28 - Liver
Editors: Mills, Stacey E.
Title: Histology for Pathologists, 3rd Edition
Copyright 2007 Lippincott Williams & Wilkins
> Table of Contents > IX - Genitourinary Tract > 35 - Urinary Bladder, Ureter, and Renal Pelvis
35
Urinary Bladder, Ureter, and Renal Pelvis
Victor E. Reuter
Introduction
The urinary bladder is an epithelial-lined muscular viscus that has the ability to distend and accommodate up to 400 to 500 mL of urine without a change in intraluminal pressure. In addition, it is able to initiate and sustain a contraction until the organ is empty. Interestingly, micturition may be initiated or inhibited voluntarily despite the involuntary nature of the organ. The ureters are epithelial lined muscular tubes designed to transport urine from the kidneys to the urinary bladder with the aid of peristalsis. The renal pelvis represents the expanded proximal end of the ureter and serves to collect the urine excreted from the kidney and to transport it to the ureter proper.
Embryology
The cloaca is divided by the urorectal septum into a dorsal rectum and a ventral urogenital sinus (1,2). It is this urogenital sinus that will give rise to the majority of the urinary bladder; it is aided by the caudal migration of the cloacal membrane, which will close the infraumbilical portion of the abdominal wall. The caudal portions of the mesonephric ducts become dilated and eventually fuse with the urogenital sinus in the midline dorsally, contributing to the formation of the bladder trigone. While these ducts contribute initially to the formation of the mucosa of the trigone, this is subsequently entirely replaced by endodermal epithelium of the urogenital sinus. The gradual absorption of the mesonephric ducts bring about the separate opening of the ureters into the urinary bladder in the area of the trigone. During embryologic development, the allantois regresses to completely form a thick, epithelial-lined tube, the urachus, which extends from the umbilicus to the apex (dome) of the bladder (1). Before or shortly after birth, the urachus involutes further becoming simply a fibrous cord. Pathologists commonly refer to this fibrous cord, which extends from the dome of the bladder to the umbilicus, as the urachal remnant, but it should be called the median umbilical ligament since urachal remnant refers to remnants of the epithelial lining of the urachus that occasionally persist
P.910
within the median umbilical ligament (Figure 35.1). The epithelial lining of the urachus is urothelium, similar to that of the urinary bladder and ureter, but it frequently undergoes metaplastic change that is mostly of a glandular nature.
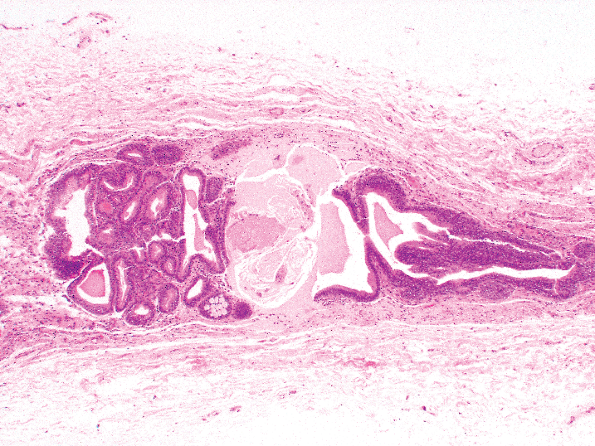 |
Figure 35.1 Urachal remnants within the median umbilical ligament. |
The epithelium of the urinary bladder is endodermally derived from the cranial portion of the urogenital sinus in continuity with the allantois. The lamina propria, the muscularis propria, and the adventitia develop from the adjacent splanchnic mesenchyme. These facts are important in understanding the histogenesis and nomenclature of lesions arising from the epithelial surface, as well as the bladder wall. For example, glandular features within benign (cystitis glandularis, nephrogenic adenoma) and malignant (adenocarcinoma) urothelium is not due to mesodermal or m llerian rests within the trigone but come about through a process of metaplasia and are a reflection of histologic plasticity (multipotentiality) of the urothelium. Since the mesonephric ducts involute totally during embryologic development, it is wrong to refer to tumors with mixed epithelial and sarcomatoid features arising in the bladder epithelium as mesodermal mixed tumors. They are, in fact, endodermal mixed tumors and are usually called sarcomatoid carcinomas (3). Very rarely, m llerian rests may be identified in the wall of the bladder and ureters or in the surrounding soft tissues in the form of endometriosis, endocervicosis, or endosalpingiosis (so-called m llerianosis) (4,5,6,7) (Figure 35.2).
The ureters develop by branching and elongation of the ureteric bud (metanephric diverticulum), which begins as a dorsal bud from the mesonephric duct (1,2). The stalk of the ureteric bud becomes the ureter, while the cranial end forms the renal pelvis, calyces, and collecting tubules. The epithelium of the ureter and renal pelvis, although histologically identical to that of the bladder, is of mesodermal derivation.
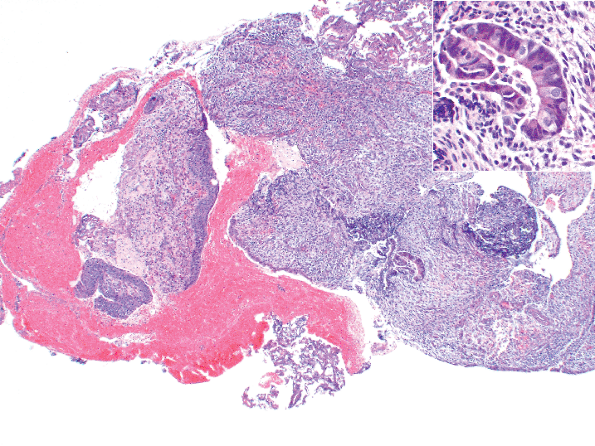 |
Figure 35.2 Endometriosis involving the ureteral wall. This female patient presented with hematuria and was thought to have a primary ureteral neoplasm. Insert shows an endometriotic gland at higher magnification. |
Anatomic Considerations
Bladder
In the adult, the empty urinary bladder lies within the anteroinferior portion of the pelvis minor, inferior to the peritoneum. In infants and children, it is located in part within the abdomen, even when empty (8). It begins to enter the pelvis major at about 6 years of age and will not be found entirely within the pelvis minor until after puberty. Nevertheless, in adults, as the bladder fills it will distend and ascend into the abdomen, at which time it may reach the level of the umbilicus.
The bladder lies relatively free within the fibrofatty tissues of the pelvis except in the area of the bladder neck, where it is firmly secured by the pubovesical ligaments in the female and the puboprostatic ligaments in the male (8,9). The relative freedom of the rest of the bladder permits expansion superiorly as the viscus fills with urine.
