10. The Musculoskeletal System
Authors: Corwin, Elizabeth J.
Title: Handbook of Pathophysiology, 3rd Edition
Copyright 2008 Lippincott Williams & Wilkins
> Table of Contents > Unit III - Integrated Control and Dysfunction > Chapter 10 - The Musculoskeletal System
Chapter 10
The Musculoskeletal System
Skeletal muscles and bones support and move the body. Bones protect the internal organs and are moved by the muscles. Muscles are responsible for vascular tone, gut contractions, genitourinary function, and the beating of the heart. Some muscles function relatively independently of neural or hormonal stimulation, whereas other muscles are active only in response to neural stimulation. Diseases or injuries to the muscles and bones make movements difficult or painful. Life is impossible if the cardiac or respiratory muscles are destroyed.
Physiologic Concepts
There are three types of muscles: skeletal, cardiac, and smooth. The basic processes of contraction are similar in all three types, but important differences exist. Although the focus of this chapter is the skeletal-muscular system, the unique characteristics of the cardiac and smooth muscles will be presented briefly.
Skeletal Muscle
Skeletal muscles are connected to bones through tendons. Tendons move the bones by contraction of the skeletal muscles, which is controlled by lower motor neurons from the spinal cord. One motor neuron may innervate several muscle fibers. A motor neuron and all the muscle fibers it innervates are called a motor unit. In general, the muscles over which we have fine control have only a few muscle fibers innervated by a single motor
P.281
neuron. Muscles that do not need fine control (i.e., the support muscles of the back) are composed of many muscle fibers per motor neuron.
Skeletal muscle cells are highly differentiated cells whose growth during embryogenesis and later in life is under the control of growth factors, hormones, and physical stimuli. During embryogenesis, skeletal muscle cells undergo both hyperplasia (increase in cell number) and hypertrophy (increase in cell size). After embryogenesis, skeletal muscle cells continue to undergo hypertrophy in response to certain stimuli, including exercise, but no longer undergo hyperplasia. The protein myostatin, also known as growth and differentiating factor-8 , has been identified as playing a key role in the regulation of skeletal muscle growth before and after birth, by limiting the growth and reproduction of muscle cell fibers. In animals that lack the gene coding for myostatin, muscle hypertrophy and hyperplasia occur both before and after birth, resulting in increased muscle mass and strength.
Skeletal Muscle Structure
Each skeletal muscle is made up of many muscle cells, called muscle fibers. A given muscle may have a few hundred or several thousand fibers. The more muscle fibers present in a muscle, the greater the potential strength of that muscle.
Skeletal muscle is called striated muscle because of the banding that can be seen throughout the muscle with a light microscope. The striations reflect the subunits of each muscle fiber: the myofibrils. A single muscle cell is made up of many myofibrils. The myofibrils are composed of smaller subunits called myofilaments; myofilaments are the functional units of the muscle cell. They are composed of thick and thin contractile proteins, grouped together into a repeating pattern, called a sarcomere.
The Sarcomere
Three sarcomeres are aligned together in Figure 10-1. Each sarcomere contains thick and thin filaments. The thick filaments are located in the central region of the sarcomere, and are composed of several hundred copies of the contractile protein myosin. The thin filaments are attached to the edges of the sarcomere and are composed of the proteins actin, tropomyosin, and troponin.
The area of the sarcomere where only thick filaments are present is called the H zone. The area where only thin filaments are present is termed the I zone. The A band is the section where the thin and thick filaments overlap. The Z lines are the borders of the sarcomere, where the actins attach. Each sarcomere spans from one Z line to the next.
Cross-Bridges
Each myosin molecule is composed of six peptide chains: two heavy chains that twist together to form a long tail with two globular heads, and four light chains that group, two to a head, with the myosin heads. The heads form small projections that extend from the myosin filament. These projections
P.282
are called cross-bridges. When a muscle is relaxed, the myosin cross-bridges are unattached in the sarcomere. During this relaxed state, an adenosine triphosphate (ATP) molecule binds to myosin and is split into ADP and a high-energy phosphate (P). The ADP and P remain bound to the myosin, without releasing the energy generated by the splitting of ATP.
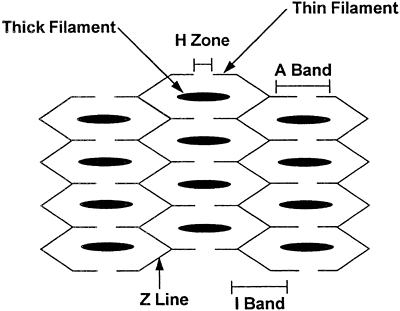 |
Figure 10-1. The sarcomere. |
Muscle Contraction
Contraction of a muscle occurs when the myosin cross-bridges bind to specific sites on the actin proteins. When this occurs, energy that has been stored in the myosin head from the previous splitting of an ATP molecule is released. The released energy is used to swing the cross-bridges, causing the actin and myosin filaments to slide over each other. This shortens and contracts the muscle. With cross-bridge swinging, the remaining ADP and P release from myosin.
During muscle contraction, the lengths of the actin and myosin filaments do not change, but the I band and the H zone shorten. The different regions of the sarcomere are described in Table 10-1; the thick filaments, myosin heads, and thin filaments are shown in Figure 10-2. Each muscle contraction involves several repeated cycles of filament sliding to provide the tension necessary for the muscle to do work.
Excitation-Contraction Coupling
Given that myosin and actin are ready to bind, and energy is available to be released to swing the cross-bridges, the question becomes: What prevents contraction of the skeletal muscle from happening all the time? The simple answer is that myosin and actin cannot always bind to each other, so the cross-bridges cannot always swing. Skeletal muscle contraction only
P.283
occurs in response to neural stimulation and the subsequent release of intracellular calcium.
Table 10-1. Sarcomere Composition and Changes During Contraction | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
When an action potential is delivered by a motor neuron to a skeletal muscle fiber, the neuron releases acetylcholine (ACh) into the neuromuscular junction. ACh diffuses to a specialized area of the muscle cell, called the end plate. Muscle cell end plates are concentrated with receptors for ACh. ACh binds to the receptors, causing the opening of sodium channels present in the muscle cell. With the opening of these channels, sodium ions rush into the cell, depolarizing it (making the cell positively charged on the inside) and initiating an action potential. The action potential passes along the entire muscle fiber, depolarizing the fiber. Depolarization
P.284
spreads into the fiber through small tubules, called transverse (T) tubules, which run along the juncture between the A and I bands. When the inside of the cell becomes positive, calcium ion is released from intracellular bags of calcium (called lateral sacs) that lie adjacent to the T tubules. The lateral sacs are outpouchings of a large intracellular calcium storage compartment: the sarcoplasmic reticulum (SR). High levels of intracellular calcium released from the SR initiate muscle contraction.
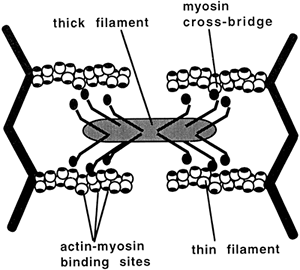 |
Figure 10-2. Diagrammatic representation of myosin molecules lined up in thick filaments with globular heads extending in to form cross-bridges with overlying and underlying actin molecules. |
The Role of Intracellular Calcium in Initiating Muscle Contraction
When a skeletal muscle fiber is at rest, the myosin heads are prevented from binding to the actin molecules by the presence of the other two proteins of the thin filaments: tropomyosin and troponin. Without myosin binding to actin, energy from ATP cannot be released, cross-bridges cannot swing, and the muscle cannot contract. Elevated intracellular calcium changes the interaction of these proteins and causes contraction.
At rest, tropomyosin is attached to the actin molecules in such a way that it blocks the sites on actin where the myosin cross-bridges would bind. Troponin attaches to both the actin and the tropomyosin molecules. It also has a binding site for calcium. When calcium concentration inside the cell increases, calcium binds to troponin, causing troponin to shift its position on the tropomyosin molecule. This causes tropomyosin to shift its position on actin, uncovering the binding site for myosin. Once the binding site on actin is uncovered, the myosin heads immediately bind actin and release their stored energy, and the cross-bridges swing. The filaments slide past each other and the muscle contracts. The greater the number of cross-bridges connected and swinging at one time, the greater the tension produced by the muscle. Excitation-contraction coupling and the role of calcium are outlined in Figure 10-3.
After each cross-bridge swinging, a new ATP molecule binds to the myosin molecule (the old ADP and P have already been released). This causes the myosin cross-bridges to separate from actin and the fiber to relax. Once relaxed, the new ATP molecule is split, and its energy is again stored in the myosin head. If calcium is still high intracellularly, the myosin cross-bridge will again bind actin, and this energy will be released, leading to a second contraction. Excitation-contraction coupling occurs when intracellular calcium levels increase from a resting molar concentration of less than 10-7 to approximately 10-5. During a typical action potential, calcium concentration is approximately 2 10-4 molar; this is approximately 10 times the level required to maximally contract the muscle.
