I - Cutaneous Tissue
Editors: Mills, Stacey E.
Title: Histology for Pathologists, 3rd Edition
Copyright 2007 Lippincott Williams & Wilkins
> Table of Contents > I - Cutaneous Tissue > 1 - Normal Skin
function show_scrollbar() {}
1
Normal Skin
Min Li
Carlos D. Urmacher
Introduction
The skin accounts for about 15% of the total body weight and is the largest organ of the body. It is composed of three layers: (a) epidermis, (b) dermis and (c) the subcutaneous adipose tissue. Each component has its unique and complex structure and function (1,2,3), with variation according to age, gender, race, and anatomic location. Functions of the skin are extremely diverse. It serves as a mechanical barrier against external physical, chemical, and biological noxious substances and as an immunologic organ. It participates in body temperature and electrolyte regulation. It is an important organ of sensuality and psychological well-being. In addition, it is a vehicle that expresses not only primary diseases of the skin, but also diseases of the internal organs. An understanding of the skin's normal histology is essential to the understanding of pathologic conditions.
Embryology
Epidermis
Embryologically, the ectoderm gives rise to epidermis and its appendages. The mesoderm provides the mesenchymal elements of the dermis and subcutaneous fat (4,5).
At first, the embryo is covered by a single layer of ectodermal cells. By the sixth to eighth week of development, it differentiates into two layers, the basal layer and an overlying second layer called periderm. The surface of the periderm is covered by microvilli and is in contact with the amniotic fluid. The mitotic activity of the basal layer predominates over that of the periderm, and soon the basal layer becomes the germinative layer. Additional rows of cells develop from this proliferating layer, forming a multilayer of cells between ectoderm and periderm (5). By the
P.4
twenty-third week, keratinization has taken place in the upper stratum, and the cells of the periderm have already been shed (5,6,7). Interestingly, it has been demonstrated that many of the cell junction proteins are expressed in the early two-layered embryonic epidermis and as early as the eighth week of estimated gestational age (8). By the end of the first trimester, the dermal epidermal junction with its component is ultrastructually similar to that of mature skin (9). Thus, the characteristic neonatal epidermis is well developed by the fourth month.
The majority of cells in the epidermis are keratinocytes (90 to 95%). The rest of the epidermal cells are nonkeratinocytes (5 to 10%), and they include melanocytes, Langerhans cells, and Merkel cells. The nonkeratinocytes are seen in the epidermis of 8- to10-week-old embryos. The precursor cells of melanocytes migrate from the neural crest to the dermis and then to the epidermis, where they differentiate into melanocytes during the first three months of development. During this migration, melanocytes can reside in other organs and tissue. Ultrastructually, recognizable melanosomes in melanocytes may be seen in the fetal epidermis at 8 to10 weeks of gestational age (10).
Langerhans cells are derived from the CD34+ hematopoietic precursor cell of the bone marrow. The characteristic cytoplasmic marker, the Birbeck granule, is seen ultrastructurally in 10-week-old embryos (11). The expression of a more characteristic immunohistochemical marker, CD1a is completed by 12 to 13 weeks of estimated gestational age (12,13).
Merkel cells can also be seen in the epidermis of 8- to 10-week-old embryos. The origin of Merkel cells is debatable. Some have suggested a neural crest derivation (14), whereas others suggest epidermal origin through a process of differentiation from neighboring keratinocytes (15,16,17,18). Merkel cells in the epidermis are initially numerous and later diminish with increasing gestational age, which suggests their role in growth and development (19).
Dermis
The dermis is derived from the primitive mesenchyme underlying the surface ectoderm. The papillary and reticular dermis is recognized by 15 weeks of intrauterine life (20,21).
As described by Breathnach (20), three types of cells are recognized in 6- to 14-week old embryos. Type I cells are stellate-dendritic cells with long slender processes. These are the most numerous primitive mesenchymal cells and probably give rise to the endothelial cells and the pericytes. Type II cells have less extensive cell processes; the nucleus is round, and the cytoplasm contains large vacuoles. They are classified as phagocytic macrophages of yolk-sac origin. Type III cells are round with little or no membrane extension, but they contain numerous vesicles, some with an internal content suggestive of granule-secretory type of cells. These cells could be melanoblasts on their way to the epidermis, or they could be precursors of mast cells; Schwann cells associated with neuroaxons, but lacking basal lamina, are also identified during this period.
The type II mesenchymal cells are rarely seen after week 14 of development. However, another cell type with ultrastructure of histiocyte or macrophage is frequently seen during this time. Well-formed mast cells are also seen in the dermis.
In 14 to 21 weeks of development, fibroblasts are numerous and active. Fibroblasts are recognized as elongated spindle cells with abundant rough endoplasmic reticulum. They are the fundamental cell of the dermis and synthesize all types of fibers and ground substance (1). Type III collagen fibers are abundantly present in the matrix of fetus, whereas type I collagen fibers are more prominent in adult skin (21). Elastic fibers appear in the dermis after the collagen fiber during the twenty-second week of gestational age; and, by week 32, a well-developed network of elastic fiber is formed in the dermis (5).
Initially, the dermis is organized into somites, but soon this segmental organization ends and the dermis of the head and neck and extremities organizes into dermatomes along the segmental nerves that are being formed (22). From the twenty-fourth week to term, fat cells develop in the subcutaneous tissue from the primitive mesenchymal cells.
Epithelial Skin Appendages
Most epithelial cells of skin appendages derive from follicular epithelial stem cells localized in the basal layer of epidermis at the prominent bulge region of the developing human fetal hair follicles. Furthermore, such multipotent stem cells may represent the ultimate epidermal stem cell (23). In 10-week-old embryos, a group of mesenchymal cells of the developing dermis aggregate beneath a budding group of tightly packed basal cells (24). These epidermal cells grow both downward to the dermis and upward through the epidermis to form the opening of the hair canal. As the growing epithelial cells reach the subcutaneous fat, the lower portion becomes bulbous and partially encloses the mesenchymal cells, descending with them to form the dermal papillae of the hair follicle. The descending epidermal cells around the dermal papillae constitute the matrix cells from which the hair layers and inner root sheath will develop. The outer root sheath derives from downward growth of the epidermis. The first hairs appear by the end of the third gestational month as lanugo hair around the eyebrow and the upper lip. The lanugo hair is shed around the time of birth. The developing hair follicle gives rise to the sebaceous and apocrine glands.
The sebaceous glands originate as epithelial buds from the outer root sheath of the hair follicles and are
P.5
developed at approximately the thirteenth to fifteenth gestational week (25). Differentiated sebaceous gland with a hair protruding through the skin surface are present at the eighteenth week of gestational age (26). They respond to maternal hormones and are well developed at the time of birth.
The apocrine glands also develop as epithelial buds from the outer sheath of the hair follicles in 5- to 6-month-old fetuses (22,24) and continue into late embryonic life as long as new hair follicles develop.
The eccrine glands develop from the fetal epidermis independent of the hair follicles (22). Initially, they are seen as regularly spaced undulations of the basal layer. At 14 to 15 weeks, the tips of the primordial eccrine glands have reached the deep dermis, forming the eccrine coils (27). At the same time, the eccrine epithelium grows upward into the epidermis. The primordial eccrine epithelium acquires a lumen by the seventh to eighth fetal month, and thus the first eccrine unit is formed. Both ducts and secretory portions are lined by two layers of cells. The two layers in the secretory segment undergo further differentiation; the luminal cells into tall columnar secretory cells, and the basal layer into secretory cells or myoepithelial cells (5). The first glands are formed on the palms and soles by the fourth month, then in the axillae in the fifth month, and finally on the rest of the hairy skin (28).
