25 - Psychiatric Disorders
Editors: McPhee, Stephen J.; Papadakis, Maxine A.; Tierney, Lawrence M.
Title: Current Medical Diagnosis & Treatment, 46th Edition
Copyright 2007 McGraw-Hill
> Table of Contents > 28 - Lipid Abnormalities
28
Lipid Abnormalities
Robert B. Baron MD, MS
For patients with known cardiovascular disease (secondary prevention), cholesterol lowering leads to a consistent reduction in total mortality and recurrent cardiovascular events in men and women and in middle-aged and older patients. Among patients without cardiovascular disease (primary prevention), the data are less conclusive, with rates of cardiovascular events, heart disease mortality, and all-cause mortality differing among studies. Nonetheless, treatment algorithms have been designed to assist clinicians in selecting patients for cholesterol-lowering therapy based on their lipid levels and their overall risk of developing cardiovascular disease.
Lipids & Lipoproteins
The two main lipids in blood are cholesterol and triglyceride. They are carried in lipoproteins, globular particles that also contain proteins known as apoproteins. Cholesterol is an essential element of all animal cell membranes and forms the backbone of steroid hormones and bile acids; triglycerides are important in transferring energy from food into cells. Why lipids are deposited into the walls of large and medium-sized arteries an event with potentially lethal consequences is not known.
Lipoproteins are usually classified on the basis of density, which is determined by the amounts of triglyceride (which makes them less dense) and apoproteins (which makes them more dense). The least dense particles, known as chylomicrons, are normally found in the blood only after fat-containing foods have been eaten. They rise as a creamy layer when nonfasting serum is allowed to stand. The other lipoproteins are suspended in serum and must be separated using a centrifuge. The densest (and smallest) family of particles consists mainly of apoproteins and cholesterol and is called high-density lipoproteins (HDL). Somewhat less dense are the low-density lipoproteins (LDL). Least dense are the large, very-low-density lipoproteins (VLDL), consisting mainly of triglyceride. In fasting serum, most of the cholesterol is carried on LDL particles and is therefore referred to as LDL cholesterol; most of the triglyceride is found in VLDL particles. Specific apoproteins are associated with each lipoprotein class.
Chylomicrons are made in the gut and travel via the portal vein into the liver and via the thoracic duct into the circulation. They are normally completely metabolized, transferring energy from food into muscle and fat cells. The liver manufactures VLDL particles from its own stores of fat and carbohydrate. VLDL particles transfer triglyceride to cells; after losing enough, they eventually become LDL particles, which provide cholesterol for cellular needs. Excess LDL particles are taken up by the liver, and the cholesterol they contain is then excreted into the bile. HDL particles are made in the liver and intestine and appear to facilitate the transfer of apoproteins among lipoproteins. They also participate in reverse cholesterol transport, either by transferring cholesterol into other lipoproteins or directly into the liver.
Lipoproteins & Atherogenesis
The plaques in the arterial walls of patients with atherosclerosis contain large amounts of cholesterol. The higher the level of LDL cholesterol, the greater the risk of atherosclerotic heart disease; conversely, the higher the HDL cholesterol, the lower the risk of coronary heart disease (CHD). This is true in men and women, in different racial and ethnic groups, and at all ages up to age 75 years. Because most cholesterol in serum is LDL, high total cholesterol levels are also associated with an increased risk of CHD. Middle-aged men whose serum cholesterol levels are in the highest quintile for age (above about 230 mg/dL) have a risk of coronary death before age 65 years of about 10%; men in the lowest quintile (below about 170 mg/dL) have a 3% risk. Death from CHD before age 65 years is less common in women, with equivalent risks one-third those of men. In men, each 10-mg/dL increase in cholesterol (or LDL cholesterol) increases the risk of CHD by about 10%; each 5-mg/dL increase in HDL reduces the risk by about 10%. The effect of HDL cholesterol is greater in women, whereas the effects of total and LDL cholesterol are smaller. All of these relationships diminish with age.
The exact mechanism by which LDL particles result in the formation of atherosclerotic plaques or the means whereby HDL particles protect against their formation is not known. The model of LDL carrying cholesterol into the walls of arteries with HDL removing it is simple but not established. The natural oxidation of LDL particles may be particularly atherogenic. Receptors on the surface of macrophages within atherosclerotic plaques bind and accumulate oxidized LDL. The formation of antibodies to oxidized LDL may also
P.1267
be important in plaque formation. The size of the LDL molecule may also influence atherogenesis; at the same LDL concentrations, individuals with large numbers of smaller particles appear to be at higher risk for CHD.
The relationship of VLDL cholesterol to atherogenesis is less certain. The number, size, or subtype of VLDL particles in addition to the total amount in serum may be important. In addition, HDL and VLDL levels are inversely related. Patients with a high VLDL level are likely to have a low HDL level and thus be at increased risk for CHD for that reason alone.
There are several genetic disorders that provide insight into the pathogenesis of lipid-related diseases. Familial hypercholesterolemia, rare in the homozygous state (about one per million) is a condition in which the cell-surface receptors for the LDL molecule are absent or defective, resulting in unregulated synthesis of LDL. Patients with two abnormal genes (homozygotes) have extremely high levels up to eight times normal and present with atherosclerotic disease in childhood. Homozygotes may require liver transplantation to correct their severe lipid abnormalities. Those with one defective gene (heterozygotes) have LDL concentrations twice normal; persons with this condition may develop CHD in their 30s or 40s.
Another rare condition is caused by an abnormality of lipoprotein lipase, the enzyme that enables peripheral tissues to take up triglyceride from chylomicrons and VLDL particles. Patients with this condition, one cause of familial hyperchylomicronemia, have marked hypertriglyceridemia with recurrent pancreatitis and hepatosplenomegaly in childhood.
