5. The Immune System
Authors: Corwin, Elizabeth J.
Title: Handbook of Pathophysiology, 3rd Edition
Copyright 2008 Lippincott Williams & Wilkins
> Table of Contents > Unit II - Effective and Ineffective Health Protection > Chapter 5 - The Immune System
function show_scrollbar() {}
Chapter 5
The Immune System
The immune system works to protect the body from infection by microorganisms, to assist in healing, and to remove or repair damaged cells if an infection or injury does occur. The immune system also identifies self from non-self: the cells, tissues, and organs of the host versus the cells and tissues of foreign origin. In addition, the immune system recognizes and eliminates host cells that have been altered by intracellular viruses or cancer. Abnormal responses by the immune system can cause an attack against the body's own cells, the development of cancer, or an inability to fight or heal from infection.
Physiologic Concepts
Cells of the Immune Response
The immune response is under the control of specialized cells, the white blood cells. White blood cells protect the body from infection and cancer and assist in healing. White blood cells include the neutrophils, eosinophils, basophils, monocytes, and macrophages, and the B and T lymphocytes. Platelets are fragments of cells that also play a role in healing. All white blood cells and platelets derive from a basic stem (originator) cell, called the pluripotent stem cell, in the bone marrow. From this cell, succeeding generations of stem cells differentiate and commit to producing one type of cell. Blood cells and their differentiation are discussed in Chapter 12.
P.125
Neutrophils, Eosinophils, and Basophils
Neutrophils, eosinophils, and basophils are called granulocytes because of the cytoplasmic granules that dominate their appearance under the microscope. The granulocytes remain in the bone marrow or circulation until they are drawn to an area of infection, inflammation, or trauma by substances released from damaged tissues, by microorganisms, or by the B or T lymphocytes. The cytoplasmic granules contain enzymes that break down and destroy microorganisms and digest cellular debris. Once granulocytes complete their function, they die. In a serious infection, granulocytes may only survive a few hours.
The neutrophils are the first white blood cells to arrive at an area of injury or infection and are key players in the processes of inflammation. Neutrophils arriving on the scene begin phagocytizing cells and debris immediately. They also release chemicals that attract other white blood cells to the area, in a process called chemotaxis. Neutrophils initiate the inflammatory responses of vasodilation and increased capillary permeability. Clinically, neutrophils are often referred to as polymorphonuclear cells (PMNs) or segmented neutrophils ( segs ) because of the segmented appearance of their multi-lobed nuclei. In adults, approximately 50% of circulating white blood cells are neutrophils.
Eosinophils have several functions. First, they are involved in the allergic response (described later). Second, they are important in the defense against parasitic (helminthic) infections. They also appear to perform a protective role for the host by helping to end the inflammatory reaction. The eosinophils phagocytize cell debris, although to a lesser degree than do neutrophils. Eosinophils normally account for only 1 to 3% of circulating white blood cells. The level of eosinophils may increase during an allergic response or in response to helminthic infection.
Basophils circulate in the bloodstream and, when activated by injury or infection, release histamine, bradykinin, and serotonin. These substances increase capillary permeability and blood flow to the area, bringing to the area other mediators required to eliminate infection and promote healing. Basophils secrete the natural anti-clotting substance heparin, which ensures that clotting and coagulation pathways do not continue unchecked. Basophils are also involved in producing allergic responses. They are similar in function to important initiators of tissue inflammation, the mast cells, but unlike mast cells, basophils circulate in the blood. Basophils account for up to 1% of circulating white blood cells.
Monocytes and Macrophages
Monocytes circulate in the blood and enter injured tissue across capillary membranes that become permeable as part of the inflammatory reaction (described later). Monocytes are not phagocytic, but after several hours in the tissue area, they mature into macrophages. Macrophages are large cells capable of ingesting large quantities of cell debris and bacteria. Macrophages can phagocytize lysed red blood cells and other white blood
P.126
cells. Some macrophage cells colonize tissues such as skin, lymph nodes, and lungs for months or years. These cells are readily available to scavenge microorganisms that may enter the body through those routes. The monocyte-macrophage cell system is called the reticuloendothelial system. Circulating monocytes account for approximately 3 to 7% of white blood cells in adults.
Lymphocytes
Lymphocytes include the B and T lymphocytes and a type of cell called the natural killer (NK) cell. Lymphocytes are produced in the bone marrow and mature there or in other lymphoid tissues.
B lymphocytes (B cells) mature in the bone marrow. After maturation, a B cell circulates in the blood in an inactive state and becomes active only after exposure to a specific molecule, usually a protein or large carbohydrate of foreign origin, to which it has been genetically programmed during fetal development to respond. When activated, the B cell becomes a plasma cell, a specialized cell that mounts an immune response against the molecule that activated it. B lymphocytes comprise the humoral immune system, meaning that they circulate in the blood (the humor).
T lymphocytes comprise the cellular immune system. T cell maturation occurs during passage through the thymus gland. Like a B cell, the mature T cell stays inactive until it encounters the specific molecule to which it has been programmed during development to respond; once it does so, it becomes activated and may directly attack and destroy the cell expressing that molecule. The T cell may also release chemicals that alert B cells to the presence of the invader, thereby initiating a humoral response. T cells can stimulate or in some circumstances inhibit the inflammatory responses via the release of pro- or anti-inflammatory peptides known as cytokines. T cells are important for recognizing and destroying parasites and viruses that hide intracellularly, where the B cells are unable to encounter them.
The natural killer (NK) cells also react to foreign molecules, but do not demonstrate specificity, that is, they may respond to more than one foreign molecule. The natural history of NK cell origin and maturation is unclear. In total, circulating lymphocytes account for approximately 30% of white blood cells in adults.
Platelets
Platelets are not cells, but cytoplasmic fragments that develop from specialized cells in the bone marrow called megakaryocytes. Like white blood cells, platelets are drawn to an area of inflammation. Once the platelets arrive at the site of injury, they adhere to the vessel wall, forming aggregates or plugs. Adhering to the vessel wall activates the platelets, causing them to release several biochemical mediators, including serotonin and histamine, which temporarily decrease blood flow and bleeding. This vasoconstriction is short lived, however, and soon blood flow increases to deliver other white blood cells to the area. If the injury is small,
P.127
the platelet plug is usually sufficient to allow for healing. Platelets circulate in the blood for about 10 days before they become non-functioning and are phagocytized by neutrophils and monocytes. If a person has too few platelets, he or she is at increased risk of developing multiple small hemorrhages under the skin and throughout the body.
The Specific and Innate Immune Responses
The immune system includes both specific and innate responses. Specific immune responses are those that involve activation of the B and T lymphocytes. B and T lymphocytes are capable of responding with specificity and precision to virtually any foreign molecule an individual may encounter in a lifetime. Once the original response is made, the B or T cell retains a memory of it. If a second encounter with that molecule occurs, the B- or T-cell response will be faster and more effective than before.
The innate immune response, in contrast, includes the inflammatory responses to infection or injury and the white blood cells that participate in those responses: the neutrophils, the basophils, eosinophils, and monocytes and macrophages. The inflammatory response is stimulated after tissue injury or infection, with the goal of delivering white blood cells and platelets to the tissues to limit damage and promote healing. The inflammatory reactions are not characterized by specificity or memory, but they are fast and effective.
Antigens
An antigen is any molecule that can stimulate a specific immune response against itself or the cell that carries it. Billions of B and T lymphocytes are produced during fetal development with the potential to bind to at least 100 million distinct antigens. Antigens that can bind to a T or B lymphocyte include those present on the cell wall of bacteria or mycoplasmas, the coat of a virus, or on certain pollens, dusts, or foods. Every cell of a person has surface proteins that would be recognized as foreign antigens by B or T lymphocytes from another person. If an antigen causes either the B or T lymphocyte to become activated and to multiply or differentiate further, it is an immunogenic antigen.
B-Lymphocyte Response to an Antigen
When a B lymphocyte encounters its specific antigen, it binds to it in a lock and key fashion, causing the B cell to differentiate into a plasma cell. The plasma cell in turn begins to secrete millions of molecules of antibodies made specifically against that antigen. Once produced, the antibodies, also called immunoglobulins, circulate throughout the bloodstream seeking to eliminate the antigen that stimulated their production. Antibody-mediated responses are important for defense against bacteria and circulating viruses and against toxins released from bacteria.
P.128
Immunoglobulins/Antibodies
There are five specific immunoglobulins produced in response to an antigen: IgG, IgM, IgA, IgE, and IgD.
IgG is the most common immunoglobulin and represents approximately 80% of all circulating antibodies. IgG is the main antibody that crosses the placenta from the mother to the fetus during pregnancy. Levels of IgG increase slowly during the primary (first) exposure to an antigen, but increase immediately and to a much greater extent with a second exposure (Fig. 5-1).
IgM antibody is produced first and in highest concentration during the primary exposure to an antigen. IgM is the largest antibody in size.
IgA antibody is most concentrated in secretions such as saliva, vaginal mucus, breast milk, gastrointestinal (GI) and lung secretions, and semen. IgA acts locally rather than through the systemic circulation. Maternal IgA passes to an infant during breastfeeding (as do IgG and IgM to a lesser extent).
IgE is responsible for allergic reactions. It is also the antibody most stimulated during a parasitic infection.
IgD exists in low concentration in the plasma. Its role in the immune response is not completely clear, although it appears to be important for the maturation and differentiation of all B cells.
