Table of Contents
Editors: McPhee, Stephen J.; Papadakis, Maxine A.; Tierney, Lawrence M.
Title: Current Medical Diagnosis & Treatment, 46th Edition
Copyright 2007 McGraw-Hill
> Table of Contents > 3 - Preoperative Evaluation & Perioperative Management
3
Preoperative Evaluation & Perioperative Management
Joshua S. Adler MD
Lee Goldman MD, MPH
Each year, tens of millions of patients in the United States undergo a surgical procedure requiring general or spinal-epidural anesthesia. A disproportionate and increasing number of these patients are over age 65. Recent estimates suggest that 15 23% of Americans over the age of 65 undergo surgery in any given year. Operative mortality has declined over the past 10 20 years, probably because of improvements in surgical, anesthetic, and monitoring techniques. Furthermore, most patients do not suffer significant morbidity as a result of the surgical procedure or the anesthetic. The rate of major complications varies from less than 1% to nearly 20% depending on the patient's age, the presence of underlying disease, and the type of surgery. Postoperative complications have significant impact. In patients over 70 years of age, the occurrence of a postoperative complication is associated with decreased long-term functional status and survival. Cardiac, pulmonary, infectious, and neurologic complications account for most of the perioperative morbidity and mortality.
The role of the medical consultant includes evaluating the severity and stability of the patient's medical conditions and providing a surgical risk assessment. The consultant's most important contribution, however, is recommending perioperative measures to reduce surgical risk.
Centers for Disease Control and Prevention National Center for Health Statistics: http://www.cdc.gov/nchs
Fleischmann KE et al: Association between cardiac and noncardiac complications in patients undergoing noncardiac surgery; outcomes and effects on length of stay. Am J Med 2003;115:515.
Lawrence VA et al: Functional independence after major abdominal surgery in the elderly. J Am Coll Surg 2004;199:762.
Manku K et al: Prognostic significance of postoperative in-hospital complications in elderly patients. I. Long-term survival. Anesth Analg 2003;96:583.
Physiologic effects of Anesthesia & Surgery
Both general and spinal or epidural anesthetic agents usually cause peripheral vasodilation, and most of the commonly used general anesthetic regimens also decrease myocardial contractility. These effects often result in transient mild hypotension or, less frequently, prolonged or more severe hypotension. The decrease in tidal volume caused by general and spinal-epidural anesthesia can close small airways and lead to atelectasis. Epinephrine, norepinephrine, and cortisol levels increase during surgery and remain elevated for 1 3 days. Serum antidiuretic hormone levels may be elevated for up to 1 week postoperatively. There is mounting evidence that anesthesia and surgery may be associated with a relative hypercoagulable and inflammatory state mediated by increases in plasminogen activator-1, factor VIII, and platelet reactivity, and increased levels of tumor necrosis factor, interleukins 1 and 6, and C-reactive protein. These effects may be less evident with spinal or epidural anesthesia compared with general anesthesia. The degree to which these hypercoagulable or inflammatory states contribute to perioperative morbidity is not known.
There is no definitive evidence that spinal or epidural anesthesia is preferable to general anesthesia in terms of overall surgical outcomes in large clinical trials. However, subgroup analyses in patients undergoing thoracic, abdominal, and vascular surgery have consistently demonstrated superior analgesia in the spinal/epidural group and a reduction in specific adverse outcomes (cardiac, pulmonary, or neurologic). Similarly, routine use of invasive hemodynamic monitoring with pulmonary artery catheters does not improve surgical outcomes. There are now two randomized trials that have shown a reduction in postoperative cardiac outcomes through the maintenance of normothermia during the perioperative period. In general, the choice of anesthetic technique or agent, the decision to use invasive hemodynamic monitoring, and the regulation of body temperature should be left to the anesthesiologist.
Harvey S et al: Assessment of the clinical effectiveness of pulmonary artery catheters in management of patients in intensive care (PAC-Man): a randomised controlled trial. Lancet 2005;366:472.
Sandham JD et al: A randomized controlled trial of pulmonary artery catheters in high risk surgical patients. N Engl J Med 2003;348:5.
P.38
Evaluation of the Asymptomatic Patient
Patients without significant medical problems especially those under age 50 are at very low risk for perioperative complications. The preoperative evaluation of these patients should include a complete history and physical examination. Special emphasis is placed on the assessment of functional status, exercise tolerance, and cardiopulmonary symptoms and signs in an effort to reveal previously unrecognized disease (especially cardiopulmonary disease) that may require further evaluation prior to surgery. In addition, a directed bleeding history (Table 3-1) should be taken to uncover disorders of hemostasis that could contribute to excessive surgical blood loss. Routine preoperative testing of asymptomatic healthy patients under age 50 has not been found to predict risk or to aid in reducing adverse perioperative outcomes.
Patients who are older than 50 years and those with risk factors for coronary artery disease should have a 12-lead ECG because evidence of clinically silent coronary artery disease should prompt further cardiac evaluation. Minor ECG abnormalities such as bundle branch block, T wave changes, and premature ventricular contractions do not predict adverse postoperative outcomes.
Garcia-Miguel FJ et al: Preoperative assessment. Lancet 2003; 362:1749.
Liu LL et al: Preoperative electrocardiogram abnormalities do not predict postoperative cardiac complications in geriatric surgical patients. J Am Geriatr Soc 2002;50:1186.
Cardiac Risk Assessment
The cardiac complications of noncardiac surgery are a major cause of perioperative morbidity and mortality. The most important perioperative cardiac complications are myocardial infarction (MI), congestive heart failure (CHF), and cardiac death. Older age, preexisting coronary artery disease, and CHF are the principal risk factors for development of these complications.
Major abdominal, thoracic, and vascular surgical procedures (especially abdominal aortic aneurysm repair) tend to carry a higher risk of postoperative cardiac complications than other procedures. Emergency operations are generally associated with more cardiac complications than elective operations. These high-risk procedures are more often associated with major fluid shifts, hemorrhage, and hypoxemia, which may predispose to cardiac complications.
