11. The Senses
Authors: Corwin, Elizabeth J.
Title: Handbook of Pathophysiology, 3rd Edition
Copyright 2008 Lippincott Williams & Wilkins
> Table of Contents > Unit III - Integrated Control and Dysfunction > Chapter 11 - The Senses
function show_scrollbar() {}
Chapter 11
The Senses
Our senses link us to each other and to the world. Each sense allows us to respond to subtle and not-so-subtle stimuli with precision and recognition. Because of the way our senses bring the external environment to us, the day-to-day implications of losing a sense are enormous. The senses include sight, hearing, taste, smell, and touch.
Physiologic Concepts
Sight
Stimulation of light-sensitive receptors in the eye, called photoreceptors, leads to the sense of sight. The response of the photoreceptors is transmitted to the brain by way of electrical signaling that passes through several levels of increasingly complex cell networks. Once the signals reach the brain, they are interpreted as a particular visual image based on the complexity of firing patterns, the rate of firing frequency, and the coding of color. Therefore, to have the sense of sight, one needs a functioning eye to receive a stimulus, cells capable of coding it electrically, intact neural pathways to transmit the electrical signal, and a cerebral cortex capable of interpreting the signal as a meaningful image.
Structure of the Eye
A diagrammatic representation of the eye is presented in Figure 11-1. The outermost (anterior) portion of the eye consists of a tough white membrane covering the eyeball, called the sclera. At the center of the eye, the sclera
P.318
becomes a transparent membrane, the cornea. Light rays enter the eye through the cornea. By way of its natural curvature, the cornea bends the rays, causing the light to become less scattered and more focused on the underlying tissue. The image projected through the cornea is upside down and reversed right to left where it strikes the back of the eye.
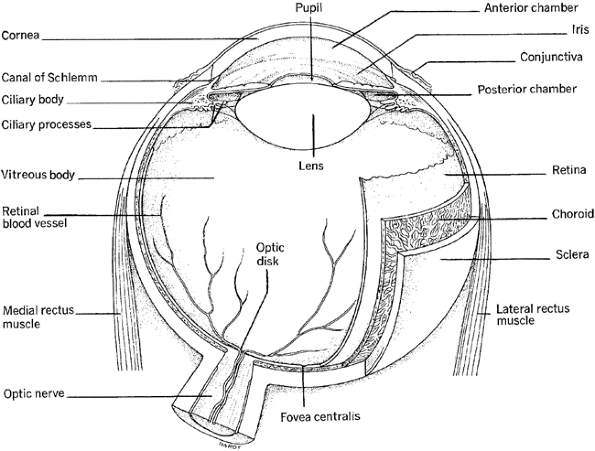 |
Figure 11-1. Transverse section of the eyeball. (From Porth, C. [2005]. Pathophysiology: Concepts of altered health states [7th ed.]. Philadelphia: Lippincott Williams & Wilkins. ) |
The choroid, a pigmented membrane lying under the sclera, helps reduce light scatter. Directly under the cornea, the choroid becomes the iris. The iris is a colored membrane that gives the eye its tint. At the center of the iris is an area without pigment: the pupil. The cornea focuses light rays on the pupil. The diameter of the pupil is controlled by smooth muscles that innervate the iris. These muscles cause the pupil to contract in the light and to dilate in the dark. The muscles controlling the diameter of the pupil are innervated by parasympathetic and sympathetic nerves. Parasympathetic stimulation causes the pupil to constrict, and sympathetic stimulation causes it to dilate. Varying pupil diameter determines the amount of light that passes deeper into the eye.
Posterior to the iris and pupil is the lens. The lens is a curved transparent structure that further bends the light rays. By passage through the lens, light rays are focused exactly on the most posterior and sensitive portion of the eye, the retina. The shape of the lens is controlled by a muscle that allows the lens to focus both distant and close objects precisely on the retina. The retina contains the photoreceptors of the eye, the rods and cones that change light rays into the electrical messages that the brain
P.319
interprets as vision. In the center of the retina is the macula, the site of the most acute and finely detailed vision. The retina also contains the cells that join together to form the optic nerve the pathway by which the visual signal travels to the brain. The fovea centralis is a depression in the macula corresponding to the point of central vision. Between the lens and the retina, the eyeball is filled with blood vessels and a gelatinous fluid, called vitreous fluid.
![]() eriatric Consideration
eriatric Consideration
The center of the lens receives no direct capillary supply. Therefore, as a person ages, the cells at the center of the lens are the oldest and least oxygenated. When the cells in the center of the lens die, they are not replaced. This loss tends to make the lens stiff and less transparent. The lens becomes less able to change its shape to focus an object on the retina, which causes the object to appear out of focus. Visual quality is often reduced in the elderly. The lens may also become opaque (cloudy) with age, a condition known as a cataract. Cataracts further limit visual quality.
Cells of the Retina
The structures of the eye anterior to the retina function primarily to focus the light rays scattered by a particular image exactly on the retina. Once the light rays strike the retina, the cells there have the job of changing the light signal into an electrical signal and then passing the signal to the brain. The cells of the retina that receive and transform the signal are the photoreceptors: the rods and the cones. The second class of cells in the pathway is the bipolar cells; bipolar cells receive the electrical signal from the rods and the cones and pass that signal to the third class of cells in the retina: the ganglion cells. The axons of the ganglion cells travel to the central nervous system as the optic nerve.
Rods and Cones
Rod-shaped photoreceptors are heavily concentrated on the periphery of the retina. Each rod is connected by way of a chemical synapse to a bipolar cell. The rods contain the photosensitive chemical rhodopsin, which is one of the four photopigments present in the retina (the other three are found in the cones). Rhodopsin decomposes when struck by light. When rhodopsin is decomposed, sodium permeability in the rod is reduced, leading to a hyperpolarization of the rod (i.e., the inside becomes more negative). Hyperpolarization decreases the firing rate of the rod on the bipolar cell. Normally, the rod inhibits the firing of the bipolar cell. When the rod is hyperpolarized by light, bipolar cell inhibition is removed, and the bipolar cell depolarizes. Depolarization of the bipolar cell causes an action potential to fire in the ganglion cell. Action potentials produced by the ganglion cells are sent to the brain via the optic nerve. Rods fire even with low levels of light and so function to provide night vision. They do not provide information regarding color.
P.320
Cone photoreceptors are heavily concentrated in the center of the eye. They are the only photoreceptors present in the fovea, an area located at the center of the macula. Each cone cell contains one of the other three photopigments, which, like rhodopsin, decompose when struck by light. As in a rod cell, decomposition of the photopigment in the cone cell hyperpolarizes the cone cell, removing its inhibitory influence from the bipolar cell and causing an action potential to fire in the bipolar cell. As before, the bipolar cell transmits this action potential to a ganglion cell that sends the signal to the brain via the optic nerve. Unlike rods, cones are insensitive to faint light and so contribute little to night vision. The photopigments present in the cone cells are color sensitive, allowing cone cells to transmit information regarding color, as described below. Vitamin A is an important component of both rhodopsin and the cone photopigments.
Reactivation of the photopigments
Immediately after decomposition, the original structure of the photopigments is returned. The rods and cones again inhibit the firing of the bipolar cells and are ready to respond to another light signal.
Differences between rods and cones
Rods are capable of responding to low levels of light; therefore, they provide limited vision in the dark. Many rods usually converge on one bipolar cell. The convergence of many rods on one bipolar cell reduces the acuity of rod vision, but increases its sensitivity. Rods are not color sensitive. Therefore, all stimulation is perceived in shades of gray. Cone stimulation requires higher levels of light and so cones do not fire in the dark or in near dark. Because few cones converge on any one bipolar cell, there is increased acuity of cone vision.
Color vision
The different photopigments of the cones allow for color vision because of their sensitivity to the colors red, blue, and green. The ratio of red, blue, and green cones activated at any one time results in color vision. Which cones are stimulated determines which bipolar cells depolarize and which ganglion cells fire action potentials. Ganglion cells may receive information from several different bipolar cells activated by one color-specific cone or a few different color-specific cones. Ganglion cells may be activated by one color, but inactivated by a second color. These variations and levels of stimuli allow for fine discrimination between many shades of color.
