4 - Acute Exacerbations of Heart Failure - Initial Evaluation and Management in the Acute Care Setting
Editors: Peacock, W. Frank
Title: Short Stay Management of Heart Failure, 1st Edition
Copyright 2006 Lippincott Williams & Wilkins
> Table of Contents > 4 - Acute Exacerbations of Heart Failure: Initial Evaluation and Management in the Acute Care Setting
4
Acute Exacerbations of Heart Failure: Initial Evaluation and Management in the Acute Care Setting
Douglas S. Ander
Introduction
Heart failure (HF) is a common diagnosis, facing approximately 4,900,000 people.1 Over the past 3 decades we have seen a significant rise in the number of hospital discharges for HF,1 and because the majority of admitted patients enter through the emergency department (ED),2 proper initial management is important. Several studies on the use of the B-type natriuretic peptide (BNP) assay have indicated that earlier treatment in the ED can significantly impact hospital length of stay and cost.3
Patients may present to the ED with minimal symptoms, such as mild dyspnea or weight gain. Conversely, they may present to the ED with overt pulmonary edema. In addition to their fluid status, evaluation for adequate perfusion or evidence of shock should be considered. Recognition that HF patients may fall into different hemodynamic categories allows the clinician to tailor treatment based on the initial assessment. Using components of the history and physical examination, the clinician can develop a treatment plan individualized to that patient. Diuretics, vasodilators, angiotensin-converting enzyme inhibitors (ACEI), noninvasive ventilation, and natriuretic peptides can be used for hemodynamic and symptomatic needs.
Because of improvements in the treatment of HF that have resulted in improved survival, the emergency physician will see increased instances of acute exacerbations of HF. This chapter provides the reader with an understanding of the principles of initial acute HF management and examine specific therapeutic agents.
P.28
Initial Stabilization
The first step when a patient presents with presumed HF is to stabilize the clinical condition. This is followed by an evaluation including laboratory tests, radiographs, consideration of advance directives, and a search for reversible causes including ischemia and arrhythmias. Finally, treatment is instituted based on an assessment of the hemodynamic status.
Patients presenting with presumed HF should be initially assessed and stabilized. Those with impending respiratory failure should be treated with supplemental oxygen and the physician should consider the use of noninvasive ventilation. Patients who are unable to control their airway, cannot tolerate noninvasive ventilation, or worsen despite these measures should be considered for endotracheal intubation.
When HF is considered the most likely etiology of the patient's symptoms, the initial workup should include establishment of intravenous (IV) access, a focused history and physical examination, assessment of the degree of fluid overload and perfusion, evaluation of oxygenation, assessment of the cardiac rhythm, evaluation for cardiac ischemia with a 12-lead electrocardiogram, and a chest radiography. Blood work should be based on clinical suspicion and may include a complete blood cell count, electrolytes, BNP or N-terminal pro-BNP levels, and cardiac markers.
Advance directives should be discussed with the patient or family, especially in those with severe exacerbations of chronic disease. This information will help direct the management plan.
The clinician should look for precipitants of the HF (Table 4-1). The identifiable reversible causes should be treated in conjunction with the treatment of the HF. Unfortunately, in one study the authors could not identify a precipitant in 40% of the HF presentations.4 Patient-related factors such as noncompliance with medication and diet should be addressed. In several studies, poor compliance was the precipitant of the acute exacerbation, ranging from 21% to 41.9%.4,5 Patient and family education cannot be underestimated. Several studies have investigated the impact of education and find that it significantly decreases hospital readmissions.6, 7 and 8
Cardiac ischemia is a leading cause of HF and an important cause of acute exacerbation.9 Although not every exacerbation of chronic HF will require a workup of an ischemic etiology, the clinician should be aware of the potential and consider it accordingly. Cardiac arrhythmias are associated with a worse prognosis in HF.10 In the acute care setting, cardiac arrhythmias such as atrial fibrillation, ventricular arrhythmias, and conduction abnormalities can be precipitants of exacerbations. Treatment should be focused on the hemodynamic effects and not correction of every arrhythmia. Patients with chronic HF are at significant risk for ventricular arrhythmias.
Principles of Treatment
The classic picture of an HF patient with acute pulmonary edema frothing at the mouth and appearing cyanotic is only one possible presentation. Some individuals present with respiratory distress, but others may have
P.29
only mild symptomatic dyspnea and others have only fatigue. Some present hypotensive and others with significant hypertension.
