54 - Anatomy of the Pleura
Editors: Shields, Thomas W.; LoCicero, Joseph; Ponn, Ronald B.; Rusch, Valerie W.
Title: General Thoracic Surgery, 6th Edition
Copyright 2005 Lippincott Williams & Wilkins
> Table of Contents > Volume I - The Lung, Pleura, Diaphragm, and Chest Wall > Section XI - The Pleura > Chapter 66 - Technique of Extrapleural Pneumonectomy for Diffuse Malignant Pleural Mesothelioma
Chapter 66
Technique of Extrapleural Pneumonectomy for Diffuse Malignant Pleural Mesothelioma
Michael Y. Chang
David J. Sugarbaker
The technique of extrapleural pneumonectomy was described in the late 1940s, initially by Sarot (1949), for the treatment of tuberculous empyema, and in the 1980s and 1990s for malignant pleural diseases, such as pleural mesothelioma. Worn (1974) and Butchart (1976), DeLaria (1978), DaValle (1986), Ruffie (1989), Allen (1994) and their colleagues, as well as Rusch and Venkatraman (1996), have described the surgical approach, pathophysiologic consequences, and results of extrapleural pneumonectomy for malignant pleural mesothelioma. Most researchers have reported higher rates of perioperative mortality and morbidity with extrapleural pneumonectomy than with standard (nonextrapleural) pneumonectomy or pleurectomy and decortication (Table 66-1). However, one of us (DJS), and colleagues (1999), has reported a series of 183 patients who underwent extrapleural pneumonectomy followed by adjuvant chemotherapy and radiation therapy for malignant pleural mesothelioma. The operative mortality in this large series is 3.8%, which is a significant improvement over previous reports. This reduction in operative mortality is believed to be the result of improvements in patient selection, preoperative preparation, intraoperative management, surgical technique, and postoperative care of patients with this extremely complex disease.
This chapter describes our approach to patient selection for extrapleural pneumonectomy in patients with malignant pleural mesothelioma. The surgical technique for right extrapleural pneumonectomy is explained in detail, with a supplement on the variations required for left-sided resection. The clinical results from our 12-year experience at the Brigham and Women's Hospital Thoracic Surgery Division and Dana-Farber Thoracic Oncology Program are also described.
TRIMODALITY THERAPY
Until recently, attempts to improve survival using surgical therapy alone have largely been unpromising. Many groups have combined other treatment modalities with surgery. Treatment algorithms include radical pleurectomy with adjuvant chemotherapy or radiation therapy, and extrapleural pneumonectomy with adjuvant chemotherapy or radiation therapy. These treatment algorithms have all failed to demonstrate median survival beyond 18 months, with many studies demonstrating median survival of 12 months or less. A treatment strategy involving a trimodality approach was introduced by one of us (DJS) and co-workers (1991, 1993, 1996, 1997). This approach achieves local control by using extrapleural pneumonectomy followed by radiation therapy and control of systemic disease with chemotherapy, with a median survival of 19 months. One of us (DJS) and colleagues (1999) showed that trimodality therapy is safe and appears to improve survival in malignant pleural mesothelioma, particularly in patients with favorable prognostic indicators.
PATIENT SELECTION
To be considered for extrapleural pneumonectomy, a patient must have a Karnofsky performance status higher than 70, normal liver and renal function tests, a room air arterial Pco2 of less than 45 mm Hg, and a room air arterial Po2 of more than 65 mm Hg. Although there is no specific age limit, we are reluctant to perform extrapleural pneumonectomy in patients over the age of 75 years.
As a result of the obliterative nature of the pleural disease, preoperative pulmonary function tests alone, that is, notably
P.923
forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC), may not fully predict a patient's postoperative pulmonary function. Patients with malignant pleural mesothelioma may perform on these tests as though the diseased lung is functionally absent. For patients in whom lung function is borderline (i.e., FEV1 <2 L), the use of quantitative ventilation-perfusion scanning combined with preoperative FEV1 and FVC results in a reliable prediction of the lung function after surgery. Patients with a predicted postoperative FEV1 of less than 1.0 L are considered for pleurectomy and decortication, rather than extrapleural pneumonectomy.