The empty bladder in an adult has the shape of a four-sided inverted pyramid and is enveloped by the vesical fascia (8). The superior surface faces superiorly and is covered by the pelvic parietal peritoneum (Figures 35.3,35.4). The posterior surface, also known as the base of the bladder, faces posteriorly and inferiorly. It is separated from the rectum by the uterine cervix and the proximal portions of the vagina in females and by the seminal vesicles and the ampulla of the vasa deferentia in males. These posterior anatomic relationships are very important clinically. Since the majority of vesicle neoplasms arise in the posterior wall adjacent to the ureteral orifices, invasive tumor may extend into adjacent soft tissue and organs (Figure 35.5A). The intimate relationship to the previously mentioned organs explains why hysterectomy and partial vaginectomy are commonly performed at the time of radial cystectomy in
P.911
women. Similarly, we know that perivesical and seminal vesicle involvement is a bad prognostic sign in bladder carcinoma in males (10,11,12), a reflection of high pathologic stage. It is important to note that seminal vesicles may contain carcinoma without invasion, and this occurs in cases of in situ urothelial carcinoma involving prostatic and ejaculatory ducts and extending into the seminal vesicle epithelium. The latter is a rare occurrence, but these patients do not appear to have a similarly bad prognosis unless prostatic stromal invasion is present. The two inferolateral surfaces of the bladder face laterally, inferiorly, and anteriorly and are in contact with the fascia of the levator ani muscles. The most anterosuperior point of the bladder is known as the apex, and it is located at the point of contact of the superior surfaces and the two inferolateral surfaces. The apex (dome) marks the point of insertion of the median umbilical ligament and consequently is the area where urachal carcinomas are located (Figures 35.3,35.4).
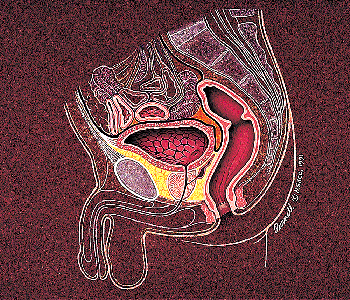 |
Figure 35.3 Anatomical relationships of the urinary bladder in males. |
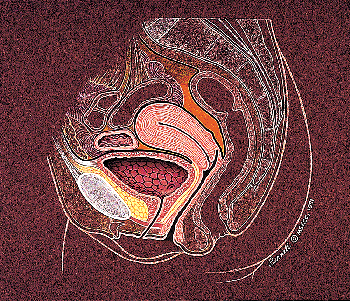 |
Figure 35.4 Anatomical relationships of the urinary bladder in females. |
The trigone is a complex anatomic structure located at the base of the bladder and extending to the posterior bladder neck. In the proximal and lateral aspects of the trigone, the ureters enter into the bladder (ureteral orifices) obliquely. The muscle underlying the mucosa in this region is a combination of smooth muscle of the longitudinal layer of the intramural ureter and detrusor muscle (13,14,15,16,17). The intramural ureter is surrounded by a fibromuscular sheath (Waldeyer's sheath) that is fused into the ureteral muscle. This fibromuscular tissue fans out in the area of the
P.912
trigone and mixes with the detrusor muscle, thus fixing the intramural ureter to the bladder. As the bladder distends, the surrounding musculature exerts pressure on the obliquely oriented intramural ureter, producing closure of the ureteral lumen and thus avoiding reflux of urine.
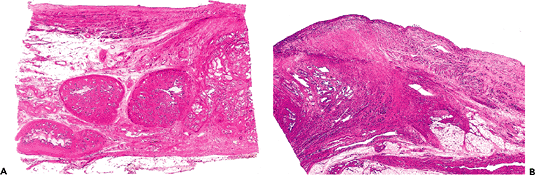 |
Figure 35.5 Bladder neck and distal trigone. A. The seminal vesicles are separated from the muscularis propria of the trigone by a scant amount of soft tissue. B. The muscularis propria merges with the prostate in the bladder neck area. The central (circular) fibers will predominate in this area and form the internal sphincter. The outer longitudinal layer contributes somewhat to the formation of the prostate musculature. |
The most distal portion of the bladder is called the bladder neck, and it is marked by the area where the posterior and the inferolateral walls converge and open into the urethra. In the male, the bladder neck merges with the prostate gland, and one may occasionally observe several prostatic ducts present in this area. It is important to recognize the existence of these ducts since their involvement by carcinoma should not be mistaken with invasive carcinoma. The bladder neck is formed with contributions from the trigonal musculature (inner longitudinal ureteral muscle and Waldeyer's sheath), the detrusor musculature, and the urethral musculature (13,14,15,16,17,18). The internal sphincter is located in this general area, with major contributions from the middle circular layer of the detrusor muscle (Figure 35.5B).
The bladder bed (structures on which the bladder neck rests) is formed posteriorly by the rectum in males and vagina in females (Figures 35.3 and 35.4). Anteriorly and laterally it is formed by the internal obturator and levator ani muscles, as well as the pubic bones. These structures may be involved in advanced tumors occupying the anterior, lateral, or bladder neck regions and render the patient inoperable.
The main arterial blood supply of the bladder comes from the inferior vesical arteries that branch from the internal iliacs (19,20). The umbilical arteries and its branches (the superior vesical arteries) also supply the bladder, as do the obturator and inferior gluteal arteries and, in females, the uterine and vaginal arteries. The veins of the urinary bladder drain into the internal iliac veins and form the vesical venous plexus. In the male, this plexus envelops the bladder base, prostate, and seminal vesicles and connects with the prostatic venous plexus. In females, it covers the bladder neck and urethra and communicates with the vaginal plexus. Lymphatic drainage is through the external and internal lymph nodes although drainage of portions of the bladder neck region may be through the sacral or common iliac nodes.
The urinary bladder is supplied by both sympathetic and parasympathetic nerves, which form the vesical nerve plexus (19,20). The former are derived from T11 through L2 nerves and play no role in micturition. On the other hand, the parasympathetic nerves come from S2 through S4 and travel to the bladder via the pelvic nerve and inferior hypogastric plexus. These nerves are important to micturition since they contract the fibers of the muscularis propria, which in turn produces traction upon the bladder neck and opens the internal sphincter of the bladder. In fact, it is believed that micturition is initiated by voluntary relaxation of the perineal muscles and the striated muscle of the external sphincter located along the urethra. This action decreases urethral resistance and triggers contraction of the smooth muscle of the trigone and remaining bladder, closing the ureteral orifices and increasing the hydrostatic pressure within the viscus (9,21). These facts account for the difficulty in starting micturition while whistling. The bladder also contains sensory nerves that travel along the pelvic and hypogastric nerves and account for the sensation of pain as the bladder becomes too distended.