TABLE 4-1 Precipitants of Heart Failure Exacerbations | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Categorizing HF patients using a hemodynamic classification system (Figure 4-1) is a useful approach. Based on the initial history and physical examination, patients can be placed into a hemodynamic category and then appropriate therapy can be selected. Fluid overload can be assessed by the presence or absence of dyspnea, orthopnea, pulmonary rales, elevated jugular venous pressure, the presence of a third heart sound S3, and hepatomegaly. Perfusion can be estimated by evaluating for the presence or absence of fatigue, nausea, symptomatic hypotension, and cool extremities (Table 4-2).
The physician must recognize that these signs and symptoms can be inaccurate and have a low inter-rater reliability.11 Assessment using these historical and physical diagnostic clues needs to be considered in aggregate and supplemented with more objective diagnostic testing (chest radiograph, BNP or pro-BNP level, bioimpedance measurements, digital S3detection) to improve accuracy. At this point, this clinical assessment strategy provides the best estimate of the patient's hemodynamic status.
P.30
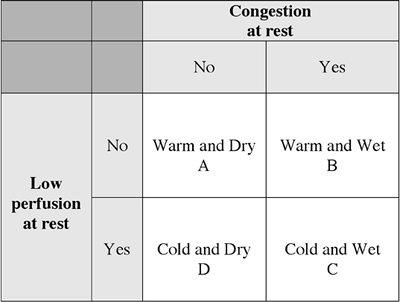 |
FIGURE 4-1 Bedside assessment of hemodynamic status and corresponding therapeutic intervention. (Adapted from Stevenson et al.) |
Most patients presenting with HF are fluid overloaded but have adequate perfusion, that is, wet and warm. These patients require diuretics and vasodilation. The fluid-overloaded patient could also present with diminished perfusion, called wet and cold. Those with elevated systemic vascular resistance (SVR) will require vasodilation. Those with symptomatic hypotension and diminished SVR may require inotropic support. The least common patient is the patient that has been overdiuresed and has evidence of hypoperfusion, the dry and cold. If manifesting symptomatic hypotension, this patient may require a fluid bolus and the addition of inotropic support. As a guiding principle, chronic HF patients are best served by lower blood pressures, with a systolic blood pressure as low as 80 to 90 mm Hg. As long as they are asymptomatic, continue to mentate, and have appropriate urine output, no further blood pressure treatment is necessary.
P.31
TABLE 4-2 Clinical Bedside Assessment of Heart Failure | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
This hemodynamic classification system provides the physician with a framework for the initial assessment and treatment of the HF patient. However, this framework is based on imprecise measures; therefore, the initial treatment must be guided by good clinical judgment and must be adjusted depending on the initial response to the therapeutic interventions. Until better and more accurate diagnostic modalities are available, the physician should use the framework presented in this section to develop the therapeutic plan.
Specific Therapeutic Agents and Modalities
Diuretics
Patients presenting with evidence of fluid overload, the wet patient, benefit by the use of a diuretic. Loop diuretics act through the inhibition of sodium reabsorption at the loop of Henle to promote diuresis. The resultant volume reduction causes a decrease in filling pressures and pulmonary congestion. This provides the patient with symptomatic improvement. Peak diuretic effect is typically seen within 30 minutes of administration. Physicians should be aware that some studies have demonstrated adverse physiologic effects of furosemide administration including initial increases in filling pressures prior to diuresis and adverse neurohormonal effects by increasing renin, norepinephrine, and vasopressin levels.12
P.32
The usual starting dose of furosemide is 20 to 80 mg intravenously. A patient with no previous exposure to furosemide may have adequate response to lower doses. If a patient is currently on furosemide, using the current enteral dose as an IV bolus is a good starting point. If no response is seen within 30 to 60 minutes, the dose can be doubled. Treatment with diuretics may lead to hypokalemia and hypomagnesemia; therefore, monitoring and treatment of electrolyte abnormalities is important. Despite some of the potential hemodynamic and neurohormonal effects of loop diuretics, diuresis and symptomatic improvement seen with furosemide administration make it the drug of choice for the fluid-overloaded patient.
