain prepregnancy blood levels.There is less experience with tacrolimus, although to date no serious concerns exist, and it appears similar to cyclosporine with respect to an association with premature delivery.
Although limited animal data regarding fetal effects of mycopheolate mofetil were a concern, no structural malformations have been noted among offspring exposed to this drug, although exposures are limited to less than 10 reported pregnancies. For this reason we do not recommend it during pregnancy. Limited experience exists with rapamycin, and this author is not aware of published reports of use during pregnancy.
Dialysis. Fertility is reduced in dialysis patients, and few women conceive once they have started dialysis. Most pregnancies occur during the first few years on dialysis. Although previously the outcomes of such pregnancies were poor, with only approximately 25% resulting in surviving infants, new information suggests that pregnancy in dialysis patients is successful as often as 30% to 50% of the time in pregnancies that reach the second trimester. Considerable problems exist in such pregnancies, however, and we do not encourage women on maintenance dialysis to conceive.
The National Registry for Pregnancy in Dialysis Patients has reported 318 pregnancies in women on dialysis. Prematurity is common, and approximately 85% of infants born to women who conceive after starting dialysis are born before 36 weeks' gestation. More than one-third of the infants reported by the registry weighed less than 1,500 g at birth, and growth restriction was common. Maternal complications are also common, including accelerated hypertension, and an increased maternal mortality rate. Management of pregnant women on dialysis should include attempts to minimize the uremic environment. In women with chronic renal disease, prophylactic dialysis should be considered when renal function is poor. In women already on dialysis, treatment time should be increased to five to seven dialysis sessions per week, with minimal heparinization and slow ultrafiltration to avoid dialysis hypotension and volume contraction. If peritoneal dialysis is used, decreasing exchange volumes and increasing exchange frequency is recommended. Adequate calorie and protein intake should be encouraged and supplemental vitamins prescribed. Antihypertensive therapy should be adjusted to be appropriate for pregnancy. Anemia should be corrected with iron, folic acid, and erythropoietin. Adequate calcium supplements should be administered but the dose of vitamin D may have to be decreased. And metabolic acidosis should be prevented. No evidence exists to support the superiority of one dialysis modality over another with respect to pregnancy outcome.
Hypertensive disorders of pregnancy. Hypertension during gestation remains a major cause of morbidity and death in both mother and child.
Of the many classifications proposed for hypertension complicating pregnancy, that of the Committee on Terminology of the American College of Obstetricians and Gynecologists (1972) has been the most useful. The National High Blood Pressure Education Program in the United States endorsed this system in 1990 and again in 2000. The four categories of hypertensive disorders in pregnancy are:
Preeclampsia. Preeclampsia, characterized by hypertension, proteinuria, edema, and, at times, coagulation and liver function abnormalities, occurs in late pregnancy (after 20 weeks), primarily in nulliparas. Third-trimester hypertension is defined as a BP of 140/90 mm Hg or greater (Korotkoff V) sustained for 4 to 6 hours.
Attempts have been made to categorize this disease as severe (e.g., diastolic and systolic pressures of 110 and 160 mm Hg or greater, heavy
P.232
proteinuria, oliguria, and neurologic symptoms) or mild. Because a seemingly mild preeclamptic patient (e.g., a teenage gravida with a systolic blood pressure of 140/85 mm Hg and trace proteinuria) may suddenly convulse (in which case the disease is called eclampsia, a complication associated with maternal mortality), terms such as mild and severe may be misleading. Hypertension during late pregnancy in a nullipara, whether or not other signs are present, is sufficient reason to consider hospitalization and treatment as if the patient were potentially preeclamptic.Chronic hypertension. Most women in this category have essential hypertension, but in some the elevated BP is secondary to such conditions as renal artery stenosis, coarctation of the aorta, renal disease, primary aldosteronism, and pheochromocytoma. Evidence of arteriolar disease and knowledge that hypertension was present before conception or early in gestation is helpful in establishing a diagnosis. Cocaine abuse may masquerade as chronic hypertension in pregnancy. Pheochromocytoma has a catastrophic outcome during pregnancy; thus, measurement of urinary catecholamines or vanillylmandelic acid excretion should be considered in selected hypertensive gravidas not previously evaluated.