Lateral Neurons
Two types of neurons, the horizontal cells and the amacrine cells, lie laterally (sideways) in the retina and fire in such a way that they modify and control the messages being passed from the rods and cones to the bipolar cells and from the bipolar cells to the ganglion cells. Horizontal cells connect rods and cones to each other and to bipolar cells. Horizontal cells fine-tune the transmission of signals to the bipolar cells, thereby refining visual acuity. The amacrine cells fine-tune signals between the ganglion cells, functioning to
P.321
sharpen transient responses. Given the large number of lateral neurons in the retina, their importance in fine visual discrimination must be great indeed, although their exact mechanism of action is poorly understood.
Optic Nerve
Ganglion cell axons join together to form the eye's optic nerve (cranial nerve II). The optic nerve leaves the eye as a bundle through an area of the retina called the optic disk. The optic disk lacks rods or cones; therefore, it does not participate in the response to light (i.e., it is a blind spot). The central artery of the retina enters the eye through the optic disk. An area called the physiologic cup is at the center of the optic disk.
When the optic nerve reaches the brainstem, some fibers from the left eye cross and project to the right side of the brain. At the same time, some fibers from the right eye cross and project to the left side of the brain. This crossing allows both cerebral hemispheres access to information from each eye. Other fibers do not cross sides. The optic nerves terminate in the thalamus, in an area called the dorsal lateral geniculate nucleus, and there activate other neurons that then project to the occipital lobe. It is in the occipital lobe the area of the brain that interprets the electrical signals as a meaningful visual image that the visual cortex of the brain resides (Fig. 11-2).
P.322
The integrity of the image from the dorsal lateral geniculate nucleus to the occipital lobe is ensured because each cell in the dorsal lateral geniculate nucleus passes the information in the same exact spatial arrangement to the visual cortex. The pathway of vision is shown in Figure 11-3.
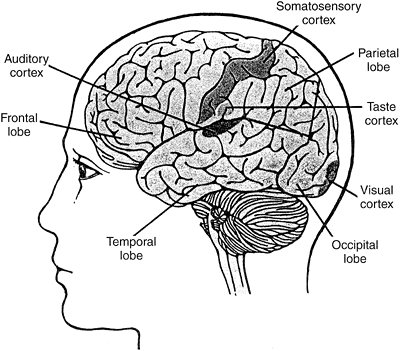 |
Figure 11-2. Primary sensory areas of the cerebral cortex. (Used with permission from Vander, A.J., Sherman, J., & Luciano, D. [1998]. Human physiology [7th ed.]. Boston: McGraw-Hill. ) |
Descending Inhibition to the Dorsal Lateral Geniculate Nucleus
Descending fibers from higher brain centers can influence the transmission of signals from the dorsal lateral geniculate nucleus to the visual cortex. These inhibitory and excitatory fibers come from the visual cortex itself and from areas of the brainstem. Descending stimulation can limit or accentuate what visual information is allowed to pass into consciousness.
Integration of the Visual Pathways
Integration of the pathways of vision occurs at each level of the retina: between the rods and the cones, at the horizontal cells, at the amacrine cells, at the bipolar cells, and at the ganglion cells. At each level, some cells fire with certain stimuli, such as specific on-off fields or various horizontal and vertical patterns, while the same stimuli turn off the firing of other cells. Ganglion cells have a complex pattern of firing that depends on the various on-off firing patterns of the bipolar cells and the amacrine cells. Bipolar cells are activated by unique combinations of the firings of rods, cones, and horizontal cells. Continued integration of signals in the brain further refines the original, simple photoreceptor decomposition that initiated the cascade.
Optics of Vision
It is essential that the cornea and lens direct the light rays exactly toward the cells of the retina so that well-focused images can appear. Although the cornea must bend the rays initially, it is by changing the shape of the lens that the image is made to land exactly on the retina. This process is called accommodation. Accommodation occurs by the contraction and relaxation of the muscles that control the shape of the lens. A progressive loss in the ability of the lens to accommodate and focus the light rays occurs by approximately the fifth decade of life and is responsible for the loss of near vision in middle age (called presbyopia). Accommodation, pupil size, and eye movements are controlled by cranial nerves II, III, IV, and VI.
Intraocular Pressure
Pressure in the chamber of the eye is called intraocular pressure. Intraocular pressure is tightly controlled at approximately 15 millimeters of mercury (mmHg). The pressure is determined by the amount of a fluid, called aqueous humor, which fills the space between the lens and the retina. Aqueous humor is formed by a small gland, the ciliary body, located behind the iris. Once formed, aqueous humor flows through the
P.323
eye, bathing all structures and providing nutrients to the lens and other cells. The aqueous humor then exits the eye through a venous channel between the cornea and the iris. This venous channel, the canal of Schlemm, joins extraocular veins that carry blood and fluid away from the eye.
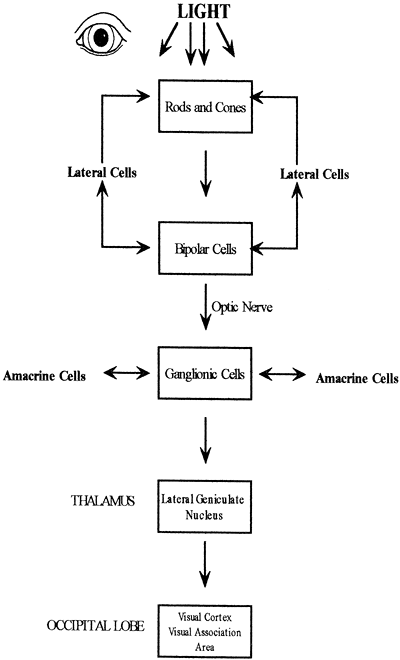 |
Figure 11-3. Pathway of vision. |
P.324
External Structures of the Eye
The lacrimal glands (tear ducts), conjunctiva (mucous membrane layer on the outside of the eye), and eyelids make up the external structures of the eye. These structures protect the eye from irritants and injury. Tears also contain anti-microbial chemicals.
Hearing
The sense of hearing occurs when sound waves enter the external structure of the ear, pass through the middle ear to the inner ear, and stimulate specific receptor cells in the inner ear that fire action potentials, which in turn are carried to the brain. The action potentials are transmitted via the cochlear nerve (part of cranial nerve VIII) to the auditory cortex, a structure located in the temporal lobe of the brain (see Fig. 11-2), where they are interpreted as sounds. A diagrammatic representation of the ear is presented in Figure 11-4.
External Ear
The external ear consists of the auricle (outside cartilage) and the external ear canal. The auricle gathers the sound waves and projects them into the external canal. The external ear canal is a tube through which the sound waves travel to the middle ear. Separating the external ear from the middle ear is the tympanic membrane, also called the eardrum. A part of the temporal bone, the mastoid process, lies behind and below the external canal.
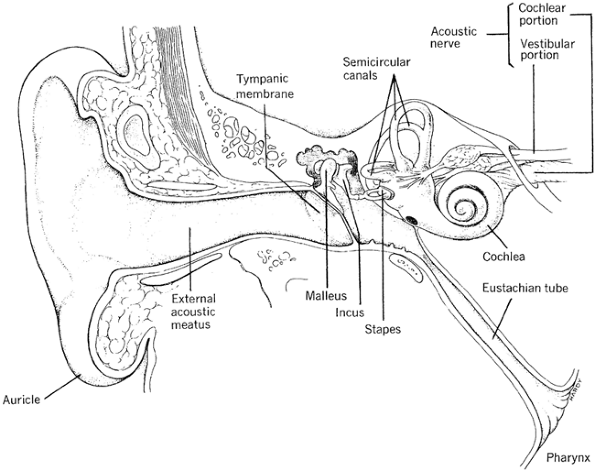 |
Figure 11-4. External, middle, and internal subdivisions of the ear. (From Porth, C. [2005]. Pathophysiology: Concepts of altered health states [7th ed.]. Philadelphia: Lippincott Williams & Wilkins. ) |
P.325
Middle Ear
The tympanic membrane is stretched tightly across the end of the external canal. When sound waves strike the eardrum, it is pushed in, or bowed, toward the middle ear. The degree of bowing of the eardrum depends on the loudness of the sound. After one sound wave, the eardrum returns to its previous position. The eardrum can be pushed in again and again if the sound waves continue, causing the drum to vibrate. The frequency with which the eardrum vibrates depends on the frequency of the sound waves.
