7. Acamprosate - preclinical data
Editors: Spanagel, Rainer; Mann, Karl F.
Title: Drugs for Relapse Prevention of Alcoholism, 1st Edition
Copyright 2005 Springer
> Table of Contents > Acamprosate: preclinical data
Acamprosate: preclinical data
Philippe De Witte1
Daniel Bachteler2
Rainer Spanagel2
1 UCL - Biologie du Comportement, Place Croix du Sud, 1, 1348 Louvain-la-Neuve, Belgium
2 Department of Psychopharmacology, Central Institute of Mental Health, University of Heidelberg (CIMH), J5, 68159 Mannheim, Germany
Introduction
Acamprosate (calcium-bis-acetyl-homotaurinate, Campral ) was introduced to the European market in the time between 1990 and 2000 and since then it has been successfully applied in clinical relapse prevention as an anti-craving/anti-relapse drug in detoxified alcoholics. Numerous preclinical studies on alcohol drinking and relapse behavior have been performed with acamprosate [1, 2]. Besides the demonstration of its anti-relapse and anti-craving properties it has been demonstrated that acamprosate neither produces discriminative stimulus properties [3] nor does it produce place conditioning [4]. This implies that acamprosate is lacking in any rewarding properties and cannot be used as a substitution drug (see Chapter by Bachteler and Spanagel). The primary site of action is the glutamatergic system [1, 2, 5] and it has been shown that N-methyl-D-aspartate (NMDA) receptor subunits are modulated by acamprosate [6]. Until the present, the detailed mode of action is still not clear; however, a functional interplay with other glutamate receptors has recently been described. Thus, Harris et al. [7, 8] described a novel site of acamprosate action at metabotropic glutamate receptors, in particular at mGluR5. Furthermore, a series of in vivo microdialysis studies on the interaction of ethanol and acamprosate with brain taurine levels suggest that acamprosate reduces the preference for alcohol in dependent animals through the release of taurine [9, 10, 11, 12 and 13]. The following paragraphs review animal studies on the behavioral effects of acamprosate and provide insights into the latest research on acamprosate, especially its relation to brain taurine and its action on the glutamate system.
Animal studies - general characterization
As a therapeutic drug, acamprosate combines important characteristics: it neither exerts its actions by imitating alcohol's reinforcing effects, nor does it alter ethanol-induced hypothermia, taste-aversion or motor impairment [14].
P.74
Rewarding effects of the compound itself could not be demonstrated in conditioned place preference experiments in rats [4, 15], nor could it substitute for or antagonize ethanol effects in drug discrimination experiments [3]. Furthermore, its effects are not psychotropic, sedative, anti-depressant or anxiolytic [16]. However, it is suggested that acamprosate reduces craving and relapse that are associated with a hyper-glutamatergic state in the brain and the phenomenon of conditioned withdrawal [17] and thus could be used as an anticraving/anti-relapse drug.
Attenuation of ethanol intake and relapse
The initial study on the effects of acamprosate was performed by Boismare et al. [18], reporting a significant decrease in voluntary ethanol intake in rats, daily treated with 0.26 and 0.52 mMol/kg Ca-acetylhomotaurine, respectively. This effect could be inhibited by simultaneous administration of the -aminobutyric acid (GABA) antagonist bicuculline, thus leading to the assumption of an involvement of the GABAergic system. Acamprosate injections had no consequences on total fluid intake, but selectively affected alcohol preference.
These first results were confirmed by other studies [19, 20]. Although different experimental protocols were used, all studies consistently reported a significant, dose-dependent reduction of ethanol consumption after chronic treatment with doses of 100 and 200 mg/kg or acute treatment with 200 and 400 mg/kg acamprosate, respectively. A dose of only 50 mg/kg produced no significant effect [21]. Water intake during chronic treatment remained unaffected. However, Heyser et al. [22] reported an increasing water consumption after an acute high dose of 400 mg/kg. The aforementioned effects were confirmed in more recent studies under operant and non-operant conditions [21, 23, 24, 25 and 26].