Ureters
The ureters measure approximately 30.0 cm in length, equally divided between the abdomen (retroperitoneum) and pelvis (22,23,24,25,26,27). The abdominal ureter takes a vertical course downward and medially on the anterior surface of the psoas muscle, covered by adventitia that is in fact an extension of Gerota's fascia. The pelvic ureter can be subdivided into a longer parietal and a shorter intravesical portion. The parietal portion is intimately related to the peritoneum. It descends posterolaterally; and, as it approaches the bladder base, it becomes medially directed to reach the urinary bladder. The ureters enter the base of the bladder obliquely and empty into the bladder at the ureteral orifices. The distal parietal portion and the intravesical segments are enveloped in a fibromuscular sheath (Waldeyer's sheath), which aids in fixing the ureter to the bladder (see description of the trigone).
The ureteral blood supply is quite diverse (19,22). Depending on the anatomic level, it receives blood from branches of the renal, abdominal aortic, gonadal, hypogastric, vesical, and uterine arteries, which form a richly intercommunicating plexus of vessels surrounding the tube. Venous drainage is variable but tends to follow a pattern similar to the arterial distribution. Lymphatic drainage is also quite complex. The upper portions drain into the lateral aortic lymph nodes, the middle portion drains into the common iliac lymph nodes, and the inferior portion drains into either the common, external, or internal iliac lymph nodes.
Renal Pelvis
As previously mentioned, the renal pelvis has its origin in the cranial portion of the ureteric bud, together with the calyces and collecting ducts. The renal pelvis lies primarily within the renal hilum, a space formed medially when one draws a vertical plane through the medial aspects of the upper and lower poles of the kidney (Figure 35.6). Within the hilum is the renal sinus, a space within the medial and antral portion of the kidney occupied by the renal pelvis, renal vessels and nerves, renal calyces, and fat. The fibrous capsule that lines the kidney passes over the lips of the hilum and lines the renal sinus, becoming continuous with the renal calyces. Within the renal sinus, the renal pelvis divides into two and rarely three major calyces, which in turn divide into 7 to 14 minor calyces. Urine from the distal collecting ducts within the renal
P.913
medulla (ducts of Bellini) flows into the minor calyces at the tips of the renal papillae (area cribrosa) (Figures 35.6 and 35.7). The blood supply of the renal pelvis comes from branches of the renal arteries, and the venous drainage follows a similar distribution. Its lymphatic drainage is into the renal hilar lymph nodes.
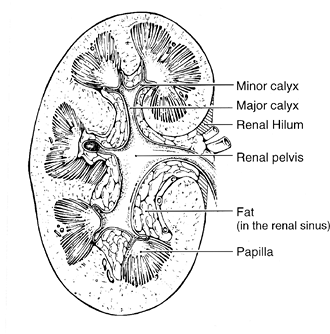 |
Figure 35.6 Anatomical relationships of the renal pelvis. Notice that the pelvis is mostly within the renal hilum (medial shaded area) and the renal sinus. |
Microscopic Anatomy
The urinary bladder, ureter, and renal pelvis for the most part have a similar anatomic composition, the innermost layer being an epithelial lining and, extending outward, a lamina propria, smooth muscle (muscularis propria), and adventitia. The superior surface of the bladder comes in contact with parietal peritoneum and hence has a serosa. The anatomic landmarks are used clinically and pathologically to stage patients with urothelial cancer in order to choose therapy and estimate survival (Figure 35.8). For this reason, it is important to accurately identify them microscopically.
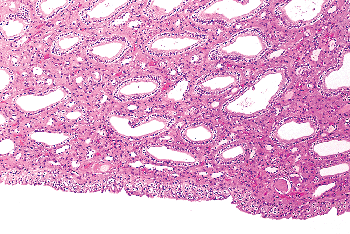 |
Figure 35.7 Renal papilla. Distal collecting ducts open into the urothelium covering the papillae. |
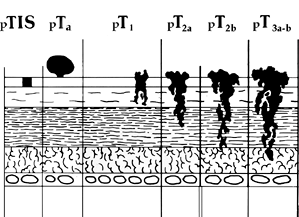 |
Figure 35.8 Pathologic staging of bladder cancer. This classification follows the recommendations of the American Joint Committee on Cancer (AJCC). In addition, prostatic stromal invasion is considered stage pT4. |
Urothelium
The urinary bladder, ureters, and renal pelvis are lined by so-called transitional epithelium. This name was coined because its histologic appearance was transitional between nonkeratinizing squamous and pseudostratified columnar. Many histologists and pathologists have suggested urothelium as a more appropriate term.
The thickness of the urothelium will vary according to the degree of distension and anatomical location. It may be only two or three cell layers thick along the minor calyces. In the contracted bladder, it is usually six to seven cells thick and in the ureter three to five cells thick. One can identify three regions: the superficial cells that are in contact with the urinary space, the intermediate cells, and the basal cells that lie on a basement membrane (28,29) (Figure 35.9). In the distended bladder, the urothelium
P.914
may be only two to three cells thick and flattened with their long axis horizontal to the basement membrane. In practice, the thickness of the urothelium is dependent not only in the degree of distension but also on the plane on which the tissue is cut. If the cut is tangential to the basement membrane, it is possible to generate an artificially thick mucosa. For these and other reasons, we feel that urothelial thickness is of marginal or no utility in the assessment of urothelial neoplasms.
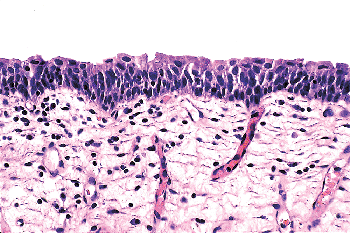 |
Figure 35.9 Normal urothelium. The mucosa may be up to seven cells thick in the bladder, but thickness will vary as a consequence of distension and other factors. The superficial (umbrella) cells have ample eosinophilic cytoplasm. |
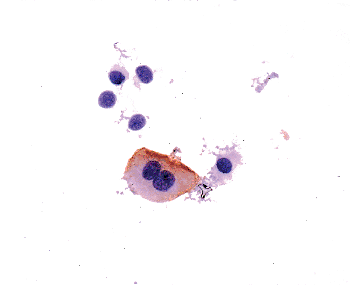 |
Figure 35.10 Urine cytology preparation stained with monoclonal antibody CD-15. A large, binucleated umbrella cell expresses the antigen identified by this antibody, while other normal urothelial cells do not. |
Superficial cells are in contact with the urinary space. They are large, elliptical cells that lie umbrella-like over the smaller intermediate cells (28,29,30,31). They may be binucleated and have abundant eosinophilic cytoplasm (Figure 35.10). In the distended bladder, they become flattened and barely discernible. While the presence of these cells is taken as a sign of normalcy of the urothelium, one must be aware that they may become detached due to superficial erosion during instrumentation or tissue processing in the prosecting area. Conversely, it is possible to see umbrella cells overlying frank carcinoma. In summary, the presence or absence of superficial cells cannot be used as a determining factor of malignancy.