Muscle Fiber Summation
Each calcium pulse lasts approximately 1/20th of a second and produces what is called a single muscle twitch. Summation occurs if calcium is maintained in the intracellular compartment by repeated neural stimulation of the muscle. Summation means individual twitches are added together, causing increased contraction strength. If stimulation is prolonged, the individual
P.285
twitches blend together until the strength of contraction is at a maximum. At this point, the muscle is said to have reached tetany, which is characterized by a smooth, continued contraction. Summation and tetany in an individual muscle fiber are shown in Figure 10-4.
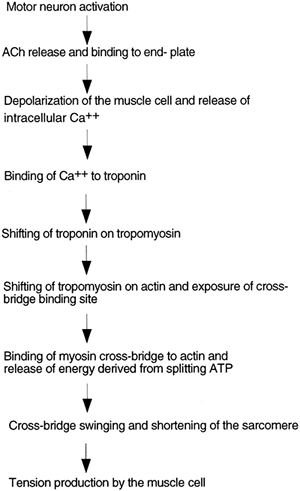 |
Figure 10-3. Flow diagram showing excitation-contraction coupling in a muscle cell, initiated by release of ACh from a motor neuron. |
Whole-Muscle Summation: Multiple-Fiber Summation
The total amount of tension produced by an entire muscle is the result of the summation of the tension produced by each muscle fiber. An increase in the number of fibers stimulated to contract will increase the amount of tension produced by the entire muscle. This is called multiple-fiber summation. Multiple-fiber summation occurs when additional motor units are activated, leading to the contraction of more muscle fibers.
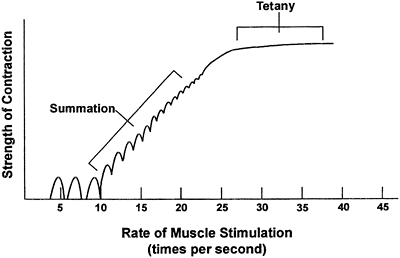 |
Figure 10-4. Summation and tetany. |
P.286
Relaxation of the Muscle
Muscle fibers relax when calcium is pumped out of the cytoplasm back into the sarcoplasmic reticulum. Calcium pumping is an active process occurring in the membrane of the sarcoplasmic reticulum. This process uses energy derived from splitting a different ATP molecule. When calcium levels decrease to approximately 10-7 molar, troponin returns to its original position on the tropomyosin molecule, and tropomyosin again inhibits the binding of actin and myosin, which causes muscle contraction to stop.
Muscle Metabolism and Muscle Fatigue
Muscle contraction depends on the production of ATP from one of three sources: (1) creatinine phosphate (CP) stored in the muscle, (2) oxidative phosphorylation of foodstuffs stored in or delivered to the muscle, and (3) anaerobic glycolysis. Muscle fatigue results when the use of ATP in a muscle becomes excessive.
When a muscle first starts contracting, it begins to use its stores of CP to drive contraction. CP contains a high-energy phosphate molecule that it transfers to ADP to produce ATP:
CP + ADP = C + ATP.
This source of ATP is rapidly accessed, but is limited by the amount of CP present in the cell at the start of contraction. After several seconds, the muscle begins to rely mostly on oxidative phosphorylation. Sources of fuel for oxidative phosphorylation include glycogen stored in the muscle and, later, glucose and fatty acids delivered to the muscle in the blood supply. This source of energy is available for 30 minutes or so, depending on the intensity of contraction. If the intensity of exercise is very high, or the duration is very long, the muscle begins to rely increasingly on anaerobic glycolysis. Anaerobic glycolysis produces a limited amount of ATP from the
P.287
metabolism of muscle glycogen and circulating blood glucose. A muscle using anaerobic glycolysis for a large part of its ATP production rapidly fatigues. Muscle fatigue can be predicted experimentally by depletion of glycogen stored in the muscle. Lactic acid is a by-product of anaerobic glycolysis and may accumulate in the muscle and blood with intense or prolonged muscle contraction, contributing to fatigue. Lactic acid also may contribute to the muscle pain felt a day or two after intense exercise.
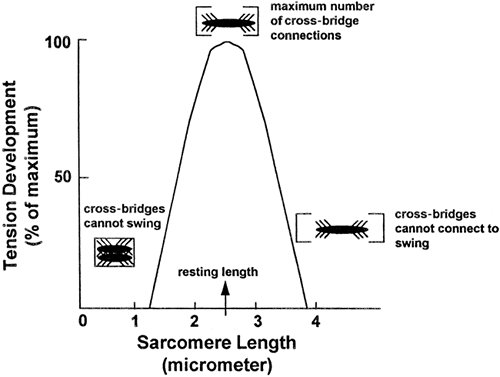 |
Figure 10-5. Length-tension curve. |
Length-Tension Relationship
The resting length of a muscle fiber determines the maximum amount of tension it can produce. This relationship is shown in Figure 10-5. Muscle fiber length affects tension production as a result of stretching of the sarcomere. If a sarcomere is stretched beyond an optimum length, as shown on the right side of the figure, some myosin cross-bridges will be too far away to connect with the actin sites and therefore will not swing. This will reduce total tension. In contrast, if the sarcomere is less than optimally stretched, as shown on the left side of the figure, the cross-bridges will be bunched too closely together to be able to swing freely, thus limiting filament sliding and again reducing total tension. In a normal skeletal muscle, muscle length at rest will produce the maximum amount of tension.
Isometric Contraction
Isometric contractions are those in which cross-bridge swinging occurs and tension is produced without shortening of the muscle. An isometric
P.288
contraction occurs when an individual is trying to lift a load that requires greater tension than can be produced by the muscle. No mechanical work is performed. Tension is produced, but the muscle does not shorten.
Isotonic Contraction
Isotonic contractions occur when a muscle shortens against a constant load. Work is done to lift the load. An example of an isotonic contraction is when a weightlifter lifts a barbell. Most muscle contractions include both isotonic and isometric periods.
Series Elastic Elements
There is typically a delay between excitation of a muscle and an isotonic contraction. A delay occurs because the elastic components of the muscle, including the tendons and the attachments of the sarcomeres, must be shortened before the muscle itself shortens. The elastic components of a muscle are called the series elastic elements. If a second contraction of the muscle occurs before the series elastic elements relax, there is no delay, and muscle tension can be increased immediately. This concept of the series elastic element may be easier understood by picturing a spring connected to an object. Before the object can be lifted by pulling the spring, the spring must first be stretched. This delays lifting of the object.
Fast-Twitch and Slow-Twitch Fibers
Different muscles contain different types of muscle fibers, depending on the range of jobs performed. Muscles that must function continually, such as those of the respiratory system, must have long endurance and an ample supply of oxygen. Others function briefly and intensely and then relax; these muscles must be able to produce short bursts of high energy. Usually, a muscle will contain a mixture of fiber types, with one fiber type predominating but not exclusively. Two main divisions of muscle fibers are fast-twitch and slow-twitch fibers.
Fast-twitch fibers release calcium rapidly from the sarcoplasmic reticulum, and rapidly split ATP to ADP on the myosin head. This causes the rate of cross-bridge swinging to be fast. Fast-twitch fibers may depend primarily on either oxidative phosphorylation or anaerobic glycolysis for energy, depending on what type of work they typically perform.
Fast-twitch fibers that frequently produce large amounts of energy for quick bursts of tension have large stores of glycolytic enzymes and produce a great deal of their ATP from anaerobic glycolysis. These are usually large fibers. They require less vascularization because they rely less on oxidative phosphorylation; therefore, they appear white. These fibers are called fast-glycolytic fibers. Fast-glycolytic fibers tire rapidly and predominate in the muscles of weightlifters and sprinters.
Fast-twitch fibers that rely mostly on oxidative phosphorylation, called fast-oxidative fibers, are well vascularized and contain elevated stores of
P.289
the muscle protein myoglobin. Myoglobin combines in the muscle with oxygen, serving as an oxygen storage bank. Fast-oxidative fibers tire less rapidly and predominate in muscles of longer distance runners.
Slow-twitch fibers are small, highly vascularized fibers that depend predominantly on oxidative phosphorylation for the production of ATP. Muscles with slow-twitch fibers look red because of their high vascularity and the presence of the protein myoglobin. Slow-oxidative fibers have long endurance and predominate in muscles required to produce tension for prolonged periods, such as back muscles.
Stretch Reflex
Many skeletal muscles contain special muscle fibers that act as stretch receptors, called muscle spindle fibers. Muscle spindle fibers are fibers wrapped by afferent nerve endings, which increase their rate of firing when the muscle is stretched. The impulses are transmitted to the spinal cord by an afferent neuron. In the spinal cord, the afferent neuron synapses directly on the motor neuron supplying the muscle (a monosynaptic reflex) or on an interneuron, which then stimulates the motor neuron (a multisynaptic reflex). Activation of the motor neuron causes the muscle to contract, thus removing the stretch on the muscle spindles and returning the nerves' firing rate to normal. This process is called the stretch reflex. The opposite occurs if stretch on the spindles is suddenly reduced (called the negative stretch reflex). The result of either type of stretch reflex is maintenance of the muscle at a resting length. Voluntary muscle movement involves simultaneous contraction of regular muscle fibers and muscle spindle fibers. This contraction allows movements to be fluid. The afferent neurons that innervate the muscle spindle fibers are called gamma neurons. The monosynaptic stretch reflex that results in the knee jerk response is shown in Figure 10-6.