A generally accepted method for assessing relapse-like drinking behavior in rats is the alcohol-deprivation model. Ethanol consumption in long-term ethanol-experienced animals is measured after a period of imposed abstinence, leading to a strong transient increase in alcohol intake, termed the alcohol deprivation effect (ADE) [27, 28] (see also Chapter by Spanagel). Using this model, administration of acamprosate to long-term alcohol-drinking rats dose-dependently reduced the alcohol deprivation effect, and at the highest dose (200 mg/kg), alcohol intake even dropped significantly below baseline levels [29]. It should be emphasized that alcohol consumption after a deprivation effect is more effectively reduced than during baseline drinking [25]. Furthermore, chronic injections of acamprosate were more effective in reducing the alcohol deprivation effect than acute single administrations [22]. In conclusion, acamprosate reduces not only voluntary ethanol self-administration under free-choice and operant paradigms, but is also effective in reducing relapse behavior, as measured by the alcohol deprivation effect.
P.75
Only recently the anti-craving effect of acamprosate was demonstrated by Bachteler et al. [30] in the reinstatement model, which in contrast to the ADE model (relapse), measures the animals' motivation to get the drug and thus reflects craving for alcohol [31]. A stimulus is said to reinstate drug seeking, if the animal restarts to lever press for the drug after an extinction phase, but without the primary reinforcer ethanol being available. Administration of acamprosate (100, 200 mg/kg) significantly and selectively attenuated responding for ethanol at both doses after presentation with the ethanol-related stimulus. It should however be noted that in the aforementioned reinstatement paradigm, only the behavioral outcome of alcohol craving can be assessed. The subjective state, associated with an incentive motivation to administer the drug, remains unknown. In addition, it should be kept in mind that not all patients are responding to acamprosate treatment, leading to relapse despite medication. The potential role of acamprosate as a neuroprotective agent in humans could furthermore support its use, as already proposed in different in vitro models [32, 33 and 34], but also needs further investigation.
Involvement of the glutamatergic system in the action of acamprosate
The exact mechanism as to how acamprosate diminishes alcohol consumption and reduces the likelihood of relapse is still not clear. Different neurobiological pathways have been implicated in the etiology of alcohol dependence and one pathway seems to involve the glutamatergic system [1, 35, 36 and 37], where chronic alcohol intake leads to compensatory changes. It is suggested that acamprosate acts mainly on a hyper-glutamatergic state, yet having only little effect on a normal glutamatergic state [17, 33, 38, 39].
Depending on the brain region and the rat strain, low doses of ethanol can increase glutamate levels in the brain, whereas high intoxicating doses can decrease glutamate levels [40]. The mechanism behind the inhibitory effect of high intoxicating doses of ethanol on glutamate release is not clear. Although multiple mechanisms have been implicated in this action, considering the general inhibitory influence of GABA on glutamatergic neurotransmission, it may be suggested that the inhibitory effect of ethanol on glutamate is due to an initial increase of GABA release, which in turn inhibits the release of glutamate. Whatever the mechanism might be, the inhibitory effect of high intoxicating doses of ethanol leads to several adaptive responses within the glutamatergic system following its chronic administration. Indeed, extracellular glutamate levels are enhanced during withdrawal [39, 41] and long-lasting alterations in glutamate release mechanisms following chronic alcohol intake have also been demonstrated. Thus, following a period of abstinence, ethanol-conditioned stimuli can induce an increase in extracellular glutamate levels in the amygdala [42], suggesting that conditioned responses to extracellular glutamate may participate in environmental cue-induced craving and relapse behavior.