Numerous other genetic abnormalities of lipid metabolism are named for the abnormality noted when serum is electrophoresed (eg, dysbetalipoproteinemia) or from combinations of lipid abnormalities in families (eg, familial combined hyperlipidemia). Thus, family members of patients with severe lipid disorders are appropriately studied. Other patients have abnormalities in the production of apoproteins, such as increased apoprotein B and its affiliated lipoproteins, LDL and VLDL; reduced apoprotein AII and its affiliated particle; or excess lipoprotein(a). Other mutations occur in lipoprotein lipase and in the gene encoding for cholesterol efflux regulatory protein.
Berneis K et al: Low-density lipoprotein size and subclasses are markers of clinically apparent and non-apparent atherosclerosis in type 2 diabetes. Metabolism 2005;54:227.
Carmena R et al: Atherogenic lipoprotein particles in atherosclerosis. Circulation 2004;109:III2.
Deb A et al: Lipoprotein(a): new insights into mechanisms of atherogenesis and thrombosis. Clin Cardiol 2004;27:258.
Drexel H et al: Is atherosclerosis in diabetes and impaired fasting glucose driven by elevated LDL cholesterol or by decreased HDL cholesterol? Diabetes Care 2005;28:101.
Marks D et al: A review on the diagnosis, natural history, and treatment of familial hypercholesterolaemia. Atherosclerosis 2003;168:1.
Moreno PR et al: New aspects in the pathogenesis of diabetic atherothrombosis. J Am Coll Cardiol 2004;44:2293.
Nam BH et al: Search for the optimal atherogenic lipid risk profile: from the Framingham study. Am J Cardiol 2006;97: 372.
Rader DJ: Regulation of reverse cholesterol transport and clinical implications. Am J Cardiol 2003;92:42J.
Lipid Fractions & The Risk of Coronary Heart Disease
In fasting serum, cholesterol is carried primarily on three different lipoproteins the VLDL, LDL, and HDL molecules. Total cholesterol equals the sum of these three components:
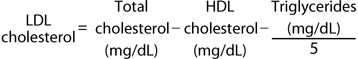
Most clinical laboratories measure the total cholesterol, the total triglycerides, and the amount of cholesterol found in the HDL fraction, which is easily precipitated from serum. Most triglyceride is found in VLDL particles, which contain five times as much triglyceride by weight as cholesterol. The amount of cholesterol found in the VLDL fraction can be estimated by dividing the triglyceride by 5:
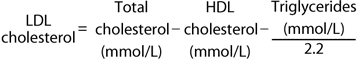
Because the triglyceride level is used as a proxy for the amount of VLDL, this formula works only in fasting samples and when the triglyceride level is less than 400 mg/dL. At higher triglyceride levels, LDL and VLDL cholesterol levels can be determined after ultracentrifugation or by direct chemical measurement.
The total cholesterol is reasonably stable over time; however, measurements of HDL and especially triglycerides may vary considerably because of analytic error in the laboratory and biologic variation in a patient's lipid level. Thus, the LDL should always be estimated as the mean of at least two determinations; if those two estimates differ by more than 10%, a third lipid profile is obtained and is estimated as follows:
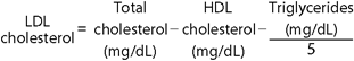
When using SI units, the formula becomes
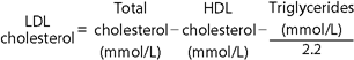
Understanding the relationships of the different lipid fractions leads to a more accurate understanding of a patient's lipid-related coronary risk than the total cholesterol. Two persons with the same total cholesterol
P.1268
of 275 mg/dL may have very different lipid profiles. One may have an HDL cholesterol of 110 mg/dL with a triglyceride of 150 mg/dL, giving an estimated LDL cholesterol of 135 mg/dL; the other may have an HDL cholesterol of 25 mg/dL with a triglyceride of 200 mg/dL and an LDL cholesterol of 210 mg/dL. The second would have more than a tenfold higher CHD risk than the first, assuming no differences in other factors. Because of high HDL cholesterol levels in women, many with apparently high total cholesterol levels have favorable lipid profiles. Thus, evaluation of the lipid fractions is essential before therapy is initiated.
Some authorities use the ratio of the total to HDL cholesterol as an indicator of lipid-related coronary risk: the lower this ratio is, the better. (In the example above, the first person would have a ratio of 275 110 = 2.5, while the second would have a much less favorable ratio of 275 25 = 11.) Although ratios are useful predictors within populations of patients, they may obscure important information in individual patients. (A total cholesterol of 300 mg/dL and an HDL of 60 mg/dL result in the same ratio of 5 as a total cholesterol of 150 mg/dL with an HDL of 30 mg/dL.) Moreover, errors in the measurement of HDL cholesterol are common in many laboratories, and the total cholesterol-to-HDL cholesterol ratio magnifies their importance.
There is no true normal range for serum lipids. In Western populations, cholesterol values are about 20% higher than in Asian populations and exceed 300 mg/dL in nearly 5% of adults. About 10% of adults have LDL cholesterol levels above 200 mg/dL. Total and LDL cholesterol levels tend to rise with age in persons who are otherwise in good health.
Declines are seen in acute illness, and lipid studies in such patients are of little value with the exception of the serum triglyceride level in a patient with pancreatitis. Cholesterol levels (even when expressed as an age-matched percentile rank, such as the highest 20%) do not remain constant over time, especially from childhood through adolescence and young adulthood. Thus, children and young adults with relatively high cholesterol may have lower levels later in life, whereas those with low cholesterol may show increases.
Therapeutic effects of Lowering Cholesterol
Most studies of the effect of cholesterol lowering have distinguished between primary and secondary prevention. Primary prevention trials enroll healthy subjects who have relatively low rates of coronary disease but in whom other causes of morbidity and mortality are proportionately more common. Secondary prevention trials, on the other hand, follow patients who have a high rate of subsequent coronary disease; other causes of mortality are relatively less important.