Antibody Structure
All antibodies are similar in appearance. They consist of two long heavy chains called the Fc portion, and two small heads called the Fab portion. The Fc portion is identical for all antibodies of a single class (e.g., IgG or IgM). The Fab portion is specific for each antibody and contains the specific binding site for an antigen. Binding of antigen to the Fab portion of the
P.129
antibody activates the Fc portion, leading to destruction of the microorganism or other antigen-bearing cell. An antibody is shown in Figure 5-2.
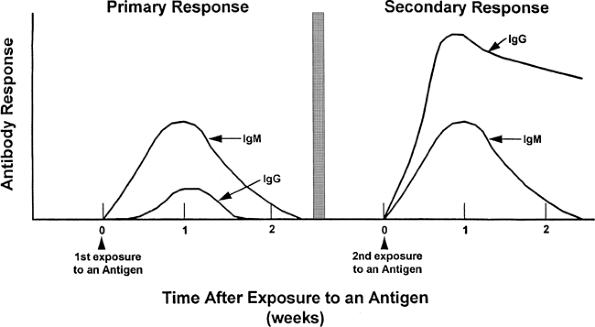 |
Figure 5-1. Primary and secondary antibody responses to an antigen. |
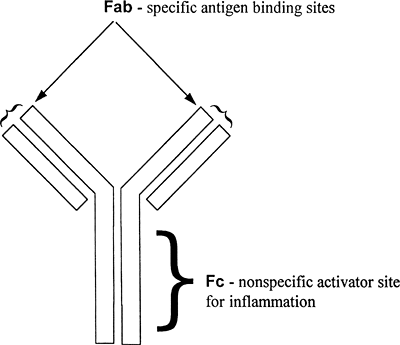 |
Figure 5-2. Diagrammatic representation of an antibody, showing sites for antigen binding and inflammatory activation. |
Antibody Destruction of a Microorganism
Antibodies cause the destruction of bound antigen by a variety of mechanisms. Usually, the antibody does not kill the cell, but instead coordinates the attack by turning on NK cells, activating complement, and enhancing phagocytosis. Under some circumstances, an antibody may directly inactivate an antigen.
NK cell activation occurs when binding of the antigen to the Fab (specific) portion of the antibody allows an NK cell to establish connections with the Fc (nonspecific) portion, thus linking up the NK cell with the antigen (Fig. 5-3). The NK cell then releases toxic chemicals that directly kill the antigen target.
Complement activation will be described in more detail shortly. Complement is a series of molecules that, when activated, leads to the initiation of an inflammatory response and the killing of the antigen-bearing cell. Like NK cell activation, binding of the antigen to the Fab portion of the antibody allows the first molecule in the complement chain (C1) to bind nonspecifically to the Fc portion. Such binding hooks up the antigen-bearing cell with complement, ultimately leading to the destruction of the antigen-bearing cell.
Phagocytic stimulation occurs similarly; when the antigen binds to the Fab portion of the antibody, this allows a phagocytic cell (usually a
P.130
macrophage or neutrophil) to bind to the nonspecific Fc portion, stimulating phagocytosis of the linked antigen and the cell that bears it.
 |
Figure 5-3. Activation of NK cell after binding of antibody to bacterial antigen. Activated NK cell secretes lytic components that lyse the bacterial cell. Similar patterns occur after binding of complement or phagocyte. |
Direct effects of an antibody may occur if, for example, an antibody binds to a virus at the same site the virus uses to bind to and enter a susceptible cell. This would inactivate the virus. Similarly, the antibody may bind to a bacterial toxin at the same site that the toxin would use to interact with susceptible cells. This would eliminate the effect of the toxin.
Opsonization
Binding of an antibody to an antigen on a bacterium causes opsonization, a change in the bacterial cell wall that renders otherwise impenetrable bacteria susceptible to phagocytosis. Complement also serves as an opsonin (an agent that can cause opsonization).
The Role of the T Cell in B-Cell Response to an Antigen
To mount an antibody attack against a microorganism, T cell support is almost always required. As described below, cytokines released by activated T lymphocytes trigger B cell proliferation and differentiation into antibody-secreting plasma cells.
Memory Cells
Some B lymphocytes do not become antibody-secreting plasma cells after antigenic stimulation, but rather become memory cells. Memory cells circulate indefinitely in the blood and become active immediately upon repeat exposure to the antigen.
P.131
The first time a B lymphocyte is exposed to its antigen (the primary exposure), production of antibodies against the antigen can take 2 weeks to more than 1 year, although normally antibodies to an antigen are detectable in the blood within 3 to 6 months. Because of memory cells, the next time the antigen is encountered, the antibody response occurs almost immediately (see Fig. 5-1).
T-Lymphocyte Response to an Antigen
When a T lymphocyte binds to an immunogenic antigen, it is stimulated to mature and reproduce. This reproduction results in up to four subtypes of T cells capable of acting in response to the antigen: cytotoxic T cells, helper T cells, regulatory T cells, and memory T cells. The T-cell response to antigen is called a cell-mediated response, because the T cells respond directly; they do not need to become plasma cells and secrete antibody to destroy the antigen.
Cytotoxic T cells directly destroy the antigen by releasing toxic chemicals. These chemicals punch holes in the membranes of the cells carrying the foreign antigen. Cytotoxic T cells also are called CD8 cells because of a specific protein present on their plasma membrane.
Helper T cells secrete peptides, called cytokines, which act as cell messengers to coordinate the response of cytotoxic T cells and B cells. There are two general categories of helper T (Th) cells: Th1 and Th2 cells. Th1 cells release cytokines that are pro-inflammatory, in that they draw neutrophils and monocytes to the area of injury or infection and stimulate macrophage phagocytosis. Th1 cytokines increase production of prostaglandins, leading to increased blood flow and interstitial edema, and induce systemic symptoms of inflammation, including fever. Th1 cytokines favor the production of cytotoxic T cells and induce cell-mediated immune responses. Th2 cells generally secrete anti-inflammatory cytokines, which put the brakes on potentially dangerous inflammatory reactions. Th2 cells favor activation of humoral (B cell driven) responses. Normally, Th1 and Th2 immune responses are in balance with each other.
Regulatory T cells act to suppress the host's immune response, a function which under some circumstances may increase the risk of infection, but under other circumstances may serve to protect the host against an overzealous immune system. Although their mechanism of action is still under investigation, regulatory T cells appear to suppress immune function via direct contact with B cells or other T cells, and/or by releasing anti-inflammatory cytokines. A deficiency in regulatory T cells has been suggested to play a role in the development of autoimmune disease, while overactive regulatory T cells may protect tumor cells from immune attack. Some evidence suggests that certain viruses, including the human immunodeficiency virus (HIV), exploit regulatory T cells' ability to dampen the body's antiviral response. Regulatory T cells are characterized by C25 receptors on their cell membranes.
P.132
Memory T cells circulate in the bloodstream until the specific antigen that stimulated their production is encountered again. Subsequent responses to that antigen occur rapidly.
![]() eriatric Consideration
eriatric Consideration
As an individual ages, the number and function of immune cells decrease, resulting in an increased prevalence of infection and malignancy in the elderly. Moderate exercise may improve immunocompetence in the elderly by increasing the number of NK and cytotoxic T cells.
Recognition of Self Versus Foreign Antigens
It is essential that the immune system recognizes self-antigens and only initiates attack against non-self or damaged cells. To ensure only appropriate immune responses, potential antigens are always presented to the immune system, and to the T cells in particular, in combination with self-antigens, called major histocompatibility complex (MHC) proteins.
Self-Antigens
Each individual possesses cell surface antigens that are unique to that individual. These antigens, the MHC proteins, serve as a sort of cellular fingerprint. (In humans, these proteins are sometimes called histocompatibility antigens.) There are two groups of MHC proteins: MHC I and MHC II. The MHC I proteins are found on nearly all cells of the body except the red blood cells. The MHC II proteins are found only on the surface of macrophages and B cells. MHC proteins have two functions: (1) they present self-antigens to T cells, and (2) they bind foreign antigens and present these to T cells. The MHC I molecules bind and present antigens only to cytotoxic T cells. The MHC II molecules bind and present antigens only to helper T cells (both Th1 and Th2 types).
The MHC Genes
The MHC proteins are inherited as four closely linked loci (groups of genes) on chromosome 6. These genes, called the major histocompatibility complex, are usually inherited together, with one set of loci received from each parent. There are many different possible alleles for each loci, resulting in more than one trillion possible antigen combinations. Therefore, it is virtually impossible for two unrelated individuals to match MHC proteins. Identical twins will have the same proteins, and an individual's siblings and offspring will likely have MHC proteins that are more similar than those of unrelated individuals.
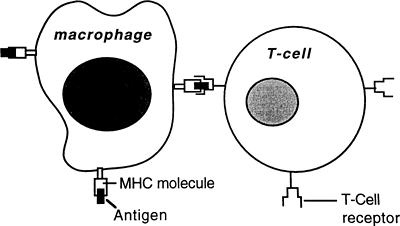 |
Figure 5-4. Antigen presentation to a T cell. The macrophage presents processed antigen along with the MHC self molecule to the T cell, which becomes activated against the antigen. |
P.133
The Role of the MHC Proteins in Controlling Immunity
After a foreign or unknown cell has been phagocytized by a macrophage or has become bound to a B cell, antigens from the cell are expressed on the macrophage or B cell adjacent to host MHC II antigens. The foreign antigens and the MHC II antigens are presented together to passing helper T (CD4) cells. Each passing helper T cell compares the foreign or unknown antigen to the host's MHC II antigens (Fig. 5-4). If in comparing the unknown antigen to the MHC II antigens, a helper T cell recognizes the antigen as foreign, the helper T cell will secrete cytokines that activate the B cell to become an antibody-secreting plasma cell. If the antigen presented is seen by helper T cells as too similar to the MHC II proteins on the B cell or macrophage, the helper T cells will not become activated, or perhaps may become T regulatory cells, and the antigen will not be attacked.