Table 66-1. Reported Mortality with Extrapleural Pneumonectomy | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Echocardiography is performed to measure left and right ventricular function, assess preexisting alterations in ventricular function, and evaluate for valvular disease. Furthermore, echocardiography is used to rule out pulmonary hypertension, a contraindication to pneumonectomy. Patients with suspected pulmonary hypertension by echocardiography undergo right heart catheterization to obtain direct pulmonary artery pressure measurements.
Preoperative chest magnetic resonance (MR) imaging and computed tomography (CT) are essential in determining the extent of intrathoracic and extrathoracic disease. Determining the presence of transdiaphragmatic extension, contralateral hemithorax involvement, or invasion of vital structures (vena cava, trachea, esophagus, aorta) is required during the preoperative evaluation of the patient. The presence of abdominal disease, contralateral disease, or mediastinal invasion precludes surgical resection. Chest MR imaging has been shown by Patz and associates (1992) to be of value in addition to CT scanning in making these determinations. We routinely perform both chest MR imaging and CT on all patients evaluated for surgery.
Although both chest MR imaging and CT are useful to rule out distant disease, these modalities can be unreliable for chest wall invasion. Because we have had numerous instances of both false-positive and false-negative readings of chest wall invasion, we offer surgery to patients who have a suggestion of chest wall invasion on radiographic imaging, but no other evidence of extrathoracic spread. The same is true of diaphragmatic invasion. It is often impossible to distinguish between a tumor invading through the diaphragm and a tumor that is pushing caudad on the abdominal cavity but without violating the peritoneum. Unless there is obvious abdominal involvement or peritoneal ascites, we recommend abdominal exploration prior to extrapleural pneumonectomy. Our liberal policy of surgical exploration is shared by Rusch and Venkatraman (1999).
Often patients will present with a diagnosis of malignant mesothelioma based entirely on cytologic examination of pleural fluid. Although a cytologic diagnosis of mesothelioma is possible, we require histologic confirmation with pleural biopsy. Differentiating mesothelioma from adenocarcinoma or sarcoma may be particularly difficult and often requires a large pleural sample. Extrapleural pneumonectomy is deferred until pleural biopsy is obtained via a single-port thoracoscopy procedure. If contralateral involvement is suggested on the preoperative imaging studies, confirmatory pleural biopsies are obtained.
TECHNIQUE OF EXTRAPLEURAL PNEUMONECTOMY IN MALIGNANT PLEURAL MESOTHELIOMA
Right Extrapleural Pneumonectomy
Extrapleural pneumonectomy is conducted in five basic steps via a single incision: (a) incision and exposure of the
P.924
parietal pleura, (b) dissection in the extrapleural plane to separate the tumor from the chest wall, mediastinum, and peritoneum, (c) division of the hilar structures, resulting in the en bloc resection of the lung, pleura, pericardium, and diaphragm, (d) radical mediastinal lymph node dissection, and (e) reconstruction of the diaphragm and pericardium.
The patient undergoes standard intraoperative monitoring, including arterial line, continuous oximetry, and central venous access. A thoracic epidural catheter is placed preoperatively and is used for intraoperative management and postoperative pain control. After induction of anesthesia and placement of a left-sided double-lumen endotracheal tube, the patient is positioned in a left lateral decubitus position in preparation for an extended right posterolateral thoracotomy. A nasogastric tube is placed to facilitate palpation of the esophagus during the surgical procedure, and is left in place postoperatively to maintain a decompressed stomach and to minimize the risk for aspiration.
If transdiaphragmatic involvement is suggested by preoperative imaging, we explore the abdomen through a limited subcostal incision. This small incision is made in line with the thoracotomy incision just over the costal margin. Once abdominal involvement is ruled out, the extended posterolateral thoracotomy incision is made. Laparoscopic exploration may be preferred by surgeons with experience in this technique. If peritoneal involvement is discovered, the procedure is terminated and no thoracic resection is performed. The diaphragm may be displaced inferiorly by the tumor mass, but frank invasion into the peritoneal cavity should be documented histologically with biopsy prior to abandoning the procedure.
The thoracotomy incision is made over the sixth rib, as suggested by Garcia and associates (1998) (Fig. 66-1). The incision is taken from 2 cm lateral to the costovertebral junction, following the bed of the sixth rib, to the costochondral junction. After periosteal stripping, the sixth rib is resected to provide adequate exposure. The posterior periosteum, in the bed of the excised sixth rib, is then incised. A widely based extrapleural dissection is advanced superiorly toward the apex of the thorax using mostly blunt digital dissection, but also combined with sharp and cautery dissection as needed. While the dissection is advanced, previously dissected areas are packed with surgical pads to promote hemostasis and minimize blood loss. After adequate initial dissection, two chest retractors are positioned anteriorly and posteriorly in the thoracotomy.