The middle ear has three bony processes, which are connected in series to the eardrum: the malleus, the incus, and the stapes. Vibrations of the eardrum are transmitted from one small bone to the next, eventually striking the oval window. The oval window is a small membrane at the entrance to the inner ear. Because the oval window is smaller than the tympanic membrane, the force of the sound waves on the oval window per unit of area is significantly magnified.
The middle ear is connected to the nose and throat via the eustachian tube. Although normally closed, the eustachian tube opens with yawning or swallowing. This opening allows the pressure in the middle ear to remain equal to atmospheric pressure.
Inner Ear
The inner ear is a complex organ consisting of two mazelike structures: the outer bony labyrinth and the inner membranous labyrinth. The bony labyrinth is separated from the membranous labyrinth by thick fluid called perilymph. The membranous labyrinth is filled with a slightly different fluid called endolymph. The bony labyrinth contains the cochlea, the vestibule, and the semicircular canals. The cochlea is the organ responsible for changing sound waves to action potentials. The vestibule and the semicircular canals maintain equilibrium and balance.
Cochlea
The cochlea is a shell-shaped organ, filled with perilymph. The cochlea is separated down the middle by a structure called the basilar membrane. On the basilar membrane is a blanket of hair cells that, together with the basilar membrane, compose the organ of Corti. The hair cells on the basilar membrane depolarize when deformed or bent. Each hair cell synapses upon an afferent neuron, the axons of which make up the acoustic nerve. Depolarization of a hair cell initiates a receptor potential, which, if large enough, stimulates an action potential in the afferent neuron. The hair cells are covered by an overhanging membrane, called the tectorial membrane. It is against the tectorial membrane that the hair cells bend when a sound wave is passed into the inner ear.
Sound Wave Transmission
When a sound wave strikes the oval window, a pressure wave is generated in the fluid-filled inner ear. The pressure wave causes a wavelike displacement
P.326
of the basilar membrane against the overhanging tectorial membrane. As the hair cells rub against the tectorial membrane, they are bent. This bending leads to the depolarization of the hair cell and the production of a receptor potential. With significant deformation, the afferent nerves synapsed upon by the hair cells are stimulated to fire action potentials and the signal is transmitted to the auditory cortex. The pathway of hearing is shown in Figure 11-5.
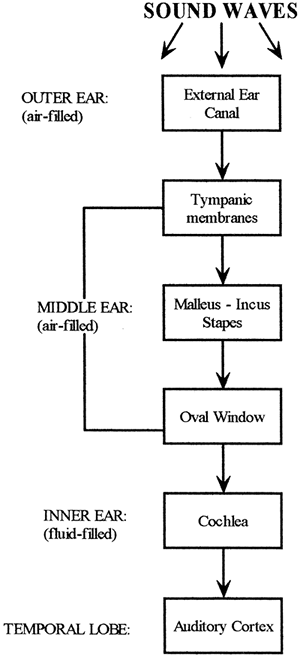 |
Figure 11-5. Pathway of hearing. |
P.327
The frequency of the pressure wave determines which hair cells are displaced and, consequently, which afferent neurons fire action potentials. For instance, the hair cells lying on the part of the basilar membrane nearest to the oval window are most displaced by high-frequency sounds, whereas the hair cells lying on the basilar membrane farthest from the oval window are most displaced by low-frequency sounds. The brain interprets the pitch of a sound by which neurons are activated. The brain interprets the intensity of a sound by the frequency of neuronal impulses and the number of afferent neurons firing.
![]() eriatric Consideration
eriatric Consideration
Hearing acuity generally decreases with age. Causes include atherosclerosis and poor blood flow to the structures of the ear, stiffening of the middle-ear bones, and loss of receptor cells in the inner ear. Cerumen (wax) also builds up, which decreases sound transmission. Concomitant systemic disease, such as diabetes mellitus, may reduce hearing as well by compromising blood flow or diminishing neural transmission.
Vestibular System and Equilibrium
The vestibule and the semicircular canals also contain hair cell receptors that are sensitive to movement and position. When the head is turned, the hair cells are bent as they pass through the thick endolymph surrounding them. As in the organ of Corti, bending of a hair cell in the vestibule and semicircular canals causes depolarization of the cell and the firing of an action potential. Action potentials initiated in the vestibule and semicircular canals are carried via the vestibular nerve to the parietal lobe of the brain, converging near the somatosensory area, where information on joint and muscle position is integrated (see Fig. 11-2). The semicircular canals and the vestibular apparatus work together with other tactile and visual systems to determine the current position of the body and any change in motion or direction.
Taste
Receptors for taste are called taste buds. Taste buds are located in a pattern on the tongue and are depolarized in response to specific chemical stimulation. Depolarization of the taste buds leads to action potentials and the firing of cranial nerves V, VII, IX, and X. These nerves send their information to the taste cortex in the parietal lobe (Fig. 11-2), where the sensation is identified. The pathway of taste is shown in Figure 11-6.
There are specific taste buds for many different taste sensations, some of which are as yet unidentified. Known taste receptors are usually divided into those which respond to sweet, bitter, salty, and sour tastes. Activation of different receptors to varying degrees by substances found in food allows for a wide range of tastes. The sense of taste initiates digestion and provides a stimulus to eat. There can be adaptation (decreased firing) of
P.328
the taste buds if exposure to a chemical stimulus is prolonged. Certain drugs, including nicotine, may sensitize some receptors while desensitizing others, causing taste sensations to change.
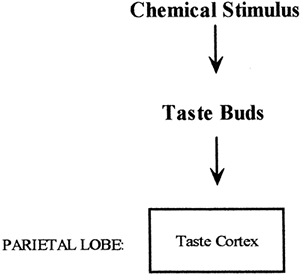 |
Figure 11-6. Pathway of taste. |
Olfaction
The sense of smell is provided by receptor cells, called olfactory cells, which line the membranes of the nasal mucosa. The olfactory cells contain cilia that depolarize when bound by certain chemicals corresponding to specific odors in the air. A few types of cilia hyperpolarize in response to any one specific odor. Significant depolarization or hyperpolarization of the cilia leads to the firing of action potentials in the neurons of the olfactory nerve (cranial nerve I) that terminate in the olfactory bulbs of the frontal lobe. From there, the signal is passed to the olfactory cortex in the limbic system of the brain (see Fig. 11-2). The olfactory receptor cells adapt rapidly to a continuing smell. The pathway of smell is shown in Figure 11-7.
![]() eriatric Consideration
eriatric Consideration
Loss of taste and smell acuity occur with normal aging. Concomitant disease, including Alzheimer's disease, and certain medications taken by the elderly may worsen the normal loss of taste and smell. Reduced taste and smell may contribute to the poor appetite seen in some elderly individuals and may partially explain why the elderly often oversalt their food. Interestingly, the perception of sweet taste does not disappear with age, which may contribute to the weight gain seen in some individuals.
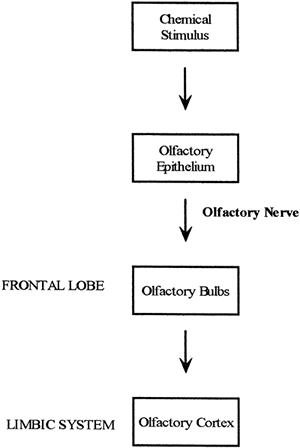 |
Figure 11-7. Pathway of smell. |
P.329
Touch
Tactile sensations include the body's recognition of touch, pressure, and vibration. Each of these sensations appears to be mediated by receptors that vary only in location; touch receptors are located in or near the skin, whereas pressure receptors are found deeper in the tissues. Vibration is sensed as rapidly repeating stimuli activating both touch and pressure receptors.
Tactile Receptors
There are several types of tactile receptors spread over the body. Tactile receptors are mechanoreceptors, cells that respond to physical deformation and compression with depolarization, causing a receptor potential. If the depolarization is great enough, the nerve fiber attached to the receptor
P.330
fires an action potential and transmits the information to the spinal cord and the brain. Different tactile receptors vary in sensitivity and in the velocity with which they send their impulses. They also vary as to which type of nerve fiber transmits their signal to the spinal cord and the brain.
