18. The Genitourinary System
Authors: Corwin, Elizabeth J.
Title: Handbook of Pathophysiology, 3rd Edition
Copyright 2008 Lippincott Williams & Wilkins
> Table of Contents > Unit V - Nutrition, Elimination, and reproductive function and dysfunction > Chapter 18 - The Genitourinary System
function show_scrollbar() {}
Chapter 18
The Genitourinary System
Although the kidney is sometimes thought of as simply being an organ for waste elimination, it is much more than that. The kidney is essential in maintaining water, salt, and electrolyte balance and is an endocrine gland that secretes at least three hormones. The kidney helps control blood pressure and is especially susceptible to damage if blood pressure is too high or too low.
Physiologic Concepts
Structure
The kidneys lie outside the peritoneal cavity in the upper posterior portion of the abdominal wall, one on each side of the body. Each kidney is made up of approximately one million functional units, each of which is called a nephron. As shown in Figure 18-1, the nephron begins as a capillary tuft, called the glomerulus. Plasma is filtered across the glomerulus by the process of bulk flow and enters the twisting, looping tubule of the nephron. Of the plasma that enters the tubule, only a small fraction is excreted as urine. The remaining plasma, compared with what entered the tubule across the glomerular capillary, has its final composition and volume drastically altered by the processes of renal reabsorption and secretion.
Each kidney is divided anatomically into an outer cortex containing all the glomerular capillaries and some short tubular segments, and an inner medulla where most of the tubular segments are located. The progression
P.605
of tubular segments from the glomerulus to the proximal tubule, to the distal tubule, and finally to the collecting tubule is shown in Figure 18-1. Each nephron's collecting tubule joins other collecting tubules to become several hundred large collecting ducts. The large collecting ducts are located in the renal papillae, which are located in the innermost portion of the kidney, the renal medulla. The large collecting ducts feed into a central draining area, called the renal pelvis, and from there empty into the ureter. The ureter from each kidney is connected to the bladder (Fig. 18-2). The bladder stores urine until it is released outside the body in the process of micturition (urination). Micturition occurs through a single tube called the urethra.
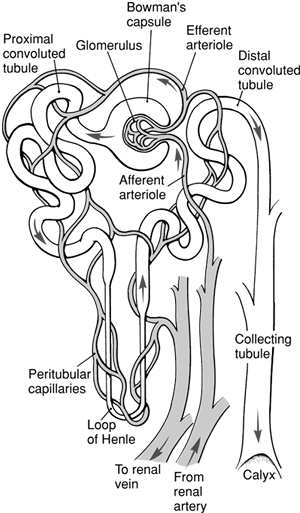 |
Figure 18-1. Structure of the nephron. (From Bullock, B.A., & Henze, R.L. [2000]. Focus on pathophysiology. Philadelphia: Lippincott Williams & Wilkins. ) |
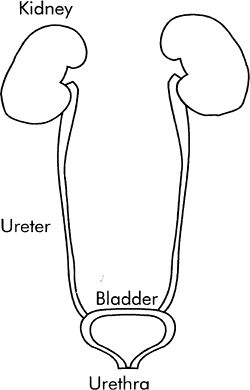 |
Figure 18-2. Urinary tract. |
P.606
Renal Blood Flow
The kidneys receive approximately 1 L of blood per minute one-fifth of the cardiac output. This high rate of blood flow is not required for meeting extraordinary energy demands, but for allowing the kidney to adjust the blood composition continually. By adjusting the blood composition, the kidney is able to maintain blood volume; ensure sodium, chloride, potassium, calcium, phosphate, and pH balance; and eliminate products of metabolism such as urea and creatinine.
Blood flows to the kidneys via the renal arteries, one renal artery to each kidney. In the kidney, the renal artery branches many times, ending as several afferent arterioles. Each afferent arteriole becomes the glomerular capillary that supplies a nephron with blood.
The glomerular capillary reforms not to become a venule as most capillaries do, but to form the efferent arteriole. This is shown in Figure 18-1. The efferent arteriole soon branches into a second capillary network, the peritubular capillaries, which surround and support the nephron tubules themselves. At the end of each nephron, the peritubular capillaries finally reform to venules. The venules join to become veins. Blood leaves the kidney and heads back to the vena cava to be recirculated. The peritubular capillaries surrounding the long loop of the nephron (the loop of Henle) are called the vasa recta.
Filtration, Reabsorption, and Secretion
Filtration refers to the bulk flow of plasma across the glomerular capillary into the interstitial fluid space surrounding the start of the nephron, an area called Bowman's space. At the glomerulus, approximately 20% of the
P.607
plasma is continually filtered into Bowman's space. This filtrate is of the same composition as the plasma, except that protein molecules are not usually filtered. The initial filtrate diffuses across Bowman's space and into the beginning section of the tubule, Bowman's capsule, to begin its journey through the rest of the tubule.
Most of the substances that enter the tubule at Bowman's capsule do not remain in the tubule. Instead, they move (or are moved) back into the blood across the peritubular capillaries by the process of reabsorption. Other substances are added to the urine filtrate, also across the peritubular capillaries, by the process of secretion. It is by reabsorption and secretion that the nephrons manipulate the composition and volume of the initial urine filtrate to produce the final urine.
Glomerular Filtration
Glomerular filtration is the process by which approximately 20% of the plasma entering the glomerular capillary moves across the capillary into the interstitial space and from there into Bowman's capsule. Neither red blood cells nor plasma proteins are more than minimally filtered in healthy kidneys.
The process of filtration across the glomerulus is similar to that which occurs across all capillaries, as described in Chapter 13. What is different in the kidney is that the glomerular capillaries have increased permeability to small solutes and water. Also, unlike other capillaries, the forces favoring filtration of plasma across the glomerular capillary into Bowman's space are greater than the forces favoring reabsorption of fluid back into the capillary. Therefore, net filtration of fluid into Bowman's space occurs. This fluid then diffuses into Bowman's capsule and begins its journey through the rest of the nephron.
In the glomerulus, the primary force favoring filtration is capillary pressure. In most other capillaries, this pressure averages 18 mmHg; in the glomerulus the average pressure is almost 60 mmHg. This higher capillary pressure occurs as a result of decreased resistance to flow offered by the afferent arteriole feeding the glomerulus, compared with arterioles elsewhere. Therefore, the hydrostatic pressure reaching the glomerulus is greater, as shown in Figure 18-3.
Interstitial fluid pressure in Bowman's space is also much greater than in normal interstitial spaces (approximately 15 mmHg versus approximately -3 mmHg). This greater pressure is a result of the high fluid volume entering Bowman's space from the glomerulus, thus opposing further glomerular filtration. Capillary concentration of protein (plasma colloid osmotic pressure) is the same in the glomerulus as in other capillaries. The plasma colloid osmotic pressure increases throughout the length of the glomerulus as protein-free filtrate is pushed into Bowman's space, averaging approximately 28 mmHg overall; this force opposes glomerular filtration. The interstitial fluid colloid osmotic pressure (the pressure exerted by interstitial proteins) is normally approximately 8 mmHg; this pressure favors glomerular filtration.
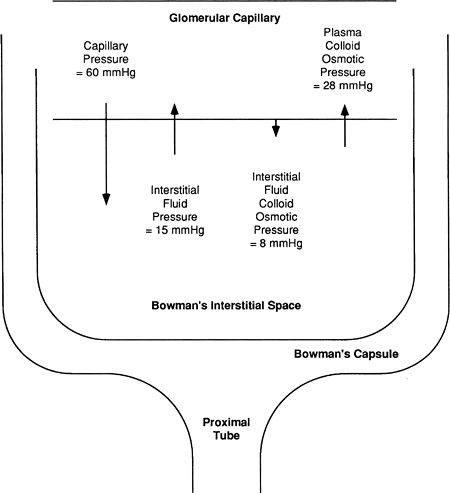 |
Figure 18-3. Forces favoring filtration and reabsorption across the glomerular capillary. |
P.608
Adding up the forces favoring filtration across the glomerulus (60 mmHg + 8 mmHg) and the forces favoring reabsorption (28 mmHg + 15 mmHg), a net force results of approximately 25 mmHg favoring the filtration of plasma into Bowman's space. This filtrate enters Bowman's capsule, moves through the tubule, and a portion of it becomes urine.
Glomerular Filtration Rate
The glomerular filtration rate (GFR) is defined as the volume of filtrate entering Bowman's capsule per unit of time. GFR is nearly constant and gives a good indication of the health of the kidneys. GFR depends on the four forces determining filtration and reabsorption (capillary pressure, interstitial fluid pressure, plasma colloid osmotic pressure, and interstitial fluid colloid osmotic pressure). Therefore, any change in these forces can alter GFR. Likewise, GFR depends on the available surface area of the glomerulus for filtration. Therefore, a loss of glomerular surface area decreases GFR.
P.609
An average value for GFR in an adult is 180 L/day (125 mL/min). A normal plasma volume is approximately 3 L (out of a total blood volume of approximately 5 L). This means that the kidney filters the plasma approximately 60 times each day! Equally remarkable is the fact that of the 180 L/day filtered into Bowman's capsule, only approximately 1.5 L/day are excreted from the body as urine. The rest is reabsorbed back into the blood across the peritubular capillaries.
Measurement of Glomerular Filtration Rate
GFR measurement is possible if one has a substance (call it x) that is freely filterable at the glomerulus and then is not reabsorbed, secreted, or changed in any way before it appears in the urine. To calculate the GFR from this substance, one would measure its concentration in a plasma sample (Px), its concentration in a urine sample (Ux), and the urine volume over a certain period of time (V). Given these values, the equation for GFR, in milliliters per minute, can be solved as shown in Equation 18-1:
![]()
The classic substance that fits the criteria described above for substance x is the polysaccharide inulin. However, inulin is not normally present in the body, which means using inulin to measure GFR involves infusing it into an individual for an extended period. This offers a highly accurate but impractical method for measuring GFR. Instead, what is usually measured in plasma and urine is the concentration of creatinine, which is a naturally produced protein.
Creatinine is produced as a result of normal daily protein metabolism, a process that is assumed to occur at a nearly constant rate. This assumption may not hold true, e.g., it may increase after muscle trauma or intense exercise. To measure GFR using creatinine, a blood sample is drawn along with a timed urine sample, and creatinine concentrations in the blood and urine are measured.
GFR measured from creatinine concentration and urine volume is only an estimate of the true GFR, because a small amount of creatinine actually is secreted into the lumen of the tubule from the peritubular capillaries. Therefore, GFR estimated by creatinine will be slightly high, because more creatinine will be excreted in the urine than was filtered at the glomerulus. Measurement of GFR is important because it offers a clue to nephron function. In conditions of disease leading to renal failure, GFR falls.
![]() eriatric Consideration
eriatric Consideration
GFR declines with age due to a 30 to 50% loss of functional nephrons and reduced renal blood flow. Such a decline means that when drugs normally cleared by the kidneys are given to an elderly individual, their dosage should be adjusted to reflect declining renal function. However, because muscle mass, and therefore serum creatinine, also declines with age, the increase in serum creatinine level that normally indicates a fall in GFR may not be apparent. Since serum creatinine levels are frequently used to determine drug dosing, elderly individuals may receive inappropriately high doses of drugs despite reduced kidney function. This problem can have severe toxic consequences. In order to adjust for age on GFR, the following equation has been developed by Cockcoft and Gault:
![]()
P.610
Renal Clearance
The concentration of a substance totally cleared from the blood into the urine over time is known as the renal clearance. The GFR described above for inulin is really the clearance of inulin, because all filtered inulin is cleared by the kidneys (it is neither reabsorbed nor secreted). For creatinine, clearance is actually slightly greater than the GFR, because some creatinine is secreted into the urine as well as filtered.
Other substances not normally excreted in the urine, such as glucose, have zero clearance. Although glucose is freely filtered across the glomerulus, it is normally totally reabsorbed by the tubules and none appears in the urine (i.e., none is cleared). Substances that are partially reabsorbed back into the plasma, for example, sodium and chloride ion, are cleared at a rate less than the GFR but greater than zero. Substances that are secreted from the blood into the tubule are cleared at a rate greater than the GFR.
Measuring the clearance of any substance is done by the same technique as measuring GFR. The concentration of the substance in the plasma and urine is determined, as is the urine volume over a given period. The equation expressing the clearance of any substance is UV/P, where U is the concentration of the substance in the urine (milligrams [mg] per milliliter [mL]), V is the volume per time of urine (mg per mL), and P is the concentration of the substance in the plasma (mg per mL).