Chronic hypertension with superimposed preeclampsia. Hypertensive women are at increased risk for the development of superimposed preeclampsia, and, when this occurs, maternal and fetal morbidity and mortality are greater than when preeclampsia develops in a previously normotensive woman. Many of the maternal deaths attributable to hypertensive disease occur in previously hypertensive women with superimposed preeclampsia
Gestational hypertension, which is high BP appearing first after midpregnancy, is distinguished from preeclampsia by the absence of proteinuria. This category is broad, and includes women who later develop diagnostic criteria for preeclampsia, as well as women with chronic hypertension in whom BP decreased in early pregnancy, masking the true diagnosis. Gestational hypertension that resolves postpartum, and which was not in retrospect preeclampsia, is more likely to occur in women who develop essential hypertension later in life.
The physician should be aware of two forms of hypertension that complicate the puerperium. One entity is called late postpartum eclampsia (hypertension and convulsions 48 hours to weeks after delivery). This disorder may be elicited by the administration of bromocriptine, a drug prescribed to suppress lactation. The second entity is postpartum hypertension in normotensive gravidas, presenting 2 weeks to 6 months after delivery, with normalization by the end of year 1. Its etiology is poorly understood.
Pathophysiology of preeclampsia. Preeclampsia is syndrome whose manifestations affect many organ systems, including brain, liver, kidney, blood vessels, and placenta. Thus, whereas the focus may be on hypertension and proteinuria, we must always be aware that such signs and symptoms may be minimal while other, life-threatening syndromes develop, including convulsions and liver failure, both often associated with thrombocytopenia, as well as signs of DIC.
The placenta may be critically involved in the genesis of preeclampsia, and failure of cytotrophoblastic invasion of the uterine spiral arteries is one of the earliest changes in this disorder. Thus, these vessels do not undergo the expected transformation into the dilated blood vessels characteristic of normal placentation. This aberration may underlie the poor placental perfusion and growth retardation characteristic of preeclampsia. The reason for the failure of the trophoblast to invade the uterine spiral arteries is obscure. Recent research has focused on the abnormal modulation of cytotrophoblast adhesion molecules, integrins, and abnormal vascular endothelial growth factor receptor-ligand interactions. The abnormal placentation leading to the maternal syndrome of preeclampsia is believed to occur early in pregnancy
P.233
(10 to 20 weeks gestation). Finally, recent evidence has implicated the production of antiangiogenic factors in the genesis of preeclampsia.The mediators of hypertension in preeclampsia are not clearly understood. Evidence suggests that vasoconstriction results from a complex interplay of hormonal and vascular alterations. With respect to hormonal factors, the renin-angiotensin system is stimulated in normal pregnancy and relatively suppressed in women with preeclampsia. However, preeclamptic patients are more sensitive to the pressor effects of angiotensin II, and therefore this pressor peptide may play a role in their elevated BP. Aldosterone levels are also lower in preeclamptic women than in women with normal pregnancies, although still higher than nonpregnant levels.
Recent evidence suggests that alterations in vascular endothelial cell function are important features of the pathophysiology of preeclampsia. Endothelial cells produce a variety of substances important in modulating vascular tone and coagulation [e.g., nitric oxide (NO), prostacyclin, and endothelin]. Animal studies of gestational hypertension and limited clinical investigations suggest that decreased NO and prostacyclin, and increased endothelin, all sequelae of endothelial cell dysfunction, cause the vasoconstriction, platelet aggregation, and increased intravascular coagulation that then leads to the maternal clinical manifestations of preeclampsia. More recently soluble vascular endothelial growth factor variants have been postulated to modulate endothelial dysfunction.
