14. The Respiratory System
Authors: Corwin, Elizabeth J.
Title: Handbook of Pathophysiology, 3rd Edition
Copyright 2008 Lippincott Williams & Wilkins
> Table of Contents > Unit IV - Oxygen Balance and Deficiencies > Chapter 14 - The Respiratory System
Chapter 14
The Respiratory System
The respiratory system has a vital charge: to provide for the exchange of oxygen and carbon dioxide between the air and the blood. Oxygen is required by all cells so that the life-sustaining energy source, adenosine triphosphate (ATP), can be produced. Carbon dioxide is produced by metabolically active cells and forms an acid that must be removed from the body. For gas exchange to be performed, the cardiovascular and respiratory systems must work together. The cardiovascular system is responsible for perfusion of blood through the lungs. The respiratory system performs two separate functions: ventilation and respiration.
Physiologic Concepts
Alveolus
The functional unit of the lungs is the alveolus (plural, alveoli). There are more than a million alveoli in each lung. Alveoli are small, air-filled sacs across which oxygen and carbon dioxide and other gases diffuse. The large number of small alveoli ensures that the total area available for the diffusion of gas in each lung is enormous. If the airflow into an alveolus is blocked, it collapses and is unavailable for gas exchange. If airflow into several alveoli is blocked, exchange of gases may be impaired to the extent that the person becomes hypoxic or unconscious or dies.
P.465
Ventilation
The movement of air from the atmosphere into and out of the lungs is called ventilation. Ventilation occurs by bulk flow. Bulk flow is the movement of a gas or a fluid from high to low pressure.
Factors That Affect Ventilation
Ventilation is determined by the variables in Equation 14-1:
![]()
where F is the bulk flow of air, P is the difference in pressure between the atmosphere and the alveoli, and R is the resistance offered by the conducting airways.
Pressure
Alveolar pressure varies with each inspiration and drives the flow of air. With the onset of inspiration, the thoracic cavity expands. As the thoracic cavity expands, the lungs also expand. According to Boyle's law, if the volume of an air-filled chamber increases, the pressure of the air in the chamber decreases. Therefore, as the lungs expand, pressure in the alveoli decreases to below atmospheric pressure, and air rushes into the lungs from the atmosphere (from high pressure to low pressure). At the end of inspiration, the thoracic cavity relaxes, causing pressures in the alveoli, which are filled with the air of inspiration, to be higher than in the atmosphere. Air then flows out of the lungs and down the pressure gradient.
Bronchial Resistance
Resistance of the airways is usually low. Resistance is increased when the smooth muscle of the bronchiolar tubes constricts. Constriction of the bronchi results in a decrease in airflow into the lungs. Resistance is inversely proportional to the radius of a vessel to the fourth power. This means that if the radius of a bronchiolar tube decreases by one-half, the resistance to airflow in that tube increases by 16 (i.e., 24). Therefore, when the air passages constrict even slightly, resistance to airflow goes up significantly.
Bronchiolar resistance is determined by parasympathetic and sympathetic nervous system innervation of the smooth muscle of the bronchi and local chemical mediators.
Parasympathetic nerves are carried to the bronchial smooth muscle by way of the vagus nerve and cause contraction or narrowing of the airways, increasing resistance and reducing airflow. Parasympathetic nerves release the neurotransmitter acetylcholine (ACh). ACh acts by binding to cholinergic receptors on the smooth muscle of the bronchi.
Sympathetic innervation of the bronchial smooth muscle occurs by way of nerve fibers from the upper thoracic and cervical ganglia and causes relaxation of the bronchi. This reduces resistance and increases
P.466
airflow. Sympathetic nerves release the neurotransmitter norepinephrine. Norepinephrine acts by binding to 2 adrenergic receptors on the smooth muscle of the bronchi.
Nervous Control of Respiration
Ventilation is controlled by the respiratory center in the lower brainstem areas of the medulla and pons. In the medulla, there are inspiratory and expiratory neurons that fire at opposite times in a preset pattern of rate and rhythm. Respiratory neurons drive ventilation by exciting motor neurons that innervate the main muscle of inspiration (the diaphragm) and the accessory muscles (the intercostal muscles).
Central Chemoreceptors
Central chemoreceptors in the brain respond to changes in the hydrogen ion concentration of the cerebral spinal fluid. Increased hydrogen ion concentration increases the firing rate of the chemoreceptors, while decreased hydrogen ion concentration decreases the firing rate of the chemoreceptors. Information from the central chemoreceptors is delivered to the respiratory center in the brain, which in response increases or decreases the breathing pattern. Hydrogen ion concentration usually reflects carbon dioxide concentration. Therefore, when carbon dioxide levels rise, hydrogen ion levels rise, and the firing rate of inspiratory neurons is increased, causing an increase in respiratory rate. This is an example of negative feedback, because with an increase in the rate of breathing, the excess carbon dioxide and hydrogen ion will be blown off. With low carbon dioxide and low hydrogen ion levels, the firing rate of the inspiratory neurons returns toward baseline, and respiration slows.
Peripheral Chemoreceptors
Peripheral chemoreceptors exist in the carotid and the aortic arteries, and monitor oxygen concentration in arterial blood. These receptors, called the carotid and the aortic bodies, send their impulses to the respiratory center of the medulla and pons primarily to increase the rate of ventilation when oxygen is low. They are less sensitive than the central chemoreceptors. The peripheral chemoreceptors also respond with an increase in firing rate to increased hydrogen ion dissolved in the blood. This is important because under certain circumstances free hydrogen ion increases without causing a change in carbon dioxide concentration (e.g., during conditions of metabolic acidosis caused by prolonged diarrhea or diabetes mellitus). Free hydrogen ion is relatively impermeable across the blood-brain barrier, so it is unable to activate the central chemoreceptors directly.
Motor Neurons Driving Respiration
The major motor neuron controlling respiration is the phrenic nerve. When activated by the central inspiratory neurons, the phrenic nerve causes the diaphragm muscle to contract and the chest to expand. As the
P.467
chest expands, air begins to flow from the atmosphere into the lungs. Airflow into the lungs is called inspiration. As inspiration continues, firing of the central inspiratory neurons slows and firing of the expiratory neurons increases, causing cessation of motor neuron activity and relaxation of the diaphragm. Chest expansion reverses and air flows out of the lungs. Airflow out of the lungs is called expiration.
Respiration
Respiration refers to the diffusion of gases between an alveolus and the capillary that perfuses it. Respiration occurs by diffusion, which involves the movement of a gas down its concentration gradient.
Factors That Affect Respiration
The rate of diffusion of a gas (e.g., oxygen and carbon dioxide) is determined with Equation 14-2:
![]()
where [D with dot above] is the rate of diffusion, Xa is the concentration of gas in the alveolus, Xc is the concentration of gas in the capillary, SA is the surface area available for diffusion, T is the temperature of the solution, d is the distance across which diffusion must occur, and k is a physical constant that takes into account non-variable characteristics of the gas such as its molecular weight and its specific solubility coefficient.
Concentration of Oxygen and Carbon Dioxide in the Alveolus and the Capillary
Alveolar oxygen concentration reflects atmospheric oxygen, whereas pulmonary capillary oxygen concentration reflects the oxygen concentration of systemic venous blood. Because systemic venous blood is blood returning from the peripheral circulation, where much of the oxygen has been used by the cells of the body, it has a low oxygen concentration. The atmosphere is typically well supplied with oxygen. Therefore, oxygen is normally in higher concentration in the alveolus than it is in the pulmonary capillary. Values for oxygen concentration are directly proportional to the partial pressure of the gas and are usually expressed in millimeters of mercury (mm Hg).
At sea level, the partial pressure of oxygen is approximately 100 mmHg in the alveolus and 40 mmHg in the pulmonary capillary. Because alveolar oxygen concentration in the alveolus is greater than in the capillary, oxygen diffuses down its concentration gradient from the alveolus into the capillary. This is how deoxygenated blood is replenished with oxygen by respiration.
Carbon dioxide normally diffuses in the opposite direction. It is in low concentration in the atmosphere and thus in low concentration in the alveolus (40 mmHg). Pulmonary capillary blood reflects systemic venous
P.468
blood. Because carbon dioxide is a waste product of metabolizing cells, the concentration of carbon dioxide in the capillary is high (46 mmHg). Therefore, in the lungs, carbon dioxide diffuses down its concentration gradient, from the blood into the alveolus, where it will be expired.
Under some circumstances, concentration gradients of oxygen and carbon dioxide between the blood and the alveolus may be increased or decreased; magnified concentration gradients affect the diffusion rate of the gas. For example, during exercise, oxygen concentration in the blood entering the pulmonary capillaries may be less than 40 mmHg because the exercising muscles have increased their oxygen usage. Carbon dioxide concentration would be greater in blood flowing to the lungs from exercising tissue because its metabolic production would be increased. In this situation, diffusion rates for both gases would be increased, allowing more oxygen to diffuse into the blood and more carbon dioxide to diffuse out of the blood.
Surface Area
Surface area (SA) refers to the expanse of alveolar and capillary membranes available for gas diffusion. Surface area is normally high in the lungs. Some diseases, including emphysema, tuberculosis, and lung cancer, can decrease the surface area available for diffusion, thus reducing the diffusion rates of oxygen and carbon dioxide.
Distance for Diffusion
The distance (d) across which oxygen and carbon dioxide must diffuse is normally quite small. Alveolar and capillary membranes are close to each other, separated only by a thin layer of interstitial fluid (Fig. 14-1).
Certain conditions, including pneumonia, can increase the distance of diffusion by causing edema and swelling of the interstitial space. This
P.469
decreases the diffusion rate of the gases (Fig. 14-2). Interstitial fibrosis (scarring) can also increase the distance between the alveoli and the capillaries and so slow diffusion.
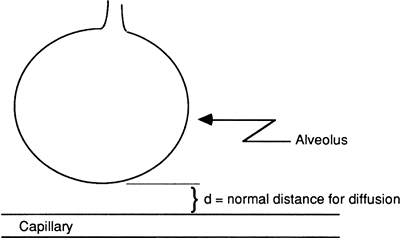 |
Figure 14-1. Normal alveolus capillary distance allows for efficient diffusion of oxygen and carbon dioxide between the capillary and alveolus. |
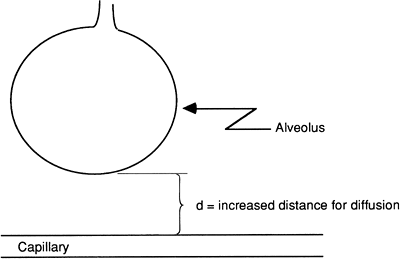 |
Figure 14-2. With interstitial edema, the alveolus capillary distance is increased, resulting in reduced diffusion of oxygen and carbon dioxide between the capillary and the alveolus. |
Temperature
A decrease in temperature (T) would decrease the diffusion rate of oxygen and carbon dioxide. An increase in T would increase the diffusion rate of both gases. An increase in temperature may play a role in meeting the increased metabolic demands during fever.
Permeability Coefficient
Carbon dioxide and, to a lesser extent, oxygen have high permeability. Because the variable k in the equation is fixed for each gas, k does not play an active role in determining respiration.
Oxygen Carrying in the Blood
Oxygen is carried in the blood in dissolved form and bound to hemoglobin. The amount of oxygen dissolved in the blood depends on the partial pressure of oxygen in the air entering the alveoli and the solubility of oxygen. Normally the amount carried dissolved is small (only approximately 3 mL/L). Instead, most oxygen (98%) is carried bound to hemoglobin.
Hemoglobin is a protein molecule composed of four subunits, each combining a globulin molecule with a molecule of iron. Each iron molecule has a binding site for oxygen. Dissolved oxygen combines with hemoglobin until all four sites are saturated. At normal arterial oxygen concentration of 100 mmHg, nearly 100% of hemoglobin molecules are saturated with oxygen. Even in the venous blood, with a reduced oxygen
P.470
concentration of 40 mmHg, hemoglobin is still at least 75% saturated with oxygen (Fig. 14-3). The ability of hemoglobin to bind oxygen is reduced by increased hydrogen ion concentration, increased temperature, and increased amount of a substance produced by red blood cells during glycolysis, 2,3-diphosphoglycerate (DPG). A reduced affinity for oxygen means that hemoglobin releases oxygen to the tissues more readily. Increases in hydrogen ion, temperature, and DPG occur during periods of increased metabolism; therefore, decreased hemoglobin affinity releases more oxygen to a cell and allows it to meet its elevated metabolic demands.
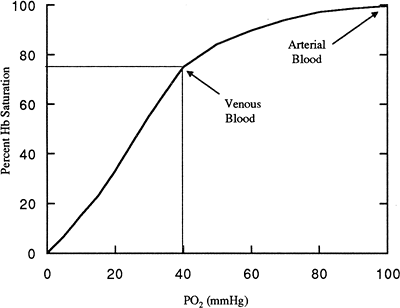 |
Figure 14-3. Oxygen-hemoglobin saturation curve. Notice that at arterial O2 levels (PO2), nearly 100% of hemoglobin is saturated. Even at PO2 of 60 mmHg, 90% of Hb is saturated. In venous blood (PO2 40), 75% of Hb is saturated with blood. |
Perfusion
For the respiratory system, perfusion refers to the movement of blood in the pulmonary vascular system, past the alveolar capillaries. Perfusion, like blood flow and like ventilation, occurs by bulk flow. In the lungs, perfusion and ventilation usually are well matched. This ensures that there is adequate oxygen available in each alveolus to replenish the blood flowing past it and adequate blood flow to support each alveolus.
Circulations That Provide Blood Flow to the Lungs
Two separate blood circulations supply blood flow to the lungs from the heart: the pulmonary circulation and the bronchial circulation.