P.76
In several in vivo microdialysis studies, it has been shown that acamprosate can reduce enhanced glutamate levels. Thus, rats which were alcoholized by ethanol inhalation, exhibited enhanced extracellular glutamate levels in the nucleus accumbens, whereas rats which were simultaneously alcoholized and treated orally by acamprosate (400 mg/kg/day), failed to present the increase in glutamate during ethanol withdrawal [41]. Acamprosate was also able to decrease augmented glutamate release in the hippocampus following repeated withdrawal episodes [39]. On the behavioral level, acamprosate reduced context-dependent ethanol effects [43]. In conclusion, enhanced glutamate levels which occur during withdrawal or conditioned withdrawal and which reflect a hyper-glutamatergic state of the brain can be effectively blocked by acamprosate. This hypothesis has recently received further support by studying transgenic mice, which exhibit a hyper-glutamatergic state in their brain and show enhanced alcohol consumption compared to control wild-type mice [44]. A threshold dose of acamprosate, which had no effect on alcohol drinking in control wild-type mice, produced a strong reduction of alcohol intake in the transgenic mice [44], demonstrating a causal relationship between a hyper-glutamatergic system and enhanced alcohol intake as well as the action of acamprosate.
Up to now it is not clear how acamprosate interferes with enhanced glutamate levels. However, several studies have shown an interaction of acamprosate with glutamate receptors and it has been suggested that acamprosate binds to the polyamine binding site of the NMDA receptor [45, 46] and to the metabotropic mGlu5 receptor [7, 8]. This in turn might induce a cascade of genomic processes [1, 6], which could finally alter glutamate release and reuptake mechanisms.
Acamprosate and taurine
Acamprosate presents chemical structural similarities with taurine and particularly shares a similar sulfur group (see Appendix: Chemical structures). Interactions between the endogenous amino acid taurine and ethanol in the central nervous system have been identified in recent years [38, 47]. The amino acid taurine is synthesized from cysteine, and is present in cells at relatively high concentrations in the mM range. Taurine is one of the most abundant amino acids in the brain and plays an integral role in physiological processes such as osmoregulation and neuromodulation. Thus, taurine exerts positive allosteric modulatory effects on neuronal ligand-gated chloride channels (i.e., GABAA and glycine receptors) as well as inhibitory effects on other ligand- and voltage-gated cation channels (i.e., NMDA and Ca2+ channels). Neuroprotective and neuromodulatory functions have also been attributed to taurine. Taurine has been shown to modulate cell excitability [48], to prevent neuronal excitotoxicity [49], to protect neuronal membranes against different toxic damages [50] and to regulate calcium homeostasis [51]. Together, all
P.77
these protective effects result in the membrane stabilization properties of taurine [52]. Behavioral evidence suggests that taurine can alter the locomotor stimulatory, sedating, and motivational effects of ethanol in a dose-dependent manner [38, 47]. Furthermore, microdialysis studies have revealed that in response to ethanol, elevated extracellular levels of taurine can be observed in numerous brain regions, an effect which is shared by acamprosate administration (Figs 1 and 2).
The administration of acute ethanol to rats increases the level of the sulphonated amino acid taurine in many brain regions, including nucleus accumbens [9, 10] (Fig. 1), hippocampus, frontal cortex [41] and amygdala [11, 42]. The increase in taurine microdialysate level seems thus to represent a global answer of the brain to an acute administration of ethanol. Chronic administration of ethanol to rats leads to a progressive increase of the basal level of taurine within the entire brain [10]. Exposure of primary astrocyte culture to iso-osmotic ethanol from 10 to 100 mM leads to cell swelling and the release of taurine. Ethanol-induced cell swelling probably activates volumesensitive channels, and taurine passively diffuses outside the cells along its concentration gradient [12].
The effect of ethanol on taurine has also been tested in Sardinian ethanol-preferring (sP) and non-preferring (sNP) rats. The sP and sNP rats have been selectively outbred from a population of Wistar rats for their high and low
P.78
ethanol consumption respectively [53]. Results indicate that ethanol at high doses (2 g/kg) induced an immediate and significant increase in taurine microdialysate content in the nucleus accumbens of both sP and sNP rats. However, this ethanol-induced taurine release was significantly higher in sNP rats in comparison to sP rats (Fig. 3). Furthermore, the differences in taurine responsiveness to ethanol in sP and sNP rats was not caused by differences in their ethanol absorption, redistribution and elimination, as their blood ethanol elimination followed a similar time course. It is noteworthy that sNP rats displayed an ethanol-induced taurine release similar to that observed in Wistar rats, while the responsiveness of taurine to ethanol administration in sP rats was significantly lower. It is interesting to relate this to the pattern of ethanol consumption in these rats, as Wistar and sNP rats are reluctant to consume ethanol, while sP rats voluntarily drink large quantities of ethanol in a free-choice procedure [53], suggesting that there is an inverse relationship between the intensity of ethanol-induced extracellular taurine release and ethanol preference.