To activate cytotoxic (CD8) cells, MHC I proteins must be presented in association with the unknown or foreign antigen. All cells express MHC I proteins; therefore, any cell can present foreign antigens to CD8 cells for comparison. Cells infected with a virus make abnormal proteins, as do cancerous cells. These abnormal proteins are recognized as antigens and presented to CD8 cells along with host MHC I proteins. When cytotoxic T cells encounter abnormal proteins compared to the MHC I proteins, they are stimulated to initiate killing of the cells.
Graft Rejection and MHC Proteins
A poor match of MHC proteins is the major cause of graft rejection. Not all cellular antigens need to match; however, the closer the MHC profile match between donor and host, the greater the chance a graft will be accepted. Graft rejection is an example of cell-mediated immunity.
P.134
Development of Self-Tolerance
During gestation, hundreds of thousands of T and B cells are formed. Some of these T and B cells fit lock and key with host antigens and are therefore capable of reacting against them. To eliminate the potential of attack against host cells, T cells residing in the thymus and B cells in the bone marrow are exposed during a critical period of embryogenesis to a multitude of host antigens. If, during this time, a B or T cell encounters an antigen to which it matches, the B or T cell is programmed to undergo apoptosis and self-destruct. This leaves behind only cells tolerant to host antigens. This theory of tolerance is called the clonal deletion theory, because it explains the elimination of clones of immune cells that react with self-antigens.
A second method also exists to ensure the elimination of cells with the potential to attack host antigens. This mechanism, called clonal inactivation, occurs outside the thymus during fetal development and throughout life. In this scenario, MHC II antigens are presented to helper T cells. If a helper T cell encounters the specific antigen to which it matches among the MHC proteins, the helper cell undergoes apoptosis.
Because helper T cells are essential in activating B cells to become plasma cells, clonal deletion and clonal inactivation of T cells can eliminate humoral and cellular immunity against self-antigens. In these processes, tolerance is recognized as an active process, essential for the survival of the host. Occasionally, tolerance to host cells may be lost, which leads to the development of an immune response against those cells and may cause autoimmune disease.
Exceptions to Clonal Elimination
Some tissues grow during fetal development without exposure to immature T cells. These tissues normally are kept sequestered from the immune system after birth. If they are later exposed to immune cells, however, an attack against them may occur. Cells that are normally kept sequestered from the immune system include certain cells of the testes and eye.
B- and T-Lymphocyte Responses to a Foreign Antigen
In a B-cell response, B cells bind a foreign antigen. Helper T cells are presented pieces of the antigen, either by the B cell directly or by macrophages that have begun phagocytizing the antigen. If the antigen is different enough from the MHC II proteins expressed by the B cell or macrophage, the helper T cell releases cytokines that activate the B cell, causing it to become an antibody-secreting plasma cell. The antibody will bind the antigen throughout the body and orchestrate its destruction. The T cells also stimulate macrophages to increase phagocytosis of the organism and activate other white blood cells and complement to assist in the defense response.
In a cell-mediated response, cells of any type infected with an intracellular organism or cells that have become cancerous present foreign
P.135
proteins to cytotoxic T cells along with their own MHC I proteins. This activates the cytotoxic cells to destroy the cells carrying the foreign protein.
Red Blood Cell Antigens
There are at least 80 different antigens present on the red blood cells. The most important of these are the ABO antigens and the Rh antigens. Blood types are referred to as ABO and Rh.
ABO Antigens
The ABO blood group consists of A and B antigens. An individual receives from each parent an A antigen gene and a B antigen gene, or neither (called the O antigen). The A and B antigen genes are each dominant over the O antigen gene, but are codominant with each other. Thus, an individual who receives an A antigen gene from one parent and an A or an O from the other (AA or AO) will have type A blood. An individual who receives a B antigen gene from one parent and a B or an O from the other (BB or BO) will have type B blood. An individual who receives an A antigen gene from one parent and a B antigen gene from the other will have AB blood. Type O blood is possible only if an individual receives neither the A nor B antigen gene from either parent (OO).
Rh Antigens
The Rh antigens are a complex group of antigens present on the red blood cell. If a particular type of Rh antigen is present on the red blood cells, an individual is considered to be Rh positive. If this antigen is not present, an individual is considered Rh negative. Each individual receives one Rh gene from each parent. The Rh-positive gene dominates, such that an individual with one Rh-positive gene and one Rh-negative gene is Rh positive. Rh antigens, when mismatched between mother and fetus, can be responsible for a severe reaction in the fetus. This reaction is characterized by red blood cell lysis and anemia (hemolytic disease of the newborn and erythroblastosis fetalis), which occurs when an Rh-negative mother produces antibodies against the red cells of an Rh-positive fetus. Antibodies may cross the placenta and cause destruction of fetal red blood cells before or during birth.
Immune Reactions Against Mismatched Blood
An individual with type A blood given a transfusion of type B blood may develop a severe immune reaction against the B blood, called a transfusion reaction. In a transfusion reaction, lysis and agglutination of the donated red blood cells occur. Inflammation, blood clotting, and death may result. A similar reaction would occur if an individual with type B blood were to receive a donation of type A blood. An individual with Rh-negative blood who receives an Rh-positive blood transfusion may also have an immunologic reaction, although typically the response is less intense.
P.136
Individuals with type A or type B blood can safely receive type O blood because type O blood will not stimulate an antibody reaction. Individuals with O-negative blood are called universal donors because they can supply blood to anyone. Individuals with AB-positive blood are universal recipients because they will not react against any type blood. O-negative is the blood of choice for an emergency trauma patient who has not been cross-matched (blood type determined).
Immunity
Immunity is the state in which one is protected from disease development. Immunity may be innate, passive, or acquired after exposure to a microorganism or toxin.
Innate Immunity
Innate immunity is the immunity that exists due to an organism's natural resistance. Innate immunity includes barriers to infection provided by the skin, gastrointestinal juices, and tears, as well as the protection provided by the nonspecific mediators of inflammation discussed previously.
Passive Immunity
Passive immunity refers to immunity provided to an individual by transfer of antibodies from someone else or by delivery of a prepared antitoxin. Antitoxins are antibodies produced specifically against a certain bacterial toxin (e.g., diphtheria antitoxin). An example of passive immunity is when antibodies produced in one individual against the hepatitis B virus are harvested and given to another individual who has been exposed to the virus, but whose cells have not yet been infected by it. Clinically, the transferred antibodies are referred to as hepatitis B immunoglobulin. Likewise, an individual bitten by a rattlesnake may be given antivenom. The antivenom consists of antibodies produced by a person who was bitten by a rattlesnake and survived the bite. The hope in this situation is that the passively provided antibodies will take out of circulation the poisonous venom before the person is killed or seriously affected. Passive immunity also occurs when maternal IgG antibodies cross the placenta and when IgA and other antibodies are provided to the infant in breast milk.
Passive immunity works by providing a person with preformed, specific antibodies when that person is incapable of forming antibodies (a fetus or infant) or when there is insufficient time for antibodies to be produced before infection or death occurs. Passive immunity is temporary and does not provide a memory response.
Active Immunity
Active immunity is the cellular and humoral immune response developed by an individual who has been significantly exposed to a microorganism or
P.137
toxin. Exposure may occur in a disease process or as a result of an immunization. Active immunity is characterized by memory in the B and T lymphocytes and the production of specific antibodies and T cells. An antibody titer (level) may be measured in a serum sample to document the development of immunity to a microorganism or toxin. A positive titer (except in an infant) implies active immunity.
![]() ediatric Consideration
ediatric Consideration
Vaccines provide protection to the immunized individual and to other members of the community who have not been immunized (herd immunity). Childhood immunization is by far the most cost-effective public health program available today. Vaccines have significantly reduced the incidence of many illnesses previously common in childhood, including measles, rubella, chicken pox, and polio. Pediatric hepatitis B immunization has reduced the incidence of liver cancer worldwide.
Immune Status of the Fetus and Newborn
Cell-mediated (T-cell) immunity begins in utero. A primary humoral immune response (IgM) to various microorganisms can be stimulated in the fetus in the last trimester of gestation. Other immune responses to an antigen (IgG and IgA), neutrophil and macrophage phagocytosis, and the production of inflammatory mediators, however, are not significantly present until 6 to 8 months after birth. This makes the fetus and newborn vulnerable to infection and disease. In utero, maternal IgG antibodies are actively transported across the cells of the placenta and can be detected in the newborn for at least 6 months after birth. These antibodies offer the fetus and infant passive immunity against various microorganisms. IgA and other immunoglobulins may pass to the newborn through breast milk.
A time of particular vulnerability for a newborn is at approximately 5 to 6 months after birth when maternal IgG levels are being cleared, yet the infant's own immune system is not yet working at its peak. This vulnerability is especially true if the infant is not breastfeeding.
