26 - Endocrinology
Editors: McPhee, Stephen J.; Papadakis, Maxine A.; Tierney, Lawrence M.
Title: Current Medical Diagnosis & Treatment, 46th Edition
Copyright 2007 McGraw-Hill
> Table of Contents > 29 - Nutrition
function show_scrollbar() {}
29
Nutrition
Robert B. Baron MD, MS
Nutritional Requirements
Approximately 40 nutrients are required by the human body. Nutrients are essential if they cannot be synthesized by the body and if a deficiency causes recognizable abnormalities that disappear when the deficit is corrected. Required nutrients include the essential amino acids, water-soluble vitamins, fat-soluble vitamins, minerals, and the essential fatty acids. The body also requires an adequate energy substrate, a small amount of metabolizable carbohydrate, indigestible carbohydrate (fiber), additional nitrogen, and water.
Nutritional requirements have been most commonly expressed by recommended dietary allowances (RDAs). Published and periodically reviewed by the Food and Nutrition Board of the National Academy of Sciences, the RDAs were initially designed to meet the known nutritional needs of practically all healthy persons. RDAs have been established for carbohydrate and protein; the water-soluble vitamins thiamine, riboflavin, niacin, vitamin B6, folate, vitamin B12, and vitamin C; the fat-soluble vitamins A, D, and E; and the minerals copper, phosphorus, magnesium, iron, zinc, iodine, molybdenum, and selenium.
The Food and Nutrition Board has since developed a broader approach to defining nutritional adequacy. Known as dietary reference intakes (DRIs), these new guidelines go beyond the prevention of classic nutritional deficiency diseases and address the role of nutrients and other food components in long-term health and the reduction of risk of chronic diseases. The DRIs consist of four reference intakes: the RDA, the estimated average requirement (EAR), the tolerable upper intake level (UL), and the adequate intake (AI). The RDA remains the dietary intake that is sufficient to meet the nutritional requirements of nearly all individuals in an age- and gender-specific group. RDAs are intended as goals for individuals. The EAR is the intake value that is estimated to meet the requirements of 50% of individuals in an age- and gender-specific group. The UL is the maximum level of daily nutrient intake that is unlikely to pose health risks to most individuals. The AI is determined when insufficient data are available to establish the EAR and RDA for a given nutrient. It is based on fewer data and more expert opinion but is also intended as goals for individuals. The DRIs for vitamins, elements, and macronutrients are shown in Tables 29-1, 29-2 and 29-3.
Energy
The body requires energy to support normal functions and physical activity, growth, and repair of damaged tissues. Energy is provided by oxidation of dietary protein, fat, carbohydrate, and alcohol. Oxidation of 1 g of each provides 4 kcal of energy from protein and carbohydrate, 9 kcal from fat, and 7 kcal from alcohol.
In healthy adults, energy expenditure is primarily determined by three factors: basal energy expenditure (BEE), thermic effect of food (TEF), and physical activity.
The BEE is the amount of energy required to maintain basic physiologic functions. It is measured while the subject is resting in a warm room, not having eaten for 12 hours. In healthy persons, the BEE (in kcal/24 h) can be estimated by the Harris-Benedict equation, which will correctly predict measured BEE in 90% 10% of healthy subjects (See Nutritional Support, below). In clinical practice, patients rarely meet the strict criteria for BEE measurement. Instead, energy expenditure is measured in individuals at rest without food for 2 hours. This measurement, the resting energy expenditure (REE), is about 10% greater than BEE.
TEF, the amount of energy expended during and following the ingestion of food, averages approximately 10% of the BEE.
Physical activity has a major impact on energy expenditure. The average energy expenditure per hour by adults engaged in typical activities is shown in Table 29-4.
Protein
Protein is required for growth and for maintenance of body structure and function. Although the nutritional requirement is commonly stated in grams of protein, the true requirement is for nine essential amino acids plus additional nitrogen for protein synthesis. The essential amino acids are leucine, isoleucine, lysine, methionine, phenylalanine, threonine, tryptophan, valine, and histidine.
Adequate protein must be consumed each day to replace essential amino acids lost through protein turnover. On a protein-free diet, the average male loses 3.8 g of nitrogen
P.1280
P.1281
P.1282
P.1283
per day equivalent to 24 g of protein. Allowing for differences in protein quality and utilization and for individual variability, the RDA for protein is 56 g/d for men and 45 g/d for women.
Table 29-1. Dietary reference intakes: recommended intakes for individuals, vitamins.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 29-2. Dietary reference intakes: recommended intakes for individuals, elements.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 29-3. Dietary reference intakes: recommended intakes for individuals, macronutrients.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Protein and energy requirements are closely related. Diets that provide insufficient energy will require additional protein to maintain nitrogen equilibrium.
Carbohydrate
A small amount of carbohydrate approximately 100 g/d is necessary to prevent ketosis. In practice, however, a substantial portion of dietary energy should be provided by carbohydrate. The average American diet contains 45% of calories as carbohydrate. Dietary carbohydrates include simple sugars, complex carbohydrates (starches), and indigestible carbohydrates (dietary fiber). The bulk of dietary carbohydrates should be derived from starches as found in whole grains and from sugars as found in fruits and vegetables. Sucrose and other forms of added sugar such as high fructose corn syrup are concentrated sources of calories without other sources of essential nutrients and contribute to excess calorie consumption. Sucrose consumption is also thought to be an important factor in the development of tooth decay. Starches, when unrefined, provide carbohydrate calories and vitamins, minerals, and dietary fiber.
Table 29-4. Average energy kilocalories expended per hour by adults at selected weights engaged in various activities. | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||
P.1284
Dietary fiber is that portion of plant foods that cannot be digested by the human intestine. Fiber increases the bulk of the stool and facilitates excretion. Diets high in dietary fiber are associated with a lower incidence of digestive and cardiovascular diseases. The more insoluble fibers, such as those found in wheat bran, have the greatest effect on colonic function. Soluble fibers such as those found in legumes, oats, and fruit result in lower blood sugar levels in diabetics and lower blood cholesterol.
Fat
Dietary fat is the most concentrated source of food energy. Like energy from dietary carbohydrate, energy derived from fat can support protein synthesis. Dietary fat also provides the essential fatty acid linoleic acid. Other than the need for adequate quantities of linoleic acid, there is no specific requirement for dietary fat as long as the diet provides adequate nutrients oxidizable for energy. Although the average American diet contains 35 40% of calories as fat, current recommendations are to limit dietary fat to 20 35% of total calories. Diets containing as little as 5 10% of total calories as fat appear to be safe and well tolerated.
Dietary fats are composed primarily of fatty acids and dietary cholesterol. Fatty acids contain either no double bonds (saturated), one double bond (monounsaturated), or more than one double bond (polyunsaturated). Saturated fatty acids are associated with increased serum cholesterol, whereas polyunsaturated and monounsaturated fatty acids lower serum cholesterol. Trans-fatty acids, a particular form of unsaturated fat found in partially hydrogenated vegetable oils, also raise serum cholesterol levels. Saturated fats are solid at room temperature and in general are derived from animal foods; unsaturated fats are liquid at room temperature and in general are derived from plant foods.
The polyunsaturated fatty acid linoleic acid is an essential nutrient, required by the body for the synthesis of arachidonic acid, the major precursor of prostaglandins. Deficiency of linoleic acid results in dermatitis, hair loss, and impaired wound healing. For individuals with average energy requirements, approximately 5 g of linoleic acid per day 1 2% of total calories is required to prevent essential fatty acid deficiency.
Cholesterol is a major constituent of cell membranes. It is synthesized by the body and is not an essential nutrient. Diets that contain large amounts of cholesterol partially inhibit endogenous cholesterol synthesis but result in a net increase in serum cholesterol concentrations because of suppression of synthesis of low-density lipoprotein receptors. Average American diets contain approximately 450 mg/d of cholesterol, but 300 mg or less per day is recommended.
Vitamins
Vitamins are a heterogeneous group of organic molecules required by the body for a variety of essential metabolic functions. They are grouped as water-soluble vitamins: thiamine, riboflavin, niacin, vitamin B6 (pyridoxine), vitamin B12 (cobalamin), folate, pantothenic acid, biotin, and vitamin C (ascorbic acid); and fat-soluble vitamins: A, D, E, and K. Disorders of vitamin metabolism are discussed below.
Minerals
The body also requires a number of inorganic minerals, commonly grouped as the major minerals calcium, magnesium, and phosphorus; the electrolytes sodium, potassium, and chloride; and the trace elements iron, zinc, copper, manganese, molybdenum, fluoride, iodine, cobalt, chromium, and selenium. Important characteristics of major minerals and electrolytes are summarized in Table 29-5.
Table 29-5. Essential macrominerals: summary of major characteristics. | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||
Drug-Nutrient Interactions
Many medications affect nutritional requirements. A variety of drugs induce nutrient deficiencies by appetite
P.1285
suppression, intestinal malabsorption, and alterations in nutrient metabolism or excretion. The effects of selected drugs on nutrient absorption and metabolism are summarized in Table 29-6.
Dietary Guidelines
Over the past two and a half decades, numerous authorities have published dietary guidelines that make specific nutritional recommendations. Dietary Guidelines for Americans were published by the U.S. Department of Health and Human Services and the U.S. Department of Agriculture in January 2005. These guidelines reinforce many long-standing nutritional principles while also placing greater emphasis on several recent observations. Key recommendations in the new guidelines include the following: limit the intake of saturated fat, trans fat, cholesterol, added sugars, salt, and alcohol; balance calories from food and beverages with calories expended; engage in regular physical activity at least 30 60 minutes most days of the week; consume greater quantities (up to 9 servings per day) and more varieties of fruits and vegetables per day; consume at least half of the daily grains as whole grains; consume 3 cups per day of low fat milk or milk products; consume less than 10% of calories as saturated fat, less than 300 mg/d of cholesterol, and as little trans-fatty acids as possible; consume fiber-rich fruits, vegetables, and whole grains; use little added sugars or caloric sweeteners; consume less than 2300 mg of sodium per day; use alcohol sensibly and in moderation; and prepare food safely. The guidelines also provide detailed suggestions of foods to consume to meet recommended nutrient intakes for 12 different calorie levels (Table 29-7).
Butte NF et al: Energy requirements of women of reproductive age. Am J Clin Nutr 2003;77:630.
Department of Health and Human Services and Department of Agriculture: Dietary Guidelines for Americans 2005. http://www.healthierus.gov.
Dwyer J et al: National Health and Nutrition Examination Survey. Estimation of usual intakes: What We Eat in America NHANES. J Nutr 2003;133: 609S.
Howard BV et al: Low-fat dietary pattern and risk of cardiovascular disease: the Women's Health Initiative Randomized Controlled Dietary Modification Trial. JAMA 2006;295: 655.
Institute of Medicine: Committee on Use of Dietary Reference Intakes in Nutrition Labeling. Dietary reference intakes: guiding principles for nutrition labeling and fortification. National Academy of Sciences, Washington, D.C., 2003.
Niedert KC; American Dietetic Association: Position of the American Dietetic Association: Liberalization of the diet prescription improves quality of life for older adults in long-term care. J Am Diet Assoc 2005;105:1955.
Nicklas TA et al: The 2005 Dietary Guidelines Advisory Committee: developing a key message. J Am Diet Assoc 2005;105: 1418.
Rand WM et al: Meta-analysis of nitrogen balance studies for estimating protein requirements in healthy adults. Am J Clin Nutr 2003;77:109.
Table 29-6. Effect of drugs on nutrient absorption and metabolism. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 29-7. USDA food guide. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
P.1286
P.1287
P.1288
P.1289
Assessment of Nutritional Status
No single biochemical test or clinical technique is sufficiently accurate to serve as a reliable test for malnutrition. Techniques of nutritional assessment utilize a combination of methods, including evaluation of dietary intake, anthropometric measurements, clinical examination, and laboratory tests.
Dietary History
Patients undergoing a history and physical examination should be asked questions to help identify those high-risk patients who require further evaluation for malnutrition. Of particular importance are the regularity and availability of meals; who does the shopping and food preparation; recent changes in appetite, intake, or body weight; use of special diets or dietary supplements; use of alcohol, drugs, or medications; food preferences and food allergies; and the presence of illnesses affecting nutritional intakes, losses, or requirements. Elderly and adolescent patients, pregnant or lactating women, and the poor and socially isolated are at particular risk for nutritional problems.
