5.2 The Quality Manual Controversy
|
5.2 The Quality Manual Controversy
5.2.1 An Issue of Content
In the attempt to create a fully responsive QMS, we have found that the content of the manual is the most controversial element in the creation of an effective ISO 9000 documentation and implementation system. It is the root cause of misunderstanding and misapplication of the Standard.
By contrast, procedural documentation and forms, whether effective or not, are more easily understood by those who either create or use them. The reason for this wide discrepancy is not mysterious. The vast majority of our language deals with how things are done, how they are resolved, or how things are put together and taken apart. We seldom speak in terms of policy or principle unless litigation is involved. Then we throw up our hands and turn the problem over to our lawyers.
In the case of manual design, it is necessary to carefully consider the manner in which the ISO 9000 elements are discussed with regard to policy; the degree of detail required to describe such policies; the location of policy within the manual versus a location in lower tier documents; the use of paraphrasing instead of clear, descriptive text; and the choice of presentation as either a stand-alone document or an integrated set of documents.
This issue of content existed for the 1987 and 1994 versions of the international Standard and has already shown to be the same for the 2000 version. In fact, there were ISO 9001:2000-paraphrased manual software advertisements on the market before the Standard's final draft was even released.
All of these topics are discussed in detail within this book and the reader is presented with a number of alternative approaches to the manual's content—all of which, we believe, are in full compliance with the Standard.
Our observations with regard to manual content are based on the review of over 100 manuals during 10 years of firsthand experience in the accredited certification of more than 100 companies ranging in size from eight to 2,000 employees—in industries as diverse as printed wiring assembly contract manufacturing, medical device houses, manufacturers of electronic systems, and the growth of laboratory-bred animals. We have also observed the same issue in the areas of QS-9000 for automotive and ISO 14000 for environmental, based on a much smaller firsthand sample.
This wide interpretive disparity of the ISO 9000 guidelines—especially the interpretation of the ISO 10013:1995 "Guidelines for Developing Quality Manuals"—has proven to be counterproductive for both suppliers and ISO 9000 practitioners. Unfortunately, the clients have been placed in the middle of the conflict and have become a captive audience who must agonize over what is best for their organizations based on controversial information. This situation is hardly conducive to the elusive customer satisfaction we wish to achieve.
5.2.2 Manual's Value
We believe that a clear controversy exists with regard to what constitutes a fully responsive manual and thus an effective QMS. The question about whether we should exert our efforts to resolve the controversy is not as clear. As a result, we first need to examine the value of the manual to the organization in the ISO 9001:2000 QMS and clarify its strategic role in both the certification process and the development of an effective QMS. That the manual is considered a significant document as part of quality management has been established in part through rigorous analysis [5].
This book asserts that it is through the flow of prescriptive, quality policy statements into the lower tier documentation—and the resultant operational feedback by users of that documentation to the organization's top management—that completeness, closure, and continuous QMS informational growth occurs.
5.2.3 Major Gate
As indicated in Figure 5.1, once the strategic plan is created and the stewardship established, the manual is the first critical documentation gate an organization must pass through to complete their QMS.
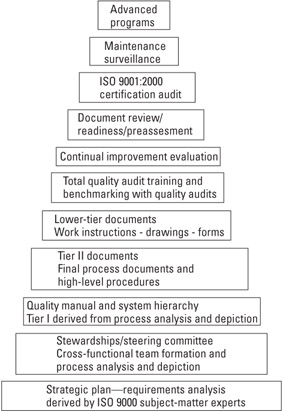
Figure 5.1: Typical ISO 9001—2000 certification gates.
We note that the manual is derived from an analysis of the organization's total processes—from its strategic front-end core competencies to its after-sales service activities (e.g., from marketing and sales to engineering to manufacturing to the repair of returned product).
Most importantly, to satisfy the ISO 9001:2000 requirements, the manual must include a description of the interaction between the several processes of the QMS. Thus, the manual becomes the unifier of strategic organizational thought. It is the glue that binds the various operational areas together. It is the first document that fully encompasses the manner in which the organization carries out its core competencies.
Once the manual is complete, the process/procedural documents can be finalized and a total quality auditing system can be put in place to monitor the effective implementation of the QMS. By total quality auditing, we mean audits at the system, process, and product level—internally via the organization's employees and also by customer and third-party auditors, and externally at the subcontractor's/supplier's facilities [6].
In addition, the manual is the first document seen by the registrar's auditors when the time comes to schedule the initial (certification) assessment. Unless the manual is acceptable, the registrar normally can go no further into the process. The manual review (a central part of the document review process) is often accomplished by the registrar as either an off-site activity or part of a documentation review in your facility prior to either the on-site initial assessment or the optional on-site preassessment.