The ability to excrete sodium may be impaired in preeclampsia, but the degree to which this occurs varies, as severe disease can occur in the absence of edema (the dry preeclamptic patient). Even when edema is marked, plasma volume is below that for normal pregnancy, and hemoconcentration is often present. This latter phenomenon may relate to the development of a leaky vasculature (thus, hypoalbuminemia in this disease may have three components: renal protein loss, liver dysfunction, and extravasation from the intravascular to the interstitial space). A decrement or suboptimal increase in intravascular volume also appears to precede the onset of overt hypertension.
Cardiac output is often decreased, and central venous and pulmonary capillary wedge pressures are normal or low. Thus, high BP is maintained by a marked increase in peripheral resistance. The alterations in cardiac output, combined with the decrements in intravascular volume, and the fact that placental perfusion is decreased in preeclampsia are major reasons that most authorities discourage use of diuretics in this disease.
In one variant of preeclampsia, HELLP, coagulation abnormalities and liver dysfunction predominate, whereas hypertension and proteinuria may be minimal. This syndrome is life threatening, because platelet counts may plunge far below 100 to 103 per mm3, whereas transaminase and lactic acid dehydrogenase levels rise above 1,000 units per L and evidence of a marked microangiopathic hemolytic anemia appears on the peripheral blood smear, all in less than 24 hours. Early recognition of this HELLP variant and prompt termination of gestation are important; such action avoids substantial maternal morbidity.
The pathogenesis of the eclamptic convulsion is also poorly understood. Vasospasm, ischemia, and local hemorrhage may all play a role. The importance of hypertension per se in the genesis of the seizures is debated, because convulsions may be observed in women whose BP is only mildly elevated.
Kidney function and morphology in preeclampsia
GFR and RPF. Both GFR and RPF decrease in preeclampsia. The decrements approximate 25% in most instances, so that the GFR of preeclamptic women often remains above pregravid values. However, in rare instances, large decreases in function may occur and, on occasion, lead to acute tubular or cortical necrosis.
Uric acid. Changes occur in the renal handling of urate in preeclampsia. A decrease in the clearance of uric acid, accompanied by increments
P.234
in blood levels of this solute, may occur weeks before any clinical signs of the disease appear. In pregnancy, serum urate levels above 4.5 mg per dL are suspect [to convert to SI units ( mol/L), multiply mg/dL by 59.48]. The level of hyperuricemia also correlates with the severity of the preeclamptic renal lesion, as well as with fetal outcome.Increased proteinuria, which may be moderate or heavy, is a feature of preeclampsia, and the diagnosis is suspect in its absence. The magnitude of proteinuria does not appear to affect maternal prognosis, but protein excretion in the nephrotic range is associated with greater fetal loss.
Calcium. Studies have demonstrated that renal calcium handling is altered in preeclampsia, and that in contrast to normotensive gravidas, or those with chronic or transient hypertension, patients with preeclampsia demonstrate marked hypocalciuria. The basis for this abnormality is unknown.
Preeclampsia is accompanied by a characteristic histologic lesion: glomerular capillary endotheliosis (Fig. 14-2). In women diagnosed clinically as preeclamptic, this lesion is present in only about 85% of biopsies obtained from primiparas and in considerably fewer biopsies from multiparas. The remainder of patients have evidence of nephrosclerosis or another parenchymal disease. Some claim that preeclampsia is a cause of focal glomerular sclerosis, but others believe preeclampsia lesions to be completely reversible, with the presence of focal glomerular sclerosis reflecting preexisting nephrosclerosis or primary renal disease. Women with glomerular endotheliosis alone tend to have uneventful subsequent gestations, but when focal glomerular sclerosis or alterations in the renal vessels are present, hypertension is more likely to recur in later pregnancies.