P.471
Pulmonary Circulation
The pulmonary circulation consists of deoxygenated blood traveling in the pulmonary artery from the right side of the heart. This blood perfuses the respiratory portions of the lungs and participates in the exchange of oxygen and carbon dioxide across the capillaries and alveoli. After picking up oxygen and releasing carbon dioxide, the blood returns to the heart by way of the pulmonary vein. Pressure and resistance to flow in the pulmonary circulation are usually low, with a mean pulmonary pressure of approximately 12 mmHg compared with a mean systemic pressure of approximately 90 mmHg. The pulmonary circulation is compliant and can accommodate large variations in blood volume. Therefore, the pulmonary circulation can act as a reservoir for blood that can be called upon in times of decreased systemic blood volume or pressure.
Bronchial Circulation
The bronchial circulation carries blood from the left side of the heart to the lungs through the thoracic aorta. The bronchial circulation accounts for approximately 8 to 9% of the total cardiac output. Blood in the bronchial circulation is well oxygenated and supplies oxygen to the structures of the lungs not involved in the exchange of gases, including the connective tissue and the large and small bronchi. Blood returns to the left side of the heart through the pulmonary vein. Returning bronchial blood is deoxygenated because it has been used by metabolically active cells of the lungs but has not been involved in gas exchange. This blood mixes with the well-oxygenated blood coming from the pulmonary circulation also back to the left side of the heart, and slightly reduces the overall oxygen concentration of that blood.
Ventilation:Perfusion Ratio
Ventilation refers to air moving into and out of the lungs. Perfusion is the blood passing through the pulmonary circulation to be oxygenated. The ventilation:perfusion ratio, V/Q, is the ratio of airflow into the lungs divided by the pulmonary blood flow. In this expression, V is the volume of air moved with each breath, expressed as milliliters per minute (mL/min), and Q is the rate of blood flow in the pulmonary circulation, also expressed as mL/min. Normally, perfusion is slightly greater than ventilation and the V/Q ratio is approximately 0.8. Therefore, the alveoli receiving oxygen are well perfused by blood, allowing optimal conditions for gas exchange.
Elasticity
Elasticity of the respiratory system refers to the degree to which the lungs resist inflation or stretching. The alveoli and other lung tissue normally resist stretching and recoil after the force causing the stretch or expansion is removed. This situation is partially caused by the surface tension of each
P.472
alveolus and partially by the presence of elastic fibers throughout the lungs, which tend to recoil after stretch. Conditions such as emphysema reduce the elastic recoil of the lungs, resulting in chronic overinflation.
The reciprocal of elasticity of the lungs is termed lung compliance. Compliance refers to the ease of inflation or stretching of the lungs. Lung compliance is reduced by fibrosis, infection, or adult respiratory distress syndrome (ARDS).
Pleural Pressure
The lungs are surrounded by a thin membrane called the pleura. The outer layer of the pleural membrane is attached to the wall of the thoracic cavity. The inner layer of the pleura is attached to the lungs. With expansion of the thoracic cavity during inspiration, the outer layer is pulled out; this force is transmitted to the inner layer, which expands the lungs. In between the inner and outer layers of the pleura is the pleural space. This space is filled with a few milliliters of fluid that surround and lubricate the lungs. The pleural fluid is at negative pressure and opposes the elastic recoil (collapse) of the lungs. This helps keep the lungs expanded.
Surface Tension
Surface tension refers to the tendency of water molecules to pull toward each other and to collapse a sphere. Because each alveolus is lined with a thin water layer, the surface tension within each alveolus could be high, making it extremely difficult to expand an alveolus. With each breath, a certain pressure must be exerted to overcome the surface tension of the water layer. The amount of pressure needed to expand the alveolus is described by Laplace's law in Equation 14-3:
![]()
where P is the pressure needed to expand the alveolus, T is the surface tension of the water molecules, and r is the radius of the alveolus. As shown in this equation, the smaller the alveolus, the greater the pressure required to expand it. An inability to overcome the surface tension of an alveolus could lead to alveolar collapse. Normally, however, the surface tension of an alveolus is kept low by the presence of surfactant.
Surfactant
Certain cells inside the alveolus, called type II alveolar cells, produce an important substance called surfactant that helps reduce the surface tension of the alveolus, making it easier to inflate. Surfactant is a phospholipid that acts like a detergent to intersperse between water molecules in the alveolus, thereby weakening the bonds between them. This reduces surface tension and the tendency of the sphere to collapse.
P.473
When surfactant is present, a small alveolus actually requires less pressure to inflate than a large one because the surfactant is packed tightly together, greatly reducing the surface tension of the alveolus. This serves to compensate for the effect of small radius in Laplace's law.
Tests of Pulmonary Function: Lung Volumes
Spirometry is the measurement of the volume of air moving into and out of the lungs and is measured as an individual inhales and exhales into a closed chamber. It is used to determine lung volumes, including tidal, inspiratory reserve, expiratory reserve, and residual volumes, and, calculated from these, vital capacity (Fig. 14-4). The average values presented in the figure for each of these volumes are for an adult male. Values for adult females are approximately 20 to 25% less.
Tidal Volume
The amount of air entering or leaving the lungs during a single breath is the tidal volume. The amount of air inspired at rest (inspiratory volume) usually equals the amount expired (expiratory volume). Tidal volume averages approximately 500 mL at rest.
 |
Figure 14-4. Approximate lung volumes per breath for a 70-kg male. Average lung volumes are proportional to body mass index. |
P.474
Inspiratory Reserve Volume
The amount of air above the normal inspiration that can be maximally inspired with each breath is the inspiratory reserve volume. It averages approximately 3,000 mL.
Expiratory Reserve Volume
The maximum amount of air that can be exhaled beyond normal exhalation is the expiratory reserve volume. This value averages approximately 1,000 mL.
Residual Volume
The air remaining in the lungs after maximum exhalation is the residual volume. The normal value is approximately 1,000 mL.
Vital Capacity
The maximum amount of air that an individual can inspire and expire during a single breath is the vital capacity. It is the sum of the normal tidal volume and the inspiratory reserve volume and the expiratory reserve volume. It is measured by having an individual take a maximum breath and then exhale as much as possible into the measurement chamber. In restrictive pulmonary disorders (e.g., resulting from neuromuscular disease, fibrosis, or loss of surfactant-producing cells), vital capacity is reduced.
A common test of pulmonary function is to plot the volume of air an individual can expire in the first second of expiration, called the forced expiratory volume in one second (FEV1). A healthy individual can expire approximately 80% of vital capacity as fast as possible in the first second (FEV1/vital capacity). In obstructive pulmonary diseases such as asthma and emphysema, expiration is particularly affected, and the amount of air an individual can forcefully expire in the first second is reduced. In patients who have restrictive airway disease, expiration is usually normal. Therefore, whereas overall vital capacity is reduced in those who have restrictive airway disease, FEV1 is normal.
Anatomic Dead Space
The amount of air in each breath that is measured as part of the tidal volume but that does not actually participate in gas exchange is the anatomic dead space. This air fills the conducting passages of the nose, mouth, pharynx, larynx, trachea, bronchi, and the bronchioles. With rapid, shallow respirations, a greater percentage of each breath is wasted simply moving air in and out of the anatomic dead space compared with that seen with slow, deeper breathing.
P.475
Pathophysiologic Concepts
Atelectasis
The collapse of either a lung or an alveolus is called atelectasis. Collapsed alveoli are airless, and therefore do not participate in gas exchange. This results in a reduction in the surface area available for diffusion, and respiration is decreased. Newborns may be born with alveoli collapsed at birth. This condition is called primary atelectasis. The collapse of previously expanded alveoli is called secondary atelectasis.
![]() ediatric Consideration
ediatric Consideration
As described later, primary atelectasis of the alveoli results in poor oxygenation of the newborn and is associated with significant morbidity and mortality. The cause of alveolar collapse is usually inadequate production of surfactant, resulting in high surface tension in the alveoli. The infant must work hard with each breath to overcome the surface tension and expand the alveoli. This can lead to exhaustion and an ever-worsening exchange of gases.
The two main types of atelectasis are compression atelectasis and absorption atelectasis.
Compression Atelectasis
Compression atelectasis occurs when a source outside the alveolus exerts enough pressure on the alveolus to collapse it. This occurs if the chest wall is punctured or opened because atmospheric pressure is greater than the pressure holding the lungs expanded (pleural pressure), and with exposure to atmospheric pressure the lungs will collapse. Compression atelectasis can also occur if there is pressure exerted on the lungs or alveoli from a growing tumor, abdominal distention, or edema and swelling of the interstitial space around an alveolus.
Absorption Atelectasis
Absence of air in the alveolus results in absorption atelectasis. If flow of air into an alveolus is blocked, the air currently inside eventually diffuses out and the alveolus collapses. Blockage usually occurs after mucus buildup and obstruction of airflow through a bronchus supplying a given group of alveoli. Any situation that results in mucus accumulation, such as cystic fibrosis, pneumonia, or chronic bronchitis, increases the risk of absorption atelectasis. Surgery is also a risk factor for absorption atelectasis because of the mucus-producing effects of anesthesia as well as a resultant hesitancy to cough up accumulated mucus after surgery. This is especially true if the
P.476
surgery was in the abdominal or thoracic area, where pain associated with coughing is intense. Prolonged bed rest after surgery increases the risk of developing absorption atelectasis, because lying down causes a pooling of mucus secretions in dependent areas of the lung, decreasing ventilation to those areas. Mucus accumulation increases the risk of pneumonia because mucus can act as a breeding ground for growth of microorganisms.
Absorption atelectasis also can be caused by anything that reduces the production or concentration of surfactant. Without surfactant, surface tension in the alveolus is high, increasing the likelihood of alveolar collapse. Premature birth is associated with a reduction in surfactant and a high incidence of absorption atelectasis. Near drowning may dilute out surfactant and thus may be associated with absorption atelectasis as well.
Damage to the type II alveolar cells that produce surfactant also can lead to absorption atelectasis. These cells are destroyed by the breakdown of the alveolar wall that occurs during some forms of respiratory disease, as well as by high oxygen therapy for a period longer than 24 hours. With loss of these cells, surfactant production is reduced.
Hypoxemia
The condition of reduced oxygen concentration in arterial blood is called hypoxemia. There are many causes of hypoxemia. Hypoxemia can occur if there is decreased oxygen in the air (hypoxia) or if hypoventilation occurs because of decreased lung compliance or atelectasis. Hypoxemia related to hypoperfusion (decreased blood flow past the alveoli) can occur from pulmonary hypertension, a pulmonary embolus, or a myocardial infarct. Hypoxemia may also occur if there is a problem with diffusion of oxygen across the alveolus into the capillary. This may happen with destruction of the alveolar-capillary interface or with edema of the alveolar-capillary interstitial space.
Because oxygen is carried in the red blood cell bound to hemoglobin, any decrease in hemoglobin concentration or carrying capacity can result in hypoxemia. Hemoglobin concentration is reduced in certain types of anemia. Binding sites for oxygen on the hemoglobin molecule may be occupied by other gases (e.g., carbon monoxide) that would also decrease the carrying capacity of hemoglobin for oxygen.
Cyanosis
Bluish discoloration of the blood that occurs if large amounts of hemoglobin in the blood are not completely bound with oxygen molecules is called cyanosis. There are four sites on hemoglobin where oxygen may bind. Hemoglobin fully bound with oxygen at all four sites is called saturated. Desaturated or deoxygenated hemoglobin is not fully bound with oxygen.
If the hemoglobin concentration in the blood is normal, but the availability of oxygen to bind to hemoglobin is reduced, the hemoglobin molecules
P.477
will be deoxygenated. Normally there are approximately 15 g of hemoglobin per 100 mL of blood. In arterial blood, more than 98%, or 14.7 g per l00 mL of blood, will be saturated with oxygen. Venous blood normally has an oxygen saturation of 75%; this comes to 11.3 g of hemoglobin per 100 mL of blood saturated with oxygen and about 4 g per 100 mL unsaturated. If arterial hemoglobin oxygen saturation falls below 70%, resulting in 5 g or more of unsaturated hemoglobin per 100 mL of blood, cyanosis will be apparent.
In hypoxemia caused by low hemoglobin concentration, such as with microcytic hypochromic anemia, cyanosis will not develop because there will not be greater than 5 g of deoxygenated hemoglobin per 100 mL of blood. Cyanosis will not occur with carbon monoxide poisoning because the hemoglobin binding sites will still be saturated, although with the carbon monoxide molecule rather than oxygen. In both of these situations, hypoxemia would be present in the absence of cyanosis.
Alterations in Ventilation:Perfusion Ratio
A decrease in ventilation may occur when delivery of air to some alveoli is obstructed, for example, with mucus or by foreign-body aspiration. With decreased ventilation (V), the ventilation:perfusion ratio is decreased, because the blood flow (Q) will pass by underventilated alveoli. This mismatch in ventilation and perfusion is not beneficial for gas exchange, and is an example of a right-to-left shunt of blood. A right-to-left shunt, described in Chapter 13, is characterized by delivery of deoxygenated blood to the systemic circulation. A decrease in the ventilation:perfusion ratio, however, will not last long in the lungs because of the pulmonary arteriolar response when exposed to low oxygen.
Pulmonary arterioles vasoconstrict in response to low oxygen concentration in underventilated alveoli. This serves to decrease blood flow to those alveoli, returning the ventilation:perfusion ratio back toward 1.0. This response is called hypoxic vasoconstriction. Hypoxic vasoconstriction is effective only if the extent of underventilated alveoli is limited. In conditions such as chronic bronchitis, alveolar obstruction is so widespread that a normal ventilation:perfusion ratio cannot be maintained. Hypoxic vasoconstriction of the pulmonary arterioles can lead to pulmonary hypertension as described later.