Ultrastructural studies have shown the superficial urothelial cells to be quite unique. The luminal surface is lined by a cytoplasmic membrane that is three layers thick, having two electron-dense layers and a central lucent layer (Figure 35.11A). The two dense layers are said to be unequal thickness; for this reason, the membrane is known as the asymmetric unit membrane (AUM) (30,31,32,33,34). In reality, while the trilaminar arrangement of the cytoplasmic membrane can be readily observed, it is difficult to see the asymmetry of the dense layers. The membrane contains frequent invaginations, giving it a scalloped
P.915
appearance. The superficial (luminal) cytoplasm contains vesicles that are also lined by AUM (Figure 35.11B). During the process of distention, these invaginations and vesicles are incorporated into the surface membrane, thus increasing the surface area and maintaining the structural integrity of the urothelium.
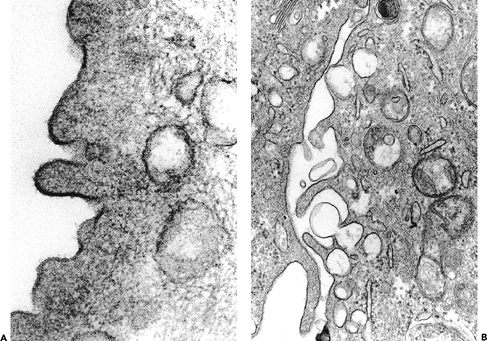 |
Figure 35.11 Ultrastructure of the urothelium. A. Detail of the luminal surface of a urothelial cell from a 1-year-old infant. The trilaminar cell membrane does not appear to be asymmetric ( 136,000). B. Vesicles communicating with the apical/lateral surface of a urothelial cell ( 54,000). |
The intermediate cell layer may be up to five cells thick in the contracted bladder, where they are oriented with the long axis perpendicular to the basement membrane. The nuclei are oval and have finely stippled chromatin with absent or minute nucleoli. There is ample cytoplasm, which may be vacuolated. The cytoplasmic membranes are distinct, and these cells are attached to each other by desmosomes. In the distended state, this layer may be inconspicuous or only one cell thick and flattened. The basal layer is composed of cuboidal cells that are evident only in the contracted bladder and which lie on a thin but continuous basement membrane composed of a lamina lucida, lamina densa, and anchoring fibrils (35). All normal urothelial cells may contain glycogen, but only the superficial cells are occasional mucicarminophilic.
Urothelial Variants and Benign Urothelial Proliferations
While the above microscopic and ultrastructural features describe normal urothelium, we know there are many benign morphologic variants. Koss studied 100 grossly normal bladders obtained postmortem (36). Of these, 93% had Brunn's nests, cystitis cystica, or squamous metaplasia.
The most common urothelial variant is the formation of Brunn's nests, which represent invaginations of the surface urothelium into the underlying lamina propria (Figure 35.12). In some cases these solid nests of benign-appearing urothelium may lose continuity with the surface. They become cystic due to accumulation of cellular debris or mucin, and the term cystitis cystica has been coined to describe this phenomenon. The lining epithelium of these small cysts is composed of one or several layers of flattened transitional or cuboidal epithelium. In some cases, the epithelial lining undergoes glandular metaplasia, giving rise to what is called cystitis glandularis (Figure 35.12). The cells become cuboidal or columnar and mucin secreting; some are transformed into goblet cells. These processes also occur in the renal pelvis and ureter, where they are called pyelitis or ureteritis cystica (or glandularis), respectively (Figure 35.13).
Brunn's nests, cystitis cystica, and cystitis glandularis represent a continuum of proliferative or reactive changes seen along the entire urothelial tract, and it is common to see all three in the same tissue sample (Figure 35.12). Most investigators believe that they occur as a result of local inflammatory insult (36,37,38). Nevertheless, these proliferative changes are seen in the urothelium of patients with no evidence of local inflammation, so that it is possible that they also represent either normal histologic variants or the residual effects of old inflammatory processes (39,40). The high incidence of these proliferative changes in normal bladder suggests that they are not likely to be premalignant changes and that there is no cause-and-effect relationship between their presence and the appearance of bladder cancer. It is true that one or all of these changes are commonly present in biopsy specimens containing bladder cancer; but, the coexistence may be coincidental, or the cancer itself may be producing the local inflammatory insult that causes them. The fact that exceptional cases may occur in which carcinoma clearly arises within the epithelium of these reactive lesions does not alter this argument (41,42).
 |
Figure 35.12 Bladder urothelium exhibiting proliferative changes, including Brunn's nests and cystitis glandularis. |
Metaplasia refers to a change in morphology of one cell type into another that is considered aberrant for that location. Urothelium frequently undergoes either squamous or glandular metaplasia, presumably as a response to chronic inflammatory stimuli such as urinary tract infection, calculi, diverticula, or frequent catheterization (37,40).
 |
Figure 35.13 Brunn's nests involving a ureter. Notice that the nests are more numerous and irregularly oriented compared to what is commonly seen in the bladder. |
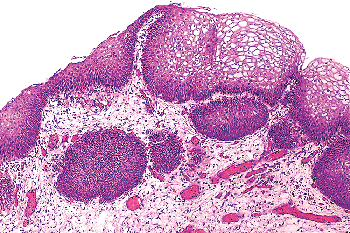 |
Figure 35.14 Squamous epithelium in the trigone of a woman. In this setting squamous epithelium is so common that it is considered to be a normal urothelial variant. |
P.916
Squamous epithelium in the area of the trigone is a common finding in women. It is characterized by abundant intracytoplasmic glycogen and lack of keratinization, making it histologically similar to vaginal or cervical squamous epithelium (Figure 35.14). In this particular setting, most of us believe that squamous epithelium should be regarded as a variant of urothelium rather than metaplasia. Squamous metaplasia may occur at other sites and at times may undergo keratinization and even exhibit parakeratosis and a granular layer. Squamous metaplasia is not preneoplastic per se, but patients with keratinizing squamous metaplasia must be monitored closely since some may progress to squamous carcinoma (43).
The most common site of glandular metaplasia of the urothelium is the bladder, in the form of cystitis glandularis. Nevertheless, it may also occur within surface urothelium elsewhere in the urinary tract, usually as a response to chronic inflammation or irritation and also in cases of bladder exstrophy (44,45). The epithelium is composed of tall columnar cells with mucin-secreting goblet cells (Figure 35.15), strikingly similar to colonic or small intestinal epithelium in which one might identify even Paneth's cells. In this setting, we should use the term intestinal metaplasia. As with squamous metaplasia, glandular metaplasia is not of itself a precancerous lesion but may eventually undergo neoplastic transformation in exceptional cases (45). Patients should be monitored accordingly.
So-called nephrogenic adenoma is a distinct metaplastic lesion characterized by aggregates of cuboidal or hobnail cells with clear or eosinophilic cytoplasm and small discrete nuclei without prominent nucleoli (46). These cells line thin papillary fronds on the surface or form tubular structures within the lamina propria of the bladder (Figure 35.16). The tubules are often surrounded by a thickened and hyalinized basement membrane. Variable numbers of acute and chronic inflammatory cells are commonplace within the bladder wall.