Reducing cholesterol levels in healthy middle-aged men without CHD (primary prevention) reduces their risk in proportion to the reduction in LDL cholesterol and the increase in HDL cholesterol. Treated patients have statistically significant and clinically important reductions in the rates of myocardial infarctions, new cases of angina, and need for coronary artery bypass procedures. The West of Scotland Study showed a 31% decrease in myocardial infarctions in middle-aged men treated with pravastatin compared with placebo. The Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS) study showed similar results with lovastatin. As with any primary prevention interventions, large numbers of healthy patients need to be treated to prevent a single event. The numbers of patients needed to treat (NNT) to prevent a nonfatal myocardial infarction or a coronary artery disease death in these two studies were 46 and 50, respectively. The Anglo-Scandinavian Cardiac Outcomes Trial (ASCOT) study of atorvastatin in subjects with hypertension and other risk factors but without CHD also demonstrated a convincing 36% reduction in CHD events.
Primary prevention studies have found a less consistent effect on total mortality. The West of Scotland study found a 20% decrease in total mortality, tending toward statistical significance. The AFCAPS/TexCAPS study with lovastatin showed no difference in total mortality. The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT-LLT) also showed no reduction either in all-cause mortality or in CHD events when pravastatin was compared with usual care. Subjects treated with atorvastatin in the ASCOT study had a 13% reduction in mortality, but the result was not statistically significant. This study, however, was stopped early due to the marked reduction in CHD events.
In patients with CHD, the benefits of cholesterol lowering are more clear. Major studies with statins have shown significant reductions in cardiovascular events, cardiovascular deaths, and all-cause mortality in men and women with coronary artery disease. The NNT to prevent a nonfatal myocardial infarction or a coronary artery disease death in these three studies were between 12 and 34. Aggressive cholesterol lowering with these agents causes regression of atherosclerotic plaques in some patients, reduces the progression of atherosclerosis in saphenous vein grafts, and can slow or reverse carotid artery atherosclerosis. Meta-analysis suggests that this latter effect results in a significant decrease in strokes. Results with other classes of medications have been less consistent. For example, gemfibrozil treatment subjects had fewer cardiovascular events, but there was no benefit in all-cause mortality when compared with placebo.
The disparities in results between primary and secondary prevention studies highlight several important points. The benefits and adverse effects of cholesterol lowering may be specific to each type of drug; the clinician cannot assume that the effects will generalize to other classes of medication. Second, the net benefits from cholesterol lowering depend on the underlying risk of CHD and of other disease. In patients with atherosclerosis,
P.1269
morbidity and mortality rates associated with CHD are high, and measures that reduce it are more likely to be beneficial even if they have no effect or even slightly harmful effects on other diseases.
Ashen MD et al: Low HDL cholesterol levels. N Engl J Med 2005;353:1252.
Baigent C et al: Efficacy and safety of cholesterol-lowering treatment: prospective meta-analysis of data from 90,056 participants in 14 randomised trials. Lancet 2005;366:1267.
Cannon CP et al: Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004; 350:1495.
Corvol JC et al: Differential effects of lipid-lowering therapies on stroke prevention: a meta-analysis of randomized trials. Arch Intern Med 2003;163:669.
de Lemos JA et al: Early intensive vs. a delayed conservative simvastatin strategy in patients with acute coronary syndromes: phase Z of the A to Z trial. JAMA 2004;292:1307.
Grundy SM et al for the Coordinating Committee of the NCEP: Implications of recent clinical trials for the national cholesterol education program adult treatment panel III guidelines. Circulation 2004;110:227.
Larosa JC et al: Intensive lipid lowering with atorvastatin in patients with stable coronary disease. N Engl J Med 2005; 352:1425.
Sever PS et al: Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than-average cholesterol concentrations, in the Anglo-Scandinavian Cardiac Outcomes Trial-Lipid Lowering Arm (ASCOT-LLA): a multicentre randomized controlled trial. Lancet 2003;361:1149.
Whitney EJ et al: A randomized trial of a strategy for increasing high-density lipoprotein cholesterol levels: effects of progression of coronary heart disease and clinical events. Ann Intern Med 2005;142:95.
Secondary Conditions that affect Lipid Metabolism
Several factors, including drugs, can influence serum lipids (Table 28-1). These are important for two reasons: Abnormal lipid levels (or changes in lipid levels) may be the presenting sign of some of these conditions, and correction of the underlying condition may obviate the need to treat an apparent lipid disorder. Diabetes and alcohol use, in particular, are commonly associated with high triglyceride levels that decline with improvements in glycemic control or reduction in alcohol use, respectively. Thus, secondary causes of high blood lipids should be considered in each patient with a lipid disorder before lipid-lowering therapy is started. In most instances, special testing is not needed: a history and physical examination are sufficient. However, screening for hypothyroidism in patients with hyperlipidemia is cost effective.
Table 28-1. Secondary causes of lipid abnormalities. | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||
Clinical Presentations
Most patients with high cholesterol levels have no specific symptoms or signs. The vast majority of patients with lipid abnormalities are detected by the laboratory, either as part of the workup of a patient with cardiovascular disease or as part of a preventive screening strategy. Extremely high levels of chylomicrons or VLDL particles (triglyceride level above 1000 mg/dL) result in the formation of eruptive xanthomas (red-yellow papules, especially on the buttocks). High LDL concentrations result in tendinous xanthomas on certain tendons (Achilles, patella, back of the hand). Such xanthomas usually indicate one of the underlying genetic hyperlipidemias. Lipemia retinalis (cream-colored blood vessels in the fundus) is seen with extremely high triglyceride levels (above 2000 mg/dL).