![]() ediatric Consideration
ediatric Consideration
Before maternal antibodies are cleared from the infant's bloodstream, it is impossible to tell whether an infant showing IgG antibodies against a specific microorganism is reflecting maternal infection or whether the infant is him or herself actively infected by the microorganism. Maternal antibodies begin to decrease after 6 months; therefore, the infant's antibody titer (level) after 6 months should be measured to identify true infection versus passive immunity. This is important to keep in mind when identifying which infants of mothers infected with HIV are infected and which only carry maternal antibodies to the virus. Infants who are infected with the virus may benefit from drug
P.138
therapies that would be unnecessary and possibly dangerous to uninfected infants. Measuring the presence of HIV virus or viral antigens in an infant, rather than antibodies against HIV, allows for earlier diagnosis of infant HIV status.
The Inflammatory Response
The inflammatory response occurs after tissue injury or infection. Inflammation may precede a specific immune response or be initiated by one. There are two stages in an acute inflammatory reaction: vascular and cellular (see page C3 for illustrations).
Vascular Stage of Inflammation
The vascular stage of inflammation begins almost immediately after an injury or in response to infection or toxin exposure. Arterioles at or near the site briefly constrict and then undergo a prolonged vasodilation (relaxation). The brief constriction pulls the endothelial cells lining the arterioles apart, allowing for passage of early arriving white blood cells. The subsequent vasodilation occurs primarily as a result of mast cell degranulation and the release of chemical mediators (described later). Dilation of the arterioles causes increased fluid pressure in the downstream capillaries. At the same time, histamine and other chemicals cause an increase in permeability of surrounding capillaries. Increased permeability combined with increased blood flow leads to increased movement of a plasma filtrate into the interstitial space. The result is swelling and edema of the interstitial space and increased viscosity of the blood left behind in the capillary. Occasionally, red blood cells may move into the area surrounding injured cells.
Cellular Stage of Inflammation
The cellular stage of inflammation begins with the movement of white cells in the blood to the area of injury or infection. These cells and platelets are drawn to the area by chemicals released from injured cells, from mast cells, by complement activation, and by cytokine production that occurs after antibody-antigen binding. Attraction of white blood cells to an area of injury is called chemotaxis. Once at the site of injury, the various stimulants cause the capillary endothelial cells and the white blood cells, especially neutrophils and later monocytes, to express complementary adhesion molecules. The endothelial cells become sticky for the white blood cells, causing the white blood cells to move to the periphery of the capillary, in a process called margination. This leads to the emigration of the white blood cells through the capillaries to surround and phagocytize the damaged cells. Platelets entering the area stimulate clotting to isolate the infection and control bleeding. Cells brought to the site will eventually be responsible for healing the injured area.
P.139
The Mast Cell
Mast cells are specialized cells filled with vasoactive chemicals. These cells are found throughout the loose connective tissue surrounding the blood vessels, especially in the lungs, gastrointestinal tract, and skin. Inflammation begins when mast cells release their intracellular contents during tissue injury, exposure to toxins, activation of the proteins of the complement cascade, and antibody-antigen binding. Releasing the contents of the mast cell is called mast cell degranulation. Histamine, serotonin, and other substances are released with mast cell degranulation and contribute to vasodilation, increased capillary permeability, and the drawing of white blood cells and platelets to the area.
Chemical Mediators of Inflammation
The main chemical mediator of inflammation is histamine, which is released by basophils, platelets, and mast cells. Histamine participates in both steps of the vascular response to inflammation: relaxation of blood vessels leading to increased blood flow and increased capillary permeability. Histamine is also active in nonvascular tissues. In the respiratory passages, histamine causes constriction of bronchiolar smooth muscles. In the gut, histamine stimulates acid secretion. Histamine also causes itching. Histamine binds to H1 receptors in the respiratory passages and the vascular system, and to H2 receptors in the gut. Binding to the H1 receptors causes a decrease in further release of histamine from the mast cells, an example of a negative feedback response.
Neutrophil and eosinophil chemotactic factors are chemicals released from white blood cells (neutrophils or eosinophils) that draw other cells to the area.
Prostaglandins, especially of the E series, are important mediators of inflammation. Prostaglandins are produced when a cell membrane is damaged, or a cell bursts, and arachidonic acid, a main constituent of the cell membrane, is metabolized by the cycloxygenase (COX) enzymes I and II. Prostaglandins (PGE1 and PGE2) increase blood flow and increase capillary permeability. They also augment the effects of histamine, cause fever in response to infection, and stimulate pain receptors. Other substances produced from arachidonic acid metabolism include vasoconstrictive prostaglandins and thromboxane A2, a promoter of platelet aggregation and vasoconstriction. Prostaglandin synthesis is inhibited by non-steroidal anti-inflammatory drugs (NSAIDs), such as aspirin and ibuprofen, which act by blocking the COX I and COX II enzymes.
Leukotrienes also are end-products of arachidonic acid metabolism. These substances increase vascular permeability and increase the adhesion of white blood cells to the capillary during injury or infection. They also act as chemoattractant chemicals. One type of leukotriene, called slow-reacting substance of anaphylaxis, plays an important role in the bronchiolar constriction of asthma and in allergic reactions. Leukotriene production is not blocked by COX enzyme inhibitors.
P.140
Cytokines are a family of peptides produced by a variety of immune and inflammatory cells, including macrophages, monocytes, neutrophils, and B and T lymphocytes. They are also produced by non-inflammatory cells, including fibroblasts and endothelial cells. Cytokines are often referred to by specific names related to their function or numbered following the general term interleukin. Cytokines function as local hormones that affect the host defense response to injury or infection. Each cytokine may have multiple effects on a variety of interrelated processes. In general, cytokines serve as communication links between different arms of the immune and inflammatory responses. Cytokines are discussed further in Chapter 7.
There are two general categories of cytokines: pro-inflammatory and anti-inflammatory. Pro-inflammatory cytokines (e.g., interleukin-1, interleukin-2, interleukin-6, tumor necrosis factor, and interferon-gamma) promote the inflammatory responses and cause fever and malaise. These pro-inflammatory cytokines are released from macrophages and monocytes (interleukin-1) or activated Th1-type lymphocytes (interleukin-2, interleukin-6, tumor necrosis factor, and interferon-gamma). Interleukin-2 and tumor necrosis factor stimulate cytotoxic T cells to attack and kill cancer cells or cells infected with a virus. They also alert macrophages to increase phagocytosis. A variety of other cytokines are important for stimulating the bone marrow to increase white and red blood cell production (hematopoietic colony-stimulating factors). These cytokines cause the increase in white blood cells that typically accompanies infection. Anti-inflammatory cytokines (e.g., interleukin-4 and interleukin-10) decrease the secretion of the pro-inflammatory cytokines and stimulate activation of B cells.
Interferons are a type of cytokine specific for preventing intracellular infection by viruses or parasitic organisms. They are produced by T cells (interferon-gamma) or other white blood cells (interferon-alpha) or fibroblasts (interferon-beta) and function to alert neighboring cells to secrete chemicals that will prevent them from becoming infected. The interferons have been used clinically to stimulate the immune system of patients with cancer and other diseases.
Chemokines are a type of cytokine that act as chemotactic agents to regulate leukocyte movement. They may act to attract all types of white blood cells, or may be specific for certain white blood cells. Chemokines interact with target white cells by binding to receptors on the cell membrane. They work by a variety of mechanisms, one of which is to cause the cell to express adhesion molecules complementary to those expressed by capillary endothelial cells. These adhesion molecules make white blood cells sticky for the capillary, resulting in migration and emigration of the cells.
Complement System
The complement system consists of 20 or more plasma proteins that are activated one by one in a domino fashion when the first protein (C1, the
P.141
classical pathway) or the third protein (C3, the alternative pathway) is activated. The C1 protein is activated when the Fc portion of an IgG or IgM antibody is turned on after antigen binding to the Fab portion. The C3 protein is usually activated by pieces of bacterial or fungal cell wall released during phagocytosis. Activation of complement is an effective mechanism for destroying extracellular microbes.
The first five complement proteins (C1 C5) stimulate mast cell degranulation, white blood cell chemotaxis, and opsonization of bacteria. Activation of complement proteins 6 through 10 (C6 C10) causes bacterial cell lysis by making the cell wall leaky to water.
Coagulation Pathway
The coagulation pathway involves another series of at least 13 proteins that activate in a step-by-step cascade. The coagulation cascade functions like the platelets (but more powerfully) to stop bleeding. The result of the coagulation cascade is the formation of an insoluble clot and a meshwork of fibers that entrap microorganisms and prevent the spread of infection.
The coagulation cascade can be stimulated by many substances present with inflammation or injury. The intrinsic pathway is activated when one of the plasma proteins, factor XII (the Hageman factor), comes into contact with an injured blood vessel. The extrinsic pathway is stimulated when a different plasma protein, factor VII, comes into contact with a substance called tissue thromboplastin, which is released by injured cells. Both pathways result in the formation of a fibrin clot. The coagulation pathway requires calcium ion for most steps. Coagulation is kept in check by a series of natural anticoagulants that act with naturally occurring heparin (from platelets) to stop the coagulation cascade and prevent uncontrolled thrombus formation.
Pathophysiologic Concepts
Local Signs of Inflammation
Local characteristics of inflammation include the following:
Rubor, the redness that accompanies inflammation, results from increased blood flow to the inflamed area.
Calor, the heat that accompanies inflammation, results from increased blood flow.
Turgor, the swelling of an inflamed tissue, results from increased capillary permeability, which allows plasma proteins and exudate to enter the interstitial space.
Dolor, the pain of inflammation, results from stretching of nerves caused by swelling and the stimulation of nerve endings by mediators of inflammation.