Only for a substance like inulin (freely filtered, neither reabsorbed nor secreted) is the GFR equal to the clearance. For all other substances, clearance is either more or less than the GFR. Measuring the clearance of a plasma substance that is 100% excreted by the kidneys allows one to estimate renal plasma flow, and from there, renal blood flow.
Measurement of Renal Plasma Flow and Renal Blood Flow
Measuring renal plasma flow usually involves measuring the clearance of a substance called para-aminohippurate (PAH). PAH is freely filtered at the glomerulus. It is not reabsorbed, but is actively secreted into the urine filtrate. Therefore, all PAH in the plasma (100%) is cleared by the kidneys. The clearance of PAH gives an estimate of renal plasma flow. Because the
P.611
plasma is approximately 40 to 50% of the total blood volume, this allows one to estimate renal blood flow.
It is only possible to estimate renal blood flow from clearance of PAH because not all plasma entering the kidney goes through a glomerular capillary. Approximately 10 to 15% of renal blood flow feeds non-filtering tissue such as renal fat and connective tissue. Therefore, clearance of PAH is said to give the effective renal plasma flow (ERPF), which is 10 to 15% less than total renal plasma flow, as shown in Equation 18-3:
![]()
From the ERPF, the effective renal blood flow (ERBF) can be found with Equation 18-4:
![]()
where Vc is the measured hematocrit of the blood sample (the amount of blood occupied by red blood cells, not plasma).
Regulation of Renal Blood Flow
Maintenance of adequate renal blood flow is essential for kidney survival and for control of plasma volume and electrolytes. Changes in renal blood flow may increase or decrease the glomerular hydrostatic pressure, affecting GFR. The kidney has several mechanisms for controlling renal blood flow. These mechanisms serve to maintain both kidney function and GFR constant in spite of systemic blood pressure changes.
Renal blood flow is controlled by intrarenal and extrarenal mechanisms. Intrarenal mechanisms include the inherent ability of the afferent and the efferent arterioles to dilate or constrict, thereby controlling blood flow through the kidney. This inherent ability is called autoregulation. Extrarenal mechanisms regulating renal blood flow include the direct effects of increased or decreased mean arterial pressure and the effects of the sympathetic nervous system. A third mechanism regulating renal blood flow that has both intrarenal and extrarenal components involves a hormone produced by the kidney that affects the entire systemic circulation. This hormone, called renin, exerts its effects through the production of a potent vasoconstrictor, angiotensin II.
Autoregulation
Autoregulation is the intrinsic response of vascular smooth muscle to changes in blood pressure. Like many arterioles, smooth muscle cells of the afferent and the efferent arterioles respond to their own stretch with reflex constriction. When systemic blood pressure is increased, stretch on afferent arterioles is increased. Stretching the afferent arterioles causes them to constrict, reducing the blood flow and returning renal blood pressure back toward normal. In contrast, when systemic blood pressure is decreased, stretch on the afferent and the efferent arterioles is reduced,
P.612
and the arterioles respond by relaxing and dilating to increase flow. As a result of autoregulation, renal blood flow remains nearly constant over a range of blood pressures between 80 and 180 mmHg.
Autoregulation is especially effective during blood pressure increases. The bottom limit of autoregulation, 80 mmHg, however, is reached more frequently than the upper limit. Therefore, GFR may decrease with severe hypotension.
Sympathetic Nervous System
Sympathetic nerves innervate both the afferent and the efferent arterioles of the kidney and can override autoregulation when stimulated. As is true in most arterioles, stimulation of the sympathetic nerves causes constriction of the afferent arterioles, leading to increased resistance to flow. As a result, blood flow through the glomerulus decreases, causing a decrease both in capillary hydrostatic pressure and in GFR. Simultaneous sympathetic stimulation of the efferent arterioles, however, and their subsequent constriction, causes blood flow to back up in the glomerulus. This backup can actually increase capillary hydrostatic pressure and glomerular filtration. The net result of sympathetic stimulation to the kidneys is a significant decrease in renal blood flow (because blood going both in and out is reduced) but a lesser decrease in GFR. The sympathetic nervous system is stimulated when there is a decrease in systemic blood pressure.
Decreased renal blood flow in response to decreased systemic blood pressure is adaptive and helps the organism survive a hypotensive crisis. With hypotension, less water and salt are filtered at the glomerulus, causing less to be lost in the urine. This helps to increase blood volume and restore blood pressure.
In conditions of increased blood pressure, sympathetic stimulation to all arterioles is reduced. The afferent and the efferent arterioles dilate, and renal blood flow and GFR both increase. This change results in increased loss of water and salt in the urine, which helps to reduce blood volume and return blood pressure toward normal.
Note that sympathetic input dominates over autoregulatory mechanisms of the kidney. If sympathetic stimulation increases, renal blood flow decreases despite attempts by the kidney to autoregulate its flow.
Renin
Renin is a hormone released from the kidney in response to either a decrease in blood pressure or a decrease in plasma sodium concentration. Cells that synthesize and secrete renin and control its release are a particular group of cells of the nephron called the juxtaglomerular (JG) apparatus. This group of cells includes smooth muscle cells of the afferent arteriole and cells of the macula densa. The smooth muscle cells synthesize renin and act as baroreceptors monitoring blood pressure. Macula densa cells are part of the thick ascending limb of the nephron. These cells sense plasma sodium concentration. The macula densa cells and the afferent
P.613
arteriolar cells are in close approximation to each other where the ascending limb of the distal tubule nearly touches the glomerulus. When the macula densa cells sense a change in plasma sodium, they pass that message on to the renin-secreting cells.
When blood pressure falls, the smooth muscle cells increase renin release. When blood pressure increases, the smooth muscle cells decrease their release of renin. If plasma sodium levels decrease, macula densa cells signal the renin-producing cells to increase their activity. If plasma sodium levels increase, macula densa cells signal the smooth muscle cells to decrease renin release.
Sympathetic nerves also stimulate the JG apparatus to secrete renin. Thus, decreased blood pressure causes increased renin both directly, via the JG baroreceptors, and indirectly, via the sympathetic nerves.
Once released, renin circulates in the blood and acts to catalyze the breakdown of a small protein, angiotensinogen, to a 10 amino-acid protein, angiotensin I (AI). Angiotensinogen is produced by the liver and is highly concentrated in the blood. Renin release is thus the rate-limiting step in the reaction. The conversion of angiotensinogen to AI occurs throughout the plasma, but primarily in the pulmonary capillaries. AI has few effects of its own, but it is quickly acted upon by another enzyme readily available in the bloodstream angiotensin-converting enzyme. Angiotensin-converting enzyme splits AI into an 8 amino-acid peptide, angiotensin II (AII).
Angiotensin II
AII is a potent vasoconstrictor that acts throughout the vascular system to increase smooth muscle contraction, thereby decreasing vessel diameter and increasing total peripheral resistance (TPR). An increase in TPR directly increases systemic blood pressure (see Chapter 13). AII is also a potent hormone that circulates in the blood to the adrenal glands, causing the synthesis of the mineralocorticoid hormone aldosterone.
Aldosterone
Aldosterone circulates in the blood and binds to cells of the cortical collecting duct. The binding of aldosterone increases sodium reabsorption from the urine filtrate, causing sodium to return into the peritubular capillaries. Since water often follows sodium movement, increased sodium reabsorption allows for increased water reabsorption, causing increased plasma volume. An increase in plasma volume increases venous return to the heart, thereby increasing the stroke volume and cardiac output. Increased cardiac output, like increased TPR, directly increases systemic blood pressure.
Other stimuli for aldosterone release, besides angiotensin II, are high plasma potassium level and a hormone from the anterior pituitary, adrenocorticotropic hormone (ACTH). In addition to affecting sodium reabsorption, aldosterone stimulates the secretion (and therefore the excretion) of potassium from the cortical collecting duct into the urine
P.614
filtrate. Aldosterone affects sodium and potassium transport across the gut, in the same manner as it does across the collecting duct.
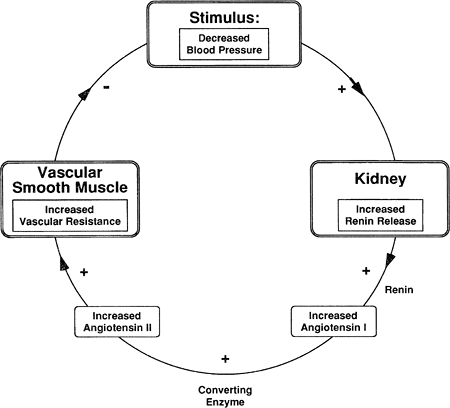 |
Figure 18-4. Response of renin, AI, and AII to a decrease in blood pressure. |
Renin-Angiotensin Reflex Response to Changes in Blood Pressure
With a decrease in blood pressure, the JG cells release renin, which in turn causes an increase in AII. AII constricts arterioles throughout the body, including the afferent and the efferent arterioles. AII-induced constriction increases total peripheral resistance and a return of blood pressure back toward normal (Fig. 18-4). Renal blood flow is reduced, which causes less urine to be produced. Decreased urine output contributes to increased plasma volume and blood pressure.
The opposite occurs with increased blood pressure. With an increase in blood pressure, renin release decreases, as do AII levels. This leads to dilation of systemic arterioles, a reduction of total peripheral resistance, and a return of blood pressure back toward normal. Decreased AII causes afferent and efferent arterioles to relax, leading to an increase in renal blood flow and urine output, which also serves to decrease blood pressure.
Renin-Angiotensin-Aldosterone Response to Decreased Sodium
The second stimulus for renin release is plasma sodium concentration. Decreased sodium in the tubular fluid passing the cells of the macula densa causes increased renin release. As shown in Figure 18-5, increased
P.615
renin leads to increased AII, which stimulates aldosterone synthesis and therefore increases sodium reabsorption. Increased sodium reabsorption reduces the stimulus for further renin release. The opposite is true if there is increased plasma sodium passing the macula densa cells.
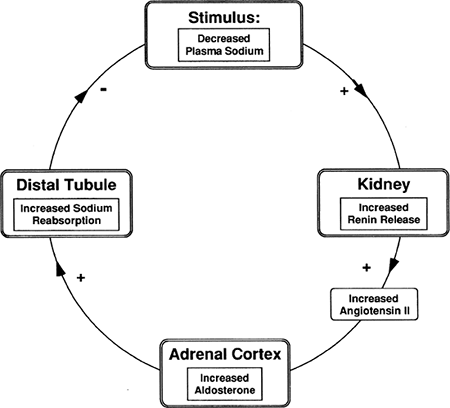 |
Figure 18-5. Response of renin, AI, and AII to a decrease in plasma sodium. |
Renal Reabsorption
Reabsorption is the second process by which the kidney determines the concentration of a substance filtered from the plasma. Reabsorption refers to the active (requiring energy and always being mediated by a carrier) or the passive (no energy required) movement of a substance filtered at the glomerulus back into the peritubular capillaries. Reabsorption may be total (e.g., glucose) or partial (e.g., sodium, urea, chloride, and water).
Reabsorption of Glucose
Glucose is freely filtered at the glomerulus. All of the filtered glucose is normally reabsorbed by active transport, primarily in the proximal tubule.
Because carriers are involved, a transport maximum (Tm) for glucose can be reached. The Tm is the amount of a substance that can be transported per unit of time. For glucose, at a certain filtered load (GFR plasma concentration), all carriers become occupied. Any glucose filtered beyond that load is not reabsorbed, but is instead excreted in the urine. The Tm for glucose is approximately 375 mg/min of filtered glucose. The concentration
P.616
of glucose that results in this filtered load, given a GFR of 125 mL/min, is 3.0 mg/mL of plasma because glucose concentration clinically is frequently expressed as per 100 mL of blood, or, 300 mg/dL. However, glucose begins to appear in the urine even before this plasma level is reached, because each nephron has a slightly different Tm and the carrier transport rate may accelerate at the highest glucose concentrations. Plasma glucose seldom gets high enough that glucose Tm is reached unless an individual has diabetes mellitus (see Chapter 16). Note that the kidney does not control blood glucose levels; it simply filters and reabsorbs all it can. The pancreas, via insulin release, controls blood glucose.