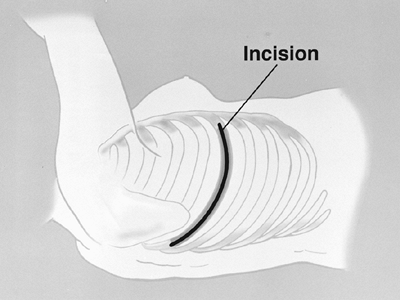 |
Fig. 66-1. The extended right thoracotomy incision. From Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998. With permission. |
The anterior component of the dissection is completed first, both superiorly toward the apex of the lung and inferiorly toward the diaphragm. Anteriorly, the internal mammary artery and vein must be protected from injury, because they are easily avulsed from the superior vena cava and the subclavian artery (Fig. 66-2). The posterior dissection is executed after adequate exposure has been obtained anteriorly. This provides a safe view of the mediastinal structures during the posterior portion of the dissection. The brachial triangle is exposed carefully to avoid avulsion of the subclavian artery and vein. The dissection proceeds over and around the apex of the lung, and the tumor is dissected away from the mediastinum. During this phase of the dissection, care is taken to avoid traction injury to the superior vena cava and azygos vein (Fig. 66-3).
The dissection is continued in an extrapleural plane until the right main-stem and upper lobe bronchi are visualized. Posteriorly, the absence of aortic or esophageal invasion is confirmed by palpation of these structures. The esophagus is separated from the specimen using blunt and sharp scissor dissection. The pericardium is opened anteriorly and the
P.925
pericardial space is palpated to rule out invasion. Without signs of unresectable disease, attention is turned to the diaphragmatic resection.
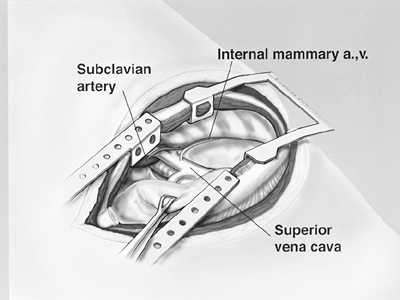 |
Fig. 66-2. Internal mammary artery (a.) and vein (v.) originating from the subclavian artery and superior vena cava, respectively. From Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998. With permission. |
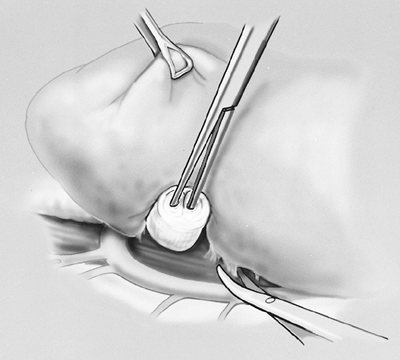 |
Fig. 66-3. Right lung dissected medially away from azygos vein. From Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998. With permission. |
The diaphragm is incised at its anterior margin and extended in a circumferential fashion laterally and posteriorly (Fig. 66-4). The diaphragm muscle fibers may be divided with cautery or simply avulsed from its insertion. The diaphragm muscle fibers are dissected off the peritoneum by placing Babcock clamps on the diaphragm to provide cephalad retraction and blunt caudad dissection on the peritoneum using a sponge stick (Fig. 66-4, inset). Care is taken to avoid violating the peritoneum. If an opening is created, the peritoneum is closed prior to diaphragm reconstruction.
