3. Group I introns form a characteristic secondary structure
23.2 Group I introns undertake self-splicing by transesterification |
Group I introns are found in diverse locations. They occur in the genes coding for rRNA in the nuclei of the lower eukaryotes Tetrahymena thermophila (a ciliate) and Physarum polycephalum a (slime mold). They are common in the genes of fungal mitochondria. They are present in three genes of phage T4 and also are found in bacteria. Group I introns have an intrinsic ability to splice themselves. This is called self-splicing or autosplicing. (This property is found also in the group II introns discussed in 22 Nuclear splicing and RNA processing. (Belfort et al., 1985))
Self-splicing was discovered as a property of the transcripts of the rRNA genes in T. thermophila. The genes for the two major rRNAs follow the usual organization, in which both are expressed as part of a common transcription unit. The product is a 35S precursor RNA with the sequence of the small rRNA in the 5′ part, and the sequence of the larger (26S) rRNA toward the 3′ end.
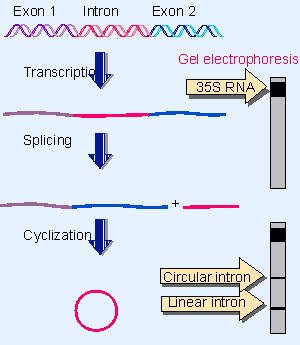 |
Figure 23.1 Splicing of the Tetrahymena 35S rRNA precursor can be followed by gel electrophoresis. The removal of the intron is revealed by the appearance of a rapidly moving small band. When the intron becomes circular, it electrophoreses more slowly, as seen by a higher band. |
In some strains of T. thermophila, the sequence coding for 26S rRNA is interrupted by a single, short intron. When the 35S precursor RNA is incubated in vitro, splicing occurs as an autonomous reaction. The intron is excised from the precursor and accumulates as a linear fragment of 400 bases, which is subsequently converted to a circular RNA. These events are summarized in Figure 23.1.
The reaction requires only a monovalent cation, a divalent cation, and a guanine nucleotide cofactor. No other base can be substituted for G; but a triphosphate is not needed; GTP, GDP, GMP, and guanosine itself all can be used, so there is no net energy requirement. The guanine nucleotide must have a 3′ VOH group (Cech et al., 1981).
The fate of the guanine nucleotide can be followed by using a radioactive label. The radioactivity initially enters the excised linear intron fragment. The G residue becomes linked to the 5′ end of the intron by a normal phosphodiester bond.
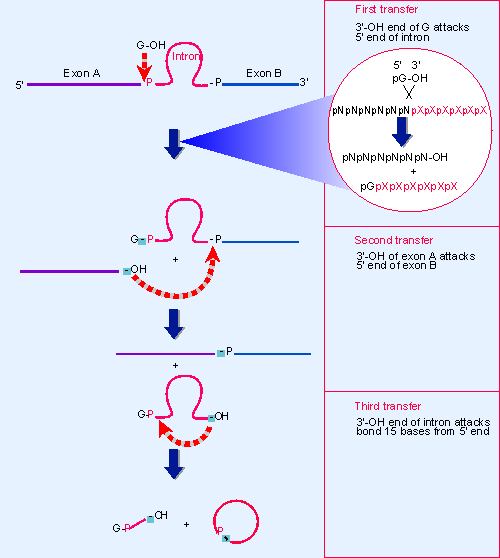 |
Figure 23.2 Self-splicing occurs by transesterification reactions in which bonds are exchanged directly. The bonds that have been generated at each stage are indicated by the shaded boxes. |
Figure 23.2 shows that three transfer reactions occur. In the first transfer, the guanine nucleotide behaves as a cofactor that provides a free 3′ VOH group that attacks the 5′ end of the intron. This reaction creates the G-intron link and generates a 3′ VOH group at the end of the exon. The second transfer involves a similar chemical reaction, in which this 3′ VOH then attacks the second exon. The two transfers are connected; no free exons have been observed, so their ligation may occur as part of the same reaction that releases the intron. The intron is released as a linear molecule, but the third transfer reaction converts it to a circle.
Each stage of the self-splicing reaction occurs by a transesterification, in which one phosphate ester is converted directly into another, without any intermediary hydrolysis. Bonds are exchanged directly, and energy is conserved, so the reaction does not require input of energy from hydrolysis of ATP or GTP. (There is a parallel for the transfer of bonds without net input of energy in the DNA nicking-closing enzymes discussed in 14 Recombination and repair.)
If each of the consecutive transesterification reactions involves no net change of energy, why does the splicing reaction proceed to completion instead of coming to equilibrium between spliced product and nonspliced precursor? The concentration of GTP is high relative to that of RNA, and therefore drives the reaction forward; and a change in secondary structure in the RNA prevents the reverse reaction (see later).
The in vitro system includes no protein so the ability to splice is intrinsic to the RNA. The RNA forms a specific secondary/tertiary structure in which the relevant groups are brought into juxtaposition so that a guanine nucleotide can be bound to a specific site and then the bond breakage and reunion reactions shown in Figure 23.2 can occur. Although a property of the RNA itself, the reaction is assisted in vivo by proteins, which stabilize the RNA structure (Kruger et al., 1982).