Peripheral Nerve Fibers That Transmit Tactile Information
Tactile sensation is carried to the spinal cord by one of three types of sensory neurons: large type A beta ( ) fibers, smaller type A delta ( ) fibers, and small type C fibers. Both types of A fibers are myelinated, transmitting action potentials rapidly; the larger fibers transmit faster than the smaller fibers. Tactile information carried in the A fibers is typically well-localized and pinpoint. The small C fibers are unmyelinated, transmitting action potentials to the spinal cord much more slowly than the A fibers. Tactile information carried in the C fibers is poorly localized.
Transmission of Tactile Information in the Spinal Cord
Virtually all information on touch, pressure, and vibration enters the spinal cord via the dorsal roots of the corresponding spinal nerve. After synapsing in the spine, highly localized information carried in the fast-firing A fibers (both and ) is sent to the brain by way of the dorsal column lemniscal system. Nerve fibers in this system cross over left to right in the brainstem and travel through the thalamus before synapsing in the somatosensory cortex (see Fig. 11-2). Information on temperature and poorly localized touch is carried to the spinal cord by way of the slow-firing C fibers. This information is sent to the reticular area of the brainstem and then to higher centers via fibers carried in the anterolateral system. Pain and some sexual sensations are transmitted in the anterolateral tracts.
Types of Tactile Receptors
There are six basic types of tactile receptors: free nerve endings, Meissner's corpuscles, expanded-tip tactile receptors, hair end-organ receptors, Ruffini's end-organ receptors, and Pacinian corpuscles. These six types of tactile receptors are discussed individually in the following sections.
Free Nerve Endings
Receptors that respond to touch are found all over the skin and are called free nerve endings. Most send their information to the spinal cord via the small type A fibers. From the spinal cord, information from the free nerve endings is sent through the thalamus to the somatosensory area (parietal lobe) of the cortex. Some free nerve endings send their information to the cord via the slow type C fibers. Free nerve endings respond to stimuli perceived as painful.
Meissner's Corpuscles
Touch receptors found on areas of the body not covered with hair, especially the fingertips and lips, are called Meissner's corpuscles. These receptors
P.331
allow for precise discrimination concerning the location of a touch. Information from Meissner's corpuscles is carried to the spinal cord via fast-firing type A nerve fibers. From the spinal cord, information from Meissner's corpuscles is sent through the thalamus to the somatosensory area of the cortex.
Expanded-Tip Tactile Receptors
The expanded-tip tactile receptors are present in association with Meissner's corpuscles, as well as on areas of the body that do have hair. These receptors provide information on continuous touch, responding with a strong signal when a touch is initiated and continuing with a weak signal for as long as the touch remains. These receptors send their information to the spinal cord via the type A fibers, allowing for fine discrimination concerning the location and the quality of the touch. From the spinal cord, information is delivered through the thalamus to the somatosensory area of the cortex.
Hair End-Organ Receptors
Each hair follicle on the body has a nerve fiber at its base that acts as a touch receptor. When a hair is bent, the nerve fires an action potential. The hair receptors send their information to the spinal cord via type A fibers and then on through the thalamus to the somatosensory area of the cortex.
Ruffini's End Organs
Nerve fibers located deep in the skin and underlying tissues are known as Ruffini's end organs. These receptors fire continuously in response to deformation. Ruffini's end organs are present in the joints and provide information on joint position and movement. They send their information to the spinal cord via type A fibers and then on through the thalamus to the somatosensory area of the cortex.
Pacinian Corpuscles
Pacinian corpuscles are rapidly adapting fibers located under the skin and in other body tissues such as the penis, clitoris, and nipples. These corpuscles fire quickly with the onset of a touch, especially one involving deep pressure or high-frequency vibration, and then adapt. They send information via type A fibers to the spinal cord and then on through the thalamus to the somatosensory area of the cortex. The pathways of touch are shown in Figure 11-8.
Temperature Sensation
Temperature is sensed by specific warm and cold receptors located immediately under the skin. There are more cold receptors than warm receptors. Pain receptors also participate in temperature sensation.
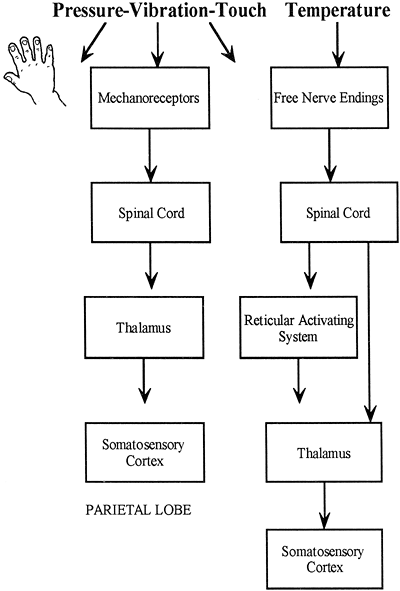 |
Figure 11-8. Pathway of touch. |
P.332
Warm receptors are poorly understood but appear to be free nerve endings that depolarize with a warm stimulus. Cold receptors have been identified as free nerve endings of the small type A fibers. From the spinal cord, nerves carrying temperature information pass through the reticular activating system on their way to the thalamus. A few fibers continue to the somatosensory area of the cortex. Temperature receptors are not mechanoreceptors. They are activated chemically by substances produced by cells after temperature-induced changes in metabolic activity.
![]() eriatric Consideration
eriatric Consideration
Temperature sensation is decreased in the elderly. Decreased sensation may result in accidental burns from heating pads or hot baths.
P.333
Pathophysiologic Concepts
Amblyopia
The loss of visual acuity in an eye that appears to be structurally intact is known as amblyopia. With amblyopia, the central nervous system becomes unable to identify visual stimuli; that is, the signals are sent from the eye but are not recognized in the brain. Often, amblyopia develops from the disuse of one eye (lazy eye), due to conditions of abnormal binocular interaction (e.g., strabismus or infantile cataracts). Amblyopia occurs under these conditions because normal development of the visual areas of the thalamus and the visual cortex require binocular visual stimuli during a critical period of development (0 to 5 years of age). Occasionally, amblyopia may result from ingestion of toxins such as alcohol or tobacco or may be associated with a systemic disease such as renal failure or diabetes mellitus. Although amblyopia may be irreversible, in some cases intensive visual re-training may allow even adults to obtain some vision in the eye.
Strabismus
The condition called strabismus is a deviation in the position of the eyes relative to each other. With strabismus, the eyes may appear to be crossed. An individual who has strabismus often complains of double vision.
Strabismus may result from a congenital inability to use both eyes together. This condition is called nonparalytic strabismus and is treated by patching the eye that can fix on an object (the good eye). Patching forces the deviating eye to focus. Without treatment, ambylopia develops and visual activity in the deviating eye is lost by approximately age 6.
Paralytic strabismus usually occurs later in life after paralysis of one or more of the muscles controlling eye movement. Tumor, injury, or infection may cause paralytic strabismus.
Nystagmus
The involuntary, rhythmic movement of one or both eyes is called nystagmus. The movement may be jerking, rotating, or pendular. Causes of nystagmus include damage to the vestibular system; injury of cranial nerves III, IV, or VI; cerebellar disturbance; and drug intoxication. Rotating nystagmus is frequently associated with dizziness and nausea. Miners exposed to years of working in the dark may develop pendular nystagmus.
P.334
Myopia
Myopia, also called nearsightedness, occurs when the eye is unable to accommodate sufficiently to objects that are far away. Myopia may result from developmental elongation of the eyeball that causes the image to be focused in front of the retina. Myopia has a genetic predisposition and frequently develops in late childhood. It is especially common in children who read extensively, perhaps as a result of changes in the length of the eyeball after prolonged focusing on near objects. Myopia is treated with a concave lens in eyeglasses or contact lenses. A surgical procedure, laser-assisted in situ keratomileusis (LASIK), also can significantly improve myopia. LASIK surgery involves the use of a laser to permanently change the shape of the cornea to improve vision, thereby reducing or eliminating the individual's need to wear corrective lenses.