Cardiac Muscle
Cardiac muscle contraction is similar to skeletal muscle contraction, with the following differences:
Cardiac cells are capable of spontaneous contraction; that is, contraction without neural stimulation. Neural stimulation can increase or decrease the rate of cardiac contraction.
Cardiac muscle fibers are connected to each other through areas of low resistance, called intercalated disks. Intercalated disks allow depolarization, beginning in one cardiac muscle fiber, to pass rapidly to neighboring fibers, ensuring simultaneous contraction of all cardiac muscle fibers at one time. Simultaneous contraction is required for the maintenance of cardiac output and blood pressure.
There are two sources of calcium involved in producing a cardiac muscle cell contraction. In cardiac muscle, as in skeletal muscle, calcium ions are released intracellularly from the sarcoplasmic reticulum, but they
P.290
also enter the cell from the extracellular fluid through sodium-calcium channels present in the T tubules. These channels also are voltage sensitive but are slow to open and thus prolong the duration of the cardiac action potential. The strength of cardiac contraction, therefore, is highly dependent on extracellular calcium level. In contrast, skeletal muscle contraction does not depend on extracellular calcium.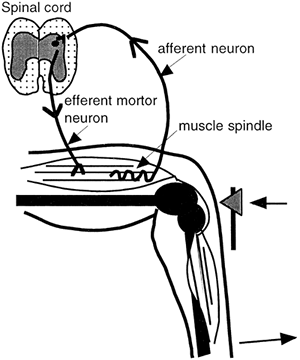
Figure 10-6. The knee-jerk reflex occurs when a muscle spindle fiber is stretched artificially. This sends a signal to the spinal cord leading to rapid contraction of the muscle.
Because of the slow calcium channels, cardiac muscle cell contraction lasts approximately 10 times as long as skeletal muscle contraction. As a result, cardiac muscle is unable to fire action potentials rapidly and does not undergo summation or tetany. If the cardiac muscle were to achieve a state of maintained contraction, the heart would be unable to fill with blood.
At rest, cardiac muscle cells are stretched less than is required to produce maximum tension, which allows the heart to increase tension when it is stretched during times of increased filling (e.g., during exercise).
Smooth Muscle
Smooth muscle contraction and skeletal muscle contraction have some similarities and some important differences. Smooth muscle contraction is not the same in all smooth muscles. Some characteristics of smooth muscle contraction include the following:
Smooth muscle is innervated and stimulated by the sympathetic and parasympathetic nerves of the autonomic nervous system. These nerves
P.291
do not innervate the smooth muscle at specific end plates, but branch over the muscle cells and diffusely release transmitter substances onto the fibers.Some smooth muscles function as a unit composed of millions of fibers. These fibers contract in response to action potentials produced from mechanical stretch, local chemical mediator release, or neural or hormonal stimulation. Spontaneous firing of action potentials can also occur. In this type of smooth muscle, action potentials generated from any source pass from one cell to another across gap junctions. This type of smooth muscle is called single-unit smooth muscle. It is found in the gut, throughout the genitourinary tract, and in many blood vessels.
Some smooth muscle fibers contract individually and only in response to neural stimulation. These fibers are usually innervated by one neuron that releases ACh or norepinephrine. These fibers depolarize and contract, but usually do not fire action potentials. This type of smooth muscle is called multi-unit smooth muscle. It is found in the muscles of the eyes and in the muscles that surround hair follicles. When contracted, these muscles cause the hair to stand up on the skin.
Although smooth muscle contains actin and myosin and splits ATP to produce tension, the thin filaments in smooth muscle fibers do not contain troponin. When intracellular calcium levels increase in smooth muscle fibers, calcium binds to a protein called calmodulin, resulting in phosphorylation of one of the light chains of the myosin heads. Phosphorylation of the light chain allows the myosin head to bind to actin and split ATP.
The sarcomeres of smooth muscle do not show striations under the microscope, but are more diffuse and less regular in pattern, allowing the muscle to contract over a wide range of lengths. There are many more actin molecules than myosin molecules in smooth muscle fibers, although maximal tension production is similar.
In smooth muscle, most calcium enters from the extracellular fluid through voltage-sensitive calcium channels. Some calcium is released from the sarcoplasmic reticulum. In some smooth muscles, intracellular calcium levels always are sufficient to maintain a low level of cross-bridge connection. This results in a resting muscle tone in these muscles.
The speed of cross-bridge cycling and muscle contraction is reduced in smooth muscle compared to skeletal muscle, most likely because myosin heads contain less ATPase. Therefore, it takes longer for ATP to be split, prolonging the amount of time myosin is attached to actin. A longer period of attachment results in increased production of tension. The slow speed of the calcium pumps in smooth muscle also prolongs contraction.
A latch mechanism in smooth muscle allows muscle contraction to be maintained for long periods of time at a fraction of the energy expenditure of skeletal muscle. This mechanism is probably related to the length of time myosin remains attached to actin.
P.292
Tendons
Tendons are bundles of collagen fibers that attach the muscles to the bones. Tendons transmit force generated by the contracting muscle to the bone and thereby move the bone. The collagen fibers are considered connective tissue and are produced by fibroblast cells.
Ligaments
Ligaments are strong fibrous connections between bones, usually at joints. Ligaments allow and limit joint movement.
Bones
Bone Structure
Mature bone is composed of 30% organic (living) material and 70% salt deposits. The organic material is called the matrix, and is composed of more than 90% collagen fibers and less than 10% proteoglycans (proteins plus polysaccharides). The salt deposits are primarily calcium and phosphate, with small amounts of sodium, potassium carbonate, and magnesium ions. The salts cover the matrix and are bound to the collagen fibers by the proteoglycans. The organic matrix gives bone its tensile strength (resistance to being pulled apart). The bone salts give bone its compressional strength (ability to withstand compression).
Exchangeable Calcium
Some calcium ion in bone is non-crystallized. This non-crystalline salt is considered exchangeable calcium, in that it can rapidly move between the bone, interstitial fluid, and blood.
Bone Formation
Bone formation is ongoing and can involve lengthening and thickening of the bone. The rate of bone formation changes throughout the lifespan. Bone formation is determined by hormonal stimulation, dietary factors, and the amount of stress put on a bone, and results from activities of the bone-forming cells, the osteoblasts.
Osteoblasts are found on the outer surface and on the inside of bones. Osteoblasts respond to various chemical signals to produce the organic matrix. When the organic matrix is first produced, it is called the osteoid. Within a few days, calcium salts begin to precipitate on the osteoid and the bone hardens over the next several weeks or months. Some osteoblasts remain part of the osteoid, and are called osteocytes or true bone cells. As the bone forms, osteocytes in the matrix send out projections to each other, forming a system of microscopic canals (canaliculi) in the bone.
P.293
Osteoblast activity is affected by diet, hormonal stimulation, and exercise. These factors interact and are dynamic, resulting in different rates of bone formation throughout a lifetime.
Exercise and Osteoblast Activity
Osteoblastic activity is stimulated by exercise and weight-bearing, as a result of the electrical currents produced when stress is applied to the bone. Bone fracture dramatically stimulates osteoblast activity, but the exact mechanism is unclear.
Hormonal Stimulation and Osteoblast Activity
Estrogen, testosterone, and growth hormone enhance osteoblast activity and bone growth. Bone growth is accelerated during puberty as a result of surging levels of these hormones. Estrogen and testosterone eventually cause the long bones to stop growing by stimulating closure of the epiphyseal plate (growing end of the bone). When estrogen levels decrease after menopause, osteoblastic activity is reduced. Deficiencies in growth hormone impede bone formation.
Diet and Osteoblast Activity
An adequate diet during childhood and adolescence is essential for maximal bone growth. Calcium ion deficiency during adolescence will result in bones less dense than optimum later in life. Most of the calcium present in bones in an individual's lifetime is deposited before the age of 20.
Vitamin D Control of Osteoblast Activity
Vitamin D stimulates bone calcification directly by acting on the osteoblasts, and indirectly by stimulating calcium absorption across the gut. Increased calcium absorption increases blood calcium concentration, which promotes bone calcification. Thus, vitamin D is essential in order to ensure adequate calcium absorption across the gut. Very large amounts of vitamin D, however, may increase bone breakdown in an attempt to liberate calcium in order to increase serum calcium levels. Large amounts of vitamin D without adequate calcium in the diet actually can promote bone resorption.
![]() ediatric Consideration
ediatric Consideration
Proper nutrition to maximize bone health begins very early in life, as suggested by studies in which vitamin D supplementation during infancy was associated with increased bone density years later.
Bone Breakdown
Bone breakdown, called resorption, occurs simultaneously with bone formation and is also ongoing throughout life. Bone resorption results
P.294
from the activity of cells called osteoclasts. Osteoclasts are multinucleated, large phagocytic cells derived from monocytes (white blood cells) present in the bone. Osteoclasts secrete various acids and enzymes that digest the bone and allow for its phagocytosis. Osteoclasts also secrete various cytokines that further stimulate resorption. Osteoclasts are usually present in only one small section of bone at a time, and phagocytize the bone section by section. Once they finish in one area, the osteoclasts disappear and osteoblasts arrive. The osteoblasts begin to fill in the clear section with new bone. This process allows old, weakened bone to be replaced with new, stronger bone.
Factors that control osteoclast activity include parathyroid hormone and calcitonin.