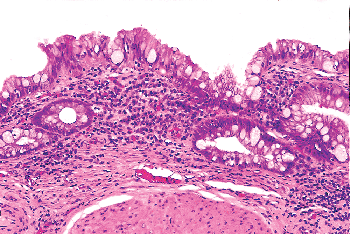 |
Figure 35.15 Intestinal metaplasia. The individual cells are morphologically identical to intestinal-type epithelium, even at the electron microscopic level. |
Nephrogenic adenoma is thought to be secondary to an inflammatory insult or local injury (46,47,48,49,50). It was originally described in the trigone and given its name because it was thought to arise from mesonephric rests. We now know that nephrogenic adenoma may occur anywhere in the urothelial tract, although it is most common in the bladder. It is important in that it may present as an exophytic mass mimicking carcinoma grossly and suggesting adenocarcinoma microscopically. The benign histologic appearance of the cells arranged in characteristic tubules surrounded by a prominent basement membrane should provide the correct diagnosis. A very interesting publication described nephrogenic adenomas of the bladder in patients who underwent renal transplantation (51). The authors demonstrated the adenomatous lesions and the donor kidneys were clonal, suggesting that they developed through a process of shedding of donor renal tubule cells followed by implantation and proliferation within
P.917
the bladder. While this is interesting, it is certainly not the usual mechanism by which they develop.
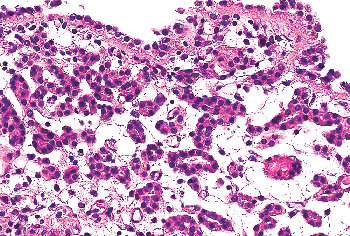 |
Figure 35.16 Nephrogenic adenoma. This proliferative urothelial lesion is characterized by aggregates of cuboidal cells with scant eosinophilic cytoplasm forming small tubules within the lamina propria. It may exhibit an exophytic, papillary growth. |
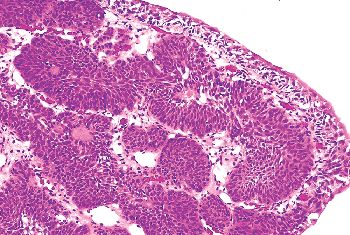 |
Figure 35.17 Inverted papilloma. This proliferative urothelial lesion is characterized by invaginated cords and nests of transitional epithelium within the lamina propria. |
Inverted papillomas are relatively rare lesions that may occur anywhere along the urothelial tract and may be confused clinically and pathologically with transitional-cell carcinoma (52,53). In order of decreasing frequency, they occur in the bladder, renal pelvis, ureter, and urethra (54,55,56,57,58,59,60). Patients usually present with hematuria. Cystoscopically, the lesions are polypoid and either sessile or pedunculated. The mucosal surface is smooth or nodular and without villous or papillary fronds. Microscopically, the surface epithelium is compressed but otherwise unremarkable. It is undermined by invaginated cords and nests of transitional epithelium that occupy the lamina propria (Figure 35.17). The accumulation of these endophytic growths gives the lesion its characteristic polypoid gross appearance. The urothelial cells forming the cords are benign, exhibiting normal maturation and few mitoses. They are similar to the cells of bladder papillomas, differing only in that the epithelial cords are endophytic and consequently more closely packed. Frequently the cells are oval or spindle-shaped. Epithelial nests may become centrally cystic, dilated, and even lined by cuboidal epithelium.
These cords of transitional epithelium in the lamina propria represent invagination, not invasion. As such, there are no fibrous reactive changes within the stroma. Although mitotic figures can be seen, they are rare, regular, and located at or near the basal layer of the epithelium. Inverted papillomas are discrete lesions and do not exhibit an infiltrative border (54,55). One must be careful not to confuse a nested type of urothelial carcinoma infiltrating the lamina propria with an inverted papilloma.
The etiology of inverted papilloma is unclear. Most investigators feel that, similar to other proliferative lesions such as Brunn's nests and cystitis cystica, they are a reactive, proliferative process secondary to a noxious insult. They are not premalignant, although in exceptional cases they have been associated with carcinoma (56,57,58). Given the rarity of this association, we consider it incidental.
Lamina Propria
The lamina propria lies between the mucosal basement membrane and the muscularis propria. It is composed of dense connective tissue containing a rich vascular network, lymphatic channels, sensory nerve endings, and a few elastic fibers (20,28,32). In the deeper aspects of the lamina propria, of the urinary bladder and ureter, the connective tissue is loose, allowing the formation of thick mucosal folds when the viscus is contracted (Figures 35.18,35.19). Its thickness varies with the degree of distention and is generally thinner in the areas of the trigone and bladder neck. In fact, in patients with urinary outflow obstruction (i.e., prostatic hyperplasia), the bladder neck may contain muscularis propria directly beneath the mucosa, with the lamina propria being virtually indiscernible (Figure 35.5B). Lamina propria is also absent beneath the urothelium lining the renal papillae in the renal pelvis and is quite thin along the minor calyces (Figure 35.20).
In the midportion of the lamina propria of the bladder lie intermediate-sized arteries and veins. Wisps of smooth muscle are commonly found in the lamina propria and usually are associated with these vessels (61,62) (Figures 35.21A and 35.21B). These fascicles of smooth muscle are not connected to the muscularis propria; they appear as isolated bundles but may form a discontinuous thin layer of muscle. The anatomic relationship of these fibers to the overlying urothelium can be severely disrupted by inflammation or prior therapeutic intervention when they may be seen juxtaposed to the basement membrane (Figure 35.21C). Uncommonly, these muscle fibers may present as a continuous layer of muscle within the lamina propria, thus forming a true muscularis mucosae (62). In evaluating surgical and
P.918
biopsy material, every effort should be made to distinguish these superficial muscle fascicles from muscularis propria since a failure to do so will lead to errors in tumor staging and treatment. A pathologist should not sign out a biopsy as transitional cell carcinoma invading muscle because he/she is not giving useful information as to the depth of invasion. In fact, many urologists are unaware of the existence of a superficial muscle layer (muscularis mucosae) so that the above diagnosis will lead the urologist to treat the patient as a deeply invasive tumor (stage pT2 or greater) when in fact the patient has superficially invasive disease (stage pT1). Occasionally one may encounter fat within the
P.919
lamina propria and muscularis (63) (Figure 35.22). At this time, it is unclear whether this is due to the patient's body habitus, but its presence should not be misinterpreted by pathologists as evidence of perivesical fat.