Table 3-1. Factors suggestive of a bleeding disorder. | |
|---|---|
|
Table 3-2. Characteristics defining patients with known or suspected coronary artery disease. | ||
|---|---|---|
|
Coronary Artery Disease
Prior to the routine use of preoperative prophylactic -blocking agents and the widespread use of less invasive surgical techniques, there were approximately 50,000 perioperative MIs annually. Current rates of perioperative MI are likely to be lower. Patients without coronary artery disease are at extremely low risk (< 0.5%) for perioperative ischemic cardiac complications. Patients with known or suspected coronary artery disease, as defined in Table 3-2, have a threefold to 25-fold increased risk of cardiac complications.
The estimated risk of cardiac complications in patients with coronary artery disease can be refined through an assessment of the severity of anginal symptoms, the use of multifactorial indices, and the judicious use of noninvasive tests for ischemia. The severity of anginal symptoms is most accurately assessed using a standardized scale such as that shown in Table 3-3. Multifactorial indices combine several clinical parameters to estimate an overall risk of cardiac complications. The Revised Cardiac Risk Index (RCRI), presented in Table 3-4, is the most recently developed and has been validated in several subsequent studies.
Preoperative Noninvasive Ischemia Testing
Noninvasive tests for myocardial ischemia such as exercise treadmill testing, dipyridamole-thallium scintigraphy, and dobutamine stress echocardiography have been shown to improve upon the clinical risk assessment and help optimize perioperative management in selected patients. Most patients can be accurately stratified through an assessment of anginal symptoms and use of a multifactorial index. Patients who have mild symptoms, defined as Canadian Cardiovascular Society (CCS) class I or II angina, and a low or intermediate multifactorial index score are at low risk for cardiac complications. Noninvasive testing in these patients is
P.39
generally unnecessary. Patients with severe symptoms, CCS class III or IV angina, or a high multifactorial index score are likely to be at high risk for cardiac complications. Stress echocardiography in this group of patients may be able to identify a low-risk subgroup. In the case of high-clinical-risk vascular surgery patients, the absence of a regional wall motion abnormality on stress echocardiography predicts a low risk of perioperative cardiac death or MI.
Table 3-3. Canadian Cardiovascular Society angina class. | ||
|---|---|---|
|
Noninvasive cardiac testing may also be useful in patients with known or suspected coronary artery disease and an unknown functional status. In patients who are able to walk, jog, or use a supine bicycle, the absence of ischemia at or above 85% of their maximal predicted heart rate on exercise ECG predicts a low risk for perioperative cardiac complications.
In patients who cannot exercise, a normal dipyridamole-thallium scan or stress echocardiogram predicts a low risk of complications (comparable to that of patients with a low-risk clinical assessment), whereas evidence of thallium redistribution or stress-induced echocardiographic wall motion abnormalities predicts a much higher risk (comparable to that of patients with a high-risk clinical assessment).
Table 3-4. Revised cardiac risk index (RCRI). | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
Of note in patients with known coronary artery disease who cannot exercise but have other clinical indicators predicting a high risk of cardiac complications (such as a high multifactorial index score), dobutamine stress echocardiography is the preferred noninvasive test.
Any patient who is considered a candidate for noninvasive ischemia testing independent of the planned noncardiac surgery should generally have such testing prior to surgery if the test result may lead to coronary revascularization. This is particularly true for patients found to be at high risk on clinical assessment.
Left ventricular systolic dysfunction, moderate or severe left ventricular hypertrophy, and a peak aortic gradient greater than 40 mm Hg on resting echocardiography are associated with an increased risk of cardiac complications in selected patients. Resting echocardiography and radionuclide ventriculography, however, are not recommended for routine perioperative risk assessment.
Preoperative Management of Patients with Coronary Artery Disease
Patients with stable coronary disease have a 1 5% risk of MI and about a 1% mortality rate whereas patients with unstable symptoms or signs are at much higher risk. An approach to the assessment and management of patients with known or suspected coronary artery disease is shown in Figure 3-1.
A. Medications
Preoperative antianginal medications, including -blockers, calcium channel blockers, and nitrates, should be continued preoperatively and during the postoperative period. There have been several clinical trials demonstrating a reduction in perioperative cardiac morbidity with prophylactic -blocking agents. Atenolol, metoprolol,
P.40
and bisoprolol have been the most frequently studied agents. Comparative trials are lacking, and these drugs are currently believed to be equally effective. The optimal dosing schedules have not been determined. Suggested regimens based on the available clinical trials are presented in Table 3-5. It is thought that heart rate control is one mechanism by which -blockers reduce cardiac morbidity. The -blocker dosage and route of administration should be adjusted to maintain a heart rate of 50 60 beats per minute. The patients most likely to benefit have two or more RCRI criteria.
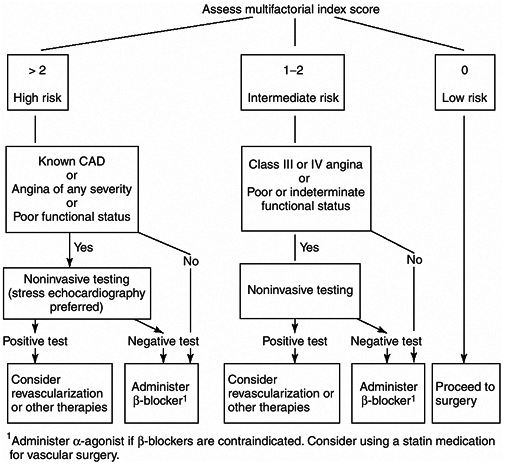 |
Figure 3-1. Assessment and management of patients with known or suspected coronary artery disease (CAD) undergoing major noncardiac surgery. |
A meta-analysis of six randomized trials that studied the use of prophylactic 2-agonists (clonidine or mivazerol) showed that perioperative cardiac mortality was reduced. The effect was statistically significant only in vascular surgery patients. In the most recent trial, patients were randomized to receive clonidine (0.2 mg/d orally or via transdermal patch) or placebo for 4 days beginning the night before a variety of noncardiac procedures. There were no statistically significant differences in the groups during the original hospitalization. However, the 2-year mortality in the clonidine group was 15%, compared with 29% in the placebo group. Based on these data, an 2-agonist should be considered in patients undergoing vascular surgery who are unable to take -blocking agents. Suggested dosing for these agents is shown in Table 3-5.