The manual is also the primary document requested by your customer/client in their evaluation of your QMS. A fully responsive manual will often remove the necessity of an on-site audit, while a manual that simply repeats the Standard's text (a paraphrased manual) leaves your customer/client with no alternative but to visit you to better judge your conformance to the Standard if you are still considered a viable supplier. There are, of course, situations where the customer actually requests a trivialized manual. If that is what the customer wants, that is what the customer gets. However, that does not mean that the operational manual used by you should be the trivialized one.
The manual is the major driver for effectiveness in the QMS because it mandates the policies followed during management review, corrective and preventive action with customer complaints, analysis of data, and internal quality auditing.
5.2.4 Competitive Advantage
We have observed in practice that a fully compliant manual—that reflects both the personality and technical competence of the organization—significantly enhances the organization's competitive position. By comparison, we have observed that an inadequate manual has served as a competitive disadvantage and a source of delay in the certification process.
5.2.5 Rationale for an Ineffective Manual
In juxtaposition to the intrinsic value of a prescriptive manual, the following rationale is used by practitioners to answer why it doesn't matter if the manual is useless:
-
Disinterest on the part of the customer;
-
The only important documents are the operational ones;
-
Disinterest on the part of the distributor;
-
Protection of proprietary information;
-
Disinterest on the part of top management;
-
Wish to minimize the documentation effort;
-
Insensitivity to those who want to understand the enterprise's strategic market position;
-
Disinterest on the part of the employees;
-
Misunderstanding of the manual's value as the primary navigator to find documentation;
-
The position that the employees don't need to read the manual anyway;
-
The customer simply wants a summary manual.
Of course, there are elements of truth in each of these positions. For example, selling product directly to either a high-volume end user or to a service provider can negate the need for a sophisticated quality policy manual. The warranty is of more importance. This situation, however, does not negate the need for the organization to have a dynamic internal QMS as a way to increase profitability through reduction of variance.
5.2.6 Conclusion
Based on this discussion, we see that although the manual is of an intrinsic value to the organization because it describes the strategic organizational viewpoint, a lack of definitive criteria establishes what constitutes a fully responsive manual. This interpretive issue has always existed with the previous ISO 9000 versions, and we already see a worsening situation for ISO 9001:2000, which requires even more interpretive aid than the previous versions [7]. The tendency is to continue to trivialize the manual through a slightly revised restatement of the Standard's text. We have concluded that the controversy needs much attention.
5.2.7 Observed Root Causes
We believe that the controversy is a result of rapid growth in the ISO 9000 industry. For example, approximately 10 years ago, in the very early days of United States entry into the world of ISO 9000, we found that the manual's structure was essentially a nonissue. We believe that this was due to the following:
-
Relatively few suppliers that were large, multidivisional companies with established quality-assurance and control departments [8];
-
The involvement of basically only high-tech organizations;
-
The involvement of a limited number of registrars;
-
Lead assessors shared similar quality-assurance backgrounds;
-
A strong TQM influence;
-
Basic quality programs formed from Mil-Q-9858A and FDA/GMP 820 standards.
We observed that the manual controversy grew slowly after 1994, and then in the period 1995 to the present accelerated into what we believe is a major issue. We have concluded that this change is due in part to the following:
-
The explosion of candidates in small, medium, and large organizations in extremely diverse fields;
-
Candidates in widely ranging levels of technology;
-
A plethora of registrars and consulting groups and more on the way [9];
-
A broad spectrum of lead assessors with varied backgrounds;
-
Enhancements to TQM—for example, reengineering, quality function deployment (QFD), six sigma programs, enhanced Statistical Process Control (SPC), best practices management; business process engineering;
-
The introduction of both integrated Standards—for example, QS-9000/ISO/TS 16949 (automotive), ISO 14000 (environmental), TL-9000 (telecommunications), AS9000 (aerospace), FDA/CGMPs (USA) medical devices, Medical Device Directive (International); and a profusion of ISO 9000 guidelines.
We offer our approach to ISO 9000 QMS design in an attempt to harmonize such widely disparate perspectives. We feel that our design rules form a common ground for discussion and agreement.
[5]See, for example, Kannan, V., et al., "Tools and Techniques of Quality Management: An Empirical Investigation of Their Impact on Performance," QMJ, Vol. 6, No. 3, 1999.
[6]See, for example, Russell, J. P., The Quality Audit Handbook, Second Edition, Milwaukee, WI: ASQ Quality Press, 2000.
[7]See, for example, "First Interpretations Stall Over Possible Lapse in Process," Quality Systems Update, Volume 11, Number 11, November 2001, at http://www.qsuonline.com.
[8]This observation is supported in part by data presented on p. 20 of Quality, (January 1997), which notes that, in general, larger companies have been registered longer than smaller companies.
[9]The Registered Company Directory, North America, 1999, McGraw-Hill Companies, publisher of Quality Systems Update, Columbus, OH (e-mail: <customer.service@mcgraw-hill.com>) lists the profiles of 84 accredited registrars and four unaccredited registrars in North America.
|
EAN: 2147483647
Pages: 155