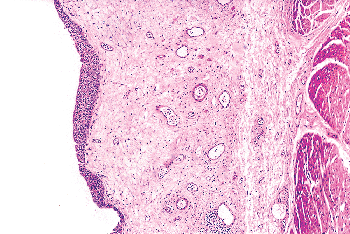 |
Figure 35.18 Lamina propria. It is composed of connective tissue, vascular structures, sensory nerves, and elastic fibers. Notice that the superficial connective tissue is denser than the deep portion. |
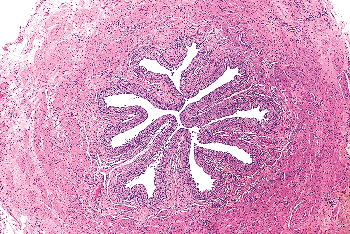 |
Figure 35.19 Cross section of mid-ureter. The elastic fibers and loose connective tissue within the lamina propria impart a festooned appearance to the urothelium. Notice that the different layers of the muscularis propria are indiscernible. |
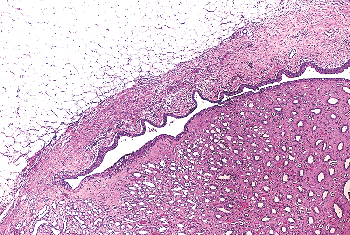 |
Figure 35.20 Junction of the renal papilla with the minor calyx. Notice the absence of the lamina propria along the papilla and a very thin lamina propria and muscularis propria along the minor calyx. |
 |
Figure 35.21 Lamina propria of the urinary bladder. A. and B. Discontinuous smooth muscle fascicles are adjacent to intermediate size vessels within the lamina propria (anti actin monoclonal antibody). C. Disorganized wisps of superficial smooth muscle lie directly beneath the urothelium at the site of a prior biopsy. (Transurethral resection biopsy [TURB] specimen.) |
 |
Figure 35.22 Mature adipose tissue within the lamina propria of the urinary bladder. |
Pathologists are surprised to learn that, in terms of prognosis and treatment, urologists and urologic oncologists lump noninvasive (Ta) and superficially invasive (T1) into a single category. It is my opinion that this is due greatly to the fact that we, as pathologists, do not agree as to what constitutes lamina propria invasion. There are many cases of pT1 disease that are unequivocal, but there are an equal number in which invasion is, at best, questionable. Pathologists' interpretation in the latter group is inconsistent and not reproducible. While this confusion is partly due to the lack of orientation of transurethral biopsy specimens and to disruption of the normal histologic architecture by tumor or prior therapy, it is clear that better parameters are needed to make this distinction.
Muscularis Propria
The muscularis propria is said to be composed of three smooth muscle coats, inner and outer longitudinal layers, and a central circular layer. In fact, these layers can only be identified consistently in the area of the bladder neck. In other areas, the longitudinal and circular layers mix freely and have no definite orientation. In the ureter, the muscularis propria is thicker distally, and the proximal portion contains only two layers (64). In the renal pelvis, the muscularis propria becomes thinner along the major and minor calyces, and no orientation of the muscle fibers is evident. No muscular fibers are evident between the urothelium and the renal medulla at the level of the renal papillae (Figures 35.7, 35.20). Within the renal sinus, the muscularis propria is surrounded by variable amounts of fat (Figures 35.6, 35.23). This fact is rarely mentioned by pathologists at the time of evaluating urothelial tumors arising in the renal pelvis. Many cases are signed out as invading renal hilar fat or invading perirenal fat, when in fact the invasion is solely into the fat within the renal sinus. The significance of this finding remains to be determined (65).
In the contracted bladder, the muscle fibers are arranged in relatively coarse bundles that are separated from each other by moderate to abundant connective tissue containing blood vessels, lymphatics, and nerves. Mature adipose tissue may also be present. Very infrequently, one may see nests of paraganglia, usually associated with neural or vascular structures (Figure 35.24A). The cells are arranged in discrete nests or cords and have clear or granular cytoplasm with round or vesicular nuclei. They should not be confused with invasive carcinoma. Immunohistochemical stains for cytokeratins are negative but for chromogranin are positive (Figure 35.24B).
Similar to other layers, the thickness of the muscularis propria will vary from patient to patient, with age, and with the degree of distention (Figures 35.25A B). In fact, Jequier and Rousseau (66) performed sonographic measurements of the bladder wall thickness in 410 urologically normal children and 10 adults. They found that the bladder wall thickness varied mostly with the state of bladder filling and only minimally with age and gender. The bladder wall had a mean thickness of 2.76 mm when empty and 1.55 mm when distended.
For staging purposes, the muscularis propria has been divided into two segments, superficial and deep (T2a and T2b, respectively) (Figure 35.8). No anatomical landmarks can be used to make this distinction so it must be done by direct visualization on the light microscope. Prior transurethral resection will alter the anatomy of the site and mask normal landmarks, making proper staging difficult if not impossible.
Bladder diverticula are relatively common, yet their etiology remains controversial. Most investigators agree that they occur secondary to increased intravesical pressure as a
P.920
result of obstruction distal to the diverticulum (67,68,69). The obstruction brings about compensatory muscle hypertrophy and eventual mucosal herniation in areas of weakness. Others feel that at least some diverticula are a consequence of congenital defects in the bladder musculature, citing as evidence cases of diverticula in young patients without evidence of obstruction (69,70). The most common sites of diverticula are (a) adjacent to the ureteral orifices, (b) the bladder dome (probably related to an urachal remnant), and (c) the region of the internal urethral orifice. Grossly, one sees distortion of the external surface of the bladder. The diverticula may be widely patent but are usually narrow in symptomatic patients. The mucosa adjoining the diverticulum is usually hyperemic or ulcerated. There may be epithelial hyperplasia and hypertrophy of the muscularis propria. Very commonly, there is inflammation involving the lamina propria and muscularis. The wall of the diverticulum itself consists of urothelium and underlying connective tissue, similar to the bladder mucosa with lamina propria (Figure 35.26). Few, if any, muscle fascicles of the muscularis propia will be identified in the majority of cases of acquired diverticula, although muscularis mucosae may be present. The true congenital diverticulum contains a thinned outer muscle layer. Infrequently, the epithelium lining the sac will undergo squamous or glandular metaplasia due to local irritation associated with urine stasis, infection, or stone. In these cases, it is not unusual for the diverticular wall to become extensively fibrotic.