Nitrates
The patient with fluid overload and reasonable perfusion ( wet and warm ) can be successfully treated with a reduction in preload, which decreases ventricular workload. Nitroglycerin (NTG) achieves the reduction in filling pressures by dilation of the venous system.13 At higher doses, there is dilation of the arterial system, causing decreased left ventricular (LV) work. NTG also dilates epicardial coronary arteries, increasing collateral blood flow.14 Some evidence exists showing that high doses of nitrates results in better outcomes, with decreased need for mechanical ventilation and fewer myocardial infarctions.15,16 As long as there is adequate blood pressure, NTG can be administered sublingually as either a spray or a pill (0.4 mg) at 1-minute intervals. This can be done effectively before institution of IV access. This may be followed by transdermal NTG paste (1 to 2 inches), if the patient is perfusing adequately to permit absorption. If continued reductions in preload and afterload are required, an NTG drip may be started and titrated to effect. Dosing can start at 10 to 20 g per minute and titrated quickly every 3 to 5 minutes. Doses can reach 200 to 300 g per minute to achieve the desired effect. Adverse effects of nitrates include headache, sinus tachycardia, abdominal pain, tachyphylaxis, and hypotension. Despite a minimal number of prospective trials, nitrates with diuretics form the basis behind the treatment of the fluid-overloaded patient.
Nitroprusside
Some patients with extreme elevations of SVR require additional afterload reduction not provided by NTG. Nitroprusside (NTP) can be added if further afterload reduction is warranted. NTP is a potent vasodilator that relaxes both arteriolar and venous smooth muscle, with resultant improvement in cardiac output and stroke volume.17 The decrease in preload and afterload results in augmentation of cardiac output and stroke volume. IV infusion dose starts at 0.1 to 0.2 g/kg/minute and can be increased every 5 minutes. Dangerous hypotension can occur precipitously, mandating continuous blood pressure monitoring. Other adverse effects include the theoretic coronary steal syndrome if a fixed epicardial coronary
P.33
artery obstruction exists. Patients with renal failure are at risk for thiocyanate toxicity with prolonged administration of NTP. Finally, a reflex tachycardia may adversely affect myocardial oxygen consumption.
Morphine
Morphine sulfate has potential hemodynamic benefits in the treatment of HF by causing an increase in venous capacitance and a decrease in preload.18 Morphine is administered in incremental doses of 2 to 10 mg every 5 to 15 minutes. It should be titrated to effect with careful observation of its respiratory and hemodynamic effects. Patients with acute pulmonary edema present with significant air hunger. In this population, morphine has the ability to decrease anxiety and may be beneficial. Unfortunately, very limited data support the therapeutic advantages of using morphine. One retrospective study has associated morphine with an increased need for intensive care unit admission.19
Fluids
Patients with poor perfusion and who have been overdiuresed, the cold and dry, typically report an increased use of their diuretics. They can be recognized by symptoms of increased fatigue, nausea, cool extremities, and hypotension, without any evidence of fluid overload. Although counterintuitive to the treatment of HF, these patients may benefit from a fluid bolus of 250 to 500 mL of isotonic fluid. This should be done cautiously with continuous reassessment of the patient's response and fluid status.
Inotropic/Pressor Support
In the patient with poor perfusion and signs of fluid overload, the cool and wet, inotropic support is sometimes indicated. Vasodilators and diuretics are first-line agents, but if the patient does not have an adequate response and has evidence of poor perfusion without overt shock, pure beta agonists such as dobutamine or a phosphodiesterase inhibitor such as milrinone may be required. The initial dose of dobutamine is 2 to 5 g/kg/minute, which can be titrated up to 20 g/kg/minute. Dobutamine, as a beta-1 receptor agonist, acts to increase myocardial contractility and improve cardiac output. In most patients, this should be sufficient to overcome the vasodilator effects of the beta-2 activity. Caution should be exercised because, in patients with borderline hypotension, the addition of dobutamine may lead to significant hypotension.
If cardiogenic shock is present, a mixed catecholamine agonist such as dopamine may be added to the regimen. One should remember that cardiogenic shock implies evidence of poor perfusion and is not dependent on the blood pressure. Intra-aortic balloon pump placement may also be considered as a bridge to cardiac transplantation.