Further quantification of dietary intake can be performed using a variety of techniques. Twenty-four-hour diet recalls provide rough estimates of nutrient intakes. Patients are asked to describe their dietary intake over the preceding day, including snacks, beverages, and alcohol. Problems with this technique include poor patient recall, difficulties in estimating serving sizes, and the inaccuracy associated with generalizing from a single day's intake. More accurate information can be obtained by asking patients to complete a 3- to 5-day diet record. Nutrient composition can then be analyzed with the aid of standard handbooks or computer software. Although prospective and less likely to be invalidated by memory lapses, omissions are still common, and the usual difficulties in estimating serving sizes persist.
Clinical Examination
A nutritionally focused physical examination should be performed on each patient at risk for nutritional problems. The examination targets body weight, muscle wasting, fat stores, volume status, and signs of micronutrient deficiencies (Table 29-8).
Table 29-8. Clinical signs that may be due to nutrient deficiency. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Evaluation of body weight is particularly useful. Body weight in relation to height can be assessed as the body mass index (BMI) weight (in kilograms)/height (in meters)2 (Table 29-9). In adult patients, however, a recent unintentional change in body weight is usually a better index of undernutrition than a low BMI. This change is best expressed as a percentage of usual weight lost per unit of time. A weight loss of 10% or more of usual weight within a period of 1 2 months is generally considered to be predictive of a poor clinical outcome.
Evaluation of body composition particularly fat stores and skeletal muscle can be performed by visual inspection or, more quantitatively, by using anthropometric measurements. The most commonly used are the triceps skin fold and mid arm muscle circumference. Because of variations in measurement, they have limited clinical utility.
A number of more sophisticated techniques are available for assessment of body composition. Most have little role in patient care. These include bioelectrical impedance, dual-energy x-ray absorptiometry, air-displacement plethysmography, hydrodensitometry, spectroscopy and mass spectrometry, neutron activation analysis, and MRI and body line scanners.
Laboratory Tests
Serum albumin is the most important laboratory test for the diagnosis of protein-calorie undernutrition. Most patients with severe protein depletion will have low serum albumin levels. Many nonnutritional conditions can also reduce serum albumin particularly liver disease and severe illness in general. Other serum proteins with shorter half-lives (such as transferrin, transthyretin, and prealbumin) may reflect short-term changes in nutritional status but suffer from similar shortcomings.
Tests of cellular immunity are also abnormal in many patients with protein-calorie undernutrition. Measurements of the total lymphocyte count and delayed hypersensitivity reactions to common skin test antigens are nonspecific; abnormalities may be due to nonnutritional factors.
Despite their poor specificity, these tests are useful prognostically. Patients with abnormal nutritional assessment parameters have a markedly increased risk of poor clinical outcomes.
Blanck HM et al: Laboratory issues: use of nutritional biomarkers. J Nutr 2003;133(Suppl 3):888S.
Cupisti A et al: Skeletal muscle and nutritional assessment in chronic renal failure patients on a protein-restricted diet. J Intern Med 2004;255:115.
Kotler D: Challenges to diagnosis of HIV-associated wasting. J Acquir Immune Defic Syndr 2004;37:S280.
Mackerras D et al: 24-hour national dietary survey data: how do we interpret them most effectively? Public Health Nutr 2005; 8:657.
Muhlberg W et al: Low total protein increases injury risk in the elderly. J Am Geriatr Soc 2004;52:324.
Sahyoun NR et al: Nutritional status of the older adult is associated with dentition status. J Am Diet Assoc 2003;103: 61.
Singh H et al: Malnutrition is prevalent in hospitalized medical patients: Are housestaff identifying the malnourished patient? Nutrition 2006; January 31 Epub.
Yeh SS et al: Risk factors relating blood markers of inflammation and nutritional status to survival in cachectic geriatric patients in a randomized clinical trial. J Am Geriatr Soc 2004;52:1708.
P.1290
Nutritional Disorders
Protein-Energy Malnutrition
![]() Essentials of Diagnosis
Essentials of Diagnosis
History of decreased intake of energy or protein, increased nutrient losses, or increased nutrient requirements.
Manifestations range from weight loss and growth failure to distinct syndromes, kwashiorkor, and marasmus.
In severe cases, virtually all organ systems are affected.
Protein loss correlates with weight loss: 35 40% total body weight loss is usually fatal.
General Considerations
Protein-energy malnutrition occurs as a result of a relative or absolute deficiency of energy and protein. It may
P.1291
be primary, due to inadequate food intake, or secondary, as a result of other illness. For most developing nations, primary protein-energy malnutrition remains among the most significant health problems. Protein-energy malnutrition has been described as two distinct syndromes. Kwashiorkor, caused by a deficiency of protein in the presence of adequate energy, is typically seen in weaning infants at the birth of a sibling in areas where foods containing protein are insufficiently abundant. Marasmus, caused by combined protein and energy deficiency, is most commonly seen where adequate quantities of food are not available.
Table 29-9. Body mass index chart.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
In industrialized societies, protein-energy malnutrition is most often secondary to other diseases. Kwashiorkor-like secondary protein-energy malnutrition occurs primarily in association with hypermetabolic acute illnesses such as trauma, burns, and sepsis. Marasmus-like secondary protein-energy malnutrition typically results from chronic diseases such as chronic obstructive pulmonary disease (COPD), congestive heart failure, cancer, or AIDS. These syndromes have been estimated to be present in at least 20% of hospitalized patients. A substantially greater number of patients have risk factors that could result in these syndromes. In both syndromes, protein-energy malnutrition is caused either by decreased intake of energy and protein, increased nutrient losses, or increased nutrient requirements dictated by the underlying illness. For example, diminished oral intake may result from poor dentition or various gastrointestinal disorders. Loss of nutrients results from malabsorption and diarrhea as well as from glycosuria. Nutrient requirements are increased by fever, surgery, neoplasia, and burns.
Pathophysiology
Protein-energy malnutrition affects every organ system. The most obvious results are loss of body weight, adipose stores, and skeletal muscle mass. Weight losses of 5 10% are usually tolerated without loss of physiologic function; losses of 35 40% of body weight usually result in death. Loss of protein from skeletal muscle and internal organs is usually proportionate to weight loss. Protein mass is lost from the liver, gastrointestinal tract, kidneys, and heart.
As protein-energy malnutrition progresses, organ dysfunction develops. Hepatic synthesis of serum proteins decreases, and depressed levels of circulating proteins are observed. Cardiac output and contractility are decreased, and the electrocardiogram (ECG) may show decreased voltage and a rightward axis shift. Autopsies of patients who die with severe undernutrition show myofibrillar atrophy and interstitial edema of the heart.
P.1292
Respiratory function is affected primarily by weakness and atrophy of the muscles of respiration. Vital capacity and tidal volume are depressed, and mucociliary clearance is abnormal. The gastrointestinal tract is affected by mucosal atrophy and loss of villi of small intestine, resulting in malabsorption. Intestinal disaccharidase deficiency and mild pancreatic insufficiency also occur.
Changes in immunologic function are among the most important changes seen in protein-calorie undernutrition. T lymphocyte number and function are depressed. Changes in B cell function are more variable. Impaired complement activity, granulocyte function, and anatomic barriers to infection are noted, and wound healing is poor.
Clinical Findings
The clinical manifestations of protein-energy malnutrition range from mild growth retardation and weight loss to a number of distinct clinical syndromes. Children in the developing world manifest marasmus and kwashiorkor. In secondary protein-energy malnutrition as seen in industrialized nations, clinical manifestations are affected by the degree of protein and energy deficiency, the underlying illness that resulted in the deficiency, and the patient's nutritional status prior to illness.
In marasmus-like secondary protein-energy malnutrition, most patients typically develop progressive wasting that begins with weight loss and proceeds to more severe cachexia. In the most severe form of this disorder, most body fat stores disappear and muscle mass decreases, most noticeably in the temporalis and interosseus muscles. Laboratory studies may be unremarkable serum albumin, for example, may be normal or slightly decreased, rarely decreasing to < 2.8 g/dL. In contrast, owing to its rapidity of onset, kwashiorkor-like secondary protein-energy malnutrition may develop in patients with normal subcutaneous fat and muscle mass or, if the patient is obese, in patients with excess fat and muscle. The serum protein level, however, typically declines and the serum albumin is often < 2.8 g/dL. Dependent edema, ascites, or anasarca may develop. As with primary protein-energy malnutrition, combinations of the marasmus-like and kwashiorkor-like syndromes can occur simultaneously, typically in patients with progressive chronic disease in whom a superimposed acute illness develops.
Treatment
The treatment of severe protein-energy malnutrition is a slow process requiring great care. Initial efforts should be directed at correcting fluid and electrolyte abnormalities and infections. Of particular concern are depletion of potassium, magnesium, and calcium and acid-base abnormalities. The second phase of treatment is directed at repletion of protein, energy, and micronutrients. Treatment is started with modest quantities of protein and calories calculated according to the patient's actual body weight. Adult patients are given 1 g of protein and 30 kcal/kg. Concomitant administration of vitamins and minerals is obligatory. Either the enteral or parenteral route can be used, although the former is preferable. Enteral fat and lactose are withheld initially. Patients with less severe protein-calorie undernutrition can be given calories and protein simultaneously with the correction of fluid and electrolyte abnormalities. Similar quantities of protein and calories are recommended for initial treatment.
Patients treated for protein-energy malnutrition require close follow-up. In adults, both calories and protein are advanced as tolerated, adults to 1.5 g/kg/d of protein and 40 kcal/kg/d of calories.
Patients who are refed too rapidly may develop a number of untoward clinical sequelae. During refeeding, circulating potassium, magnesium, phosphorus, and glucose move intracellularly and can result in low serum levels of each. The administration of water and sodium with carbohydrate refeeding can overload hearts with depressed cardiac function and result in congestive heart failure. Enteral refeeding can lead to malabsorption and diarrhea due to abnormalities in the gastrointestinal tract.
Refeeding edema is a benign condition to be differentiated from congestive heart failure. Changes in renal sodium reabsorption and poor skin and blood vessel integrity result in the development of dependent edema without other signs of heart disease. Treatment includes reassurance, elevation of the dependent area, and modest sodium restriction. Diuretics are usually ineffective, may aggravate electrolyte deficiencies, and should not be used.
The prevention and early detection of protein-energy malnutrition in hospitalized patients require awareness of its risk factors and early symptoms and signs. Patients at risk require formal assessment of nutritional status and close observation of dietary intake, body weight, and nutritional requirements during the hospital stay.
Cooper BA et al: Protein malnutrition and hypoalbuminemia as predictors of vascular events and mortality in ESRD. Am J Kidney Dis 2004;43:61.
Delano MJ et al: The origins of cachexia in acute and chronic inflammatory diseases. Nutr Clin Pract 2006;21:68.
Faintuch J et al: Severe protein-calorie malnutrition after bariatric procedures. Obes Surg 2004;14:175.
Wanke C: Pathogenesis and consequences of HIV-associated wasting. J Acquir Immune Defic Syndr 2004;37(Suppl 4):S277.
Obesity
![]() Essentials of Diagnosis
Essentials of Diagnosis
Excess adipose tissue, resulting in BMI > 30.
Upper body obesity (abdomen and flank) of greater health consequence than lower body obesity (buttocks and thighs).
Associated with multiple metabolic and structural disorders, including diabetes mellitus, hypertension, and hyperlipidemia.
P.1293
General Considerations
Obesity is one of the most common disorders in medical practice and among the most frustrating and difficult to manage. Little progress has been made in prevention or treatment, yet major changes have occurred in our understanding of its causes and its implications for health.
Definition & Measurement
Obesity is defined as an excess of adipose tissue. Accurate quantification of body fat requires sophisticated techniques not usually available in clinical practice. Physical examination is usually sufficient to detect excess body fat. More quantitative evaluation is performed by calculating the BMI.
The BMI closely correlates with excess adipose tissue. It is calculated by dividing measured body weight in kilograms by the height in meters squared (Table 29-9).
The National Institutes of Health (NIH) define a normal BMI as 18.5 24.9. Overweight is defined as BMI = 25 29.9. Class I obesity is 30 34.9, class II obesity is 35 39.9, and class III (extreme) obesity is BMI > 40. Factors other than total weight, however, are also important. Upper body obesity (excess fat around the waist and flank) is a greater health hazard than lower body obesity (fat in the thighs and buttocks). Obese patients with increased abdominal circumference (> 102 cm in men and 88 cm in women) or with high waist-hip ratios (> 1.0 in men and > 0.85 in women) have a greater risk of diabetes mellitus, stroke, coronary artery disease, and early death than equally obese patients with lower ratios. Further differentiation of the location of excess fat suggests that visceral fat within the abdominal cavity is more hazardous to health than subcutaneous fat around the abdomen.