Screening for High Blood Cholesterol
All patients with CHD or CHD risk equivalents (other clinical forms of atherosclerosis such as peripheral artery disease, abdominal aortic aneurysm, and symptomatic carotid artery disease; patients with diabetes mellitus; and patients with multiple risk factors that confer a greater than 20% 10-year risk for developing CHD) should be screened for elevated lipids. The only exceptions are patients in whom lipid lowering is not indicated or desirable for other reasons. Patients
P.1270
who already have evidence of atherosclerosis are the group at highest risk of suffering additional manifestations in the near term and thus have the most to gain from reduction of blood lipids. Additional risk reduction measures for atherosclerosis are discussed in Chapter 10; lipid lowering should be just one aspect of a program to reduce the progression and effects of the disease.
In patients with cardiovascular disease, a complete lipid profile (total cholesterol, HDL cholesterol, and triglyceride levels) after an overnight fast should be obtained as a screening test. Those whose estimated LDL cholesterol level is high should have at least one repeat measurement. Specific treatments for high LDL cholesterol levels are discussed below. The goal of therapy should be to reduce the LDL cholesterol to below 100 mg/dL or optimally to below 70 mg/dL. Recent evidence suggests that treatment with a statin is effective even if the starting LDL cholesterol is below 100 mg/dL. These data suggest that most patients with CHD or CHD risk equivalents should be treated with statin therapy.
The best screening and treatment strategy for adults who do not have atherosclerotic cardiovascular disease is less clear. Several algorithms have been developed to guide the clinician in treatment decisions, but management decisions are individualized based on the patient's risk.
Although the National Cholesterol Education Program (NCEP) recommends screening of all adults aged 20 years or older for high blood cholesterol, the United States Preventive Services Task Force (USPSTF) suggests beginning at age 35 years in men and age 45 years in women unless there are other risk factors for CHD. This strategy focuses cholesterol screening on those at most immediate risk of coronary artery disease and increases the cost effectiveness of cholesterol screening.
Individuals without cardiovascular disease can then be stratified according to risk factors as defined by the NCEP. Those with two or more risk factors are considered to be at intermediate risk of coronary artery disease, and those with less than two are at low risk. These include age and gender (men aged 45 years or older, women aged 55 years or older), a family history of premature CHD (myocardial infarction or sudden cardiac death before age 55 years in a first-degree male relative or before age 65 years in a first-degree female relative), hypertension (whether treated or not), current cigarette smoking (10 or more cigarettes per day), and low HDL cholesterol (< 40 mg/dL). Because HDL cholesterol is protective against CHD, a risk factor is subtracted if the level is greater than 60 mg/dL. Patients with two or more risk factors are then further stratified by evaluating their 10-year risk of developing CHD using Framingham projections of 10-year risk (Table 28-2). Because risk factors alone are an imprecise measure of CHD risk, estimating the 10-year risk using Framingham data is likely to be helpful even in patients with one or no risk factors.
Several strategies for obtaining the initial cholesterol measurement have been proposed, including (1) measuring total cholesterol alone, (2) measuring total cholesterol and HDL cholesterol, or (3) measuring LDL and HDL cholesterol. Each is acceptable, but treatment decisions are based on the LDL and HDL cholesterol levels. Measurement of the total cholesterol alone is the least expensive strategy and is adequate for low-risk individuals; those with total cholesterol greater than 200 mg/dL should then be reevaluated with a fasting LDL and HDL cholesterol measurement. Measurement of the total cholesterol and HDL cholesterol allows for better characterization of the risk factor profile but also requires reevaluation if the total cholesterol is greater than 200 mg/dL as recommended by the USPSTF. Initial measurement of the LDL and HDL cholesterol is least likely to lead to patient misinformation and misclassification and is the strategy recommended by the NCEP.
Treatment decisions are based on the LDL cholesterol, and the patient's risk factor profile (including the HDL cholesterol level) and estimated 10-year risk. Patients in the intermediate-risk group (two or more risk factors) are selected for diet therapy (therapeutic lifestyle changes) if LDL cholesterol is greater than 130 mg/dL. If the 10-year risk of CHD is < 10%, drug treatment is recommended if LDL is > 160 mg/dL; if the 10-year CHD risk is between 10% and 20%, drug treatment is recommended if LDL is > 130 mg/dL. Low-risk individuals with one or no risk factors and estimated 10-year CHD risk less than 10% are selected for diet therapy if LDL cholesterol is greater than 160 mg/dL and for drug therapy if it is greater than 190 mg/dL (Table 28-3).
Screening in Women
The foregoing screening and treatment guidelines, based largely on LDL cholesterol levels, are designed for both men and women. Yet several observational studies suggest that a low HDL cholesterol is a more important risk factor for CHD in women than a high LDL cholesterol. Meta-analysis of studies including women with known heart disease, however, has found that medications that primarily lower LDL cholesterol do prevent recurrent myocardial infarctions in women. There is insufficient evidence to be certain of a similar effect from LDL-lowering therapy in women without evidence of CHD. Although most experts recommend application of the same primary prevention guidelines for women as for men, clinicians should be aware of the uncertainty in this area. Using estimates of 10-year CHD risk may be particularly helpful in women since a larger percentage of women than men will have estimated 10-year CHD risks below 10% per year and thus women are less likely to benefit from therapy unless their LDL cholesterol is extremely high (greater than 190 mg/dL).
Screening in Older Patients
Meta-analysis of evidence relating cholesterol to CHD in the elderly suggests that cholesterol is not a risk factor
P.1271
P.1272
for CHD for persons over age 75 years. Clinical trials have rarely included such individuals. One exception is the Prospective Study of Pravastatin in the Elderly at Risk (PROSPER). In this study, elderly patients with cardiovascular disease (secondary prevention) benefited from statin therapy, whereas those without cardiovascular disease (primary prevention) did not. Although the NCEP recommends continuing treatment in the elderly, many clinicians will prefer to stop screening and treatment in patients age 75 years or older who do not have CHD. In patients age 75 years or older who have CHD, LDL-lowering therapy can be continued as recommended for younger patients with the disease. Decisions to discontinue therapy should be based on overall functional status and life expectancy, comorbidities, and patient preference and should be made in context with overall therapeutic goals and end-of-life decisions.