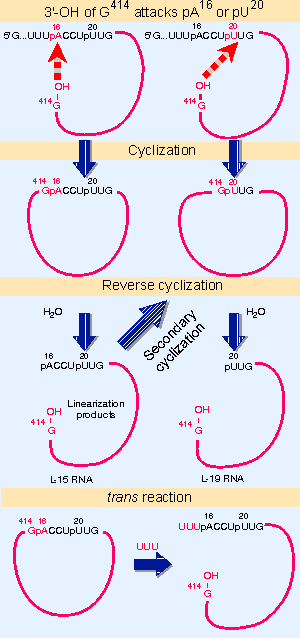 |
Figure 23.3 The excised intron can form circles by using either of two internal sites for reaction with the 5 F end, and can reopen the circles by reaction with water or oligonucleotides. |
The ability to engage in these transfer reactions resides with the sequence of the intron, which continues to be reactive after its excision as a linear molecule. Figure 23.3 summarizes its activities:
The intron can circularize when the 3′ terminal G attacks either of two positions near the 5′ end. The internal bond is broken and the new 5′ end is transferred to the 3′ VOH end of the intron. The primary cyclization usually involves reaction between the terminal G414 and the A16. This is the most common reaction (shown as the third transfer in Figure 23.2). Less frequently, the G414 reacts with U20. Each reaction generates a circular intron and a linear fragment that represents the original 5′ region (15 bases long for attack on A16, 19 bases long for attack on U20). The released 5′ fragment contains the original added guanine nucleotide.
Either type of circle can regenerate a linear molecule in vitro by specifically hydrolyzing the bond (G414 VA16 or G414 VU20) that had closed the circle. This is called a reverse cyclization. The linear molecule generated by reversing the primary cyclization at A16 remains reactive, and can perform a secondary cyclization by attacking U20.
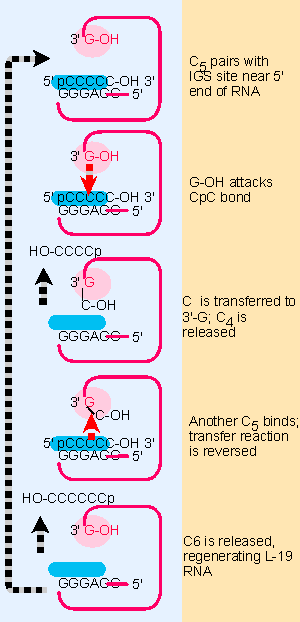 |
Figure 23.8 The L-19 linear RNA can bind C in the substrate-binding site; the reactive G-OH 3 F end is located in the G-binding site, and catalyzes transfer reactions that convert 2 C5 oligonucleotides into a C4 and a C6 oligonucleotide. |
The final product of the spontaneous reactions following release of the intron is the L-19 RNA, a linear molecule generated by reversing the shorter circular form. This molecule has an enzymatic activity that allows it to catalyze the extension of short oligonucleotides (not shown in the figure, but see Figure 23.8).
The reactivity of the released intron extends beyond merely reversing the cyclization reaction. Addition of the oligonucleotide UUU reopens the primary circle by reacting with the G414 VA16 bond. The UUU (which resembles the 3′ end of the 15-mer released by the primary cyclization) becomes the 5′ end of the linear molecule that is formed. This is an intermolecular reaction, and thus demonstrates the ability to connect together two different RNA molecules.
This series of reactions demonstrates vividly that the autocatalytic activity reflects a generalized ability of the RNA molecule to form an active center that can bind guanine cofactors, recognize oligonucleotides, and bring together the reacting groups in a conformation that allows bonds to be broken and rejoined. Other group I introns have not been investigated in as much detail as the Tetrahymena intron, but their properties are generally similar (Been and Cech, 1986; for review see Cech, 1985; Cech, 1987).
The autosplicing reaction is an intrinsic property of RNA in vitro, but to what degree are proteins involved in vivo? Some indications for the involvement of proteins are provided by mitochondrial systems, where splicing of group I introns requires the trans-acting products of other genes. One striking case is presented by the cyt18 mutant of N. crassa, which is defective in splicing several mitochondrial group I introns. The product of this gene turns out to be the mitochondrial tyrosyl-tRNA synthetase! One possible implication is that the intron might take up a tRNA-like tertiary structure that is recognized by the synthetase. Binding of the enzyme might be directly involved in splicing, but more probably has an indirect effect such as stabilizing the RNA conformation.
This relationship between the synthetase and splicing is consistent with the idea that splicing originated as an RNA-mediated reaction, subsequently assisted by RNA-binding proteins that originally had other functions. The in vitro self-splicing ability may represent the basic biochemical interaction. The RNA structure creates the active site, but is able to function efficiently in vivo only when assisted by a protein complex.
| Reviews | |
| Cech, T. R. (1985). Self-splicing RNA: implications for evolution. Int. Rev. Cytol. 93, 3-22. | |
| Cech, T. R. (1987). The chemistry of self-splicing RNA and RNA enzymes. Science 236, 1532-1539. | |
| Research | |
| Been, M. D. and Cech, T. R. (1986). One binding site determines sequence specificity of Tetrahymena pre-rRNA self-splicing, trans-splicing, and RNA enzyme activity. Cell 47, 207-216. | |
| Belfort, M., Pedersen-Lane, J., West, D., Ehrenman, K., Maley, G., Chu, F., and Maley, F. (1985). Processing of the intron-containing thymidylate synthase (td) gene of phage T4 is at the RNA level. Cell 41, 375-382. | |
| Cech, T. R. et al. (1981). In vitro splicing of the rRNA precursor of Tetrahymena: involvement of a guanosine nucleotide in the excision of the intervening sequence. Cell 27, 487-496. | |
| Kruger, K. et al. (1982). Self-splicing RNA: autoexcision and autocyclization of the rRNA intervening sequence of Tetrahymena. Cell 31, 147-157. | |
- Structures, Processes and Relational Mechanisms for IT Governance
- An Emerging Strategy for E-Business IT Governance
- Linking the IT Balanced Scorecard to the Business Objectives at a Major Canadian Financial Group
- A View on Knowledge Management: Utilizing a Balanced Scorecard Methodology for Analyzing Knowledge Metrics
- Technical Issues Related to IT Governance Tactics: Product Metrics, Measurements and Process Control