Current U.S. survey data demonstrate that 65% of Americans are overweight and 30.4% are obese. Women in the United States are more apt to be obese than men, and African-American and Mexican-American women are more obese than whites. The poor are more obese than the rich regardless of race.
Health Consequences of Obesity
Obesity is associated with significant increases in both morbidity and mortality. A great many disorders occur with greater frequency in obese people. The most important and common of these are hypertension, type 2 diabetes mellitus, hyperlipidemia, coronary artery disease, degenerative joint disease, and psychosocial disability. Approximately 60% of individuals with obesity in the United States have the metabolic syndrome (including three or more of the following factors: elevated abdominal circumference, blood pressure, blood triglycerides, and fasting blood sugar, and low high-density lipoprotein [HDL] cholesterol). Certain cancers (colon, rectum, and prostate in men; uterus, biliary tract, breast, and ovary in women), thromboembolic disorders, digestive tract diseases (gallstones, reflux esophagitis), and skin disorders are also more prevalent in the obese. Surgical and obstetric risks are greater. Obese patients also have a greater risk of pulmonary functional impairment, endocrine abnormalities, proteinuria, and increased hemoglobin concentration.
In young and middle-aged adults, mortality from all causes and mortality from cardiovascular disease increase in proportion to the degree of obesity. The relative risk associated with obesity, however, decreases with age, and weight is no longer a risk factor in adults over age 75 years.
Etiology
Until recently, obesity was considered to be the direct result of a sedentary lifestyle plus chronic ingestion of excess calories. Although these factors are undoubtedly the principal cause in some cases, there is now evidence for strong genetic influences on the development of obesity. Adopted children demonstrate a close relationship between their body mass index and that of their biologic parents. No such relationship is found between the children and their adoptive parents. Twin studies also demonstrate substantial genetic influences on BMI with little influence from the childhood environment. As much as 40 70% of obesity may be explained by genetic influences.
Genetic determinants of some types of obesity have now been established. Five genes affecting control of appetite have been identified in mice. Mutations of each gene result in obesity, and each has a human homolog. One gene codes for a protein expressed by adipose tissue leptin and another for the leptin receptor in the brain. The other three genes affect brain pathways downstream from the leptin receptor. Numerous other candidate genes for human obesity have been identified. Only a small percentage (4 6%) of human obesity is thought to be due to single gene mutations. Most human obesity undoubtedly develops from the interactions of multiple genes, environmental factors, and behavior.
Medical Evaluation of the Obese Patient
Historical information should be obtained about age at onset, recent weight changes, family history of obesity, occupational history, eating and exercise behavior, cigarette and alcohol use, previous weight loss experience, and psychosocial factors including assessment for depression and eating disorders. Particular attention should be directed at use of laxatives, diuretics, hormones, nutritional supplements, and over-the-counter medications.
P.1294
Physical examination should assess the degree and distribution of body fat, overall nutritional status, and signs of secondary causes of obesity.
Less than 1% of obese patients have an identifiable secondary, nonpsychiatric, cause of obesity. Hypothyroidism and Cushing's syndrome are important examples that can usually be diagnosed by physical examination in patients with unexplained recent weight gain. Such patients require further endocrinologic evaluation, including serum thyroid-stimulating hormone (TSH) determination and dexamethasone suppression testing (see Chapter 26).
All obese patients should be assessed for medical consequences of their obesity by screening for the metabolic syndrome. Blood pressure, waist circumference, fasting glucose, low-density lipoprotein (LDL) and HDL cholesterol, and triglycerides should be measured.
Treatment
Using conventional techniques, only 20% of patients will lose 20 lb and maintain the loss for over 2 years; 5% will maintain a 40-lb loss. Continued close provider-patient contact appears to be more important for success of treatment than the specific features of any given treatment regimen. Careful patient selection will improve success rates and decrease frustration of both patients and therapists. Only sufficiently motivated patients should enter active treatment programs. Specific attempts to identify motivated patients eg, requesting a 3-day diet record are often useful.
Most successful programs employ a multidisciplinary approach to weight loss, with hypocaloric diets, behavior modification to change eating behavior, aerobic exercise, and social support. Emphasis must be on maintenance of weight loss.
Dietary instructions for most patients incorporate the same principles that apply to healthy people who are not obese, ie, a low-fat, high-complex carbohydrate, high-fiber diet. This is achieved by emphasizing intake of a wide variety of predominantly unprocessed foods. Special attention is usually paid to limiting foods that provide large amounts of calories without other nutrients, ie, fat, sucrose, and alcohol. There is no special advantage to diets that restrict carbohydrates, advocate relatively larger amounts of protein or fats, or recommend ingestion of foods one at a time. In some instances, however, diets that are restricted in carbohydrates can be effective in achieving a lower total calorie intake. Several studies have demonstrated that low-carbohydrate diets can be used safely for weight loss for up to 1 year without adverse effects on lipids or other metabolic parameters. Meal replacement diets can also be used effectively and safely to achieve weight loss.
Long-term changes in eating behavior are required to maintain weight loss. Although formal behavior modification programs are available to which patients can be referred, the clinician caring for obese patients can teach a number of useful behavioral techniques. The most important technique is to emphasize planning and record keeping. Patients can be taught to plan menus and exercise sessions and to record their actual behavior. Record keeping not only aids in behavioral change, but also helps the provider to make specific suggestions for problem solving. Patients can be taught to recognize eating cues (emotional, situational, etc) and how to avoid or control them. Reward systems and refundable financial contracts are also useful for many patients. Regular self-monitoring of weight is also associated with improved long-term weight maintenance.
Exercise offers a number of advantages to patients trying to lose weight and keep it off. Aerobic exercise directly increases the daily energy expenditure and is particularly useful for long-term weight maintenance. Exercise will also preserve lean body mass and partially prevent the decrease in BEE seen with semistarvation. Up to 1 hour of moderate exercise per day is associated with long-term weight maintenance in individuals who have successfully lost weight. Social support is essential for a successful weight loss program. Continued close contact with clinicians and involvement of the family and peer group are useful techniques for reinforcing behavioral change and preventing social isolation.
Patients with severe obesity may require more aggressive treatment regimens. Very-low-calorie diets ( 800 kcal/d) result in rapid weight loss and marked initial improvement in obesity-related metabolic complications. Patients are commonly maintained on such programs for 4 6 months and lose an average of 2 4 lb per week. Most programs use meal replacement diets to achieve the very-low-calorie intake. Long-term weight maintenance is less predictable and requires concurrent behavior modification, long-term use of low-calorie diets, careful self-monitoring, and regular exercise. Side effects such as fatigue, orthostatic hypotension, cold intolerance, and fluid and electrolyte disorders are observed in proportion to the degree of calorie reduction and require regular supervision by a physician. Other less common complications include gout, gallbladder disease, and cardiac arrhythmias. Although weight loss is more rapidly achieved with very-low-calorie diets as compared with traditional diets, long-term outcomes are equivalent.
Medications for the treatment of obesity are available both over the counter and by prescription. Considerable controversy exists as to the appropriate use of medications for obesity. NIH clinical obesity guidelines state that obesity drugs may be used as part of a comprehensive weight loss program for patients with BMI > 30 or those with BMI > 27 with obesity-related risk factors. Nonetheless, use of medications has decreased in the United States since dexfenfluramine and fenfluramine were withdrawn from the market in 1997 after multiple reports of medication-associated valvular heart disease. Although studies have since estimated the risk of valvular heart disease to be substantially
P.1295
less than the 30% prevalence first reported, this experience has led to considerable caution in the use of anorectic medications.
Anorectic medications can be classified as catecholaminergic or serotonergic. Catecholaminergic medications include amphetamines (with high abuse potential) and the nonamphetamine schedule IV appetite suppressants phentermine, diethylpropion, and mazindol. The selective serotonin reuptake inhibitor (SSRI) antidepressants, eg, fluoxetine and sertraline, have serotonergic activity but are not approved by the Food and Drug Administration (FDA) for weight loss.
Several medications remain available for treatment of obesity. Older catecholaminergic medications (eg, phentermine, diethylpropion, mazindol) are approved for short-term use only and have limited utility. Two newer medications are approved for weight loss: sibutramine and orlistat. Sibutramine blocks uptake of both serotonin and norepinephrine in the central nervous system. Orlistat reduces fat absorption in the gastrointestinal tract.
Sibutramine, typically at doses of 10 mg/d, results in average weight losses of 3 5 kg more than placebo in studies extending over 6 12 months. Sibutramine also appears to improve 1-year outcomes in patients on very-low-calorie diets. Side effects include dry mouth, anorexia, constipation, insomnia, and dizziness. In some patients (< 5%), sibutramine may substantially increase blood pressure.
Orlistat is the first approved medication for obesity that works in the gastrointestinal tract rather than the central nervous system. By inhibiting intestinal lipase, orlistat reduces fat absorption. As expected, orlistat may result in diarrhea, gas, and cramping and perhaps also reduced absorption of fat-soluble vitamins. In randomized trials with up to 2 years of follow-up, orlistat has resulted in 2 4 kg greater weight loss than placebo. The recommended dose of orlistat is 120 mg three times daily with meals. Despite FDA approval of sibutramine and orlistat and NIH guidelines supporting their use, long-term clinical benefits have not been demonstrated. Although these medications result in some additional weight loss at the end of 1- and 2-year clinical trials and, in some studies, improved obesity-related metabolic parameters, the impact of these medications on obesity-related clinical outcomes is unknown.
Bariatric surgery is an increasingly prevalent treatment option for patients with severe obesity. In the United States, gastric operations are considered the procedures of choice. Most popular is the roux-en-Y gastric bypass (GBP). In most centers, the operation can be done laparoscopically. GBP typically results in substantial amounts of weight loss close to 50% of initial body weight in some studies. Complications occur in up to 40% of subjects undergoing bariatric surgery and include peritonitis due to anastomotic leak, abdominal wall hernias, staple line disruption, gallstones, neuropathy, marginal ulcers, stomal stenosis, wound infections, thromboembolic disease, and various nutritional deficiencies and gastrointestinal symptoms. Within 30 days operative mortality rates are nil to 1% in low-risk populations but have been reported to be substantially higher in Medicare beneficiaries. One-year mortality rates have been reported as high as 7.5% in men with Medicare. The surgical volume (the number of cases performed by the surgeon or hospital) has been demonstrated to be an important predictor of outcome. NIH consensus panel recommendations are to limit obesity surgery to patients with BMIs over 40, or over 35 if obesity-related comorbidities are present. Recent studies suggest that the procedure is cost-effective for patients with severe obesity and some third-party payers now cover the procedure.
American Medical Association: Assessment and Management of Adult Obesity: A Primer for Physicians, 2003. http://www.ama-assn.org/ama/pub/category/10931.html.
Avenell A et al: What are the long-term benefits of weight reducing diets in adults? A systematic review of randomized controlled trials. J Hum Nutr Diet 2004;17:317.
Buchwald H et al: Bariatric surgery: a systematic review and meta-analysis. JAMA 2004;292:1724.
Dansinger ML et al: Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: a randomized trial. JAMA 2005;293:43.
Flegal KM et al: Excess deaths associated with underweight, overweight, and obesity. JAMA 2005;293:1861.
Flum DR et al: Early mortality among Medicare beneficiaries undergoing bariatric surgical procedures. JAMA 2005;294:1903.
Hedley AA et al: Prevalence of overweight and obesity among US children, adolescents, and adults, 1999 2002. JAMA 2004;291:2847.
Howard BV et al: Low-fat dietary pattern and weight change over 7 years: the Women's Health Initiative Dietary Modification Trial. JAMA 2006;295:39.
Hu FB et al: Adiposity as compared with physical activity in predicting mortality among women. N Engl J Med 2004;351:2694.
Mathus-Vliegen EM et al: Health-related quality-of-life in patients with morbid obesity after gastric banding for surgically induced weight loss. Surgery 2004;135:489.
Noakes M et al: Meal replacements are as effective as structured weight-loss diets for treating obesity in adults with features of metabolic syndrome. J Nutr 2004;134:1894.
Norris S et al: Pharmacotherapy for weight loss in adults with type 2 diabetes mellitus. Cochrane Database Syst Rev 2005;(1):CD004096.
Pi-Sunyer FX et al; RIO-North America Study Group: Effect of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients: RIO-North America: a randomized controlled trial. JAMA 2006;295:761.