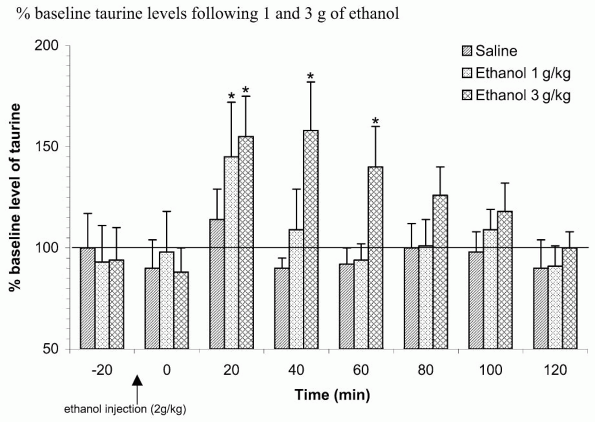 |
Figure 1. Effects of an acute IP injection of 1 and 3 g/kg ethanol (IP) or saline on extracellular taurine levels in the nucleus accumbens in na ve rats. Data are presented as means + S.E.M. * indicates significant differences to baseline. |
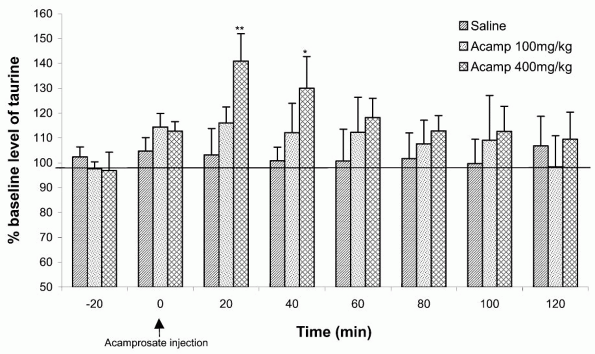 |
Figure 2. Effects of an acute IP injection of 100 and 400 mg/kg acamprosate (IP) or saline on extracellular taurine levels in the nucleus accumbens in na ve rats. Data are presented as means + S.E.M. * indicates significant differences to baseline. |
Another in vivo microdialysis study was conducted to study the effect of ethanol administration on brain taurine levels in High-Alcohol Sensitive (HAS) and Low-Alcohol Sensitive (LAS) rats. These rat lines were used to test whether the effects of acute ethanol on extracellular concentrations of taurine might be related to genetic differences in ethanol sensitivities. HAS and LAS rats from the University of Colorado were genetically selected according to their differential sensitivities to the hypnotic effects of acute ethanol [54]. Using microdialysis in awake and freely moving animals, HAS rats displayed a reduced ethanol-induced taurine release in comparison to LAS rats (Fig. 4).
P.79
Taurine responsiveness to ethanol was therefore inversely related to initial ethanol sensitivity in these genetically selected rat lines. This result is unlikely to be due to pharmacokinetic differences between HAS and LAS rats in ethanol absorption, distribution and elimination, since no differences in blood ethanol concentrations in these two lines of rats were observed [55]. Taurine may contribute to the reduction of several ethanol-adverse effects and this may thus explain why HAS rats show a reduced release of this regulatory amino acid following ethanol administration. On the contrary, in LAS, a higher taurine release may oppose some of the adverse effects of ethanol, thereby contributing to their overall lower sensitivity to ethanol.