In the kidney, glucose reabsorption is coupled with the reabsorption of sodium ions from the urine filtrate into the tubular cells. At some point this movement is driven by the splitting of adenosine triphosphate (ATP) by the sodium-potassium ATPase. It is a process that requires energy. It is this secondary use of energy that makes glucose transport an active (energy-requiring) process.
Reabsorption of Sodium
Sodium reabsorption occurs throughout the tubule by a combination of simple diffusion and active transport. Approximately 65% of sodium reabsorption occurs across the proximal tubule and 25% across the loop of Henle. Therefore, only approximately 10% of the filtered sodium remains in the tubule by the time the filtrate reaches the distal convoluted tubule. The final concentration of sodium in the urine is usually less than 1% of the total amount filtered at the glomerulus.
Unlike glucose, plasma sodium concentration is regulated by the kidney. Although sodium is freely filtered and 98 to 99% is normally reabsorbed, the final 1 to 2% of its reabsorption can vary. Plasma sodium concentration is 145 mmol/L, and the amount of filtered sodium is approximately 18 mmol/min (supposing a GFR of approximately 180 L/day). This amounts to approximately 1,500 grams of sodium filtered each day. Even if only 2% of this amount 30 g/day is excreted, it is a considerable amount. This final 1 to 2% is controlled by the presence or absence of the hormone aldosterone.
Transport of sodium out of the nephron and back into the capillaries may either be coupled in the same direction to the reabsorption of another substance (co-transport), or it may be coupled in the opposite direction with another substance (counter-transport). Substances co-transported with sodium include glucose, amino acids, and chloride. Hydrogen ion (H+) is counter-transported and thus secreted into the urine when a sodium ion is reabsorbed.
Reabsorption of Chloride
Chloride reabsorption can be active or passive and is nearly always coupled to sodium transport. It is affected by the electrical gradient across the tubule. Like sodium, most chloride reabsorption (65%) occurs across
P.617
the proximal tubule, less across the loop of Henle (25%), and the rest (10%) between the distal convoluted tubule and the collecting-duct system.
Reabsorption of Potassium
Most potassium in the body is present intracellularly. Therefore, although plasma potassium is freely filtered across the glomerulus, its concentration in Bowman's capsule is low. Most potassium that is filtered is reabsorbed: 50% across the proximal tubule, 40% in the thick ascending limb, and the remaining 10% in the final part of the nephron, the medullary collecting duct. Most potassium reabsorption occurs by passive diffusion.
Potassium is also secreted into the tubule by active transport across the cells of the proximal tubule, the descending limb of the loop of Henle, and the collecting ducts. The amount of secreted potassium is variable and depends on the amount of potassium ingested in the diet. An individual on a high-potassium diet filters, reabsorbs, and secretes potassium. An individual on a low-potassium diet only filters and reabsorbs, but does not secrete, potassium. Potassium secretion by the collecting ducts is stimulated by the hormone aldosterone released from the adrenal cortex.
Reabsorption of the Amino Acids
Amino acids filtered at the glomerulus are actively reabsorbed in the proximal tubule. All reabsorption of amino acids is carrier-mediated. The Tm for the carriers is well above the amounts of amino acids normally filtered, so none are normally present in the urine.
Reabsorption of Plasma Proteins
Very few plasma proteins are filtered across the glomerulus. Those that are filtered are actively reabsorbed across the proximal tubule. Because the GFR is so high, the filtration of even a few molecules of plasma protein, such as albumin, would result in a significant daily loss of protein if reabsorption did not occur.
The few proteins filtered at the glomerulus are not reabsorbed. They are degraded by tubular cells and excreted in the urine. Examples of these proteins include the protein hormones, such as growth hormone and luteinizing hormone, both of which are secreted from the anterior pituitary.
Reabsorption of Urea
Urea is produced in the liver as an end-product of protein metabolism. It is freely filtered at the glomerulus. Because urea is highly permeable across most (but not all) of the nephron, it diffuses back into the peritubular capillaries. It follows water as water is reabsorbed from the urine filtrate moving through the nephron. By the end of the proximal tubule, approximately 50% of the filtered urea has been reabsorbed. From the end of the proximal tubule to the medullary collecting ducts, the proximal tubule is impermeable to urea. Along this route, some portions of the tubule begin
P.618
to secrete urea into the filtrate. Thus, at the point the filtrate reaches the medullary collecting ducts, urea concentration has again reached what it was in the original glomerular filtrate. At the medullary collecting ducts, urea once more becomes permeable and again follows water reabsorption out of the tubule. As the filtrate leaves the kidney, approximately 40% of the original filtered urea remains and is excreted.
Note that urea reabsorption depends on water reabsorption. If water reabsorption is low, more urea will be excreted, and vice versa.
Acid-Base Handling
The kidney plays a pivotal role in maintaining acid-base balance. Most metabolic processes in the body produce acid (see Chapter 19). These processes include oxidative phosphorylation, which produces the volatile acid carbon dioxide, and the metabolism of proteins, which produce non-volatile acids such as sulfuric and phosphoric acids. Although the lungs normally excrete all carbon dioxide produced by oxidation, the kidney is the only organ capable of eliminating non-volatile acids. More importantly, the kidneys have the essential job of reabsorbing large quantities of the base bicarbonate, which is freely filtered at the glomerulus. Without this function, fatally low blood pH would occur. The kidneys assist in eliminating acid produced by cell metabolism in individuals who have lung disease by increasing the secretion and excretion of acid and by reabsorbing increased amount of base.
Reabsorption of Bicarbonate
Reabsorption of bicarbonate is an active process that occurs primarily in the proximal tubule (and to a lesser extent in the collecting ducts). As shown in Figure 18-6, reabsorption occurs when a molecule of water breaks down in the proximal tubular cell into an H+ and a hydroxyl molecule (OH-). The H+ is actively secreted into the lumen of the tubule and joins with a bicarbonate molecule that has been filtered at the glomerulus. Hydrogen plus bicarbonate results in carbonic acid (H2CO3), which, in the presence of the enzyme carbonic anhydrase, breaks down to carbon dioxide and water. These diffuse back into the proximal tubular cell to be used again as this cycle repeats.
By this process, the filtered bicarbonate is saved from being excreted in the urine. The reaction of hydrogen with bicarbonate is reversible, as shown in Equation 18-5:
![]()
The OH- produced in the proximal tubule cell joins with an intracellular carbon dioxide molecule. In the presence of the enzyme carbonic anhydrase, it too proceeds to a bicarbonate ion. This bicarbonate also returns into the peritubular capillary as shown in Figure 18-6. The enzyme carbonic anhydrase is readily available.
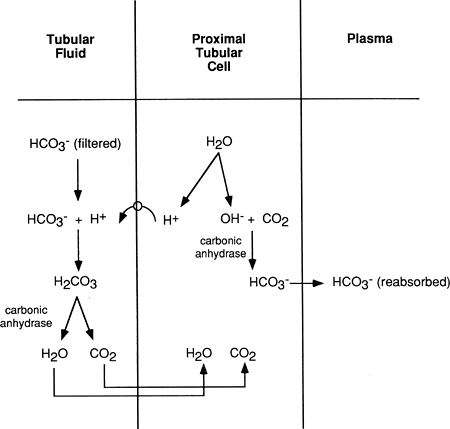 |
Figure 18-6. Reabsorption of filtered bicarbonate by the proximal tubular cells. |
P.619
Secretion and Excretion of Acid
The above reactions only serve to reabsorb filtered bicarbonate. They do not eliminate acid. The kidney does actively secrete and excrete H+ in the urine as well, which allows it to rid the blood of metabolically produced nonvolatile acids. As shown in Figure 18-7, H+ excretion occurs after most of the filtered bicarbonate has been reabsorbed. In this case, the H+ produced in the proximal tubule cell from the breakdown of water moves into the lumen of the tubule and combines with filtered phosphate ions (or to a lesser extent, sulfate ions) and is then excreted in the urine.
The effect of excreting hydrogen bound to phosphate is not only the loss of acid in the urine but a net gain of bicarbonate. This net gain occurs because a bicarbonate ion is still produced in the proximal tubule when carbon dioxide joins with OH-. This bicarbonate is returned to the plasma.
A second mechanism by which the kidney excretes acid is by active secretion of ammonium ion (NH4+) into the tubular fluid (Fig. 18-8). Ammonium ion is produced in the proximal tubular cell as a result of the metabolism of glutamine. Glutamine enters the cell from the peritubular capillary and from the tubular lumen, after being filtered across the glomerulus. Once in the tubule, ammonium ion cannot return into the
P.620
proximal tubular cells; therefore, it is excreted in the urine. The bicarbonate produced from glutamine metabolism diffuses back into the peritubular capillary, thereby returning base to the blood. Finally, a small amount of H+ is excreted free in the urine, causing the urine to normally have an acidic pH.
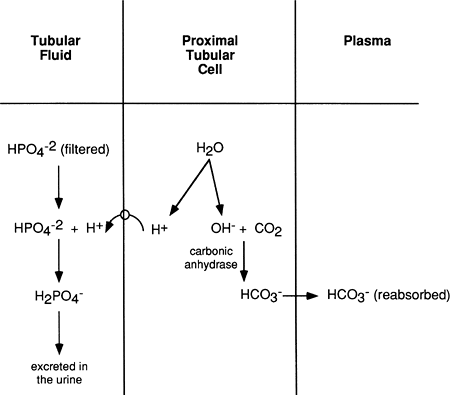 |
Figure 18-7. Excretion of H+ bound to filtered phosphate. |
Secretion of Bicarbonate
Under conditions of alkalosis (excess base), the kidney can secrete bicarbonate, thus ridding the plasma of base and returning the pH toward normal. Secretion of bicarbonate is an active process occurring in the cortical collecting duct. However, even under conditions of alkalosis, bicarbonate reabsorption in the proximal tubule is ongoing and essential. Loss of all filtered bicarbonate would be fatal.
Renal Concentrating Mechanism: The Countercurrent System
To survive periods without water, animals, including humans, must excrete a concentrated (hypertonic) urine. They must eliminate waste products, including urea, without losing much water in the process. In contrast, under conditions of water excess, animals must excrete large amounts of water in a dilute (hypotonic) urine. The kidney has adapted to
P.621
handle day-to-day variations in water consumption by developing the countercurrent multiplier system. For this system to work, the hormone antidiuretic hormone (ADH), also called vasopressin, is required.
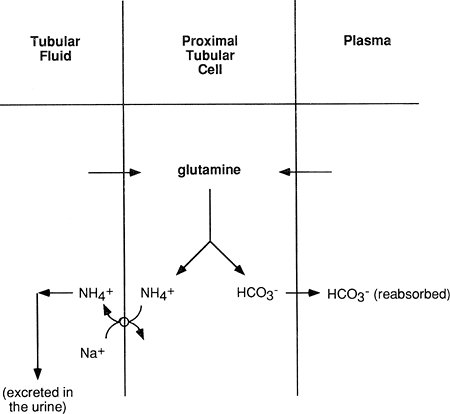 |
Figure 18-8. Excretion of H+ as ammonium ion. Glutamine diffuses into the proximal tubular cell from the plasma and from the tubular fluid. The active transport of ammonium into the tubular fluid occurs as a result of a sodium-ammonium counter-transport system. |
The countercurrent multiplier system exists in the loop of Henle, a long, curving portion of the nephron located between the proximal and distal tubules. The multiplier system has five basic steps and depends on active transport of sodium (and chloride) out of the ascending part of the loop. It also depends on impermeability of this part of the loop to water, which keeps water from following sodium out. Finally, this system relies on the permeability of collecting ducts to water. The five steps are outlined in the following section and are shown graphically in Figure 18-9.
Steps of the Countercurrent Multiplier System
When sodium is transported out of the ascending limb, the interstitial fluid surrounding the loop of Henle becomes concentrated.
Because water is impermeable across the ascending limb, water cannot follow sodium out of the ascending limb. Thus, the remaining filtrate becomes progressively diluted.
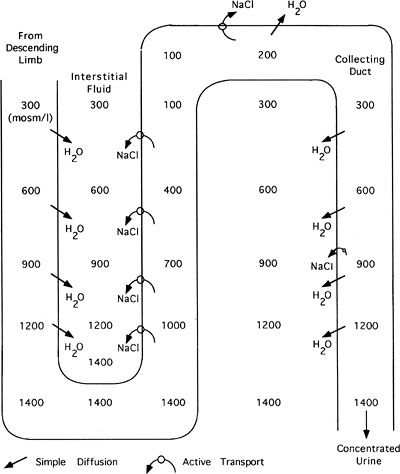
Figure 18-9. Formation of a concentrated urine in the presence of ADH. With ADH, water diffuses out of the collecting duct into the concentrated interstitium.