Sjostrom L et al and the Swedish Obese Subjects Study Scientific Group: Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med 2004;351:2683.
Tsai AG et al: Systematic review: an evaluation of major commercial weight loss programs in the United States. Ann Intern Med 2005;142:56.
P.1296
Wadden RA et al: Randomized trial of lifestyle modification and pharmacotherapy for obesity. N Engl J Med 2005;353:2111.
Eating Disorders
Anorexia Nervosa
![]() Essentials of Diagnosis
Essentials of Diagnosis
Disturbance of body image and intense fear of becoming fat.
Weight loss leading to body weight 15% below expected.
In females, absence of three consecutive menstrual cycles.
General Considerations
Anorexia nervosa typically begins in the years between adolescence and young adulthood. Ninety percent of patients are females, most from the middle and upper socioeconomic strata. The diagnosis is based on weight loss leading to body weight 15% below expected, a distorted body image, fear of weight gain or of loss of control over food intake, and, in females, the absence of at least three consecutive menstrual cycles. Other medical or psychiatric illnesses that can account for anorexia and weight loss must be excluded.
The prevalence of anorexia nervosa is greater than previously suggested. In Rochester, Minnesota, for example, the prevalence per 100,000 population is estimated to be 270 for females and 22 for males. Many other adolescent girls have features of the disorder without the severe weight loss.
The cause of anorexia nervosa is not known. Although multiple endocrinologic abnormalities exist in these patients, most authorities believe they are secondary to malnutrition and not primary disorders. Most experts favor a primary psychiatric origin, but no hypothesis explains all cases. The patient characteristically comes from a family whose members are highly goal and achievement oriented. Interpersonal relationships may be inadequate or destructive. The parents are usually overly directive and concerned with slimness and physical fitness, and much of the family conversation centers around dietary matters. One theory holds that the patient's refusal to eat is an attempt to regain control of her body in defiance of parental control. The patient's unwillingness to inhabit an adult body may also represent a rejection of adult responsibilities and the implications of adult interpersonal relationships. Patients are commonly perfectionistic in behavior and exhibit obsessional personality characteristics. Marked depression or anxiety may be present.
Clinical Findings
A. Symptoms and Signs
Patients with anorexia nervosa may exhibit severe emaciation and may complain of cold intolerance or constipation. Amenorrhea is almost always present. Bradycardia, hypotension, and hypothermia may be present in severe cases. Examination demonstrates loss of body fat, dry and scaly skin, and increased lanugo body hair. Parotid enlargement and edema may also occur.
B. Laboratory Findings
Laboratory findings are variable but may include anemia, leukopenia, electrolyte abnormalities, and elevations of blood urea nitrogen (BUN) and serum creatinine. Serum cholesterol levels are often increased. Endocrine abnormalities include depressed levels of luteinizing and follicle-stimulating hormones and impaired response of luteinizing hormone to luteinizing hormone-releasing hormone.
Diagnosis & Differential Diagnosis
The diagnosis can be difficult, since many common social and cultural factors promote and maintain anorexic behavior. The diagnosis depends on identification of the common behavioral features and exclusion of medical disorders that would account for weight loss.
Behavioral features required for the diagnosis include intense fear of becoming obese, disturbance of body image, weight loss of at least 15%, and refusal to exceed a minimal normal weight.
The differential diagnosis includes endocrine and metabolic disorders such as panhypopituitarism, Addison's disease, hyperthyroidism, and diabetes mellitus; gastrointestinal disorders such as Crohn's disease and celiac sprue; chronic infections and cancers such as tuberculosis and lymphoma; and rare central nervous system disorders such as hypothalamic tumors.
Treatment
The goal of treatment is restoration of normal body weight and resolution of psychological difficulties. Hospitalization may be necessary. Treatment programs conducted by experienced teams are successful in about two-thirds of cases, restoring normal weight and menstruation. One-half continue to experience difficulties with eating behavior and psychiatric problems. Occasional patients with anorexia develop obesity after treatment. Two to 6% of patients die from the complications of the disorder or commit suicide.
Various treatment methods have been used without clear evidence of superiority of one over another. Supportive care by physicians and nurses is probably the most important feature of therapy. Structured behavioral
P.1297
therapy, intensive psychotherapy, and family therapy may be tried. A variety of medications including tricyclic antidepressants, SSRIs, and lithium carbonate are effective in some cases; overall, however, clinical trial results have been disappointing. Patients with severe malnutrition must be hemodynamically stabilized and may require enteral or parenteral feeding. Forced feedings should be reserved for life-threatening situations, since the goal of treatment is to reestablish normal eating behavior.
American Dietetic Association: Position of the American Dietetic Association: Nutrition intervention in the treatment of anorexia nervosa, bulimia nervosa, and eating disorder not otherwise specified (EDNOS). http://www.eatright.org/adap0701.html.
Birch K: Female athlete triad. BMJ 2005;330:244.
Claudino A et al: Antidepressants for anorexia nervosa. Cochrane Database Syst rev 2006;(1):CD004365.
Fairburn CG et al: Eating disorders. Lancet 2003;361:407.
Fisher M: The course and outcome of eating disorders in adults and in adolescents: a review. Adolesc Med 2003;14:149.
McIntosh VV et al: Three psychotherapies for anorexia nervosa: a randomized, controlled trial. Am J Psychiatry 2005;162:741.
Pompili M et al: Suicide in anorexia nervosa: a meta-analysis. Int J Eat Disord 2004;36:99.
Rome ES: Eating disorders. Obstet Gynecol Clin North Am 2003;30:353.
Wadden TA et al: Dieting and the development of eating disorders in obese women: results of a randomized controlled trial. Am J Clin Nutr 2004;80:560.
Wilson GT et al: Eating disorders guidelines from NICE. Lancet 2005;365:79.
Yager J et al: Clinical practice. Anorexia nervosa. N Engl J Med 2005;353:1481.
Bulimia Nervosa
![]() Essentials of Diagnosis
Essentials of Diagnosis
Uncontrolled episodes of binge eating at least twice weekly for 3 months.
Recurrent inappropriate compensation to prevent weight gain such as self-induced vomiting, laxatives, diuretics, fasting, or excessive exercise.
Overconcern with weight and body shape.
General Considerations
Bulimia nervosa is the episodic uncontrolled ingestion of large quantities of food followed by recurrent inappropriate compensatory behavior to prevent weight gain such as self-induced vomiting, diuretic or cathartic use, or strict dieting or vigorous exercise.
Like anorexia nervosa, bulimia nervosa is predominantly a disorder of young, white, middle- and upper-class women. It is more difficult to detect than anorexia, and some studies have estimated that the prevalence may be as high as 19% in college-aged women.
Clinical Findings
Patients with bulimia nervosa typically consume large quantities of easily ingested high-calorie foods, usually in secrecy. Some patients may have several such episodes a day for a few days; others report regular and persistent patterns of binge eating. Binging is usually followed by vomiting, cathartics, or diuretics and is usually accompanied by feelings of guilt or depression. Periods of binging may be followed by intervals of self-imposed starvation. Body weights may fluctuate but generally are within 20% of desirable weights.
Some patients with bulimia nervosa also have a cryptic form of anorexia nervosa with significant weight loss and amenorrhea. Family and psychological issues are generally similar to those encountered among patients with anorexia nervosa. Bulimics, however, have a higher incidence of premorbid obesity, greater use of cathartics and diuretics, and more impulsive or antisocial behavior. Menstruation is usually preserved.
Medical complications are numerous. Gastric dilatation and pancreatitis have been reported after binges. Vomiting can result in poor dentition, pharyngitis, esophagitis, aspiration, and electrolyte abnormalities. Cathartic and diuretic abuse also cause electrolyte abnormalities or dehydration. Constipation and hemorrhoids are common.
Treatment
Treatment of bulimia nervosa and bulimarexia requires supportive care and psychotherapy. Individual, group, family, and behavioral therapy have all been utilized. Antidepressant medications may be helpful. The best results have been with fluoxetine hydrochloride and other SSRIs. Although death from bulimia is rare, the long-term psychiatric prognosis in severe bulimia is worse than that in anorexia nervosa.
Duncan AE et al: Are there subgroups of bulimia nervosa based on comorbid psychiatric disorders? Int J Eat Disord 2005;37:19.
Hay PJ et al: Psychotherapy for bulimia nervosa and binging. Cochrane Database Syst Rev 2003;(1):CD000562.
Palmer R: Bulimia nervosa: 25 years on. Br J Psychiatry 2004;185:447.
Schneider M: Bulimia nervosa and binge-eating disorder in adolescents. Adolesc Med 2003;14:119.
Walsh BT et al: Treatment of bulimia nervosa in a primary care setting. Am J Psychiatry 2004;161:556.
P.1298
Disorders of Vitamin Metabolism
Deficiencies of single vitamins are less often encountered than those of multiple vitamins. Although any cause of protein-calorie undernutrition can result in concurrent vitamin deficiency, most deficiencies are associated with malabsorption, alcoholism, medications, hemodialysis, total parenteral nutrition, food faddism, or inborn errors of metabolism.
Vitamin deficiency syndromes develop gradually. Symptoms are commonly nonspecific, and the physical examination is rarely helpful in early diagnosis. Most characteristic physical findings are seen late in the course of the syndrome. Other characteristic physical findings, such as glossitis and cheilosis, are seen with deficiencies of many B vitamins. Such abnormalities suggest the presence of a nutritional deficiency but do not indicate which nutrient is deficient.
Despite the relative ease of meeting the recommended daily allowances with a mixed diet, many adults in the United States take vitamin supplements. In fact, syndromes of vitamin excess may be more common than deficiency syndromes, particularly those due to excess of vitamins A, D, and B6. Most claims for significant health benefits of such supplements, particularly those taken in megadoses, remain unsubstantiated.
Some vitamins can be used efficaciously as drugs. Derivatives of vitamin A are used to treat cystic acne and, more recently, skin wrinkles. Niacin is an effective medication for hyperlipidemia. Vitamin-responsive inborn errors of metabolism also commonly require pharmacologic doses of vitamins.
Millen AE et al: Use of vitamin, mineral, nonvitamin, and nonmineral supplements in the United States: The 1987, 1992, and 2000 National Health Interview Survey results. J Am Diet Assoc 2004;104:942.
Radimer K et al: Dietary supplement use by US adults: data from the National Health and Nutrition Examination Survey, 1999 2000. Am J Epidemiol 2004;160:339.
Thiamine (B1) Deficiency
![]() Essentials of Diagnosis
Essentials of Diagnosis
Most common in patients with chronic alcoholism.
Early symptoms of anorexia, muscle cramps, paresthesias, irritability.
Advanced syndromes of high output heart failure ( wet beriberi ), peripheral nerve disorders, and Wernicke-Korsakoff syndrome ( dry beriberi ).
Clinical Findings
Most thiamine deficiency in the United States is due to alcoholism. Patients with chronic alcoholism may have poor dietary intakes of thiamine and impaired thiamine absorption, metabolism, and storage. Thiamine deficiency is also associated with malabsorption, dialysis, and other causes of chronic protein-calorie undernutrition. Thiamine deficiency can be precipitated in patients with marginal thiamine status with intravenous dextrose solutions.
Early manifestations of thiamine deficiency include anorexia, muscle cramps, paresthesias, and irritability. Advanced deficiency primarily affects the cardiovascular system ( wet beriberi ) or the nervous system ( dry beriberi ). Wet beriberi occurs in thiamine deficiency accompanied by severe physical exertion and high carbohydrate intakes. Dry beriberi occurs in thiamine deficiency accompanied by inactivity and low calorie intake.
Wet beriberi is characterized by marked peripheral vasodilation resulting in high-output heart failure with dyspnea, tachycardia, cardiomegaly, and pulmonary and peripheral edema, with warm extremities mimicking cellulitis.
Dry beriberi involves both the peripheral and the central nervous systems. Peripheral nerve involvement is typically a symmetric motor and sensory neuropathy with pain, paresthesias, and loss of reflexes. The legs are affected more than the arms. Central nervous system involvement results in Wernicke-Korsakoff syndrome. Wernicke's encephalopathy consists of nystagmus progressing to ophthalmoplegia, truncal ataxia, and confusion. Korsakoff's syndrome includes amnesia, confabulation, and impaired learning.
Diagnosis
A variety of biochemical tests are available to assess thiamine deficiency. In most instances, however, the clinical response to empiric thiamine therapy is used to support a diagnosis of thiamine deficiency. The most commonly used and widely available biochemical tests are measurement of erythrocyte transketolase activity and urinary thiamine excretion. A transketolase activity coefficient greater than 15 20% suggests thiamine deficiency.