P.34
Natriuretic Peptides
The warm and wet patient, and even the patient with some degree of hypoperfusion, may benefit from vasodilation. Nesiritide (Scios, Tremont, CA) is an IV human BNP and provides a combination of neurohormonal and hemodynamic effects that may be beneficial in the initial management of HF. Neurohormonal effects include improved sodium and water excretion and decreased endothelin, norepinephrine, aldosterone, renin, and angiotensin levels.20 Hemodynamic effects include decreasing filling pressures and SVR and an increase in cardiac output, without an increase in heart rate.20, 21 and 22
Several trials have studied the effects of nesiritide on inpatients with HF. Colucci et al.23 reported a dose-dependent decrease in filling pressures and improved symptoms. The Vasodilation in the Management of Acute HF trial compared the addition of IV nesiritide or IV NTG to standard HF therapy.24 Nesiritide, administered as a 2 g per kilogram bolus, followed by a maintenance infusion of 0.01 g/kg/minute resulted in decreased filling pressures and improvement in dyspnea compared with standard therapy. In the observation unit, nesiritide is safe and its use resulted in decreased 30-day readmissions to the hospital as compared with standard therapy.25
Although no large-scale trials exist using nesiritide, it has comparable or slightly better hemodynamic attributes than NTG, does not need to be titrated, has no associated tachycardia or tachyphylaxis, is nonarrythmogenic, and attenuates the neurohormonal derangements of HF. Based on two recent meta-analyses of nesiritide, clinicians should be cautious when instituting therapy.26,27 Before starting nesiritide, there should be clear indications of decompensated HF in patients who have failed standard therapy. Future large studies are necessary to elucidate the best and safest use of nesiritide.
Angiotensin-Converting Enzyme Inhibitors
The renin-angiotensin-aldosterone system (RAAS) plays an important role in the neurohormonal derangements of HF. Activation of the RAAS, although initially beneficial, leads to many of the adverse hemodynamic consequences of HF. The ability of ACE inhibitors (ACEIs) to relax both arterial resistance and venous vessels and to lower impedance to LV ejection provides the patient with a hemodynamic advantage. Patients with asymptomatic LV dysfunction and chronic HF derive a mortality reduction from therapy with ACEI.28,29 Several studies of severe chronic HF demonstrate dramatic improvements in hemodynamics immediately after administration of an ACEI.30, 31 and 32
Some preliminary evidence exists supporting the use of an ACEI for acute exacerbations of HF.33,34 In a small prospective, randomized, double-blind, placebo-controlled trial of 48 patients, Hamilton and Gallagher34 evaluated the role of sublingual captopril for acute pulmonary edema. They measured HF severity using a composite of four parameters: patient-reported dyspnea, physician judgment of respiratory distress, diaphoresis,
P.35
and level of bed elevation tolerance. Using this composite score, they demonstrated that sublingual captopril results in better scores during the first 40 minutes of therapy. However, there was no change in mortality, admissions, or intubation rates and this study was too small to evaluate safety.
ACEIs can be administered as captopril 25 mg by mouth or sublingually. Oral captopril has an onset of action within 15 to 30 minutes. Sublingual captopril decreases pulmonary capillary wedge pressure significantly after 10 minutes.35 Captopril should probably be avoided if the patient's systolic blood pressure is less than 90 mm Hg.
Despite the lack of definitive, large-scale trials, some centers advocate the use ACEIs as a routine component of their therapeutic regimen.19 Based on hemodynamic data and one ED study, addition of ACEIs in patients with high SVR appears to be advantageous. Because there are no large placebo-controlled trials using ACEIs in the acute care setting, their use cannot be recommended as a standard of care.
Respiratory Considerations
All patients presenting with HF should receive supplemental oxygen. Patients with respiratory distress and dyspnea will typically respond to aggressive medical management. Failure of medical management and worsening respiratory status may lead to endotracheal intubation. Two noninvasive ventilatory techniques are continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BiPAP), which may be used to treat the respiratory distress.
CPAP, when added to standard therapy in HF patients, does not change overall hospital mortality36, 37 and 38 but does decrease endotracheal intubations. A pooled systematic review comparing standard therapy with and without CPAP reported a trend in the reduction of the risk of mortality of -6.6% (95% CI: -16% to +3%) and a reduction in the risk of intubation of -26% (95% CI: -13% to -38%).39
A pooled review comparing BiPAP to standard therapy revealed intubation rates for BiPAP ranging from 0% to 44% and mortality rates from 0% to 22%.39 A more recent trial, not included in the pooled analysis, reported an intubation rate for BiPAP of 23.8% versus a 41.2% standard therapy rate.40 In the BiPAP group, there was a lower myocardial infarction rate as compared with standard therapy, 19% versus 29.4%, respectively. Only one randomized trial has compared CPAP directly with BiPAP.41 The researchers found no difference in hospital mortality or intubation rates. The study was halted early due to a high rate of myocardial infarction in the BiPAP group compared with the CPAP cohort, 71% versus 31%, p = 0.006, respectively. It is unclear whether this phenomenon is unique to the BiPAP or due to a higher number of patients in the BiPAP group with chest pain potentially indicating underlying ischemia.