Hyperopia
Hyperopia, also called farsightedness, occurs when the eye is unable to accommodate sufficiently to close objects, causing the object to be focused past the retina. Hyperopia may be present early in life or may develop later, typically after the fourth decade of life (presbyopia). In older persons, hyperopia is due to inflexibility of the aging lens. Hyperopia is treated with convex eyeglasses or contact lenses. LASIK surgery can significantly improve hyperopia as well.
Astigmatism
In astigmatism, light rays are scattered rather than focused on the retina because of an asymmetric curvature of the cornea. The image is distorted or blurred. Astigmatism may occur with myopia or hyperopia. Specially constructed lenses are required.
Color Blindness
Color blindness is usually a sex-linked genetic disorder caused by a deficiency in one of the three photopigments. Color-blind individuals see only colors formed by the activity of the other two types of cones. Color blindness is passed on the X chromosome; therefore, it usually affects males. In extreme cases, more than one color cone can be deficient.
![]() eriatric Consideration
eriatric Consideration
With age, most people experience a decline in color vision caused by yellowing of the lens. The decline in color vision may interfere with visual cues and contribute to falls.
P.335
Papilledema
Papilledema is the swelling of the optic disk where the optic nerve leaves the eye and enters the brain. Because the optic disk is in communication with the brain, papilledema can occur in any condition that causes severely increased intracranial pressure. Such conditions may include brain tumor, infection, or injury. Papilledema is often an important diagnostic clue in severe brain pathology.
Conductive Hearing Loss
Conductive hearing loss is a decrease in hearing caused by a blockage in the conduction of sound waves in the external or middle ear. Conductive hearing loss may occur if a foreign object is present in the ear or if there is an excessive wax or fluid buildup in the external or middle ear. Middle-ear infections (otitis media) may cause conductive hearing loss. A hearing aid may offer improvement.
![]() ediatric Consideration
ediatric Consideration
A child who suffers repeated episodes of otitis media may develop a speech deficiency if hearing is reduced during critical periods of language development. Repeated episodes of middle-ear infections may cause scarring of the eardrum and permanent hearing loss.
Sensorineural Hearing Loss
Sensorineural hearing loss is a decrease in hearing caused by dysfunction of the organ of Corti, the auditory nerve, or the brain. The organ of Corti may become damaged from prolonged exposure to high levels of noise or after the use of ototoxic (ear-damaging) drugs. Ototoxic drugs include aminoglycoside antibiotics (gentamicin, neomycin, and streptomycin), analgesics (aspirin), tobacco, and alcohol. Systemic diseases, including diabetes mellitus and syphilis, may also cause sensorineural hearing loss.
![]() ediatric Consideration
ediatric Consideration
Congenital sensorineural hearing loss may occur after fetal exposure to rubella or maternal drug exposure (including to the aminoglycosides). Congenital sensorineural hearing loss may also be inherited.
![]() eriatric Consideration
eriatric Consideration
The basilar membrane of the cochlea stiffens with age, resulting in sensorineural hearing loss called presbycusis. Receptor hair cells die and are not replaced. Loss of receptors in the high-frequency range is especially common. Because of these changes, the elderly person is better able to hear deep voices compared with higher-pitched voices.
P.336
Tinnitus
Described as a ringing in one or both ears, tinnitus may accompany ear wax buildup or presbycusis. Aspirin overdose or other drugs may induce tinnitus. Middle-ear infection, M ni re's disease, or otosclerosis (irregular ossification of middle-ear bones) may also cause tinnitus.
Vertigo
The sensation of motion or spinning, often described as a feeling of being off balance, is called vertigo. Vertigo is sometimes accompanied by nausea, weakness, and mental confusion. Inner-ear inflammation, especially of the semicircular canals, is the most common cause of vertigo. Cranial nerve disorders may also cause vertigo. Drugs such as the aminoglycosides, aspirin, and loop diuretics may cause dizziness and imbalance, also related to damaging effects on the inner ear.
Hyposmia
A decrease in the sensation of smell is called hyposmia. Hyposmia may be bilateral or unilateral. If all smells are affected, congestion of the nasal passages is most likely the cause. Hyposmia of a specific smell suggests nerve tract damage. Individuals who suffer a frontal-lobe injury often suffer hyposmia.
Hypogeusia
A decrease in taste sensation is called hypogeusia. Those affected may lose sensation of one specific taste or of all tastes. It may indicate damage to one of the cranial nerves innervating either the tongue or the palate. Sometimes tastes previously enjoyed are suddenly perceived as distasteful. This phenomenon is called parageusia and may occur with drug therapy, including chemotherapeutic drugs, or with liver dysfunction. In the elderly, hypogeusia sometimes occurs spontaneously. Cigarette smoking may affect taste.
Conditions of Disease or Injury
Conjunctivitis
Inflammation of the conjunctiva of the eye caused by an infectious process, physical irritation, or an allergic response is known as conjunctivitis. With inflammation, the conjunctiva becomes red, swollen, and tender. Conjunctivitis stemming from a bacterial infection is sometimes called pink eye. Pink eye may exist alone, or it may coexist with an ear
P.337
infection. Viral conjunctivitis is often caused by adenovirus infection. Bacterial and viral conjunctivitis are highly contagious. Allergic conjunctivitis occurs as part of the inflammatory reaction to an environmental allergen. Physical stimulation by a foreign object in the eye will irritate and inflame the conjunctiva as well, causing inflammation and pain.
Clinical Manifestations
Red, swollen conjunctiva. With infectious or allergic conjunctivitis, both eyes are usually affected.
Photophobia (an aversion to light).
A purulent discharge is characteristic of bacterial conjunctivitis. Infection and discharge often begin in one eye and spread to the other. The eyes may be matted shut by a greenish crust.
A clear, watery discharge is characteristic of viral conjunctivitis. Viral conjunctivitis frequently accompanies an upper respiratory tract infection.
Burning and itching of the eyes is characteristic of allergic conjunctivitis.
Conjunctivitis due to a foreign object is associated with discomfort and a feeling of sand or grit in the eye. Usually with a foreign object, only one eye is affected.
Diagnosis
Diagnosis follows history and physical examination. Cultures may be required in some circumstances.
A foreign object in the eye should be visualized with the use of a special lamp, called a Wood's lamp.
Complications
Certain bacterial infections (gonorrhea, some types of chlamydial conjunctivitis) and viral infections may cause permanent damage to the eye if untreated.
A foreign body in the eye may lead to corneal abrasion and scarring.
Conjunctivitis may be an early symptom of the severe systemic disease Kawasaki's disease. This disease is one of widespread vasculitis that affects many organs of the body, including the heart, brain, joints, liver, and eyes. It begins acutely with a high fever, followed shortly by bilateral conjunctivitis that is notable for its lack of discharge and its prolonged course. A rash and swelling of hands and feet accompany these early symptoms. Early diagnosis is important to prevent damage to the coronary arteries. Treatment for Kawasaki's disease involves the use of aspirin and gamma globulin.
Treatment
Bacterial conjunctivitis is usually treated with antibiotic eye drops or cream, but it often resolves within approximately 2 weeks, even without treatment. Because it is highly contagious among family members and schoolmates, excellent hand washing techniques and separate towels
P.338
for infected individuals are required. Family members should not share bed linens or pillows.Conjunctivitis co-associated with otitis media is treated with systemic antibiotics. Warm compresses placed on the eyes may remove the discharge.
Viral conjunctivitis is usually treated with warm compresses. Excellent hand washing techniques are required to prevent transmission.
Allergic conjunctivitis is treated by avoidance of the allergen if possible. Antihistamines or steroid-containing eyedrops may be used to reduce itching and inflammation.
Conjunctivitis caused by an irritant is treated by removal of any foreign object, followed by the use of antibacterial medication.
![]() ediatric Consideration
ediatric Consideration
Newborns may develop conjunctivitis during the birth process. The causative microorganism is often chlamydia, which may colonize in the mother's cervix, or gonorrhea. Both of these diseases are sexually transmitted. Pregnant women with a sexually transmitted disease should be treated with antibiotics before giving birth. Untreated neonatal eye infections may lead to blindness.
Cataracts
A cataract is a progressive loss in the transparency of the lens. The lens becomes cloudy or gray-white in color, and visual acuity is reduced. Cataracts occur when proteins in the normally transparent lens break down and coagulate on the lens.