Data from four retrospective studies showed an association between the use of statin medications during the perioperative period and lower rates of nonfatal MI and cardiac death. In the only published randomized trial, patients received either atorvastatin (20 mg/d) or placebo for 45 days, beginning at least 2 weeks before vascular surgery. At 6 months, the combined endpoint of nonfatal MI or cardiac death occurred in 8% of the atorvastatin group and 26% of the placebo group. Although there are insufficient data to make a general recommendation regarding the use of prophylactic statin medications, it seems reasonable to use them in vascular surgery patients.
Prophylactic intraoperative intravenous nitroglycerin may decrease the frequency of ischemia but has not been shown to reduce the rate of postoperative complications. This medication may be considered for high-risk patients. Too little is known about the effects of the prophylactic use of calcium channel blockers to make any recommendations.
B. Coronary Revascularization
The potential value of preoperative coronary revascularization in high-risk coronary artery disease patients has been a controversial issue. Studies of registries of patients who had previously undergone revascularization
P.41
with coronary artery bypass grafting (CABG) surgery or percutaneous coronary interventions (PCI) have shown that such patients may undergo subsequent noncardiac surgery with a relatively low risk of cardiac morbidity and mortality. However, the use of intracoronary stents in the immediate preoperative period actually may increase the risk of perioperative cardiac complications. When the interval between intracoronary stenting and noncardiac surgery is less than 6 weeks, perioperative mortality is significantly higher than that observed when the interval is more than 6 weeks. The presumed mechanism of this increased mortality is acute stent thrombosis that results from discontinuation of anticoagulant therapy before the standard 4 6 week period. Therefore, it is prudent to delay elective surgery for at least 6 weeks after intracoronary stenting. However, these data do not support a strategy of prophylactic coronary revascularization, especially since the mortality rate for CABG surgery is roughly 1.5% and that for percutaneous transluminal coronary angioplasty (PTCA) ranges from 0.5% to 1.5%. For example, a recent trial randomized over 500 patients with definitive coronary artery disease on angiography to two groups: preoperative prophylactic revascularization with either CABG or PCI and no revascularization before vascular surgery. Postoperative nonfatal MI, 30-day mortality, and mortality at 2.7 years were similar in the two groups, suggesting that truly prophylactic revascularization before noncardiac surgery does not reduce the risk of cardiac complications. However, it seems prudent that patients who are candidates for coronary revascularization independent of the planned surgery undergo the revascularization before the elective noncardiac surgery.
Table 3-5. Preoperative prophylactic cardioprotective medications. | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||
C. Patients with Unstable Coronary Artery Disease
Surgery should be postponed in this group of patients, except in emergency situations, to allow for stabilization of ischemic symptoms. For patients with a recent MI and evidence of ongoing ischemia, delaying surgery allows for appropriate stabilization, and therapy may significantly reduce perioperative mortality and morbidity rates. Patients with unstable angina should be evaluated and treated as indicated by their cardiac status prior to surgery and then reevaluated with respect to severity of symptoms and functional status. Patients with severe stable angina or worsening angina may be managed in a variety of ways. Like patients with less severe angina, those who are potential candidates for coronary revascularization independent of the planned noncardiac surgery should certainly undergo this procedure before the noncardiac surgery. For patients who are not obvious candidates for revascularization, one approach is to optimize their antianginal medications and reevaluate their symptoms. This approach assumes that an improvement in symptoms correlates with a reduction in perioperative cardiac complication rates an assumption that is without clear validation at present. All high-risk patients should be treated with a prophylactic -blocking medication, and perhaps a statin medication if they are not already taking one and if it is not otherwise contraindicated. If a -blocker is contraindicated, an 2-agonist, should be considered.
Chf & Left Ventricular Dysfunction
Decompensated CHF, manifested by an elevated jugular venous pressure, an audible third heart sound, or
P.42
evidence of pulmonary edema on physical examination or chest radiography, significantly increases the risk of perioperative pulmonary edema (roughly 15%) and cardiac death (2 10%). It has been estimated that roughly one-third of perioperative cardiac deaths are a result of CHF. Similarly, patients who were hospitalized for CHF within 1 year prior to noncardiac surgery had a twofold increase in perioperative mortality compared with patients without a recent hospital stay for CHF. Preoperative control of CHF, including the use of diuretics and afterload reducing agents, is likely to reduce the perioperative risk. Clinicians must be cautious not to give too much diuretic, since the volume-depleted patient will be much more susceptible to intraoperative hypotension. Although spironolactone, -adrenergic blocking agents, and angiotensin receptor-blocking agents have been shown to reduce long-term mortality in patients with heart failure, starting these medications in the immediate preoperative period has not been studied and is not recommended as routine practice.
Patients with compensated left ventricular dysfunction are at increased risk for perioperative pulmonary edema but are not at excess risk for other cardiac complications. One large study found that patients with a left ventricular ejection fraction of less than 50% had an absolute risk of 12% for postoperative CHF compared with 3% for patients with an ejection fraction greater than 50%. Such patients should continue taking all medications for chronic heart failure up to and including the day of surgery. Patients receiving digoxin and diuretics should routinely have serum electrolyte and digoxin levels measured prior to surgery because abnormalities in these levels may increase the risk of perioperative arrhythmias. Preoperative echocardiography or radionuclide angiography to assess left ventricular function should be considered for patients with evidence of left ventricular dysfunction who have not had an objective assessment of left ventricular function, and in patients for whom the cause of left ventricular dysfunction is in question. The surgeon and anesthesiologist should be made aware of the presence and severity of left ventricular dysfunction so that appropriate decisions can be made regarding perioperative fluid management and intraoperative monitoring.
Valvular Heart Disease
There are few data available regarding the perioperative risks of valvular heart disease independent of associated coronary artery disease or CHF. Patients with severe symptomatic aortic stenosis are clearly at increased risk for cardiac complications. Such patients who are candidates for valve replacement surgery or, if only short-term relief is needed, for balloon valvuloplasty independent of the planned noncardiac surgery should have the corrective procedure performed prior to noncardiac surgery. In the most recent series of patients with aortic stenosis who underwent noncardiac surgery, the combined endpoint of death or nonfatal MI was 31% in patients with severe aortic stenosis (aortic valve area < 0.7 cm2), 11% in those with moderate aortic stenosis (aortic valve area 0.7 1.0 cm2), and 2% in those without aortic stenosis. Other studies have found that patients with asymptomatic aortic stenosis appeared to be at lower risk than patients with symptomatic aortic stenosis. Noncardiac surgery in patients with severe aortic stenosis must be approached with great caution and requires close consultation with the anesthesiologist.