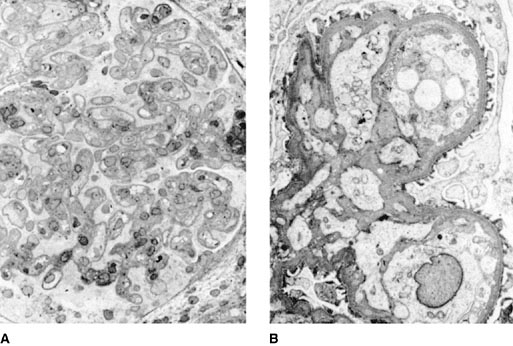
Figure 14-1. A: Electron micrograph demonstrating complete capillary obliteration by a swollen endothelial cell. Note, however, that the basement membrane is normal and the epithelial foot processes are intact. B: Micrograph showing glomerulus from a preeclamptic kidney. Swollen endothelial and mesangial cells that display prominent vacuolization encroach on the capillary lumina. (Courtesy of B. H. Spargo, M.D.)
Management of preeclampsia
Hospitalization. Ambulatory treatment is risky in the management of preeclampsia. Thus, suspicion of the disease is sufficient to consider hospitalization. Such an approach diminishes the frequency of convulsions and other consequences of diagnostic error. In general, fetal maturity is evaluated; if the gestation is near term, induction is the therapy of choice, whereas attempts to temporize are made if the pregnancy is at an earlier stage. Rest is an extremely important part of the therapeutic regimen, which must be prescribed rather than suggested. Termination of pregnancy should be considered when signs of impending eclampsia (e.g., hyperreflexia, headaches, epigastric pain) develop or persist; BP cannot be controlled; serum creatinine, urea nitrogen, and uric acid rise; laboratory evidence suggests DIC or abnormal liver function (increased transaminases); or specific obstetric tests suggest fetal jeopardy. When signs of impending convulsions (eclampsia) are present, parenteral magnesium sulfate is the drug of choice.
Treatment of hypertension. The approach to treatment of high BP in gravidas is disputed. As noted, morphologic examination of preeclamptic placentae demonstrates decreased trophoblastic invasion of the uterine spiral arteries, causing these vessels to be more constricted than normal. Thus, perfusion of the placenta is compromised. Therefore, aggressive reduction in maternal BP may decrease uteroplacental perfusion even further (i.e., poor autoregulation of uterine blood flow). Thus, large decrements in the mother's mean pressure should be avoided, especially in acute emergencies. Data on human pregnancy are limited, but they suggest that decrements in maternal pressure may indeed reduce placental perfusion. Others have argued that, based on evidence obtained from animal studies, uterine blood flow is autoregulated, and thus hypertension should be aggressively treated. Assuming that autoregulation of uterine blood flow exists, a critical but unanswered question is how quickly it takes place, because fetuses may be damaged by short periods of ischemia. In documented instances, precipitous decreases of pressure in response to diazoxide (even to diastolic levels above 85 mm Hg) were followed by immediate monitor signs of fetal distress. Thus, we prescribe the careful use of parenteral hydralazine or labetalol, in addition to close maternal scrutiny and fetal monitoring, when acute hypertension exceeds diastolic levels of 100 mm Hg or systolic levels of 150 mm Hg (Table 14-5). This approach is successful in most gravidas.
Calcium channel blockers have also been used to treat acute hypertension associated with preeclampsia. However, these agents should be considered only after hydralazine or labetalol has been unsuccessful, because anecdotal reports exist of precipitous decreases in BP when calcium channel blockers have been used concomitantly with magnesium sulfate.
Diazoxide can be used in rare resistant cases and should be administered only in small doses (30 mg at a time). Sodium nitroprusside should be avoided, because cyanide poisoning and fetal death have been observed in laboratory animals. ACE inhibitors and angiotensin receptor blockers (ARBs) should not be used in pregnancy.
Treatment of the eclamptic convulsion. Several large clinical trials have demonstrated that magnesium sulfate is superior to other anticonvulsants for prevention of recurrent eclamptic convulsions, and also, for primary prevention of eclampsia in women with preeclampsia. The usual protocol is to administer a loading dose of 4 g magnesium sulfate, infused over 15 minutes, followed by a sustaining infusion of 1 to 2 g per hour, aiming to achieve plasma levels of 2 to 4 mol per L. Because the incidence of convulsion is highest in the immediate puerperium, it is common practice to begin magnesium sulfate immediately after delivery and continue it for 24 hours.