Causes of Cataracts
Most cataracts, called senile cataracts, develop from degenerative changes associated with aging. Lifelong exposure to sunlight and a hereditary predisposition contribute to their development.
Cataracts also may occur at any age after trauma to the lens, eye infection, or exposure to radiation or certain drugs. Fetal exposure to rubella virus may cause cataracts. Individuals who have long-term diabetes mellitus frequently develop cataracts, most likely caused by poor blood flow to the eye and by the altered handling and metabolism of glucose.
Clinical Manifestations
Progressively decreased visual acuity.
Blurring of vision, glaring, and loss of color perception occur.
Diagnosis
Cataracts are diagnosed following a history and physical examination, during which whitish opacities on the lens may be seen.
In infants, there may be an absence of the red reflex on eye examination.
P.339
Complications
Loss of vision may occur if untreated.
Treatment
Treatment may involve excision of the entire lens and replacement with an artificial lens, or fragmentation of the lens by ultrasound or laser, followed by aspiration of the fragments and lens replacement.
Glaucoma
Glaucoma is a condition of the eye usually caused by an abnormal increase in intraocular pressure (to greater than 20 mm Hg). The high pressures, sometimes reaching 60 to 70 mm Hg, cause compression of the optic nerve as it leaves the eyeball, leading to death of the nerve fibers. In some cases, glaucoma may develop even though intraocular pressure is normal. This type of glaucoma is associated with other causes of optic nerve damage. Glaucoma is a main cause of blindness in the United States and is the second most common cause of blindness worldwide.
Blindness caused by glaucoma usually develops gradually as intraocular pressures slowly increase, but may occur within a few days if intraocular pressures suddenly become high. Typically, loss of peripheral vision occurs first, followed by loss of central vision. The blindness caused by glaucoma is irreversible. The two main types of glaucoma are acute angle closure glaucoma and primary open angle glaucoma.
Causes of Glaucoma
Glaucoma is usually caused by an obstruction of aqueous humor flow out of the eye chamber. Acute angle closure glaucoma is caused by sudden obstruction to flow through the angle between the cornea and the iris, which may occur with infection or injury or even for unknown reasons. In contrast, primary open angle glaucoma develops more gradually, usually from age-related fibrosis of the angle or gradual obstruction of other channels involved in the flow of aqueous humor. In these cases, there is a progressive increase in intraocular pressure. Occasionally, an increase in the production of aqueous humor may cause increased intraocular pressure. Risk factors for glaucoma include age (10% at age >80), positive family history, African Caribbean origin, thin corneas, myopia, and genetic mutation.
Clinical Manifestations
Acute glaucoma is characterized by severe eye pain and sudden loss of the visual field. The individual may report seeing halos of light around objects. Enlargement of the eye may occur.
Chronic glaucoma is characterized by a slow decrease in visual acuity and blurring, beginning with peripheral vision. Headache and eye pain
P.340
may develop as the condition worsens. The eye may be red and tender to the touch.
Diagnosis
Glaucoma can be diagnosed from history and physical examination. Gradual or sudden reductions in visual field may be reported. Intraocular pressure readings will usually be high, and close inspection of the optic nerve may show characteristic color changes and cupping of the retinal rim.
Early diagnosis of glaucoma is essential to reduce the risk of blindness.
Complications
Blindness may develop with any type of glaucoma. Acute angle closure glaucoma is a medical emergency.
Topical agents used to treat glaucoma may have adverse systemic effects, especially in the elderly. These effects may include worsening of cardiac, respiratory, or neurologic conditions.
Treatment
Eyedrops are applied to decrease intraocular pressure. The most common medications include beta blockers to reduce the production of aqueous humor or parasympathomimetic drugs to constrict the pupil and increase the flow of aqueous humor out of the eye.
With acute angle closure, diuretics may be used to decrease intraocular pressure. Surgery may be required. Intraocular pressures should be monitored annually in individuals older than age 40 or anyone who has an increased risk of the disorder.
Surgery that includes iridectomy for angle closure glaucoma, drainage surgery, or laser trabeculoplasty may be employed to improve aqueous outflow.
Otitis
Otitis is an inflammation of the ear. Inflammation may be of the external ear canal, called otitis externa, or of the middle ear, called otitis media.
Otitis externa may develop in susceptible individuals after swimming or following other types of exposure of the external ear to water. Otitis media is divided into two classes: (1) acute otitis media, characterized by recent abrupt onset, fluid in the middle ear space, acute infection with fever, and ear pain, and (2) otitis media with effusion, characterized by fluid within the middle ear space without signs or symptoms of acute infection (see page C7 for illustrations).
Acute otitis media often results from a bacterial infection, usually by Streptococcus pneumoniae, Haemophilus influenzae, or Staphylococcus aureus. Acute otitis media may also result from a viral infection. Immaturity of the immune system or gastroesophageal reflux disease in young children
P.341
may be causative as well. Acute otitis media occurs when the eustachian tubes that normally drain middle-ear secretions to the throat become blocked or full, causing middle-ear secretions and fluid to accumulate. When the tubes reopen, pressure in the congested ear can draw contaminated nasal secretions through the eustachian tubes into the middle ear, leading to infection.
Otitis media with effusion refers to the accumulation of fluid in the middle ear that often results from an allergy. In some circumstances, a secondary bacterial infection may develop.
Clinical Manifestations
Pain in the affected ear is the most common symptom of acute otitis media.
In an infant or toddler, fever, fussing, and pulling on the ear may signify acute otitis media.
Anorexia, vomiting, and diarrhea may accompany acute otitis media.
An uncomfortable feeling of fullness in the ear is common with otitis media with effusion.
Pain with manipulation of the external structures of the ear suggests otitis externa.
Diagnostic Tools
Otoscopic examination provides information on the eardrum that can be used to diagnose otitis media. Acute otitis media presents with a reddened, bulging eardrum when examined otoscopically. Bony landmarks and the light reflex may be obscured. Otitis media with effusion may present as a gray eardrum, either bulging or depressed inward. Otitis externa is diagnosed by the observance of a reddened, inflamed external canal.
The use of a pneumonic device with the otoscope (pneumatic otoscope) further assists diagnosis of otitis media. By squeezing an air-filled bulb connected to the otoscope, a small bolus of air can be injected into the external ear. The mobility of the tympanic membrane can be observed by the examiner through the otoscope. With acute otitis media and otitis media with effusion, tympanic mobility is reduced.
A tympanogram, a test that involves placing a small probe in the external ear and measuring the movement of the tympanic membrane (eardrum) after the presentation of a fixed tone, can also be used to evaluate tympanic mobility. With acute otitis media and otitis media with effusion, the mobility of the eardrum is reduced.
Audiologic testing may show a hearing deficit, which is an indication of fluid buildup (infectious or allergic).
Complications
Repeated or untreated otitis media may cause scarring of the eardrum and permanent reduction in hearing acuity.
Rare complications of acute otitis media include meningitis, otogenic brain abscess, or infection of the mastoid bone.
P.342
Treatment
Pain diagnosis and management with acetaminophen or other analgesics are recommended for acute otitis media.
Acute otitis media is usually treated with antibiotics, although a period of watchful waiting may be appropriate. Repeated episodes of acute otitis media may lead to insertion of a tympanostomy tube in an attempt to prevent future infections.
Although otitis media with effusion usually will resolve on its own over 3 to 4 months, close observation by the health care provider is needed. When hearing loss is involved, the patient is referred to an otolaryngologist for evaluation on the use of tympanostomy tube placement.
Otitis externa is treated with anti-inflammatory drops, antimicrobial drops, or both.
![]() ediatric Consideration
ediatric Consideration
Infants and young children are most susceptible to middle-ear infections because their eustachian tubes are shorter and straighter than those of older children and adults. Prevention of acute otitis media in infants includes not putting a baby to bed with a bottle and feeding an infant with the head raised. Young children who have repeated ear infections may have reduced hearing acuity or experience language delays. To prevent this, tubes may be placed in the ear to assist drainage.
M ni re Disease
A chronic disorder of the semicircular canals and labyrinths of the inner ear is called M ni re disease. It is named after Dr. Prosper M ni re, who first reported the syndrome in a young girl in 1861. This disease is associated with severe attacks of vertigo (sense of spinning or disequilibrium), often accompanied by nausea. The cause of M ni re disease is unknown, but it appears related to an overproduction of endolymph in the inner ear. Elevation in antidiuretic hormone may be involved in some cases. Occurrences of M ni re disease may follow middle-ear infection or head trauma or may be associated with systemic illness such as thyroid disease. The condition may also show a genetic predisposition. Typically, the disorder is unilateral (only one ear is affected).