The severity of valvular lesions should be defined prior to surgery to allow for appropriate fluid management and consideration of invasive intraoperative monitoring. Echocardiography should also be considered in patients with a previously unexplained heart murmur for those procedures in which a valvular abnormality would require antibiotic prophylaxis. For specific recommendations regarding antibiotic prophylaxis, see Chapter 33.
Arrhythmias
Several early studies on cardiac risk factors reported that both atrial and ventricular arrhythmias were independent predictors of an increased risk of perioperative complications. Subsequent data have shown these rhythm disturbances to be frequently associated with underlying structural heart disease, especially coronary artery disease and left ventricular dysfunction. The finding of a rhythm disturbance on preoperative evaluation should prompt consideration of further cardiac evaluation, particularly when the finding of structural heart disease would alter perioperative management. Patients found to have a rhythm disturbance without evidence of underlying heart disease are at very low risk for perioperative cardiac complications.
Management of patients with arrhythmias in the preoperative period should be guided by factors independent of the planned surgery. In patients with atrial fibrillation, adequate rate control should be established. Symptomatic supraventricular and ventricular tachycardia must be controlled prior to surgery. There is no evidence that the use of antiarrhythmic medications to suppress an asymptomatic arrhythmia alters perioperative risk.
It seems prudent for patients who have indications for a permanent pacemaker to have it placed prior to noncardiac surgery. When surgery is urgent, these patients may be managed perioperatively with temporary transvenous pacing. Patients with bundle branch block who do not meet recognized criteria for a permanent pacemaker do not require pacing during surgery.
Hypertension
Severe hypertension, defined as a systolic pressure greater than 180 mm Hg or diastolic pressure greater than 110 mm Hg, appears to be an independent predictor of perioperative cardiac complications, including MI and CHF. Mild to moderate hypertension immediately preoperatively is associated with intraoperative blood pressure lability
P.43
and asymptomatic myocardial ischemia but does not appear to be an independent risk factor for adverse cardiac outcomes. It seems wise to delay surgery in patients with severe hypertension until blood pressure can be controlled, although it is not known whether the risk of cardiac complications is reduced with this approach. It is unlikely that treatment of mild to moderate hypertension in the immediate preoperative period will significantly reduce the risk of cardiac complications. However, medications for chronic hypertension should be continued up to and including the day of surgery.
Auerbach A et al: Assessing and reducing the cardiac risk of noncardiac surgery. Circulation 2006;113:1361.
Boersma E et al: Perioperative cardiovascular mortality in noncardiac surgery: Validation of the Lee cardiac risk index. Am J Med 2005;118:1134.
Devereaux PJ et al: Perioperative cardiac events in patients undergoing noncardiac surgery: a review of the magnitude of the problem, the pathophysiology of the events and the methods to estimate and communicate risk. CMAJ 2005;173:627.
Devereaux PJ et al: Surveillance and prevention of major perioperative ischemic cardiac events in patients undergoing noncardiac surgery: a review. CMAJ 2005;173:779.
Durazzo AE et al: Reduction in cardiovascular events after vascular surgery with atorvastatin: a randomized trial. J Vasc Surg 2004;39:967.
Hernandez AF et al: Outcomes in heart failure patients after major noncardiac surgery. J Am Coll Cardiol 2004;44:1446.
Kertai MD et al: Aortic stenosis: an underestimated risk factor for perioperative complications in patients undergoing noncardiac surgery. Am J Med 2004;116:8.
McFalls EO et al: Coronary-artery revascularization before elective major vascular surgery. N Engl J Med 2004;351:2795.
Sandham JD et al: A randomized controlled trial of the use of pulmonary artery catheters in high risk surgical patients. N Engl J Med 2003;348:5.
Wallace AW et al: Effect of clonidine on cardiovascular morbidity and mortality after noncardiac surgery. Anesthesiol 2004; 101:284.
Wilson SH et al: Clinical outcomes of patients undergoing non-cardiac surgery in the two months following coronary stenting. J Am Coll Cardiol 2003;42:234.
Pulmonary Evaluation in Non-Lung Resection Surgery
Pneumonia and respiratory failure requiring prolonged mechanical ventilation are the most important postoperative pulmonary complications and occur in 2 19% of surgical procedures. The occurrence of a postoperative pulmonary complication has been associated with a significant increase in hospital length of stay.
Risk Factors for the Development of Postoperative Pulmonary Complications
Numerous series have investigated the risk factors for the development of postoperative pulmonary complications. The risk of developing a pulmonary complication is highest in patients undergoing cardiac, thoracic, and upper abdominal surgery, with reported complication rates ranging from 9% to 19%. The risk in patients undergoing lower abdominal or pelvic procedures ranges from 2% to 5%, and for extremity procedures the range is less than 1 3%. The pulmonary complication rate for laparoscopic procedures appears to be much lower than that for open procedures. In one series of over 1500 patients who underwent laparoscopic cholecystectomy, the pulmonary complication rate was less than 1%.
Three patient-specific factors have been repeatedly found to increase the risk of postoperative pulmonary complications: chronic lung disease, morbid obesity, and tobacco use. Patients with chronic obstructive pulmonary disease (COPD) have a twofold to fourfold increased risk compared with patients without COPD. In a single large prospective cohort of US military veterans, additional risk factors for the development of postoperative pneumonia included age over 60 years, dependent functional status, impaired sensorium, and prior stroke. In addition, three studies found that placement of a nasogastric tube postoperatively increases the risk of pneumonia. In two studies, a positive cough test was associated with an increased risk of complications. The cough test is performed by asking the patient to take a deep inspiration and cough once. A positive test is defined as recurrent coughing after the first cough.
Patients with asthma are at increased risk for bronchospasm during tracheal intubation and extubation and during the postoperative period. However, if patients are at their optimal pulmonary function (as determined by symptoms, physical examination, or spirometry) at the time of surgery, they do not appear to be at increased risk for other pulmonary complications.
Postoperative pneumonia is approximately twice as likely to develop in morbidly obese patients those weighing over 113 kg (250 lb) than in patients weighing less. Mild obesity does not appear to increase the risk of clinically important pulmonary complications.