Water is permeable across the descending limb of the loop. Water leaves this section and flows down its concentration gradient into the surrounding interstitial space. This concentrates the descending limb fluid. As the fluid loops into the ascending limb, it is progressively diluted as sodium is pumped out.
The net and key result is the concentration of the interstitial fluid surrounding the loop of Henle. Concentration is highest surrounding the bottom of the loop, becoming more dilute as the ascending limb is followed up.
At the top of the ascending limb, tubular fluid is isotonic (equal in concentration to the plasma) or even hypotonic (more dilute compared with plasma).
P.622
P.623
Result of the Countercurrent Multiplier System
The goal of the countercurrrent system is to concentrate the interstitial fluid surrounding the loop of Henle (as described in step 4). This is vital because the final filtrate passes down the collecting ducts through this fluid. Permeability of the collecting ducts to water is variable. If permeability to water is high (as shown in Fig. 18-9), as the water moves down through the concentrated interstitium, it will diffuse out of the collecting duct and back into the peritubular capillary. The result is little water excretion and concentrated urine. In contrast, if permeability to water is low at this portion of the nephron, water will not diffuse out of the collecting duct and instead will be excreted in the urine. The urine will be dilute, as shown in Figure 18-10.
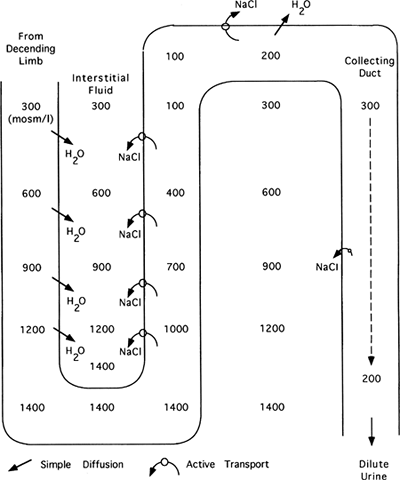 |
Figure 18-10. Formation of a dilute urine if ADH is absent. Note that because there is actually some NaCl transported out of the collecting duct, urine osmolality can be less than 300 mOsm/L. |
P.624
Role of Antidiuretic Hormone in Concentrating the Urine
Whether the collecting ducts are permeable to water or not is determined by the circulating level of the posterior pituitary hormone, ADH (also called vasopressin). Release of ADH from the posterior pituitary is increased in response to a decrease in blood pressure or an increase in extracellular osmolarity (decreased water concentration). ADH acts on the collecting tubules to increase water permeability. If blood pressure is low or plasma osmolarity is high, ADH release will be stimulated and water will diffuse into the peritubular capillaries, increasing blood volume and pressure, and decreasing extracellular osmolarity. In contrast, if blood pressure is too high or extracellular fluid is too dilute (decreased osmolarity), ADH release will be inhibited, causing the collecting ducts to be impermeable to water, and thus more water will be excreted in the urine, decreasing blood volume and pressure, and increasing extracellular osmolarity.
Sensors that measure blood pressure and control ADH release include the carotid and the aortic baroreceptors and a group of receptors in the left atrium. Sensors that measure extracellular osmolarity lie in the hypothalamus, adjacent to the cells that actually synthesize ADH. After synthesis in the hypothalamus, ADH is stored in the posterior pituitary.
Approximately 1,400 mOsmol/L is the most concentrated human urine can become. The most dilute concentration is less than 200 mOsmol/L.
Renal Endocrine Function
The kidney functions as an endocrine organ, not only with the production and release of renin but also with the production and release of two other hormones: 1,25-dihydroxyvitamin D3, important for bone mineralization; and erythropoietin, required for red blood cell production.
1,25-Dihydroxyvitamin D3
The kidney acts in conjunction with the liver to produce an active form of vitamin D, called 1,25-dihydroxyvitamin D3, from an inactive precursor consumed in the diet. The inactive form of vitamin D can also be produced in a reaction catalyzed by sunlight on a precursor present in the skin. Vitamin D is essential for maintenance of plasma calcium levels required for bone formation. The active form of vitamin D acts as a hormone by circulating in the blood and stimulating absorption of calcium and, to a lesser extent, phosphate across the small intestine and across the kidney tubules. Vitamin D also stimulates bone resorption (breakdown). Bone resorption releases calcium, and thus plasma calcium is increased by this mechanism as well.
Parathyroid hormone is the stimulus for the kidney to play its role in activating vitamin D3. Parathyroid hormone is released from the parathyroid gland in response to decreased plasma calcium. This is an example of a negative feedback cycle: decreased plasma calcium leads to increased parathyroid hormone, which leads to increased renal activation of vitamin D3.
P.625
Activation of vitamin D3 increases gut and kidney absorption of calcium, increasing plasma calcium and removing the stimulus for parathyroid release. Parathyroid hormone also directly stimulates bone resorption to release calcium into the plasma when necessary. Individuals who have renal disease frequently develop brittle, easily broken bones as a result of too little active vitamin D3.
Erythropoietin
The hormone that stimulates the bone marrow to increase the production of erythrocytes (red blood cells) is called erythropoietin. The cells of the kidney responsible for synthesizing and releasing erythropoietin respond to renal hypoxia. Individuals who have renal disease frequently demonstrate chronic and debilitating anemia.
Micturition
Micturition is the process of urination, which is the elimination of urine from the body. Micturition occurs when the internal and the external urethral sphincters at the base of the bladder are relaxed.
The bladder is composed of smooth muscle (the detrusor muscle), innervated by sensory neurons that respond to stretch, and parasympathetic fibers that travel from the sacral area to the bladder. An area of smooth muscle at the base of the bladder (the internal sphincter) is also innervated by parasympathetic nerves. An external sphincter composed of skeletal muscle is just below the internal sphincter and at the top of the urethra. The external sphincter is innervated by motor neurons from the pudendal nerve. When urine accumulates, stretch of the bladder is sensed by afferent fibers that send the information to the spinal cord. Parasympathetic nerves to the bladder are activated, causing contraction of the smooth muscle and opening of the internal sphincter. At the same time, the motor neurons going to the external sphincter are inhibited and the external sphincter is relaxed, causing micturition to occur.
Micturition, however, can be voluntarily inhibited. This is possible because at the same time that the afferent nerves are conveying information on bladder stretch to the spinal cord, they are also sending information up the cord to the brainstem and cortex, allowing one to be conscious of the need to void. Descending neurons from the brain can inhibit or stimulate the spinal reflex to void. These descending pathways inhibit urination by causing contraction of the skeletal muscles of the pelvis as well as the external sphincter. Descending pathways also block the firing of parasympathetic nerves to the internal sphincter. For urination to be facilitated, skeletal muscles can be voluntarily relaxed. Voluntary control over micturition becomes functional in children by or before the time they become 3 or 4 years of age. However, it may become interrupted at any time by central nervous system disease or injury or from spinal cord trauma.
P.626
Tests of Renal Function
Blood Urea Nitrogen
Urea is a nitrogenous waste product of protein and amino acid metabolism. One important job of the kidney is to eliminate this potentially toxic substance from the body. With declining renal function, blood urea nitrogen (BUN) levels increase. Measuring BUN therefore provides an indication of kidney health.
BUN, however, is not only determined by renal function. It can also be affected by circumstances not associated with the kidney, such as increased or decreased dietary protein intake, or any unusual cause of an increased protein breakdown, such as a muscle injury. Likewise, liver disease may decrease BUN, because the liver is necessary to convert ammonia to urea. Because BUN levels are affected by these other factors, BUN level alone may be an indiscriminate indicator of renal disease. Therefore, often the ratio of BUN to serum creatinine is reported as well. Normally BUN and creatinine co-vary, keeping this ratio at approximately 10:1. If BUN is affected by other than renal factors, however, this ratio may change. Ratios greater than 15:1 suggest a non-renal cause of urea elevation. Ratios less than 10:1 occur with liver disease.
Serum Creatinine
Creatinine is a product of muscle breakdown. Creatinine is excreted by the kidney through a combination of filtration and secretion. The concentration of creatinine in the plasma remains nearly constant from day to day. It varies slightly from approximately 0.7 mg/100 mL of blood in a small woman to 1.5 mg/l00 mL in a muscular man. Levels greater than these suggest the kidney is not clearing creatinine and indicate renal disease. Serum creatinine is very indicative of renal function. As a rough guide, a doubling of serum creatinine levels indicates a 50% reduction in renal function. Likewise, a tripling of normal creatinine levels indicates a 75% reduction in renal function. The clearance of creatinine may be used to estimate GFR.
Urinalysis
A urine sample may be easily obtained and evaluated for the presence of red blood cells, protein, glucose, and leukocytes, all of which are normally minimal to absent in the urine. Urine casts, which occur in the presence of high amounts of urine protein, may also be observed under some conditions of renal disease or injury. Urine osmolality (specific gravity) is measurable and should range between 1.015 and 1.025. Dehydration causes increased urine osmolality as more water is reabsorbed back into the peritubular capillaries. Overhydration results in decreased urine osmolality.
P.627
Cystoscopy
Cystoscopy is the process in which a lighted scope (cystoscope) is inserted up the urethra into the bladder. Bladder lesions, stones, and biopsy samples may be taken.
Voiding Cystourethrography
Voiding cystourethrography involves bladder catheterization and infusion of a radioactive dye to study the shape and size of the bladder. It can be used to detect and grade the degree of vesicoureteral reflux. If used inappropriately, cystourethrography may spread an unresolved bladder infection into the ureters or kidney.
Intravenous Urography
Intravenous urography is a technique in which a radiologic dye is injected intravenously, and x-ray films are taken sequentially as the dye filters through the kidney. Obstructions to flow in the glomeruli or tubules, vesicoureteral reflux, and stones may be visualized. A drawback to the use of this technique is the finding that some individuals are allergic to the dye and may suffer an anaphylactic reaction. High doses of radiation are involved.
Renal Ultrasound
Renal ultrasound uses the reflection of sound waves to identify renal abnormalities, including structural abnormalities, kidney stones, tumors, and other masses. Because it is non-invasive and does not involve radiation exposure, this technique is frequently used to evaluate renal function in children who have had a urinary tract infection. It does not, however, offer sufficient detail to evaluate vesicoureteral reflux, renal scarring, or inflammation.
Pathophysiologic Concepts
Alterations in Glomerular Filtration
Glomerular filtration depends upon the summation of forces favoring filtration of plasma out of the glomerulus and forces favoring reabsorption of filtrate into the glomerulus. Anything that affects the forces of filtration or the forces of reabsorption affects net glomerular filtration. Forces favoring filtration are capillary pressure and interstitial fluid colloid osmotic pressure. Forces favoring reabsorption are interstitial fluid pressure and plasma colloid osmotic pressure.
Alterations in Capillary Pressure
Capillary pressure depends on mean arterial pressure. Increased mean arterial pressure increases capillary pressure, tending to increase glomerular
P.628
filtration. A decrease in mean arterial pressure decreases capillary pressure and tends to decrease glomerular filtration. Autoregulation of afferent and efferent arterioles minimizes these changes unless the mean arterial blood pressure becomes too high (180 mmHg) or too low (80 mmHg).
Increased sympathetic activity and increased AII constrict afferent and efferent arterioles. These stimuli decrease the capillary pressure somewhat. However, because afferent and efferent arterioles are both affected, the responses tend to cancel each other out and GFR is nearly unaffected. Because the efferent arteriole response to sympathetic stimulation is especially sensitive, with heavy sympathetic stimulation, GFR may actually increase, as blood in the glomerulus backs up due to greater constriction of the efferent compared to afferent arteriole.
Alterations in Interstitial Fluid Colloid Osmotic Pressure
Interstitial fluid colloid osmotic pressure is low because very few plasma proteins or red blood cells filter out of the glomerulus into the interstitial space. With injury to the glomerulus or the peritubular capillaries, interstitial fluid colloid osmotic pressure may increase. If the interstitial fluid osmotic pressure increases, fluid is drawn out of the glomerulus and peritubular capillaries, and swelling and edema occur in Bowman's space and the interstitial space surrounding the tubule. Swelling in Bowman's space or around the tubule can interfere with further glomerular filtration and tubular reabsorption because of increasing interstitial fluid pressure. Swelling and edema may also collapse the delicate glomeruli or peritubular capillaries, leading to hypoxia and death of the nephrons in extreme situations.