Under some circumstances, it is possible for ventilation of an alveolus to be adequate but capillary perfusion to be compromised. The result is a decrease in Q and an increase in the ventilation:perfusion ratio. This situation could occur as a result of a pulmonary embolus. A myocardial infarct would also cause decreased perfusion of the alveoli.
Pulmonary Hypertension
Elevated blood pressure in the pulmonary vascular system is called pulmonary hypertension. It is a common condition in serious respiratory or cardiovascular disease.
P.478
Causes of Pulmonary Hypertension
The pulmonary circulation is usually a low-pressure, low-resistance circulation. Anything that causes (1) a prolonged increase in pulmonary blood flow, (2) an increase in pulmonary resistance to flow, or (3) an impediment in pulmonary vascular outflow can result in pulmonary hypertension.
Increased Pulmonary Blood Flow
If excessive blood volume is delivered to the lungs, increased pulmonary blood flow occurs. For example, with a left-to-right shunt, blood from the left side of the heart goes back to the lungs rather than to the systemic circulation, thus overloading the lungs.
Increased Pulmonary Resistance to Flow
Anything that obstructs the passage of blood into or through the lungs causes increased pulmonary resistance to flow. This includes pulmonary fibrosis (scarring) and the changes in the structure of the lungs that accompany chronic obstructive pulmonary disease (COPD). Long-term pulmonary hypoxic vasoconstriction is also a significant cause of increased pulmonary resistance and hypertension. Hypoxic vasoconstriction of the pulmonary arterioles occurs when the pulmonary circulation is exposed to low oxygen, causing the vascular smooth muscle of the pulmonary arterioles to constrict. This can be useful because it allows the ventilation:perfusion ratio to return toward 1.0. However, if the condition is chronic or extensive, hypertrophy of the arterioles and increased pulmonary resistance can result.
Impediment to Outflow
This condition occurs with left-heart failure, as blood backs up in the left side of the heart, opposing continued flow out of the lungs. Other causes of impediment to outflow are mitral or aortic stenosis or incompetence, which also interfere with blood leaving the heart.
Consequences of Pulmonary Hypertension
Pulmonary hypertension can make it more difficult for the right side of the heart to pump. A type of heart failure called cor pulmonale can result. Cor pulmonale is right-sided heart failure caused by chronic lung disease. Pulmonary hypertension can also result in pulmonary edema because the capillary hydrostatic force favoring filtration is increased. Edema of the pulmonary interstitial space leads to a decreased diffusion rate of oxygen from the alveolus to the capillary because of increased distance for diffusion.
Bronchiectasis
Bronchiectasis is abnormal dilation of a bronchus or bronchi. Bronchiectasis occurs from long-standing pulmonary obstruction of the lower airways by tumors, chronic infections, mucus accumulation as seen in cystic fibrosis,
P.479
and exposure to toxins. The bronchi fill with mucus, resulting in atelectasis and development of abnormal connections between the bronchi. Ventilation of the alveoli is impaired.
Central Nervous System Depression
Central nervous system depression is a depressed respiratory drive resulting from alteration in function of the respiratory centers of the brain. Central nervous system depression can occur with hypoxemia or chronic elevation of carbon dioxide concentration, both of which occur when there is decreased pulmonary ventilation or perfusion. The respiratory center of the medulla and pons, which normally drives respiration, requires adequate oxygenation to function. Although the normal stimulus to breathe is the carbon dioxide concentration of the cerebral spinal fluid (as reflected in hydrogen ion concentration), high carbon dioxide levels can depress the respiratory center enough to cause a cessation of breathing.
Conditions of Disease or Injury
Upper Respiratory Tract Infections
Infections caused by any microorganism of the non gas-exchanging upper structures of the respiratory tract, including the nasal passages, the pharynx, and the larynx, are known as upper respiratory tract infections. Upper respiratory tract infections include the common cold, pharyngitis or sore throat, laryngitis, and uncomplicated influenza. Most upper respiratory tract infections are caused by viruses, although bacteria may also be involved either initially or secondary to a viral infection. All types of infections activate the immune and the inflammatory responses, leading to swelling and edema of the infected tissue. The inflammatory reaction leads to increased mucus production, contributing to the symptoms seen with upper respiratory tract infections, including congestion, excess sputum, and nasal discharge. Headache, low-grade fever, and malaise may also occur as a result of the inflammatory reaction.
Respiratory Defenses Against Infection
Although the upper respiratory tract is directly exposed to the environment, infections are uncommon and seldom progress to lower respiratory tract infections involving the lower airways and alveoli. Protective mechanisms abound throughout the respiratory tract to prevent infection. The cough reflex expels foreign bodies and microorganisms, and removes accumulated mucus. The mucociliary blanket consists of cells, located from the level of the bronchi up, which make mucus, and cilia cells that line the mucus-producing cells. The mucus-producing cells trap foreign particles, and the cilia beat rhythmically to propel the mucus and any trapped particles up the respiratory tree to the nasopharynx, where they
P.480
can be expelled as sputum, blown out the nose, or swallowed. This complex is sometimes referred to as the mucociliary escalator system. The cilia are delicate structures that can be paralyzed or injured by a variety of noxious stimuli, including cigarette smoke, as described later.
If microorganisms evade these defense mechanisms and colonize the upper respiratory tract, a third important line of defense, the immune system, is in position to prevent their passage to the lower respiratory tract. This response is mediated by lymphocytes, but also involves other white blood cells such as macrophages and neutrophils brought to the area by the inflammatory process. If there is a breakdown of a defense mechanism of the respiratory system or if the microorganism is especially virulent, a lower respiratory tract infection can result.
Effects of Cigarette Smoking on Respiratory Defenses
Cigarette smoke is known to alter the effectiveness of some respiratory defense mechanisms. Products of cigarette smoke stimulate mucus production while paralyzing the cilia. This leads to the accumulation of thick mucus and any trapped particles or microorganisms in the airways, decreasing the movement of air and increasing the risk of microbial growth. A smoker's cough is an attempt to expel this thick mucus out of the respiratory tract. Lower respiratory tract infections are more common in smokers and in those exposed to secondhand smoke, especially infants and children.
![]() ediatric Consideration
ediatric Consideration
Infants and children exposed to cigarette smoke before or after birth experience increased rates of upper respiratory tract infections, lower respiratory tract infections such as pneumonia, and childhood asthma compared with infants and children of parents who do not smoke. Urinary output of nicotine metabolites is grossly elevated in children whose parents smoke compared with those whose parents do not. Several metabolites of nicotine are known carcinogens as well as pulmonary irritants.
Clinical Manifestations
Clinical indications of upper respiratory tract infections depend on the infection site as well as the microorganism responsible for the infection. All clinical manifestations result from the inflammatory processes and any direct damage the microorganism inflicts. Clinical manifestations include:
Cough
Sneezing and nasal congestion
Mucus production and drainage from the nose and down the throat
Headache
Low-grade fever
Malaise (physical discomfort)
P.481
Diagnostic Tools
A good history and physical will assist diagnosis.
Complications
Sinusitis and acute otitis media may develop.
Lower respiratory tract infections, including pneumonia and bronchitis, may follow an upper respiratory infection.
Treatment
Rest to reduce the body's metabolic demands.
Extra hydration helps liquefy the thick mucus, making it easier to move it out of the respiratory tract. This is important because mucus accumulation offers a breeding ground for secondary bacterial infection.
Decongestants, antihistamines, and cough suppressants may provide some symptom relief.
Some studies suggest zinc lozenges or increased vitamin C consumption may reduce the severity or likelihood of certain viral infections.
Antibiotics are required if the infection is bacterial either initially or secondary to a viral infection.
![]() ediatric Consideration
ediatric Consideration
In children, croup, a viral infection of the larynx or trachea, and epiglottitis, a bacterial infection of the epiglottis, may occur. Like adults, children develop significant inflammation and swelling of the respiratory tract with infections. In fact, children may demonstrate more drastic clinical manifestations with upper airway infections because the upper airways are much narrower to begin with, resulting in a significant increase in resistance to airflow with even slight swelling and airway blockage. Symptoms of croup include a barking cough, hoarse voice, and stridor. Treatment for children who have mild-to-moderate croup may include a vaporizer, mist tent, or oxygen therapy. Those who have moderate-to-severe croup are likely to benefit from intramuscular or nebulized glucocorticoids. The inflammation seen with epiglottitis may result in total obstruction to airflow, significant anxiety, and death. Children typically sit forward and may drool. For children who have epiglottitis, hospitalization and perhaps intubation or tracheotomy may be required. Children who have epiglottitis should be kept as calm as possible (and so should their parents) to maintain airway patency until emergency support can be given.
Lower Respiratory Tract Infections
Pneumonia
Pneumonia, an acute infection of the lung tissue by a microorganism, is a lower respiratory tract infection. Most pneumonias are bacterial in origin,
P.482
occurring as a primary condition or secondary to a previous viral infection. The most common cause of bacterial pneumonia is the gram-positive bacterium Streptococcus pneumoniae, which is responsible for pneumococcal pneumonia. The bacteria Staphylococcus aureus and group A beta-hemolytic streptococci are also frequent causes of pneumonia, as is Pseudomonas aeruginosa. Other pneumonias are caused directly by viruses, such as that seen occasionally with influenza. Young children especially are susceptible to viral pneumonia, usually from infection with respiratory syncytial virus (RSV), parainfluenza, adenovirus, or rhinovirus. Mycoplasmal pneumonia, a relatively common pneumonia, is caused by a microorganism that is, in some respects, between a virus and a bacterium. Individuals who have acquired immunodeficiency syndrome (AIDS) frequently develop an otherwise rare pneumonia called Pneumocystis carinii pneumonia. Individuals exposed to aerosols of previously standing water, for instance, from air-conditioning units or dirty humidifiers, may develop Legionella pneumonia. Finally, individuals who aspirate stomach contents after vomiting or who aspirate water in an experience of near-drowning may develop aspiration pneumonia. For these individuals, the aspirated material itself rather than a microorganism may cause pneumonia by stimulating an inflammatory reaction. Subsequent bacterial infection may also develop.
The risks of developing the pneumonias described above are greater for the young, the old, or for anyone immunocompromised or weakened by another disease or disability. Risk of death after pneumonia is also stratified based on age (over 50 or young, especially newborn) and the presence of coexisting illness such as congestive heart failure, neoplastic disease, or renal disease.
Much of the damage to the lung tissue after successful colonization of the lungs by a microorganism is the result of the usually vigorous immune and inflammatory reaction mounted by the host. In addition, toxins released by bacteria in bacterial pneumonia can directly damage cells of the lower respiratory system, including the surfactant-producing type II alveolar cells. Bacterial pneumonia results in the most striking immune and inflammatory response, the course of which has been well described for pneumococcal pneumonia.
Stages of Bacterial Pneumonia
For pneumococcal pneumonia, four stages of disease have been described. What occurs in these four stages is similar for the other types of pneumonia.
Stage 1, called hyperemia, refers to the initial inflammatory response occurring in the area of lung infection. It is characterized by increased blood flow and increased capillary permeability at the site of infection. It occurs as a result of inflammatory mediators released from mast cells after immune cell activation and tissue injury. These components include histamine and prostaglandin. Mast cell degranulation also activates the
P.483
complement pathway. Complement acts with histamine and prostaglandin to vasodilate the pulmonary vascular smooth muscle, leading to increased blood flow to the area and increased capillary permeability. This results in movement of plasma exudate into the interstitial space, causing swelling and edema between the capillary and the alveolus. Fluid buildup between the capillary and the alveolus increases the distance over which oxygen and carbon dioxide must diffuse, thereby decreasing the rate of gas diffusion. Because oxygen is less soluble than carbon dioxide, its movement into the blood is most affected, often leading to a decrease in hemoglobin oxygen saturation. During this first stage of pneumonia, infection spreads to neighboring tissue as a result of increased blood flow and breakdown of neighboring alveolar and capillary membranes as the inflammatory processes continue.
Stage 2 is called red hepatization. It occurs when the alveoli fill with red blood cells, exudate, and fibrin, produced by the host as part of the inflammatory reaction.
Stage 3, called gray hepatization, occurs as white blood cells colonize the infected part of the lung. Then, fibrin deposits accumulate throughout the area of injury and phagocytosis of cell debris occurs.
Stage 4, called the resolution stage, occurs when the inflammatory and immune responses wane; cell debris, fibrin, and bacteria are digested; and macrophages, the cleanup cells of the inflammatory reaction, dominate.
![]() ediatric Consideration
ediatric Consideration
In the newborn period, pneumonia is most often caused by infection with group B streptococcal disease transmitted in utero. This disease can have a devastating effect, with an infant developing severe illness within hours of delivery. Treatment requires hospitalization, oxygen therapy, and intravenous antibiotics. This terrible disease may be reduced by prenatal screening of expectant mothers and treatment of women shown to be infected.
![]() eriatric Consideration
eriatric Consideration
The most common cause of pneumonia in the elderly is pneumococcal pneumonia. The elderly are at the greatest risk of dying from pneumonia, usually related to preexisting disease, poor nutrition, and reduced immune responsiveness. Those in long-term care facilities are especially susceptible to outbreaks of pneumococcal pneumonia. The risk of acquiring pneumococcal pneumonia can be reduced or eliminated by immunization; it is recommended that those who are older than 65 years of age or who live in a nursing home be vaccinated. Reports indicate, however, that less than 30% of those over 65 have been vaccinated against pneumococcal pneumonia.
P.484
Clinical Manifestations
Symptoms are similar for all types of pneumonia but are usually most pronounced for those of bacterial origin.
Significantly increased respiratory rate. Normal and abnormal respiratory rates vary with age, with young infants and children having more rapid normal rates of breathing than older children and adults.
Fever and chills from the inflammatory processes and a cough that is often productive, purulent, and present throughout the day; infants may grunt in an attempt to improve airflow.