Several studies have shown that current cigarette smoking is associated with an increased risk of postoperative atelectasis. In a single study, cigarette smoking was also found to double the risk of postoperative pneumonia, even when controlling for underlying lung disease. A summary of the known risk factors for pulmonary complications is presented in Table 3-6.
Pulmonary Function Testing & Arterial Blood Gas Analysis
The majority of studies have shown that preoperative pulmonary function testing in unselected patients is not helpful in predicting postoperative pulmonary complications. The data are conflicting regarding the usefulness of preoperative pulmonary function testing in certain selected groups of patients: the morbidly
P.44
obese, those with COPD, and those undergoing upper abdominal or cardiothoracic surgery. Assessment of the severity of COPD using pulmonary function tests has not been shown to improve upon the clinical risk assessment with the exception that patients with a forced expiratory volume in 1 second (FEV1) under 500 mL or an FEV1 below 50% of the predicted value appear to be at particularly high risk. At present, definitive recommendations regarding the indications for preoperative pulmonary function testing cannot be made. In general terms, such testing may be helpful to confirm the diagnosis of COPD or asthma, to assess the severity of known pulmonary disease, and perhaps as part of the risk assessment for patients undergoing upper abdominal surgery, cardiac surgery, or thoracic surgery.
Table 3-6. Risk factors for postoperative pulmonary complications. | |
|---|---|
|
Arterial blood gas measurement is not routinely recommended except in patients with known lung disease and suspected hypoxemia or hypercapnia.
Perioperative Management
The goal of perioperative management is to reduce the likelihood of postoperative pulmonary complications. Smoking cessation for at least 4 weeks prior to thoracic surgery reduced the incidence of pulmonary complications by 25%. Incentive spirometry (IS), continuous positive airway pressure (CPAP), intermittent positive-pressure breathing (IPPB), and deep breathing exercises (DBE) have all been shown to reduce the incidence of postoperative atelectasis and, in a small number of studies, to reduce the incidence of postoperative pulmonary complications. In most comparative trials, these methods were equally effective. However, in a randomized trial of patients undergoing resection of the esophagus or stomach, those who received postoperative CPAP had a lower risk of prolonged mechanical ventilation or reintubation compared with those who did IS or DBE. Given the higher cost of CPAP and IPPB, IS and DBE are the preferred methods for most patients. CPAP may be preferable for patients undergoing esophageal or gastric resection. IS must be performed for 15 minutes every 2 hours. DBE must be performed hourly and consist of 3-second breath-holding, pursed lip breathing, and coughing. These measures should be started preoperatively and be continued for 1 2 days postoperatively. Most studies suggest that postoperative epidural opioid and local anesthetic agents provide excellent pain control but do not appreciably reduce pulmonary complication rates.
There is some evidence that the incidence of postoperative pulmonary complications in patients with COPD or asthma may be reduced by preoperative optimization of pulmonary function. Patients who are wheezing will probably benefit from preoperative therapy with bronchodilators and, in certain cases, corticosteroids. Antibiotics may be of benefit for patients who cough with purulent sputum if the sputum can be cleared prior to surgery. On the other hand, the use of antibiotics in unselected patients undergoing head and neck cancer surgery did not reduce the occurrence of pulmonary complications. Patients receiving oral theophylline should continue taking the drug during the intraoperative and postoperative periods, using intravenous theophylline when necessary.
Block BM et al: Efficacy of postoperative epidural anesthesia: a meta-analysis. JAMA 2003;290:2455.
Fagevik Olsen M et al: Randomized clinical study of the prevention of pulmonary complications after thoracoabdominal resection by two different breathing techniques. Br J Surg 2002;89:1228.
Fisher BW et al: Predicting pulmonary complications after nonthoracic surgery: a systematic review of blinded studies. Am J Med 2002;112:219.
McAlister FA et al: Incidence of and risk factors for pulmonary complications after nonthoracic surgery. Am J Respir Crit Care Med 2005;171:514.
Ong SK et al: Pulmonary complications following major head and neck surgery with tracheostomy: a prospective, randomized, controlled trial of prophylactic antibiotics. Arch Otolaryngol Head Neck Surg 2004;130:1084.
Evaluation of the Patient with Liver Disease
Patients with serious liver disease are generally thought to be at increased risk for perioperative morbidity and demise. Appropriate preoperative evaluation requires consideration of the effects of anesthesia and surgery on postoperative liver function and of the complications associated with anesthesia and surgery in patients with preexisting liver disease.
The Effects of Anesthesia & Surgery on Liver Function
Postoperative elevation of serum aminotransferase levels is a relatively common finding after major surgery. Most of these elevations are transient and not associated with hepatic dysfunction. Studies in the 1960s and early 1970s showed that patients with liver disease are at increased relative risk for postoperative deterioration in hepatic function, although the absolute risk is not known. General anesthetic agents may cause deterioration
P.45
of hepatic function via intraoperative reduction in hepatic blood flow leading to ischemic injury. It is important to remember that medications used for spinal and epidural anesthesia produce similar reductions in hepatic blood flow and thus may be equally likely to lead to ischemic liver injury. Intraoperative hypotension, hemorrhage, and hypoxemia may also contribute to liver injury.
Risk Factors for Surgical Complications
Surgery in the patient with serious liver disease has been associated in several series with a variety of significant complications, including hemorrhage, infection, renal failure, and encephalopathy, and with a substantial mortality rate. A key limitation in interpreting these data is our inability to determine the contribution of the liver disease to the observed complications independent of the surgical procedure.
In three small series of patients with acute viral hepatitis who underwent abdominal surgery, the mortality rate was roughly 10%. Cirrhotic patients undergoing portosystemic shunt surgery who have evidence of alcoholic hepatitis on the preoperative liver biopsy have a significantly increased surgical mortality rate compared with patients without alcoholic hepatitis. Although data are quite limited, it seems reasonable to delay elective surgery in patients with acute viral or alcoholic hepatitis, at least until the acute episode has resolved. These data are not sufficient to warrant substantial delays in urgent or emergent surgery.