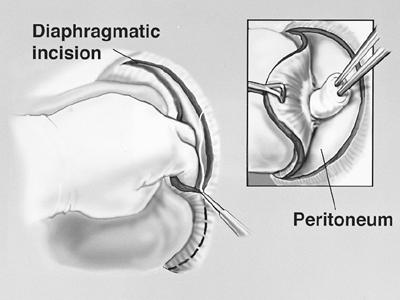 |
Fig. 66-4. Dissection of the pleural envelope off the diaphragm. From Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998. With permission. |
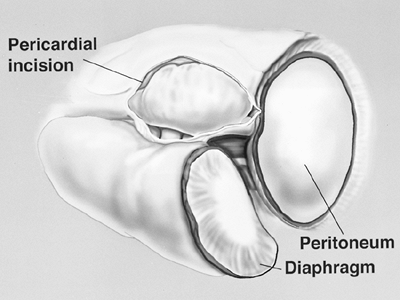 |
Fig. 66-5. Pericardium is opened anteromedially to the phrenic nerve and hilar vessels. From Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998. With permission. |
After the diaphragm dissection has proceeded medially to the pericardium, the incision in the pericardium is extended sufficiently to view and protect the inferior vena cava during division of the medial aspect of the diaphragm. Palpation of the nasogastric tube during the posterior portion of the dissection helps prevent inadvertent injury to the esophagus and facilitates mobilization of the specimen away from the aorta and esophagus. Lateral to the inferior vena cava and the esophagus, the posterior diaphragmatic attachments are divided. A 2-cm rim of the crus is preserved with the esophagus. This tissue can be used as a point of attachment of the diaphragm patch during the reconstruction phase of the operation. The diaphragmatic resection is now complete.
The specimen is retracted posteriorly and the pericardium is opened to the level of the hilar vessels (Fig. 66-5). The main pulmonary artery is then dissected circumferentially, isolating it from the superior vena cava and superior pulmonary vein. In a technique described previously by one of us (DJS) and associates (1997), a soft, flanged catheter (endoleader) is passed around the pulmonary artery to guide subsequent safe passage of an endovascular stapler (United States Surgical, Norwalk, CT, U.S.A.). One jaw of the endovascular stapler is placed in the flange of the endoleader catheter. This catheter delivers the stapler safely around the vessel (Fig. 66-6). The pulmonary artery is divided intrapericardially. Similarly, the superior and inferior pulmonary
P.926
veins are each isolated and stapled with the endovascular stapler using the endoleader technique.
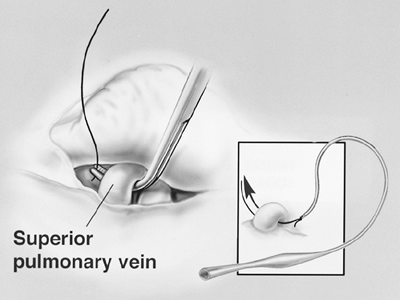 |
Fig. 66-6. The superior pulmonary vein is dissected within the pericardium. Inset. Illustration of a safe technique for dissection of the hilar vessels using a pliable, plastic, self-dilating guidance catheter. One jaw of the endoscopic stapler fits into the end of this catheter, thus allowing safe placement of the stapler. From Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998. With permission. |
The surgical specimen is retracted anteriorly, and the esophagus is retracted medially as the posterior pericardium is divided. Division of the pericardium posterior to the hilum completes the pericardial resection. At this point, we perform a subcarinal node dissection. The right main bronchus is dissected as far as the carina and encircled with a heavy-gauge wire bronchial stapler (TA-30, Ethicon Endo-Surgery, Johnson & Johnson, Cincinnati, OH, U.S.A.) (Fig. 66-7). After closing the stapler, but prior to firing, the contralateral lung is gently hand-bag ventilated to ensure that there is no encroachment on the contralateral bronchus. The stapler is fired and division of the bronchus completes the resection. The surgical specimen is removed. Paratracheal, hilar, paraesophageal, inferior pulmonary ligament, peridiaphragmatic, and subcarinal nodes are resected for complete staging. Resection of aortopulmonary nodes is added for left-sided resections. If a small area of gross disease cannot be resected en bloc with the pneumonectomy specimen, it is delineated using radiopaque clips to facilitate targeting for postoperative radiation therapy.
A pericardial fat pad based on a superior vascular pedicle is raised and rotated to the bronchial stump. It is secured circumferentially around the stump using self-absorbing 3 0 sutures (polygalactin, Vicryl, Ethicon, Somerville, NJ, U.S.A.). The pericardial fat pad provides a vascularized buttress to cover the airway and to separate the bronchial stump from the pulmonary artery stump. Warm saline is placed into the chest, and the airway pressure is increased to 30 mm Hg to detect dehiscence in the bronchial staple line. The entire hemithorax is inspected for hemostasis. Liberal use of the argon beam coagulator (Valleylab, Boulder, CO, U.S.A.) on the chest wall facilitates hemostasis. The hilum and the divided edges of the diaphragm are inspected closely for hemostasis, because bronchial arteries and phrenic vessels are frequent sources of bleeding.