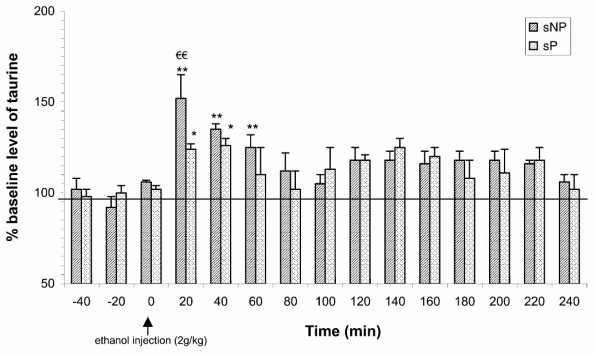 |
Figure 3. Time course of extracellular taurine content of the microdialysate from the nucleus accumbens before and after an acute intraperitoneal injection of 2.0 g/kg ethanol to either sNP or sP rats. Data are expressed as mean (+ S.E.M.) percentage of baseline level which was calculated for each rat by averaging the concentration of the three sample values before injection. *P < 0.05 relative to respective control group injected with saline; **P < 0.01 relative to respective control group injected with saline; and P < 0.01 relative to sP rats. |
With respect to the effects of ethanol on brain taurine levels, similar observations have been made with acamprosate. Thus, the IP injection of 400 mg/kg acamprosate induced a significant increase of extra-cellular taurine concentration in the nucleus accumbens lasting for 40 min (Fig. 2). An IP injection of 1 g/kg acamprosate induced an even larger increase in brain taurine lasting at least 3 h following injection [38]. Pretreatment with acamprosate orally for 30 days in the drinking bottle at 400 mg/kg induced an augmentation of taurine release in both sP and sNP rats. In this experiment, the acamprosate-pretreated sP group obtained brain taurine levels following ethanol injection similar to the brain taurine levels in sNP rats, without a pretreatment of acamprosate suggesting that the release of taurine after acute administration of ethanol in the
P.80
sNP group could be involved in their avoidance of alcohol intake. These studies suggest that acamprosate reduces the preference for alcohol in sP rats through the release of brain taurine.
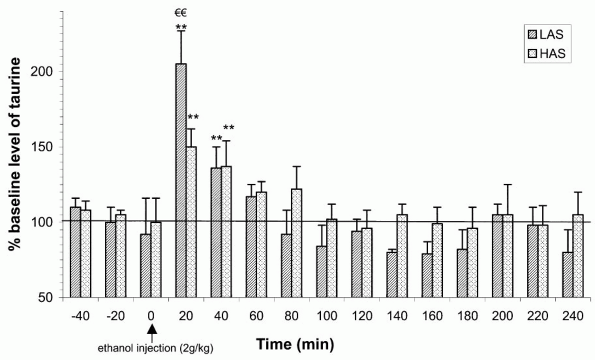 |
Figure 4. Time course of extracellular taurine content of the microdialysate from the nucleus accumbens before and after an acute intraperitoneal injection of 2.0 g/kg ethanol to either LAS or HAS rats. Data are expressed as mean (+S.E.M.) percentage of baseline level which was calculated for each rat by averaging the concentration of the three sample values before injection. ** P < 0.01 relative to respective control group injected with saline; and P < 0.01 relative to HAS rats. |
In summary, the endogenous taurine system may be an important modulator of the effects of ethanol on the central nervous system and might counteract some of ethanol's adverse effects. Externally added taurine, such as in taurine-supplementation studies [13], might therefore modulate ethanol consumption, probably by preventing ethanol-adverse effects. The action of acamprosate on ethanol consumption might also be mediated, at least in part, by taurine. However, it is still unclear how and where taurine acts, as is the relationship between taurine and glutamate.
Conclusion
To achieve its therapeutic effects, acamprosate may act on several neurochemical systems. However, there is a clear interaction with the glutamatergic system and there is convincing evidence that acamprosate effectively reduces a hyper-glutamatergic state, which may trigger, at least in some alcoholic patients, craving and relapse. Whether this dampening effect on a hyper-glutamatergic state is due to binding on the polyamine site of the NMDA receptor
P.81
or mGluR5 and the thereby induced cascade of genomic processes, remains to be further investigated. Furthermore, the relationship between the effects of acamprosate on the endogenous taurine system and a hyper-glutamatergic state is still not well understood; however, it might be due to taurine producing a protective effect in this respect. A better understanding of the precise mode of action of acamprosate would help in identifying treatment responders (individually adapted pharmacotherapy; see also project PREDICT in the Chapter by Mann) and could lead to compound optimization.