There are few data regarding the risks of surgery in patients with chronic hepatitis. In a series of 272 patients with chronic hepatitis undergoing a variety of surgical procedures for variceal hemorrhage, the in-hospital mortality rate was less than 2%. It is of note that patients with Child-Turcotte-Pugh class C cirrhosis (see Chapter 15) or with serum aminotransferase levels over 150 units/L were excluded. In a study of patients undergoing hepatectomy for hepatocellular carcinoma, patients with both cirrhosis and active hepatitis on the preoperative liver biopsy had a fourfold increase in mortality (8.7%) compared with patients with cirrhosis alone or active hepatitis alone. In a study of patients with chronic viral hepatitis who underwent liver resection for cancer, a preoperative hyaluronic acid level greater than 200 ng/mL was associated with a substantially increased risk of postoperative hepatic failure.
Substantial data exist regarding surgery in patients with cirrhosis. In several series from the 1960s and 1970s, patients with cirrhosis undergoing abdominal surgery had substantial mortality rates. Biliary surgery was especially risky. Patients with Child-Turcotte-Pugh class C cirrhosis who underwent portosystemic shunt surgery, biliary surgery, or trauma surgery during the 1970s and 1980s had a 50 85% mortality rate. Patients with Child-Turcotte-Pugh class A or B cirrhosis who underwent abdominal surgery during the 1990s, however, had relatively low mortality rates (hepatectomy 0 8%, open cholecystectomy 0 1%, laparoscopic cholecystectomy 0 1%). In the most recent study of patients with cirrhosis who underwent major abdominal nonhepatic surgery, the Child-Turcotte-Pugh class was the most important predictor of perioperative complications and mortality. Patients with Child-Turcotte-Pugh class A cirrhosis were similar to noncirrhotic patients with respect to postoperative complications, mortality, and length of hospital stay. Patients with Child-Turcotte-Pugh class B and C cirrhosis had substantially higher complication and mortality rates. Cirrhosis-related complications (ascites, encephalopathy, gastrointestinal bleeding, renal and hepatic failure) were far more common in these patients. However, pulmonary, cardiac, and infectious complications were no more frequent than in the noncirrhotic patients. In recent years, the Model for End Stage Liver Disease (MELD) score has been used to predict survival in patients awaiting liver transplantation. The MELD score has now been compared to the Child-Turcotte-Pugh class in several series of cirrhotic patients undergoing abdominal surgery. In general, these scoring systems are comparable in predicting postoperative decompensation of liver function or mortality. A MELD score of > 7 seems to identify a very high risk group. A conservative approach would be to avoid elective surgery in patients with class C cirrhosis, those with class A or B cirrhosis and concomitant active hepatitis, and those with a MELD score > 7. In addition, when surgery is elective, it is prudent to attempt to reduce the severity of ascites, encephalopathy, and coagulopathy preoperatively.
Befeler AS et al: The safety of intra-abdominal surgery in patients with cirrhosis: model for end-stage liver disease score is superior to Child-Turcotte-Pugh classification in predicting outcome. Arch Surg 2005;140:650.
del Olmo JA et al: Risk factors for nonhepatic surgery in patients with cirrhosis. World J Surg 2003;27:647.
Nanashima A et al: Preoperative serum hyaluronic acid level as a good predictor of posthepatectomy complications. Surg Today 2004;34:913.
Perkins L et al: Utility of preoperative scores for predicting morbidity after cholecystectomy in patients with cirrhosis. Clin Gastroenterol Hepatol 2004;12:1123.
Preoperative Hematologic Evaluation
Several hematologic disorders may have an impact on the outcomes of surgery. A detailed discussion of the preoperative management of patients with complicated hematologic disorders is beyond the scope of this section. Two of the more common clinical situations faced by the medical consultant are the patient with preexisting anemia and the assessment of bleeding risk.
The key issues in the anemic patient are to determine the need for preoperative diagnostic evaluation and the need for transfusion. When feasible, the diagnostic evaluation of the patient with previously unrecognized anemia should be done prior to surgery because certain types of anemia (particularly sickle cell
P.46
disease and immune hemolytic anemias) may have implications for perioperative management. Anemia is common before surgery, with a prevalence of 5 75%. Most data suggest that morbidity and mortality increase as the preoperative hemoglobin level decreases, although none of these data were corrected for the presence of preexisting diseases. Hemoglobin levels below 7 or 8 g/dL appear to be associated with significantly more perioperative complications than higher levels. In patients with ischemic heart disease and with Child-Turcotte-Pugh class B or C cirrhosis, a preoperative hemoglobin level below 10 g/dL has been associated with an increased perioperative mortality rate. It is not known, however, whether preoperative transfusion reduces the risk for perioperative complications. Determination of the need for preoperative transfusion in an individual patient must consider factors other than the absolute hemoglobin level, including the presence of cardiopulmonary disease, the type of surgery, and the likelihood of surgical blood loss.
The most important component of the bleeding risk assessment is a directed bleeding history (see Table 3-1). Patients who are reliable historians and who reveal no suggestion of abnormal bleeding on directed bleeding history and physical examination are at very low risk for having an occult bleeding disorder. Laboratory tests of hemostatic parameters in these patients are generally not needed. When the directed bleeding history is unreliable or incomplete or when abnormal bleeding is suggested, a formal evaluation of hemostasis should be done prior to surgery and should include measurement of the prothrombin time, the activated partial thromboplastin time, the platelet count, and the bleeding time.
Armas-Loughran B et al: Evaluation and management of anemia and bleeding disorders in surgical patients. Med Clin North Am 2003;87:229.
Shander A et al: Prevalence and outcomes of anemia in surgery: a systematic review of the literature. Am J Med 2004;116 (Suppl 7A):58S.
Neurologic Evaluation
Delirium occurs after major surgery in approximately 9% of patients over the age of 50 years. Postoperative delirium has been associated with higher rates of major postoperative cardiac and pulmonary complications, poor functional recovery, and increased length of hospital stay. In addition, postoperative delirium may be associated with an increased risk of subsequent dementia. Several prospective series of hip fracture patients have shown that postoperative delirium is associated with an increased likelihood of functional and cognitive decline in the 3 12 months following surgery and an increased mortality at 1 and 5 years. There were similar findings in a series of older patients undergoing abdominal surgery. Several preoperative and postoperative factors, particularly age and preoperative dementia, have been associated with the development of postoperative delirium (Table 3-7). Patients with multiple risk factors are at especially high risk.