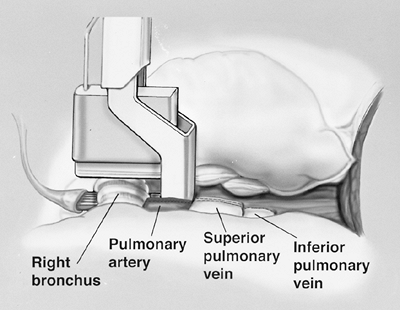 |
Fig. 66-7. The right main-stem bronchus is transected with a heavy-gauge stapler near the carina after the pulmonary artery and veins have been divided. Note the guidance catheter (full view in Fig. 66-6) is also used to place the bronchial stapler. From Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998. With permission. |
The last step is reconstruction of the diaphragm and pericardium. The diaphragm is reconstructed using 2.0-mm thick prosthetic material (Gore-Tex DualMesh, WL Gore and Associates, Inc., Flagstaff, AZ, U.S.A.). An oversized patch is created by sewing together two pieces of 20 30 cm DualMesh. We believe the use of a loose, floppy patch decreases the incidence of patch dehiscence and subsequent herniation of abdominal contents into the chest. Contrary to expectation, we have not observed a loose patch to result in a high hemi-diaphragm, which would interfere with application of adjuvant external-beam radiation to the hemithorax. The diaphragmatic patch is secured anteriorly, laterally, and posteriorly to the chest wall using nine interrupted heavy silk sutures. The silk sutures are first placed through the patch, then both ends of each silk suture are brought through the intercostal space and tied to a 14-mm polypropylene button (Ethicon) as a buttress (Fig. 66-8). Medially, the diaphragm patch is sewn to the diaphragmatic edge of the divided pericardium and to the fibers of the crus using interrupted 2 0 Ethibond sutures (Ethicon) (Fig. 66-9).
The pericardium is always reconstructed with a prosthetic patch to prevent cardiac herniation. The heart is tethered
P.927
by the cavae and projected into the left hemithorax; therefore, the heart can rotate or herniate into an empty right hemithorax. The pericardial patch is a 15 20 cm piece of 0.1-mm thick synthetic membrane (Gore-Tex Pericardial Membrane, WL Gore and Associates). It is secured to the anterior and posterior pericardial margins with interrupted 2 0 Ethibond sutures. Inferiorly, it is sewn to the diaphragm patch or the inferior pericardial margin (Fig. 66-10). The pericardial patch should be fenestrated to prevent cardiac tamponade, a condition caused by fluid accumulating postoperatively in a closed pericardial space (Fig. 66-11).
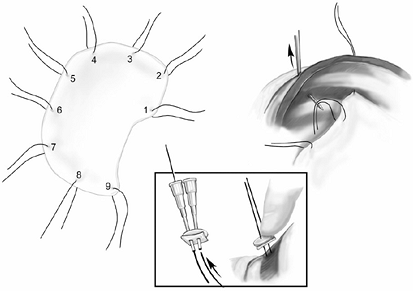 |
Fig. 66-8. The diaphragm patch is contoured to the shape of the hemithorax. Nine silk sutures are placed thorough the patch. The silk sutures are passed through the chest wall using a suture guide. Each end of the suture is passed through a polypropylene button prior to tying. |
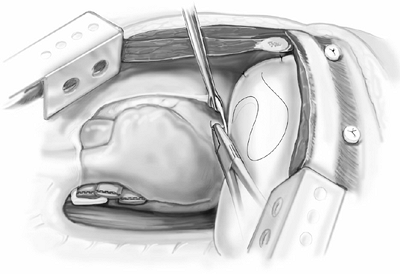 |
Fig. 66-9. The diaphragm patch is secured to the diaphragmatic edge of the pericardium and to the crus of the diaphragm. |
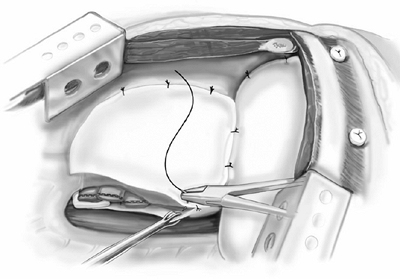 |
Fig. 66-10. The pericardial patch is secured to the anterior and posterior edge of the percardium. Inferiorly the pericardial patch is sutured to the diaphragm patch. |
The thoracotomy is then reapproximated in the usual fashion to ensure an airtight closure. A small (e.g., No. 12F) red rubber catheter is placed in the pneumonectomy space to permit mediastinal positioning. Immediately after closure of the thoracotomy, the pneumonectomy space is reduced in size by removal of 1,000 mL of air in men or 750 mL in women with the use of a 60-mL syringe attached to the red rubber catheter with a three-way stopcock. After flexible bronchoscopy to inspect the bronchial stump and to clear the airway of secretions and blood, the patient is extubated in the operating room. Upon admission to the intensive care unit, a radiograph of the chest is obtained to check for midline position of the heart, nasogastric tube, and mediastinal structures. If these are not midline on the chest radiograph,
P.928
the mediastinum can be balanced by introducing or removing air via the red rubber catheter. If hemothorax is encountered during the postoperative period, a standard chest tube is inserted and placed on water-seal. The chest tube is used to monitor drainage and prevent mediastinal compression by shed blood. Continued high chest tube output mandates operative exploration for bleeding.