Noninvasive ventilation to avoid endotracheal intubation has some promise in the treatment of severe respiratory distress. Data with large-scale
P.36
studies is lacking, and some evidence even exists that noninvasive ventilation may be harmful. The clinician should use caution when considering the use of noninvasive ventilation in HF, especially in the patient with possible ischemic heart disease.
Conclusion
The management of patients with acute exacerbations of HF is difficult because the history, physical examination, and chest radiography lack the accuracy clinicians need to accurately make the diagnosis and determine the degree of decompensation. Clinicians must use the tools at hand in the acute care setting to accurately diagnose and categorize the hemodynamic derangements. Once the patient's clinical condition has been stabilized, he or she can be treated based on hemodynamic classification. Treatment should be focused on improving symptoms, decreasing volume overload, and improving perfusion. In the majority of cases, this can be done with diuretics and vasodilators. In some, the addition of inotropic agents and pressor agents may be needed to improve perfusion.
Ongoing research on the diagnosis, management, and disposition of patients presenting with ADHF is needed, especially in the acute care environment. Applying the results from research in chronic HF or from the inpatient cohort to the acute care population is fraught with potential for error. As this research grows, clinicians can expect to become more accurate in diagnosing HF and providing treatment specifically designed to combat the neurohormonal and hemodynamic changes of HF.
References
1. American Heart Association. Heart disease and stroke statistics 2005 update. Dallas, TX: American Heart Association, 2005.
2. ADHERE, Acute decompensated heart failure national registry, Vol. 4th quarter 2002 National Database. Sunnyvale, CA: ADHERE, 2003.
3. Mueller C, et al. Use of B-type natriuretic peptide in the evaluation and management of acute dyspnea [see comment]. N Engl J Med 2004;350:647 654.
4. Opasich C, et al. Precipitating factors and decision-making processes of short-term worsening heart failure despite optimal treatment (from the IN-CHF Registry). Am J Cardiol 2001;88:382 387.
5. Michalsen A, Konig G, Thimme W. Preventable causative factors leading to hospital admission with decompensated heart failure [see comment]. Heart 1998;80:437 441.
6. Philbin EF. Comprehensive multidisciplinary programs for the management of patients with congestive heart failure [comment]. J Gen Intern Med 1999;14:130 135.
7. Rich MW, et al. A multidisciplinary intervention to prevent the readmission of elderly patients with congestive heart failure [comment]. N Engl J Med 1995;333:1190 1195.
8. West JA, et al. A comprehensive management system for heart failure improves clinical outcomes and reduces medical resource utilization. Am J Cardiol 1997;79:58 63.
9. Goldberger JJ, et al. Prognostic factors in acute pulmonary edema. Arch Intern Med 1986;146:489 493.
P.37
10. Dries DL, et al. Atrial fibrillation is associated with an increased risk for mortality and heart failure progression in patients with asymptomatic and symptomatic left ventricular systolic dysfunction: a retrospective analysis of the SOLVD trials. Studies of Left Ventricular Dysfunction. J Am Coll Cardiol 1998;32:695 703.
11. Badgett RG, Lucey CR, Mulrow CD. Can the clinical examination diagnose left-sided heart failure in adults? [see comments.]. JAMA 1997;277:1712 1719.
12. Francis GS, et al. Acute vasoconstrictor response to intravenous furosemide in patients with chronic congestive heart failure. Activation of the neurohumoral axis. Ann Intern Med 1985;103:1 6.
13. Bussmann WD, Schupp D. Effect of sublingual nitroglycerin in emergency treatment of severe pulmonary edema. Am J Cardiol 1978;41: 931 936.
14. Cohen MCDJ, Sonnenblick EH, Kirk ES. The effects of nitroglycerin on coronary collaterals and myocardial contractility. J Clin Invest 1973;52: 2836 2847.
15. Cotter G, et al. Randomised trial of high-dose isosorbide dinitrate plus low-dose furosemide versus high-dose furosemide plus low-dose isosorbide dinitrate in severe pulmonary oedema. Lancet 1998;351:389 393.
16. Sharon A, et al. High-dose intravenous isosorbide-dinitrate is safer and better than Bi-PAP ventilation combined with conventional treatment for severe pulmonary edema [comment]. J Am Coll Cardiol 2000;36:832 837.