Alterations in Plasma Colloid Osmotic Pressure
Plasma colloid osmotic pressure depends on the protein concentration of plasma. Plasma protein levels can decrease as a result of liver disease, protein loss in the urine, or protein malnutrition. Plasma colloid osmotic pressure is the major force favoring reabsorption of fluids back into the capillaries. If it decreases, less fluid reenters the capillaries. Fluid accumulates in the tubular and the peritubular (surrounding) areas. Again, swelling around the tubule can collapse the tubule and the surrounding peritubular capillaries, leading to hypoxia and death of the nephron.
Alterations in Interstitial Fluid Pressure
Interstitial fluid pressure in Bowman's space and surrounding the tubule can increase dramatically if the glomerular or peritubular capillaries are damaged. Increased interstitial fluid pressure opposes further glomerular filtration. Increased interstitial fluid pressure can cause collapse of the surrounding nephrons and the peritubular capillaries, leading to hypoxia and renal cell injury or death. When cells die, they release intracellular
P.629
enzymes that stimulate immune and inflammatory reactions (see Chapter 5), which further contribute to swelling and edema. Edema worsens interstitial fluid pressure. With loss of glomerular filtration, blood volume and electrolyte composition cannot be regulated.
Tubular Obstruction
One cause of increased interstitial fluid pressure is tubular obstruction. Obstruction present in the nephron causes fluid to back up into Bowman's capsule and the interstitial space. Unrelieved tubular obstruction can collapse the nephrons and capillaries and can lead to irreversible damage, especially to the renal papillae, which are the final site for urine concentration. Causes of obstruction include renal calculi (stones) and scarring from repeated kidney infections.
Azotemia
Azotemia refers to abnormal elevation of nitrogenous waste products in the blood such as urea, uric acid, and creatinine. Azotemia indicates a decrease in GFR, occurring either acutely or with chronic renal failure. Azotemia is an early sign of renal damage.
Uremia
Uremia is not a single event, but rather a syndrome (a constellation of symptoms) that develops in an individual who has end-stage renal disease. Because the kidney is pivotal in maintaining water, acid-base, and electrolyte balance and in removing toxic waste products, the symptoms of uremia are widespread and affect all the organs and tissues of the body. Common symptoms include fatigue, anorexia, nausea, vomiting, and lethargy. Intractable itching (pruritus) may occur. Hypertension, osteodystrophy, and uremic encephalopathy develop as well, with central nervous system changes, including confusion and psychosis, characterizing end stages. The range of symptoms appears to be caused by acidosis, anemia from decreased erythropoietin, and the buildup of all waste products.
Nephrotic Syndrome
Nephrotic syndrome is the loss of 3.5 g or more of protein in the urine per day. Under normal circumstances, virtually no protein is lost in the urine. Nephrotic syndrome usually indicates severe glomerular damage. Diabetic nephropathy is the most common cause of nephrotic syndrome. In individuals who do not have diabetes, different glomerular diseases may account for the disorder.
Loss of plasma proteins into the urine leads to sodium retention, hypoalbuminemia, and hypoimmunoglobulinemia. Thromboembolic complications are common. Clinical manifestations may include increased
P.630
susceptibility to infections (caused by hypoimmunoglobulins) and generalized edema, called anasarca. Hyperlipidemia (elevated plasma lipids) is associated with hypoalbuminemia, perhaps as a hepatic response to low levels of albumin.
Treatment consists of mechanisms to reduce proteinuria. These mechanisms include a soy-based protein, low-fat diet, with salt restrictions. Angiotensin-converting enzyme (ACE) inhibitors reduce proteinuria and have become a mainstay of treatment. Diuretics may be prescribed to increase fluid loss. Protein supplements may be provided to prevent malnutrition unless renal failure is suspected, in which case they are contraindicated as protein worsens renal failure.
Anasarca
Defined as a generalized edema in individuals suffering from hypoalbuminemia as a result of nephrotic syndrome or other conditions, anasarca is caused by a systemic decrease in capillary osmotic pressure. With a decrease in this major force favoring reabsorption of interstitial fluid back into the capillaries, edema of the interstitial space throughout the body occurs. The edema is usually soft and pitting and occurs early in the periorbital (surrounding the eye) regions, the ankles, and the feet.
Renal Osteodystrophy
Demineralization of bone occurring with renal disease is known as renal osteodystrophy. Renal osteodystrophy has many causes, including decreased renal activation of vitamin D3, leading to decreased calcium absorption across the gut, and subsequent reduced serum calcium levels. In addition, decreased renal function leads to an accumulation of phosphate ions, and hyperphosphatemia causes the secretion of parathyroid hormone, which leads to bone breakdown (resorption). Decreased serum calcium levels also stimulate parathyroid hormone release. An elevated bone breakdown contributes to easy bone fracturing.
Renal osteodystrophy also occurs as a result of the role bone plays in acting as a buffer for plasma H+. Bone buffering means bone takes up H+ and removes it from the general circulation to help maintain plasma pH. In taking up H+, calcium (which is also positively charged) is leached from the bone to maintain electrical balance in the bone. With chronic acidosis of advanced renal disease, bone buffering of H+ increases and the leaching of bone calcium becomes significant.
Treatment of renal osteodystrophy is aimed toward calcium and vitamin D supplementation. A phosphate-restricted diet is necessary.
Metabolic Acidosis/Renal Acidosis
Metabolic acidosis is a decrease in plasma pH not caused by a respiratory disorder. Chronic renal disease results in metabolic acidosis as a result of
P.631
reduced H+ excretion and altered bicarbonate reabsorption. The result is increased plasma H+ and lowered pH.
Increased H+ concentration contributes to bone resorption and causes neural and muscular function changes. The respiratory system is stimulated by the increase in hydrogen. Tachypnea (increased respiratory rate) occurs in an attempt to blow off the excess hydrogen as carbon dioxide. The respiratory response to renal acidosis is called respiratory compensation.
Uremic Encephalopathy
Uremic encephalopathy refers to neurologic changes seen in severe renal disease. Symptoms include fatigue, drowsiness, lethargy, seizures, muscle twitching, peripheral neuropathy (pain in the legs and feet), decreases in memory, and coma. Uremic encephalopathy appears to be caused by accumulation of toxins, alterations in potassium balance, and decreased pH. Treatment involves renal replacement with dialysis or transplantation if the condition is irreversible.
Renal Dialysis
The process of adjusting blood levels of water and electrolytes in a person who has poor or non-functioning kidneys is called renal dialysis. In this procedure, blood is directed past an artificial medium containing water and electrolytes in predetermined concentrations. The artificial medium is the dialyzing fluid. In the United States, more than 300,000 people require dialysis to survive.
By simple diffusion across a selectively permeable membrane, water and electrolytes in the blood move down their individual concentration gradients into or out of the dialyzing solution. As a result of simple diffusion, the final blood levels of these substances can be manipulated to be near normal. For example, sodium concentration in the dialyzing fluid can be adjusted to cause net loss or gain of sodium from the blood. Glucose is added to dialyzing fluid, at the same concentration present in blood, to ensure that glucose is not lost during dialysis. Urea is kept very low in the dialyzing fluid so that urea diffuses down its concentration gradient, out of the blood and into the artificial medium. There are two types of dialysis: hemodialysis and peritoneal dialysis.
Hemodialysis
In hemodialysis, dialysis is performed outside the body. Blood is passed from the body, through an arterial catheter, into a large machine. Two chambers separated by a semipermeable membrane are inside the machine. Blood is delivered to one chamber, dialyzing fluid is placed in the other, and diffusion is allowed to occur. Blood is returned to the body via a venous shunt.
P.632
Hemodialysis takes about 3 to 5 hours and is required approximately three times per week. By the end of the 2- to 3-day interval between treatments, salt, water, and pH balance are again abnormal, and the individual usually feels unwell. Hemodialysis contributes to problems of anemia because some red blood cells are destroyed in the process. Infection is also a risk.
Peritoneal Dialysis
In peritoneal dialysis, the individual's own peritoneal membrane is used as a natural, semipermeable barrier. Prepared dialysate solution (approximately 2 L) is delivered into the peritoneal cavity through an indwelling catheter placed under the skin of the abdomen. The solution is allowed to remain in the peritoneal cavity for a predetermined amount of time (usually between 4 and 6 hours). During this time, water and electrolytes diffuse back and forth between the circulating blood. The person can usually continue activity while the exchange takes place.
Peritoneal dialysis must be performed approximately four times per day. Because the procedure is performed daily (at home or at work), the fluctuations in plasma composition seen between hemodialysis treatments are minimized and convenience is increased. Unlike with hemodialysis, individuals usually feel well on a daily basis. However, peritoneal dialysis may lead to infections from the indwelling catheter or catheter malfunction.
Heart disease is common among patients who have renal failure for several reasons, including increasing age and the high incidence of diabetes mellitus or hypertension in patients on dialysis. Recent studies have shown that patients on dialysis who suffer a myocardial infarct have high mortality and poor long-term survival.
Kidney Transplantation
Defined as a form of kidney replacement available to patients who have renal failure, kidney transplantation involves placement of a donor kidney into the abdominal cavity of an individual suffering from end-stage renal disease. Transplanted kidneys can come from living or dead donors. The more similar the antigenic properties of the donated kidney are to the patient, the more likely the transplantation will be successful. With appropriate follow-up, approximately 94% of kidneys transplanted from cadavers and 98% from living donors function well after surgery. Long-term graft survival (10 years) is similar for both (approximately 78% for grafts from living donors versus 76% for grafts from cadavers). Each year, about 4,000 kidney patients die while waiting for an organ transplant.
Individuals receiving kidney donation must remain on a variety of immunosuppressant medications for life to prevent organ rejection. Ideally, immunosuppressive therapy should be individualized to match
P.633
the characteristics of the donor kidney (donor source, age, status of the donor kidney) and the characteristics of the recipient (age, race, reactive antibodies present, transplant number, and tolerance toward immunosuppressive therapy). In addition, the degree of histocompatibility between donor and recipient must be considered. If rejection does occur, it may happen during the very early postoperative period, through the first 3 months, or it may be delayed months or years after the transplantation. All individuals on immunosuppressive therapy are subject to increased risk of infection. Infections may be kidney related or independent of the renal system.
![]() ediatric Consideration
ediatric Consideration
Children who receive kidney transplantation frequently experience growth retardation. Whether this is a result of immunosuppressive therapy or a loss of a vital renal function such as erythropoietin production or vitamin D3 activation is unclear. Studies suggest that erythropoietin replacement can reduce growth complications of kidney transplantation. For all patients, medical therapy with recombinant human erythropoietin (rHuEPO) can also drastically reduce the symptoms of anemia, especially fatigue, and can improve quality of life.
Conditions of Disease or Injury
Hypospadias and Epispadias
Hypospadias is a congenital defect in males involving misplacement of the opening of the urethra from the tip of the penis to the ventral side. This condition may be slight or extreme. Some infants demonstrate the urethral meatus (opening) in the scrotal or perineal area. Epispadias, the congenital misplacement of the urethral opening to the dorsal side of the penis, is less common than hypospadias.
Clinical Manifestations
Difficulty or inability to urinate adequately while standing.
Chordee (a bending of the penis) may accompany hypospadias.
Inguinal hernia or undescended testes may accompany hypospadias.
Diagnostic Tools
Diagnosis is made on physical examination of the newborn or infant. Because other abnormalities may accompany hypospadias and epispadias, a thorough examination, including chromosomal studies, is recommended.
P.634
Complications
Ejaculatory dysfunction in the adult male may occur. If chordee is severe, penetration during sexual intercourse may be impossible.
If the dorsal urethral opening is extensive in epispadias, bladder exstrophy (exposure through the skin) is possible.
Treatment
Surgical correction may be necessary, preferably before the child is 1 or 2 years old. Circumcision should be avoided in the newborn so that the foreskin may later be used for repair.
Renal agenesis
Failure of the kidneys to develop during gestation is called renal agenesis. Renal agenesis may be unilateral or bilateral. Bilateral agenesis is incompatible with life.