Chest pain as a result of pleural irritation. The pain may be diffuse or referred to the abdominal area.
Sputum that is rust colored (for Streptococcus pneumoniae), pink (for Staphylococcus aureus), or greenish with a particular odor (for Pseudomonas aeruginosa).
Crackles, a poplike sound when the airways open suddenly, are indicative of lower airway infection.
Wheezing, the high-pitched sound heard when air rushes through a narrow orifice, signifies obstruction to airflow.
Fatigue, from both inflammatory reactions and hypoxia, if the infection is serious.
Pleural pain from inflammation and edema.
The subjective response of dyspnea is common. Dyspnea is a feeling of air hunger or a reported difficulty in breathing, which can be attributed in part to decreased gas exchange.
Hemoptysis, the coughing up of blood, may occur as a result of direct toxin injury to the capillaries or as a result of the inflammatory reaction and subsequent capillary breakdown.
![]() eriatric Consideration
eriatric Consideration
Geriatric patients may not demonstrate these typical signs of pneumonia. Instead, complaints of fatigue or disorientation, or both, may be made by the patient or the caregiver.
Diagnostic Tools
White blood cell count generally increases (unless the patient is immunodeficient). This is especially true for bacterial pneumonia.
Edema of the interstitial space is often apparent on chest radiograph (x-ray). Arterial blood gases may be abnormal.
Complications
Cyanosis with accompanying hypoxia may develop.
Ventilation may be reduced because of mucus accumulation, which may lead to absorption atelectasis.
Respiratory failure and death may occur in extreme cases and may be related to either exhaustion or sepsis (spread of the infection in the blood).
P.485
Treatment
The causative agent as determined by a pretreatment sputum sample determines the treatment for pneumonia. Such treatment includes:
Antibiotics, especially for a bacterial pneumonia. Other pneumonias may be treated with antibiotics to reduce the risk that a bacterial infection will develop secondary to the original infection.
Rest.
Hydration to help loosen secretions.
Deep-breathing techniques to increase ventilation of alveoli and to reduce the risk of atelectasis.
Other drugs specific for the type of microorganism identified in a sputum culture.
Tuberculosis
Tuberculosis (TB) is another example of a lower respiratory tract infection. It is caused by the microorganism Mycobacterium tuberculosis, which usually infects by inhalation of droplets, person to person, and colonizes the respiratory bronchioles or alveoli. It can also enter the body through the gastrointestinal tract, by means of ingestion of contaminated unpasteurized milk, or, occasionally, through a skin lesion.
After nearly 30 years of decline, starting in the mid-1980s, the number of cases of tuberculosis diagnosed in the United States began to climb. Reasons for this included increasing numbers of immigrants from areas where tuberculosis is endemic, increased poverty and homelessness in this country, and the advent of HIV/AIDS and a surge of immunocompromised persons. Although this increase has begun to taper off, the U.S. Center for Disease Control and Prevention noted that despite a low TB rate reported in 2004 (4.9 cases per 100,000 population), the rate of decline for 2003 and 2004 were the smallest since 1993.
If a significant amount of the mycobacterium bypasses the defense mechanisms of the respiratory system and successfully implants in the lower respiratory tract, the host mounts a vigorous immune and inflammatory response. Because of this vigorous response, which is primarily T-cell mediated, only approximately 5% of people exposed to the bacillus develop active tuberculosis. Only those individuals who develop an active tuberculin infection are contagious to others and only during the time of active infection.
Racial and Ethnic Implications
TB rates greater than the U.S. average were observed in certain racial/ethnic populations in 2004: Hispanics, blacks, and Asians living in the U.S. have 7.5, 8.3, and 20.0 times higher frequency of infection than
P.486
whites, respectively. In 2004, TB was reported more frequently among Hispanics (an increase of 1.2% from 2003 to 2004) than among any other racial/ethnic population in the U.S. Slightly more than half (53.7%) of U.S. cases were in foreign-born persons. To address the high rate of TB among foreign-born persons living in the U.S., custom and immigration efforts are directed to improve overseas screening of immigrants and refugees, strengthen the current notification system about the arrival of those suspected of having TB, ensure completion of treatment among TB patients who cross the border, test recent arrivals from high-incidence countries for latent infection, and treat to completion. March 24 of each year is being celebrated as World TB Day.
Risk Factors for Tuberculosis Exposure and Infection
Those most at risk of exposure to the bacillus are those living in close quarters with someone who has an active infection. This includes homeless individuals living in shelters where tuberculosis is present, as well as family members of infected individuals. Children may be especially susceptible. Immigrants to this country from developing nations frequently arrive with active or latent infection.
Also at risk of exposure to and development of tuberculosis are health care workers caring for the infected, and those individuals using the same health care clinics or hospital units as people who have active infection. Of those exposed to the bacillus, individuals who have inadequate immune systems, including the undernourished, the elderly and the young, individuals receiving immunosuppressant drugs, and those infected with the human immunodeficiency virus (HIV), are most likely to become infected. The virulence of the strain also affects transmission, with certain highly infective strains identified. TB control is hindered by the emergence of multi-drug resistance and the synergistic effect of HIV/AIDS. A significant number of TB cases in Africa have been linked to HIV infection.
Immune Response to Tuberculosis
Because the tuberculosis bacillus is difficult to destroy once colonization of the lower respiratory tract occurs, the goal of the immune response is to surround and seal off the bacilli, rather than to kill them. The cell-mediated response involves T cells as well as macrophages. Macrophages encircle the bacilli, after which T cells and fibrous tissue wall off the bacilli and macrophage complex. This complex of bacilli, macrophages, T cells, and scar tissue is called a tubercle. The tubercle eventually becomes calcified and is called Ghon's complex, which can be seen on chest radiograph. Before engulfment of the bacteria is complete, the material liquefies. At this time, viable microorganisms can gain access to the tracheobronchial system and spread airborne to infect others. Even when adequately walled off, the bacillus may survive within the tubercle. It is believed that because of this viability, approximately 5 to 10% of individuals who do not initially
P.487
develop tuberculosis may have a clinical demonstration of the disease at some other time in their lives, perhaps when they have become immunocompromised by age, other infection, or the need for anti-inflammatory medications. In fact, many if not most cases of active tuberculosis occur in individuals whose primary infection occurred decades earlier.
Among those infected, damage to the lung is caused by the bacilli as well as by a vigorous immune and inflammatory reaction. Interstitial edema and permanent scarring of the alveoli increase the distance for diffusion of oxygen and carbon dioxide, decreasing gas exchange. Also, the deposition of scar tissue and production of tubercles decrease surface area available for gas diffusion, decreasing diffusion capacity. If the disease is extensive, abnormalities in the ventilation:perfusion ratio occur that can lead to hypoxic vasoconstriction of pulmonary arterioles and pulmonary hypertension. Decreased lung compliance occurs with scar tissue.
Multi-Drug Resistant Tuberculosis
A recent worldwide and serious complication of tuberculosis is the development of tuberculin bacilli resistant to many drug combinations. Resistance develops when individuals do not complete the course of their therapy, and mutations of the bacillus make it non-responsive to the antibiotics that were used for a short time. The tuberculin bacillus mutates rapidly and often. Drug-resistant tuberculosis can also occur if an individual cannot mount an effective immune response, for instance, as seen in AIDS patients or in the malnourished. In these cases, antibiotic therapy is only partially effective. Health care workers or others who are exposed to these strains of bacillus also may develop drug-resistant tuberculosis, which can result in years of morbidity and frequently even in death. Those who have multi-drug-resistant tuberculosis will need to undergo more toxic and expensive treatments that are more likely to fail.
Clinical Manifestations
Clinical indications of tuberculosis may be absent with initial infection and may never be present if active infection does not occur. If active tuberculosis develops, an individual usually demonstrates the following:
Fevers, especially in the afternoon.
Malaise.
Night sweats.
Loss of appetite and weight loss.
A productive, purulent cough accompanied by chest pain is common with active infection.
Diagnostic Tools
A positive skin test for tuberculosis demonstrates cell-mediated immunity and is evidence only of previous exposure of the lower respiratory tract to the bacillus. It is not evidence that active tuberculosis ever developed.
Active tuberculosis is diagnosed by collection of a sputum sample followed by microscopic examination for the presence of acid-fast bacilli or culturing of the cells followed by identification and drug susceptibility testing of the isolates. Microscopy suffers from low sensitivity, especially in extrapulmonary tuberculosis and conditions of low bacillus count, which are common among HIV-infected individuals. Sputum culture of an actively infected individual will reveal the bacillus but takes a significantly longer time to complete.
Drug-resistance testing is traditionally performed using conventional methods in either solid or liquid media. More recently, molecular techniques based on PCR in conjunction with electrophoresis, sequencing, or hybridization are being used to detect gene mutations associated with the development of drug resistance. These molecular techniques have been used to complement smear results and clinical diagnosis.
Chest radiograph demonstrates current or previous tubercle formation.
P.488
Complications
Severe disease may lead to overwhelming sepsis, respiratory failure, and death.
Multi-drug-resistant TB may develop. Passage to others of the drug-resistant strain may occur.
Treatment
Treatment of individuals who have active tuberculosis is lengthy because the bacillus is resistant to most antibiotics and rapidly mutates when exposed to antibiotics to which it is sensitive. Currently, treatment of individuals who have an active infection includes a combination of four drugs and lasts at least 9 months or longer. If the person does not respond to those drugs, other drugs will be tried and different protocols will be followed.
Individuals who develop a positive tuberculosis skin test after having been previously negative, even if they show no symptoms of active disease, are usually put on a 6- to 9-month antibiotic regimen to support their immune response and to increase the likelihood that the bacillus will be eradicated completely.
If drug-resistant tuberculosis develops, more toxic drugs will be administered. The patient may be kept in the hospital or under some type of forced quarantine if compliance with the medical therapy is unlikely or impossible.
Pneumoconiosis
Pneumoconiosis, which is defined as a restrictive pulmonary disease, results from occupational inhalation of dust, usually from stone, coal
P.489
plants, or artificial fibers. Pneumoconiosis usually only develops after many years of dust inhalation.
Dust that reaches the lower respiratory tract stimulates an immune and inflammatory reaction resulting in the accumulation of dust-filled macrophages and the development of diffuse pulmonary fibrosis. Pulmonary fibrosis increases the distance across which diffusion of gases must occur, resulting in a decrease in gas exchange. Fibrosis also limits chest compliance and reduces ventilation. Additional influences such as cigarette smoking, which incapacitates the mucociliary escalator system, promote the likelihood of dust reaching the lower respiratory system and increasing its damage.
Examples of diseases from dust inhalation include black lung disease, seen in coal miners; silicosis, which occurs in stone workers, including masons and potters; and brown-lung disease, seen in those exposed to cotton dust. Asbestos exposure also leads to fibrosis and may cause lung cancer.
Clinical Manifestations
Dyspnea
A generally nonproductive cough unless chronic bronchitis develops.
A severe restriction of inspiratory volume.
Cyanosis may develop from decreased ventilation coupled with decreased diffusion rate.
Diagnostic Tools
A complete history and physical examination.
Chest x-ray.
Complications
Pulmonary hypertension leading to cor pulmonale may develop from severe fibrosis and decreased alveolar ventilation.
Pneumonias may repeatedly occur as restrictive disease contributes to atelectasis and poor gas exchange.
Treatment
A reduction of further exposure and avoidance of additive influences such as smoking.
Prevention and treatment of pneumonia with antibiotic therapy is also important.
Pneumothorax
Pneumothorax is the collapse of all or part of a lung that occurs when air or another gas enters the pleural space surrounding the lungs. There are different types of pneumothorax: open, spontaneous, and tension.
P.490
Open and Spontaneous Pneumothorax
An open pneumothorax occurs when the chest wall has been opened and air is allowed into the pleural space from the atmosphere. Atmospheric pressure is greater than pleural pressure and collapses the lungs. A spontaneous pneumothorax occurs when the chest wall is intact, but the lungs spontaneously develop a leak (primary) or are injured and begin to leak air into the pleural space (secondary). Air entering the pleural space from the lungs can cause the underlying alveoli to collapse. Causes of open pneumothorax include stab and gunshot wounds, rib fractures, and penetrating and non-penetrating trauma to the chest wall.
Tension Pneumothorax
It is also possible to have a tension pneumothorax in which there is one-way movement of air from the lung into the pleural space through a small hole in the lung structure. In this case, air leaves the lung and enters the pleural space during inspiration. However, air cannot move back into the lungs with expiration because the small hole collapses as the lungs deflate. It is also possible for air to enter the pleural cavity from damage to the tracheobronchial tree. Any tension pneumothorax is a life-threatening situation as it results in increased pressure in the pleural space. As pleural pressure increases, widespread compression atelectasis can occur. Displacement of the heart and great vessels in the thoracic cavity may also occur, resulting in severe alterations of cardiovascular function.
Clinical Manifestations
Acute onset of pain in the thoracic area resulting from pleural trauma.
Rapid, shallow breathing (tachypnea) and dyspnea are common.
If the pneumothorax is extensive, or if it is a tension pneumothorax and air is accumulating in the pleural space, the heart and large blood vessels may be displaced toward the other lung, which would give the chest the appearance of asymmetry. Tracheal deviation may also be apparent.
Diagnostic Tools
Blood gases and hemoglobin saturation will indicate hypoxia.
Radiographs can identify a collapsed lung.
Complications
A tension pneumothorax may collapse blood vessels, leading to reduced cardiac filling and causing a fall in blood pressure. The other lung may also be affected.
A pneumothorax may lead to hypoxia and severe dyspnea. Death may occur.
P.491
Treatment
A tension pneumothorax is a life-threatening condition because the buildup of air in the pleural space can eventually collapse the underlying lungs and blood vessels. It must be treated immediately with insertion of a chest tube or a large-bore needle into the pleural space with subsequent suction of the air out of the space.