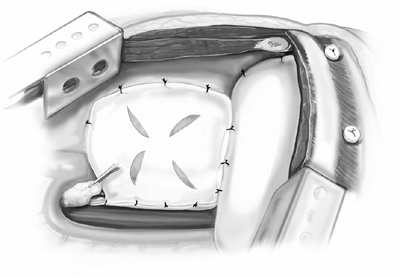 |
Fig. 66-11. Fenestrations in the pericardial patch are made to prevent tamponade. A pericardial fat pad is place over the bronchial stump. |
Left Extrapleural Pneumonectomy
Extrapleural pneumonectomy for a diffuse mesothelioma of the left hemithorax is technically similar to that on the right; however, some important differences exist. For left extrapleural pneumonectomy, lung isolation is achieved by placement of either a left endobronchial blocker or a right double-lumen endotracheal tube. The initial broad-based extrapleural dissection is performed in similar fashion to a right-sided resection. The undersurface of the aortic arch is dissected carefully to avoid injury to the left recurrent laryngeal nerve. During dissection of the posteromedial aspect of the specimen, it is imperative to enter the correct preaortic plane to prevent avulsion of the intercostal vessels. The esophagus in its location anterior to the aorta should be identified and protected, particularly during dissection at the base of the diaphragm.
Garcia and associates (1998) have recommended that the left main pulmonary artery, being much shorter than the right main pulmonary artery, be dissected and divided in an extrapleural, extrapericardial plane to prevent encroachment on the right pulmonary artery (see Fig. 66-10). The pulmonary veins may be divided intrapericardially. As with right-sided resections, a rim of crus is preserved around the esophagus. The left main-stem bronchus must be dissected to the carina, deep to the aortic arch, to ensure a short bronchial stump.
During the early experience with this procedure, we did not routinely reconstruct the pericardium on left-sided resections. However, Byrne and associates (2002) reported seven patients who developed constrictive cardiac physiology after left-sided extrapleural pneumonectomy. All seven patients required cardiac decortication (epicardiectomy) to alleviate symptoms. Histologic examination of the cardiac peel revealed fibrotic tissue with chronic inflammation. Only one patient had malignant cells in the decorticated tissue. We now routinely reconstruct the pericardium after left-sided resections, using a technique similar to right-sided reconstructions.
During dissection and exploration of the hemithorax, a number of situations can be encountered which would preclude surgical resection. The operation is abandoned if extensive chest wall invasion is observed, or if the tumor has infiltrated into the chest wall, thereby obliterating the extrapleural plane. If unexpected invasion of vital structures, such as the aorta, vena cavae, esophagus, epicardium, or trachea, is identified, the patient is not a candidate for extrapleural pneumonectomy. Evidence of transdiaphragmatic spread of tumor also precludes resection.