17. Rossen RM, Alderman EL, Harrison DC. Circulatory response to vasodilator therapy in congestive cardiomyopathy. Br Heart J 1976;38:695 700.
18. Vismara LA, Leaman DM, Zelis R. The effects of morphine on venous tone in patients with acute pulmonary edema. Circulation 1976;54:335 337.
19. Sacchetti A, et al. Effect of ED management on ICU use in acute pulmonary edema. Am J Emerg Med 1999;17:571 574.
20. Abraham WT, et al. Systemic hemodynamic, neurohormonal, and renal effects of a steady-state infusion of human brain natriuretic peptide in patients with hemodynamically decompensated heart failure. J Cardiac Fail 1998;4:37 44.
21. Marcus LS, et al. Hemodynamic and renal excretory effects of human brain natriuretic peptide infusion in patients with congestive heart failure. A double-blind, placebo-controlled, randomized crossover trial. Circulation 1996;94:3184 3189.
22. Hobbs RE, et al. Hemodynamic effects of a single intravenous injection of synthetic human brain natriuretic peptide in patients with heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol 1996;78:896 901.
23. Colucci WS, et al. Intravenous nesiritide, a natriuretic peptide, in the treatment of decompensated congestive heart failure. Nesiritide Study Group. N Engl J Med 2000;343: 246 253.
24. Publication Committee for the V.I. Intravenous nesiritide vs nitroglycerin for treatment of decompensated congestive heart failure: a randomized controlled trial. JAMA 2002;287:1531 1540.
25. Peacock WF 4th, Holland R, Gyarmathy R, et al. Observation unit treatment of heart failure with nesiritide: results from the proaction trial. J Emerg Med 2005;29: 243 252.
26. Sackner-Bernstein JD, Skopicki HA, Aaronson KD. Risk of worsening renal function with nesiritide in patients with acutely decompensated heart failure [see comment]. Circulation 2005;111:1487 1491.
27. Sackner-Bernstein JD, et al. Short-term risk of death after treatment with nesiritide for decompensated heart failure: a pooled analysis of randomized controlled trials. JAMA 2005;293:1900 1905.
28. Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). The CONSENSUS Trial Study Group. N Engl J Med 1987;316:1429 1435.
P.38
29. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. The SOLVD Investigators. N Engl J Med 1991;325:293 302.
30. Haude M, Erbel R, Meyer J. Sublingual administration of captopril versus nitroglycerin in patients with severe congestive heart failure. Int J Cardiol 1990;27: 351 359.
31. Powers ER, Stone J, Reison DS, et al. The effect of captopril on renal, coronary, and systemic hemodynamics in patients with severe congestive heart failure. Am Heart J 1982;104:1203 1210.
32. Kubo SH, Laragh JH, Prida XE, et al. Immediate converting enzyme inhibition with intravenous enalapril in chronic congestive heart failure. Am J Cardiol 1985;55:122 126.
33. Annane D, et al. Placebo-controlled, randomized, double-blind study of intravenous enalaprilat efficacy and safety in acute cardiogenic pulmonary edema. Circulation 1996;94:1316 1324.
34. Hamilton RJ, Gallagher EJ. Rapid improvement of acute pulmonary edema with sublingual captopril. Acad Emerg Med 1996;3:205 212.
35. Barnett JC, Touchon RC. Sublingual captopril in the treatment of acute heart failure. Curr Ther Res 1991;49:274 281.
36. Bersten AD, et al. Treatment of severe cardiogenic pulmonary edema with continuous positive airway pressure delivered by face mask. N Engl J Med 1991;325:1825 1830.
37. Lin M, et al. Reappraisal of continuous positive airway pressure therapy in acute cardiogenic pulmonary edema. Short-term results and long-term follow-up. Chest 1995;107: 1379 1386.
38. Rasanen J, et al. Continuous positive airway pressure by face mask in acute cardiogenic pulmonary edema. Am J Cardiol 1985;55:296 300.
39. Pang D, et al. The effect of positive pressure airway support on mortality and the need for intubation in cardiogenic pulmonary edema: a systematic review. Chest 1998;114: 1185 1192.
40. Levitt MA. A prospective, randomized trial of BiPAP in severe acute congestive heart failure. J Emerg Med 2001;21:363 369.
41. Mehta S, et al. Randomized, prospective trial of bilevel versus continuous positive airway pressure in acute pulmonary edema. Crit Care Med 1997;25:620 628.
EAN: 2147483647
Pages: 18