Table 3-7. Risk factors for the development of postoperative delirium. | ||
|---|---|---|
|
Delirium is particularly common after hip fracture repair, occurring in 35 65% of patients. In a randomized controlled trial of hip fracture surgery patients, those who received daily visits and targeted recommendations from a geriatrician had a lower risk of postoperative delirium (32%) than the control patients (50%). The most frequent interventions to prevent delirium were maintenance of the hematocrit greater than 30%; minimizing the use of benzodiazepines and anticholinergic and antihistamine medications; maintenance of regular bowel function; and early discontinuation of urinary catheters.
Stroke may occur in 1 6% of patients undergoing cardiac or carotid artery surgery, but it occurs in less than 1% of all other surgical procedures. Most of the available data on postoperative stroke are in cardiac surgery patients. Stroke after cardiac surgery is associated with significantly increased mortality, up to 22% in some studies. The risk factors for stroke after cardiac surgery include age > 60 years, a calcified aorta, prior stroke, carotid stenosis > 50%, peripheral vascular disease, cigarette smoking, diabetes mellitus, and renal failure. Most studies suggest that asymptomatic carotid bruits are associated with little or no increased risk of stroke in noncardiac, noncarotid surgery. The importance of asymptomatic carotid artery stenoses > 50% is not known.
Prophylactic carotid endarterectomy in most patients with asymptomatic carotid artery disease is unlikely
P.47
to be beneficial. On the other hand, patients with carotid disease who are candidates for carotid endarterectomy anyway (see Chapter 12) should probably have the carotid surgery prior to the elective surgery. Some patients require both cardiac and carotid surgery. The ideal timing of these two procedures is not certain and must be decided individually for each patient. In general, the more symptomatic and threatening condition should be addressed first. In a recent observational study of 1566 patients who underwent carotid endarterectomy, the use of a statin medication was associated with a significant reduction in the rate of perioperative stroke and overall mortality. Given the broad indications for statin therapy in patients with vascular disease, it is prudent to begin statin treatment in patients with appropriate indications prior to their undergoing carotid surgery. Nonstroke neurologic complications including coma, seizures, memory loss, and diminished intellectual function occur in up to 7% of patients after cardiac surgery. The risk factors for these complications include a calcified aorta, age > 70 years, pulmonary disease, diabetes, and neurologic disease.
Bitsch MS et al: Pathogenesis of and management strategies for postoperative delirium after hip fracture. Acta Orthop Scand 2004;75:378.
Lundstrom M et al: Dementia after delirium in patients with femoral neck fractures. J Am Geriatr Soc 2003;51:1002.
McGirt MJ et al: 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitors reduce the risk of perioperative stroke and mortality after carotid endarterectomy. J Vasc Surg 2005; 42:829.
McKhann GM et al: Encephalopathy and stroke after coronary artery bypass grafting: incidence, consequences, and prediction. Arch Neurol 2002;59:1422.
Management of Endocrine Diseases
Diabetes Mellitus
Patients with diabetes are at increased risk for postoperative infections, particularly those involving the surgical site. Furthermore, diabetic patients are more likely to have cardiovascular disease and thus are at increased risk for postoperative cardiac complications. The most challenging issue in diabetics, however, is the maintenance of glucose control during the perioperative period.
The increased secretion of cortisol, epinephrine, glucagon, and growth hormone during surgery is associated with insulin resistance and hyperglycemia in diabetic patients. The goal of management is the prevention of severe hyperglycemia or hypoglycemia in the perioperative period.
The ideal blood glucose level during surgery is not known. In vitro studies have shown that cellular immunity may be impaired when the blood glucose level exceeds 250 mg/dL. In several trials of patients undergoing cardiac surgery, patients with mean postoperative glucose levels < 180 mg/dL had fewer serious surgical site infections, a lower risk of renal failure, and a shorter hospital stay than patients with higher mean levels. Based on these data, it is advisable to maintain perioperative glucose levels between 100 mg/dL and 180 mg/dL.
All diabetic patients should have serum electrolyte levels measured and abnormalities in any of these levels corrected prior to surgery. Serum creatinine and urea nitrogen levels should also be measured to assess renal function. The specific pharmacologic management of diabetes during the perioperative period depends on the type of diabetes (insulin-dependent or not), the level of glycemic control, and the type and length of surgery. In general, patients who require insulin to control the diabetes (whether type 1 or type 2) will need intraoperative insulin with any surgical procedure. Patients with type 2 diabetes who take oral agents generally require insulin during major or prolonged surgery.
Perioperative management of all diabetic patients requires frequent blood glucose monitoring to prevent hypoglycemia and to ensure prompt treatment of hyperglycemia (Tables 3-8 and 3-9). Specific recommendations for glycemic control in patients who do not need intraoperative insulin are shown in Table 3-8. For patients who require intraoperative insulin, no single regimen has been found to be superior in comparative trials. Three commonly used insulin administration methods are shown in Table 3-9. The subcutaneous route is used most often because it is easier to access and
P.48
is less expensive. Intravenous insulin, which offers more rapid onset, shorter duration of action, and ease of dose titration, may be preferable in patients with poorly controlled diabetes, patients in the intensive care unit, and in those undergoing cardiac surgery.
Table 3-8. Management of patients who do not need insulin during surgery. | ||||||
|---|---|---|---|---|---|---|
|
Table 3-9. Intraoperative insulin administration methods. | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||
Corticosteroid Replacement
Perioperative complications (predominantly hypotension) resulting from primary or secondary adrenocortical insufficiency are rare. It is not known whether the administration of high-dose corticosteroids during the perioperative period in patients at risk for adrenocortical insufficiency decreases the risk of these complications. In a trial comparing high-dose corticosteroid therapy with simply administering long-term corticosteroid medications in patients with secondary adrenal suppression, there were no differences in perioperative complications. Therefore, definitive recommendations regarding perioperative corticosteroid therapy cannot be made. The most conservative approach would be to consider any patient to be at risk for having adrenocortical insufficiency who has received either the equivalent of 20 mg of prednisone daily for 3 weeks or the equivalent of 7.5 mg of prednisone daily for 1 month within the past year. A commonly used regimen is 50 100 mg of hydrocortisone given intravenously every 8 hours beginning on the morning of surgery and continuing for 48 72 hours. Tapering the dose is not necessary. Patients being maintained on long-term corticosteroids should then resume their usual dose.