Prevention of preeclampsia. Many strategies to prevent preeclampsia have been attempted over the years, including sodium restriction, diuretics, high-protein diets, and antihypertensive medication all have been unsuccessful. In the last decade, considerable resources have been allocated toward determining whether either low-dose aspirin or calcium supplementation are effective preventive strategies.
Several small studies conducted in the mid-1980s suggested that low-dose aspirin (50 to 150 mg per day), administered early in gestation may prevent preeclampsia. Since 1993, several large trials have now been completed, and most have not demonstrated a beneficial effect of aspirin for the prevention of preeclampsia. In a National Institutes of Health (NIH) sponsored randomized placebo-controlled trial of 3,135 healthy nulliparous women, 4.6% of the aspirin group and 6.3% of the placebo group developed preeclampsia, a barely significant difference. There was a slight increase in placental abruption in the group taking aspirin. A larger trial conducted largely in the United Kingdom (the CLASP trial) of 9,309 women at slightly increased risk failed to demonstrate any decrease in the incidence of preeclampsia in the treated group. In this study, there was no increase in placental abruption in the aspirin-treated patients. Several subsequent large trials in Brazil, Barbados, Jamaica, and the United States, with a total of more than 13,000 women at moderate or high risk found no significant differences in the incidence of preeclampsia, fetal growth restriction, preterm births, or adverse outcomes between women receiving aspirin and controls. A summary of all large trials conducted suggests that aspirin treatment may prevent the occurrence of early onset preeclampsia in approximately 15% of women at risk and reduce fetal growth restriction by 18%. A recent systematic review of 14 trials including over 12,000 women showed a small but significant benefit of aspirin therapy in reducing perinatal death (OR .79) and preeclampsia (OR .86), and an increase of 215 g in mean birth weight. Aspirin therapy is safe and not associated with increased risk of bleeding or placental abruption.
It has been suggested that the women enrolled in the earlier trials were too low risk to detect a significant benefit of aspirin; however, a multicenter trial of women with high-risk pregnancies (chronic hypertension, diabetes, multiple gestation) did not demonstrate a reduced incidence of preeclampsia in the treated group. The present data therefore do not support the prophylactic use of low-dose aspirin in pregnant women at low or medium risk for preeclampsia, or in women with mild chronic hypertension. However, low-dose aspirin may be effective in a defined group of predominantly parous women at high risk, not only in preventing preeclampsia but in reducing its severity.
Another preventive strategy that has received considerable attention is calcium supplementation. Low calcium intake has been implicated in the pathogenesis of preeclampsia, and a trial of 1,094 nulliparous pregnant women, as well as a meta-analysis, suggested that calcium supplementation with 2 g of calcium carbonate daily can prevent preeclampsia. Again, a large NIH multicenter randomized clinical trial of 4,589 women failed to support these earlier findings. However, subsequent trials conducted outside the United States have continued to demonstrate a beneficial effect of calcium supplementation for the prevention of preeclampsia. A possible explanation for the discrepancy in these results is the dietary calcium intake in these populations. In the NIH trial, both the placebo and treated groups had a baseline calcium intake of greater than 1,000 g per day. In contrast, in those trials in which calcium has been beneficial, the calcium intake in the population was well below the recommended dietary allowance for pregnancy. Thus, calcium supplementation may be beneficial in women with low calcium intake but may not be necessary if calcium intake is adequate.
Most other approaches to prevention, including fish oil, diuretics, sodium restriction, and magnesium supplementation have not been successful.