Physiology
Epidermis
The skin not only serves as a physical barrier between internal organs and the environment, but also functions as an active immune organ (29). Langerhans cells, the antigen-presenting cells of the epidermis, function as immunologic cells by recognizing antigens on the skin and presenting them to naive T lymphocytes. They become mature after contact with the antigen (30). Melanocytes produce melanin from the substrate tyrosine, using tyrosinase, and store it in melanosomes. Mature melanosomes are then transported to adjacent keratinocytes, where they provide protection against the harmful effects of solar radiation.
The keratinocytes are responsible for the process of keratinization. The formation of keratin filaments, in association with desmosomes, hemidesmosomes, and the basement membrane provides the structural integrity of the epidermis (31). Keratinocytes produce immunologic molecules, such as interleukins, interferons, and growth factors (32). It is recognized that the epidermal keratinocytes have immune properties.
Different types of keratin intermediate filaments are expressed in fetal and adult skin, and this process is, in part, regulated by apoptosis.
Apoptosis
Apoptosis, or programmed cell death, is the mechanism by which cells are deleted in normal tissue (33) and is the process responsible in establishing the final normal architecture of adult skin (34).
Terminal differentiation of the epidermis into a stratified squamous layer can be considered a specialized form of apoptosis (34). Apoptosis also participates in the cycling of the hair follicle (35,36,37) and is the principal mechanism by which catagen hair is formed (38,39,40). The bcl-2 proto-oncogen is a protein that blocks apoptosis and is expressed in basal cell keratinocytes and in the dermal papillae, protecting the latter from apoptosis (38).
Apoptosis affects individual cells not groups of cells, as in necrosis (33). The basic morphologic changes include fragmentation of the nucleus, chromatin compaction, and budding of the cells to produce membrane-bound apoptotic bodies, which are ingested by neighboring cells. No inflammation is seen with the process of apoptosis (33).
By light microscopy, apoptotic cells are seen as isolated cells with bright eosinophilic cytoplasms and dark, pyknotic and fragmented nuclei (Figure 1.1).
In routine hematoxylin-eosin (H&E) stained sections, apoptotic bodies are seen in a large variety of inflammatory and neoplastic diseases, such as graft-versus-host disease, lichen planus, erythema multiforme, squamous carcinoma, and malignant melanoma.
Recent study suggests that the mitogen-activated protein kinase signal transduction pathways are important in regulating the balance between keratinocyte cell proliferation, survival, apoptosis, and cell differentiation. Furthermore, it is suggested that extracellular regulated kinases induce keratinocyte proliferation and survival, whereas p38
P.6
mitogen-activated protein kinase promote differentiation and apoptosis (41).
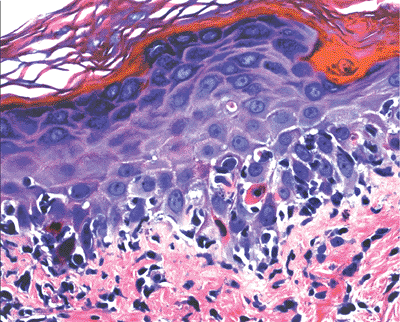 |
Figure 1.1 Apoptotic cell in a case of erythema multiforme. Note the eosinophilic cytoplasm and condensed nucleus. |
Dermis
The dermis is a supportive, connective tissue composed of cells, fibrous molecules, and ground substance (1). The mesenchymal component of dense fibrous connective tissue provides the mechanical support, rigidity, and thickness to the skin. Collagenous and elastic fibers are closely associated with each other in the dermis. Collagen presumably provides the skin with tensile strength, whereas elastin posseses the retractile properties of the skin (42). The dermis also has immunologic functions because it contains dendritic cells, lymphocytes, other migrant leukocytes, mast cells, and tissue macrophages. Mast cells react to inflammatory process and also participate in wound healing.
Except for the epidermis, the skin is rich in a vascular network. In addition to providing nutrients to the skin, blood vessels are involved in thermal regulation, wound healing, immune response, and control of blood pressure. The lymphatic system is important in regulating the pressure of interstitial fluid (1).
The skin is supplied with autonomic nerves and sensory nerves. Small and large nerve plexuses participate in the innervations, which are responsible for the detection of touch, pressure, vibration, pain, temperature, and itching, as well as sweat secretion and piloerection.
Eccrine and Apocrine Glands
The most important function of the eccrine glands is in the processes of thermoregulation and electrolytic balance (43). The eccrine glands are the true sweat glands, and their function begins in the neonatal period. Eccrine glands produce colorless and odorless hypotonic sweat composed of predominantly water and the same electrolytes that are present in the plasma. There are two types of secretory cells, clear and dark cells. The clear cells of the eccrine coil, responding predominantly to cholinergic stimuli, and to a lesser degree to sympathetic stimulation (44,45,46), produce an isotonic sweat. When it reaches the duct, sodium and chloride ions are reabsorbed, delivering a hypotonic solution to the surface. The function of the dark cells is still not known with certainty. It has been suggested that they permit reabsorption of sodium, potassium, and chloride (24) and may secrete sialomucin (47) to the sweat.
In addition, the eccrine duct has the important function of delivering parenteral or orally administered drugs to the surface of the skin (48). Ductal epithelium also participates in the process of wound healing (22).
The major function of the myoepithelial cells is mechanical support against a high hydrostatic pressure. The contraction of myoepithelial cells aids in delivery of sweat to the skin surface (49).
The exact role of apocrine glands and the mechanisms regulating apocrine secretory process in humans are still under investigation. Apocrine glands might also play a role in thermal regulation (50,51). In nonhuman mammals, apocrine glands are found over the entire skin surface; they are believed to serve as identifying or sexual organs (24). Apocrine secretion has a milky color and is sterile and odorless; however, when it reaches the surface of the skin, the action of regional microorganisms on the apocrine secretion makes it odorous (24). The most abundant odor component, known to be E-3-methyl-2-hexanoic acid (E-3M2H), is liberated from nonodorous apocrine secretion by microorganisms (52).
Alteration in the rate and amount of sweat secretion manifests as anhidrosis, hypohidrosis, and hyperhidrosis (53,54). Cystic fibrosis (44,49) is the disorder with alterations in the electrolyte composition of eccrine sweat. Few morphologic changes are seen in association with these diseases.
Light Microscopy
Epidermis
The epidermis is a stratified keratinizing squamous epithelium that dynamically renews itself but maintains its normal thickness by the process of desquamation. The cells in the epidermis include: (a) keratinocytes, (b) melanocytes, (c) Langerhans cells, and (d) Merkel cells. In addition, the epidermis contains the openings for the eccrine ducts (acrosyringium) and hair follicles. Recent immunohistochemical studies have demonstrated that the epidermis contains free nerve axons in association with Langerhans cells (55).