A small spontaneous pneumothorax or a pneumothorax resulting secondarily to chest trauma is treated by insertion of a chest tube connected to a drainage tube that is kept in place until the pleural injury is healed. Any penetrating wound should be covered or closed.
Respiratory Failure
Inadequate exchange of gas that results in hypoxia, hypercapnia (increased arterial carbon dioxide concentration), and acidosis is called respiratory failure. It frequently develops when breathing becomes so difficult that exhaustion sets in and the individual no longer has enough energy to breathe. Respiratory failure becomes a vicious cycle; the more difficult it is to breath, the less the alveoli themselves are oxygenated, leading to death of the surfactant-producing cells, and an increased resistance to expansion. This means that the work of breathing is even harder, and the cycle continues and worsens. Respiratory failure can develop after a variety of respiratory diseases, including widespread pneumonia, sepsis, and infection with certain viruses such as Hantavirus.
Clinical Manifestations
Cyanosis.
Severe dyspnea.
Diagnostic Tools
Respiratory failure is defined clinically as a partial pressure of oxygen in arterial blood of less than 50 mmHg, and a partial pressure of carbon dioxide in arterial blood of greater than 50 mmHg, with a pH less than or equal to 7.25.
Complications
Poor oxygenation of other organs may lead to multi-organ failure.
Individuals in respiratory failure are at high risk of dying.
Treatment
Oxygen support, including artificial ventilation, is required. In general, the sooner a person is put on ventilatory support, the better the prognosis.
P.492
Adult Respiratory Distress Syndrome (ARDS)
ARDS is a disease characterized by widespread breakdown of the alveolar and/or pulmonary capillary membranes. ARDS occurs after a major pulmonary, cardiovascular, or system-wide insult.
Causes of Adult Respiratory Distress Syndrome
ARDS can occur as a result of direct injury to the capillaries of the lungs or to the alveoli. However, because the capillary and the alveolus are so intimately connected, extensive destruction of one typically leads to destruction of the other. This destruction occurs because of the release of lytic enzymes when cells die; it also occurs with activation of the inflammatory reaction subsequent to cell injury and death. Examples of conditions that affect the capillaries and/or the alveoli and can lead to ARDS are presented in the following sections.
Capillary Destruction
If breakdown is initially of the capillary membrane, movement of plasma and red blood cells into the interstitial space occurs. This increases the distance across which oxygen and carbon dioxide must diffuse, decreasing the rate of gas exchange. Fluid accumulating in the interstitial space moves into the alveoli, diluting surfactant and increasing surface tension. The exertion of pressure needed to inflate the alveoli is vastly increased. Increased surface tension coupled with edema and swelling of the interstitial space leads to widespread compression atelectasis, resulting in a loss of lung compliance, significantly decreased ventilation, and hypoxia. Causes of pulmonary capillary breakdown include septicemia, pancreatitis, venoms, and uremia. Pneumonia, smoke inhalation, trauma, and near drowning can also destroy the capillary membrane and initiate ARDS.
Alveolar Destruction
When the alveoli are the initial damage site, the surface area available for gas exchange is reduced, and, again, the rate of gas exchange is decreased. Causes of alveolar damage include pneumonia, aspiration, and smoke inhalation. Oxygen toxicity, which occurs after 24 to 36 hours of high-oxygen treatment, can also be a cause of alveolar membrane damage through the production of oxygen free radicals and by damaging the surfactant-producing cells.
Without oxygen, vascular and pulmonary tissues become hypoxic, leading to further cell injury and death. Once the alveoli and capillaries are damaged, inflammatory reactions, including macrophage and neutrophil infiltration and the release of various cytokines, are initiated that lead to swelling and edema of the interstitial space and damage to the neighboring capillaries and alveoli. Within 24 hours of ARDS onset, hyaline membranes form within the alveoli. These are white fibrin deposits that progressively accumulate and further decrease gas
P.493
exchange. Eventually, fibrosis obliterates the alveoli. Ventilation, respiration, and perfusion are all compromised. Mortality associated with ARDS is approximately 50%.
Clinical Manifestations
Significant dyspnea.
Decreased lung compliance.
Rapid shallow breathing initially, resulting in respiratory alkalosis as carbon dioxide is blown off. Later, as the person fatigues, breathing may become slow and infrequent.
Diagnostic Tools
Arterial blood-gas analysis demonstrates reduced arterial oxygen concentration despite oxygen therapy. Oxygen therapy is ineffective in ARDS, regardless of the amount of oxygen supplied, because diffusion of the gas is limited owing to fibrin accumulation, edema, and capillary and alveolar breakdown.
Complications
Respiratory failure may develop as the disease progresses and the individual has to work harder to overcome decreased compliance of the lungs. Eventually, exhaustion sets in and ventilation slows. This results in respiratory acidosis as carbon dioxide accumulates in the blood. Respiratory slowing and a fall in arterial pH are indications of impending respiratory failure and possible death.
Pneumonia may develop after ARDS because of fluid accumulation in the lungs and poor lung expansion.
Renal failure and gastrointestinal stress ulcer can occur as a result of hypoxia.
Disseminated intravascular coagulation may develop because of the large amount of tissue that can be destroyed during ARDS.
Treatment
Initially, treatment of ARDS is geared toward prevention, because ARDS is never a primary disease but always occurs after a major body catastrophe. When present, ARDS is treated as follows:
Diuretics to decrease fluid load and cardiostimulatory drugs to increase cardiac contractility and stroke volume are used. These interventions serve to reduce fluid buildup in the lungs and to reduce the likelihood of right-heart failure.
Oxygen therapy and mechanical ventilation are often initiated.
Anti-inflammatory drugs to reduce the damaging effects of inflammation are occasionally used, although their effectiveness is questionable.
P.494
Severe Acute Respiratory Syndrome (SARS)
SARS was first recognized as a global threat by the World Health Organization in mid-March 2003. The SARS coronavirus (SARS-CoV) is the etiological agent and is believed to be an animal virus that crossed the species barrier to humans. The natural reservoir of SARS-CoV has not been identified, but a number of wildlife species, including the Himalayan masked palm civet (Paguma larvata), the Chinese ferret badger (Melogale moschata), and the raccoon dog (Nyctereutes procyonoides), are suspect. The most probable sources of human infection with the SARS-CoV are either from these or other animal reservoirs or from exposure in laboratories where the virus is used or stored for diagnostic and research purposes.
Clinical Manifestations
During the first week, patients develop influenza-like prodromal symptoms such as fever, malaise, myalgia, headache, and lymphopenia.
Dry cough, dyspnea, and large-volume diarrhea may be present in the first to second weeks.
Transmission occurs during the second week.
Severe cases develop rapidly progressing respiratory distress and oxygen desaturation, with about 20% requiring intensive care.
![]() eriatric Consideration
eriatric Consideration
Afebrile illness or concurrent bacterial sepsis/pneumonia have been seen among SARS-infected elderly (those over 60 years of age). Those among the elderly with coexisting conditions should be excluded from caring for SARS patients and handling SARS-CoV.
![]() ediatric Consideration
ediatric Consideration
SARS-CoV is seen less frequently in children and has a milder presentation. The reasons for this are unknown.
Diagnostic Tools
Laboratory Tests
Diagnosis requires reverse transcription-polymerase chain reaction (RT-PCR) positive for the SARS-CoV, using a validated method from at least two different clinical specimens (e.g., nasopharyngeal and stool) or from the same clinical specimen collected on two or more occasions during the course of the illness (e.g., sequential nasopharyngeal aspirates). Two
P.495
different assays or repeat RT-PCR are required, using a new RNA extract from the original clinical sample on each occasion of testing.Seroconversion by ELISA. Negative antibody test on acute serum (collected prior to the development of antibody) followed by positive antibody test on convalescent phase serum tested in parallel.
Virus Isolation
Isolation in cell culture from any clinical specimen and identification of the SARS-CoV using a validated method such as RT-PCR.
Laboratory Findings
Proliferation of the cytokine interferon-gamma has been identified after SARS-CoV infection. It is hypothesized that this cytokine might be involved in immunopathological damage in SARS patients.
Lymphopenia, thrombocytopenia, and elevated lactate dehydrogenase and creatine kinase levels. Age, male sex, high lactate dehydrogenase level, high creatine kinase level, and high initial absolute neutrophil count are significant predictive factors for intensive care unit admission and death.
Hyponatremia, hypokalemia, hypomagnesemia, and hypocalcemia have been reported.
Complications
Respiratory failure and death may occur.
Those who survive may experience compromised pulmonary function for months after.
For pregnant women who develop SARS, there is an increase in fetal loss in early pregnancy and maternal mortality in later pregnancy.
Treatment
The anti-inflammatory agent dexamethasone and intravenous immunoglobulin are given. Dexamethasone inhibits cytokine production and delayed chemokine-recruited inflammation, while the immunoglobulin appears to modulate cytokine over-action and inhibit lymphocyte or macrophage activation.
Respiratory Distress Syndrome of the Newborn
Respiratory distress syndrome (RDS) of the newborn, also called hyaline membrane disease, is a condition of pulmonary hypoxia and injury resulting from widespread primary atelectasis. Primary atelectasis refers to the state of substantial alveolar collapse seen in a newborn. With alveolar collapse, ventilation is decreased. Hypoxia develops, leading to
P.496
pulmonary injury and a subsequent inflammatory reaction with the accumulation of white blood cells and the release of various cytokines. The inflammatory reaction leads to edema and swelling of the interstitial space, further reducing gas exchange between capillaries and any functioning alveoli. Inflammation also results in the production of hyaline membranes, which are white fibrin accumulations lining the alveoli. Fibrin deposits further decrease gas exchange and reduce lung compliance. With a decrease in lung compliance, the work of breathing is increased.
Decreased alveolar ventilation results in a decreased ventilation: perfusion ratio and pulmonary arteriolar vasoconstriction. Pulmonary vasoconstriction can lead to an increase in right-heart volume and pressure, resulting in a shunting of blood from the right atrium through the still-patent foramen ovale of the newborn, and directly to the left atrium. Likewise, high pulmonary resistance can result in deoxygenated blood bypassing the lungs and being delivered directly to the left side of the body via the ductus arteriosus. Both of these blood flow routes are considered right-to-left shunts, in that they bypass the lungs and so deliver poorly oxygenated blood to the systemic circulation. These examples of shunting worsen the condition of hypoxia, leading to significant cyanosis.
With each attempt to ventilate the collapsed alveoli, the infant must exert a large amount of energy. Such energy expenditure results in a correspondingly large oxygen demand, contributing to the evident cyanosis. With increased oxygen demand, the infant is caught in a positive-feedback cycle as shown in Figure 14-5.
At first the infant demonstrates rapid, shallow breathing in an attempt to meet this high oxygen demand, causing initial blood gases to indicate respiratory alkalosis as carbon dioxide is blown off. However, the infant soon tires from the extraordinarily difficult alveolar and lung expansion and is unable to keep up the respiratory effort. When this occurs, respiratory effort slows and blood gases reflect respiratory acidosis (buildup of carbon dioxide) and the onset of respiratory failure.
 |
Figure 14-5. When the work of breathing is increased, oxygen demand increases, which further increases the work of breathing. |
P.497
Risk Factors for Respiratory Distress Syndrome
The primary risk factor for the development of RDS of the newborn is prematurity. Between 5 and 10% of premature infants suffer from this syndrome. The more premature the infant, the more likely RDS will develop. The mechanism whereby prematurity is associated with RDS is threefold.
Most significantly, the type II alveolar cells that produce surfactant do not mature until between 28 and 32 weeks of gestation. Therefore, any infant born before surfactant is present in the alveoli encounters high alveolar surface tension with each breath. This contributes significantly to the primary atelectasis seen in RDS and results in decreased alveolar ventilation and hypoxia. Second, alveoli of premature infants are small and unfolded. By Laplace's law, this factor also contributes to the increased pressure that must be exerted to overcome their surface tension. Third, premature infants have weak, immature chest muscles, making it almost impossible for an infant without surfactant to successfully expand his alveoli, breath after breath for hours.
Another group of newborns at risk of developing RDS are infants born to insulin-dependent diabetic mothers. It appears that insulin provided by injection interferes with the development of type II alveolar cells.
Clinical Manifestations
The following clinical manifestations are usually present at birth:
Increased respiratory rate.
Duskiness of the skin caused by hypoxia.
Intercostal or chest retractions with each breath.
Nasal flaring with each breath.
Many infants survive RDS, and in these cases the symptoms lessen and resolve, usually within 3 days.
Diagnostic Tools
Diagnosis is usually made from the clinical appearance of the infant at birth coupled with the pregnancy history.
Arterial blood gases may be drawn to assist in diagnosis and management.
Chest radiograph typically shows diffuse granular densities within hours of birth.
Complications
Some infants who survive RDS go on to develop bronchopulmonary dysplasia (BPD), which is a chronic respiratory disease characterized by alveolar scarring, inflammation of the alveoli and the capillaries, and pulmonary hypertension. Effects of BPD may continue for years.
Signs of dyspnea and hypoxia may continue and proceed to infant exhaustion, respiratory failure, and death, usually within 3 days.
P.498
Treatment
Prevention is the first treatment of RDS. This includes behavioral and pharmacologic attempts to delay or stop labor and accurate dating of pregnancy to minimize delivery of premature infants by cesarean section.
Delay of parturition (delivery of an infant) for even 24 to 48 hours has been shown to reduce the incidence and severity of RDS. This is because the stress of labor increases maternal and fetal cortisol release from the adrenal cortex. Naturally occurring increases in cortisol have been shown to stimulate the type II alveolar cells to produce surfactant.
Maternal injections of corticosteroids at least 24 hours before a premature infant is delivered can significantly reduce the incidence of RDS. Concerns exist, however, about the long-term effects of exposing newborns to high levels of steroids.