P.238
Currently under investigation is the ability of antioxidants (e.g. vitamins C and E) to prevent preeclampsia. Nevertheless, although prevention of preeclampsia is usually not possible, avoidance of severe complications may be accomplished by early recognition of the disease before such complications develop. If early signs are detected, hospitalization should be strongly considered to permit close monitoring of the patient. If preeclampsia is detected early, bedrest and close monitoring of maternal and fetal condition may enable prolongation of pregnancy in some cases.The hypertensive patient without preeclampsia. Increased risk is associated with pregnancy in women with chronic hypertension. Complications include superimposed preeclampsia, placental abruption, acute tubular and cortical necrosis, retardation of intrauterine growth, and midtrimester fetal death. Such events seem to correlate with the age of the gravida and the duration of her high BP. Thus, the majority of these complications occur in women over the age of 30 or with evidence of end-organ damage. Conversely, most women (approximately 85%) with essential hypertension have uncomplicated and successful gestations.
Women with chronic hypertension often have reductions in BP by midpregnancy, so that their BP may not exceed that observed in normotensive pregnant women. The failure of this decrement to occur, or increases in BP in early or midtrimester pregnancy, indicates a guarded prognosis for the gestation. Fetal outcome is poorer in hypertensive women with superimposed preeclampsia than in previously normotensive women with this complication, and the combination of chronic hypertension and preeclampsia seems responsible for most cases of cerebral hemorrhage in pregnancy. Patients with both conditions should be hospitalized and their hypertension controlled, but even with these precautions, most authorities consider delaying actions too risky and terminate the gestation.
Antihypertensive therapy. Guidelines for antihypertensive therapy during gestation are less clear than those for nonpregnant hypertensive women. For the latter, compelling data exist from large population studies to document the benefits of lowering BP with medication, even in women with only mild hypertension. During pregnancy, however, although maternal safety remains the primary concern, there is also a desire to minimize exposure of the fetus to drugs, given their unknown long-term effects on growth and development. A systematic review of clinical trials of hypertension found only 13 randomized clinical trials comparing antihypertensive therapy to either no treatment or placebo in women with chronic hypertension. The most commonly used drug, methyldopa, was given to just over 200 subjects. Six trials showed no reduction in perinatal mortality with antihypertensive treatment, whereas three reported a trend toward lower perinatal mortality with treatment. A debatable issue is whether lowering BP will prevent superimposed preeclampsia, but little or no convincing evidence supports this contention. Thus, it is permissible to tolerate higher BP levels during gestation that do no harm in the short term, while limiting use of antihypertensive drugs. In this respect, most pregnant women with chronic hypertension have only mild or very moderate elevations in BP and require little or no medication at all. However, appropriate or tolerable levels of BP for these patients during gestation seem to have been set empirically, and multicenter clinical trials are needed to support or reject such practices.
Whether mild degrees of hypertension during gestation should be treated is not generally agreed on. Although one group claimed that such therapy decreases the incidence of superimposed preeclampsia, most authorities would withhold treatment until diastolic levels are at least 15 mm Hg above borderline hypertension (defined in our clinics as 75 mm Hg
P.239
in the second trimester and 85 mm Hg in late gestation). Very young gravidas, however, may require treatment at lower levels. Tables 14-6 and 14-7 summarize current knowledge on the use of antihypertensive drugs in pregnancy. Only since the mid-1980s have serious efforts been undertaken to determine the safety and efficacy of these medications in pregnant women, and only a handful of these efforts have involved controlled trials. Future endeavors should increase the availability of this information.Table 14-6. Guidelines for Treating Severe Hypertension Near Term or During Labor