Parathyroid Hormone and Osteoclastic Activity
Osteoclast activity is primarily controlled by parathyroid hormone, which is released by the parathyroid glands located directly behind the thyroid gland. Parathyroid hormone release increases in response to decreased serum calcium levels. Parathyroid hormone increases osteoclastic activity and stimulates bone breakdown, liberating free calcium into the blood. Increased serum calcium acts in a negative feedback manner to reduce further release of parathyroid hormone. It has been hypothesized that estrogen reduces bone resorption by inhibiting the effect of parathyroid hormone on osteoclasts; the mechanism of this is unknown.
Other Effects of Parathyroid Hormone
Parathyroid hormone increases serum calcium by decreasing excretion of calcium by the kidneys. Parathyroid hormone also increases renal excretion of phosphate ion, thereby decreasing blood phosphate levels. Renal activation of vitamin D depends on parathyroid hormone.
Calcitonin and Osteoclastic Activity
Calcitonin is a hormone secreted by the thyroid gland in response to high serum calcium. Calcitonin has a weak effect on inhibiting osteoclastic activity and formation. These effects increase bone calcification, thereby reducing serum calcium levels.
Remodeling
The balance between osteoblast and osteoclast activity continually remodels, or renews, the bone. In children and teenagers, osteoblastic activity outpaces osteoclastic activity, leading to thickening and lengthening of the skeleton. Osteoblastic activity also outpaces osteoclastic activity in bones healing from fracture. In a young adult, osteoblastic activity and osteoclastic activity are typically in equilibrium, resulting in a constant total amount of bone mass. By middle age, osteoclastic activity outpaces osteoblastic activity and bone density begins to decrease. Osteoclastic activity is also accelerated in immobilized bones. By the seventh or eighth
P.295
decade of life, dominance of osteoclastic activity may cause the bones to become brittle, leading to increased fractures. Osteoclastic activities are controlled by several physical and hormonal factors.
Types of Bones
Bone is classified as long, short, flat, or irregular. Long bones are found in the extremities, whereas short bones are found in the ankles and wrists. Flat bones are found in the skull and rib cage. Irregular bones include the vertebrae, the bones of the face, and the jaw.
A long bone consist of a long, thick shaft, called the diaphysis, and two ends, called the epiphyses. Proximal to each epiphysis is the metaphysis. In between the epiphysis and the metaphysis is an area of growing cartilage, called the epiphyseal or growth plate. Long bones grow by means of the accumulation of cartilage at the epiphyseal plate. Cartilage is replaced by the osteoblasts, and the bone elongates. By the end of the teen years, the cartilage is used up, the epiphyseal plate fuses, and the bones stop growing. Growth hormone, estrogen, and testosterone stimulate growth of long bones. Estrogen, in conjunction with testosterone, stimulates fusion of the epiphyseal plates. The shaft of a long bone is hollowed out along the medullary canal, which is filled with bone marrow.
Bone Marrow
Bone marrow consists of cells involved in blood cell formation (red marrow) and fat cells (yellow marrow). Marrow is found in long and flat irregular bones. Bone marrow biopsy is performed on flat bones.
Joints
Joints are areas of the body where two bones come together. A joint may be freely movable, called a diarthrodial joint, or may be immobile, called a synarthrodial joint.
In a diarthrodial joint, the two ends of the bone are not connected directly, but come together in a fibrous joint capsule that surrounds and supports the joint. There are two layers of the joint capsule: an outer layer and an inner membrane layer called the synovium or synovial membrane. The synovial membrane secretes a slippery fluid, called synovial fluid, which lubricates the joint. The synovial membrane also covers the tendons that connect the bone to muscle, and the ligaments that connect the bones to each other. There is a well-developed vascular supply to the synovium, which may be damaged with joint trauma, leading to swelling, bruising, and pain surrounding the joint. In some joints, the synovial membrane forms a closed sac external to the joint, called a bursa. Bursae are found at areas where the bones are physically close together, or where a tendon runs over the bone. Bursae too may become inflamed, a condition called bursitis. Most joints in the body are diarthrodial joints, including the sacroiliac joint, the interphalangeal joints, the hip and knee joints, and the
P.296
shoulder and elbow joints. Although all diarthrodial joints are considered movable, some of these joints move more than others (i.e., the sacroiliac joint is nearly fixed, whereas the shoulder joint is capable of moving in several different directions).
In synarthroses, the bones are held together by connective tissue, cartilage, ligaments, or other bones; thus, their positions are, to a large degree, fixed. Examples of synarthroses are the joints of the skull bones, ribs, and intervertebral disks.
Pathophysiologic Concepts
Atrophy
Atrophy is the decrease in size of a cell or tissue. Muscle atrophy may result from muscle disuse or severing of the nerve supplying the muscle. With muscular atrophy, the size of the myofibrils is reduced. Although bones do not atrophy, bone density can decrease with disuse or metabolic deficiencies or disease.
![]() eriatric Consideration
eriatric Consideration
Muscle mass, muscle strength, and bone density decrease in the elderly, usually as a result of disuse. Decreased muscle mass, muscle strength, and bone density can be reversed, even in the very elderly (those 85 years or older), with moderate, regular weight-bearing exercise. Weight-bearing exercise coupled with mild aerobic exercise such as walking also improves balance and coordination in the elderly and may help prevent falling.
Strains
A strain is trauma to a muscle or tendon, usually occurring when the muscle or tendon is stretched beyond its normal limit. Strains may involve tissue tears or ruptures. Inflammation occurs with injury to muscles or tendons, leading to pain and swelling of tissue. Healing may take several weeks.
Sprains
A sprain is trauma to a joint, usually related to a ligament injury. In a severe sprain, the ligament may be torn. Sprains lead to inflammation, swelling, and pain. Healing may take several weeks.
Joint Dislocation
Dislocation of a joint occurs when a bone is displaced from its position in a joint. A subluxation is a partial dislocation of the joint. Joint dislocation
P.297
typically occurs after a severe trauma, which disrupts the ability of the ligament to hold the bone in place. Dislocation of a joint may also occur congenitally; for example, dislocation of the hip is sometimes seen in a newborn (developmental hip dysplasia). For a trauma-induced dislocation, there is associated marked pain, swelling, and loss of range of motion of the joint. Sometimes a popping noise may be heard or felt at the time of occurrence or during physical examination; in the newborn examination, manipulation of the joint to reproduce the sound or feeling of dislocation is used to diagnose the condition. Dislocation of a joint will usually be apparent on a radiograph and is treated by manipulation or surgical repair followed by immobilization until the joint structures are healed.
Rhabdomyolysis
Rhabdomyolysis, also called myoglobinuria, is the presence of large amounts of muscle protein (myoglobin) in the urine. Rhabdomyolysis usually occurs after major muscle trauma, especially a muscle crush injury. Long-distance running, certain severe infections, and exposure to electrical shock can cause extensive muscle damage and excessive release of myoglobin. Rhabdomyolysis may cause renal failure if the myoglobin gets trapped in the delicate capillaries or tubules of the kidney, interfering with renal blood flow.
Rigor Mortis
Rigor mortis is stiffening or contraction of muscles that occurs several hours after death. The condition results from ATP depletion in the muscle cells. Without ATP to bind to myosin, the actin-myosin cross-bridges that are connected in the muscle at the time of death cannot disengage, and the muscle stays contracted. Within a day, muscle proteins are destroyed by local enzymes released as cells degenerate, causing the muscles to relax.
Conditions of Disease or Injury
Muscular Dystrophy
Muscular dystrophy refers to a variety of diseases characterized by wasting of the muscles. The disorders are not caused by neural, hormonal, or blood flow abnormalities, but rather are inherited disorders involving an enzymatic or metabolic defect. With the defect, muscle cells die and are phagocytized by cells of the inflammatory system, leading to scar tissue buildup and loss of muscle function.
Duchenne's Muscular Dystrophy
The most common form of muscular dystrophy is Duchenne's muscular dystrophy, a sex-linked disorder passed on the X chromosome and seen almost exclusively in males. In approximately 50% of cases, the disease
P.298
shows a clear family history and is passed from mother to son. The other 50% occur as spontaneous mutations on the X chromosome before or during conception. Because males only have one X chromosome, the defective gene that causes the disease is not compensated for by a healthy gene on another X chromosome.
Duchenne's muscular dystrophy results from a defect in the gene that produces the protein dystrophin. Dystrophin appears to act as an anchor for the actin filaments; without it, the muscle fiber is literally pulled apart with repeated contractions. Without dystrophin, muscle cells die and are then phagocytized and replaced by fatty tissue.
Muscle cell weakness begins in the pelvic region by the time an affected child is approximately 2 or 3 years old. The weakness spreads to the legs and upper body within 3 to 5 years. When the muscle cells die, scar tissue and fat cells replace the dead cells, causing the muscles (especially the calf muscles) to appear strong and well muscled (called pseudohypertrophy), when in actuality they are weak and poorly functioning. Eventually, the skeleton begins to deform and the child becomes progressively immobile and finally restricted to a wheelchair. Cardiac muscle is often involved, and approximately 50% of affected children develop heart failure. Respiratory effort is progressively compromised, related to dysfunction of the diaphragm and other respiratory muscles as well as the inability to expand the chest because of severe kyphosis. Smooth muscle dysfunction may cause GI disturbance. Mental retardation also may be present. Death usually occurs as a result of respiratory or cardiac complications in the 20s or younger.