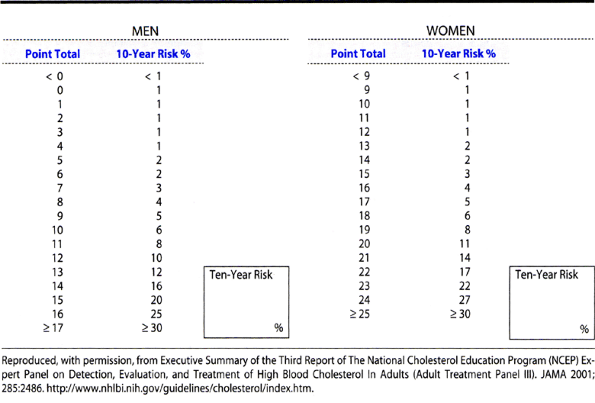 |
Table 28-2. Framingham 10-year coronary heart disease risk projections. Calculate the number of points for each risk factor. Sum the total risk score and estimate the 10-year risk. |
Ballantyne CM et al: Role of lipid and lipoprotein profiles in risk assessment and therapy. Am Heart J 2003;146:227.
Grundy SM et al, for the Coordinating Committee of the NCEP: Implications of recent clinical trials for the national cholesterol education program adult treatment panel III guidelines. Circulation 2004;110:227.
Mora S et al: Justification for the use of statins in primary prevention: An intervention trial evaluating rosuvastatin (JUPITER)-Can C-reactive protein be used to target statin therapy in primary prevention? Am J Cardiol 2006;97:33.
Mosca L et al: Evidence-based guidelines for cardiovascular disease in women. Circulation 2004;109:672.
Nissen SE: Halting the progression of atherosclerosis with intensive lipid lowering: results from the reversal of atherosclerosis with aggressive lipid lowering (REVERSAL) trial. Am J Med 2005;118 Suppl 12A:22.
O'Keefe JH Jr et al: Optimal low-density lipoprotein is 50 to 70 mg/dl: lower is better and physiologically normal. J Am Coll Cardiol 2004;43:2142.
Pletcher M et al: Primary prevention of cardiovascular disease in women: New guidelines and emerging strategies. Adv Studies in Medicine 2005;5:412.
Sabharwal AK et al: Low-density lipoprotein reduction: Is the risk worth the benefit? Curr Atheroscleros Rep 2006;8:19.
Sepulveda JL et al: C-reactive protein and cardiovascular disease: a critical appraisal. Curr Opin Cardiol 2005;20:407.
Walsh JM et al: Drug treatment of hyperlipidemia in women. JAMA 2004;291:2243.
Treatment of High Low-Density Lipoprotein Cholesterol
Reduction of LDL cholesterol is just one part of a program to reduce the risk of cardiovascular disease. Other measures including smoking cessation, hypertension
P.1273
control, and aspirin are also of central importance. Less well studied but of potential value is raising the HDL cholesterol level. Quitting smoking reduces the effect of other cardiovascular risk factors (such as a high cholesterol level); it may also increase the HDL cholesterol level. Exercise (and weight loss) may reduce the LDL cholesterol and increase the HDL. Modest alcohol use (1 2 ounces a day) also raises HDL levels and appears to have a salutary effect on CHD rates. Although the clinician may not wish to recommend alcohol use to patients, its use in moderation need not be discouraged.
Table 28-3. LDL goals and treatment cutpoints: recommendations of the NCEP Adult Treatment Panel III. | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
Diet Therapy
Studies of nonhospitalized adults have reported only modest cholesterol-lowering benefits of dietary therapy, typically in the range of a 5 10% decrease in LDL cholesterol, with even less in the long term. The effect of diet therapy, however, varies considerably among individuals, as some patients will have striking reductions in LDL cholesterol up to a 25 30% decrease whereas others will have clinically important increases. Thus, the results of diet therapy should be assessed about 4 weeks after initiation.
Cholesterol-lowering diets may also have a variable effect on lipid fractions. Diets very low in total fat or in saturated fat may lower HDL cholesterol as much as LDL cholesterol. It is not known how these diet-induced changes affect coronary risk.
Several nutritional approaches to diet therapy are available. Most Americans currently eat over 35% of calories as fat, of which 15% is saturated fat. Dietary cholesterol intake averages 400 mg/d. A cholesterol-lowering
P.1274
diet recommends reducing total fat to 25 30% and saturated fat to less than 7% of calories. Dietary cholesterol should be limited to less than 200 mg/d. These diets replace fat, particularly saturated fat, with carbohydrate. In most instances, this approach will also result in fewer total calories consumed and will facilitate weight loss in overweight patients. Other diet plans, including the Dean Ornish Diet, the Pritikin Diet, and most vegetarian diets, restrict fat even further. Low-fat, high-carbohydrate diets may, however, result in reductions in HDL cholesterol.
An alternative strategy is the Mediterranean diet, which maintains total fat at approximately 35 40% of total calories but replaces saturated fat with monounsaturated fat such as that found in canola oil and in olives, peanuts, avocados, and their oils. This diet is equally effective at lowering LDL cholesterol but is less likely to lead to reductions in HDL cholesterol. Recent studies have demonstrated that this approach may also be associated with reductions in endothelial dysfunction, insulin resistance, and markers of vascular inflammation and may result in better resolution of the metabolic syndrome than traditional cholesterol-lowering diets.