If an infant is born with RDS, treatment is supportive and consists of oxygen therapy, maintenance of a quiet, warm environment to decrease oxygen requirements, nutritional support, and repeated evaluation of blood gases and acid-base status.
A major treatment advance has been the development of artificial surfactant. Surfactant can be delivered directly into the lower respiratory tract of infants demonstrating signs of RDS and has been shown to be successful in reducing clinical manifestations of the disease. This treatment, combined with maternal corticosteroid injections, offers the best hope for reducing the morbidity and mortality caused by RDS.
Diabetic women are closely monitored throughout pregnancy.
Mechanical ventilation may be employed to treat RDS. However, it is associated with increased risk of developing BPD.
Sudden Infant Death Syndrome
Characterized by the unexpected and the unexplained death of a previously healthy infant, sudden infant death syndrome (SIDS) typically occurs when the infant is between 1 week and 1 year of age. The highest incidence of SIDS is between 2 and 4 months of age, and occurs primarily during the night. At risk of developing SIDS are premature infants, infants born small for gestational age, and those born in multiple-birth pregnancies. Males are at slightly increased risk. In some cases, there is also a history of an upper respiratory tract infection during the week before death. Also at increased risk of developing SIDS are siblings of a child who has died of SIDS, and infants who have already experienced an episode of prolonged apnea, a near-miss occurrence of SIDS.
The incidence of SIDS has declined in recent years. This decline appears to be because of an understanding of behavioral risk factors as well as more
P.499
restrictive diagnostic criteria. SIDS, however, remains the major cause of death in otherwise healthy infants during the first year of life.
Causes of Sudden Infant Death Syndeome
The cause of SIDS is unknown. Some evidence suggests that an immature central nervous system fails to respond appropriately to increasing levels of carbon dioxide. Normally, increasing carbon dioxide is a stimulation to breathe, until high levels eventually depress ventilation. Healthy infants show occasional periods of apnea, during which carbon dioxide levels rise, stimulating the infant to breathe. Infants who experience SIDS or who experience near-miss episodes of SIDS appear not to respond to rising carbon dioxide with a stimulation of ventilation. Instead, they may show only a depression of ventilation in response to carbon dioxide. In these infants, apnea would occur, carbon dioxide levels would rise, and instead of stimulating breathing, the apneic episode would continue. Eventually, the high carbon dioxide levels completely suppress ventilation and the child dies.
Another phenomenon that may be involved in the development of SIDS in some infants is a prolongation of the cardiac QT interval (time before ventricular repolarization). It is suggested that this may lead to a fatal arrhythmia. Both respiratory and cardiac causes of SIDS are believed to have developmental as well as genetic influences.
Other Causes of Unexpected Infant Death
The major behavioral intervention that has resulted in a significant reduction in the occurrence of SIDS has to do with the positioning of infants during sleep periods. It appears that many infants who were assumed to have died of SIDS in the past actually suffocated while lying on mattresses or pillows that were too soft. When an infant lies face down, it has an increased risk of suffocation caused by its inability to move its head and face freely out of a confining position. This is especially true if the infant is weak because of prematurity or if the bed is too soft. In addition, if a baby's face is pressed into a small, soft depression on a mattress or pillow, it exhales into that depression and then rebreathes the same air. The carbon dioxide concentration in this pocket of rebreathed air increases, leading to central nervous system depression and cessation of breathing. Thus, it is recommended that infants should be propped on their back, not placed face down, and never left unattended on a water bed, soft mattress, pillow, or fur rug.
Another cause of infant death that is occasionally diagnosed as SIDS is child abuse. Injuries to the infant brainstem can occur with even moderate shaking, leading to respiratory depression and death. This condition is known as shaken-baby syndrome.
P.500
Clinical Manifestations
Death by suffocation or heart failure caused by respiratory arrest.
Symptoms of near-miss apneic episodes are cyanosis and, upon revival, gasping for air.
Treatment
Treatment of SIDS is aimed at prevention. Infants at high risk (as defined by having suffered a near-miss episode or having had a sibling who experienced SIDS) may wear monitors to signal apneic episodes.
If a cardiac tendency to dysrhythmia is documented, antiarrhythmic drugs or monitors may be used.
Prevention of other causes of infant death include careful placement of an infant to avoid face-down positions and vigilant prevention of child abuse.
Cystic Fibrosis
Cystic fibrosis is a hereditary disease characterized by alterations of exocrine gland function throughout the body. It results in production of large amounts of thick mucus and increased concentration of sodium and chloride in the sweat. Cystic fibrosis is a relatively common genetic disease, affecting mostly whites (1 in 3200 born in the U.S.). It is much less common in other races, although it does happen. Cystic fibrosis occurs as a result of a mutation in the 230 kb gene, a gene located in the middle of chromosome 7. This gene normally produces a protein called the cystic fibrosis transmembrane regulator protein, which functions as a chloride channel in epithelial cell membranes, thus affecting the flow of chloride through virtually all cells of the body. Without this protein, secretions dry out and become thick and obstructive. Cystic fibrosis is typically inherited as an autosomal-recessive disease. Only individuals who carry two copies of the defective gene, one from each parent, will have the disease. Over 1,000 mutations in this gene have been described, although the most common mutation is one that results in defective processing of the protein. Carriers of one cystic fibrosis gene and one normal gene are heterozygotic for the trait and do not demonstrate the disease.
Effects of Cystic Fibrosis
When the cystic fibrosis transmembrane regulator protein is absent, copious amounts of mucus and heavily concentrated sweat are produced. The main body systems affected by the mucus accumulation are the pulmonary and the gastrointestinal systems. Other organs also are victims of the excess mucus, including the liver and the reproductive organs. Sweat glands over-secrete sodium chloride, and sweat accumulates on the skin.
P.501
Pulmonary Effects of Cystic Fibrosis
In the lungs, the thick mucus increases the risk of repeated pneumonias and chronic bronchitis. It also blocks alveolar ventilation, leading to absorption atelectasis. In addition, an exaggerated and extended inflammatory response to pathogens, characterized by neutrophil-dominated airway inflammation, is chronically present. This leads to edema of the capillary and alveolar interface that accelerates progression of disease. Bronchial scarring and fibrosis progressively destroy the bronchial passages. Lung compliance is reduced and ventilation impaired.
Gastrointestinal Effects of Cystic Fibrosis
In the gastrointestinal tract, thick mucus accumulates, blocking digestion and absorption of nutrients. The pancreatic duct becomes clogged, thereby preventing the pancreatic digestive enzymes from reaching the small intestine and further compromising digestion and absorption of nutrients. Failure to thrive, a downward deviation in weight of an infant or a child from a previously recorded maximum, crossing one or two percentile lines and persisting for longer than 1 month, is a common complication of the disease. Poor nutritional status contributes to the frequency and severity of pulmonary infections. Frequently, the pancreas is destroyed, resulting in decreased insulin secretion and diabetes.
Clinical Manifestations
A protuberant abdomen may be apparent soon after birth, resulting from an inability to pass meconium in the first stool.
Salty taste when kissed, caused by salt buildup from sweat on the skin.
Repeated occurrences of respiratory tract infections throughout infancy and early childhood.
Chronic rhinitis (or nasal drainage), and chronic cough and sputum production.
Failure to thrive because nutrients are poorly absorbed.
Diagnostic Tools
In the U.S., 50% of patients with cystic fibrosis are diagnosed by 6 months of age, and 90% are diagnosed by 8 years. While early diagnosis may improve weight gain and early growth of infants via improved nutritional management, whether early diagnosis affects long-term outcome is unclear.
Testing for excess chloride in a sample of sweat; a positive result is indicative of disease. A concentration of sweat chloride greater than 60 mmol/L on repeated analysis is diagnostic.
Genetic testing can be used to confirm the diagnosis.
Abnormal pancreatic enzymes support the diagnosis.
Cystic fibrosis may be diagnosed prenatally by amniocentesis in couples who are known heterozygotes for the disease. Even eggs fertilized
P.502
in vitro can be identified as being heterozygotic or homozygotic for the cystic fibrosis gene at the eight-cell stage. The option to implant the embryo or abort the fetus is available.
Complications
Most individuals who have cystic fibrosis become progressively worse, and many die in their 20s or 30s, usually of bacterial pneumonia. Pulmonary hypertension can also develop as a result of decreased ventilation-perfusion, leading to cor pulmonale.
Frequent respiratory infections are present throughout life.
Nearly 98% of men with cystic fibrosis are infertile. Aspermia is present secondary to atrophied or absent vas deferens or seminal vesicles. Spermatogenesis and sexual functioning are normal, however, and men have successfully become fathers with assisted technology. Female reproductive function is normal, although cervical mucus may be especially thick.
Treatment
Prevention of bacterial lung infection is a primary aim of treatment. The use of improved antibiotic treatment strategies both for prophylaxis and in response to acute infection is the main reason for the increased life span currently seen. Lung injury and scarring are reduced.
Drugs that block inflammation are being tested as a mechanism to reduce pulmonary damage, including drugs that reduce the production of pro-inflammatory cytokines or increase anti-inflammatory cytokine levels. Other drugs have been developed that target specific components of neutrophils or block enzymes that accumulate at sites of neutrophil degradation. These enzymes are largely responsible for the damage to associated airway tissues. Clinical trials are underway to test the efficacy of such drugs, including recombinant human DNase (dornase alpha). Early studies suggest cystic fibrosis patients have improved lung function and minimal pulmonary exacerbations following use of DNase. DNase appears to facilitate expectoration of mucus and more effective clearance of neutrophils and by-products from the lungs.
Daily chest physiotherapy is essential to assist the individual in clearing the respiratory passages of mucus accumulations. Techniques include chest percussion and postural drainage, and frequent rest periods are advised to decrease energy demands.
Increased nutrient intake is important to maintain a well-functioning immune system and to minimize growth deficiencies. Dietary education and digestive enzyme supplements can improve nutritional balance and growth.
Double lung or heart-lung transplantation is a treatment option for some patients with end-stage lung disease. Survival is poorer than for
P.503
other types of organ transplantation, however, with a 3-year survival of about 60%. Better results are seen in adults than in children.There is ongoing genetic research aimed at correcting cystic fibrosis at the gene level. Experimental techniques include introducing into the lungs of an individual with cystic fibrosis the gene for transmembrane regulator protein, via exposure of the person to a genetically engineered virus that has been designed to carry that gene in its own DNA. It is intended that the virus, typically an attenuated cold virus, when delivered into the lower respiratory tract will incorporate its DNA into the host's respiratory cell DNA. This is the normal pattern of viral infection in the lungs. However, in this case, the virus would also be incorporating the genetically altered DNA into the host's genome, replacing the host's missing gene. Preliminary results of these initial trials have been unsuccessful, although hope is that in the future their success will be a standard for cure.
Additional studies are underway to investigate the use of supplements, including the following:
Glutathione, a naturally occurring antioxidant that protects the lungs from germs and pollutants. Patients with cystic fibrosis appear to have low levels of pulmonary glutathione. Glutathione administered by inhalation or as an oral supplement may reduce oxidative stress in patients, thereby reducing cell damage and lung inflammation.
Cucumin, the component of the spice turmeric that gives it a bright yellow color and strong taste. In animal studies, cucumin appeared to stimulate the function of the cystic fibrosis transmembrane conductance regulator. Phase 1 clinical trials are underway to test if similar effects will be manifested in humans.
Asthma
Asthma is a progressive respiratory disease characterized by inflammation of the respiratory tract and spasm of airway bronchiolar smooth muscle. This results in excess mucus production and accumulation, obstruction to airflow, and a decrease in ventilation of the alveoli. See page C10 for illustrations.
Asthma occurs only in certain individuals who aggressively respond to various airway irritants. Risk factors for this type of hyper-responsiveness include a family history of asthma or allergy, suggesting a genetic tendency. Repeated or intense exposure to some irritating stimuli, perhaps at a key developmental time, may also increase risk of this disease. Although most cases of asthma are diagnosed in childhood, adults may develop asthma without a previous history of the disease. Stimulation of adult-onset asthma often appears related to a worsening of previous allergies. Repeated upper respiratory infections may also trigger adult-onset asthma, as can occupational exposure to dusts and irritants.
![]() ediatric Consideration
ediatric Consideration
Being exposed to cigarette smoke in utero or in early childhood is considered a risk factor for childhood asthma. Infection in infancy with RSV may also be a risk factor for childhood asthma. Children may outgrow asthma, although a tendency toward allergies often remains.
P.504
Inflammatory Reaction in Asthma
The pathophysiology of asthma involves a hyper-responsiveness of the airways after exposure to one or more irritating stimuli. Known stimuli for inducing an asthmatic reaction include viral infections; an allergic response to dust, pollen, mites, or pet dander; exercise; cold exposure; and gastrointestinal reflux. With airway irritability and hyper-responsiveness, both an inflammatory reaction and bronchoconstriction result. Although bronchoconstriction and a feeling that the air passages are closing may be the first symptom of an asthmatic attack, it is the delayed inflammatory reaction that makes asthma the serious disease it is.
The primary mediators of inflammation in an asthmatic reaction are the eosinophils, a type of white blood cell. Eosinophils concentrate in the area and release chemicals that stimulate mast-cell degranulation. They also draw other white blood cells to the area, including basophils and neutrophils; stimulate the production of mucus; and increase swelling and edema of the tissues. The inflammatory response begins with the initial stimulus, but it may take as long as 12 hours to become apparent.
More acutely felt is the effect of the chemical histamine on the bronchiolar smooth muscle. Histamine is released with IgE-mediated mast-cell degranulation and quickly causes bronchiolar smooth muscle constriction and spasm. Histamine also stimulates mucus production and increased capillary permeability, further contributing to the more delayed congestion and swelling of the interstitial spaces.