P.142
Fever
Fever is the elevation of the temperature set-point in the hypothalamus. With an increase in set-point, the hypothalamus sends out signals to increase body temperature. The body responds by shivering and increasing the basal metabolic rate.
Fever occurs in response to production of certain cytokines, including interleukin-1, interleukin-6, and tumor necrosis factor. These cytokines are considered to be endogenous pyrogens (heat producers). The pyrogenic cytokines are released by several different cells, including monocytes, macrophages, T-helper cells, and fibroblasts, in response to tissue infection or injury. The endogenous pyrogens cause fever by producing a prostaglandin, probably PGE, that raises the hypothalamic thermoregulatory set-point. When the source of the pyrogen is removed (e.g., after a successful response of the immune system against a microorganism), its level decreases, which returns the set-point to normal. For a short time, body temperature will lag behind the return of the set-point and the hypothalamus will perceive the body temperature as too high. In response, the hypothalamus will stimulate responses such as sweating to cool the body. Aspirin and other non-steroidal anti-inflammatory drugs inhibit fever by blocking prostaglandin synthesis.
Although the function of endogenous pyrogens has been recognized for more than 30 years, it is still uncertain exactly how they transmit the message of infection from the periphery to the central nervous system (CNS). The primary hypothesis proposed to explain this occurrence is that locally produced interleukin-1 stimulates firing of the vagus nerve, which then transmits the information to the CNS. Once the signal reaches the CNS, brain prostaglandin is produced and raises the hypothalamic thermoregulatory set-point. An alternative hypothesis is that the interleukins may themselves cross the blood-brain barrier, and directly stimulate hypothalamic PGE production.
Fever has been shown to occur in every observed animal, suggesting an evolutionary role in species survival. Research suggests fever helps an organism fight infection and thus is beneficial to the host. However, high fevers may damage cells, especially those of the CNS.
Leukocytosis
Leukocytosis is an increase in circulating white blood cells (leukocytes). An increase in neutrophils is responsible for the initial leukocytosis that accompanies an infection or inflammation. With infection, the number of immature cells (myeloid cells) in the blood increases as the mature neutrophils and other mature granulocytes are used up. This shift toward immature cells is called a left shift. With resolution of inflammation or infection, a right shift occurs as mature cells are released from the marrow and again dominate in the circulation.
P.143
Chronic Inflammation
Chronic inflammation is an inflammatory reaction lasting longer than 2 weeks. Chronic inflammation may follow acute inflammation, for example, an unresolved infection or a poorly healed wound. Chronic inflammation may also occur without a preceding acute inflammation, for example, if the body encounters a microorganism it cannot kill, it often encloses the microorganism within a wall to isolate it. Examples of microorganisms that may lead to chronic inflammation include the mycobacteria responsible for tuberculosis and leprosy. These bacteria survive in macrophages, which group together to form a protective capsule of cells called a granuloma.
Hypersensitivity Reactions
Hypersensitivity reactions are abnormal immune and inflammatory responses. There are four types of hypersensitivity reactions.
Type I Hypersensitivity Reactions
Type I hypersensitivity reactions are allergic reactions mediated by the IgE antibody. In type I reactions, an antigen called an allergen to which the individual is sensitive is recognized by a B cell, which is then stimulated to make IgE antibodies against the antigen. IgE binds the antigen as well as a nearby basophil or mast cell via a high-affinity IgE receptor present on those cells. The inciting allergen is typically multi-valent (has many IgE binding sites), so the allergen actually links several IgE antibodies together. This linking triggers a cascade of signals that cause degranulation of the mast cells and basophils, and the release of histamine, cytokines, chemokines, and leukotrienes. These mediators, as well as activated complement and eosinophil chemotactic factor, cause peripheral vasodilation and increased capillary permeability, leading to localized swelling and edema. Symptoms are specific according to where the allergic response occurs. Binding of the antigen in the nasal passages causes allergic rhinitis with nasal congestion and inflammation of the tissues, while binding of an antigen in the gut may cause diarrhea or vomiting.
A severe type I hypersensitivity reaction is termed an anaphylactic reaction. Anaphylaxis involves a rapid IgE mast cell response after exposure to an antigen to which the individual is highly sensitive. Histamine-induced dilation of the entire systemic vasculature can occur, leading to collapse of the blood pressure. A severe decrease in systemic blood pressure during an anaphylactic reaction is called anaphylactic shock. Because histamine is a potent constrictor of bronchiolar smooth muscle, anaphylaxis involves closure of the respiratory passages. Anaphylaxis in response to some drugs such as penicillin, or in response to a bee sting, may be fatal in highly sensitized individuals, as a result of circulatory collapse or respiratory failure. Symptoms of an anaphylactic reaction include itching, abdominal cramps, flushing of the skin, gastrointestinal (GI) upset, and breathing difficulties.
P.144
Type II Hypersensitivity Reactions
Type II hypersensitivity reactions occur when IgG or IgM antibodies attack tissue antigens. Type II reactions result from a loss of self-tolerance and are considered autoimmune reactions. The target cell is usually destroyed.
In a type II reaction, antibody-antigen binding causes complement activation, mast cell degranulation, interstitial edema, tissue destruction, and cell lysis. Type II hypersensitivity reactions lead to macrophage phagocytosis of the host cells. Examples of type II autoimmune diseases include Graves' disease, which involves antibodies produced against the thyroid gland; autoimmune hemolytic anemia, which involves antibodies produced against red blood cells; transfusion reactions, which involve antibodies produced against donor blood cells; and autoimmune thrombocytopenic purpura, which involves antibodies produced against platelets. Systemic lupus erythematosus (SLE) also has aspects of type II reactions (described later).
Type III Hypersensitivity Reactions
Type III hypersensitivity reactions occur when circulating antibody-antigen complexes precipitate out in a blood vessel or in downstream tissue. Antibodies are not directed against those particular tissue sites, but are trapped in their capillary meshwork. In some cases, foreign antigens may adhere to tissues, causing formation of antibody-antigen complexes at those sites.
Type III reactions activate complement and mast cell degranulation, causing damage to the tissue or capillaries where they occur. Neutrophils are drawn to the area and begin to phagocytize the injured cells, causing release of cellular enzymes and the accumulation of cell debris. This continues the inflammation cycle.
Examples of type III hypersensitivity reactions include serum sickness, in which antibodies form against foreign blood, often in response to intravenous drug use. The antibody-antigen complexes deposit in the vascular system, joints, and kidneys. With glomerulonephritis, antibody-antigen complexes form in response to an infection, often by streptococcal bacteria, and deposit in the glomerular capillaries of the kidneys. With systemic lupus erythematosus, antibody-antigen complexes form against collagen and cellular DNA, and deposit in multiple sites of the body.
Type IV Hypersensitivity Reactions
Type IV hypersensitivity reactions are T cell-mediated reactions, in that cytotoxic (CD8) or helper (CD4) T cells are activated by an antigen, leading to destruction of the cells involved. Cytotoxic cell-mediated reactions are often against virally infected cells and can lead to extensive tissue damage. CD4 cell-mediated reactions are delayed, taking 24 to 72 hours to develop. They are characterized by the production of pro-inflammatory cytokines that stimulate macrophage phagocytosis and increase swelling and edema.
P.145
Examples of conditions caused by type IV reactions include autoimmune thyroiditis (Hashimoto's disease), in which T cells are produced against thyroid tissue, graft and tumor rejection, and delayed allergic reactions, such as the reaction to poison ivy. The tuberculin skin test indicates the presence of delayed cell-mediated immunity against the tuberculin bacillus.
Immune and Inflammatory Deficiencies
Immune and inflammatory deficiencies inhibit the body's ability to respond to infection or injury, and may result from impaired function of any or all white blood cells. Complement or coagulation proteins may also be deficient. Immune and inflammatory deficiencies may be congenital (present at birth) or acquired after illness, infection, or prolonged stress. The deficiencies may be temporary or permanent.
Congenital Immunodeficiency
Congenital immunodeficiency occurs as a result of a genetic defect. Congenital immunodeficiency may involve one type of T or B cell, all the T cells (DiGeorge syndrome), or all the B cells (Bruton's agammaglobulinemia). Most commonly, one immunoglobulin (usually IgA or IgG) is missing. Individuals with selective immunoglobulin deficiency may have an increased susceptibility to certain infections or may be asymptomatic. Severe cases of IgG deficiency may be treated with replacement injections. Typically, selective IgA deficiency is not treated because patients may develop IgG antibodies to administered IgA, which may cause anaphylaxis. With total B-cell deficiency, the missing immunoglobulins can be provided to the individual by intravenous administration. Infants with primary T-cell deficiency have severely impaired ability to fight infection because T cells are required not only for cellular immunity but humoral immune responses as well. If the pluripotential bone marrow stem cells are dysfunctional, T and B cells and all other white blood cells may be deficient. This condition is called severe combined immunodeficiency syndrome (SCIDS). SCIDS used to be fatal in early childhood, but treatment with harvested stem cells is yielding promising results.
Congenital immunodeficiency may also occur if an individual is born without certain MHC proteins. Without these proteins, dysfunctional self-antigen presentation to the T cells occurs, leading to a failure of T cell immune function. This condition usually causes death in early childhood.