Clinical Manifestations
M ni re disease is characterized by extreme vertigo, lasting several minutes to a few hours. The episodes come and go, often with several months between attacks.
Fluctuating tinnitus and hearing loss accompany the attacks.
Nausea, vomiting, hypotension, and sweating often occur with attacks.
P.343
Diagnosis
M ni re disease is usually diagnosed from history and physical examination. Tests of vestibular function, including balance testing, and tests of nystagmus eye movements may help confirm the diagnosis.
Diagnosis needs to rule out other causes of vertigo and tinnitus, including autoimmune disease, auditory nerve damage, or tumor.
Complications
M ni re disease may progress to unilateral nerve deafness.
Treatment
Symptoms may decrease if the patient lies down or sits still, making no sudden movements.
Treatments to reduce fluid volume, including diuretics and a low-salt diet, are suggested. Antihistamines and steroid hormones have been used with varying degrees of success.
Medications that can reduce nausea are available.
Surgical placement of a shunt to drain excess endolymph may be performed.
Prescription of vestibulotoxic drugs, including systemic administration of streptomycin or intratympanic delivery of streptomycin and gentamicin, is used for severe cases.
Pain
Pain is a subjective sensation of unpleasantness usually associated with actual or potential tissue damage. Pain can be protective, in that it causes an individual to back away from a dangerous stimulus, or it can serve no function, as is the case with chronic pain. Pain is sensed when specific pain receptors are activated. Description of pain is subjective and objective, based on the duration, the speed of sensation, and the location.
Receptors for Pain
Pain receptors are called nociceptors. Nociceptors include the free nerve endings, which respond to many stimuli, including mechanical pressure, deformation, temperature extremes, and various chemicals. With intense stimuli, other receptors such as the Pacinian corpuscles and Meissner's corpuscles also send information perceived as painful. Chemicals that cause or worsen pain include histamine, bradykinin, serotonin, several prostaglandins, potassium ion, and hydrogen ion. Each of these substances accumulates at sites of cellular injury, hypoxia, or death, alerting the individual to these happenings. Although all pain receptors are capable of
P.344
responding to any type of tactile stimuli, each receptor appears to respond most readily to one specific type of stimulation.
Duration of Pain
Pain may be acute (lasting less than 6 months) or chronic (lasting longer than 6 months). Acute pain can be beneficial, serving to alert the individual to danger. Chronic pain is never beneficial.
Speed of Sensation
Fast pain is sensed less than 1 second (usually much less) after the application of a painful stimulus (e.g., touching a hot burner). Fast pain is well localized to the site and is frequently described as pricking or sharp. Fast pain is usually felt on or near the surface of the body. It is transmitted to the spinal cord by the A fibers.
Slow pain is felt 1 second or more after the application of a painful stimulus (e.g., pain that continues after a bump to the head). Slow pain is frequently described as dull, throbbing, or burning. It can intensify over the course of several minutes and may occur on the skin or in any deep tissue of the body. Slow pain can become chronic pain and lead to great disability. Slow pain is transmitted to the spinal cord by the slow C fibers. The C fibers are believed to release the neurotransmitter substance P when they synapse in the spinal cord. The neurotransmitter released by the A fibers is unknown. The pathways of pain are shown in Figure 11-9.
Location
Cutaneous pain is pain felt on the skin or in subcutaneous tissues (e.g., pain felt with a pinprick or a skinned knee). Cutaneous pain is well localized over a dermatome (an area of the skin innervated by a certain spinal cord segment) and is transmitted rapidly. Deep somatic pain is pain arising from bones and joints, tendons, skeletal muscles, blood vessels, and deep nerve pressure. A headache is considered deep somatic pain. Deep somatic pain is slow pain, which may radiate along a nerve route. Visceral pain is pain in the abdominal or thoracic cavity. Visceral pain is typically severe and may be well localized at one spot, but it may also be referred to different parts of the body. Visceral pain localizes over embryonic dermatomes and is caused by stimulation of several pain receptors.
Pain Threshold
Pain threshold is the level at which a stimulus is first perceived as pain. In general, humans have similar pain thresholds. An individual's pain threshold varies little over time.
Pain Tolerance
Pain tolerance is an individual's ability to withstand a painful stimulus without demonstrating physical signs of pain. Pain tolerance is unique for
P.345
P.346
each individual. It depends on past experience; cultural, familial, and role expectations; and the individual's current emotional and physical state. In some cultures it is considered weak to show pain, so pain tolerance is high. An individual who is depressed or anxious may have a reduced tolerance for pain. An individual who is distracted, or one who is in the middle of an emergency or an athletic challenge, may show a high tolerance for pain.
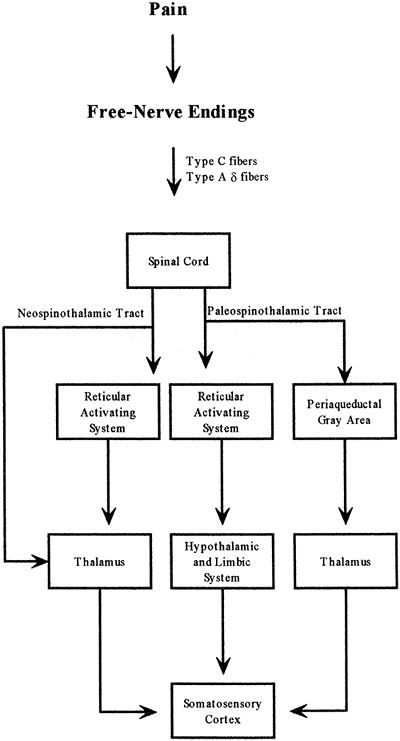 |
Figure 11-9. Pathways of pain. |
Central Nervous System Pathways for Pain
Once in the spinal cord, most pain fibers synapse on neurons in the dorsal horns of the segment they enter. However, some fibers may travel up or down several segments in the cord before synapsing. After activating cells in the spinal cord, information concerning painful stimuli is sent by one of two ascending pathways to the brain the neospinothalamic tract or the paleospinothalamic tract.
Neospinothalamic Tract
Information carried to the spine in the fast-firing A fibers is transmitted ascendingly from the spinal cord to the brain via the fibers of the neospinothalamic tract. Some of these fibers terminate in the reticular activating system, alerting one to the occurrence of pain, but most travel to the thalamus. From the thalamus, signals are sent to the somatosensory cortex where the location of the pain is well localized. Cortical stimulation is required for the conscious interpretation of the pain signal.
Paleospinothalamic Tract
Information carried to the spine in the slowly transmitting C fibers, as well as that carried in a few of the A fibers, is transmitted ascendingly to the brain via the fibers of the paleospinothalamic tract. These fibers travel to the reticular area of the brainstem and to an area of the mesencephalon called the periaqueductal gray area. Paleospinothalamic fibers that travel through the reticular area go on to activate the hypothalamus and the limbic system, influencing the function of these emotion-controlling areas. The periaqueductal gray area is an important integrating center for pain; the perception of pain is highly modified in this area. Pain carried in the paleospinothalamic tract is poorly localized and is responsible for causing the emotional distress associated with pain.
Gating of Pain in the Spinal Cord and the Brain
Experimental evidence suggests that the likelihood of transmitting painful stimuli from the spinal cord to the brain can be influenced by descending neurons firing on the cells of the spinal cord. Descending input to the spine may increase the transmission of a painful stimulus, or it might decrease the likelihood that a stimulus is perceived as painful. Reduced passage of a painful stimulus is called analgesia.
Descending neurons that affect pain transmission come from the cerebral cortex, the hypothalamus, the limbic system, and, especially,
P.347
the periaqueductal gray area. The ability of upper brain areas to influence transmission of pain in the spinal cord is called gating. Gating occurs at each level of pain transmission (across both the neospinothalamic and the paleospinothalamic tracts) and in the brain as well. Fibers from the periaqueductal gray area that diffusely innervate the cerebral cortex, the limbic system, the hypothalamus, and the reticular formation are especially important in influencing pain transmission in the brain.