Keratinocytes
The keratinocytes of the epidermis are stratified into four orderly layers from bottom to top: (a) the basal layer (stratum basalis, germinativum); (b) the squamous layer (prickle cell layer, or stratum spinosum); (c) the granular layer (stratum granulosum); (d) the cornified, or horny, layer (stratum corneum) (Figure 1.2). In histologic sections, the dermoepidermal junction has an irregular contour because of the upward extension of the papillary dermis to form the dermal papillae. The portion on the epidermis separating the dermal papillae are the rete ridges (Figure 1.3).
The Basal Layer
Basal cells are the mitotically active cells that give rise to the other keratinocytes. In histologic sections, basal cells are seen as a single layer of cells above the basement membrane that show some variation in size, shape, and melanin
P.7
content. Basal cells are columnar or cuboidal, with a basophilic cytoplasm. The nucleus is round or oval, with coarse chromatin and indistinct nucleolus. Basal cells contain melanin in their cytoplasm as a result of pigment transfer from neighboring melanocytes. Basal cells are connected to each other and to keratinocytes by specialized regions (known as desmosomes) located in the plasma cell membranes. They are aligned perpendicular to the subepidermal basement membrane and attached to it by modified desmosomes, hemidesmosomes (1).
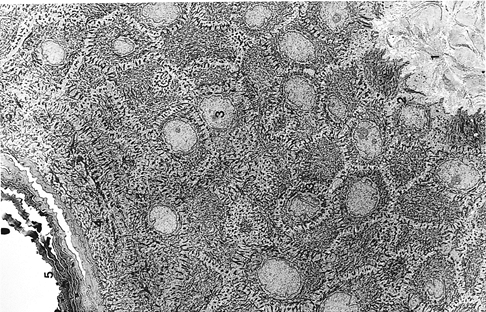 |
Figure 1.2 Electron micrograph of normal epidermis and portion of papillary dermis ( 2,100). (1, papillary dermis; 2, basal cells; 3, squamous layer; 4, granular layer; 5, cornified layer) |
Dermatitis involving the basal layer produces vacuolar alteration of the basal cells, which may progress to the formation of subepidermal vesicles as seen in diseases such as graft-versus-host disease, lupus erythematosus, and erythema multiforme.
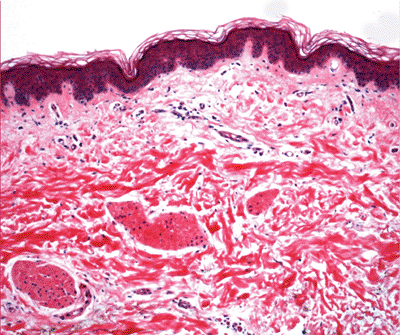 |
Figure 1.3 Normal skin showing stratified epidermis with rete ridges, papillary dermis and reticular dermis (H&E). |
The Squamous Layer
The squamous layers are composed of approximately five to ten layers of cells with keratinocytes larger than the basal cells. The suprabasal keratinocytes are polyhedral, have a somewhat basophilic cytoplasm, and with a round nucleus. Again, melanin is seen scattered in many of these keratinocytes, where it provides protection from the damaging effect of ultraviolet light. The more superficial cells are larger, flattened, eosinophilic, and oriented parallel to the surface. The keratinocytes contain one or two conspicuous nucleoli and tonofilaments within the cytoplasm.
The squamous layer is also called the spinous or prickle cell layer because of the characteristic appearance by light microscopy of short projections extending from cell to cell. These projections are the result of retraction of the plasma membrane during tissue processing whereas the desmosomes remain relatively fixed and correlate with intercellular bridges.
Desmosomes are composed of a variety of polypeptides; desmogleins and desmocollins as transmembrane constituents and the desmoplakin, plakoglobin and plakophilin as cytoplasmic components (5). In addition, other intercellular junctions (such as gap junctions and adherens junctions) are distinct from desmosomes in composition and distribution and provide alternative cell-to-cell adhesion mechanisms (56). An intercellular space of constant dimension (57) is present between each cell; acid and neutral mucopolysaccharides are present in the intercellular spaces as indicated by special stains (5). The pemphigus antigens are localized in the cell membranes (58) or in the
P.8
desmosomes of these cells (59). Antibodies to desmosomal proteins are used as additional markers for the study of neoplasm (60).
It is important to recognize that occasionally cells with clear or pale cytoplasms are seen in the squamous layer. These cells must be distinguished from the neoplastic cells of Paget's disease. Benign clear cells have a pyknotic nucleus surrounded by a clear halo and a narrow rim of clear cytoplasm (Figure 1.4). They lack the pleiomorphism, nuclear morphology, and intensity of the chromatin staining seen in Paget's cells (Figure 1.5). These benign clear cells are often seen in the epidermis of the nipple, the accessory nipple (61,62), and the pubic regions or in the milk line distribution (63). In the nipple, these clear cells, also called Toker cells, have been considered to be nonneoplastic mammary elements (61), although some authors hypothesized that these cells might be the precursors of mammary or extramammary Paget's diseases (62,64). Those outside of the nipple are considered to be the result of either abnormal keratinization or aberrant derivatives of eccrine or apocrine sweat gland epithelial cells (65,66,67). They may present as hypopigmented macules or papules in a rare disorder called clear cell papulosis (63,67). The immunohistochemical and mucin staining pattern of benign clear cells may resemble that of Paget's cells. Therefore, they must be distinguished on a morphologic basis from the neoplastic cells.
Common inflammatory changes seen in the squamous layer are: (a) spongiosis intercellular edema (e.g., allergic contact dermatitis); (b) acanthosis thickening of the epidermis (e.g., psoriasis); (c) atrophy thinning of the epidermis (e.g., discoid lupus erythematosus); (d) acantholysis detachment of keratinocytes because of changes involving intercellular junctions (e.g., pemphigus); and (e) dyskeratosis abnormal keratinization (e.g., squamous carcinoma).
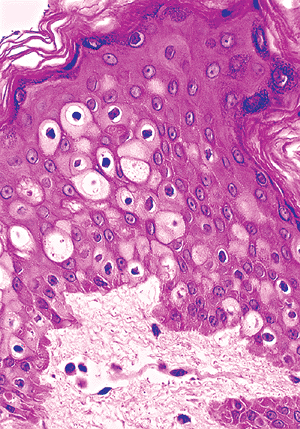 |
Figure 1.4 Clear cells of the nipple epidermis. |
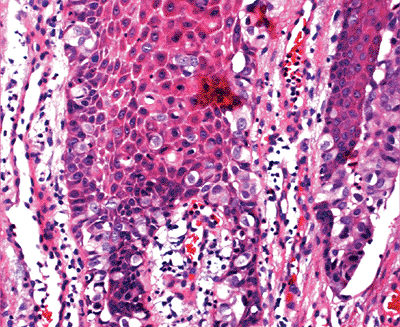 |
Figure 1.5 Paget's cells in extramammary Paget's disease. |
The Granular Layer
The granular layer is composed of one to three layers of flattened cells lying parallel to the skin surface. The cytoplasm contains intensely basophilic-stained granules known as the keratohyalin granules. In contrast, trichohyalin granules (produced by the inner root sheath of hair follicles) are stained red on routine H&E-stained sections. The keratohyalin granules are histidine-rich and are the precursors to the protein flaggrin, which promotes aggregation of keratin filaments in the cornified layer. The granular layer is rich in lysosomal enzyme, which is crucial for the autolytic changes in the granular layer (68). The increase (e.g., lichen planus) and decrease (e.g., psoriasis) in the thickness of the granular layer can be used as a clue in the diagnosis of different pathologic entities.