Acquired Immunodeficiency
Acquired immunodeficiency is reduced functioning of the immune system developing after birth. Acquired immunodeficiency may arise in response to infection, malnutrition, chronic stress, or pregnancy. Systemic illnesses such as diabetes, renal failure, and cirrhosis of the liver can cause immunodeficiency. Individuals receiving corticosteroids to prevent transplant
P.146
rejection or to reduce chronic inflammation are immunosuppressed, as are those receiving chemotherapeutic drugs and radiation therapy. Surgery and anesthesia may also depress the immune system.
Acquired immunodeficiencies can be of B or T cell function, or both. Because B cells require helper T cell stimulation to successfully fight infection, T cell deficiencies also cause dysfunction of the humoral immune system.
![]() eriatric Consideration
eriatric Consideration
The elderly are often immunodeficient, in part because of the progressive decrease in function of the thymus with aging, but also because of poor blood flow and reduced delivery of the mediators of immunity and inflammation that many elderly experience as a result of atherosclerosis. Other systemic diseases such as diabetes mellitus, which increases in incidence with age, contribute to a depressed immune response as well. Poor nutrition, caused by poverty, isolation, or bad dentition, contributes to poor immune function in the elderly.
Consequences of Immunodeficiency
Immunodeficient individuals repeatedly develop frequent severe and unusual infections and are often unable to fight them. Individuals with T-cell deficiencies frequently develop viral and yeast infections; individuals with B-cell deficiencies are especially susceptible to infections by bacteria that normally require opsonization. HIV destroys the helper T (CD4) cells and infects other white blood cells.
Conditions of Disease or Injury
Most diseases associated with alterations in immune function are presented elsewhere in this book. Only allergy, systemic lupus erythematosus (SLE), and HIV/AIDS are discussed in this chapter.
Allergy
An allergy is an overstimulation of inflammatory reactions that occurs in response to a specific environmental antigen. An antigen that causes an allergy is called an allergen. Allergic reactions may be antibody mediated or T cell mediated. Type I hypersensitivity reactions are examples of antibody-mediated allergies, whereas Type IV hypersensitivity reactions are examples of T cell mediated allergies.
An individual with a Type I hypersensitivity allergic response has developed sensitized IgE antibodies to an allergen. When the allergen is encountered by the antibody, the antibody is overexpressed, causing excessive mast
P.147
cell degranulation and release of histamine and other inflammatory mediators (leukotrienes, chemokines, and cytokines). Type IV hypersensitivity reactions occur after transdermal (across the skin) transport of an allergen that is presented to T cells sensitized to that allergen. Manifestations of an allergic response depend on where the allergen is encountered whether in food, in inhaled particles, or through the skin. The timing of an allergic reaction varies depending on if the response is type I (immediate) or type IV (delayed). A type I reaction involving the skin is called atopic dermatitis; a type IV reaction is called allergic contact dermatitis. The skin response to poison ivy is a type of allergic contact dermatitis.
Cause of Allergies
The cause of allergies is unclear, although there appears to be a genetic predisposition. A predisposition may involve excessive IgE binding, easily provoked mast cell degranulation, or excessive helper T cell response. Recent work suggests that a deficiency in T regulatory cells may contribute to over-responsiveness of the immune system and allergy. Overexposure to certain allergens at any time, including during gestation, may cause an allergic response.
![]() ediatric Consideration
ediatric Consideration
Infants and children exposed to cigarette smoke are at greater risk of developing asthma and other respiratory allergies.
Clinical Manifestations
Localized swelling, itching, and redness of the skin, with skin exposure to an allergen. Type IV reactions are often characterized by blistering and crusting over of the affected area.
Diarrhea and abdominal cramps, with exposure to a gastrointestinal allergen.
Allergic rhinitis, characterized by itchy eyes and runny nose, with exposure to a respiratory allergen. Swelling and congestion occur. Breathing difficulties may occur because of histamine-mediated constriction of the bronchiolar smooth muscle of the airways.
Diagnostic Tools
Skin tests help in diagnosing an allergy. A small amount of the suspected allergen is injected under the skin. Individuals allergic to that allergen will respond with marked erythema, swelling, and itching at the injection site.
Serum immunoglobulin analysis may indicate increased basophil and eosinophil count.
P.148
Complications
A severe allergic reaction may result in anaphylaxis, which is characterized by a decrease in blood pressure and closure of the airways. Itching, cramping, and diarrhea may occur. Without intervention, severe reactions can lead to cardiovascular shock, hypoxia, and death.
Allergic contact dermatitis (e.g., with a poison ivy reaction) may lead to secondary infection from excessive scratching.
Treatment
Antihistamines and drugs that block mast cell degranulation may reduce the allergy symptoms.
Corticosteroids, inhaled, administered nasally, or taken systemically, act as anti-inflammatory agents and can reduce the symptoms of an allergy. Inhaled or intranasal therapy needs to be used for extended periods of time before becoming effective. Inhaled corticosteroids exert their effects only on the respiratory passages and may have few systemic effects.
Inhaled mast cell stabilizers reduce mast cell degranulation and may reduce type I allergic symptoms.
Desensitization therapy, involving repeated injections of small amounts of an allergen to which an individual is sensitive, may cause the individual to build IgG antibodies, called blocking antibodies, against the allergen. These blocking antibodies also bind the allergen and by doing so interfere with the ability of the allergen to covalently link multiple IgE molecules together; this prevents mast cell degranulation so allergic symptoms are reduced. IgG antibodies are produced each time the allergen is encountered and eventually may stop the allergic response.
Systemic Lupus Erythematosus
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease in which antibodies against several different self-antigens are produced. The antibodies are usually IgG or IgM and may be produced against DNA and RNA, proteins of the coagulation cascade, skin, red blood cells, white blood cells, and platelets. Antibody-antigen complexes can precipitate in the capillary networks, causing type III hypersensitivity reactions. Chronic inflammation can follow.
Causes of SLE
The cause of SLE is unknown, although it often occurs in individuals with a genetic tendency for autoimmune disease. Additional evidence to support a genetic role is the high concurrence among identical twins, and an increased incidence in blacks compared to whites. The tendency to develop SLE may be related to alterations of specific MHC genes and how self-antigens are expressed and recognized. Women are more likely to
P.149
develop SLE than men, suggesting a role for the sex hormones. SLE can be brought on by stress, often related to pregnancy or childbearing. In some individuals, excessive exposure to ultraviolet radiation may initiate the disease. Typically affected are young women during their childbearing years. The disease may remain mild for years, or may progress and lead to death.
Clinical Manifestations
Polyarthralgia (joint pain) and arthritis (inflammation of the joint). Fever from chronic inflammation.
Facial rash, in a malar (butterfly) pattern across the nose and cheeks. The word lupus means wolf and refers to the wolf-like appearance of the rash.
Fingertip lesions and bluing caused by poor blood flow and chronic hypoxia. Sclerosis (tightening or hardening) of the skin of the fingers.
Sores on the oral or pharyngeal mucous membranes.
Scaling lesions on the head, neck, and back.
Eye and feet edema may signify renal involvement and hypertension.
Anemia, chronic fatigue, frequent infections, and bleeding are common because of the attack against the red and white blood cells and the platelets.
Diagnostic Tools
Antinuclear antibodies are present in at least 95% of individuals with SLE, but may occur in those without the disease.
Antibodies against double-stranded DNA are diagnostic for SLE.
Protein in the urine signals renal damage.
Antineuronal antibodies may be present.
Complications
Renal failure is the most common cause of death in individuals with SLE. Renal failure may develop as a result of the deposit of antibody-antigen complexes in the glomeruli with resultant complement activation leading to cellular injury, an example of a type III hypersensitivity reaction.
Pericarditis (inflammation of the sac surrounding the heart) may develop.
Inflammation of the pleural membrane surrounding the lungs can restrict respirations. Bronchitis is common.
Vasculitis of all peripheral and cerebral vessels may occur.
CNS complications include stroke and seizure. Alterations in personality, including psychosis and depression, may develop. Personality changes may be related to drug therapy or the disease.
Treatment
Anti-inflammatory drugs including aspirin or other NSAIDs are used to treat fever and arthritis.
Systemic corticosteroids are used to treat symptoms and reduce renal and CNS pathology.
Anti-inflammatory drugs, such as methotrexate, and cytotoxic drugs (azathioprine) are used if steroids are ineffective or symptoms severe.
Anti-malarial drugs are used to treat skin rashes, arthritis, and other symptoms.
P.150
Acquired Immunodeficiency Syndrome
Acquired immunodeficiency syndrome (AIDS) is a viral disease that causes collapse of the immune system. Until recently, once diagnosed with AIDS, an individual had a high probability of dying within 10 years. However, advances in treatment of patients with AIDS have made long-term survival possible. Since AIDS was first described in the early 1980s, much has been learned about how this and other viruses work and about the important roles of all white blood cells in host defense.
AIDS is caused by infection with the human immunodeficiency virus (HIV). There are at least two HIV viruses, HIV-1 and HIV-2. HIV-1 is common in the United States, whereas HIV-2 is found primarily in West Africa. HIV-1, first identified in the early 1980s, is a retrovirus, meaning that it carries in its core two single strands of RNA that, with infection of a host cell, are delivered to the nucleus and transcribed into the host DNA. Transcription of the virus into host DNA occurs by the actions of an enzyme called reverse transcriptase that also is carried as part of the virus into the host nucleus. Once integrated into host DNA, the virus replicates and mutates over the course of many years, slowly but steadily killing off cells of the immune system.