Individuals who develop asthma may have either an overabundance of eosinophils or perhaps an over-responsiveness of the mast cells to stimuli initiating degranulation. IgE antibody, responsible for allergic attacks, may overreact in response to foreign antigens, turning on the inflammatory cascade. Regardless of the source of the hypersensitivity, the final result is bronchospasm, mucus production and accumulation, edema, and obstruction to airflow. Viral infection, allergy, and reflux appear to trigger a hypersensitivity response by means of irritating the airways. Exercise might also act as an irritant because large volumes of air are moved rapidly in and out of the lungs. This air has not been adequately humidified, warmed, or cleared of particulates and can trigger an attack.
Psychological Stimuli for Asthma
Psychological stimuli may worsen an asthmatic attack. Because parasympathetic stimulation constricts bronchiolar smooth muscle, anything that
P.505
increases parasympathetic activity could worsen the symptoms of asthma. The parasympathetic system is activated with the emotions of anxiety and sometimes fear. An individual having an attack may have worsening of symptoms as his anxiety peaks. In contrast, sympathetic innervation of bronchiolar smooth muscle leads to dilation of the bronchi. Typically, sympathetic stimulation is associated with conditions of fright and flight, during which increased ventilation is an important component of escape. Many of the therapeutic interventions traditionally used to treat asthma have relied on blocking parasympathetic or stimulating sympathetic responses.
Clinical Manifestations
Significant dyspnea.
Coughing, especially at night.
Rapid, shallow breathing.
Audible wheezing can be heard on auscultation of the lungs. Typically wheezing is heard only on expiration, unless the patient's condition is severe.
An increase in the work of breathing, exemplified by chest retractions and, with a worsening of condition, nasal flaring.
Anxiety, related to the inability to get enough air.
Air trapping because of the obstruction to airflow, especially seen during expiration in patients who have asthma. This is demonstrated as prolonged expiration time.
Between asthmatic attacks, a person is usually asymptomatic. However, changes in lung function tests may be apparent even between attacks in patients who have persistent asthma.
Diagnostic Tools
Asthma is diagnosed by spirometry, a technique that measures and identifies reductions in vital capacity and reduced peak expiratory flow rates. During an asthmatic attack, maximum expiratory volume and maximum rate of expiration are reduced.
To evaluate asthma symptoms at home, peak flowmeters are available. With a peak flowmeter, maximum forced expiratory flow rate (FEV), also called peak flow, is measured during an attack and during times between asthmatic episodes. (Note: since personal flowmeters do not actually measure exhalation over just 1 second, FEV will give slightly different values than the more exact FEV1 measurements.) By comparing his or her personal best FEV with that produced during an exacerbation, an individual or a family member can recognize mild versus moderate or severe worsening of symptoms. Appropriate therapeutic or emergency interventions can then be initiated.
Individuals who have asthma typically show a significant diurnal pattern, with peak flowmeter readings significantly poorer in the early hours after midnight than in late afternoon. This may be related to
P.506
exacerbation of symptoms by exposure to the cool air of night, or related to diurnal changes in hormones such as cortisol, known to affect inflammatory reactions.The saturation of hemoglobin with oxygen (oxygen saturation) may be measured to determine how well the blood is being oxygenated in a person showing asthmatic symptoms. This technique involves placing a sensor over the finger and obtaining the information concerning the color of the blood flowing beneath it. Unsaturated hemoglobin is darker in color than saturated. This tool is easily used in the clinical setting and provides a rapid indication of a patient's ability to move air.
Blood-gas analysis may demonstrate a decrease in arterial oxygen concentration and at first respiratory alkalosis as carbon dioxide is blown off in rapid breathing. If the condition persists and worsens, respiratory acidosis may develop as a result of status asthmaticus, as described in the following section.
Complications
Status asthmaticus, a life-threatening condition of prolonged bronchiolar spasm that cannot be reversed with medication, may develop in some individuals. In this case, the work of breathing is greatly increased. When the work of breathing increases, oxygen demand also increases. Because individuals in an asthmatic attack are not meeting a normal oxygen demand, they certainly cannot meet the high-oxygen demands needed to inspire and expire against prolonged bronchiolar spasm, bronchiolar swelling, and thick mucus. This situation can lead to a pneumothorax from the enormous pressures exerted to ventilate. As the individual becomes exhausted with effort, respiratory acidosis, respiratory failure, and death can occur.
Treatment
The first step in treatment involves evaluating a patient's stage of asthma. Staging is currently separated into four levels, depending on the frequency of symptoms and the frequency with which medications are needed to provide relief. Stages of asthma include (1) mild intermittent, (2) mild persistent, (3) moderate, and (4) severe. Treatment is based on staging.
For all stages of asthma, prevention of exposure to known allergens is vital. This involves allergy-proofing the home including removal of pets as necessary avoidance of cigarette and wood-burning smoke, and the use of air conditioners to minimize the opening of windows, especially during high-pollen seasons.
Monitoring peak flow rate frequently, especially during times of increased asthma incidence, such as the cold season (winter) or the pollen season (spring) is key for early recognition of exacerbations, reduction of symptoms, and prevention of hospitalization. This is true
P.507
even for mild intermittent asthma. If a significant decrease in peak flow rate is observed, adding pharmacologic intervention immediately instead of waiting until an attack is full blown can block its progression.An important advance in prevention and treatment of an asthmatic attack has been the use of oral or inhaled corticosteroids early in the course of an attack or as preventive therapy. Corticosteroids act as potent anti-inflammatory agents. Similarly, inhaled drugs that stabilize mast cells are used to prevent an asthmatic attack. The effects of these inhaled medications appear to be localized to the respiratory system, making their use safe and effective as treatments for asthma. Because asthma is a progressive disease, it is important to maintain treatment even between episodes of asthmatic attacks. Any individual, regardless of stage of disease, may require anti-inflammatory drugs. For those who have persistent, moderate, and severe asthma, low doses of inhaled steroids may be used daily to assist in stabilizing the patient.
Bronchodilators that act by stimulating the beta-adrenergic receptors of the airways (beta agonists) are a mainstay of asthma therapy. These drugs are inhaled (or given as a liquid in young children) at the onset of an attack and between attacks as needed. Bronchodilators do not prevent the delayed inflammatory response and so are not effective alone during a moderate or severe exacerbation of asthma; too-frequent or sole reliance on them has led to a significant number of fatalities. Newly available long-lasting beta-adrenergic agonists may reduce the need for frequent use of inhalers for some patients.
Combined products containing low-dose inhaled corticosteroids and long-acting beta-2 agonists appear to improve adherence and reduce exacerbations.
Beta-agonists may also be used before exercise in those who have exercise-induced asthma.
Although potent in the prevention and treatment of asthma and allergies, corticosteroids do not affect leukotriene synthesis or release. Leukotrienes are a product of arachidonic acid metabolism and contribute to the inflammation process. The production of leukotrienes may be prevented by using a 5-lipoxygenase inhibitor (zileuton) or by blocking specific leukotriene receptors using a leukotriene receptor antagonist (LTRA) such as montelukast or zafirlukast. LTRA medications manifest both bronchodilator and anti-inflammatory properties and may be used to complement corticosteroids.
Anticholinergic drugs may be given to decrease parasympathetic effects and to relax the bronchiolar smooth muscle. However, these drugs have a narrow therapeutic range of safety and so are used infrequently in general practice.
Behavioral intervention, aimed at calming the person to reduce parasympathetic stimulation of the airways, is important. When a person who is crying stops, this also allows for a slowing and a warming of the airflow, thereby reducing stimulation to the airways.
![]() ediatric Consideration
ediatric Consideration
The daily use of oral steroids may be necessary to control conditions of severe asthma, or they may be prescribed as a burst for 5 to 7 days to help return normal function more rapidly during an exacerbation in patients who have mild or moderate asthma. The chronic use of oral steroids for the long-term treatment of childhood asthma is associated with a reduction in growth potential and a thinning of the bones (osteoporosis).
P.508
Acute Bronchitis
Bronchitis is a common, obstructive respiratory disease consisting of inflammation of the bronchi. It is usually associated with a viral or a bacterial infection or the inhalation of irritants such as cigarette smoke or chemicals present in air pollution. It is characterized by excess mucus production.
Clinical Manifestations
Cough, usually productive with thick mucus and purulent sputum.
Dyspnea.
Fever.
Hoarseness.
Crackles (discontinuous fine or coarse lung sounds), especially on inspiration.
Chest pain occasionally may be present.
Diagnostic Tools
Chest radiograph.
Complications
Repeated episodes of acute bronchitis may result in the pathologic changes characteristic of chronic bronchitis.
Treatment
Antibiotics for secondary or primary bacterial infections.
Increased fluid intake and expectorants to loosen mucus.
Rest to reduce oxygen demands.
Chronic Bronchitis
Chronic bronchitis is defined as an obstructive pulmonary disorder characterized by excessive mucus production in the lower respiratory tract and a resulting chronic cough. It must last for at least 3 consecutive months of the year for 2 consecutive years.
P.509
Excess mucus results from pathologic changes (hypertrophy and hyperplasia) of the mucus-producing cells of the bronchi. In addition, the cilia lining the bronchi become paralyzed or dysfunctional, and undergo metaplasia. These changes to the mucus-producing cells and the cilia cells derail the mucociliary escalator system and cause the accumulation of large amounts of thick mucus that cannot be easily removed from the respiratory tract. The mucus acts as a breeding ground for infection and becomes highly purulent. Inflammation sets in, resulting in edema and swelling of the tissues and changes in the pulmonary architecture. Ventilation, especially exhalation, is obstructed. Hypercapnia (increased carbon dioxide) develops, as exhalation is prolonged and difficult to accomplish through the mucus and inflammation. The decrease in ventilation causes a decrease in ventilation:perfusion ratio, with resulting pulmonary hypoxic vasoconstriction and pulmonary hypertension. Although the alveoli are normal, hypoxic vasoconstriction and poor ventilation result in decreased oxygen exchange and hypoxia.
The main risk factor for development of chronic bronchitis is cigarette smoking. Components of cigarette smoke stimulate changes in both the mucus-producing cells of the bronchi and the cilia. They also induce chronic inflammation, which is the distinguishing characteristic of chronic bronchitis.
Clinical Manifestations
A productive, purulent cough, easily worsened by inspired irritants, cold weather, or an infection.
Copious amounts of sputum production.
Air hunger and dyspnea.
Diagnostic Tools
Pulmonary function tests demonstrate a reduction in FEV1 and vital capacity.
Blood gases show decreased arterial oxygen and increased arterial carbon dioxide.
Chest radiograph may document chronic bronchitis and fibrosis of the lung tissue.
Complications
Pulmonary hypertension, resulting from chronic pulmonary hypoxic vasoconstriction, can occur, leading to cor pulmonale.
Clubbing of the end segment of the fingers, an indication of chronic hypoxic stress, may develop.
Polycythemia (an increase in red blood cell concentration) occurs due to chronic hypoxia and the stimulation of erythropoietin secretion. This, coupled with cyanosis, gives the skin a bluish coloration.
Lung cancer.
P.510
Treatment
Education on decreasing further irritant exposure, especially cigarette smoke.
Prophylactic antibiotic therapy, especially in the winter months, to reduce incidence of lower respiratory tract infections. This is important because any infectious process further increases the inflammatory outcomes of mucus production and swelling.
Because many patients experience spasms of the respiratory tract with chronic bronchitis that are similar to spasms seen in chronic asthma, bronchodilators are frequently prescribed.
Anti-inflammatory drugs reduce mucus production and relieve blockage.
Expectorants and increased fluid intake loosen the mucus.
Oxygen therapy may be required.
Vaccination against pneumococcal pneumonia is highly advised.
Emphysema
Emphysema is a chronic obstructive disease characterized by loss of lung elasticity and a reduction in alveolar surface area due the destruction of the alveolar walls and the enlargement of air spaces distal to the terminal bronchioles. Damage can be either restricted to the central part of the lobe, which results in the bronchiolar wall integrity being affected most, or it can be throughout the entire lung, which results in damage both to the bronchi and to the alveoli.
Loss of lung elasticity can affect both the alveoli and the bronchi. Elasticity is lost as a result of destruction of the elastin and collagen fibers found throughout the lung from products produced by activated alveolar macrophages. The exact cause of emphysema is unclear, but in over 80% of cases, the disease occurs after years of smoking. It appears that components of cigarette smoke directly change the structure of the elastic molecules. There may also be an effect on the elastic fibers related to repeated infectious ailments and the state of chronic inflammation that accompanies infection. As a result of the loss of elasticity, air passages and alveoli collapse, reducing ventilation. Airways collapse primarily on expiration because normal expiration occurs as a result of passive recoil after inspiration. Therefore, if there is no passive recoil, air is trapped in the lung and the airways collapse.
The walls between the alveoli, called the alveolar septa, can also be destroyed. This reduces the surface area of alveoli available for gas exchange and decreases the rate of diffusion.
Although the primary risk factor for emphysema is smoking, repeated exposure to secondhand smoke might also result in emphysema. In addition, there is a familial form of emphysema, related to a deficiency in an anti-protease, alpha-1 antitrypsin. This is a much less common cause of emphysema that occurs in individuals not necessarily exposed to cigarette
P.511
smoke, although tobacco smoke exposure worsens the course of emphysema in those with this deficiency as well.
Clinical Manifestations
Air trapping, resulting from the loss of lung elasticity and leading to expansion of the chest (increased anterior-posterior diameter).
Diminished breath sounds on auscultation.
Use of accessory muscles of respiration.
Tachypnea (increased respiratory rate) caused by hypoxia and hypercapnia. Because of the effectiveness of increasing respiratory rate in this disease, most individuals who have emphysema do not show a significant alteration in arterial blood gases until late in the course of the disease, when the respiratory rate cannot mask hypoxia or hypercapnia. Eventually, all blood-gas values deteriorate, and frank hypoxia, hypercapnia, and acidosis are present.