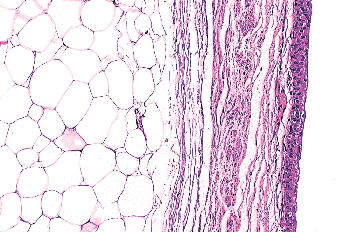 |
Figure 35.23 Urothelial wall along the minor calices. Thin layers of lamina propria and muscularis propria are surrounded by fat within the renal sinus. |
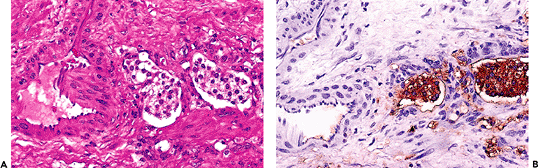 |
Figure 35.24 Nests of paraganglia within the bladder wall. A. The cells are small, have vesicular nuclei and clear cytoplasm, and are seen adjacent to neural or vascular structures. They should not be confused with invasive carcinoma. B. Immunostain for chromogranin A can clarify the issue. |
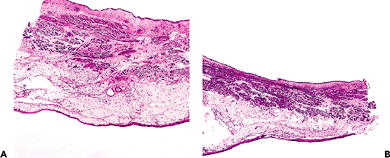 |
Figure 35.25 Full thickness section of the bladder. A. Notice the irregular thickness of the lamina propria. The three layers of muscle comprising the muscularis propria cannot be clearly defined. In contradistinction to the muscularis propria of the gut, there are ample amounts of soft tissue between muscle bundles in the bladder. B. Cross section of distended bladder. The overall thickness of the viscus is diminished as compared to the contracted bladder. Both the lamina propria and muscularis propria become more compact. |
P.921
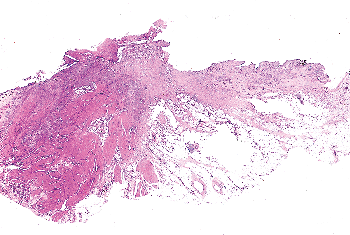 |
Figure 35.26 Bladder diverticulum. To the left is inflamed but anatomically normal bladder wall, while in the center and to the right one sees total absence of the muscularis propria. Perivesical soft tissue comes in contact with the inflamed, fibrotic, and thickened lamina propria. |
Major complications of bladder diverticula include infection, lithiasis, and carcinoma. It is believed that 2 to 7% of patients with bladder diverticula will develop an associated neoplasm, presumed secondary to the chronic inflammatory stimuli mentioned above (71,72). Ureteral diverticula are rare and are asymptomatic if uncomplicated (73). They are not seen in the renal pelvis.
References
1. Moore K. The urinary system. In: Moore K, ed. The Developing Human: Clinically Oriented Embryology. Philadelphia: WB Saunders; 1982.
2. Kissane JM. Development and structure of the urogenital system. In: Murphy WM, ed. Urological Pathology. Philadelphia: WB Saunders: 1989;1 33.
3. Eble JN, Sauter G, Epstein JI, Sesterhenn IA, eds. World Health Organization Classification of Tumors: Pathology and Genetics of Tumors of the Urinary System and Male Genital Organs. Lyon, France: IARC Press; 2004.
4. Clement PB, Young RH. Endocervicosis of the urinary bladder. A report of six cases of a benign mullerian lesion that may mimic adenocarcinoma. Am J Surg Pathol 1992;16:533 542.
5. Young RH, Clement PB. Mullerianosis of the urinary bladder. Mod Pathol 1996;9:731 737.
6. Comiter CV. Endometriosis of the urinary tract. Urol Clin North Am 2002;29:625 635.
7. Chapron C, Boucher E, Fauconnier A, Vieira M, Dubuisson JB, Vacher-Lavenu MC. Anatomopathological lesions of bladder endometriosis are heterogeneous. Fertil Steril 2002;78:740 742.
8. Moore KL. The pelvis and perineum. In: Moore KL, ed. Clinically Oriented Anatomy. 2nd ed. Baltimore: Williams & Wilkins; 1985.
9. Tanagho E. Anatomy of the lower urinary tract. In: Campbell MF and Walsh PC, eds. Campbell's Urology. 6th ed. Philadelphia: WB Saunders; 1992.
10. Mahadevia PS, Koss LG, Tar IJ. Prostatic involvement in bladder cancer. Prostate mapping in 20 cystoprostatectomy specimens. Cancer 1986;58:2096 2102.
11. Utz DC, Farrow GM, Rife CC, Segura JW, Zincke H. Carcinoma in situ of the bladder. Cancer 1980;45(suppl 7):1842 1848.
12. Ro JY, Ayala AG, el-Naggar A, Wishnow KI. Seminal vesicle involvement by in situ and invasive transitional cell carcinoma of the bladder. Am J Surg Pathol 1987;11:951 958.
13. Tanagho EA, Smith DR, Meyers FH. The trigone: anatomical and physiological considerations. II. In relation to the bladder neck. J Urol 1968;100:633 639.
14. Tanagho EA, Meyers FH, Smith DR. The trigone: anatomical and physiological considerations. I. In relation to the ureterovesical junction. J Urol 1968;100:623 632.
15. Shehata R. A comparative study of the urinary bladder and the intramural portion of the ureter. Acta Anat (Basel) 1977;98:380 395.
16. Politano VA. Ureterovesical junction. J Urol 1972;107:239 242.
17. Elbadawi A. Anatomy and function of the ureteral sheath. J Urol 1972;107:224 229.
18. Tanagho EA, Smith DR. The anatomy and function of the bladder neck. Br J Urol 1966;38:54 71.
19. Moore KL, Dalley AF. Clinically Oriented Anatomy. 5th ed. Baltimore: Lippincott Williams & Wilkins; 2005.
20. Weiss L. Cell and Tissue Biology: A Textbook of Histology. 6th ed. Baltimore: Urban & Schwarzenberg; 1988.
21. Fletcher TF, Bradley WE. Neuroanatomy of the bladder-urethra. J Urol 1978;119:153 160.
22. Olson CA. Anatomy of the upper urinary tract. In: Campbell MF and Walsh PC, eds. Campbell's Urology. 5th ed. Philadelphia: WB Saunders; 1986.
23. Hanna MK, Jeffs RD, Sturgess JM, Barkin M. Ureteral structure and ultrastructure. Part I. The normal human ureter. J Urol 1976;116:718 724.
24. Kaye KW, Goldberg ME. Applied anatomy of the kidney and ureter. Urol Clin North Am 1982;9:3 13.
25. Motola JA, Shahon RS, Smith AD. Anatomy of the ureter. Urol Clin North Am 1988;15:295 299.
26. Notley RG. Ureteral morphology: anatomic and clinical considerations. Urology 1978;12:8 14.
27. Crelin ES. Normal and abnormal development of ureter. Urology 1978;12:2 7.
28. Koss LG. Tumors of the urinary bladder. In: Atlas of Tumor Pathology. 2nd series, fascicle 11. Washington, DC: Armed Forces Institute of Pathology; 1975.
29. Fawcett DW. Bloom and Fawcett: A Textbook of Histology. 11th ed. Philadelphia: WB Saunders; 1986.
30. Hicks RM. The function of the golgi complex in transitional epithelium. Synthesis of the thick cell membrane. J Cell Biol 1966;30:623 643.
31. Battifora H, Eisenstein R, McDonald JH. The human urinary bladder mucosa. An electron microscopic study. Invest Urol 1964;12:354 361.