Cell Infection and Death
HIV only infects cells that carry certain membrane receptors to which it can bind, one of which is the CD4 antigen. On the virus is a complementary surface antigen, called group 120 antigen, which fits lock and key with the CD4 receptor. Cells that carry the CD4 antigen, and so can be infected by HIV, include macrophages, the skin immune cells called Langerhans' cells, astrocytes of the central nervous system, and the helper T cells, already identified previously as CD4 cells. Most of the macrophages and helper T cells concentrate in the lymph nodes, spleen, and bone marrow, acting as a huge reservoir of virus-containing cells that continually pass the virus on to healthy cells traveling through those sites. Because of the high density of infected cells in secondary lymph organs, the number of circulating cells infected by the virus grossly underestimates the true number of infected cells. This means that even if the virus is undetectable in the blood, it is likely to still exist in non-circulating cells.
As previously mentioned, in order to infect a cell, it is required that the virus binds the CD4 antigen; however, this requirement is not in itself sufficient for infection to occur. Besides binding the CD4 receptor, HIV must
P.151
also bind a second surface receptor before it can enter a host cell. Chemokine receptors on macrophages and helper T cells have been identified that provide the necessary second binding sites for HIV. On macrophages, this receptor is the chemokine receptor identified as CCR5; on the helper T cells, the second surface receptor is the chemokine receptor identified as CXCR4. A key point to understand is that naturally occurring HIV readily binds CCR5 receptors and so quickly infects macrophages, but ineffectively binds CXCR4 receptors. This means that at first the virus infects primarily macrophages. Once inside, HIV does not destroy the macrophage, but can survive inside the cell for years, replicating constantly, and mutating frequently. Eventually a mutated strain develops that is equally capable of binding the CXCR4 receptor, and so the virus then can infect helper T cells as well as macrophages. With this shift, the virus soon becomes deadly, because HIV kills the helper T cells it infects, eventually causing levels of helper T cells to fall to fewer than 200/ L of blood (normal levels are approximately 1,000/ L). When helper T cells fall to this level, the development of opportunistic infections and other AIDS-defining illnesses is inevitable.
HIV destroys the helper T cells when it takes over the cell's genetic machinery and begins to make new viral components, using a second enzyme protease carried by the virus into the host cell. As the virus reproduces, it destroys the host cell membrane, perhaps by interfering with the cell's ability to protect itself from free radicals or by producing a superantigen that destroys the cell. Once HIV reproduces and kills the helper T cell, many more viruses are released into the circulation. These go on to infect other cells. Contributing to the death of the helper T cells is the immune response the host killer cells mount in an attempt to eliminate the virus and infected cells. As the number of helper T cells decreases, the cell-mediated immune system becomes progressively weaker. B cell and macrophage function also fail as T cells are lost. Loss of immune function allows microorganisms that would normally be kept in check to proliferate wildly, leading to disease and death from a variety of infections. Without immune surveillance, cancers develop as well, contributing to the high death rates seen in individuals infected with AIDS.
The Course of HIV Infection
An individual infected with HIV may remain asymptomatic for 8 or more years during the time the infection is mostly restricted to the macrophages. Once the virus begins destroying helper T cells, it will progress rapidly, usually over the course of 2 to 5 years, if untreated. An individual is diagnosed as having AIDS when the T cell count decreases to fewer than 200 cells/ L, or when an opportunistic infection, cancer, or AIDS dementia develops.
It should be emphasized that HIV infection is not AIDS, and occasionally an individual infected by the virus will survive more than 12 years with no signs of AIDS developing even without treatment. However, infection
P.152
with the virus means the individual is contagious to others, whether symptoms of AIDS are present or not.
AIDS-Resistant Genes
Reports of HIV resistance have been described in the literature for many years. Approximately 10 to 20% of individuals repeatedly exposed to HIV will not become infected with the virus, and some people who become infected experience an atypically long time period without symptoms. Recently it has been shown that some of the resistance to HIV results from mutations present in certain genes, including the gene coding for the CCR5 receptor. Specifically, it has been demonstrated that approximately 10 to 14% of Caucasians carry one mutant CCR5 gene and approximately 1% of Caucasians carry two mutant genes (one from each parent). If an individual is homozygous for a mutation in the CCR5 gene, he or she is usually resistant to HIV infection. If an individual is heterozygous for the mutant gene, he or she may become infected but will show a delay in the onset of overt AIDS by at least 2 to 3 years. Rates of resistance in populations other than Caucasians are lower; only 3% of African-Americans carry a single mutated gene offering resistance, and virtually no Native Americans, native Africans, or East Asian people carry even one copy of the mutant gene. Therefore, these populations have an increased susceptibility to infection compared to Caucasians.
Because of the presence of the mutant gene in Caucasians compared to other races, it has been hypothesized that the mutation in the CCR5 gene developed relatively recently in evolutionary terms, after the major races split from each other. It has been suggested that a mutation in the CCR5 gene survived in the Caucasian population because it conferred some sort of protection against a deadly disease experienced mostly by this group; this disease has been suggested to be the bubonic plague or a similar disease that ravaged Central Europe approximately 700 years ago. Research is underway to explore this hypothesis and to determine whether the CCR5 protein can be disabled in those with two good copies, thereby offering AIDS resistance to others.
The astute reader will notice that it was mentioned earlier that an individual carrying two mutant copies of the CCR5 gene is usually incapable of becoming infected with HIV. This caveat exists because even if an individual does not have the CCR5 protein, that individual could still become infected with HIV if exposed to already mutated HIV. This situation occurs most frequently if an individual is infected with the virus from someone in the late stages of infection; if this happens, the virus can infect the T cells immediately without needing to infect the macrophages first.
Passage of HIV
HIV is passed between individuals during the exchange of body fluids, including blood, semen, vaginal fluid, and breast milk. Urine and gastrointestinal contents are not believed to be a source of HIV transmission
P.153
unless they visibly contain blood. Tears, saliva, and sweat may contain the virus, but in quantities thought to be too low to cause infection.
Individuals at Risk of Developing HIV
Whether an individual exposed to HIV becomes infected depends on several factors, including the individual's immune, nutritional, and general health status, and the amount of virus to which the individual is exposed. The age and sex of an individual also influence risk.
Individuals at high risk for becoming infected with HIV include those who exchange blood with infected persons in a blood transfusion or through contaminated needles. A contaminated needle-stick exposure may occur accidentally in the health care setting or through the sharing of needles during drug use. The risk of becoming infected after an accidental needle-stick injury with a contaminated syringe is low (0.32%). The risk of becoming infected after a single exposure to contaminated injection-drug equipment is higher (0.67%). Although the risk of becoming infected from a transfusion with infected blood is very high (almost 100%), the blood supply in industrialized countries is routinely tested for the presence of antibodies to HIV and contaminated blood is discarded. However, at certain times after infection with the virus, antibodies may not appear in infected blood, making a contaminated transfusion theoretically possible. These times include the period after infection before antibody response has developed and at the end stages of AIDS, when an individual's immune system may be so depressed that antibody levels are negligible.
Other individuals at risk of becoming infected with HIV are those exposed to semen or vaginal fluid during sexual intercourse with an infected individual. Most at risk of being infected via sexual intercourse are men who receive anal sex from other men, with the probability of HIV transmission after unprotected receptive anal intercourse estimated to be 0.8 to 3.2%. This higher risk is in part due to the breakdown and bleeding of rectal cells that occur during anal intercourse. Heterosexual transmission of the virus also occurs, however, and in fact is the primary mode of transmission in some countries, including sub-Saharan Africa and parts of the Caribbean. In general, heterosexual transmission more easily occurs from male to female, in which the probability of becoming infected from a single encounter with an infected partner is approximately 0.09% for a woman and 0.03% for a man. Although the incidence of infection in male homosexuals is still greater than in heterosexuals in the United States, the incidence of AIDS is increasing rapidly among heterosexual adults, especially women of color or Hispanic origin.
The likelihood of becoming infected with the virus during heterosexual or homosexual intercourse depends on many factors. Exposure to HIV from a partner experiencing a primary infection (i.e., before an antibody response has developed) appears to increase the risk of infection compared to exposure from a partner who has had the infection for a longer period and has already produced antibodies. Similarly, a partner
P.154
nearing the end of an infection has a higher titer of virus than at other times and may show virtually no remaining immune resistance. The presence of a sexually transmitted disease in either partner also increases the risk of sexual transmission because of the presence of open wounds with some sexually transmitted diseases and the increased number of white blood cells in an area of infection. In women, cervical ectopy, a change in the structure of the cervix that causes it to be more fragile and likely to bleed, increases the likelihood of infection from a given exposure. Cervical ectopy is more common in teenagers, pregnant women, and women taking oral contraceptives, placing women from these categories at increased risk. Sex during menstruation may increase the risk of transmission from both sexes. And finally, the incidence of HIV in uncircumcised men is eight times as high as in men who are circumcised. This increased incidence may be related to the co-occurrence of sexually transmitted diseases in uncircumcised men or to an abundance of immune cells present in the foreskin that offer the virus a quick opportunity for infection. There is also a greater risk of infection in sex partners of uncircumcised men. This may explain why some African countries reporting a cultural preference against circumcision show especially high rates of heterosexual transmission.
Spread to Women and Children
In the United States, HIV infection is increasing among women, usually after sexual intercourse with infected intravenous drug users or bisexual men. Today, women make up over 50% of all HIV-infected individuals worldwide. Women are more susceptible than men to infection during heterosexual intercourse because of the normal microscopic vaginal tears and bleeds that occur with intercourse. In addition, infected semen remains in the woman's vagina up to 48 to 72 hours longer than the amount of time the penis is exposed to vaginal secretions.