References
1 Spanagel R, Zieglgansberger W (1997) Anti-craving compounds for ethanol: new pharmacological tools to study addictive processes. Trends Pharmacol Sci 18(2): 54-59
2 Zornoza T, Cano MJ, Polache A, Granero L (2003) Pharmacology of acamprosate: an overview. CNS Drug Rev 9(4): 359-374
3 Spanagel R, Zieglg nsberger W, Hundt W (1996) Acamprosate and alcohol: III. Effects on alcohol discrimination in the rat. Eur J Pharmacol 305: 51-56
4 Kratzer U, Schmidt WJ (2003) Acamprosate does not induce a conditioned place preference and reveals no state-dependent effects in this paradigm. Prog Neuropsychopharmacol Biol Psychiatry 27: 653-656
5 Littleton J, Zieglgansberger W (2003) Pharmacological mechanisms of naltrexone and acamprosate in the prevention of relapse in alcohol dependence. Am J Addict 12 Suppl 1: S3-11
6 Rammes G, Mahal B, Putzke J, Parsons C, Spielmanns P, Pestel E, Spanagel R, Zieglgansberger W, Schadrack J (2001) The anti-craving compound acamprosate acts as a weak NMDA-receptor antagonist, but modulates NMDA-receptor subunit expression similar to memantine and MK-801. Neuropharmacology 40(6): 749-760
7 Harris BR, Prendergast MA, Gibson DA, Rogers DT, Blanchard JA, Holley RC, Fu MC, Hart SR, Pedigo NW, Littleton JM (2002) Acamprosate inhibits the binding and neurotoxic effects of trans-ACPD, suggesting a novel site of action at metabotropic glutamate receptors. Alcohol Clin Exp Res 26(12): 1779-1793
8 Harris BR, Gibson DA, Prendergast MA, Blanchard JA, Holley RC, Hart SR, Scotland RL, Foster TC, Pedigo NW, Littleton JM (2003) The neurotoxicity induced by ethanol withdrawal in mature oranotypic hippocampal slices might involve cross-talk between metabotropic glutamate type 5 receptors and N-methyl-D-aspartate receptors. Alcohol Clin Exp Res 27(11): 1724-1735
9 Dahchour A, Quertemont E, De Witte P (1994) Acute ethanol increases taurine but neither glutamate nor GABA in the nucleus accumbens of male rats: a microdialysis study. Alcohol Alcoholism 29: 485-487
10 Dahchour A, De Witte P (1996) Taurine increases in the nucleus accumbens microdialysate after acute ethanol administration to naive and chronically alcoholised rats. Brain Res 735: 9-19
11 Quertemont E, Dahchour A, Ward RJ, De Witte P (1999) Ethanol induces taurine release in the amygdala: an in vivo microdialysis study. Addict Biol 4: 47-54
12 Quertemont E, Devitgh A, De Witte P (2003) Systemic osmotic manipulations modulate ethanol-induced taurine release: a brain microdialysis study. Alcohol 29: 11-19
13 Quertemont E, Goffaux V, Vlaminck AM, Wolf C, De Witte P (1998) Oral taurine supplementation modulates ethanol-conditioned stimulus preference. Alcohol 16: 201-206
14 Le Magnen J, Tran G, Durlach J (1987) Lack of effects of Ca-acetyl homotaurinate on chronic and acute toxicities of ethanol in rats. Alcohol 4(2): 103-108
15 McGeehan AJ, Olive MF (2003) The anti-relapse compound acamprosate inhibits the development of a conditioned preference to ethanol and cocaine but not morphine. Br J Pharmacol 138: 9-12
16 Kiefer F, Wiedemann K (2002) Aktueller Stand der pharmakotherapeutischen R ckfallprophylaxe bei Alkoholabh ngigkeit mit Acamprosat und Naltrexon. Nervenheilkunde 4: 183-192
17 Littleton J (1995) Acamprosate in alcohol dependence: how does it work? Addiction 90: 1179-1188
P.82