Keratinocytes located between the squamous layer and the granular layer contain small membrane-coating granules known as lamellar granules (also called Odland bodies or keratosomes. They are composed of the acid hydrolase and of neutral sugars conjugated with proteins and lipids. These granules are present both intra- and extracellularly, are approximately 300 nm in diameter, and are not visible by light microscopy. Their functions are to provide epidermal lipids, increase the barrier property of the cornified layer against water loss, and aid in the desquamation process. This interface between the squamous and granular layer is also the site of synthesis and storage of cholesterol (57,69).
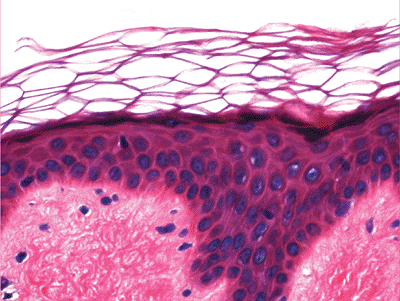 |
Figure 1.6 Basket-weave pattern of the cornified layer (also in Figure 1.3). |
P.9
The Cornified Layer
The cornified layer is composed of multiple layers of polyhedral eosinophilic keratinocytes that lack a nucleus and cytoplasmic organelles. These cells are the most differentiated cells of the keratinization system. They are composed entirely of high-molecular weight keratin filaments. In formalin-fixed section, the cornified layers are arranged in a basket-weave pattern (Figure 1.6). These cells eventually shed from the surface of the skin. The process of keratinization takes 20 to 45 days.
In histologic sections taken from the skin of the palms and soles, a homogenous eosinophilic zone, known as the stratum lucidum is present in the lowest portion of the cornified layer (above the granular layer). This additional layer is rich in protein-bound lipids contained in the lamellar granules (5), energetic enzymes and SH groups secreted by the granular cells in molecular structure (70).
Common abnormalities of the cornified layer are: (a) hyperkeratosis increased thickness in the cornified layer (e.g., ichythyosis); (b) parakeratosis presence of nuclei in the cornified layer (as usually seen in actinic keratosis); and (c) presence of fungal organisms (superficial dermatophytosis).
Basement Membrane Zone
The basement membrane zone separates the epidermal basal layer from the dermis. It is seen by light microscopy as a continuous, undulating and thin periodic acid-Schiff (PAS)-stained layer (Figure 1.7). By electron microscopy, the basal cells are attached to the basal lamina by hemidesmosomes (57). Ultrastructurally, the basement membrane zone is composed of four distinct structures, from top to bottom (Figure 1.8) (57,71):
The plasma membrane of the basal cells containing the hemidesmosomes. Bullous pemphigoid antigen 1 is localized in the intracellular component of hemidesmosomes.
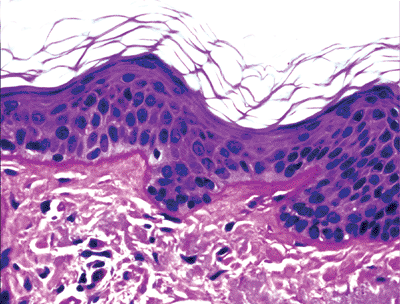
Figure 1.7 PAS-positive basement membrane.
The lamina lucida, an electron-lucent area with anchoring filaments containing various laminin isoforms (1,72). Bullous pemphigoid antigen 2 (type XVII collagen) is associated with the transmembrane component of hemidesmosome-anchoring filament complexes in
P.10
the lamina lucida. It is also the site of the blister in dermatitis herpetiformis (73).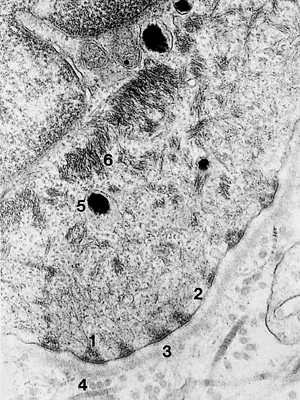
Figure 1.8 Ultrastructure of basement membrane ( 37,800). (1, hemidesmosome; 2, lamina lucida; 3, lamina densa; 4, lamina reticularis; 5, melanin; 6, tonofilaments)
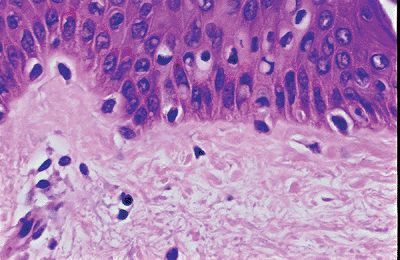
Figure 1.9 Melanocytes in the basal layer, composed of ovoid nuclei within a clear space.
The lamina densa, an electron-dense area composed of mainly type IV collagen.
The sublamina densa zone, or pars fibroreticularis, contains mainly the anchoring fibrils (74) (type VII collagen) that attach the basal lamina to the connective tissue of the dermis. Antibodies against epidermolysis bullosa aquisita react with the carboxy terminus of type VII collagen (75,76).
Inflammatory conditions of the basement membrane can be seen by light microscopy as thickening (e.g., discoid lupus erythematosus) or by the formation of subepidermal vesicles (e.g., bullous pemphigoid).
Melanocytes
Melanocytes are dendritic cells that derive from the neural crest. During migration from the neural crest, melanocytes may localize in other epithelia. In the epidermis, the melanocytes are localized in the basal layer, and their dendritic processes extend in all directions. The dendritic nature of normal melanocytes is usually not seen in routine H&E-stained sections. In H&E preparations, melanocytes are composed of elongated or ovoid nuclei surrounded by a clear space (Figure 1.9). They are usually smaller than the neighboring basal keratinocytes. Melanocytes do not contain tonofilaments and do not attach to basal cells with desmosomes (5,77). However, anchoring filaments extend from the plasma membrane of these melanocytes to the basal lamina. Laminin-5, a component of anchoring filaments, may be a ligand for melanocyte attachment to the basement membrane in vivo (78). In addition, melanocytes that are close to the basal lamina have structures resembling hemidesmosomes of basal keratinocytes (79).
Melanocytes produce and secrete melanin. Melanin can be red (pheomelanin) or yellow-black (eumelanin) (80,81). The most important function of melanin is to protect against the injurious effects of non-ionizing ultraviolet irradiation.
Melanin is formed through a complex metabolic process in which tyrosinase is the main catabolic enzyme, using tyrosine as substrate. The synthesis of melanin takes place in melanosomes, lysosome-related organelles. In the early stages of development, melanosomes are membrane-limited vesicles, located in the Golgi-associated endoplasmic reticulum. The maturation of melanosomes undergoes four stages. Stage I melanosomes are round without melanin. These are seen in balloon cell melanoma. Stage II through stage IV melanosomes are ellipsoidal with numerous longitudinal filaments (5). Melanin deposits start at stage II. In stage III, melanin deposits are prominent. Stage IV melanosomes are fully-packed, with melanin obscuring the internal structures.
The developing melanosomes, with their content of melanin, are transferred to the neighboring basal keratinocytes and hair follicular cells. The mechanism of melanin transfer is a complex process (82,83), with the end result being phagocytosis of the tip of melanocytic dendrites by the keratinocytes (Figure 1.10) in a process called pigment donation (84). The epidermal melanin unit refers to one melanocyte with associated 36 keratinocytes to which the melanocytes deliver melanosomes (1).