Treatment
Thiamine deficiency is treated with large parenteral doses of thiamine. Fifty to 100 mg/d is administered for the first few days, followed by daily oral doses of 5 10 mg/d. All patients should simultaneously receive therapeutic doses of other water-soluble vitamins. Although treatment results in complete resolution in half of patients (one-fourth immediately and another one-fourth over days), the other half obtain only partial resolution or no benefit.
P.1299
Thiamine Toxicity
There is no known toxicity of thiamine.
Day E et al: Thiamine for Wernicke-Korsakoff Syndrome in people at risk from alcohol abuse. Cochrane Database Syst Rev 2004;(1):CD004033.
Klein M et al: Fatal metabolic acidosis caused by thiamine deficiency. J Emerg Med 2004;26:301.
Loh Y et al: Acute Wernicke's encephalopathy following bariatric surgery: clinical course and MRI correlation. Obes Surg 2004;14:129.
Morcos Z et al: Wernicke encephalopathy. Arch Neurol 2004;61:775.
Selitsky T et al: Wernicke's encephalopathy with hyperemesis and ketoacidosis. Obstet Gynecol 2006;107:486.
Sgouros X et al: Evaluation of a clinical screening instrument to identify states of thiamine deficiency in inpatients with severe alcohol dependence syndrome. Alcohol Alcoholism 2004;39:227.
Riboflavin (B2) Deficiency
Clinical Findings
Riboflavin deficiency almost always occurs in combination with deficiencies of other vitamins. Dietary inadequacy, interactions with a variety of medications, alcoholism, and other causes of protein-calorie undernutrition are the most common causes of riboflavin deficiency.
Manifestations of riboflavin deficiency include cheilosis, angular stomatitis, glossitis, seborrheic dermatitis, weakness, corneal vascularization, and anemia.
Diagnosis
Riboflavin deficiency is usually treated empirically when the diagnosis is suspected. Deficiency can be confirmed by measuring the riboflavin-dependent enzyme erythrocyte glutathione reductase. Activity coefficients greater than 1.2 1.3 are suggestive of riboflavin deficiency. Urinary riboflavin excretion and serum levels of plasma and red cell flavins can also be measured.
Treatment
Riboflavin deficiency is easily treated with foods such as meat, fish, and dairy products or with oral preparations of the vitamin. Administration of 5 15 mg/d until clinical findings are resolved is usually adequate. Riboflavin can also be given parenterally, but it is poorly soluble in aqueous solutions.
Riboflavin Toxicity
There is no known toxicity of riboflavin.
Friedli A et al: Images in clinical medicine. Oculo-orogenital syndrome a deficiency of vitamins B2 and B6. N Engl J Med 2004;350:1130.
McKinley MC et al: Effect of riboflavin supplementation on plasma homocysteine in elderly people with low riboflavin status. Eur J Clin Nutr 2002;56:850.
Powers HJ: Riboflavin (vitamin B-2) and health. Am J Clin Nutr 2003;77:1352.
Niacin Deficiency
General Considerations
Niacin is a generic term for nicotinic acid and other derivatives with similar nutritional activity. Unlike most other vitamins, niacin can be synthesized from the amino acid tryptophan. Niacin is an essential component of the coenzymes nicotinamide adenine dinucleotide (NAD) and nicotinamide adenine dinucleotide phosphate (NADP), which are involved in many oxidation-reduction reactions. The major food sources of niacin are protein foods containing tryptophan and numerous cereals, vegetables, and dairy products.
Niacin in the form of nicotinic acid is used therapeutically for the treatment of hypercholesterolemia and hypertriglyceridemia. Daily doses of 3 6 g can result in significant reductions in levels of LDL and very-low-density lipoproteins (VLDL) and in elevation of HDL. Niacinamide (the form of niacin usually used to treat niacin deficiency) does not exhibit the lipid-lowering effects of nicotinic acid.
Clinical Findings
Historically, niacin deficiency occurred when corn, which is relatively deficient in both tryptophan and niacin, was the major source of calories. Currently, niacin deficiency is more commonly due to alcoholism and nutrient-drug interactions. Niacin deficiency can also occur in inborn errors of metabolism.
As with other B vitamins, the early manifestations of niacin deficiency are nonspecific. Common complaints include anorexia, weakness, irritability, mouth soreness, glossitis, stomatitis, and weight loss. More advanced deficiency results in the classic triad of pellagra: dermatitis, diarrhea, and dementia. The dermatitis is symmetric, involving sun-exposed areas. Skin lesions are dark, dry, and scaling. The dementia begins with insomnia, irritability, and apathy and progresses to confusion, memory loss, hallucinations, and psychosis. The diarrhea can be severe and may result in malabsorption due to atrophy of the intestinal villi. Advanced pellagra can result in death.
Diagnosis
In advanced cases, the diagnosis of pellagra can be made on clinical grounds. In early deficiency, diagnosis requires a high index of suspicion and attempts at confirmation of niacin deficiency. Niacin metabolites, particularly N-methylnicotinamide, can be measured in the urine. Low levels suggest niacin deficiency but may also be found in patients with generalized undernutrition.
P.1300
Serum and red cell levels of NAD and NADP are also low but are similarly nonspecific.
Treatment
Niacin deficiency can be effectively treated with oral niacin, usually given as nicotinamide. Doses ranging from 10 mg/d to 150 mg/d have been used without difficulty.
Niacin Toxicity
At the high doses of niacin used to treat hyperlipidemia, side effects are common. These include cutaneous flushing (partially prevented by pretreatment with aspirin, 325 mg/d, and use of extended-release preparations) and gastric irritation. Elevation of liver enzymes, hyperglycemia, and gout are less common untoward effects.
Canner PL et al: Benefits of niacin in patients with versus without the metabolic syndrome and healed myocardial infarction (from the Coronary Drug Project). Am J Cardiol 2006; 97:477.
Krapels IP et al: Maternal dietary B vitamin intake, other than folate, and the association with orofacial cleft in the offspring. Eur J Nutr 2004;43:7.
Miller M: Niacin as a component of combination therapy for dyslipidemia. Mayo Clin Proc 2003;78:735.
Pitsavas S et al: Pellagra encephalopathy following B-complex vitamin treatment without niacin. Int J Psychiatry Med 2004;34:91.
Whitney EJ et al: A randomized trial of a strategy for increasing high-density lipoprotein cholesterol levels: effects on progression of coronary heart disease and clinical events. Ann Intern Med 2005;142:95.
Vitamin B6 Deficiency
Clinical Findings
Vitamin B6 deficiency most commonly occurs as a result of interactions with medications especially isoniazid, cycloserine, penicillamine, and oral contraceptives or of alcoholism. A number of inborn errors of metabolism and other pyridoxine-responsive syndromes, particularly pyridoxine-responsive anemia, are not clearly due to vitamin deficiency but commonly respond to high doses of the vitamin.
Vitamin B6 deficiency results in a clinical syndrome similar to that seen with deficiencies of other B vitamins, including mouth soreness, glossitis, cheilosis, weakness, and irritability. Severe deficiency can result in peripheral neuropathy, anemia, and seizures. Recent studies have suggested a potential relationship of low vitamin B6 levels and a variety of clinical conditions including cardiovascular diseases, inflammatory diseases, and certain cancers.
Diagnosis
The diagnosis of vitamin B6 deficiency can be confirmed by measurement of pyridoxal phosphate in blood. Normal levels are greater than 50 ng/mL.
Treatment
Vitamin B6 deficiency can be effectively treated with oral vitamin B6 supplements. Doses of 10 20 mg/d are usually adequate, though some patients taking medications that interfere with pyridoxine metabolism may need doses as high as 100 mg/d. Inborn errors of metabolism and the pyridoxine-responsive syndromes often require doses up to 600 mg/d.
Vitamin B6 should be routinely prescribed for patients receiving medications (such as isoniazid) that interfere with pyridoxine metabolism to prevent vitamin B6 deficiency. This is particularly true for elderly patients, the urban poor, and alcoholics, who are more likely to have diets marginally adequate in vitamin B6.
Vitamin B6 Toxicity
A sensory neuropathy, at times irreversible, occurs in patients receiving large doses of vitamin B6. Although most patients have taken 2 g or more per day, some patients have taken only 200 mg/d.
Chiang EP et al: Abnormal vitamin B6 status is associated with severity of symptoms in patients with rheumatoid arthritis. Am J Med 2003;114:283.
Friso S et al: Low plasma vitamin B6 concentrations and modulation of coronary artery disease risk. Am J Clin Nutr 2004; 79:992.
Stott DJ et al: Randomized controlled trial of homocysteine-lowering vitamin treatment in elderly patients with vascular disease. Am J Clin Nutr 2005;82:1320.
Toole JF et al: Lowering homocysteine in patients with ischemic stroke to prevent recurrent stroke, myocardial infarction, and death: the Vitamin Intervention for Stroke prevention (VISP) randomized controlled trial. JAMA 2004;291:565.
Vitamin B12 & Folate
Vitamin B12 (cobalamin) and folate are discussed in Chapter 13. Vitamin B12 is abundant in meat and dairy products; fresh fruits and vegetables supply folic acid.
Hvas AM et al: Vitamin B12 treatment has limited effect on health-related quality of life among individuals with elevated plasma methylmalonic acid: a randomized placebo-controlled study. J Intern Med 2003;253:146.
Lange H et al: Folate therapy and in-stent restenosis after coronary stenting. N Engl J Med 2004;350:2673.
Nakamura K et al: Vitamin B12 deficiency. Arch Neurol 2004;61:960.
Quadri P et al: Homocysteine, folate, and vitamin B12 in mild cognitive impairment, Alzheimer disease, and vascular dementia. Am J Clin Nutr 2004;80:114.
Roth M et al: Oral vitamin B12 therapy in vitamin B12 deficiency. Am J Med 2004;116:358.
van Oijen MG et al: Association of aspirin use with vitamin B12 deficiency (results of the BACH study). Am J Cardiol 2004; 94:975.
P.1301
Vitamin C (Ascorbic Acid) Deficiency
Clinical Findings
Most cases of vitamin C deficiency seen in the United States are due to dietary inadequacy in the urban poor, the elderly, and chronic alcoholics. Patients with chronic illnesses such as cancer and chronic renal failure and individuals who smoke cigarettes are also at risk.
Early manifestations of vitamin C deficiency are nonspecific and include malaise and weakness. In more advanced stages, the typical features of scurvy develop. Manifestations include perifollicular hemorrhages, perifollicular hyperkeratotic papules, petechiae and purpura, splinter hemorrhages, bleeding gums, hemarthroses, and subperiosteal hemorrhages. Periodontal signs do not occur in edentulous patients. Anemia is common, and wound healing is impaired. The late stages of scurvy are characterized by edema, oliguria, neuropathy, intracerebral hemorrhage, and death.
Diagnosis
The diagnosis of advanced scurvy can be made clinically on the basis of the skin lesions in the proper clinical situation. Atraumatic hemarthrosis is also highly suggestive. The diagnosis can be confirmed with decreased plasma ascorbic acid levels, typically below 0.1 mg/dL.
Treatment
Adult scurvy can be treated with 300 1000 mg of ascorbic acid per day. Improvement typically occurs within days. Some studies have suggested that high intakes of vitamin C are associated with a decreased risk of cancer. Vitamin C may also protect against coronary heart disease by modifying blood cholesterol levels and preventing LDL cholesterol from oxidation. A decrease in all-cause and coronary heart disease mortality in individuals with high intakes (approximately 300 400 mg/d) of vitamin C has been reported.
Vitamin C Toxicity
Very large doses of vitamin C can cause gastric irritation, flatulence, or diarrhea. Oxalate kidney stones are of theoretic concern because ascorbic acid is metabolized to oxalate, but stone formation has not been frequently reported. Vitamin C can also confound common diagnostic tests by causing false-negative tests for fecal occult blood and both false-negative and false-positive tests for urine glucose.
Hampl JS et al: Vitamin C deficiency and depletion in the United States: the Third National Health and Nutrition Examination Survey, 1988 to 1994. Am J Public Health 2004;94: 870.
Salonen RM et al: Antioxidant Supplementation in Atherosclerosis Prevention Study. Six-year effect of combined vitamin C and E supplementation on atherosclerotic progression: the Antioxidant Supplementation in Atherosclerosis Prevention (ASAP) Study. Circulation 2003;107:947.