Postoperative Management
The goals of postoperative management are not dissimilar from those with patients undergoing other standard pneumonectomy procedures. These are to control pain and minimize intravascular volume shifts. Postoperatively, we routinely use thoracic epidural catheters for the first 3 to 5 days. Pain control is vital in minimizing postoperative atelectasis and promoting coughing to clear the airway of secretions. Patients are initially monitored in the thoracic intensive care unit and subsequently transferred to a thoracic intermediate intensive care unit with arterial catheters, central venous catheters, continuous oximetry, and respiratory rate monitors. Deep vein thrombosis prophylaxis is achieved with pneumatic sequential compression boots and subcutaneous heparin given 5,000 U three times daily. Patients are placed on bedrest for 48 hours postoperatively to facilitate mediastinal stability. After 48 hours, patients ambulate at least three times per day with nursing assistance. Chest physiotherapy is performed frequently throughout the day.
Patients are routinely placed on a 1 L per 24-hour fluid restriction for 3 to 5 days postoperatively. Crystalloid infusions are kept to a minimum. Volume replacement is provided by colloid solutions or transfusions. The purpose of the fluid restriction is to minimize capillary leak in the remaining lung, which can lead to desaturation. Desaturation of oxygen levels is usually treated with diuresis and chest physiotherapy. If this is not effective, bronchoscopy is performed to clear the airway of secretions. Daily chest radiographs are closely evaluated for mediastinal shift that might lead to tamponade, venous in-flow obstruction, or compression of the contralateral lung. Contralateral infiltrates are treated aggressively with bronchoscopy, chest physiotherapy, and intravenous antibiotics. Because the nasogastric tube can impede effective cough, the tube is removed on postoperative day 1 if clinical parameters are acceptable. Because aspiration is a highly morbid event in this group of patients, patients are restricted from any oral intake until return of gastric function.
CLINICAL RESULTS AND DISCUSSION
In 1999, one of us (DJS) and associates reported a series of 183 patients who underwent pleuropneumonectomy with a consistent postoperative mortality rate (3.8%) comparable with that of standard pneumonectomy, as reported by Ginsberg and associates (1983). Clearly, patient selection is an important factor in this lowered mortality, as are advances in preoperative preparation, intraoperative management, and postoperative care.
P.929
Cell type, adequate surgical resection margins, and node status in the setting of adjuvant chemotherapy and radiation therapy have been shown to have prognostic importance. As one of us (DJS) and colleagues (1999) have noted, nonepithelial mesothelioma cell type, the inability to obtain negative resection margins, or the presence of nodal involvement at the time of surgical resection are predictors of poor survival. Overall, 2- and 5-year survival rates of 38% and 15%, respectively, were noted in the 176 patients who survived pleuropneumonectomy and were treated with trimodality therapy. However, the 31 patients who had all three predictors of improved survival (epithelial histology, negative resection margins, and negative lymph nodes) had a 2- and 5-year survival of 68% and 46%, respectively.
Innovative therapeutic agents and strategies are being investigated to improve survival with this lethal disease. New chemotherapy agents, including gemcitabine (Eli Lilly and Company, Indianapolis, IN, U.S.A.) and pemtrexed (Eli Lilly), show promise against mesothelioma. Kaiser (1997) has reported using novel therapies, such as cytokines and suicide gene therapy, to target mesothelioma. Pass (1997), Moskal (1998), Schouwink (2001), and Friedberg (2003) and their colleagues have applied photodynamic therapy to the hemithorax after surgical resection. Because recurrence of mesothelioma after surgical resections tends be locoregional, rather than distant, as reported by Baldini and co-workers (1997), strategies to improve local control may be beneficial. A number of groups, including our Brigham and Women's Hospital Thoracic Surgery Division and Dana-Farber Thoracic Oncology Program, are investigating intraoperative intracavitary lavage of hyperthermic chemotherapy following extrapleural pneumonectomy.
Although mesothelioma remains a difficult disease to treat, the techniques of surgical resection for mesothelioma have improved dramatically. Extrapleural pneumonectomy can now be performed with acceptable morbidity and mortality at experienced centers.
REFERENCES
Allen KB, Faber LP, Warren WH: Malignant pleural mesothelioma. Extrapleural pneumonectomy and pleurectomy. Chest Surg Clin North Am 4:113, 1994.
Baldini EH, et al: Patterns of failure after trimodality therapy for malignant pleural mesothelioma. Ann Thorac Surg 63:334, 1997.
Butchart EG, et al: Pleuropneumonectomy in the management of diffuse malignant mesothelioma of the pleura. Experience with 29 patients. Thorax 31:15, 1976.