The number of melanocytes in normal skin is constant in all races, the ratio being one melanocyte for every 4 to 10 basal keratinocytes (77,80). Thus, the color of the skin is determined by the number and size of melanosomes present both in keratinocytes and melanocytes and not by the number of melanocytes. The number of melanocytes decreases with age. As a result, the availability of melanin to keratinocytes diminishes, so the skin becomes lighter in color and the incidence of skin cancer increases.
Melanin is both argentaffin and argyrophilic. It can be recognized by Fontana-Masson silver stains. In addition,
P.11
melanocytes and their dendritic processes are identified by the dopa reaction in histologic slides prepared from frozen sections and in paraffin-embedded sections with immunohistochemical stains with S-100 protein. The latter is highly sensitive but not specific for cells of melanocytic lineage. The S-100 protein can be detected in various types of cells, such as Langerhans cells, schwann cells, eccrine, and apocrine gland cells. Melanocytes can also be identified with monoclonal antibodies to Melan A/MART-1 (Melanoma Antigens Recognized by T cells-1), a melanocytic differentiation marker. The MART-1 antigen is expressed in normal melanocytes, common nevi, Spitz nevi, and malignant melanoma (5). Under normal conditions, the melanoma-associated antigen HMB-45 does not react with adult melanocytes (85). It is expressed in embryonic melanocytes, hair bulb melanocytes and activated melanocytes (86). It is usually seen reacting with most melanoma cells, Spitz nevi, the junctional component of common nevi, and dysplastic nevi.
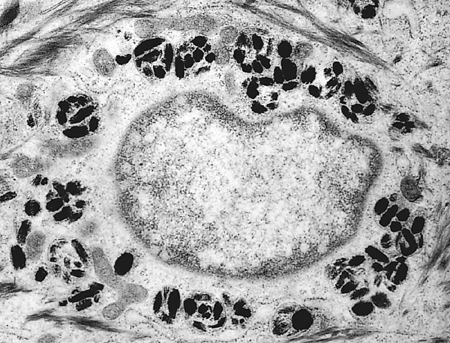 |
Figure 1.10 Electron micrograph showing membrane-bound phagocytized melanin in keratinocyte ( 19,200). |
A decrease or absent number of melanocytes is seen in vitiligo. In albinism, there is a defect in the synthesis of melanin, but the number of melanocytes is normal in a skin biopsy. Melanocytic hyperplasia is seen in lentigo, benign and malignant melanocytic neoplasm, and as a reaction pattern in a variety of neoplastic and nonneoplastic conditions (e.g., dermatofibroma). In a freckle, there is an increase in pigment donation to adjacent keratinocytes rather than melanocytic hyperplasia (84).
Langerhans Cells (LCs)
Langerhans cells (LCs), discovered by Paul Langerhans in 1868, are mobile, dendritic, antigen-presenting cells present in all stratified epithelium and predominantly in the mid to upper part of the squamous layer. In H&E-stained sections, LCs can be suggested as they appear to lie within lacunae having darkly stained nuclei with indented, reniform shape at high magnification (Figure 1.11). As with melanocytes, their dendritic nature cannot be seen in routine sections. Langerhans cells can be recognized by histoenzymatic stains for adenosine triphosphatase (ATPase); they can also be detected in formalin-fixed, paraffin-embedded tissue using immunoreactivity for S-100 protein and, more specifically, the antibody to the CD1a antigen (Figure 1.12). With histoenzymatic and immunohistochemical stains, the extensive dendritic nature of LCs becomes evident.
By electron microscopy, LCs show no desmosomes, tonofilaments, or melanosomes. They contain small vesicles, multivesicular bodies, lysosomes, and the characteristic Birbeck granule (Figure 1.13) (87), a rod-shape organelle varying in size from 100 nm to1 m (88). It has a centrally striated density and an occasional bulb at one end with a unique tennis racquet appearance. Their function is still undetermined. Langerin is a protein implicated in Birbeck granule biogenesis and represents a key molecule to trace LCs and study their function (89,90).
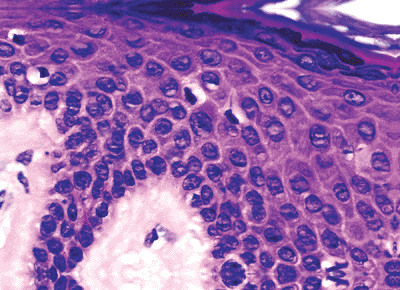 |
Figure 1.11 H&E section of possible Langerhans cells composed of elongated nuclei surrounded by a clear space in the mid-epidermis. |
Langerhans cells are also present in epithelia, lymphoid organs, and dermis and are increased in the skin in a variety of inflammatory conditions, such as contact dermatitis, where they can be seen as minute nodular aggregates in the epidermis. Langerhans' cell granulomatosis is a reactive lesion most commonly seen in bones but also appearing at other sites.
Merkel Cells (MCs)
Merkel cells (MCs), first described by F.S. Merkel in 1875, are scattered and irregularly distributed in the basal cell layer in the epidermis. They may group together in clusters coupled with enlarged terminal sensory nerve fibers to form slowly adapting mechanoreceptors; within the epidermis, they mediate tactile sensation (91,92,93). They are
P.12
located in higher concentration in the glabrous skin of the digits, lips, and oral cavity (9), in the outer root sheath of hair follicles (94), and in the tactile hair disks (95).
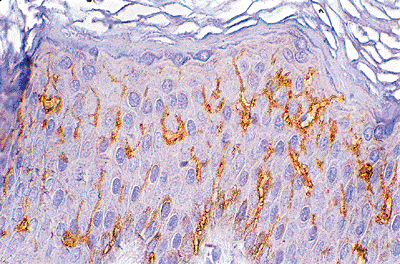 |
Figure 1.12 CD1a-specific reaction of Langerhans cells. Note the dendritic processes. |
Merkel cells are not recognized in routine histologic preparations. Electron microscopy and immunostaining are required for their identification. By electron microscopy, MCs are attached to adjacent keratinocytes by desmosomes. They have scant cytoplasms, invaginated nuclei, a parallel array of cytokeratin filaments in the paranuclear zone, and the characteristic membrane-bound dense core granules that are often, but not always, related to unmyelinated neurites.
By immunostaining techniques, normal and neoplastic MCs may express neuron-specific enolase, chromogranin, synaptophysin, neural cell adhesion molecule, and various neuropeptides and other substances (96,97,98). However, the expression of these substances in MCs is heterogenous and variable. The constant pattern seen in MCs is the presence of paranuclear aggregates of cytokeratins (16,98,99), which include low-molecular weight keratins 8, 18, 19, and 20. The most specific cytokeratin is CK20 because, in addition to MCs, they are expressed in simple epithelial cells and not in adjacent keratinocytes (100,101) (Figure 1.14).
Pilar Unit
The pilar unit is composed of the hair follicle, sebaceous gland, arrector pili muscle, and (when present) eccrine gland and apocrine gland.