Clinical Manifestations of Duchenne's Muscular Dystrophy
Clumsiness, waddling gait, and frequent falls in toddlers.
Walking on toes because of anterior tibial weakness.
Decreased deep tendon reflexes.
Pseudohypertrophy of the calf muscles.
Gowers' maneuver, whereby the child uses his arms to push up onto his legs when standing up from the floor, is seen during the toddler years.
Immobility and confinement to a wheelchair by the early teens.
Curvature of the spine (kyphoscoliosis) caused by weakness of the postural muscles.
Frequent respiratory infections from a failure to fully expand the lungs.
Diagnostic Tools
Serum levels of the muscle enzyme creatinine phosphokinase (CPK) are elevated, even before symptoms develop. CPK may be elevated in female carriers who are asymptomatic for the disease.
Muscle biopsy will demonstrate cell death, scar tissue, and fatty infiltration.
Electromyography recordings (measurements of electrical signals in a muscle) will be reduced.
Because the gene for dystrophin has been identified, prenatal testing for the disorder is possible.
P.299
Complications
Family stress, feelings of guilt or blame, anger, and grief are common.
Respiratory or cardiac failure and death are likely before adulthood.
Treatment
Support groups and family counseling are important to improve family coping.
Non-strenuous exercises are recommended to maintain mobility and function as long as possible. Strenuous exercise may hasten muscle deterioration.
Experimental studies involving intramuscular injection of dystrophin, or the gene for dystrophin, are underway in animal models. Insertion of the gene for dystrophin may be accomplished by producing a genetically engineered virus that will carry the correct gene into host muscle cells.
Experimental studies are underway in which healthy, immature muscle cells are taken from the fathers of young male children with muscular dystrophy and injected into the muscles of their sons. Whether significant improvement in muscle function will occur in the children is unclear.
The discovery that the protein myostatin limits skeletal muscle growth suggests that inhibition of the myostatin pathway may someday offer a potential treatment for muscular dystrophy.
Bone Fracture
A bone fracture is a break in a bone. Terms used to describe various types of bone fractures include:
Complete fractures Fractures that extend through the bone.
Incomplete fractures Fractures that extend partially through the bone.
Simple (closed) fractures Fractures that do not cause a break in the skin.
Compound (open) fractures Fractures that cause a break in the skin.
Open and closed fractures can be complete or incomplete. Other terms, based on the angle of the break or whether the bone buckles or bends without breaking, may also be used to describe fractures.
Causes of Bone Fractures
The most frequent cause of bone fracture is trauma, especially in children and young adults. Falls and sports injuries are common causes of traumatic fractures. In a child, abuse must be considered when evaluating a fracture, especially if there is a previous history of fractures or if the history of the current fracture is unconvincing.
Some fractures may result after minimal trauma or slight pressure if the bone is weak. These are called pathologic fractures. Pathologic fractures often occur in elderly persons who suffer from osteoporosis, or in individuals with bone tumors, infections, or other diseases.
P.300
Stress fractures may occur in normal bone as a result of prolonged or repeated low-level stress. Stress fractures, also called fatigue fractures, usually accompany a rapid increase in the training level of an athlete, or the beginning of a new physical activity. Because muscle strength increases more rapidly than bone strength, an individual may feel capable of performing beyond a previous level even though the bones may be incapable of supporting the added pressure. Stress fractures are most common in those who pursue endurance sports such as long-distance running. Stress factors may occur in weakened bone in response to only a slight increase in activity level. Individuals suffering a stress fracture should be encouraged to follow a bone-healthy diet and be screened for reduced bone density.
![]() eriatric Consideration
eriatric Consideration
Fractures resulting from even minor falls are a major cause of disability in the elderly. A large percentage of the elderly who suffer a fracture, especially a hip fracture, do not regain the same level of functioning as before the fall. When an elderly person suffers a fracture, loss of independence frequently follows, which often results in the individual being cared for in a nursing home at high cost to both the patient and society. Many frail elderly never recover from a fracture. A fear of falling is a significant concern for many elderly individuals, even those who have never fallen.
![]() ediatric Consideration
ediatric Consideration
Stress fractures are becoming increasingly common in younger athletes, as the pressure to perform in sports at an early age is increasing. Girls and young women are especially affected, perhaps because of the combined pressures for thinness and athletic excellence.
Effects of a Bone Fracture
When a bone breaks, bone cells die. Bleeding typically occurs around the site and into the soft tissues surrounding the bone. The soft tissues are usually damaged by the injury. An intense inflammatory reaction follows the break. White cells and mast cells accumulate, causing increased blood flow to the area. Phagocytosis and removal of dead cell debris begin. A fibrin clot (fracture hematoma) forms at the break and acts as a meshwork to which new cells can adhere. Osteoblastic activity is immediately stimulated and immature new bone, called callus, is formed. The fibrin clot is soon reabsorbed, and the new bone cells are slowly remodeled to form true bone. The true bone replaces the callus and is slowly calcified. Healing takes several weeks to a few months (fractures in children heal faster). Healing can be delayed or impaired if the fracture hematoma or callus is disrupted before true bone is formed, or if the new bone cells are disrupted during calcification and hardening.
P.301
Clinical Manifestations
Pain usually accompanies a traumatic bone break and soft tissue injury. Muscle spasm may follow a bone break and contribute to the pain. With a stress fracture, the pain typically accompanies activity and is relieved by rest. Pathologic fractures may not be associated with pain.
An unnatural position of the bone or a limb may be obvious.
Swelling around the site of a fracture will accompany the inflammatory processes.
Impaired sensation or tingling may occur, signaling nerve damage. Pulses distal to the fracture should be intact and equal to the non-fractured side. A loss of distal pulse may indicate compartmental syndrome (see later), although the presence of a pulse does not rule out this disorder.
Crepitus (a grating sound) may be heard with movement, as the broken ends of the bone move across each other.
Diagnostic Tools
Radiograph can reveal a bone fracture.
Bone scan can reveal a stress fracture.
Complications
Non-union, delayed union, or mal-union of the bone may occur, leading to deformity or loss of function.
Compartmental syndrome may occur. Compartmental syndrome is characterized by nerve and blood vessel damage or destruction that results from swelling and edema in the area of a fracture. With intense interstitial swelling, pressure exerted on blood vessels supplying the area may cause them to collapse. This leads to tissue hypoxia and may cause death of the nerves supplying the area. Typically, pain is intense. The individual may be unable to move his or her fingers or toes. Compartmental syndrome usually occurs in limbs that have tight volume restrictions, such as the arms. Risk of developing compartmental syndrome is greatest if muscle trauma has occurred with the break because swelling will be pronounced. Casting of a fractured limb too early or too tightly may cause increased pressure in the limb compartment, and permanent loss of function or loss of the limb may result. A cast must be immediately removed and sometimes the skin of the limb must be split. To check for compartmental syndrome, the following are evaluated frequently in an injured or cast bone: pain, pallor, paresthesias, and paralysis. A pulse may or may not be felt.
A fat embolus may occur after the break of a bone, especially a long bone. A fat embolus may be generated from exposure of the bone marrow, or may result from activation of the sympathetic nervous system leading to stimulation of free fatty acid mobilization after the trauma. A fat embolus after the break of a long bone frequently lodges in the pulmonary circulation, where it may lead to respiratory distress and failure.
P.302
Treatment
A fracture should be immediately immobilized to allow for formation of a fracture hematoma and minimize damage.
Realignment of the bone (reduction) is important to allow recovery of normal positioning and range of motion. Most reduction can be performed without surgical intervention (closed reduction). If surgery is required for fixation (open reduction), pins or screws may be inserted to maintain realignment. Traction may be required to maintain reduction and stimulate healing.
Long-term immobilization after reduction is important to allow formation of callus and new bone. Long-term immobilization is usually accomplished by casting or the use of splints.
Acute Osteomyelitis
Osteomyelitis is an acute infection of the bone that may occur from the spread of a blood-borne infection (hematogenous osteomyelitis) or, more commonly, after contamination of an open fracture or surgical reduction (exogenous osteomyelitis). A puncture wound to the soft tissue or bone, resulting from an animal or human bite or a misplaced intramuscular injection, may cause exogenous osteomyelitis. Bacteria are the usual cause of acute osteomyelitis, but viruses, fungi, and other microorganisms may be involved.
Osteomyelitis is a difficult disease to treat because local abscesses may develop. A bone abscess typically has a poor blood supply; therefore, delivery of immune cells and antibiotics is limited. Intense pain and lifelong disability may result if a bone infection is not treated immediately and aggressively.
Clinical Manifestations
Symptoms of hematogenous osteomyelitis in children may include fever, chills, and a reluctance to move a particular limb. In adults, symptoms may be vague and include fever, fatigue, and malaise. A urinary, respiratory tract, ear, or skin infection frequently precedes hematogenous osteomyelitis.
Exogenous osteomyelitis typically presents with evidence of injury and inflammation at the site of pain. Fever and regional lymph node enlargement occur.