18 Boismare F, Daoust M, Moore ND, Saligaut C, Lhuintre JP, Chretien P, Durlach J (1984) A homotaurine derivate reduces the voluntary intake of ethanol by rats. Are cerebral GABA receptors involved? Pharmacol Biochem Behav 21: 787-789
19 Le Magnen J, Tran G, Durlach J, Martin C (1987) Dose-dependent suppression of the high alcohol intake of chronically intoxicated rats by Ca-acetylhomotaurinate. Alcohol 4(2): 97-102
20 Gewiss M, Heidbreder C, Opsomer L, Durbin P, De Witte P (1991) Acamprosate and diazepam differentially modulate alcohol-induced behavioural and cortical alterations in rats following chronic inhalation of ethanol vapor. Alcohol Alcoholism 26(2): 129-137
21 Stromberg MF, Mackler SA, Volpicelli JR, O'Brien CP (2001) Effect of acamprosate and naltrexone, alone or in combination, on ethanol consumption. Alcohol 23(2): 109-116
22 Heyser CJ, Schulteis G, Durbin P, Koob GF (1998) Chronic acamprosate eliminates the alcohol deprivation effect while having limited effects on baseline responding for ethanol in rats. Neuropsychopharmacology 18: 125-133
23 Czachowski CL, Legg BH, Samson HH (2001) Effects of acamprosate on ethanol-seeking and self-administration in the rat. Alcohol Clin Exp Res 25(3): 344-350
24 Olive MF, Nannini MA, Ou CJ, Koenig HN, Hodge CW (2002) Effects of acute acamprosate and homotaurine on ethanol intake and ethanol-stimulated mesolimbic dopamine release. Eur J Pharmacol 437(1-2): 55-61
25 H lter SM, Landgraf R, Zieglg nsberger W, Spanagel R (1997) Time course of acamprosate action on operant ethanol self-administration after ethanol deprivation. Alcohol Clin Exp Res 21(5): 862-868
26 Heyser CJ, Moc K, Koob GF (2003) Effects of naltrexone alone and in combination with acamprosate on the alcohol deprivation effect in rats. Neuropsychopharmacology 28(8): 1463-1471
27 Sinclair JD, Senter RJ (1967) Increased preference for ethanol in rats following alcohol deprivation. Psycho Sci 8: 11
28 Spanagel R, H lter SM (1999) Long-term alcohol self-administration with repeated alcohol deprivation phases: an animal model of alcoholism? Alcohol Alcoholism 34: 231-243
29 Spanagel R, H lter SM, Allingham K, Landgraf R (1996) Acamprosate and alcohol: I. Effects on alcohol intake following alcohol deprivation in the rat. Eur J Pharmacol 305(1-3): 39-44
30 Bachteler D, Economidou D, Danysz W, Ciccocioppo R, Spanagel R. The effects of acamprosate and neramexane on cue-induced reinstatement of ethanol-seeking behavior in rat. Neuropsychopharmacology; in press
31 Shaham Y, Shalev U, Lu L, de Wit H, Stewart J (2003) The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology 168: 3-20
32 Al Qatari M, Khan S, Harris B, Littleton J (2001) Acamprosate is neuroprotective against glutamate-induced excitotoxicity when enhanced by ethanol withdrawal in neocotrical cultures of fetal rat brain. Alcohol Clin Exp Res 25: 1276-1283
33 Koob GF, Mason BJ, De Witte P, Littleton J, Siggins GR (2002) Potential neuroprotective effects of acamprosate. Alcohol Clin Exp Res 26(4): 586-592
34 Mayer S, Harris BR, Gibson DA, Blanchard JA, Prendergast MA, Holley RC, Littleton J (2002) Acamprosate, MK-801, and ifenprodil inhibit neurotoxicity and calcium entry induced by ethanol withdrawal in organotypic slice cultures from neonatal rat hippocampus. Alcohol Clin Exp Res 26: 1468-1478
35 Tsai G, Coyle JT (1998) The role of glutamatergic neurotransmission in the pathophysiology of alcoholism. Annu Rev Med 49: 173-184