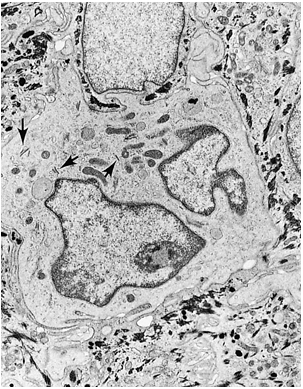 |
Figure 1.13 Electron micrograph of a Langerhans cell containing Birbeck granules (arrows) and multisegmented nucleus ( 8,000). |
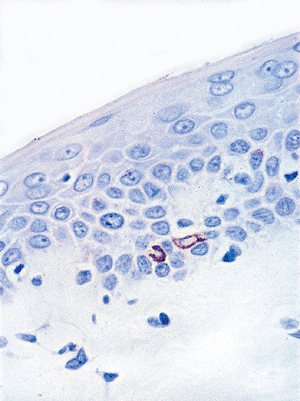 |
Figure 1.14 Cytokeratin-20 staining a Merkel cell in the basal layer of the epidermis. |
Hair Follicle
The hair follicle is divided into three segments from top to bottom: (a) the infundibulum, which extends from the opening of the hair follicle in the epidermis to the opening of the sebaceous duct; (b) the isthmus, which extends from the opening of the sebaceous duct to the insertion of the arrector pili muscle; and (c) the inferior segment, which extends to the base of the follicle. The inferior segment is bulbous and encloses a vascularized component of the dermis referred to as follicular (dermal) papilla of the hair follicle (Figure 1.15).
The microanatomy and function of the hair follicle is very complex. The cells of the hair matrix differentiate along six cell linings. Beginning from the innermost layer, they are: (a) the hair medulla; (b) hair cortex; (c) hair cuticle; and (d) three concentric layers of the inner root sheath, which are the cuticle of the inner root sheath, Hexley's layer, and Henle's layer.
The inner root sheath of the hair follicle is surrounded by the outer root sheath (Figure 1.16), which is composed of clear cells. These glycogen-rich cells are seen in some of the neoplasm with hair follicular differentiation (e.g., trichilemmoma). A PAS-positive basement membrane separates the outer root sheath from the surrounding connective tissue. Thus, the hair shaft is formed from the bulb region that occupies the hair follicular canal.
Dendritic melanocytes are present only in the upper half of the bulb, whereas inactive (amelanotic) melanocytes are present in the outer root sheath. These melanocytes can become active after injury, migrating into the upper portion of the outer root sheath and to the regenerating epidermis (5).
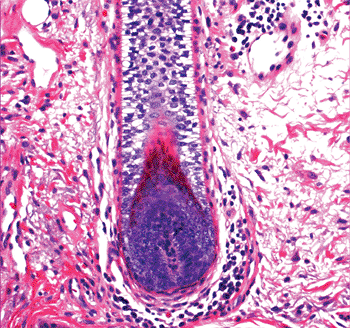 |
Figure 1.15 Inferior segment of the hair follicle, showing the hair papilla. |
P.13
At the level of the isthmus, the cells of the inner root sheath disintegrate and disappear, whereas the cells of the outer root sheath begin an abrupt sequence of keratinization. This process is called trichilemmal keratinization (102). Trichohyalin granules are red in routine H&E-stained sections, as opposed to the blue granules of the keratohyalin of epidermal keratinization and of the epithelium of the follicular infundibulum of the hair follicle. The staining features of these granules permit neoplasms and cysts to be distinguished from either pilar or epidermal origin.
Under normal circumstances, microorganisms like Staphylococcus epidermis, yeasts of Pityrosporum (Figure 1.17), and the Demodex folliculorum (Figure 1.18) mites are encountered in the follicular infundibulum.
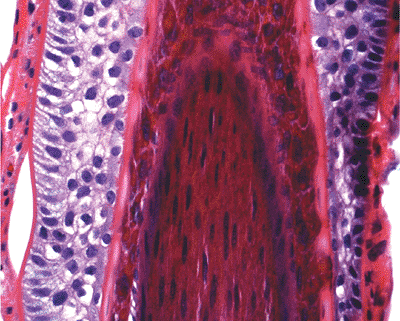 |
Figure 1.16 Hair follicle showing the hair shaft (center) surrounded by the inner root sheath, which contains trichohyalin granules. The outer root sheath is composed of clear cells. |
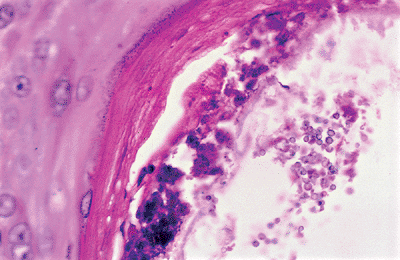 |
Figure 1.17 Yeasts of Pityrosporum in the follicular infundibulum. |
The mantle hair of Pinkus (103) is a hair follicle in which proliferation of basaloid epithelioid cells emanating from the infundibulum is seen. Sebaceous proliferation is present in those cords (Figure 1.19). The significance of this hair follicle is not known.
The hair growth is in lifelong cyclic transformation. Hormones and their receptors play prominent roles in hair cycle regulation (104). Three phases are recognized: (a) anagen active growth phase; (b) catagen involuting phase (apoptosis-driven regression); and (c) telogen relative resting phase. The histologic features previously described correspond to the anagen hair.
During the catagen phase, mitosis and melanin synthesis cease at the level of the hair bulb. The hair bulb is then replaced by a cornified sac formed by retraction of the outer root sheath around the hair bulb, and a club hair is formed. A thick glassy basement membrane surrounds the hair follicle. Apoptosis of single cells in the outer root sheath is a characteristic finding during the catagen phase (40).
During the telogen phase, the club hair and its cornified sac retract even further to the insertion of the arrector pili muscle, leaving behind the dermal papilla, which is
P.14
connected to the retracted hair follicle by a fibrous tract (Figure 1.20) (24). When the cycle is complete, a new anagen phase begins with the formation of new hair matrix.
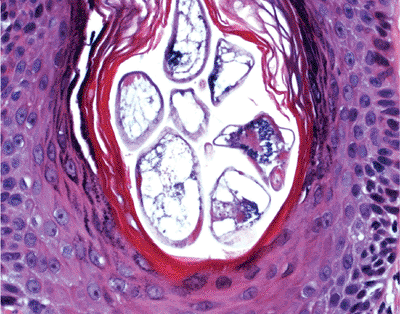 |
Figure 1.18 Demodex folliculorum mites in the follicular infundibulum. |
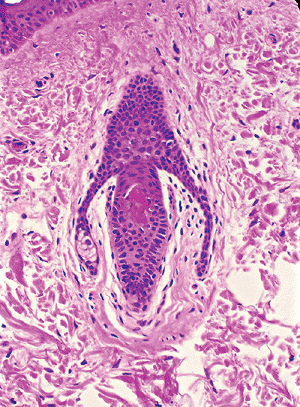 |
Figure 1.19 Mantle hair of Pinkus with lateral extensions containing sebaceous cells. |
The duration of the normal hair cycle varies. The anagen phase is measured in years for the scalp, but it is measured in shorter periods of time for the anagen cycle in other regions of the body. The length of the hair is also related to the amount of the anagen hair. More than 80% of hair present in normal scalp is anagen hair. The catagen phase takes two to three weeks, and the telogen phase may last a few months.