Central nervous system depression, resulting from high carbon dioxide levels (carbon dioxide narcosis), can occur.
One key difference between emphysema and chronic bronchitis is the lack of sputum production in emphysema.
Diagnostic Tools
Abnormal pulmonary function tests, including decreased measured FEV1, decreased vital capacity, and increased residual volume (air left in the respiratory tract after each breath) caused by loss of lung elasticity.
As the disease progresses, blood-gas analysis will first demonstrate hypoxia. Late in the disease, carbon dioxide levels may also be elevated.
Complications
Pulmonary hypertension from chronic pulmonary hypoxic vasoconstriction, leading to cor pulmonale, may occur.
A reduction in quality of life is common in severely affected individuals.
Treatment
Relieving the symptoms and preventing a worsening of the condition is the objective in emphysema treatment. There is no cure. Treatments include:
Encouraging the individual to stop smoking.
Using breathing positions and patterns to reduce air trapping.
Teaching the individual relaxation techniques and means of energy conservation.
Oxygen therapy is needed for many patients who have emphysema, so that they can complete tasks of daily living. Oxygen therapy may slow the progression of the disease and lessen morbidity and mortality.
Well-designed exercise therapy can improve symptoms.
P.512
Chronic Obstructive Pulmonary Disease
Individuals who have long-standing emphysema also usually have chronic bronchitis and demonstrate indications of both diseases. This condition is called chronic obstructive pulmonary disease (COPD). Chronic asthma in association with either emphysema or chronic bronchitis may also result in COPD.
Clinical Manifestations
Symptoms of both emphysema and chronic bronchitis are usually present.
Dyspnea is constant.
Diagnostic Tools
History and physical examination.
Chest x-ray.
Complications
Pulmonary hypertension leading to cor pulmonale.
Pneumothorax.
Treatment
Long-acting beta-2 agonists (LABAs), rather than short-acting beta-2 agonists, have the potential to improve the mucociliary clearance and act as bronchodilators. A combination therapy consisting of LABA and an inhaled corticosteroid provides anti-inflammatory activity and improves mucociliary clearance.
In general, COPD treatment is as described for chronic bronchitis and emphysema, with the exception that oxygen therapy must be closely monitored. Individuals who have COPD have chronic hypercapnia that causes the central chemoreceptors, which normally respond to carbon dioxide, to adapt. What keeps these individuals breathing is the low oxygen concentration of the arterial blood that continues to stimulate the less-sensitive peripheral chemoreceptors. These peripheral chemoreceptors only fire if the arterial partial pressure of oxygen decreases to less than 50 mmHg. Therefore, if oxygen therapy were to result in a partial pressure of oxygen of greater than 50 mmHg, this remaining drive to breathe would be extinguished. Individuals who have COPD typically have low oxygen levels and cannot be treated with high-oxygen therapy. This severely affects quality of life.
Phosphodiesterase 4 (PDE4) inhibitors are a promising and potent drug class that controls the inflammatory process in patients with COPD by reducing the number of bronchial mucosal CD8+ T cells and CD68+ macrophages and neutrophils.
P.513
Lung Cancer
Lung cancer is defined as a cancer of the epithelial lining of the respiratory tract (bronchogenic carcinoma). It can occur anywhere in the lung. There are four general types of lung cancer: three non small cell carcinomas and one small cell carcinoma. The non small cell carcinomas are squamous cell carcinoma, adenocarcinoma, and large cell carcinoma.
Squamous cell carcinoma accounts for about 30% of lung cancers. This cancer is clearly associated with cigarette smoking and exposure to environmental toxins, such as asbestos and components of air pollution. Squamous cell tumors are usually located in the bronchi at the site where the bronchi enter the lungs, called the hila, and from there extend down into the bronchi. Because the bronchi are to some degree obstructed, absorption atelectasis and pneumonia, as well as decreased ventilatory capacity, can occur. This tumor grows relatively slowly and has the best prognosis for a 5-year survival if diagnosed prior to metastasis.
Adenocarcinoma is a type of lung cancer arising from the glands of the lung. It usually occurs in the periphery of lung tissue, including the terminal bronchioles and the alveoli. This type of cancer accounts for approximately 30% of lung cancers and is increasing among women. Adenocarcinomas are typically slow growing, but they metastasize early and have a poor 5-year survival rate.
Large-cell undifferentiated cancer is highly anaplastic and associated with rapid metastasis. These tumors account for approximately 10 to 15% of all lung cancers, often occurring peripherally and spreading centrally in the lung. They are highly correlated with cigarette smoking and can cause chest pain. This type of cancer has a poor prognosis for survival.
Small cell carcinoma accounts for approximately 25% of all lung cancers. This type of tumor is also referred to as oat-cell carcinoma and usually occurs in the central areas of the lung. Small-cell carcinoma is an anaplastic, or embryonic, type of tumor, and therefore shows a high incidence of metastasis. It is often a site of ectopic tumor production and may cause early symptoms based on endocrine disturbances. Pulmonary manifestations that occur with this tumor also result from obstruction to airflow. This type of tumor is the most strongly associated with cigarette smoking, and has the worst prognosis.
Risk Factors for Lung Cancer
Although the incidence of lung cancer in the United States has begun to fall for men, it is holding steady or continues to rise among women. The reason for this is the clear fact that the primary risk factor for lung cancer in approximately 90% of cases is cigarette smoking, a behavior that peaked and then began its fall among men at least a decade or more before a similar change in behavior developed in women. Lung cancer is often associated with chronic bronchitis, because of the overlap in risk factors as well as excess mucus possibly causing abnormal epithelial cell changes. Air
P.514
pollution and exposure to chemicals and dusts, including asbestos, also contribute to the disease.
Although there are more than 50 known carcinogens in tobacco smoke, it is still not exactly understood how cigarette smoking causes cancer. It is likely, however, that these carcinogens or other metabolites of cigarette smoke interfere with the functioning of key genes regulating epithelial cell growth and development. In particular, specific mutations in certain cancer suppressor genes have been shown to occur with prolonged exposure to tobacco smoke. Without properly functioning tumor suppressor genes, uncontrolled cell division can occur and cancer may develop.
Epithelial cells change progressively with the development of lung cancer, first showing subtle signs of metaplasia, then dysplasia, and finally neoplasia. These conditions preceding neoplasia are visible histologically in individuals who have chronic bronchitis and emphysema. The bronchial epithelial cells that appear to be most damaged by toxins are those at the bronchial bifurcations. It appears that mucus and toxins accumulate there, causing the most injury to these cells. The result is that the epithelial cells become thickened, the mucus-producing cells hypertrophy, and metaplasia and dysplasia occur. Alveolar cells may also be altered in structure and function.
Clinical Manifestations
A persistent cough.
Recurring lower respiratory tract infections.
Hemoptysis (the coughing up of blood).
Weight loss.
Fatigue.
Hoarseness.
Pain or dysfunction in a distant organ reflecting metastasis may be the first clue of lung cancer.
Other symptoms seen with each particular type of lung cancer may vary, as described previously.
Diagnostic Tools
Chest x-ray followed by biopsy of suspicious lesions may diagnose the disease.
Complications
Prognosis is poor. The 5-year survival rate for all types of lung cancer is only 13%. Some types of lung cancer have an even worse prognosis. For instance, oat-cell carcinoma has a less than 5% survival rate 2 years after diagnosis.
Treatment
Any combination of surgery, radiation, and chemotherapy.
P.515
Selected Bibliography
Currie, G.P., Devereux, G.S., Lee, D.K.C., & Ayres, J.G. (2005). Recent developments in asthma management. British Medical Journal 330, 585 589.
Currie, G.P., Srivastava, P., Dempsey, O.J., & Lee, D.K.C. (2005). Therapeutic modulation of allergic airways disease with leukotriene receptor antagonists. Quarterly Journal of Medicine 98, 171 182.
Egan, M.E., Pearson, M., Weiner, S.A., Rajendran, V., Rubin. D., et al. (2004). Cucumin, a major constituent of turmeric, corrects cystic fibrosis defects. Science 304, 600 602.
File, T.M. (2003). Community-acquired pneumonia. Lancet 362, 1991 2001.
Fine, M. J., Auble, T. E. Yealy, D. M., et al. (1997). A prediction rule to identify low-risk patients with community acquired pneumonia. New England Journal of Medicine 336, 243 250.
Guntheroth, W.G., & Spiers, P.S. (1992). Sleeping prone and the risk of sudden infant death syndrome. Journal of the American Medical Association 267, 2359 2362.
Grey, V., Mohammed, S.R., Smountas, A.A., Bahlool, R., & Lands, L.C. (2003). Improved glutathione status in young adult patients with cystic fibrosis supplemented with whey protein. Journal of Cystic Fibrosis 2, 195 198.
Guan, Y., Zheng, B.J., He, Y.Q, Liu, X.L., Zhuang, Z.X., Cheung, C.L. et al. (2003). Isolation and characterization of viruses related to the SARS coronavirus from animals in Southern China. Science 302, 276 278.
Guyton, A. C. & Hall, J. (2006). Textbook of medical physiology (11th ed.). Philadelphia: W.B. Saunders.
Hallman, M. (2004). Lung surfactant, respiratory failure, and genes. New England Journal of Medicine 350, 1278 1280.
Homer, C. J. (1997). Asthma disease management. New England Journal of Medicine 337, 1461 1463.
Horsburgh, C.R. (2004). Priorities for the treatment of latent tuberculosis infection in the United States. New England Journal of Medicine 350, 2060 2067.
Huang, K.-J., Su, I.-J., Theron, M., Wu, Y.-C., Lai, S.-K., Liu, C.-C., et al. (2005). An interferon-gamma-related cytokine storm in SARS patients. Journal of Medical Virology 75, 185 194.
Jeffery, P. (2005). Phosphodiesterase 4-selective inhibition: novel therapy for the inflammation of COPD. Pulmonary Pharmacology and Therapeutics 18, 9 17.
Klass, P. (2004). Croup the bark is worse than the bite. New England Journal of Medicine 351, 1283 1284.
Ksiazek, T.G., Erdman, D., Goldsmith, C.S., Zaki, S.R., Peret, T., Emery, S. et al. (2003). A novel coronavirus associated with severe acute respiratory syndrome. New England Journal of Medicine 348, 1953 1966.
Kuiken, T., Fouchier, R.A.M., Schutten, M., Rimmelzwaan, G.F., van Amerongen, V., van Riel, D., et al. (2003). Newly discovered coronavirus as the primary cause of severe acute respiratory syndrome. Lancet 362, 263 270.
McShane, H. (2005). Co-infection with HIV and TB: double trouble. International Journal of STD and AIDS 16, 95 101.
MMWR (2005). Trends in tuberculosis- United States, 2004. MMWR 54(10), 245 249.
Poole, P.J., Saini, R., Brodie, S.M., & Black, P.N. (2005). Comparison of the effects of nebulised and inhaled salbutamol on breathlessness in severe COPD. Respiratory Medicine 99, 372 376.
Porth, C. M. (2005). Pathophysiology: Concepts of altered health states (7th ed.). Philadelphia: Lippincott Williams & Wilkins.
Ratjen, F., Paul, K., van Koningsbruggen, S., Breitenstein, S., Rietschel, E., Nikolaizik, W., et al. (2005). DNA concentrations in BAL fluid of cystic fibrosis patients with early lung disease: influence of treatment with dornase alpha. Pediatric Pulmonology 39, 1 4.
Rogers, D.F. (2005). Mucociliary dysfunction in COPD: effect of current pharmacotherapeutic options. Pulmonary Pharmacology & Therapeutics 18, 1 8.
Schwartz, P. J., Stramba-Badiale, M., Segantini, A., et al. (1998). Prolongation of the QT interval and the sudden infant death syndrome. New England Journal of Medicine 338, 1709 1714.
Shamputa, I.C., Rigouts, L., & Portaels, F. (2004). Molecular genetic methods for diagnosis and antibiotic resistance detection of mycobacteria from clinical specimens. APMIS 112, 728 752.
Snider, D. E. & Castro, K. G. (1998). The global threat of drug-resistant tuberculosis. New England Journal of Medicine 338, 1689 1690.
P.516
Taylor, J, Wentworth, D.E., Bernard, K.A., Masters, P.S., & Trimarchi, C.V. (2005). SARS coronaviruses and highly pathogenic influenza viruses: safety and occupational health for laboratory workers [conference summary]. Emerging Infectious Diseases [serial on the Internet]. Available from http://www.cdc.gov/ncidod/EID/vol11no04/04-1304.htm
Umetsu, D.T. (2005). Revising the immunological theories of asthma and allergy. Lancet 365, 98 100.
WHO Guidelines for the Global Surveillance of Severe Acute Respiratory Syndrome. (2004). Updated Recommendations October 2004. Downloaded March 30, 2005 from http://www.who.int/csr/resources/publications/SARSNEWGUIDANCE/en/.
World Health Organization. (2005). Global tuberculosis control: surveillance, planning, financing. WHO Report 2005 (WHO/HTM/TB/2005.349). Geneva: World Health Organization.
Wouters, E.F.M. (2004). Management of severe COPD. Lancet 364, 883 895.
Yu, I.T.S., Li, Y.G., Wong, T.W., Tam, W., Chan, A.T., Lee, J. H.W., et al. (2004). Evidence of airborne transmission of the severe acute respiratory syndrome virus. New England Journal of Medicine 350, 1731 1739.
Resources
Asthma and Allergy Foundation of America, 1125 15th St. N.W., Washington, DC 2005. Phone: 800-7-ASTHMA or 800-727 8462.
EAN: 2147483647
Pages: 26