A woman infected with HIV may pass the infection on to her infant across the placenta, usually during the third trimester, or after exposure of the infant to contaminated blood and amniotic fluid during the birth process. An infant born to an untreated, infected mother has at least a 25% chance of becoming infected with the virus. The use of anti-HIV drugs administered to an infected mother during pregnancy, and to an infant of an infected mother soon after birth, significantly reduces the rate of infant infection, to less than 10% in some studies. Because of this documented protection to the infant, it is recommended that all pregnant women be tested for HIV infection as soon as pregnancy is confirmed. A woman may also acquire the virus after giving birth and pass it to her infant during breastfeeding.
Clinical Manifestations
Flu-like symptoms, including a low-grade fever, aches, and chills, may develop a few weeks to a few months after infection. Symptoms resolve
P.155
after the initial immune response reduces the number of virus particles, although the virus survives in other infected cells.During the latent period, an individual infected with HIV may be asymptomatic, or in some cases may experience persistent lymphadenopathy (swollen lymph nodes).
Two to 10 years after HIV infection, most individuals, if untreated, begin to experience opportunistic infections. These illnesses indicate the onset of AIDS and include vaginal and oral yeast infections and various viral infections such as varicella zoster (chickenpox and shingles), cytomegalovirus, or persistent herpes simplex virus infection. Women may develop chronic yeast infections or pelvic inflammatory disease.
Once AIDS is established, respiratory infections, often with the opportunistic organism Pneumocystis carinii, become frequent. Multiple-drug resistant tuberculosis may develop because a patient with AIDS is unable to mount an effective immune response to fight the bacteria, even with the help of antibiotics. Patients with AIDS who develop tuberculosis typically experience a rapidly worsening course of the disease, leading to death within a few months. The disease frequently spreads to extrapulmonary sites, including the brain and bone.
Central nervous system manifestations include headaches, motor defects, seizures, personality changes, and dementia. Patients may become blind and eventually comatose. Many of these symptoms result from opportunistic viral and bacterial infections of the CNS, which cause inflammation of the brain. HIV also directly infects and injures brain cells.
Diarrhea and wasting away of body fat are common in patients with AIDS. Diarrhea results from opportunistic viral and protozoal infections. Oral and esophageal yeast infections (thrush) cause pain when chewing and swallowing, and contribute to the loss of body fat and failure to thrive.
Various cancers occur at an increased rate in patients with AIDS because of the lack of a cell-mediated immune response against neoplastic cells. Especially common in patients with AIDS is the otherwise unusual cancer, Kaposi's sarcoma, a cancer of the vascular system characterized by red skin lesions. Most individuals who develop Kaposi's sarcoma have been infected through homosexual intercourse. Recent evidence suggests that coinfection with a unique herpesvirus, human herpesvirus 8, may be required for the development of Kaposi's sarcoma. Human herpesvirus 8 is uncommon in the general population, but common in the U.S. homosexual population.
Diagnostic Tools
Immediately after infection, CD4 (helper T) cell counts may decrease, but soon return to near normal as the initial immune response contains the infection.
Antibodies against HIV usually appear 4 to 6 weeks after infection, but may take 6 months or longer in some cases to develop. If a serum sample is identified as HIV positive (having a positive antibody titer), a Western blot test will be performed to confirm infection. Uninfected infants born to infected mothers may appear HIV positive for more than a year after birth because of the presence of maternal antibodies.
Helper T cell counts eventually begin to decrease. When levels reach fewer than 200 to 300 cells/ L of blood, opportunistic infections develop. The progress of the disease and the success of various treatments are followed by measuring a patient's helper T cells over time.
Tests to measure the viral load present at any given time in the blood of an infected individual have been shown to be highly accurate in predicting the occurrence of symptoms, the prognosis, and the general state of health of an individual. Those found to have high amounts of virus have an accelerated progression of the disease regardless of the number of helper T cells. The greater the number of virus particles present in an individual, the greater the infectivity during sexual intercourse and between mother and infant.
Tests to measure HIV RNA also are predictive of host status. HIV RNA levels often are followed to evaluate the success of AIDS treatment.
P.156
Treatment
At this time, there is no cure for AIDS; thus, prevention of HIV infection is essential. Prevention means avoiding contact with HIV-contaminated body fluid. Because it is usually impossible to know in advance whether body fluid is contaminated with HIV, one should assume contamination unless proven otherwise. To avoid exposure to HIV one should:
Practice sexual abstinence or mutually monogamous sexual intercourse with a non-infected partner.
Be tested for the virus at least 6 months after the last unprotected sexual intercourse, because it may take at least 6 months after exposure to the virus to build antibodies. Oral sex may also pass the virus.
Use a latex condom during sexual intercourse with a person whose HIV status is unknown.
Refrain from sharing needles with anyone for any reason.
Prevent infection to a fetus or newborn. A woman should know her own and her partner's HIV status before pregnancy. If a pregnant woman is positive for HIV, anti-HIV drugs or antibodies can be given to her during pregnancy and to the infant after birth. Treatment in utero may also be effective in preventing transmission of the virus to the fetus or newborn. An infected mother should not breastfeed her infant. Breast pumps should not be shared.
Post-exposure prophylactic treatment with a reverse transcriptase inhibitor after accidental needle stick or sexual exposure decreases the odds of acquiring primary HIV infection.
P.157
If infection occurs, drug treatment regimens are available that can dramatically change the course of infection.
HIV/AIDS is treated by following the treatment regimen known as highly active retroviral therapy or HAART. HAART involves a combination of drugs that include one or more of the following:
A nucleoside reverse transcription inhibitor (NRTI). This type of drug (e.g., azidothymidine or AZT) interferes with viral transcription into host DNA by blocking the action of the viral enzyme reverse transcriptase by interfering with the availability of a nucleoside (thymidine).
A non-nucleoside reverse transcription inhibitor (NNRTI). This type of drug acts via non-competitive binding to block the active site on the reverse transcriptase enzyme. These drugs are effective in combination with other medications such as the NRTIs.
A protease inhibitor, which blocks the action of the HIV protease enzyme necessary for the production of mature viral particles. While effective, protease inhibitor therapy is associated with a condition known as HIV-associated lipodystrophy, characterized by hyperlipidemia, insulin resistance, and a redistribution of body fat to the abdomen, breast, and back. The etiology of this syndrome is multifactorial, and includes an effect of the protease inhibitors on fat loss from adipose tissue and on preadipocyte differentiation.
The HAART regimen does not cure AIDS, but can dramatically prolong survival time and improve the quality of life for infected individuals. Questions exist regarding when to initiate therapy, with concerns over drug side-effects and the potential for the virus to gain resistance to the medications. Recent data suggest that initiating therapy early in the course of infection may stave off most serious side effects and improve survival.
HAART appears to be safe and effective in pregnant women, although questions regarding teratogenesis remain. Current recommendations are that, if possible, therapy be discontinued during the first trimester and then resumed.
HAART is safe for use in infants born to mothers infected with HIV.
Besides the side effect described for the protease inhibitors, side effects of the NRTI and the NNRTI medications include nausea, headaches, and bone marrow suppression, leading to anemia and fatigue. Adherence to HAART is difficult and may be impossible for some patients. Drugs must be taken frequently and at certain times of the day. The cost of long-term combination therapy is high and HIV disproportionately affects the poor; thus, therapy may be impossible in some countries and for uninsured individuals.
A healthy diet and stress-free lifestyle are important. Treatment should include education to avoid alcohol, smoking, and illicit drugs. Stress, poor nutrition, alcohol, and other drugs are known to impair immune functioning.
Patients should avoid other infections because they could lead to activation of T cells and may accelerate the replication of HIV. To prevent infection, available vaccines should be administered as long as live-viruses vaccines are not used.
Treatment for specific infections and cancers should be administered as they arise.
P.158
Selected Bibliography
Cohen, J. (2005). Is an effective HIV vaccine feasible? Science 309, 99 100.
Condon, C. (2005). A vaccine in 50 days? Lancet 366, 1686 1687.
Finn, O.J. (2005). Immune response as a biomarker for cancer detection and a lot more. New England Journal of Medicine 353, 1288 1290.
Glass, R.I., and Steele, A.D. (2005). The value of cholera vaccines reassessed. Lancet 366, 7 9.
Kinet, J.-P. (2005). A new strategy to counter allergy. New England Journal of Medicine 353, 310 312.
Markel, H. (2005). The search for effective HIV vaccines. New England Journal of Medicine 353, 753 757.
Medzhitov, R., and Janeway, C., Jr. (2000). Innate immunity. New England Journal of Medicine 343, 338 344.
Peng, G., Guo, Z., Kiniwa, Y., Voo, K., Peng, W., Fu, T., et al. (2005). Toll-like receptor 8 mediated reversal of CD4+ regulatory T cell function. Science 309, 1380 1384.
Porth, C.M. (2005). Pathophysiology: Concepts of altered health states (7th ed.). Philadelphia: Lippincott Williams & Wilkins.
Soysal, A., Millington, K.A., Bakir, M., Dosanjh, D., Aslan, Y., Deeks, J.J., et al. (2005). Effect of BCG vaccination on risk of Mycobacterium tuberculosis infection in children with household tuberculosis contact: a prospective community-based study. Lancet 366, 1443 1451
Wickelgren, I. (2004). Policing the immune system. Science 306, 596 599.
EAN: 2147483647
Pages: 26