Diagnostic Tools
Bone scan using injected radiolabeled nucleotides may show an inflammatory bone site. Magnetic resonance imaging (MRI) may allow for increased diagnostic sensitivity.
Blood analysis may demonstrate elevation in complete blood count (CBC) and erythrocyte sedimentation rate, suggesting an active infection is in progress.
P.303
Complications
Chronic osteomyelitis may develop, characterized by unrelenting, severe pain and decreased function of the affected body part.
Treatment
Antibiotics may be prescribed to an individual suffering a bone break or puncture wound to the soft tissue surrounding a bone before a sign of infection develops. If a bone infection occurs, aggressive antibiotic therapy is required.
Osteoporosis
Osteoporosis is a metabolic bone disease characterized by a severe reduction in bone density, leading to easy bone fracture. Osteoporosis occurs when the rate of bone resorption greatly exceeds the rate of bone formation. Bone that is produced is normal; however, because there is too little of it, the bones are weak. All bones can be affected by osteoporosis, although osteoporosis usually develops in the bones of the hips, pelvis, wrists, and vertebral column. See page C6 for illustrations relating to osteoporosis.
Estimates suggest that in the year 2005, more than 2 million Americans developed an osteoporosis-related fracture, incurring costs of $16.9 billion dollars. Although currently seen most frequently in Caucasian women, it is predicted that in the next 20 years, the number of men and minorities with osteoporosis will increase significantly as a result of demographic changes.
Causes of Osteoporosis
The rate of bone formation decreases progressively with age, beginning at approximately age 30 or 40. The denser the bones are before that time, the less likely osteoporosis will occur. As people age into their 70s and 80s, osteoporosis becomes a common disease.
Although bone resorption begins to outpace formation by the fourth or fifth decade of life, in women the most significant thinning of the bones occurs during and after menopause. The postmenopausal decrease in estrogen appears to be primarily responsible for this development in the elderly female population. Although the mechanism by which estrogen acts to preserve bone density is unclear, it is thought that estrogen stimulates osteoblastic activity and limits the osteoclastic-stimulating effects of parathyroid hormones. Therefore, a loss of estrogen causes a pronounced shift toward osteoclastic activity. Thin women, fair-haired women, and women who smoke are especially prone to osteoporosis because their bones are less dense before menopause than those of heavier, dark-haired, nonsmoking women. Elderly men are less prone to osteoporosis because they typically have denser bones than women (by approximately 30%), and reproductive hormone levels remain high until a man is in his 80s. However, elderly men have less dense bones than younger men.
P.304
For men and women, other causes of osteoporosis include reduced physical activity and the ingestion of certain drugs, including corticosteroids and some aluminum-containing antacids that increase calcium elimination. It has been shown that even very elderly men and women can significantly increase bone density by participating in moderate forms of weight-bearing activity. Family history also plays a role in determining an individual's future risk. Bone density has been shown to decrease in breastfeeding women, although a return to near-normal density occurs after weaning.
![]() ediatric Consideration
ediatric Consideration
At particular risk for future development of osteoporosis are children and teenagers who do not consume adequate calcium or vitamin D during their bone-forming years. This is especially, but not exclusively, true of diet-conscious girls who frequently limit caloric and milk intake. Young women athletes in particular may have measurably less dense bone than their peers. This phenomenon appears related to low estrogen levels, which often accompany high or moderate physical exercise and may sometimes lead to amenorrhea (absent or infrequent menstrual cycling). Athletic women also may be thinner than their peers, as a result of the high intensity of their exercise patterns combined with an often reduced caloric intake. These women may experience severe osteoporosis in later years. The combination of amenorrhea, reduced bone mineralization and disordered eating in young women athletes is known as the female athlete triad. In addition, the increased consumption of carbonated soda drinks by both boys and girls increases later risk of osteoporosis. Not only do these drinks often replace milk in the diet, but they may contribute to bone resorption indirectly by affecting calcium handling.
Clinical Manifestations
Although insidiously advancing, osteoporosis may not be associated with any clinical manifestations unless a bone break occurs. Pain and deformity usually accompany a break.
With weakness and collapse of the vertebral bodies, an individual may shrink in height or develop kyphosis (sometimes called dowager's hump).
In 2004, the U.S. Surgeon General identified a low trauma fracture as a sentinel event indicating poor bone health that should be considered an indication for bone density screening, even in young people or others not considered at high risk of osteoporosis.
Diagnostic Tools
A careful family and personal history will identify patients at risk of developing osteoporosis. Physical examination identifying kyphosis and a demonstrable reduction in height will assist diagnosis.
Bone-mineral content of the whole body, and bone-mineral density of specific bones such as the lumbar spine, femoral neck, and shaft of the femur, tibia, fibula and distal radius, are often assessed using dual-energy
P.305
radiograph absorptiometry. This measurement offers an accurate reading of bone mass and allows a clinician to chart the rate of bone decay. Bone density testing that reveals a bone density of fewer than two standard deviations below normal (based on average of young women) is considered abnormal.
Complications
Fractures of the hips, wrists, vertebral column, and pelvis.
Hospitalization, placement in a nursing home, and loss of ability to perform activities of daily living may occur after an osteoporotic fracture.
Treatment
Prevention of osteoporosis begins in childhood and the teen years with the formation of lifelong habits of good nutrition and physical exercise to strengthen bones.
Weight-bearing exercise, even in the very elderly (>85 years), has been shown to increase bone density and muscle mass and improve balance and physical endurance. The effects of high impact exercise, such as jumping and running, on maintaining bone health in children are being studied more closely. The use of vibration to maintain bone health in children and elders with limited mobility is being studied as well.
Dietary supplements of vitamin D and calcium reduce the development of osteoporosis in the elderly and are essential components of prevention.
Cigarette smoking should be avoided.
Estrogen-progesterone replacement therapy or selective estrogen receptor modulators (SERMs) taken during and after menopause can reduce the development of osteoporosis in women. Contraindications for estrogen replacement include a family or personal history of breast cancer or a personal history of blood clots.
Drugs known as bisphosphonates (e.g., alendronate, risedronate, and ibandronate) have been shown to decrease bone resorption and prevent bone loss. These drugs, in combination with vitamin D and calcium supplements, are used for treatment and prevention of osteoporosis. The bisphosphonates significantly increase bone density in the hip and spine especially, and can be used in postmenopausal and drug-induced (glucocorticoid) osteoporosis. They also are used as chemotherapeutic adjuvants in cancer therapy because of their potential to prevent bone metastasis. Bisphosphonates are not easily absorbed by the body, so must be taken on an empty stomach with a full glass of water. The patient must remain upright and refrain from eating for a certain period afterwards, to ensure absorption and prevent gastrointestinal side effects. Adherence to bisphosphonates therefore is often problematic. Recently, a once-a-month oral preparation that may improve adherence has been approved by the FDA. In addition, clinical trials studying the efficacy of intravenous ibandronate administered once every three months are underway. The need to stay in the hospital may reduce this option's popularity. The long-term safety of such a preparation is unknown.
Patients who do not respond positively to other strategies may be given teriparatide, a synthetic parathyroid hormone that helps build bone.
Calcitonin may also be prescribed for those suffering severe osteoporosis. Intranasal administration has recently become available, increasing patient usage.
Testosterone therapy may reduce osteoporosis in men.
The use of AMG-162, a monoclonal antibody that inhibits bone resorption, is now in phase 3 clinical trials. This drug is to be injected, twice yearly. Long-term safety is unknown.
P.306
Hyperparathyroidism
Hyperparathyroidism is a disorder of bone mineralization and muscle weakness caused by high levels of circulating parathyroid hormone. Usually, the elevated parathyroid hormone results from a tumor of the parathyroid gland or another gland. With excess parathyroid hormone, bone resorption is stimulated, resulting in high serum calcium levels. Low serum phosphate accompanies high levels of parathyroid hormone. The bones become brittle and weak.
Secondary hyperparathyroidism can occur with states of hypocalcemia, caused by vitamin D deficiency or renal failure. Calcium levels remain low even with elevated parathyroid hormone.
Clinical Manifestations
Multiple pathologic fractures.
Kyphosis of the spine and vertebral compression fractures.
Fatigue and weakness because high serum calcium leads to decreased nerve and muscle cell excitability.
Diagnostic Tools
Serum assay will demonstrate elevated serum calcium levels (normal 8.6 10.5 mg/dL).
Serum phosphate levels will be low (normal 2.5 4.5 mg/dL).
Complications
Calcium-based kidney stones may develop, causing pain and inflammation of the urinary tract.
Electrocardiogram (ECG) (Chapter 13) abnormalities may develop, including premature ventricular contractions (PVCs) and sinus tachycardia, related to the effects of high serum calcium on cardiac muscle depolarization.
Treatment
Treatment depends on the cause and severity of the disease.
Fluids are essential in management.
Oral phosphate may be administered.
Specific drugs to treat hypercalcemia, including steroids and calcium-losing diuretics, may be used.
Surgical excision of the parathyroid glands may be required.
P.307
Osteomalacia and Rickets
Osteomalacia is a metabolic bone disease seen in adults. It is caused by decreased mineralization of the osteoid as a result of a deficiency of calcium, phosphate, or both. Decreased mineralization results in soft, malleable bone. Osteomalacia usually results from a vitamin D deficiency or insensitivity or from renal disease.