32. Fawcett DW, Raviola E, eds. Bloom and Fawcett: A Textbook of Histology. 12th ed. New York: Chapman & Hall; 1994.
33. Koss LG. The asymmetric unit membranes of the epithelium of the urinary bladder of the rat. An electron microscopic study of a mechanism of epithelial maturation and function. Lab Invest 1969;21:154 168.
34. Newman J, Antonakopoulos GN. The fine structure of the human fetal urinary bladder. Development and maturation. A light, transmission and scanning electron microscopic study. J Anat 1989;166:135 150.
35. Alroy J, Gould VE. Epithelial-stromal interface in normal and neoplastic human bladder epithelium. Ultrastruct Pathol 1980; 1:201 210.
36. Koss LG. Mapping of the urinary bladder: its impact on the concepts of bladder cancer. Hum Pathol 1979;10:533 548.
37. Mostofi FK. Potentialities of bladder epithelium. J Urol 1954;71:705 714.
38. Morse HD. The etiology and pathology of pyelitis cystica, ureteritis cystica and cystitis cystica. Am J Pathol 1928;4:33 50.
39. Goldstein AM, Fauer RB, Chinn M, Kaempf MJ. New concepts on formation of Brunn's nests and cysts in urinary tract mucosa. Urology 1978;11:513 517.
40. Wiener DP, Koss LG, Sablay B, Freed SZ. The prevalence and significance of Brunn's nests, cystitis cystica and squamous metaplasia in normal bladders. J Urol 1979;122:317 321.
41. Edwards PD, Hurm RA, Jaeschke WH. Conversion of cystitis glandularis to adenocarcinoma. J Urol 1972;108:568 570.
P.922
42. Lin JI, Yong HS, Tseng CH, Marsidi PS, Choy C, Pilloff B. Diffuse cystitis glandularis. Associated with adenocarcinomatous change. Urology 1980;15:411 415.
43. Tannenbaum M. Inflammatory proliferative lesion of urinary bladder: squamous metaplasia. Urology 1976;7:428 429.
44. Engel RM, Wilkinson HA. Bladder exstrophy. J Urol 1970;104:699 704.
45. Nielsen K, Nielsen KK. Adenocarcinoma in exstrophy of the bladder the last case in Scandinavia? A case report and review of literature. J Urol 1983;130:1180 1182.
46. Bhagavan BS, Tiamson EM, Wenk RE, Berger BW, Hamamoto G, Eggleston JC. Nephrogenic adenoma of the urinary bladder and urethra. Hum Pathol 1981;12:907 916.
47. Navarre RJ Jr, Loening SA, Platz C, Narayana A, Culp DA. Nephrogenic adenoma: a report of 9 cases and review of the literature. J Urol 1982;127:775 779.
48. Molland EA, Trott PA, Paris AM, Blandy JP. Nephrogenic adenoma: a form of adenomatous metaplasia of the bladder. A clinical and electron microscopical study. Br J Urol 1976;48:453 462.
49. Ford TF, Watson GM, Cameron KM. Adenomatous metaplasia (nephrogenic adenoma) of urothelium. An analysis of 70 cases. Br J Urol 1985;57:427 433.
50. Satodate R, Koike H, Sasou S, Ohori T, Nagane Y. Nephrogenic adenoma of the ureter. J Urol 1984;131:332 334.
51. Mazal PR, Schaufler R, Altenhuber-Muller R, et al. Derivation of nephrogenic adenomas from renal tubular cells in kidney-transplant recipients. N Engl J Med 2002;347:653 659.
52. DeMeester LJ, Farrow GM, Utz DC. Inverted papillomas of the urinary bladder. Cancer 1975;36:505 513.
53. Henderson DW, Allen PW, Bourne AJ. Inverted urinary papilloma: report of five cases and review of the literature. Virchows Arch A Pathol Anat Histol 1975;366:177 186.
54. Caro DJ, Tessler A. Inverted papilloma of the bladder: a distinct urological lesion. Cancer 1978;42:708 713.
55. Anderstrom C, Johansson S, Pettersson S. Inverted papilloma of the urinary tract. J Urol 1982;127:1132 1134.
56. Lazarevic B, Garret R. Inverted papilloma and papillary transitional cell carcinoma of urinary bladder: report of four cases of inverted papilloma, one showing papillary malignant transformation and review of the literature. Cancer 1978;42:1904 1911.
57. Whitesel JA. Inverted papilloma of the urinary tract: malignant potential. J Urol 1982;127:539 540.
58. Stein BS, Rosen S, Kendall AR. The association of inverted papilloma and transitional cell carcinoma of the urothelium. J Urol 1984;131:751 752.
59. Assor D. Inverted papilloma of the renal pelvis. J Urol 1976;116:654.
60. Lausten GS, Anagnostaki L, Thomsen OF. Inverted papilloma of the upper urinary tract. Eur Urol 1984;10:67 70.
61. Dixon JS, Gosling JA. Histology and fine structure of the muscularis mucosae of the human urinary bladder. J Anat 1983;136(pt 2):265 271.
62. Ro JY, Ayala AG, el-Naggar A. Muscularis mucosa of urinary bladder. Importance for staging and treatment. Am J Surg Pathol 1987;11:668 673.
63. Philip AT, Amin MB, Tamboli P, Lee TJ, Hill CE, Ro JY. Intravesical adipose tissue: a quantitative study of its presence and location with implications for therapy and prognosis. Am J Surg Pathol 2000;24:1286 1290.
64. Notley RG. The musculature of the human ureter. Br J Urol 1970;42:724 727.
65. Olgac S, Mazumdar M, Dalbagni G, Reuter VE. Urothelial carcinoma of the renal pelvis: a clinicopathologic study of 130 cases. Am J Surg Pathol 2004;28:1545 1552.
66. Jequier S, Rousseau O. Sonographic measurements of the normal bladder wall in children. AJR Am J Roentgenol 1987;149:563 566.
67. Miller A. The aetiology and treatment of diverticulum of the bladder. Br J Urol 1958;30:43 56.
68. Kertsschmer HL. Diverticula of the urinary bladder: a clinical study of 236 cases. Surg Gynecol Obstet 1940;71:491 503.
69. Fox M, Power RF, Bruce AW. Diverticulum of the bladder: presentation and evaluation of treatment of 115 cases. Br J Urol 1962;34:286 298.
70. Barrett DM, Malek RS, Kelalis PP. Observations on vesical diverticulum in childhood. J Urol 1976;116:234 236.
71. Abeshouse BS. Primary carcinoma in a diverticulum of the bladder: a report of four cases and a review of the literature. J Urol 1943;49:534 547.
72. Faysal MH, Freiha FS. Primary neoplasm in vesical diverticula. A report of 12 cases. Br J Urol 1981;53:141 143.
73. Cochran ST, Waisman J, Barbaric ZL. Radiographic and microscopic findings in multiple ureteral diverticula. Radiology 1980;137:631 636.