The color of normal hair depends on the amount and distribution of the melanin in the hair shaft (24). Normal human epidermal melanocytes may synthesize both eumelanin and pheomelanin (105). The melanins in black hair are eumelanin (characterized by the presence of ellipsoidal eumelanosomes), while those in red hair are mainly pheomelanin (ascribed to spherical pheomelanosomes) (105,106). Fewer melanosomes are produced in the bulbar melanocytes of blond hair. A relative absence of melanin and fewer melanosomes are seen in gray hair. Multiple internal or external regulatory factors are involved in hair pigmentation. There might be some correlation between tryptophan content and tyrosinase expression with hair color (107,108).
Another structure related to the pilar unit is the hair or pilar disk (the Haarscheibe). The Haarscheibe is a specialized spot in close vicinity to hairs. This structure is usually not recognized on routine histologic section. It may present as an acanthotic elevation of the epidermis, limited by two elongated rete ridges laterally (1). The epidermis in this area has more Merkel cells in the basal layer, and the dermal component is well vasculized, containing myelinized nerve fibers in contact with Merkel cells (22,95). It is considered as a highly sensitive, slowly adapting mechanoreceptor (1,109).
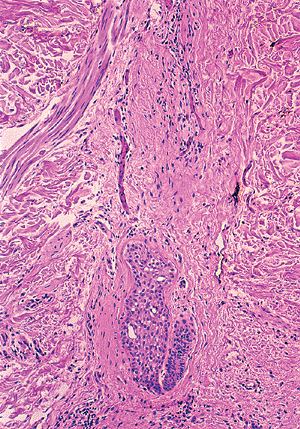 |
Figure 1.20 Catagen telogen hair follicle located entirely within the dermis. |
Sebaceous Glands
The sebaceous glands are holocrine glands associated with hair follicles. Their secretions are made of disintegrated cells. The palms and soles are the only regions devoid of sebaceous glands. Sebaceous glands are prominent in facial skin. They are also seen in the buccal mucosa, vermilion of the lip (Fordyce's spot), prepuce, labia minora, and, at times, in the parotid gland.
The sebaceous glands are lobulated structures composed of multiple acini in some locations like the head and neck; in other sites, such as chest, they are composed of a single acinus. The periphery of the lobules contains the germinative cells, which are cuboidal and flat with large nucleoli and basophilic cytoplasms without lipid droplets. As differentiation occurs, several inner layers show lipid droplet accumulation in the cytoplasm until they fill the cell.
The more differentiated cells (sebocytes) have a characteristic multivacuolated cytoplasm (Figure 1.21). The nucleus is centrally located and scalloped due to the lipid imprints. The more differentiated cells disintegrate and discharge the cellular debris (sebum) into the excretory duct, which opens into the hair follicle in the lower portion of the infundibulum. The excretory duct is short, shared by several lobules, and lined by keratinized squamous epithelium.
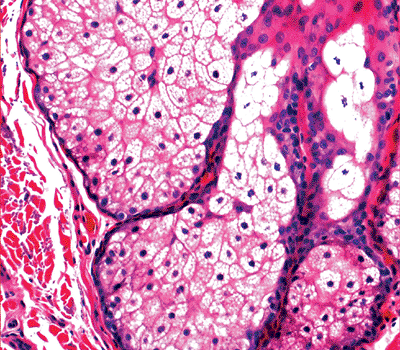 |
Figure 1.21 Sebaceous glands with peripheral germinative cells and, toward the center, the differentiated vacuolated cells. |
P.15
Within sebaceous glands, the germinative cells express appreciable quantities of keratins. Mature sebocytes demonstrate cytoplasmic reactivity for high-molecular weight keratins and epithelial membrane antigen (1).
Eccrine Glands
The eccrine glands are the true sweat glands responsible for thermoregulation. They are found in higher concentration in palms, soles, forehead, and axillae and have dual secretory and excretory functions.
The secretory portion of an eccrine gland is a convoluted tube located in the dermis, in the interface with the subcutaneous tissue, and, rarely, within the subcutaneous tissue. In cross-sections, it appears that several glandular structures with a central lumen form the secretory coils. These are seen as lobular structures often surrounded by fat even when located within the dermis (Figure 1.22).
Three types of cells are identified in the eccrine coil: clear cells, dark cells, and myoepithelial cells. The clear cells are easily seen H&E-stained sections (Figure 1.23). They rest directly on the basement membrane and on the myoepithelial cells. Clear cells are composed of pale or finely granular cytoplasms with a round nucleus usually seen in the center of the cell. Deep invaginations of the luminal membranes of adjacent clear cells form intercellular canaliculi lined with microvilli (Figure 1.24) (110). The intercellular canaliculi often persistent in neoplasms derived from eccrine glands. The clear cells contain abundant mitochondria and variable amount of PAS-positive, diastase-labile glycogen.
The dark cells border the lumen of the glands. Electron microscopy shows that they contain abundant secretory granules that have glycogen-staining characteristics. They contain sialomucin (PAS-positive, diastase-resistant (PASD) mucopolysaccharides) and high concentration of proteins (5,111). The dark cells are difficult to identify in routine H&E-stained sections. However, the acid-fast, PASD, and S-100 protein stains will highlight the granularity of the cells (Figure 1.25) (22).
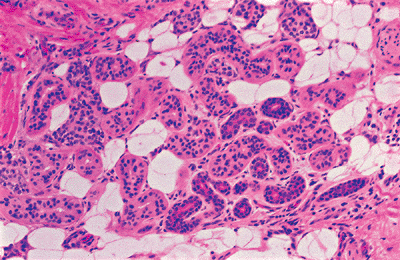 |
Figure 1.22 Eccrine lobule containing fat, glands, and ducts. |
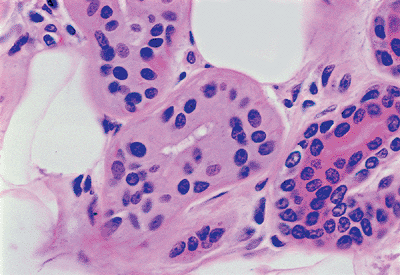 |
Figure 1.23 Clear cells of the eccrine glands. |
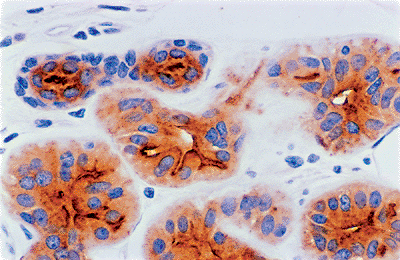 |
Figure 1.24 Intercellular canaliculi (anti-CEA). |
The myoepithelial cells are contractile spindle cells that surround the secretory coil (Figure 1.26). In turn, they are surrounded by a PAS-positive basement membrane. Elastic
P.16
fibers, fat, and small nerves are present in the adjacent stroma.
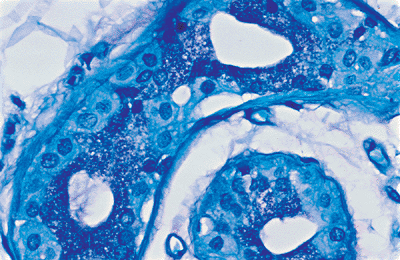 |
Figure 1.25 Dark cells with granular cytoplasm (acid-fast stain). |
The excretory component of the eccrine gland is composed of three segments: (a) a convoluted duct in close association with the secretory unit (Figure 1.27); (b) a straight dermal component; and (c) a spiral intraepidermal portion, the acrosyringium, which opens onto the skin surface (Figure 1.28). The transition between the secretory and excretory component is abrupt. Both convoluted and straight dermal ducts are histologically identical. They are narrow tubes with a slitlike lumina lined by double layers of cuboidal cells. The luminal cells have a more granular eosinophilic cytoplasm and a larger round nucleus than the peripheral row of cells. The peripheral cells are rich in mitochondria.