Shekelle P et al: Effect of supplemental antioxidants vitamin C, vitamin E, and coenzyme Q for the prevention and treatment of cardiovascular disease. Evid Rep Technol Assess (sum). 2003;1 3. http://www.ahrq.gov
Traxer O et al: Effect of ascorbic acid consumption on urinary stone risk factors. J Urol 2003;170(2 Pt 1):397.
Vitamin A Deficiency
Clinical Findings
Vitamin A deficiency is one of the most common vitamin deficiency syndromes, particularly in developing countries. In many such regions, it is the most common cause of blindness. In the United States, vitamin A deficiency is usually due to fat malabsorption syndromes or mineral oil laxative abuse and occurs most commonly in the elderly and urban poor.
Night blindness is the earliest symptom. Dryness of the conjunctiva (xerosis) and the development of small white patches on the conjunctiva (Bitot's spots) are early signs. Ulceration and necrosis of the cornea (keratomalacia), perforation, endophthalmitis, and blindness are late manifestations. Xerosis and hyperkeratinization of the skin and loss of taste may also occur.
Diagnosis
Abnormalities of dark adaptation are strongly suggestive of vitamin A deficiency. Serum levels below the normal range of 30 65 mg/dL are commonly seen in advanced deficiency.
Treatment
Night blindness, poor wound healing, and other signs of early deficiency can be effectively treated with 30,000 international units of vitamin A daily for 1 week. Advanced deficiency with corneal damage calls for administration of 20,000 units/kg for at least 5 days. The potential antioxidant effects of -carotene can be achieved with supplements of 25,000 50,000 international units of -carotene.
Vitamin A Toxicity
Excess intake of -carotenes (hypercarotenosis) results in staining of the skin a yellow-orange color but is otherwise benign. Skin changes are most marked on the palms and soles, while the scleras remain white, clearly distinguishing hypercarotenosis from jaundice.
Excessive vitamin A (hypervitaminosis A), on the other hand, can be quite toxic. Chronic toxicity usually occurs after ingestion of daily doses of over 50,000 units/d for more than 3 months. Early manifestations include dry, scaly skin, hair loss, mouth sores, painful hyperostoses, anorexia, and vomiting. More serious findings include hypercalcemia; increased intracranial
P.1302
pressure, with papilledema, headaches, and decreased cognition; and hepatomegaly, occasionally progressing to cirrhosis. Excessive vitamin A has also recently been related to increased risk of hip fracture. Acute toxicity can result from ingestion of massive doses of vitamin A, such as in drug overdoses or consumption of polar bear liver. Manifestations include nausea, vomiting, abdominal pain, headache, papilledema, and lethargy.
The diagnosis can be confirmed by elevations of serum vitamin A levels. The only treatment is withdrawal of vitamin A from the diet. Most symptoms and signs improve rapidly.
Baeten JM et al: Use of serum retinol-binding protein for prediction of vitamin A deficiency: effects of HIV-1 infection, protein malnutrition, and the acute phase response. Am J Clin Nutr 2004;79:218.
Michaelsson K et al: Serum retinol levels and the risk of fracture. N Engl J Med 2003;348:287.
Wiysonge CS et al: Vitamin A supplementation for reducing the risk of mother-to-child transmission of HIV infection. Cochrane Database Syst Rev 2005;(4):CD003648.
Vitamin D
Vitamin D is discussed in Chapter 26. The major food source of vitamin D is fortified milk, but sunlight on the skin is a prime resource as well.
Gartner LM et al: Prevention of rickets and vitamin D deficiency: new guidelines for vitamin D intake. Pediatrics 2003;111(4 Pt 1):908.
Jackson RD et al; Women's Health Initiative Investigators: Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med 2006;354:669.
Tajika M et al: Risk factors for vitamin D deficiency in patients with Crohn's disease. J Gastroenterol 2004;39:527.
Wactawski-Wende J et al; Women's Health Initiative Investigators: Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med 2006;354:684.
Vitamin E Deficiency
Clinical Findings
Clinical deficiency of vitamin E is most commonly due to severe malabsorption, the genetic disorder abetalipoproteinemia, or, in children with chronic cholestatic liver disease, biliary atresia or cystic fibrosis. Manifestations of deficiency include areflexia, disturbances of gait, decreased proprioception and vibration, and ophthalmoplegia.
Diagnosis
Plasma vitamin E levels can be measured; normal levels are 0.5 0.7 mg/dL or higher. Since vitamin E is normally transported in lipoproteins, the serum level should be interpreted in relation to circulating lipids.
Treatment
The optimum therapeutic dose of vitamin E has not been clearly defined. Large doses, often administered parenterally, can be used to improve the neurologic complications seen in abetalipoproteinemia and cholestatic liver disease. The potential antioxidant benefits of vitamin E can be achieved with supplements of 100 400 units/d. Clinical trials of supplemental vitamin E to prevent cardiovascular disease, however, have shown no beneficial effects.
Vitamin E Toxicity
Vitamin E is the least toxic of the fat-soluble vitamins. Large doses many times the recommended daily requirement have been taken for extended periods of time without apparent harm, though nausea, flatulence, and diarrhea have been reported. Large doses of vitamin E can increase the vitamin K requirement and can result in bleeding in patients taking oral anticoagulants.
Eidelman RS et al: Randomized trials of vitamin E in the treatment and prevention of cardiovascular disease. Arch Intern Med 2004;164:1552.
Lee IM et al: Vitamin E in the primary prevention of cardiovascular disease and cancer: the Women's Health Study: a randomized controlled trial. JAMA 2005;294:56.
Maras JE et al: Intake of alpha-tocopherol is limited among US adults. J Am Diet Assoc 2004;104:567.
Meydani SN et al: Vitamin E and respiratory tract infections in elderly nursing home residents: a randomized controlled trial. JAMA 2004;292:828.
Miller ER 3rd et al: Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med 2005;142:37.
Vitamin K
Vitamin K is discussed in Chapter 13. Vitamin K is synthesized by intestinal bacteria.
Diet Therapy
Specific therapeutic diets can be designed to facilitate the medical management of most common illnesses. In most cases, consultation with a registered dietitian is necessary in order to design and implement major dietary changes. Physicians should be familiar with the indications for special diets and their basic composition to facilitate patient referrals and to maximize patient compliance. Diet therapy is a difficult process, and not all patients are able to cooperate fully. Requesting the patient to record dietary intake for 3 5 days may provide useful insight into the patient's motivation.
Therapeutic diets can be divided into three groups: (1) diets that alter the consistency of food, (2) diets
P.1303
that restrict or otherwise modify dietary components, and (3) diets that supplement dietary components.
Diets that Alter Consistency
Clear Liquid Diet
This diet provides adequate water, 500 1000 kcal as simple sugar, and some electrolytes. It is fiber free and requires minimal digestion or intestinal motility.
A clear liquid diet is useful for patients with resolving postoperative ileus, acute gastroenteritis, partial intestinal obstruction, and as preparation for diagnostic gastrointestinal procedures. It is commonly used as the first diet for patients who have been taking nothing by mouth for long periods. Because of the low calorie and minimal protein content of the clear liquid diet, it is used only for short periods.
Full Liquid Diet
The full liquid diet provides adequate water and can be designed to provide adequate calories and protein. Vitamins and minerals especially folic acid, iron, and vitamin B6 may be inadequate and should be provided in the form of supplements. Dairy products, soups, eggs, and soft cereals are used to supplement clear liquids. Commercial oral supplements can also be incorporated into the diet or used alone.
This diet is low in residue and can be used in many instances instead of the clear liquid diet described above especially in patients with difficulty in chewing or swallowing, with partial obstructions, or in preparation for some diagnostic procedures. Full liquid diets are commonly used following clear liquid diets to advance diets in patients who have been taking nothing by mouth for long periods.
Soft Diets
Soft diets are designed for patients unable to chew or swallow hard or coarse food. Tender foods are used, and most raw fruits and vegetables and coarse breads and cereals are eliminated. Soft diets are commonly used to assist in progression from full liquid diets to regular diets in postoperative patients, in patients who are too weak or those whose dentition is too poor to handle a general diet, in head and neck surgical patients, in patients with esophageal strictures, and in other patients who have difficulty with chewing or swallowing.
The soft diet can be designed to meet all nutritional requirements.
Diets that Restrict Nutrients
Diets can be designed to restrict (or eliminate) virtually any nutrient or food component. The most commonly used restricted diets are those that limit sodium, fat, and protein. Other restrictive diets include gluten restriction in sprue, potassium and phosphate reduction in renal insufficiency, and various elimination diets for food allergies.
Sodium-Restricted Diets
Low-sodium diets are useful in the management of hypertension and in conditions in which sodium retention and edema are prominent features, particularly congestive heart failure, chronic liver disease, and chronic renal failure. Sodium restriction is beneficial with or without diuretic therapy. When used in conjunction with diuretics, sodium restriction allows lower dosage of the diuretic medication and may prevent side effects. Potassium excretion, in particular, is directly related to distal renal tubule sodium delivery, and sodium restriction will decrease diuretic-related potassium losses.
Typical American diets contain a minimum of 4 6 g (175 260 mEq) of sodium per day. A no-added-salt diet contains approximately 3 g (132 mEq) of sodium per day. Further restriction can be achieved with sodium diets of 2 or 1 g/d. Diets with more severe restriction are poorly accepted by patients and are rarely used.
Dietary sodium includes sodium naturally occurring in foods, sodium added during food processing, and sodium added by the consumer during cooking and at the table. About a third of current dietary intake is derived from each. Diets that allow 2000 mg of sodium daily are easiest to design and implement. Such diets generally eliminate added salt, most processed foods, and selected foods with particularly high sodium content. Patients who follow such diets for 2 3 months lose their craving for salty foods and can often continue to restrict their sodium intake indefinitely. Many patients with mild hypertension will achieve significant reductions in blood pressure (approximately 5 mm Hg diastolic) with this degree of sodium restriction. Other patients require more severe sodium restriction (approximately 1000 mg of sodium per day) for reduction in blood pressure.
Diets allowing 1000 mg of sodium require further restriction of commonly eaten foods. Special low-sodium products are now available to facilitate such diets. These diets are difficult for most people to follow and are generally reserved for hospitalized patients and highly motivated outpatients most commonly those with severe liver disease and ascites.
Fat-Restricted Diets
Traditional fat-restricted diets are useful in the treatment of fat malabsorption syndromes. Such diets will improve the symptoms of diarrhea with steatorrhea independently of the primary physiologic abnormality by limiting the quantity of fatty acids that reach the colon. The degree of fat restriction necessary to control symptoms must be individualized. Patients with severe malabsorption can be limited to 40 60 g of fat per day. Diets containing 60 80 g of fat per day can be designed for patients with less severe abnormalities.
P.1304
In general, fat-restricted diets require broiling, baking, or boiling meat and fish; discarding the skin of poultry and fish and using those foods as the main protein source; using nonfat dairy products; and avoiding desserts, sauces, and gravies.
Low-Cholesterol, Low-Saturated-Fat Diets
Fat-restricted diets that specifically restrict saturated fats and dietary cholesterol are the mainstay of dietary treatment of hyperlipidemia (see Chapter 28). Similar diets are recommended also for diabetes (see Chapter 27) and for the prevention of coronary artery disease (see Chapter 10). Current recommendations for the prevention of cancer by dietary modification also include fat restriction. The large Women's Health Initiative Dietary Modification Trial, however, did not show any significant benefit of a low-fat diet on weight control or prevention of cardiovascular disease or cancer.
The aim of these diets is to restrict total fat to less than 30% of calories and to achieve a normal body weight by caloric restriction and increased physical activity. Saturated fat is restricted to 7% of calories and dietary cholesterol to 200 mg/d. Saturated fat can be replaced either with complex carbohydrates or, if energy balance permits, with monounsaturated fats. Saturated fat, total fat, and dietary cholesterol can be restricted further, but studies suggest that more extreme restriction offers little further advantage in overall modification of serum lipids. Cholesterol-lowering diets can be further augmented with the addition of plant stanols and sterols and with soluble dietary fiber.
Protein-Restricted Diets
Protein-restricted diets are most commonly used in patients with hepatic encephalopathy due to chronic liver disease and in patients with renal failure to slow the progression of early disease and to decrease symptoms of uremia in more severe disease. Patients with selected inborn errors of amino acid metabolism and other abnormalities resulting in hyperammonemia also require restriction of protein or of specific amino acids.