Byrne JG, et al: Cardiac decortication (epicardiectomy) for occult constrictive cardiac physiology after left extrapleural pneumonectomy. Chest 122:2256, 2002.
DaValle MJ, et al: Extrapleural pneumonectomy for diffuse, malignant mesothelioma. Ann Thorac Surg 42:612, 1986.
DeLaria GA, et al: Surgical management of malignant mesothelioma. Ann Thorac Surg 26:375, 1978.
Friedberg JS, et al: A phase I study of Foscan-mediated photodynamic therapy and surgery in patients with mesothelioma. Ann Thorac Surg 75:952, 2003.
Garcia JP, Richards WG, Sugarbaker DJ: Surgical treatment of malignant mesothelioma. In Kaiser LR, Kron IL, Spray TL (eds): Mastery of Cardiothoracic Surgery. Philadelphia: Lippincott-Raven, 1998.
Ginsberg RJ, et al: Modern thirty-day operative mortality for surgical resections in lung cancer. J Thorac Cardiovasc Surg 86:654, 1983.
Kaiser LR: New therapies in the treatment of malignant pleural mesothelioma. Semin Thorac Cardiovasc Surg 9:383, 1997.
Moskal TL, et al: Operation and photodynamic therapy for pleural mesothelioma: 6-year follow-up. Ann Thorac Surg 66:1128, 1998.
Pass HI, et al: Phase III randomized trial of surgery with or without intraoperative photodynamic therapy and postoperative immunochemotherapy for malignant pleural mesothelioma. Ann Surg Oncol 4:628, 1997.
Patz EF Jr, et al: Malignant pleural mesothelioma: value of CT and MR imaging in predicting resectability. AJR 159:961, 1992.
Ruffie P, et al: Diffuse malignant mesothelioma of the pleura in Ontario and Quebec. A retrospective study of 332 patients. J Clin Oncol 7:1157, 1989.
Rusch VW, Venkatraman E: The importance of surgical staging in the treatment of malignant pleural mesothelioma. J Thorac Cardiovasc Surg 111:815, 1996.
Rusch VW, Venkatraman ES: Important prognostic factors in patients with malignant pleural mesothelioma, managed surgically. Ann Thorac Surg 68:1799, 1999.
Sarot IA: Extrapleural pneumonectomy and pleurectomy in pulmonary tuberculosis. Thorax 4:173, 1949.
Schouwink H, et al: Intraoperative photodynamic therapy after pleuropneumonectomy in patients with malignant pleural mesothelioma: dose finding and toxicity results. Chest 120:1167, 2001.
Sugarbaker DJ, et al: Extrapleural pneumonectomy, chemotherapy, and radiotherapy in the treatment of diffuse malignant pleural mesothelioma [see comments]. J Thorac Cardiovasc Surg 102:10, 1991.
Sugarbaker DJ, et al: Node status has prognostic significance in the multimodality therapy of diffuse, malignant mesothelioma. J Clin Oncol 11:1172, 1993.
Sugarbaker DJ, et al: Extrapleural pneumonectomy in the multimodality therapy of malignant pleural mesothelioma. Results in 120 consecutive patients. Ann Surg 224:288, 1996.
Sugarbaker DJ, Norberto JJ, Swanson SJ: Extrapleural pneumonectomy in the setting of multimodality therapy for diffuse malignant pleural mesothelioma. Semin Thorac Cardiovasc Surg 9:373, 1997.
Sugarbaker DJ, et al: Resection margins, extrapleural nodal status, and cell type determine postoperative long-term survival in trimodality therapy of malignant pleural mesothelioma: results in 183 patients. J Thorac Cardiovasc Surg 117:54, 1999.
Worn H: Moglichkeiten und Ergebnisse der chirurgischen Behandlung des malignen Pleuramesotheliomas. Thoraxchir Vask Chir 22:391, 1974.
EAN: 2147483647
Pages: 203
- Article 285 Transient Voltage Surge Suppressors (TVSSs)
- Article 314 Outlet, Device, Pull, and Junction Boxes; Conduit Bodies; Fittings; and Handhole Enclosures
- Article 426: Fixed Outdoor Electric De-Icing and Snow-Melting Equipment
- Article 427: Fixed Electric Heating Equipment for Pipelines and Vessels
- Example No. D5(a) Multifamily Dwelling Served at 208Y/120 Volts, Three Phase