Hypothyroidism
Severe symptomatic hypothyroidism has been associated with several perioperative complications, including intraoperative hypotension, CHF, cardiac arrest, and death. Elective surgery should be delayed in patients with severe hypothyroidism until adequate thyroid hormone replacement can be achieved. If emergency surgery is required in such patients, intravenous T3 or T4 and corticosteroids should be administered perioperatively. Conversely, patients with asymptomatic or mild hypothyroidism generally tolerate surgery well, with only a slight increase in the incidence of intraoperative hypotension; surgery need not be delayed for the month or more required to ensure adequate thyroid hormone replacement.
Coursin DB et al: Perioperative diabetic and hyperglycemic management issues. Crit Care Med 2004;32(4 Suppl):S116.
Lazar HL et al: Tight glycemic control in diabetic coronary artery bypass graft patients improves perioperative outcomes and decreases recurrent ischemic events. Circulation 2004;109:1497.
Schiff RL et al: Perioperative evaluation and management of the patient with endocrine dysfunction. Med Clin North Am 2003;87:175.
Renal Disease
The risk for development of a significant reduction in renal function, including dialysis-requiring acute renal failure, after major surgery has been estimated to be between 2% and 20%. The mortality associated with the development of postoperative acute renal failure that requires dialysis after general, vascular, or cardiac surgery exceeds 50%. Risk factors that have been associated with postoperative deterioration in renal function are shown in Table 3-10. Several medications, including renal dose dopamine, mannitol, N-acetylcysteine, and furosemide, have been evaluated in an attempt to preserve renal function during the perioperative period. None of these, however, have proved effective in clinical trials. Maintenance of adequate intravascular volume is likely to be the most effective method to reduce the risk of perioperative deterioration in renal function.
Table 3-10. Risk factors for the development of postoperative acute renal failure. | |
|---|---|
|
P.49
Although the mortality rate for elective major surgery is low (1 4%) in patients with dialysis-dependent chronic renal failure, the risk for perioperative complications, including postoperative hyperkalemia, pneumonia, fluid overload, and bleeding, is substantially increased. Postoperative hyperkalemia requiring emergent hemodialysis has been reported to occur in 20 30% of patients, and postoperative pneumonia may occur in up to 20% of patients. Patients should undergo dialysis preoperatively within 24 hours before surgery, and their serum electrolyte levels should be measured just prior to surgery and monitored closely during the postoperative period.
Bove T et al: The incidence and risk of acute renal failure after cardiac surgery. J Cardiovasc Anesth 2004;18:442.
Burns KE et al: Perioperative N-acetylcysteine to prevent renal dysfunction in high-risk patients undergoing CABG surgery: a randomized controlled trial. JAMA 2005;294:342.
Antibiotic Prophylaxis of Surgical Site Infections
The development of a postoperative surgical site infection is a common and extremely important cause of morbidity and prolonged hospital stays. There are an estimated 0.5 1 million postoperative surgical site infections annually in the United States. For most major procedures, the use of prophylactic antibiotics has been demonstrated to reduce the incidence of postoperative wound infections significantly. For example, antibiotic prophylaxis in colorectal surgery reduces the
P.50
incidence of wound infection from 25 50% to below 9%. In addition, in a case control study of Medicare beneficiaries, the use of preoperative antibiotics within 2 hours of surgery was associated with a twofold reduction in 60-day mortality. Prophylactic antibiotics are considered standard care for all but clean surgical procedures. Clean procedures are those that are elective, nontraumatic, and not associated with acute inflammation and that do not enter the respiratory, gastrointestinal, biliary, or genitourinary tract. The postoperative wound infection rate for clean procedures is thought to be roughly 2%. However, in certain clean procedures, such as those that involve the insertion of a foreign body, antibiotic prophylaxis is still recommended because the consequences of infection are serious.
Table 3-11. Recommended antibiotic prophylaxis for selected surgical procedures. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
Multiple studies have evaluated the effectiveness of different antibiotic regimens for various surgical procedures. In most cases, no single antibiotic regimen has been shown to be superior. Several general conclusions can be drawn from these data. First, there is substantial evidence to suggest that a single dose of an appropriate intravenous antibiotic or combination of antibiotics is as effective as multiple-dose regimens that extend into the postoperative period. For longer procedures, the dose should be repeated every 3 4 hours to ensure maintenance of a therapeutic serum level. One important exception is cardiac surgery, in which at least 24 hours of postoperative therapy is recommended. Second, for most procedures, a first-generation cephalosporin is as effective as later-generation agents. Third, with the exception of colorectal surgery, all prophylactic antibiotics should be given intravenously at induction of anesthesia or roughly 30 60 minutes prior to the skin incision. Although the type of procedure is the main factor determining the risk of developing a postoperative wound infection, certain patient factors have been associated with increased risk, including diabetes, older age, obesity, heavy alcohol consumption, and multiple medical comorbidities. In addition, recent evidence suggests that nasal carriage with Staphylococcus aureus is associated with a twofold to ninefold increased risk of surgical site and catheter-related infections in surgical patients. Treatment of nasal carriers of S aureus with 2% mupirocin ointment (twice daily for 3 days) prior to cardiac surgery decreases the risk of surgical site infections. Current antibiotic prophylaxis recommendations for a variety of procedures are shown in Table 3-11.
Data on the use of supplemental oxygen to prevent surgical site infections are mixed. The most recent study found an increased risk of surgical site infections with the use of 80% oxygen compared with 35% in the immediate postoperative period. At present, however, high-flow supplemental oxygen specifically to prevent these infections is not recommended.
Perl TM: Prevention of Staphylococcus aureus infections among surgical patients: beyond traditional perioperative prophylaxis. Surgery 2003;134(5 Suppl):S10.
Pryor KO: Surgical site infection and the routine use of perioperative hyperoxia in a general surgical population: a randomized controlled trial. JAMA 2004;291:79.
Silber JH et al: Preoperative antibiotics and mortality in the elderly. Ann Surg 2005;242:107.
Smith RL et al: Wound infections after elective colorectal resection. Ann Surg 2004;239:599.
EAN: 2147483647
Pages: 49