Other dietary changes may also result in beneficial changes in blood lipids. Soluble fiber, such as that found in oat bran or psyllium, may reduce LDL cholesterol by 5 10%. Garlic, soy protein, vitamin C, pecans, and plant sterols may also result in reduction of LDL cholesterol. Because oxidation of LDL cholesterol is a potential initiating event in atherogenesis, diets rich in antioxidant vitamins, found primarily in fruits and vegetables, may be helpful (see Chapter 29). Studies have suggested that when all of these elements are combined into a single dietary prescription, the impact of diet on LDL cholesterol may approach that of statin medications, lowering LDL cholesterol by close to 30%.
Anderson JW: Diet first, then medication for hypercholesterolemia. JAMA 2003;290:531.
Brunner E et al: Dietary advice for reducing cardiovascular risk. Cochrane Database Syst Rev 2005;(4):CD002128.
Ding EL et al: Chocolate and prevention of cardiovascular disease: A systematic review. Nutr Metab 2006;3:2.
Gardner CD et al: The effect of a plant-based diet on plasma lipids in hypercholesterolemic adults: a randomized trial. Ann Intern Med 2005;142:725.
Jenkins DJ et al: Direct comparison of a dietary portfolio of cholesterol-lowering foods with a statin in hypercholesterolemic participants. Am J Clin Nutr 2005;81:380.
Kelley GA et al: Exercise, lipids, and lipoproteins in older adults: a meta-analysis. Prev Cardiol 2005;8:206.
Maron DJ et al: Cholesterol-lowering effect of a theaflavin enriched green tea extract: a randomized controlled trial. Arch Intern Med 2003;163:1448.
Miettinen TA et al: Plant stanol and sterol esters in prevention of cardiovascular diseases. Ann Med 2004;36:126.
Moreyra AE et al: Effect of combining psyllium fiber with simvastatin in lowering cholesterol. Arch Intern Med 2005;165: 1161.
Mukuddem-Petersen J et al: A systematic review of the effects of nuts on blood lipid profiles in humans. J Nutr 2005;135: 2082.
Pignone MP et al: Counseling to promote a healthy diet in adults: a summary of the evidence for the U.S. Preventive Services Task Force. Am J Prev Med 2003;24:75.
Sacks FM et al: Soy protein, isoflavones, and cardiovascular health. An American Heart Association Science Advisory for professionals from the Nutrition Committee. Circulation 2006;113:1034.
Pharmacologic Therapy
All patients whose risk from CHD is considered high enough to warrant pharmacologic therapy of an elevated LDL cholesterol should be given aspirin prophylaxis at a dose of 81 mg/d unless there are contraindications such as aspirin sensitivity, bleeding diatheses, or active peptic ulcer disease. The benefit of aspirin in reducing the risk of CHD is equivalent to that of cholesterol lowering. Other CHD risk factors, such as hypertension and smoking, should also be controlled.
If the decision to treat a patient with an LDL-lowering drug is made, a goal for treatment is set. The National Cholesterol Education Program's Adult Treatment Panel III (NCEP ATP III) published revised, more intensive LDL treatment goals in 2004. For patients with CHD or CHD risk equivalents, the goal is LDL < 100 mg/dL, but for patients with very high risk, a goal of LDL < 70 mg/dL is a therapeutic option (see Table 28-3). This goal may also be appropriate for patients with very high risk who have a baseline LDL < 100 mg/dL. For patients with two or more risk factors and a 10-year CHD risk of 10 20%, the recommended goal is LDL < 130 mg/dL, but a goal of < 100 mg/dL is optional. For those with two or more risk factors and a 10-year CHD risk of < 10%, the goal is < 130 mg/dL. For those with zero or one risk factor, the goal is LDL < 160 mg/dL (see Table 28-3). In each instance, the therapeutic goal is approached slowly, watching for side effects and encouraging continued adherence to nonpharmacologic therapies. In most instances, intensity of therapy should be sufficient to achieve a 30 40% reduction in LDL cholesterol. Combinations of drugs may be necessary. Once the goal is reached, the lipid profile should be monitored periodically (every 6 12 months), with consideration given to periodic reductions in drug dose. Most lipid-lowering agents are expensive and may need to be given for decades. Thus, their cost effectiveness is low for some groups of patients, especially in primary prevention.
A. Niacin (Nicotinic Acid)
Niacin was the first lipid-lowering agent that was associated with a reduction in total mortality. Long-term follow-up of a secondary prevention trial of middle-aged men with previous myocardial infarction disclosed that about half of those who had been previously treated with niacin had died, compared with nearly 60% of the placebo group. This favorable effect on mortality was not seen during the trial itself,
P.1275
though there was a reduction in the incidence of recurrent coronary events.
Niacin reduces the production of VLDL particles, with secondary reduction in LDL and increases in HDL cholesterol levels. Studies have identified a niacin receptor in both adipocytes and hepatocytes. Niacin binding leads to reduction of lipolysis of triglycerides in adipocytes, reduced synthesis of triglycerides in the liver, and reduced hepatic production of VLDL. The average effect of full-dose niacin therapy, 3 4.5 g/d, is a 15 25% reduction in LDL cholesterol and a 25 35% increase in HDL cholesterol. Full doses are required to obtain the LDL effect, but the HDL effect is observed at lower doses, eg, 1 g/d. Niacin will also reduce triglycerides by half and will lower lipoprotein(a) (Lp[a]) levels and will increase plasma homocysteine levels. Thus, its effect on blood lipids and CHD risk is nearly optimal. Intolerance to niacin is common; only 50 60% of patients can take full doses. Niacin causes a prostaglandin-mediated flushing that patients may describe as hot flashes or pruritus and that can be decreased with aspirin (81 325 mg/d) or other nonsteroidal anti-inflammatory agents taken during the same day. Flushing may also be decreased by initiating niacin therapy with a very small dose, eg, 100 mg with the evening meal. The dose can be doubled each week until 1.5 g/d is tolerated. After rechecking blood lipids, the dose is divided and increased until the goal of 3 4.5 g/d is reached. Extended-release niacin is also available and is better tolerated by most patients. It is not known whether routine monitoring of liver enzymes results in early detection and thus reduced severity of this side effect. Niacin can also exacerbate gout and peptic ulcer disease. Although niacin may increase blood sugar in some patients, clinical trials have shown that niacin can be safely used in diabetics.