Protein restriction is intended to limit the production of nitrogenous waste products. Energy intake must be adequate to facilitate the efficient use of dietary protein. Proteins must be of high biologic value and be provided in sufficient quantity to meet minimal requirements. For most patients, the diet should contain at least 0.6 g/kg/d of protein. Patients with encephalopathy who do not respond to this degree of restriction are unlikely to respond to more severe restriction.
Diets that Supplement Nutrients
High-Fiber Diet
Dietary fiber is a diverse group of plant constituents that is resistant to digestion by the human digestive tract. Typical American diets contain about 5 10 g of dietary fiber per day. Epidemiologic evidence has suggested that populations consuming greater quantities of fiber have a lower incidence of certain gastrointestinal disorders, including diverticulitis and colon cancer. Most authorities currently recommend higher intakes of dietary fiber for health maintenance.
Diets high in dietary fiber (20 35 g/d) are also commonly used in the management of a variety of gastrointestinal disorders, particularly irritable bowel syndrome and recurrent diverticulitis. Diets high in fiber may also be useful to reduce blood sugar in patients with diabetes and to reduce cholesterol levels in patients with hypercholesterolemia. Such diets include greater intakes of fresh fruits and vegetables, whole grains, legumes and seeds, and bran products. For some patients, the addition of psyllium seed (2 tsp per day) or natural bran (one-half cup per day) may be preferable.
High-Potassium Diets
Potassium-supplemented diets are used most commonly to compensate for potassium losses caused by diuretics. Although potassium losses can be partially prevented by using lower doses of diuretics, concurrent sodium restriction, and potassium-sparing diuretics, some patients require additional potassium to prevent hypokalemia. High-potassium diets may also have a direct antihypertensive effect. Typical American diets contain about 3 g (80 mEq) of potassium per day. High-potassium diets commonly contain 4.5 7 g (120 180 mEq) of potassium per day.
Most fruits, vegetables, and their juices contain high concentrations of potassium. Supplemental potassium can also be provided with potassium-containing salt substitutes (up to 20 mEq in one-quarter tsp) or as potassium chloride in solution or capsules, but this is rarely necessary if the above measures are followed to prevent potassium losses and supplement dietary potassium.
High-Calcium Diets
Additional intakes of dietary calcium have recently been recommended for the prevention of postmenopausal osteoporosis, the prevention and treatment of hypertension, and the prevention of colon cancer. Although the evidence in each case is preliminary, authorities recommend intakes of 1 g of calcium per day for most adults and 1.5 g/d for postmenopausal women. Average American daily intakes are approximately 700 mg/d.
Low-fat and nonfat dairy products are the mainstay of supplemental calcium intakes. Patients with lactose intolerance who cannot tolerate liquid dairy products may be able to tolerate nonliquid products such as cheese and yogurt. Leafy green vegetables and canned fish with bones also contain high concentrations of calcium, although the latter is also very high in sodium.
Chao A et al: Meat consumption and risk of colorectal cancer. JAMA 2005;293:172.
P.1305
Esposito K et al: Effect of a Mediterranean-style diet on endothelial dysfunction and markers of vascular inflammation in the metabolic syndrome: a randomized trial. JAMA 2004;292:1440.
Gannon MC et al: Effect of a high-protein, low-carbohydrate diet on blood glucose control in people with type 2 diabetes. Diabetes 2004;53:2375.
Gardner CD et al: The effect of a plant-based diet on plasma lipids in hypercholesterolemic adults: a randomized trial. Ann Intern Med 2005;142:725.
Gerhard GT et al: Effects of a low-fat diet compared with those of a high-monounsaturated fat diet on body weight, plasma lipids and lipoproteins, and glycemic control in type 2 diabetes. Am J Clin Nutr 2004;80:668.
Howard BV et al: Low-fat dietary pattern and weight change over 7 years: the Women's Health Initiative Dietary Modification Trial. JAMA 2006;295:39.
Howard BV et al: Low-fat dietary pattern and risk of cardiovascular disease: the Women's Health Initiative Randomized Controlled Dietary Modification Trial. JAMA 2006;295:655.
Jackson RD et al: Calcium plus vitamin D supplementation and the risk of fractures. N Engl J Med 2006;354:669.
Little P et al: Randomized controlled factorial trial of dietary advice for patients with a single high blood pressure reading in primary care. BMJ 2004;328:1054.
MacLean et al: Effects of omega-3 fatty acids on cancer risk: A systematic review. JAMA 2006;295:403.
Moore H et al: Dietary advice for treatment of type 2 diabetes mellitus in adults. Cochrane Database Syst Rev 2004;(3):CD004097.
Park Y et al: Dietary fiber intake and risk of colorectal cancer: A pooled analysis of prospective cohort studies. JAMA 2005;294:2849.
Whelton SP et al: Meta-analysis of observational studies on fish intake and coronary heart disease. Am J Cardiol 2004;93:1119.
Nutritional Support
Nutritional support is the provision of nutrients to patients who cannot meet their nutritional requirements by eating standard diets. Nutrients may be delivered enterally, using oral nutritional supplements, nasogastric and nasoduodenal feeding tubes, and tube enterostomies, or parenterally, using lines or catheters placed in peripheral or central veins, respectively. Current nutritional support techniques permit adequate nutrient delivery to most patients. Nutrition support should be utilized, however, only if it is likely to improve the patient's clinical outcome. The financial costs and risks of side effects must be balanced against the potential advantages of improved nutritional status in each clinical situation.
Indications for Nutritional Support
The precise indications for nutritional support remain controversial. Most authorities agree that nutritional support is indicated for at least four groups of adult patients: (1) those with inadequate bowel syndromes, (2) those with severe prolonged hypercatabolic states (eg, due to extensive burns, multiple trauma, mechanical ventilation), (3) those requiring prolonged therapeutic bowel rest, and (4) those with severe protein-calorie undernutrition with a treatable disease who have sustained a loss of over 25% of body weight.
It has been difficult to prove the efficacy of nutritional support in the treatment of most other conditions. In most cases it has not been possible to show a clear advantage of treatment by means of nutritional support over treatment without such support.
The American Society for Parenteral and Enteral Nutrition (ASPEN) has published recommendations for the rational use of nutritional support. The recommendations emphasize the need to individualize the decision to begin nutritional support, weighing the risks and costs against the benefits to each patient. They also reinforce the need to identify high-risk malnourished patients by nutritional assessment.
Nutritional Support Methods
Selection of the most appropriate nutritional support method involves consideration of gastrointestinal function, the anticipated duration of nutritional support, and the ability of each method to meet the patient's nutritional requirements. The method chosen should meet the patient's nutritional needs with the lowest risk and lowest cost possible. For most patients, enteral feeding is safer and cheaper and offers significant physiologic advantages. An algorithm for selection of the most appropriate nutritional support method is presented in Figure 29-1.
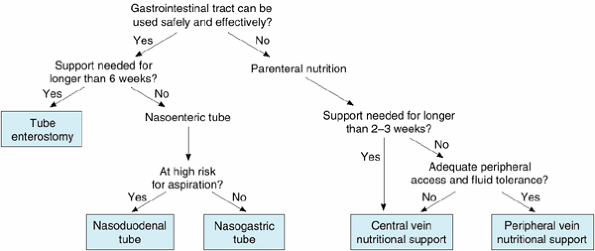 |
Figure 29-1. Nutritional support method decision tree. |
Prior to initiating specialized enteral nutritional support, efforts should be made to supplement food intake. Attention to patient preferences, timing of meals and diagnostic procedures and use of medications, and the use of foods brought to the hospital by family and friends can often increase oral intake. Patients unable to eat enough at regular mealtimes to meet nutritional requirements can be given oral supplements as snacks or to replace low-calorie beverages. Oral supplements of differing nutritional composition are available for the purpose of individualizing the diet in accordance with specific clinical requirements (see below). Fiber and lactose content, caloric density, protein level, and amino acid profiles can all be modified as necessary.
Patients unable to take adequate oral nutrients who have functioning gastrointestinal tracts and who meet the criteria for nutritional support are candidates for tube feedings. Small-bore feeding tubes are placed via the nose into the stomach or duodenum. Patients able to sit up in bed who can protect their airways can be fed into the stomach. Because of the increased risk of aspiration, patients who cannot adequately protect their airways should be fed nasoduodenally. Feeding tubes can usually be passed into the duodenum by
P.1306
leaving an extra length of tubing in the stomach and placing the patient in the right decubitus position. Metoclopramide, 10 mg intravenously, can be given 20 minutes prior to insertion and continued every 6 hours thereafter to facilitate passage through the pylorus. Occasionally patients will require fluoroscopic or endoscopic guidance to insert the tube distal to the pylorus. Placement of nasogastric and, particularly, nasoduodenal tubes should be confirmed radiographically before delivery of feeding solutions.
Feeding tubes can also be placed directly into the gastrointestinal tract using tube enterostomies. Most tube enterostomies are placed in patients who require long-term enteral nutritional support. Gastrostomies have the advantage of allowing bolus feedings, while jejunostomies require continuous infusions. Gastrostomies like nasogastric feeding should be used only in patients at low risk for aspiration. Gastrostomies can also be placed percutaneously with the aid of endoscopy. These tubes can then be advanced to jejunostomies. Tube enterostomies can also be placed surgically.
Patients who require nutritional support but whose gastrointestinal tracts are nonfunctional should receive parenteral nutritional support. Most patients receive parenteral feedings via a central vein most commonly the subclavian vein. Peripheral veins can be used in some patients, but because of the high osmolality of parenteral solutions this is rarely tolerated for more than a few weeks.
Peripheral vein nutritional support is most commonly used in patients with nonfunctioning gastrointestinal tracts who require immediate support but whose clinical status is expected to improve within 1 2 weeks, allowing enteral feeding. Peripheral vein nutritional support is administered via standard intravenous lines. Solutions should always include lipid and dextrose in combination with amino acids to provide adequate nonprotein calories. Serious side effects are infrequent, but there is a high incidence of phlebitis and infiltration of intravenous lines.
Central vein nutritional support is delivered via intravenous catheters placed percutaneously using aseptic technique. Proper placement in the superior vena cava is documented radiographically before the solution is infused. Catheters must be carefully maintained by experienced nursing personnel and used solely for nutritional support to prevent infection and other catheter-related complications.
Nutritional Requirements
Each patient's nutritional requirements should be determined independently of the method of nutritional support. In most situations, solutions of equal nutrient value can be designed for delivery via enteral and parenteral routes, but differences in absorption must be considered. A complete nutritional support solution must contain water, energy, amino acids, electrolytes, vitamins, minerals, and essential fatty acids.
Water
For most patients, water requirements can be calculated by allowing 1500 mL for the first 20 kg of body weight plus 20 mL for every kilogram over 20. Additional losses should be replaced as they occur. For average-sized adult patients, fluid needs are about 30 35 mL/kg, or approximately 1 mL/kcal of energy required (see below).
Energy
Energy requirements can be estimated by one of three methods: (1) by using standard equations to calculate BEE plus additional calories for activity and illness, (2) by applying a simple calculation based on calories per kilogram of body weight, or (3) by measuring energy expenditure with indirect calorimetry.
P.1307
BEE can be estimated by the Harris-Benedict equation: for men, BEE = 666 + (13.7 weight in kg) + (5 height in cm) (6.8 age in years). For women, BEE = 655 + (9.5 weight in kg) + (1.8 height in cm) (4.7 age in years). For undernourished patients, actual body weight should be used; for obese patients, ideal body weight should be used. For most patients, an additional 20 50% of BEE is administered as nonprotein calories to accommodate energy expenditures during activity or relating to the illness. Occasional patients are noted to have energy expenditures greater than 150% of BEE.
Energy requirements can be estimated also by multiplying actual body weight in kilograms (for obese patients, ideal body weight) by 30 35 kcal.
Both of these methods provide imprecise estimates of actual energy expenditures, especially for the markedly underweight, overweight, and critically ill patient. Studies using indirect calorimetry have demonstrated that as many as 30 40% of patients will have measured expenditures 10% above or below estimated values. For accurate determination of energy expenditure, indirect calorimetry should be used.
Protein
Protein and energy requirements are closely related. If adequate calories are provided, most patients can be given 0.8 1.2 g of protein per kilogram per day. Patients undergoing moderate to severe stress should receive up to 1.5 g/kg/d. As in the case of energy requirements, actual weights should be used for normal and underweight patients and ideal weights for patients with significant obesity.
Patients who are receiving protein without adequate calories will catabolize protein for energy rather than utilizing it for protein synthesis. Thus, when energy intake is low, excess protein is needed for nitrogen balance. If both energy and protein intakes are low, extra energy will have a more significant positive effect on nitrogen balance than extra protein.