Unilateral agenesis results in hypertrophy of the remaining kidney as it adapts to compensate functionally for the absent kidney. If the remaining kidney is malformed, successful compensation may not be possible.
Clinical Manifestations
Bilateral renal agenesis, called Potter's syndrome, is associated with facial anomalies and pulmonary disease. Infants with Potter's syndrome die in utero or soon after birth.
With unilateral renal agenesis, no symptoms are apparent if the remaining kidney is healthy. The remaining kidney may compensate and grow almost twice as big as otherwise expected. If the remaining kidney functions poorly, however, various disease manifestations may be present.
Diagnostic Tools
Prenatal ultrasound can often detect renal agenesis.
After birth, computerized axial tomography (CAT) scan or renal ultrasound is used to diagnose the condition.
Treatment
No treatment is required for unilateral agenesis if the remaining kidney is healthy.
If structural or functional defects are present in the remaining kidney, surgery may be required.
Vesicoureteral and Urethrovesical Reflux
Vesicoureteral reflux is the retrograde (backward) flow of urine from the bladder into the ureters and the kidney. Urethrovesical reflux is the backward flow
P.635
of urine from the urethra into the bladder. Vesicoureteral reflux usually occurs as a result of congenital misplacement of the ureters or urethra, which increases the likelihood of retrograde flow. Secondary causes include neurogenic bladder and repeated infections that cause structural scarring and impediment to the normal flow of urine. Urethrovesical reflux can occur during coughing or other activities that increase intra-abdominal pressure, especially in women because of the short length of the urethra.
Clinical Manifestations
Repeated urinary tract infections. These are especially suggestive of reflux in children younger than 5 years old.
Irritability and poor feeding in infants.
Diagnostic Tools
Intravenous urography and cystourethrography can help diagnose reflux. Grading of reflux determines treatment.
Complications
Renal obstruction and failure from repeated urinary tract infections may occur.
Treatment
Spontaneous remission may occur, in which case no treatment is required.
Surgery may be necessary to correct the defect if it is severe anatomically or functionally.
Prophylactic antibiotic therapy starting at birth (if the condition is known) may prevent repeated kidney infections.
Renal Calculi
Renal calculi refer to stones that occur anywhere in the urinary tract. Calculi are most commonly made up of calcium crystals. Less common components include struvite or magnesium, ammonium, uric acid, or combinations of these different substances.
Renal calculi can be caused by either increased urine pH (e.g., calcium carbonate stones) or decreased urine pH (e.g., uric acid stones). High concentration of stone-forming substances in the blood and urine and certain dietary habits or drugs can also result in stone formation. Anything that obstructs urine flow, leading to urine stasis (no movement) anywhere in the urinary tract, increases the likelihood of stone formation.
Calcium stones, usually formed with oxalate or phosphate, frequently accompany conditions that cause bone resorption, including immobilization and renal disease. Uric acid stones frequently accompany gout, a disease of increased uric acid production or decreased excretion.
P.636
Obesity and weight gain increase the risk of kidney stones due to increased excretion of overabundant calcium, oxalates and uric acid.
Clinical Manifestations
Pain is often colicky (rhythmic), especially if the stone is in the ureter or below. The pain may be intense. The location of pain depends on the site of the stone.
A stone in the kidney itself may be asymptomatic unless it causes obstruction or an infection develops.
Hematuria, caused by irritation and injury of the renal structures, is common with calculi.
Decreased urine output results if obstruction to flow occurs.
Dilute urine also results if obstruction to flow occurs, because the ability to concentrate urine may be interrupted by swelling around the peritubular capillaries.
Diagnostic Tools
Blood and urine tests may identify stone-forming substances.
Radiograph, ultrasound, or intravenous urography may locate a stone.
Complications
Urinary obstruction can occur upstream from a stone anywhere in the urinary tract. Obstruction above the bladder can lead to hydroureter, that is, abnormal distension of ureter with urine. Unrelieved hydroureter, or obstruction at or above the site where the ureter exits from the kidney, can lead to hydronephritis, swelling of the renal pelvis and collecting-duct system. Hydronephritis can cause the kidneys to be unable to concentrate the urine, leading to electrolyte and fluid imbalance.
Obstruction causes increased interstitial hydrostatic pressure and can lead to a decrease in GFR. Unrelieved obstruction can cause collapse of the nephrons and the capillaries, leading to ischemia of the nephrons as the blood supply is interrupted. Renal failure may develop if both kidneys are involved.
Anytime there is obstruction to the flow of urine (stasis), the chance of a bacterial infection increases.
Renal cancer may develop from repeated inflammation and injury.
Treatment
Increased fluid intake increases urine flow and helps wash out the stone. High fluid intake in individuals prone to calculi may prevent their formation.
If the stone content is identified, dietary modification may reduce the levels of the stone-forming substance.
Appropriate alteration of urine pH may encourage stone breakdown.
Extracorporeal (outside the body) lithotripsy (shock wave therapy) or laser therapy may be used to break apart the stone.
Surgery may be necessary to remove a large stone or to place a diversion tube around the stone to relieve obstruction.
P.637
Neurogenic Bladder
A neurogenic bladder is one that has experienced disruption of its neural connections. Neurologic disruption can be of the sensory or motor neurons; motor neurons affected may be located at the upper or lower level of the nervous system.
Interruption of sensory neurons leaves an individual unable to sense the need to void. Interruption of efferent nerves at the cortical or upper motor neuron level causes voluntary control of micturition to be lost. Because higher centers also facilitate micturition, voiding will be incomplete. If the interruption is of the lower motor neurons at the sacral area or below, the spinal reflex controlling micturition will be blocked and the bladder will not empty spontaneously.
Causes of neurogenic bladder include multiple sclerosis, which affects the cortical level; spinal cord transection; trauma; or tumors anywhere in the spinal cord. Poliomyelitis especially injures lower motor neurons, whereas diabetes mellitus is a common cause of sensory neuron damage.
Clinical Manifestations
Sensory neuron interruption leads to dribbling and overflow incontinence because bladder fullness cannot be felt.
Upper motor neuron and cortical interruption with an intact reflex arc leads to incontinence, small urine volume, and incomplete emptying. Infections may develop owing to urinary retention.
Lower motor neuron interruption, below the level of the reflex arc, leads to overflow incontinence.
Diagnostic Tools
History and physical examination will assist diagnosis.
Neuromuscular studies may help locate a lesion.
Complications
Repeated urinary tract infections may occur.
Chronic renal failure may develop from repeated infections and scarring.
Treatment
Sensory neuron interruption is treated with bladder training. The bladder is emptied at predetermined (2- to 4-hour) intervals either naturally or with a catheter.
Upper motor neuron and cortical interruption are treated by catheter drainage or manual initiation of the reflex arc by stroking the abdominal or the perineal area.
Lower motor neuron interruption is treated by catheter drainage or manual compression of the bladder.
P.638
Urinary Tract Infection
A urinary tract infection is an infection anywhere in the urinary tract, including the kidney itself, caused by proliferation of a microorganism. Most urinary tract infections are bacterial in origin, but fungi and viruses also may be implicated. The most common bacterial infection is by Escherichia coli, a fecal contaminant commonly found in the anal area.
Urinary tract infections are especially common in girls and women. One cause is the shorter urethra in the female, which allows the contaminating bacteria to gain access more easily to the bladder. The short urethra increases the likelihood that microorganisms deposited in the urethral opening during intercourse gain access to the bladder. Other factors that contribute to the frequency of urinary tract infections in girls and women include the cultural tendency for girls to delay urination and the irritation to the skin of the urethral opening that occurs during sexual intercourse. Pregnant women have a progesterone-dependent relaxation of all smooth muscle, including the bladder and the ureters, so they tend to retain urine in these parts of the tract, increasing the risk of bacterial growth. The pregnant uterus might also obstruct urine flow in some situations.
A protective factor against urinary tract infections in women is the estrogen-dependent production of a mucous coating of the bladder, which has antimicrobial functions. With menopause, estrogen levels fall and this protection is lost. Protection against urinary tract infections in both sexes is offered by the usually acidic nature of urine, which acts as an antibacterial agent.
Although urinary tract infections are less common in males, they can occur. A frequent cause in older men is benign prostatic hyperplasia (BPH) or prostatitis. The prostate is a walnut-sized gland that sits immediately below the opening of the bladder. Hyperplasia of the prostate may cause obstruction to flow, which predisposes an individual to an infection. Normally, prostatic secretions have an antimicrobial, protective effect.
Individuals who have diabetes also are at risk of frequent urinary tract infections because of the high glucose content of the urine, poor immune function, and increased frequency of neurogenic bladder. Persons who have a spinal cord injury or anyone using a urinary catheter to void are at increased risk of infection.
![]() ediatric Consideration
ediatric Consideration
In children, urinary tract infections are usually due to fecal-renal migration of E. coli. Although common, even one urinary tract infection in a child of either sex who is younger than 5 years of age is suggestive of vesicoureteral reflux and thus the child should be evaluated with renal ultrasound, cystourethrography, or intravenous urography to prevent subsequent renal damage.
![]() eriatric Consideration
eriatric Consideration
Age is a primary risk factor for urinary tract infections in both men and women, and urinary tract infections are the most common cause of infection in nursing home residents. The elderly are especially susceptible because of prostatic hypertrophy; neurogenic bladder associated with long-term diabetes mellitus; poor muscle functioning, leading to incomplete voiding; and delayed voiding because of reduced mobility getting to a bathroom.
P.639
Types of Urinary Tract Infections
Urinary tract infections may be divided into cystitis and pyelonephritis. Cystitis is an infection of the bladder, the most common site for an infection. Pyelonephritis is an infection of the kidney itself and can be either acute or chronic.
Acute pyelonephritis usually occurs as a result of an ascending bladder infection. It may also occur as a result of a blood-borne infection. Infections may be in both or in one kidney.
Chronic pyelonephritis may result from repeated infections and is usually found in individuals who have frequent calculi, other obstructions, or vesicoureteral reflux. With all kidney infections, inflammatory and immune responses cause interstitial edema and possible development of scar tissue. The tubules are most often affected and may atrophy. With chronic pyelonephritis, extensive scarring and obstruction of the tubules result. The ability of the kidneys to concentrate urine decreases as tubules are lost. The glomeruli are usually unaffected. Chronic renal failure may develop.
Clinical Manifestations
Cystitis typically presents with dysuria (pain on urination), increased frequency of urination, and a sense of urgency to urinate.
Lower back or suprapubic pain may occur, especially with pyleonephritis.
Fever accompanied by blood in the urine in severe cases.
Symptoms of infection in infants or young children may be non-specific and include irritability, fever, lack of appetite, vomiting, and very strong-smelling diapers.
Symptoms of infections in the elderly may be subtle; any elderly person who has abdominal symptoms such as nausea or vomiting should be assessed for urinary infection. Fever may or may not be present. Sometimes only increased agitation or confusion may develop, which
P.640
means that those caring for the elderly need to be especially cognizant of the frequency and subtleness of geriatric urinary tract infection. Asymptomatic infections in the elderly are also very common; there does not appear to be a benefit in treating elderly patients who have asymptomatic infection.
Acute pyelonephritis typically presents with
Fever.
Chills.
Flank pain.
Dysuria.
Chronic pyelonephritis may have manifestations similar to acute pyelonephritis. However, it can also include hypertension and may eventually lead to signs of renal failure.
Diagnostic Tools
Urine culture and sensitivity of the microorganism allow for identification and treatment.
White blood cells will be present in the urine with infection anywhere. White cell casts present in the urine suggest pyleonephritis rather than cystitis, since they indicate that white cells have been lysed in the tubules.
Complications
Renal or perirenal abscess formation may occur.
Renal failure may develop after repeated infections if both kidneys are involved.
Treatment
Women and girls in particular should be encouraged to drink fluids frequently (cranberry juice has been shown to reduce the incidence of cystitis) and go to the bathroom as needed to wash out microorganisms that may ascend the urethra.
Girls should be taught at a young age to wipe from front to back after urination to avoid contamination of the urethral opening with fecal bacteria.
Women should be encouraged to urinate after sexual intercourse to wash out ascending microorganisms.
Bubble baths are discouraged in young girls because of the irritation of the urethral opening that may occur, leading to access of bacteria to the urethra. Likewise, young girls should be discouraged from playing in the bathtub after shampooing.