Regulation of blood pressure The degree to which blood pressure should be decreased is disputed; we recommend maintaining diastolic levels between 90 and 110 mm Hg. Drug therapy Hydralazine administered intravenously is the drug of choice. Start with low doses (5 mg as an intravenous bolus), then administer 5 to 10 mg every 20 to 30 min to avoid precipitous decreases in pressure. Side effects include tachycardia and headache. Labetalol, administered intravenously, is an effective and safe agent for preeclamptic hypertension. Start with 20 mg and repeat the dose every 20 min, up to 200 mg, until desired blood pressure is achieved. Side effects include headache. Favorable results have been reported with calcium channel blockers. If magnesium sulfate is being infused, magnesium may potentiate the effects of calcium channel blockers, resulting in precipitous and severe hypotension. Diazoxide should be used only in the rare instance that hydralazine, labetalol, or calcium channel blockers have been unsuccessful. Small doses (30 mg at a time) should be used. Side effects include arrest of labor and neonatal hypoglycemia. Refrain from using sodium nitroprusside, because fetal cyanide poisoning has been reported in animals. However, maternal well-being should dictate the choice of therapy. Prevention of convulsions Parenteral magnesium sulfate is the drug of choice for preventing eclamptic convulsions. Therapy should be continued for 12 to 24 hr postpartum, because one-third of women with eclampsia have convulsions during this period. Table 14-7. Antihypertensive Drugs Used to Treat Chronic Hypertension in Pregnancy
Alpha2 adrenergic receptor agonists Methyldopa is the most extensively used drug in this group. Its safety and efficacy are supported by evidence from randomized trials and a 7.5-year follow-up study of children born to mothers treated with methyldopa Beta adrenergic receptor antagonists These drugs, especially atenolol and metoprolol, appear to be safe and efficacious in late pregnancy, but fetal growth retardation has been reported when treatment was started in early or midgestation. Fetal bradycardia can occur, and animal studies suggest that the fetus's ability to tolerate hypoxic stress may be compromised. Alpha adrenergic receptor and beta adrenergic receptor antagonists Labetalol appears to be as effective as methyldopa, but no follow-up studies of children born to mothers given labetalol have been carried out, and concern about maternal hepatotoxicity still exists. Arteriolar vasodilators Hydralazine is frequently used as adjunctive therapy with methyldopa and beta adrenergic receptor antagonists. Rarely, neonatal thrombocytopenia has been reported. Trials with calcium channel blockers look promising. The experience with minoxidil is limited, and this drug is not recommended. Calcium channel blockers Small, uncontrolled studies suggest that these agents are safe and effective in pregnancy. Angiotensin converting enzyme (ACE) inhibitors Captopril causes fetal death in diverse animal species, and several converting enzyme inhibitors have been associated with oligohydramnios and neonatal renal failure when administered to humans. Do not use in pregnancy. Angiotensin II receptor blockers These drugs have not been used in pregnancy. In view of the deleterious effects of blocking angiotensin II generation with ACE inhibitors, angiotensin II receptor antagonists are also considered to be contraindicated in pregnancy. Diuretics Many authorities discourage the use of diuretics, but others continue these medications if they were prescribed before conception or if a woman with chronic hypertension appears quite sensitive to salt. (From Cunningham FG, Lindheimer MC. Hypertension in pregnancy [see comments]. N Engl J Med 1992;326:927 932. Reprinted with permission.) I recommend the guidelines of the National High Blood Pressure Education Program's Working Group Report on High Blood Pressure in Pregnancy, in which antihypertensive drug treatment is begun only when maternal BP reaches diastolic levels of 100 mm Hg or more. Exceptions, however, include parenchymal renal disease and evidence of target organ damage (e.g., retinopathy and cardiac hypertrophy), in which therapy is recommended once levels are 90 mm Hg or more.
The argument of whether to treat is debatable when fetal well-being is considered. Some evidence suggests fetal benefits when mild to moderate hypertension is treated with antihypertensive drugs during pregnancy.
In summary, the unknown but potential hazards of antihypertensive treatment during pregnancy are sufficient reasons for withholding drug treatment when mild hypertension (diastolic levels of 90 to 99 mm Hg) is present, particularly during the initial trimester. As noted, many of these patients experience a physiologic decrease in BP that on occasion reaches normotensive levels. Otherwise normal patients whose levels are 100 mm Hg or more, however, should be treated. Patients with evidence of renal disease or end-organ damage require the initiation of treatment at lower levels (less than 90 mm Hg).