B. Bile Acid-Binding Resins
The bile acid-binding resins include cholestyramine, colesevelam, and colestipol. Treatment with these agents reduces the incidence of coronary events in middle-aged men by about 20%, with no significant effect on total mortality. The resins work by binding bile acids in the intestine. The resultant reduction in the enterohepatic circulation causes the liver to increase its production of bile acids, using hepatic cholesterol to do so. Thus, hepatic LDL receptor activity increases, with a decline in plasma LDL levels. The triglyceride level tends to increase slightly in some patients treated with bile acid-binding resins; they should be used with caution in those with elevated triglycerides and probably not at all in patients who have triglyceride levels above 500 mg/dL. The clinician can anticipate a reduction of 15 25% in the LDL cholesterol level, with insignificant effects on the HDL level.
The usual dose of cholestyramine is 12 36 g of resin per day in divided doses with meals, mixed in water or, more palatably, juice. Doses of colestipol are 20% higher (each packet contains 5 g of resin). The dose of colesevelam is 625 mg, 6 7 tablets per day.
These agents often cause gastrointestinal symptoms, such as constipation and gas. They may interfere with the absorption of fat-soluble vitamins (thereby complicating the management of patients receiving warfarin) and may bind other drugs in the intestine. Concurrent use of psyllium may ameliorate the gastrointestinal side effects.
C. Hydroxymethylglutaryl-Coenzyme A (HMG-CoA) Reductase Inhibitors (Statins)
The HMG-CoA reductase inhibitors include atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, and simvastatin. These agents work by inhibiting the rate-limiting enzyme in the formation of cholesterol. They reduce myocardial infarctions and total mortality in secondary prevention, as well as in older middle-aged men free of CHD. A meta-analysis has demonstrated significant reduction in risk of stroke. Cholesterol synthesis in the liver is reduced, with a compensatory increase in hepatic LDL receptors (presumably so that the liver can take more of the cholesterol that it needs from the blood) and a reduction in the circulating LDL cholesterol level by up to 35%. There are also modest increases in HDL levels and decreases in triglyceride levels.
Oral doses are as follows: atorvastatin, 10 80 mg/d; fluvastatin, 20 40 mg/d; lovastatin, 10 80 mg/d; pravastatin, 10 40 mg/d; rosuvastatin, 5 40 mg/d; and simvastatin, 5 40 mg/d. These agents are usually given once a day in the evening (most cholesterol synthesis takes place overnight); at the high end of the dose ranges, twice-a-day dosing may be used. Side effects include myositis, whose incidence may be higher in patients concurrently taking fibrates or niacin. Manufacturers recommend monitoring liver and muscle enzymes. Several agents (notably erythromycin, cyclosporine, and azole antifungals) reduce the metabolism of these agents.
D. Fibric Acid Derivatives
The fibric acid derivatives or fibrates approved for use in the United States are gemfibrozil and fenofibrate. Ciprofibrate and bezafibrate are also available for use internationally. Gemfibrozil reduced CHD rates in hypercholesterolemic middle-aged men free of coronary disease in the Helsinki Heart Study. The effect was observed only among those who also had lower HDL cholesterol levels and high triglyceride levels. In a recent VA study, gemfibrozil was also shown to reduce cardiovascular events in men with existing CHD whose primary lipid abnormality was a low HDL cholesterol. There was no effect on all-cause mortality.
The fibrates are peroxisome proliferative-activated receptor-alpha (PPAR-alpha) agonists that result in potent reductions of plasma triglycerides and increases in HDL cholesterol. They reduce LDL levels by about 10 15%, although the result is quite variable, and triglyceride
P.1276
levels by about 40% and raise HDL levels by about 15 20%. The usual dose of gemfibrozil is 600 mg once or twice a day. Side effects include cholelithiasis, hepatitis, and myositis. The incidence of the latter two conditions may be higher among patients also taking other lipid-lowering agents. In the largest clinical trial that used clofibrate, there were significantly more deaths especially due to cancer in the treatment group; it should not be used.
E. Ezetimibe
Ezetimibe is a lipid-lowering drug that inhibits the intestinal absorption of dietary and biliary cholesterol by blocking passage across the intestinal wall by inhibiting a newly discovered cholesterol transporter. Ezetimibe reduces LDL cholesterol between 15% and 20% when used as monotherapy and can further reduce LDL in patients taking statins who are not yet at therapeutic goal. The effects of ezetimide on CHD and its long-term safety are not yet known. The usual dose of ezetimibe is 10 mg/d orally.
Initial Selection of Medication
At present there are no absolute guidelines for selection of available lipid-modifying medications in particular patients. Nonetheless, clinical trials provide guidance (Table 28-4). For most patients who require a lipid-modifying medication, an HMG-CoA reductase inhibitor is preferred. Although niacin will also have beneficial effects on lipids in both men and women with CHD, there is less evidence demonstrating the desired effects on CHD and all-cause mortality. Resins are the only lipid-modifying medication considered safe in pregnancy.
Combinations of lipid-modifying medications may be more cost-effective than high doses of a single medication (usually an HMG-CoA reductase inhibitor) and may have beneficial effects on lipids. Combination therapy may also be needed to meet lower NCEP targets in some patients or to achieve other therapeutic goals. Low-dose niacin (0.5 1 g/d), for example, will substantially increase the HDL cholesterol when added to an HMG-CoA reductase inhibitor. Combinations, however, may increase the risk of complications of drug therapy. The combination of gemfibrozil and HMG-CoA reductase inhibitors increases the risk of myopathy more than either drug alone.