Vitamin D is required for the maintenance of calcium absorption in the gut. With vitamin D deficiency or insensitivity, decreased serum calcium develops, which in turn stimulates parathyroid hormone release. Increased parathyroid hormone stimulates bone breakdown to liberate calcium and increases renal excretion of phosphate. Without adequate mineralization of the bone, the bone becomes thinner. Abnormal amounts of non-crystallized osteoid accumulate and coat the channels of the inner bone, which leads to bone deformity.
Renal failure is associated with osteomalacia because of two factors: the inability of the kidney to activate vitamin D and the decreased ability to excrete phosphate in the urine. Increased serum phosphate also stimulates parathyroid secretion, and hence bone breakdown. Other causes of osteomalacia not directly related to dietary deficiency include the malabsorption of dietary calcium seen in Crohn's disease, malabsorption syndrome, and cystic fibrosis.
Rickets is a bone disease in children caused by vitamin D deficiency. Rickets causes disorganization of the bone, especially at the growth or epiphyseal plates, retarding growth and distorting bone development. Frank rickets is uncommon in the United States, but may be seen in cases of extreme poverty or neglect, or may be subtle in presentation.
Clinical Manifestations
Osteomalacia may be symptomless until a fracture occurs. Vertebral collapse is common, with associated changes in posture and height.
Rickets is characterized by permanent skeletal deformity, including bowed legs, lumbar lordosis (convexity of the spine), and rib and skull deformity. Children may be unable to walk without support. They may also show poor dentition.
Diagnostic Tools
Radiograph evaluation can demonstrate reduced bone ossification.
Measurements of serum calcium and phosphate will be low in severe cases.
P.308
Treatment
Vitamin D therapy with calcium supplementation is required.
If osteomalacia or rickets is caused by another disease, that disease will require treatment.
Osteoarthritis
Osteoarthritis is a degenerative bone disease characterized by loss of articular (joint) cartilage. Without cartilage buffering, the underlying bone is irritated, leading to degeneration of the joint. Osteoarthritis may develop idiopathically (for no known reason) or may occur after trauma, with repeated stress such as that experienced by a long-distance runner or ballerina, or in association with a congenital deformity. Individuals with hemophilia or other conditions characterized by chronic joint swelling and edema may develop osteoarthritis. Osteoarthritis is common in the elderly, affecting more than 70% of men and women older than the age of 65. Obesity can worsen the condition.
CLinical Manifestations
Pain and stiffness in one or more of the joints, commonly the hands, wrists, feet, knees, upper and lower spine, hips, and shoulders. Pain may be associated with tingling or numbness, especially at night.
Swelling of the affected joints, with a decreased range of motion. Joints may appear deformed.
Heberden's nodes, bony growths on the distal interphalangeal joints of the fingers, may develop.
Diagnostic Tools
Arthroscopy (visualization of the joint through a fiber-optic instrument), MRI, and CT scan may support the clinical diagnosis.
Treatment
A balance between resting and exercising the joints, geared toward minimizing inflammation but preserving range of motion, is helpful.
Analgesics and anti-inflammatory drugs to reduce swelling and inflammation.
Surgery may be required to correct a deformity or replace a joint.
Rheumatoid Arthritis
Rheumatoid arthritis (RA) is a chronic, inflammatory disease that causes degeneration of connective tissue. The connective tissue usually destroyed first is the synovial membrane, which lines the joints. In RA, the inflammation becomes unrelenting and spreads to the surrounding structures of the
P.309
joint, including the articular cartilage and the fibrous joint capsule. Eventually, the ligaments and tendons become inflamed. The inflammation is characterized by white blood cell accumulation, complement activation, extensive phagocytosis, and scarring. With chronic inflammation, the synovial membrane undergoes hypertrophy and thickens, occluding blood flow and further stimulating cell necrosis and the inflammatory response. The thickened synovium becomes covered by inflammatory granular tissue called pannus. Pannus may spread throughout the joint, leading to further inflammation and scarring. These processes slowly destroy the bone and cause great pain and deformity.
Causes of Rheumatoid Arthritis
Rheumatoid arthritis is an autoimmune disease that develops in susceptible individuals after an immune response against an unknown triggering agent. The triggering agent may be a bacterium, mycoplasma, or virus that infects the joints or resembles the joint antigenically. Typically, the original antibody response to the microorganism is IgG mediated. Although this response may successfully destroy the microorganism, individuals who develop RA begin to produce other antibodies, usually IgM or IgG, against the original IgG antibody. These self-directed antibodies are called rheumatoid factors (RFs). The RFs persist in the joint capsule, causing chronic inflammation and destruction of the tissue. RA is thought to result from a genetic predisposition to autoimmune disease. Women are more often affected than men. There is strong evidence to suggest that various cytokines, especially tumor necrosis factor alpha (TNF- ), contribute to the cycle of inflammation and joint destruction.
Clinical Manifestations
Onset of RA is characterized by general symptoms of inflammation, including fever, fatigue, body aches, and joint swelling. Joint tenderness and stiffness develop, first because of acute inflammation and then from scar formation. The metacarpophalangeal joints and the wrists are usually first involved. Stiffness is worse in the morning and affects joints bilaterally. Episodes of inflammation are interspersed with periods of remission.
Decreased range of motion, joint deformity and muscular contractions.
Extrasynovial rheumatoid nodules develop in approximately 20% of individuals with RA. These swellings consist of white blood cells and cell debris that present at areas of trauma or increased pressure. Usually, nodules develop in the subcutaneous tissue over the elbows and fingers.
Diagnostic Tools
Elevated serum rheumatoid factor in 80% of cases.
Radiograph changes including bony decalcification of the joints.
Synovial fluid aspiration may show white blood cells in a sterile culture.
P.310
Complications
Extrasynovial rheumatoid nodules may develop on cardiac valves or in the lungs, eyes, or spleen. Respiratory and cardiac function may be affected. Glaucoma may result if nodules that block outflow of ocular fluid develop in the eye.
Vasculitis (inflammation of the vascular system) may lead to thrombosis and infarction.
Loss of ability to carry out activities of daily living, depression, and family stress may accompany exacerbations of the disease.
Treatment
Rest of the inflamed joints during exacerbations.
Rest periods each day.
Alternating hot and cold packs.
Aspirin, other non-steroidal anti-inflammatory drugs, or systemic steroids. Other therapies such as gold treatments may be tried.
Anti-TNF medications are being used to block cytokine-mediated inflammation.
Surgery to remove the synovial membrane or to correct deformity.
Herbal remedies with anti-inflammatory properties have been used for generations to reduce the symptoms of rheumatoid arthritis. These include cat's claw (Uncaria tomentosa), devil's claw (Harpagophytum procumbens), and the Chinese herb lei gong teng (Trypterigium wilfordii). Practitioners should ask patients if they are using these or other over-the-counter medications, and should counsel patients regarding the lack of scientific evidence on the mechanism of action and clinical effectiveness of these herbs.
Osteogenesis Imperfecta
Osteogenesis imperfecta (OI) is a genetic disease characterized by a defect in the synthesis of connective tissue. It results in thin, poorly developed bones that fracture easily. There are a variety of genetic mutations that may result in this disease, all of which result in an abnormality in the production of procollagen proteins.
There are four general categories of OI, separated based on clinical manifestations and genetic analysis. OI is usually inherited as an autosomal-dominant disorder, meaning that an individual heterozygous for the trait will express the disease. The expression of the disease is variable. In other cases, the genetic disorder is inherited as an autosomal-recessive disease. In this case, the child is homozygous for the genetic error and the resultant pathology is more severe. These children typically die before, during, or soon after birth from fractures sustained in utero or during delivery.
P.311
Clinical Manifestations
For the autosomal-dominant condition, children may appear healthy until they begin to walk and fall as toddlers. At this time, frequent fractures with poor healing may occur. Some cases may be investigated as child abuse before the condition is diagnosed.
Children will be of short stature and may have deformed cranial structure and limbs.
Thin skin, with bluish sclera of the eye, indicates reduced collagen deposits.
Tooth development and enamel are abnormal.
Deafness frequently develops as the child ages because of bone deformity and scarring of the middle and inner ear.
Diagnostic Tools
Elevation of serum alkaline phosphatase levels. Alkaline phosphatase is released with cell injury and is high during periods of rapid bone formation.
Fibroblast analysis in a skin culture will demonstrate reduced quantity of connective tissue producing cells.
Prenatal diagnosis of OI is possible.
Complications
Infants homozygous for the autosomal-recessive condition are frequently stillborn or die within the first year of life.
Treatment
Treatment is aimed at reducing the number of fractures by teaching safety measures.
Secure stabilizing of fractures with internal fixation is frequently performed.
Moderate levels of growth hormone supplementation may improve growth outcome and reduce fracture occurrence.
Scoliosis
Scoliosis is curvature of the spine. It may result from an actual structural deformity of the vertebral column that is present at birth (congenital) or may develop as a result of a neuromuscular disease such as cerebral palsy or muscular dystrophy. Some structural scoliosis may develop for no known reason (idiopathic) or as a result of poor posture. Scoliosis results in deformity, and occasionally in pain. If the condition is untreated, respiratory and pulmonary function may be compromised.