Antibiotic therapy with a repeat urinalysis during or after drug therapy is required.
If chronic pyelonephritis is caused by an obstruction or reflux, surgical treatment specific to relieve these problems is necessary.
P.641
Glomerulonephritis
Glomerulonephritis is an inflammation of the glomerulus. Types of glomerulonephritis include acute, rapidly progressive, and chronic.
Acute Glomerulonephritis
A sudden inflammation of the glomerulus is called acute glomerulonephritis. Acute inflammation of the glomerulus occurs as a result of deposition of antibody-antigen complexes in the glomerular capillaries. Complexes usually develop 7 to 10 days after a pharyngeal or skin streptococcal infection (poststreptococcal glomerulonephritis) but may follow any infection. See page C15 for illustrations.
An inflammatory reaction is initiated in the glomerulus after deposition of antibody-antigen complexes. Inflammatory reactions in the glomeruli (or anywhere else in the body; see Chapter 5) cause complement activation and mast-cell degranulation, leading to increased blood flow, increased glomerular capillary permeability, and increased glomerular filtration. Plasma proteins and red blood cells leak through the damaged glomeruli. The eventual breakdown of the glomerular membrane causes swelling and edema of Bowman's space. This swelling and edema increase interstitial fluid pressure, which can collapse any functioning glomeruli in the area. Eventually, increased interstitial fluid pressure opposes further glomerular filtration.
Activation of the inflammatory reaction also draws white blood cells and platelets into the area of the glomerulus. Activation of coagulation factors occurs with inflammation, which can lead to fibrin deposits, scarring, and the loss of functional glomeruli. Glomerular membranes thicken and GFR decreases further.
Acute glomerulonephritis usually resolves with specific antibiotic therapy, especially in children. Some adults may not recover and may develop rapidly progressive glomerulonephritis or chronic glomerulonephritis.
Rapidly Progressive Glomerulonephritis
Rapidly progressive glomerulonephritis is an inflammation of the glomeruli that occurs so rapidly that there is a 50% decrease in GFR within 3 months of disease onset. Rapidly progressive glomerulonephritis can occur from a worsening of acute glomerulonephritis, from an autoimmune disease, or may be idiopathic (unknown) in origin.
Rapidly progressing glomerulonephritis is associated with diffuse proliferation of glomerular cells within Bowman's space. Such proliferation gives rise to the appearance of a crescent-shaped structure obliterating Bowman's space. GFR decreases, leading to renal failure.
P.642
Goodpasture's syndrome is a type of rapidly progressing glomerulonephritis caused by auto-antibodies produced against the glomerular cells themselves. Pulmonary capillaries are also attacked. Extensive scarring of the glomeruli results. Renal failure frequently occurs within weeks or months. The cause of Goodpasture's syndrome is unknown.
Chronic Glomerulonephritis
Chronic glomerulonephritis is the long-term inflammation of the glomerular cells. It may occur as a result of unresolved acute glomerulonephritis, or it might develop spontaneously. Chronic glomerulonephritis commonly occurs after years of subclinical glomerular injury and inflammation, associated with only slight hematuria (blood in the urine) and proteinuria (protein in the urine).
Common causes include diabetes mellitus and long-standing hypertension. Both of these diseases are associated with significant and repeated glomerular injury. The outcome is diffuse scarring and glomerular deterioration. Tubular atrophy frequently accompanies glomerular breakdown. Individuals with chronic glomerulonephritis who have diabetes or who are even mildly hypertensive have a poor prognosis for long-term renal function. Chronic glomerulonephritis may also accompany long-standing systemic lupus erythematosus.
Clinical Manifestations
All types of glomerulonephritis are associated with
Decreased urine volume.
Blood in the urine (brownish-colored urine), either gross or subtle.
Fluid retention.
Diagnostic Tools
Hematuria as measured by urinalysis.
Red blood cell casts in the urine.
Proteinuria greater than 3 to 5 g/day.
Decreased GFR as measured by creatinine clearance.
If the condition is caused by acute poststreptococcal glomerulonephritis, antistreptococcal enzymes, such as antistreptolysin-O and antistreptokinase, will be present.
Complications
Renal failure may develop.
Treatment
If the condition develops following acute poststreptococcal glomerulonephritis, antibiotic therapy is required.
Autoimmune destruction of the glomeruli may be treated with corticosteroids for immunosuppression.
Anticoagulants to decrease fibrin deposits and scarring can be used in rapidly progressive glomerulonephritis.
Strict glucose control in diabetics has been shown to slow or reverse the progression of glomerulonephritis. Research has shown that the angiotensin-converting enzyme (ACE) inhibitors can reduce glomerular damage in diabetics even if frank hypertension is not evident.
ACE inhibitors can reduce glomerular damage in individuals with chronic hypertension.
P.643
Myoglobinuria
Myoglobinuria or rhabdomyolysis is the presence of high levels of myoglobin in the urine. Myoglobin is an intracellular protein found in muscle. With muscle-cell damage, especially a crush injury or major body trauma, myoglobin levels in the blood can rise precipitously. A severe electrical burn may cause significant muscle damage and myoglobinuria as well. Normally, myoglobin is filtered in the urine and then is totally reabsorbed into the peritubular capillaries by active transport. However, when large amounts of myoglobin are present in the blood, its threshold for reabsorption is exceeded and it spills in the urine. Large amounts of myoglobin in the urine filtrate clog the tubules, leading to obstruction, inflammation, and tubular and glomerular injury. Renal failure can result.
Individuals have reported myoglobinuria after intense episodes of athletics or long-distance running. It appears that besides muscle breakdown, the pounding or jarring effect on the kidney may contribute to the filtration of proteins in these circumstances. Sepsis and hyperthermia may also lead to myoglobinuria.
Clinical Manifestations
Hematuria.
Diagnostic Tools
Red blood cells, protein, and protein casts are present in the urine.
Elevated levels of plasma creatine phosphokinase, a product of muscle metabolism, can be measured.
Complications
Electrolyte imbalance, as a result of potassium and phosphate release from injured muscle cells, may occur.
Renal failure may develop.
Treatment
Administration of sodium bicarbonate to facilitate renal elimination of myoglobin.
Flushing the kidney with an osmotic diuretic (mannitol).
Correction of electrolyte and volume imbalances.
Short-term dialysis, allowing time for the kidneys to recover, may be necessary.
P.644
Hemolytic Uremic Syndrome
Hemolytic uremic syndrome is a condition of injury to the endothelial cells of the glomeruli as a result of a viral, rickettsial, or bacterial infection, frequently infection by the bacteria E. coli O157 from inadequately cooked meat, especially hamburger. Damage to the glomerular endothelial cells results in swelling and edema and in a narrowing of the capillary to blood flow. Narrowing of the capillary causes injury to passing red blood cells, which are then broken down in the spleen, resulting in hemolytic anemia. Damage to the glomerular cells stimulates inflammatory reactions, including complement activation, fibrin deposition, accumulation of white blood cells, and the release of a variety of vasoactive peptides. Platelets accumulate, leading to clotting and to a decrease in their circulating levels. Blood flow to the kidney may decrease, and scarring may occur.
Clinical Manifestations
Vomiting and abdominal pain.
Bloody diarrhea.
Bruising from thrombocytopenia (decreased platelets).
Oliguria (decreased urine output).
Diagnostic Tools
Urine culture may identify the causative organism.
Obstruction and inflammation may be visible using ultrasound or radiographs.
Complications
Renal failure may occur. It may be either temporary or permanent.
Treatment
Dialysis is required if renal failure develops.
Blood transfusions may be used.
Correction of fluid and electrolyte imbalance is required.
![]() ediatric Consideration
ediatric Consideration
Children are especially susceptible to renal damage after infection with E. coli O157. Many children infected with this bacterium require dialysis or even die.
P.645
Renal Failure
Renal failure is the loss of function in both kidneys. Because the kidneys have such a vital role in maintaining homeostasis, renal failure is associated with multiple systemic effects. All attempts at preventing renal failure are essential. If renal failure does occur, it must be treated aggressively.
The U.S. National Kidney Foundation's Kidney Disease Outcomes Quality Initiative has revised and defined the stages of chronic kidney disease. Stages are based on the presence or absence of symptoms and on progressively decreasing GFR, corrected for body size (per 1.73 m2). Normal GFR for a healthy adult is approximately 120 to 130 mL/minute. The stages of kidney disease are as follows:
Stage 1: Kidney damage (pathological abnormalities or markers of damage including abnormalities in blood or urine tests or in imaging studies) with normal or near-normal glomerular filtration rate, at or above 90 mL/min ( 75% of normal).
Stage 2: Glomerular filtration rate between 60 and 89 mL/min (approximately 50% of normal), with evidence of kidney damage. This stage is considered one of diminished renal reserve. Remaining nephrons are highly susceptible to failing themselves as their load becomes overwhelming. Additional renal insults hasten the decline.
Stage 3: Glomerular filtration rate between 30 and 59 mL/min (25 to 50% of normal). This stage is considered one of renal insufficiency. Nephrons continue to die.
Stage 4: Glomerular filtration rate between 15 and 29 mL/min (12 to 24% of normal), with fewer nephrons remaining.
Stage 5: End-stage renal failure; glomerular filtration rate of less than 15 mL/min (< 12% of normal). Few functioning nephrons remain. Scar tissue and tubular atrophy are present throughout the kidneys.
Renal failure also is categorized as acute renal failure, which occurs suddenly and is usually reversible, or chronic renal failure, which is associated with progressive, irreversible loss of renal function. Chronic renal failure usually develops after years of renal disease or damage, but may occur rapidly in some situations. Chronic renal failure inevitably leads to renal dialysis, transplantation, or death.
Acute Renal Failure
Causes of acute renal failure have been separated into three general categories: prerenal, intrarenal, and postrenal. Identifying the cause of acute renal failure is accomplished by a study of the patient's history and the quantity and quality of his or her urine.
Prerenal failure is the most common cause of acute renal failure. Prerenal failure occurs as a result of conditions unrelated to the kidney but that damage the kidney by affecting renal blood flow. Causes of prerenal failure include anything that severely reduces systemic blood pressure, leading to shock, such as a myocardial infarct, an anaphylactic reaction,
P.646
severe blood loss or volume depletion, a burn, or sepsis (a blood-borne infection). Surgical procedures resulting in a prolonged decrease in renal blood flow can also cause prerenal failure. Renal autoregulation is unsuccessful with a mean systemic blood pressure below 80 mmHg.
Interruption of renal blood flow, and therefore oxygen delivery, can irreversibly damage the kidneys within 30 minutes. The tubules are most susceptible to the effects of hypoxia, and ischemic tubular necrosis (tubular cell death caused by decreased oxygenation) frequently develops.
Intrarenal failure is a type of acute renal failure that occurs as a result of primary damage to kidney tissue itself. It has many causes, including glomerulonephritis, acute pyelonephritis, and myoglobinuria.
With intrarenal failure, kidney cell damage usually occurs as a result of ischemic tubular necrosis. This tends to blur the distinction between pre-renal failure and intrarenal failure because a main cause of ischemic tubular necrosis is decreased renal blood flow.
Tubular necrosis also can result from direct action of nephrotoxic (damaging to the nephron) drugs, such as heavy metals and organic solvents. Aminoglycoside antibiotics, such as gentamicin, are also nephrotoxic. Radiopaque contrast media used for viewing the cardiac chambers or the gastrointestinal (GI) tract can be nephrotoxic in susceptible individuals. Ingestion of toxic amounts of analgesic mixtures, especially codeine and caffeine, may lead to acute tubular necrosis. Sporadic reports of elderly individuals who use non-steroidal anti-inflammatory drugs having acute renal failure after an intense athletic event (e.g., a marathon run in heat) are cause for concern.
Postrenal failure is a type of acute renal failure that occurs as a result of conditions that affect the flow of urine out of the kidneys and includes injury to or disease of the ureters, bladder, or urethra. The usual cause of postrenal failure is obstruction. Obstruction can occur in response to many factors, including untreated calculi, a tumor, repeated infections, prostatic hyperplasia, or a neurogenic bladder.
Most cases of renal failure are associated with low urine output. Occasionally, high-output failure may occur. In this case, urine production continues. This scenario is usually associated with a better outcome.