Birjmohun RS et al: Efficacy and safety of high-density lipoprotein cholesterol-increasing compounds: a meta-analysis of randomized controlled trials. J Am Coll Cardiol 2005;45: 185.
Brewer HB Jr: Increasing HDL cholesterol levels. N Engl J Med 2004;350:1491.
Brousseau ME et al: Effects of an inhibitor of cholesteryl ester transfer protein on HDL cholesterol. N Engl J Med 2004; 350:1505.
Bruckert E et al: Perspectives in cholesterol-lowering therapy: the role of ezetimibe, a new selective inhibitor of intestinal cholesterol absorption. Circulation 2003;107:3124.
Chapman MJ et al: Raising high-density lipoprotein cholesterol with reduction of cardiovascular risk: the role of nicotinic acid a position paper developed by the European Consensus Panel on HDL-C. Curr Med Res Opin 2004;20:1253.
Dale KM et al: Statins and cancer risk: a meta-analysis. JAMA 2006;295:74.
Ferdinand KC: The importance of aggressive lipid management in patients at risk: evidence from recent clinical trials. Clin Cardiol 2004;27(6 Suppl 3):III12.
Grundy SM et al: Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J Am Coll Cardiol 2004;44:720.
Kwiterovich PO: A review of lipid-modifying drugs used alone or in combination. Adv Stud Med 2005;5:475.
Miller M: Intensive versus moderate lipid lowering with statins after acute coronary syndromes. N Engl J Med 2004;351:714.
Mozaffarian D et al: Statin therapy is associated with lower mortality among patients with severe heart failure. Am J Cardiol 2004;93:1124.
Nissen SE et al: Effect of intensive compared with moderate lipid-lowering therapy on progression of coronary atherosclerosis: a randomized controlled trial. JAMA 2004;291:1071.
Thompson PD et al: Statin-associated myopathy. JAMA 2003; 289:1681.
Whitney EJ et al: A randomized trial of a strategy for increasing high-density lipoprotein cholesterol levels: effects on progression of coronary heart disease and clinical events. Ann Intern Med 2005;142:95.
Wilt TJ et al: Effectiveness of statin therapy in adults with coronary heart disease. Arch Intern Med 2004;164:1427.
High Blood Triglycerides
Patients with very high levels of serum triglycerides are at risk for pancreatitis. The pathophysiology is not certain, since pancreatitis never develops in some patients with very high triglyceride levels. Most patients with congenital abnormalities in triglyceride metabolism present in childhood; hypertriglyceridemia-induced pancreatitis first presenting in adults is more commonly due to an acquired problem in lipid metabolism.
Although there are no clear triglyceride levels that predict pancreatitis, most clinicians are uncomfortable with fasting levels above 500 mg/dL. The risk of pancreatitis may be more related to the triglyceride level following consumption of a fatty meal. Because postprandial increases in triglyceride are inevitable if fat-containing foods are eaten, fasting triglyceride levels in persons prone to pancreatitis should be kept well below that level.
The primary therapy for high triglyceride levels is dietary, avoiding alcohol, simple sugars, refined starches, saturated and trans fatty acids, and restricting total calories. Control of secondary causes of high triglyceride levels (see Table 28-1) may also be helpful. In patients with fasting triglycerides 500 mg/dL despite adequate dietary compliance and certainly in those with a previous
P.1277
episode of pancreatitis therapy with a triglyceride-lowering drug (eg, niacin, a fibric acid derivative, or an HMG-CoA reductase inhibitor) is indicated.
Table 28-4. Effects of selected lipid-modifying drugs. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Whether patients with elevated triglycerides (> 150 mg/dL) should be treated to prevent CHD is not known. Meta-analysis of 17 observational studies suggests that after adjustment for other risk factors, elevated triglycerides increased CHD risk in men by 14% and in women by 37%. Triglyceride-rich lipoproteins (partially degraded VLDL, commonly called remnant lipoproteins) have been found in human atheromas, and elevated triglycerides are associated with small dense LDL in most instances. Elevated triglycerides are also an important feature of the metabolic syndrome, found in an estimated 25% of Americans defined by three or more of the following five abnormalities: waist circumference > 102 cm in men or > 88 cm in women, serum triglyceride level of at least 150 mg/dL, HDL level of < 40 mg/dL in men or < 50 mg/dL in women, blood pressure of at least 130/85 mm Hg, and serum glucose level of at least 110 mg/dL. Other data, however, suggest that triglyceride measurements do not improve discrimination between those with and without CHD events, and clinical trial data are not available to support the routine treatment of high triglycerides in all patients.
The NCEP ATP III guidelines, however, recommend an aggressive approach to triglyceride management. For those with borderline levels (150 199 mg/dL), emphasis is placed on calorie restriction and exercise. For patients with high triglycerides (> 200 mg/dL), the non-HDL cholesterol should be measured (total cholesterol HDL cholesterol). The ATP III report recommends that non-HDL cholesterol should be treated with diet and medications to result in levels 30 mg/dL higher than the LDL goal. The ATP III report
P.1278
does not differentiate between primary and secondary prevention. It is reasonable to use this approach for patients with CHD and risk equivalents for that disease but not for lower-risk patients.
Garber AJ: The metabolic syndrome. Med Clin North Am 2004; 88:837.
Katzmarzyk PT et al: Metabolic syndrome, obesity, and mortality: impact of cardiorespiratory fitness. Diabetes Care 2005; 28:391.
Moller DE et al: Metabolic syndrome: a clinical and molecular perspective. Annu Rev Med 2005;56:45.
Park Y et al: The metabolic syndrome: prevalence and associated risk factor findings in the US population from the Third National Health and Nutrition Examination Survey, 1988 1994. Arch Intern Med 2003;163:427.
Szapary PO et al: The triglyceride-high-density lipoprotein axis: an important target of therapy? Am Heart J 2004;148:211.
EAN: 2147483647
Pages: 49