Electrolytes & Minerals
Requirements for sodium, potassium, and chloride vary widely. Most patients require 45 145 mEq/d of each. The actual requirement in individual patients will depend on the patient's cardiovascular, renal, endocrine, and gastrointestinal status as well as measurements of serum concentration.
Patients receiving enteral nutritional support should receive adequate vitamins and minerals according to the recommended daily allowances. Most premixed enteral solutions provide adequate vitamins and minerals as long as adequate calories are administered.
Patients receiving parenteral nutritional support require smaller amounts of minerals: calcium, 10 15 mEq/d; phosphorus, 15 20 mEq per 1000 nonprotein calories; and magnesium, 16 24 mEq/d. Most patients receiving nutritional support do not require supplemental iron because body stores are adequate. Iron nutrition should be monitored closely by following the hemoglobin concentration, mean corpuscular volume, and iron studies. Parenteral administration of iron is associated with a number of adverse effects and should be reserved for iron-deficient patients unable to take oral iron.
Patients receiving parenteral nutritional support should be given the trace elements zinc (about 5 mg/d) and copper (about 2 mg/d). Patients with diarrhea will require additional zinc to replace fecal losses. Additional trace elements especially chromium, manganese, and selenium are provided to patients receiving long-term parenteral nutrition.
Parenteral vitamins are provided daily. Standardized multivitamin solutions are currently available to provide adequate quantities of vitamins A, B12, C, D, E, thiamine, riboflavin, niacin, pantothenic acid, pyridoxine, folic acid, and biotin. Vitamin K is not given routinely but administered when the prothrombin time becomes abnormal.
Essential Fatty Acids
Patients receiving nutritional support should be given 2 4% of their total calories as linoleic acid to prevent essential fatty acid deficiency. Most prepared enteral solutions contain adequate linoleic acid. Patients receiving parenteral nutrition should be given at least 250 mL of a 20% intravenous fat (emulsified soybean or safflower oil) about two or three times a week. Intravenous fat can also be used as an energy source in place of dextrose.
Enteral Nutritional Support Solutions
Most patients who require enteral nutritional support can be given commercially prepared enteral solutions (Table 29-10). Nutritionally complete solutions have been designed to provide adequate proportions of water, energy, protein, and micronutrients. Nutritionally incomplete solutions are also available to provide specific macronutrients (eg, protein, carbohydrate, and fat) to supplement complete solutions for patients with unusual requirements or to design solutions that are not available commercially.
Table 29-10. Enteral solutions. | ||
|---|---|---|
|
Nutritionally complete solutions are characterized as follows: (1) by osmolality (isotonic or hypertonic), (2) by lactose content (present or absent), (3) by the molecular form of the protein component (intact proteins; peptides or amino acids), (4) by the quantity of protein and calories provided, and (5) by fiber content (present or absent). For most patients, isotonic solutions containing no lactose or fiber are preferable. Such solutions generally contain moderate amounts of fat and intact protein. Most commercial isotonic solutions contain 1000 kcal and about 37 45 g of protein per liter.
Solutions containing hydrolyzed proteins or crystalline amino acids and with no significant fat content
P.1308
are called elemental solutions, since macronutrients are provided in their most elemental form. These solutions have been designed for patients with malabsorption, particularly pancreatic insufficiency and limited fat absorption. Elemental diets are extremely hypertonic and often result in more severe diarrhea. Their use should be limited to patients who cannot tolerate isotonic solutions.
Although formulas have been designed for specific clinical situations solutions containing primarily essential amino acids (for renal failure), medium-chain triglycerides (for fat malabsorption), more fat (for respiratory failure and CO2 retention), and more branched-chain amino acids (for hepatic encephalopathy and severe trauma) they have not been shown to be superior to standard formulas for most patients.
Enteral solutions should be administered via continuous infusion, preferably with an infusion pump. Isotonic feedings should be started at full strength at about 25 33% of the estimated final infusion rate. Feedings can be advanced by similar amounts every 12 hours as tolerated. Hypertonic feedings should be started at half strength. The strength and the rate can then be advanced every 6 hours as tolerated.
Complications of Enteral Nutritional Support
Minor complications of tube feedings occur in 10 15% of patients. Gastrointestinal complications include diarrhea (most common), inadequate gastric emptying, emesis, esophagitis, and occasionally gastrointestinal bleeding. Diarrhea associated with tube feeding may be due to intolerance to the osmotic load or to one of the macronutrients (eg, fat, lactose) in the solution. Patients being fed in this way may also have diarrhea from other causes (as side effects of antibiotics or other drugs, associated with infection, etc), and these possibilities should always be investigated in appropriate circumstances.
Mechanical complications of tube feedings are potentially the most serious. Of particular importance is aspiration. All patients receiving nasogastric tube feedings are at risk for this life-threatening complication. Limiting nasogastric feedings to those patients who can adequately protect their airway and careful monitoring of patients being fed by tube should limit these serious complications to 1 2% of cases. Minor mechanical complications are common and include tube obstruction and dislodgment.
Metabolic complications during enteral nutritional support are common but in most cases are easily managed. The most important problem is hypernatremic dehydration, most commonly seen in elderly patients given excessive protein intake who are unable to respond to thirst. Abnormalities of potassium, glucose, and acid-base balance may also occur.
Parenteral Nutritional Support Solutions
Parenteral nutritional support solutions can be designed to deliver adequate nutrients to most patients. The basic parenteral solution is composed of dextrose, amino acids, and water. Electrolytes, minerals, trace elements, vitamins, and medications can also be added. Most commercial solutions contain the monohydrate form of dextrose that provides 3.4 kcal/g. Crystalline amino acids are available in a variety of concentrations, so that a broad range of solutions can be made up that will contain specific amounts of dextrose and amino acids as required.
Table 29-11. Typical parenteral nutrition solution (for stable patients without organ failure). | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Typical solutions for central vein nutritional support contain 25 35% dextrose and 2.75 6% amino acids depending upon the patient's estimated nutrient and water requirements. These solutions typically have
P.1309
osmolalities in excess of 1800 mosm/L and require infusion into a central vein. A typical formula for patients without organ failure is shown in Table 29-11.
Solutions with lower osmolalities can also be designed for infusion into peripheral veins. Typical solutions for peripheral infusion contain 5 10% dextrose and 2.75 4.25% amino acids. These solutions have osmolalities between 800 and 1200 mosm/L and result in a high incidence of thrombophlebitis and line infiltration. These solutions will provide adequate protein for most patients but inadequate energy. Additional energy must be provided in the form of emulsified soybean or safflower oil. Such intravenous fat solutions are currently available in 10% and 25% solutions providing 1.1 and 2.2 kcal/mL, respectively. Intravenous fat solutions are isosmotic and well tolerated by peripheral veins. Typical patients are given 200 500 mL of a 20% solution each day. As much as 60% of total calories can be administered in this manner.
Intravenous fat can also be provided to patients receiving central vein nutritional support. In this instance, dextrose concentrations should be decreased to provide a fixed concentration of energy. Intravenous fat has been shown to be equivalent to intravenous dextrose in providing energy to spare protein. Intravenous fat is associated with less glucose intolerance, less production of carbon dioxide, and less fatty infiltration of the liver and has been increasingly utilized in patients with hyperglycemia, respiratory failure, and liver disease. Intravenous fat has also been increasingly used in patients with large estimated energy requirements. Recent studies suggest that the maximum glucose utilization rate is approximately 5 7 mg/min/kg. Patients who require additional calories can be given them as fat to prevent excess administration of dextrose. Intravenous fat can also be used to prevent essential fatty acid deficiency. The optimal ratio of carbohydrate and fat in parenteral nutritional support has not been determined.
Infusion of parenteral solutions should be started slowly to prevent hyperglycemia and other metabolic complications. Typical solutions are given initially at a rate of 50 mL/h and advanced by about the same amount every 24 hours until the desired final rate is reached.
Complications of Parenteral Nutritional Support
Complications of central vein nutritional support occur in up to 50% of patients. Although most are minor and easily managed, significant complications will develop in about 5% of patients. Complications of central vein nutritional support can be divided into catheter-related complications and metabolic complications.
Catheter-related complications can occur during insertion or while the catheter is in place. Pneumothorax, hemothorax, arterial laceration, air emboli, and brachial plexus injury can occur during catheter placement. The incidence of these complications is inversely related to the experience of the physician performing the procedure but will occur in at least 1 2% of cases even in major medical centers. Each catheter placement should be documented by chest radiograph prior to initiation of nutritional support.
Catheter thrombosis and catheter-related sepsis are the most important complications of indwelling catheters. Patients with indwelling central vein catheters in whom fever develops without an apparent source should have their lines changed over a wire or removed immediately, the tip quantitatively cultured, and antibiotics begun empirically. Quantitative tip cultures and blood cultures will help guide further antibiotic therapy. Catheter-related sepsis occurs in 2 3% of patients even if maximal efforts are made to prevent infection.
Metabolic complications of central vein nutritional support occur in over 50% of patients (Table 29-12). Most are minor and easily managed, and termination of support is seldom necessary.
Table 29-12. Metabolic complications of parenteral nutritional support. | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Patient Monitoring During Nutritional Support
Every patient receiving enteral or parenteral nutritional support should be monitored closely. Formal nutritional support teams composed of a physician, a nurse, a dietitian, and a pharmacist have been shown to decrease the rate of complications.
Patients should be monitored both for the adequacy of treatment and to prevent complications or detect them early when they occur. Because estimates
P.1310
of nutritional requirements are imprecise, frequent reassessment is necessary. Daily intakes should be recorded and compared with estimated requirements. Body weight, hydration status, and overall clinical status should be followed. Patients who do not appear to be responding as anticipated can be evaluated for nitrogen balance by means of the following equation:
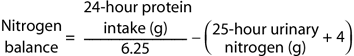
Patients with positive nitrogen balances can be continued on their current regimens; patients with negative balances should receive moderate increases in calorie and protein intake and then be reassessed. Monitoring for metabolic complications includes daily measurements of electrolytes; serum glucose, phosphorus, magnesium, calcium, and creatinine; and BUN until the patient is stabilized. Once the patient is stabilized, electrolytes, phosphorus, calcium, magnesium, and glucose should be obtained at least twice weekly. Red blood cell folate, zinc, and copper should be checked at least once a month.
August D et al: An evidence-based approach to optimal management of vascular and enteral access for home parenteral and enteral nutrition support. JPEN J Parenter Enteral Nutr 2006;30(1 Suppl):S5.
Braga JM et al: Implementation of dietician recommendations for enteral nutrition results in improved outcomes. J Am Diet Assoc 2006;106:281.
Edington J et al: A prospective randomized controlled trial of nutritional supplementation in malnourished elderly in the community: clinical and health economic outcomes. Clin Nutr 2004;23:195.
Farber MS et al: Reducing costs and patient morbidity in the enterally fed intensive care unit patient. J Parenter Enteral Nutr 2005;29(1 Suppl):S62.
Fiaccadori E et al: Enteral nutrition in patients with acute renal failure. Kidney Int 2004;65:999.
Forbes A: Parenteral nutrition. Curr Opin Gastroenterol 2006; 22:160.
Howard L et al: Nutrition in the perioperative patient. Annu Rev Nutr 2003;23:263.
Ireton-Jones C et al: Home parenteral nutrition registry: a five-year retrospective evaluation of outcomes of patients receiving home parenteral nutrition support. Nutrition 2005;21:156.
Jeejeebhoy KN: Enteral feeding. Curr Opin Gastroenterol 2005; 21:187.
Martin CM et al: Southwestern Ontario Critical Care Research Network: Multicentre, cluster-randomized clinical trial of algorithms for critical-care enteral and parenteral therapy (ACCEPT). CMAJ 2004;170:197.
McMahon MM et al: Medical and ethical aspects of long-term enteral tube feeding. Mayo Clin Proc 2005;80:1461.
Messing B et al: Guidelines for management of home parenteral support in adult chronic intestinal failure patients. Gastroenterology 2006;130(2 Suppl 1):S43.
Sayers GM et al: Parenteral nutrition: ethical and legal considerations. Postgrad Med J 2006;82:79.
Soop M et al: Randomized clinical trial of the effects of immediate enteral nutrition on metabolic responses to major colorectal surgery in an enhanced recovery protocol. Br J Surg 2004;91:1138.
Tappenden KA: Mechanisms of enteral nutrient-enhanced intestinal adaptation. Gastroenterology 2006;130(2 Suppl): S93.
EAN: 2147483647
Pages: 49