Interpretation of the Gate Theory
The gate theory of pain offers an explanation of how cultural and personal expectations, mood, and fear can influence an individual's perception and tolerance of pain. By emphasizing the ability of descending pathways to influence pain perception, the gate theory of pain explains how distraction or relaxation techniques may reduce pain, whereas focusing on a painful stimulus may increase the likelihood of the stimulus being passed into consciousness.
The gate theory of pain also explains how gating can occur with peripheral nervous stimulation to the spinal cord. Data suggest that when the large A neurons carrying skin tactile information are stimulated at the same time that the A and C fibers are transmitting painful stimuli, spinal activation of both the neospinothalamic and the paleospinothalamic tracts is reduced. This reduced activation appears to result from lateral inhibition of the cells in the dorsal spine by the large A neurons. Rubbing the head or skin after an injury stimulates the large A fibers and produces some degree of analgesia. This is an example of gating the passage of a painful stimulus.
Endorphins, Enkephalins, and Serotonin
Some of the analgesic responses described above appear to result from the central nervous system production and release of the endogenous opiates: the endorphins and the enkephalins. Serotonin, another neurotransmitter, is also involved in producing analgesia.
Enkephalin is a small peptide released in the spinal cord from neurons descending from the periaqueductal gray area. Enkephalin causes presynaptic inhibition of types C and A fibers in the spine. This inhibition reduces the passage of a painful stimulus beyond the spinal cord. Enkephalin is also present in the limbic system and the hypothalamus.
The endorphins and serotonin act as neurotransmitters in the brain to reduce the passage and perception of pain. The pituitary releases endorphin in response to intense exercise and during painful experiences such as labor and delivery. Endorphins also affect mood. Prolonged pain has been shown to deplete endorphin levels; perhaps this contributes to the despair and anguish seen in individuals who have chronic pain. Serotonin is produced in the brain and is released from descending fibers synapsing in the spinal cord. Drugs that increase brain serotonin levels, such as the tricyclic antidepressants, reduce pain perception.
![]() ediatric Consideration
ediatric Consideration
Infants and children acutely feel pain and should never be exposed to painful therapies without pain medication. Infants may express pain differently than older children and adults.
P.348
Clinical Manifestations
Acute pain is characterized by increased heart rate, increased respiratory rate, facial grimacing, withdrawal, and crying. Dilated pupils and sweating occur. Usually, a person suffering acute pain is highly focused on the pain.
Chronic pain is associated with a return of heart and respiratory rate to normal. An individual who has chronic pain may appear quiet and subdued. Depression and despair may develop.
Diagnostic Tools
Rating scales from 1 to 10 allow an individual to evaluate pain and may help a clinician recognize the intensity of a person's pain. For children, a diagram showing a range of faces, from happy to very sad and crying, may be used to help identify level of pain.
Recognizing the subtle cues shown by an individual in pain is important for responding to pain when cultural, linguistic, or age barriers to communication exist.
Complications
Pain stimulates the stress response. Stress can reduce the functioning of the immune and inflammatory systems, and thus delay or impair healing.
Acute, severe pain may lead to cardiovascular collapse and shock.
Treatment
Application of cool compresses may reduce pain associated with inflammation.
Comfort measures such as back rubs may reduce pain by stimulating the large A fibers and by activating descending pathways stimulated by distraction.
Behavioral techniques, including distraction and imaging, may stimulate descending pathways that block the transmission of painful stimuli to the brain. The Lamaze method of breathing during labor is based on this principle.
Transcutaneous electrical nerve stimulation (electrodes on the skin) may relieve pain by stimulating the large type A nerve fibers. Acupuncture may stimulate these fibers and reduce pain as well.
Analgesics such as acetaminophen can relieve mild pain, most likely by blocking the production of prostaglandins or other substances that sensitize pain receptors.
Nonsteroidal anti-inflammatory drugs, such as aspirin and ibuprofen, or steroids may be used for mild to moderate pain. These drugs block prostaglandin production both locally at sites of injury and in the central nervous system.
Narcotics, such as morphine, can reduce intense pain. Morphine binds to opiate receptors in the central nervous system and alters pain perception.
Nerve block by injection of drugs or surgery may occasionally be used to treat severe pain.
P.349
Selected Bibliography
American Academy of Pediatrics, American Academy of Family Physicians Subcommittee on Management of Acute Otitis Media. (2004). Clinical practice guideline: Diagnosis and management of acute otitis media. Pediatrics 113, 1451 1455.
American Academy of Pediatrics, American Academy of Family Physicians, American Academy of Otolaryngology Head and Neck Surgery, American Academy of Pediatrics Subcommittee on Otitis Media With Effusion. (2004). Clinical practice guideline: Otitis media with effusion. Pediatrics 113, 1412 1429.
Aoki, M., Ando, K., Kuze, B., Mizuta, K., Hayashi, T., & Ito, Y. (2005). The association of antidiuretic hormone levels with an attack of Meniere's disease. Clinical Otolaryngology 30, 521 525.
Bickley, L.S., & Szilagyi, P.G. (2007). Bates' guide to physical examination and history taking (9th ed.). Philadelphia: Lippincott Williams & Wilkins.
Carlson, L. H. (2005). Otitis media. New information on an old disease. Nurse Practitioner 30, 31 41.
De Oliveira Penido, N., Borin, A., Iha, L.C.N., Suguri, V.M., Onishi, E., Fukuda, Y., et al. (2005). Intracranial complications of otitis media. 15 years of experience in 33 patients. Otolaryngology Head and Neck Surgery 132, 37 42.
Guyton, A.C., & Hall, J. (2006). Textbook of medical physiology (11th ed.). Philadelphia: W.B. Saunders.
Hensch, T.K. (2005). Critical period plasticity in local cortical circuits. Nature Reviews Neuroscience 6, 877 888.
Khaw, P.T., Shah, P., & Elkington, A.R. (2004). ABC of eyes. Glaucoma-1: diagnosis. British Medical Journal 328, 97 99.
Khaw, P.T., Shah, P., & Elkington, A.R. (2005). Glaucoma-2: Treatment. British Medical Journal 328, 156 158.
Lee, C.A., Mistry, D., Uppal, S., & Coatesworth, A.P. (2005). Otologic side effects of drugs. Journal of Laryngology and Otology 119, 267 271.
Levi, D.M. (2005). Perceptual learning in adults with amblyopia: A reevaluation of critical periods in human vision. Developmental Psychobiology 46, 222 232.
Liu, L., Zhu, W., Zhang, Z.-S., Yang, T., Grant, A., Oxford, G., et al. (2004). Nicotine inhibits voltage-dependent sodium channels and sensitizes vanilloid receptors. Journal of Neurophysiology 91, 1482 1491.
Megale, S., Scanavini, A., Andrade, E.C., Machado Fernandes, M., & Anselmo-Lima, W.T. (2006). Gastroesophageal reflux disease: Its importance in ear, nose and throat practice. International Journal of Pediatric Otorhinolaryngology 70, 81 88.
Porth, C.M. (2005). Pathophysiology: Concepts of altered health states (7th ed.). Philadelphia: Lippincott Williams & Wilkins.
P.350
Randazzo, A., Nizzola, F., Rosetti, L., Orzalezi, N., & Vinciguerra, P. (2005). Pharmacological management of night vision disturbances after refractive surgery. Results of a randomized clinical trial. Journal of Cataract and Refractive Surgery 31, 1764 1772.
Richardson, B. (2006). Practice guidelines for pediatric nurse practitioners. St. Louis: Elsevier Mosby.
Riente, L., Bongiorni, F., Nacci, A., Migliorini, P., Segnini, G., Delle Sedie, A., et al. (2004). Antibodies to inner ear antigens in Meniere's disease. Clinical Experimental Immunology 135, 159 163.
Schiffman, S.S., & Graham, B.G. (2000). Taste and smell perception affect appetite and immunity in the elderly. European Journal of Clinical Nutrition 54, Supplement 3, S54 S63.
Watson, S.L., Bunce, C., & Allan, B.D.S. (2005). Improved safety in contemporary LASIK. Ophthalmology 112, 1375 1380.
EAN: 2147483647
Pages: 26