Clinical Manifestations
Abnormality of the usual concave-convex-concave vertebral presentation seen in descent from shoulder to buttocks.
Prominence of the ribs on the convex side.
Unevenness in the height of the iliac crest, which may cause one leg to be shorter than the other.
Asymmetry of the thoracic cage and misalignment of spinal vertebrae will be apparent when the individual bends over.
P.312
Diagnostic Tools
Physical examination and screening may allow for early diagnosis.
Lateral deviation of the vertebrae can be confirmed by radiograph.
Complications
Back pain may develop. Respiratory, cardiac, and GI complications may develop from thoracic or lumbar deformity.
Treatment
Postural scoliosis is treated by passive and active exercises. An external brace may be used to encourage compliance and speed recovery.
Structural scoliosis is treated with surgical intervention, which may include placing a flexible rod down the back to reverse the curve of the vertebral column. Fusion of the spine at different levels to correct a deformity may be performed in severe cases.
Paget's Disease
Paget's disease is a bone disorder characterized by accelerated patterns of bone remodeling. Repeated episodes of rapid bone breakdown are followed by short periods of bone formation. The new bone is thickened and rough, and the process eventually leads to structural deformity and weakness. Blood flow to bones affected by Paget's disease is increased to support high metabolic demands. The long bones and the bones of the cranium, spine, and pelvis are most commonly affected. Paget's disease is usually seen in those older than 70 years of age. There is no known cause of the disease.
Clinical Manifestations
Changes in the shape of the skull, with associated headaches, hearing abnormalities, and sometimes mental deterioration.
Pain in the long bones, spine, or pelvis.
Frequent pathologic fractures.
Diagnostic Tools
Radiographs demonstrate bone deformity and will support a clinical diagnosis.
Elevation of serum alkaline phosphatase supports the diagnosis.
Bone biopsy can rule out infection or tumor.
P.313
Complications
Heart failure may occur because of the high blood flow demands of remodeling bones (high-output failure).
Respiratory failure may occur if thoracic bones are affected and deformed.
Paget's disease is a risk factor for sarcoma (bone cancer), perhaps related to the rapid rate of cell cycling seen with the disease.
Treatment
Calcitonin may be administered to slow the rate of bone breakdown.
Anti-inflammatory agents may reduce the pain associated with growing deformity. These drugs will reduce the constant inflammation that accompanies cell breakdown. The disease has no known cure.
Talipes Equinovarus
Talipes equinovarus, also called clubfoot, is a congenital abnormality characterized by deformity of the bones and soft tissue of the foot. The front portion of the foot is abducted (turned in) whereas the rear of the foot is inverted. The foot is usually pointed down (in equinus). The individual typically walks on tiptoes. Clubfoot results from abnormal development of the foot during gestation (fetal growth). This leads to abnormality of the muscles and joints and soft tissue contracture. There may be a genetic tendency for this disorder, and other associated structural malformations may be present.
Clinical Manifestations
Clubfoot is apparent at birth.
Treatment
Casting of foot (or feet) immediately after birth with cast replacement weekly for several months.
Corrective shoes may be necessary during the toddler years.
Surgery may be performed, usually between 6 and 9 months of age.
Developmental Hip Dysplasia
Developmental hip dysplasia is dislocation of the hip present at birth (congenital) or occurring within the first year of life. The hip may be out of the hip joint or present in the joint but easily displaced. The acetabulum (joint cavity) may be abnormally shaped, which allows the head of the femur to slip. The cause of hip dysplasia is unknown, but a genetic tendency toward the disorder is apparent. Breech birth is a strong risk factor for hip dysplasia, as is any condition that limits space for the fetus in the uterus, such as multiple fetuses, abnormalities in the anatomy of the uterus, or amniotic fluid deficiency.
P.314
Clinical Manifestations
Asymmetry in leg length, leading to gait abnormality.
Asymmetry in folds of the gluteus (buttocks).
Diagnostic Tools
A thorough examination of the hips is performed during each physical examination in the newborn period and up until 18 months of age. This examination includes visual inspection of the limbs for differences in length and physical manipulation of the hip joints. In one procedure, the infant is placed on his or her back with his or her legs splayed outward. Pressure is then exerted downward on the knees, and the observer watches and feels for hip dislocation; a positive response is called Barlow's sign. A second maneuver follows in which the practitioner gently reduces the hip back into the joint, producing a telltale click, called Ortolani's sign.
Ultrasound or radiograph is used to confirm the diagnosis.
Complications
Limping and leg pain and deformity may result without treatment. In older children, undiagnosed hip instability may be a cause of delayed standing and walking.
Treatment
Early treatment is essential and may involve bracing or casting depending on the child's age and the severity of the defect. Surgery may be required for late diagnosis or if casting or braces are ineffective.
Osgood-Schlatter Disease
Osgood-Schlatter disease is a condition in which there is partial separation of the epiphysis of the tibia from the tibial tuberosity (joint area of the knee). This occurs as a result of physical stress placed on the knee during periods of rapid growth in early puberty. The stress is usually associated with sports such as running, biking, climbing, or hiking. This condition is especially common in teenage boys from the ages of 11 to 15, and girls from 8 to 13 years of age. Inflammation of the patellar tendon (tendonitis) occurs, as does the development of ossified cartilage in the tibial tuberosity. This condition is usually self-limited and symptoms resolve on closure of the tibial growth plate at the end of puberty. Occasionally, minor symptoms may continue in adulthood.
Clinical Manifestations
Pain at the front of the knee, especially during physical activity or kneeling.
P.315
Diagnostic Tools
A thorough history and physical examination is used to diagnose Osgood-Schlatter disease. MRI or radiography may be used to rule out other causes of pain.
Complications
The condition is usually self-limited. Occasionally pain may continue past puberty.
Treatment
Treatment is usually limited to the use of anti-inflammatory medications and ice packs after exercise. Rest and refraining from sports may be required during a flare-up. Knee supports or braces may be of some use.
Occasionally, surgery may be performed if the condition is severe or seems complicated by the development of bony fragments in the patellar tendon.
Bone Tumors
Bone tumors may be cancerous or benign. Bone cancer may occur as a primary disease (originating in the bone) or more commonly as a result of metastasis from another tumor.
Primary bone cancer may begin in any cell of the bone. Cancers of the bone marrow lead to leukemia or myeloma. Leukemia and myeloma are discussed in Chapter 6. Primary cancer of the osteoblast or osteocyte is called osteogenic sarcoma. Osteogenic sarcoma often occurs in a long bone, especially the femur (thigh), or in the knee. Cancers of the cartilage are called chondrosarcoma. Chondrosarcoma usually occurs in the femur or pelvis.
Clinical Manifestations
Pain related to inflammation with swelling in and around the bone.
Pathologic fracture.
Diagnostic Tools
MRI will identify tumors of the bones.
Bone biopsy will identify the neoplasm and the involved tissue.
Complications
Amputation of the limb is common.
Anxiety, fear, and family stress often accompany the diagnosis of cancer, especially in children.
P.316
Treatment
For osteosarcoma and chondrosarcoma:
Resection of the diseased part of the bone may allow successful treatment of the cancer of without amputation.
Amputation of the limb may be required.
Chemotherapy is administered.
Bone marrow cancers are treated with chemotherapy and radiation.
Selected Bibliography
Cummings, S.R., Browner, W. S., Baurer, D., Stone, K., Ensrud, K., Jamal, S., & Ettinger, B. (1998). Endogenous hormones and the risk of hip and vertebral fractures among older women. New England Journal of Medicine 339, 733 738.
Goodman, L.R., & Warren, M.P. (2005). The female athlete and menstrual function. Current Opinions in Obstetrics and Gynecology 17, 466 470.
Guyton, A.C., & Hall, J.A. (2006). Textbook of medical physiology (11th ed.). Philadelphia: W.B. Saunders.
Kuehn, B.M. (2005). Better osteoporosis management a priority. Impact predicted to soar with aging population. Journal of the American Medical Association 293, 2457 2458.
Kuehn, B.M. (2005a). Longer-lasting osteoporosis drugs sought. Journal of the American Medical Association 293, 2458.
Obermair, G.J., Kugler, G., Baumgartner, S., Tuluc, P., Grabner, M., & Flucher, B.E. (2005). The Ca2+ channel 2G-1 subunit determines Ca2+ current kinetics in skeletal muscle but not targeting of 1 or excitation-contraction coupling. Journal of Biological Chemistry 280, 2229 2237.
Setty, A.R., & Sigal, L.H. (2005). Herbal medications commonly used in the practice of rheumatology: Mechanisms of action, efficacy, and side effects. Seminars in Arthritis and Rheumatism 34, 773 784.
Tobin, J.F., & Celeste, A.J. (2005). Myostatin, a negative regulator of muscle mass: Implications for muscle degenerative diseases. Current Opinion in Pharmacology 5, 328 332.
Tosi, L.L. (1997). Osteogenesis imperfecta. Current Opinion in Pediatrics 9, 94 99.
Waldrop, J. (2005). Early identification and interventions for female athlete triad. Journal of Pediatric Health Care 19, 213 220.
Wehrens, X.H.T., Lehnart, S.E., & Marks, A.R. (2005). Intracellular calcium release and cardiac disease. Annual Review of Physiology 67, 69 98.
EAN: 2147483647
Pages: 26