P.235
P.236
P.237
P.240
Suggested Readings
Armenti VT, Ahlswede KM, Ahlswede BA, et al. Variables affecting birthweight and graft survival in 197 pregnancies in cyclosporine-treated female kidney transplant recipients. Transplantation 1995;59:476 479.
August P, Lindheimer MD. Chronic hypertension in pregnancy. In: Lindheimer MD, Roberts JM, Cunningham FG, eds. Chesley's hypertensive disorders in pregnancy, 2nd ed. Norwalk, CN: Appleton & Lange, 1999:605 633.
August P, Lindheimer MD. Pathophysiology of preeclampsia. In: Laragh JH, Brenner BM, eds. Hypertension: pathophysiology, diagnosis, and management. New York: Raven Press, 1995;2407 2426.
Burke JF. Variables affecting birthweight and graft survival in 197 pregnancies in cyclosporine-treated female kidney transplant recipients. Transplantation 1995;59:476 479.
P.241
Cadnapaphornchai MA, Ohara M, Morris KG, Knotek M, Ragachev B, Ladtkow T, Carter EP, Schrier RW. Chronic NOS inhibition reverses systemic vasodilation and glomerular hyperfiltration in pregnancy. Am J Physiol Renal Physiol 2001;280:F592 F598.
Chapman AB, Abraham WT, Zamudio S, Coffin C, et al. Temporal relationships between hormonal and hemodynamic changes in early human pregnancy. Kid Int 1998;54:2056 2063.
Conrad KP. Mechanisms of renal vasodilation in pregnancy. J Soc gynec Invest in press 2004.
Conrad KP and Lindheimer MK. Renal and cardiovascular alterations. In: Lindheimer MD, Roberts JM, and Cunningham FG, eds. Chelsey's hypertensive disorders in pregnancy, 2nd ed. Stamford, CT: Appleton & Lange, 1999:2263 2326.
Cunningham FC, Lucus MJ. Urinary tract infections complicating pregnancy. Clin Obstet Gynaecol (Bailli re) 1994;8:363 383.
Davison JM. Pregnancy in renal allograft recipients: problems, prognosis, and practicalities. Clin Obstet Gynaecol (Bailli re) 1994;8:511 535.
Davison JM, Dunlop M. Renal hemodynamics and tubular function in normal human pregnancy. Kid Int 1980;18:152.
Davison JM, Lindheimer MD; guest editors. New Developments in preeclampsia. Seminars in Nephrol in press 2004.
Hou S. Pregnancy in chronic renal insufficiency and end-stage renal disease. Am J Kidney Dis 1999;33:235 252.
Jones DC, Hayslett JP. Outcome of pregnancy in women with moderate or severe renal insufficiency. N Engl J Med 1996;335:226 232.
Jungers P, Chauveau D. Pregnancy in renal disease. Kidney Int 1997;52:871 885.
Lindheimer MD, Katz AI. Gestation in women with renal disease: prognosis and management. Clin Obstet Gynaecol (Bailli re) 1994;8:397 414.
Lindheimer MD, Katz AI. Renal physiology and disease in pregnancy. In: Seldin DW, Giebisch G, eds. The kidney: physiology and pathophysiology. 3rd ed. Philadelphia: Lippincott Williams & Wilkins, 2000:1260 1265.
National High Blood Pressure Education Program Working Group Report on High Blood Pressure in Pregnancy. Am J Obstet Gynecol 2000;183:S1 S22.
Okundaye IB, Agrinko P, Hou S. A registry for pregnancy in dialysis patients. Am J Kidney Dis 1998;31:766 773.
Pertuisset N, Gr nfeld J-P. Acute renal failure in pregnancy. Clin Obstet Gynaecol (Bailli re) 1994;8:343 361.
Roberts JM, Redman CWG. Pre-eclampsia: more than pregnancy-induced hypertension. Lancet 1993;341:1447 1451.
Sibai BM. Drug therapy: treatment of hypertension in pregnant women. N Engl J Med 1996;335:257 265.