36 Le A, Shaham Y (2002) Neurobiology of relapse to alcohol in rats. Pharmacol Ther 94(1-2): 137-156
37 Weiss F, Porrino LJ (2002) Behavioral neurobiology of alcohol addiction: recent advances and challenges. J Neurosci 22(9): 332-3337
38 Dahchour A, De Witte P (2000) Ethanol and aminoacids in the central nervous system: assessment of the pharmacological actions of acamprosate. Prog Neurobiol 60: 343-362
39 Dahchour A, De Witte P (2003) Effects of acamprosate on excitatory amino acids during multiple ethanol withdrawal periods. Alcohol Clin Exp Res 27(3): 465-470
40 Spanagel R, Bienkowski P (2002) Glutamatergic mechanisms in alcohol dependence and addiction. In: D Lodge, W Danysz, CG Parsons (eds): Therapeutic Potential of Ionotropic Glutamate Receptor Antagonists and Modulators. Graham Publishing Co., Johnson City, TN, USA, 375-403
P.83
41 Dahchour A, De Witte P, Bolo N, Nedelec JF, Muzet M, Durbin P, Macher JP (1998) Central effects of acamprosate: Part 1: acamprosate blocks the glutamate increase in the nucleus accumbens microdialysate in ethanol withdrawn rats. Psychiat Res-Neuroimag 82: 107-114
42 Quertemont E, De Neuville J, De Witte P (1998) Changes in the amygdala amino acid microdialysate after conditioning with a cue associated with ethanol. Psychopharmacology 139: 71-78
43 Quertemont E, Brabant C, De Witte P (2002) Acamprosate reduced context-dependent ethanol effects. Psychopharmacology 164(1): 10-18
44 Spanagel R, Pendyala G, Abarca C, Zghoul T, Sanchis-Segura C, Magnone MC, Lascorz J, Depner M, Holzberg D, Soyka M et al. (2005) The circadian clock gene Period2 alters the glutamatergic system and thereby modulates alcohol consumption. Nature Med; in press
45 Naassila M, Hammoumi S, Legrand E, Durbin P, Daoust M (1998) Mechanism of action of acamprosate. Part I. Characterization of spermidine-sensitive acamprosate binding site in rat brain. Alcohol Clin Exp Res 22(4): 802-809
46 Popp RL, Lovinger DM (2000) Interaction of acamprosate with ethanol and spermine on NMDA receptors in primary cultured neurons. Eur J Pharmacol 394(2-3): 221-231
47 Olive MF (2002) Interactions between taurine and ethanol in the central nervous system. Amino Acids 23(4): 345-357
48 Huxtable RJ (1989) Taurine in the central nervous system and the mammalian actions of taurine. Prog Neurobiol 32: 471-533
49 Zhao P, Huang YL, Cheng JS (1999) Taurine antagonizes calcium overload induced by glutamate of chemical hypoxia in cultured rat hippocampal neurons. Neurosci Lett 268: 25-28
50 Timbrell JA, Seabra V, Waterfield CJ (1995) The in vivo and in vitro protective properties of taurine. Gen Pharmac 26: 453-462
51 El Idrissi A, Trenkea E (1999) Growth factors and taurine protect against excitotoxicity by stabilizing calcium homeostasis and energy metabolism. J Neurosci 19: 9459-9468
52 Wright CE, Tallan HH, Lin YY, Gaull GE (1986) Taurine: biological update. Annu Rev Biochem 55: 427-453
53 Colombo G (1997) Ethanol drinking behaviour in sardininan alcohol-preferring rats. Alcohol Alcoholism 32: 443-453
54 Draski LJ, Spuhler KP, Erwin VG, Baker RC, Deitrich RA (1992) Selective breeding of rats differing in sensitivity to the effects of acute ethanol administration. Alcohol Clin Exp Res 16: 48-54
55 Dahchour A, Hoffman A, Deitrich R, De Witte P (2000) Effects of ethanol on extracellular amino acid levels in High and Low-alcohol sensitive rats: a microdialysis study. Alcohol Alcoholism 35: 548-553
EAN: 2147483647
Pages: 26