Recovery from acute renal failure typically occurs after a few weeks, but occasionally takes as long as 6 weeks after the onset of oliguria (decreased urine output). Recovery begins with diuresis (increased urine output). Although urine is being produced, alterations in electrolyte balance continue. After the diuretic phase, the recovery phase of acute renal failure follows, during which renal function and electrolyte balance return. Full recovery usually occurs within 1 to 2 years. Some individuals may never recover total renal functioning.
Clinical Manifestations
Oliguria may occur, especially if the failure is caused by ischemia or by obstruction. Oliguria results from decreased GFR.
Toxic tubular necrosis may be non-oliguric (high output) and is associated with the production of an adequate volume of dilute urine.
P.647
Diagnostic Tools
A good history identifies precipitating causes of renal failure.
Laboratory finding of azotemia (increased nitrogenous compounds in the blood), and elevated BUN and creatinine confirm diagnosis.
Laboratory findings of hyperkalemia (increased potassium in the blood) and acidosis are common.
Complications
Fluid retention from non-functioning kidneys may lead to edema, congestive heart failure, or water intoxication.
Alterations in electrolytes and pH may cause uremic encephalopathy.
If the hyperkalemia is severe ( 6.5 mEq/L), dysrhythmia and muscle weakness may occur.
Treatment
Prevention of acute renal failure is essential. Individuals experiencing shock should be quickly treated with fluid replacement to support blood pressure. Individuals at risk of developing acute renal failure, for instance, those about to undergo heart surgery, may be given an osmotic diuretic before surgery to increase renal function. Likewise, adequate hydration before nephrotoxic drugs are administered may prevent acute renal failure. For patients at high risk of suffering renal failure, the use of nephrotoxic drugs and intravenous radioactive dyes must be shown to be essential before they are employed, and their use may be contraindicated in some cases.
If renal failure does occur, data suggest that prevention of the oliguric phase results in a better prognosis. Prevention of oliguria involves:
Aggressive plasma volume expansion.
Diuretics to increase urine production.
Vasodilators, especially dopamine, given to increase renal blood flow.
Dietary restrictions on potassium and protein are often implemented in acute renal failure. High-carbohydrate intake prevents the metabolism of proteins and reduces nitrogenous waste production.
Antibiotic therapy to prevent or treat infections may be necessary because of the high rate of sepsis seen with acute renal failure.
Continuous peritoneal dialysis is often employed during the oliguric stage of acute renal failure to give the kidneys time to recover. Dialysis also prevents the buildup of nitrogenous wastes, stabilizes electrolytes, and reduces fluid overload.
Chronic Renal Failure
Chronic renal failure is the progressive, relentless destruction of renal structure. Chronic renal failure can result from virtually any of the diseases described in this chapter. In addition, analgesic nephropathy, the destruction
P.648
of the renal papillae related to the daily use of analgesic medications for many years, may lead to chronic renal failure in susceptible individuals. Regardless of the cause, unremitting deterioration of the kidneys occurs as indicated by a progressive fall in GFR.
Early in its course, fluid balance, salt handling, and waste accumulation are variable and depend on the part of the kidney in failure. Until renal function has decreased to less than 25% of normal, clinical manifestations of chronic renal failure may be minimal as surviving nephrons take over the functions of those lost. Surviving nephrons increase their rates of filtration, reabsorption, and secretion, and undergo hypertrophy in the process. As more nephrons progressively die, remaining ones have an increasingly difficult job, which leads to their own damage and eventual death. Part of this cycle of death appears related to the demands on remaining nephrons for increased protein reabsorption. With progressive loss of nephrons, scar tissue accumulates and renal blood flow may be reduced. Renin release may increase, which, coupled with fluid overload, can lead to hypertension. Hypertension accelerates renal failure, perhaps by increasing the filtration (and therefore the demands for reabsorption) of plasma proteins and by causing oxidative stress.
Failure of adequate production of erythropoietin by the kidneys frequently leads to anemia and a resultant fatigue that negatively impacts quality of life. In addition, chronic anemia leads to decreased tissue oxygenation throughout the body and activates reflexes aimed at increasing cardiac output to improve oxygenation. These reflexes involve activation of the sympathetic nervous system and an increase in cardiac output. Ultimately, these changes predispose an individual with renal failure to develop congestive heart failure, making chronic kidney disease an independent risk factor for cardiovascular disease.
Clinical Manifestations
In stage 1 renal failure, no symptoms may be apparent.
As disease progresses, reduced production of erythropoietin causes chronic fatigue, and early signs of tissue hypoxia and cardiovascular compromise may develop.
As disease progresses, polyuria (increased urine output) occurs as the kidneys are unable to concentrate the urine.
During the final stages of renal failure, urine output decreases because of low GFR.
Diagnostic Tools
Radiographs or ultrasound will show small, atrophied kidneys.
Serum BUN, creatinine, and GFR will be abnormal.
Hematocrit and hemoglobin are reduced.
Plasma pH is low.
An elevated respiratory rate indicates respiratory compensation for metabolic acidosis.
P.649
Complications
With progression of renal failure, volume overload, electrolyte imbalance, metabolic acidosis, azotemia, and uremia occur.
In stage 5 renal failure (end-stage disease), severe azotemia and uremia are present. Metabolic acidosis worsens, which significantly stimulates respiratory rate.
Hypertension, anemia, osteodystrophy, hyperkalemia, uremic encephalopathy, and pruritus (itching) are common complications.
Decreased production of erythropoietin may lead to cardiorenal anemia syndrome, a self-perpetuating triad of anemia, cardiovascular disease, and renal disease that ultimately leads to increased morbidity and mortality.
Congestive heart failure may develop.
Without treatment, coma and death result.
Treatment
Prevention of renal failure is the most important goal. Prevention includes lifestyle changes and drugs when necessary to control hypertension, good glycemic control in diabetics, and the avoidance of nephrotoxic drugs whenever possible. Long-term use of codeine-containing analgesics and possibly non-steroidal anti-inflammatory drugs (NSAIDs) should be avoided, especially in persons who have renal compromise. Early diagnosis and treatment of systemic lupus erythematosus and other diseases known to damage the kidneys is essential.
Treatments are modified as progression worsens.
For stages 1, 2, and 3 renal failure, the goals are to slow further nephron loss, primarily by the use of protein restriction and antihypertensive medications. Angiotensin-converting enzyme (ACE) inhibitors are especially helpful in slowing progression.
Because of the relationship between congestive heart failure and anemia associated with chronic kidney disease, the Renal Anemia Management Period (RAMP) has been proposed. RAMP is defined as the time following the onset of chronic kidney disease when early diagnosis and treatment of anemia will slow kidney disease progression, delay cardiovascular complications, and improve quality of life. Treatment of anemia is by administration of recombinant human erythropoietin (rHuEPO). This drug has been shown to dramatically improve quality of life and reduce the need for transfusions. It also significantly improves cardiac function.
For later stages, treatment is geared toward correcting fluid and electrolyte imbalances.
For end-stage disease, treatment includes dialysis or renal transplantation.
At all stages, prevention of infection is important.
P.650
Childhood Kidney Cancer: Wilms' Tumor
Wilms' tumor is a cancer of any part of the kidney that typically develops in children younger than 4 years of age. Because of its early onset, Wilms' tumor likely develops after one or more mutations in important tumor suppressor genes.
Wilms' tumor is a solid tumor that can grow to a large size. It may be encapsulated (contained within the capsule of the kidney). Staging and prognosis of the disease depend on encapsulation and spread. Encapsulation is associated with a favorable prognosis, whereas spread of the tumor outside of the abdominal area to the lungs is associated with a poorer outcome. Overall, prognosis is good, with an approximately 90% survival rate.
Clinical Manifestations
A large abdominal mass may be noted by parents or a health-care provider.
Vomiting, abdominal pain, and hematuria may be present.
Diagnostic Tools
A careful history can raise suspicion of Wilms' tumor.
Physical examination may identify the mass.
CT scan or ultrasound may confirm the diagnosis.
Treatment
Chemotherapy and surgery are used aggressively to destroy the tumor.
Adult kidney cancer
Most adult kidney cancer is a result of renal cell carcinoma. This cancer is especially common in the sixth or seventh decade of life, and is more common in males than in females. Risk factors for kidney cancer include repeated kidney stone irritation, smoking, and obesity. Occupational chemical exposures may also increase the risk.
Renal cell carcinoma is often symptomless in its early stages. When symptoms do occur, they often include hematuria and the presence of a flank mass. At diagnosis, tumors are staged. Treatment and outcomes depend on staging, with outcomes ranging from 85% survival for stage I tumors to less than 10% survival for stage IV tumors.
Clinical Manifestations
Hematuria is the most common manifestation. It may be frankly visible or may be microscopic and sporadic.
A flank mass may be palpable. Flank pain may be present as well.
Polycythemia may be present, reflecting alteration in the renal control of hematopoiesis.
Fever may accompany the cancer.
P.651
Diagnostic Tools
The use of CT scanning has improved the diagnosis of suspected renal cancer.
Ultrasound, renal angiography, and MRI may confirm the diagnosis.
Complications
Metastasis to the lungs or elsewhere may precede diagnosis.
Treatment
Surgery to remove the affected kidney is usually performed. In patients who already have only one functioning kidney, surgical techniques to preserve renal function in the affected kidney may be attempted.
Chemotherapy and immunotherapy may be used as well.
Use of antiangiogenic agents such as inhibitors of vascular endothelial growth factor is being evaluated in clinical trials.
Selected Bibliography
Dulczak, S., & Kirk, J. (2005). Overview of the evaluation, diagnosis, and management of urinary tract infections in infants and children. Urology Nurse 25, 185 191.
El Nahas, A.M., & Bello, A.K. (2005). Chronic kidney disease: the global challenge. Lancet 365, 331 340.
George, D.J., & Kaelin, Jr., W.G. (2003). The von Hippel-Lindau protein, vascular endothelial growth factor, and kidney cancer. New England Journal of Medicine 349, 419 421.
Go, A.S., Chertow, G.M., Fan, D., McCulloch, C.E., & Hsu, C. (2004). Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. New England Journal of Medicine 351, 1296 1305.
Guyton, A.C., & Hall, J. (2006). Textbook of medical physiology (11th ed.). Philadelphia: W.B. Saunders.
Hotoura, E., Argyropoulou, M., Papadopoulou, F., Giapros, V., Drougia, A., Nikolopoulos, P., et al. (2005). Kidney development in the first year of life in small-for-gestational-age preterm infants. Pediatric Radiology 35, 991 994.
Mathieson, P.W. (2005). Glomerulonephritis: is it worth worrying about? Clinical Medicine 5, 264 266.
McLellan, F. (2003). U.S. surgeons do first triple swap kidney transplantation. The Lancet 362, 456.
Meyer-Bahlburg, H.F.L. (2005). Gender identity outcome in female-raised 46, XY persons with penile agenesis, cloacal exstrophy of the bladder, or penile ablation. Archives of Sexual Behavior 34, 423 438.
National Kidney Foundation. (2005). 25 facts about organ donation and transplantation. Retrieved October 15, 2005 in http://www.kidney.org/news/newsroom/fsitem.cfm?id=30.
Porth, C.M. (2005). Pathophysiology: Concepts of altered health states (7th ed.). Philadelphia: Lippincott Williams & Wilkins.
Rao, M., & Pereira, B.J.G. (2005). Optimal anemia management reduces cardiovascular morbidity, mortality, and costs in chronic kidney disease. Kidney International 68, 1432 1438.
P.652
Taylor, E.N., Stampfer, M.J., & Curhan, G.C. (2005). Obesity, weight gain, and the risk of kidney stones. Journal of the American Medical Association 293, 455 462.
Weber, M., Dindo, D., Demartines, N., Ambuhl, P.M., & Clavien, P.-A. (2002). Kidney transplantation from donors without a heartbeat. New England Journal of Medicine 347, 248 255.
Wilcox, C.S. (2005). Oxidative stress and nitric oxide deficiency in the kidney: a critical link to hypertension? American Journal of Physiology(Regulatory, Integrative and Comparative Physiology) 289, R913 ssR935.
EAN: 2147483647
Pages: 26
- ERP Systems Impact on Organizations
- The Second Wave ERP Market: An Australian Viewpoint
- Data Mining for Business Process Reengineering
- Relevance and Micro-Relevance for the Professional as Determinants of IT-Diffusion and IT-Use in Healthcare
- Development of Interactive Web Sites to Enhance Police/Community Relations