The luminal cells produce a layer of tonofilaments near the luminal membrane that are often referred to as the cuticular border, which is a PASD eosinophilic cuticle (5). This cuticular border often persists in the eccrine neoplasm (e.g., eccrine poroma). There are no myoepithelial cells and peripheral hyalin basement membrane zone in the eccrine ducts (5).
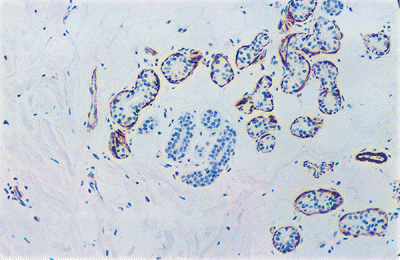 |
Figure 1.26 Glands, but not the ducts, are surrounded by myoepithelial cells (anti-HHF35). |
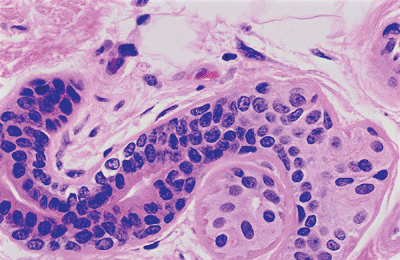 |
Figure 1.27 Eccrine duct. Note the abrupt transition from the secretory portion. |
The intraepidermal segment of eccrine duct, known as acrosyringium has a unique symmetrical and helicoidal course in the epidermis with its length correlated to the thickness of the epidermis (43). It consists of a single layer of luminal cells and two or three rows of concentrically oriented outer cells. The presence of keratohyalin granules in acrosyringium in the lower levels of the squamous layer indicates that they keratinize independently. The intraepidermal lumen is lined by acellular eosinophilic cuticle before keratinization (5,24). Melanin granules are absent.
Apocrine Glands
The apocrine gland (Figure 1.29) has a coiled secretory portion and an excretory (ductal) component. The secretory portion is much longer than its eccrine counterpart; and it may reach 200 m in diameter, compared to 20 m for the eccrine glands (77). The secretory glands are located in the
P.17
subcutaneous fat or in the deep dermis. They are lined by one layer of cuboidal, columnar, or flat cells (luminal cells), and an outer layer of myoepithelial cells, which is surrounded by a PAS-positive basement membrane. The luminal cells are composed of eosinophilic cytoplasm, which may contain lipid, iron, lipofuscin, PASD granules (24), and a large nucleus located near the base of the cell. Detached fragments of apical cytoplasm are found in the lumen of the glands. The secretion from apocrine glands releases secretory materials accompanied with loss of part of cytoplasm (112), although other forms of secretion have been observed, including melocrine (granular contents within numerous vesicles are released without loss of cytoplasm) and holocrine type (the entire cell is secreted into the glandular lumen) (50,112).
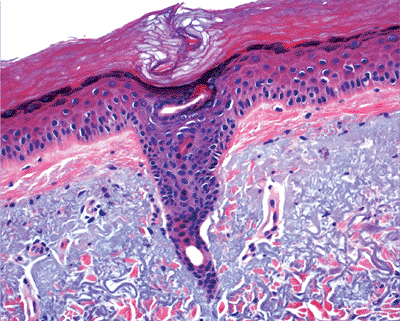 |
Figure 1.28 Acrosyringium. |
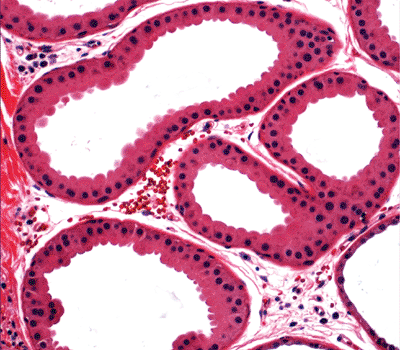 |
Figure 1.29 Secretory apocrine glands. |
Similar to the eccrine duct, the excretory (ductal) component of the apocrine gland has a double layer of cuboidal cells. Microvilli are identified on the surface of the luminal cells and keratin filaments are in their cytoplasms, the latter giving the eosinophilic hyalin appearance to the inner lining of the duct. No myoepithelial cells and peripheral basement membrane are identified in the excretory duct. Apocrine glands are always connected to a pilosebaceous follicle. The intrafollicular or intraepidermal portion of apocrine duct is straight other than the spiral as seen in acrosyringium (5).
Apocrine glands are mostly located in the axillary, anogenital areas, mammary region, eyelids (Moll's glands), and external ear canal (ceruminous glands), and their presence is characteristic in Nevus sebaceous Jadassohn.
A third type of sweat gland, so-called apoeccrine glands of the human axillae (113), are composed of a dilated secretory portion that, by electron microscopy, is indistinguishable from the apocrine glands; however, they retain the intercellular canaliculi, as well as the dark cells of the eccrine glands. The duct does not open in the hair follicle but in the epidermis. These glands, which develop from eccrine glands during puberty, account for as much as 45% of all axillary sweat glands in a young person. Recently, it was reported that the obstruction of intraepidermal apoeccrine sweat ducts by apoeccrine secretory cells might be the possible causes of Fox-Fordyce disease (114).
The most useful marker of sweat gland differentiation is carcinoembryonic antigen (CEA), found mainly in the luminal borders of secretory cells of eccrine glands and excretory eccrine ducts and, to a less extent, on apocrine glands. Gross cystic disease fluid protein-15 (GCDFP-15) and epithelial membrane antigen (EMA) are also detected in both eccrine and apocrine sweat glands (1,115). Myoepithelial cells lining the secretory sweat glands express smooth muscle actin and keratin K17 (1).
Dermis
The dermis is a dynamic, supportive connective tissue harboring cells, fibrous tissue, and ground substances with adnexal structures and vascular and nerve plexuses running through it (1). The dermis (Figure 1.30) consists of two zones: the papillary dermis and reticular dermis. The adventitial dermis (116) combines the papillary and the periadnexal dermis.
The papillary and periadnexal dermis can be recognized by a loose meshwork of thin, poorly organized collagen composed of predominantly type III collagen (117,118,119) mixed with some type I collagen and a delicate branching network of fine elastic fibers. The papillary dermis also contains abundant ground substance, fibroblasts and the capillaries of the superficial arterial and venous plexuses.
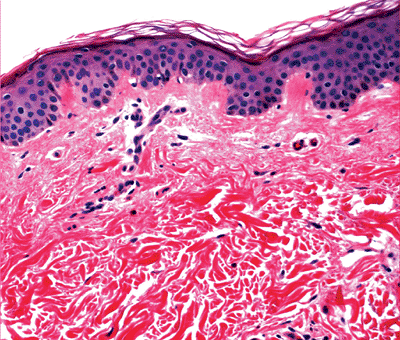 |
Figure 1.30 Dermis with papillary and thick reticular dermis. |
P.18
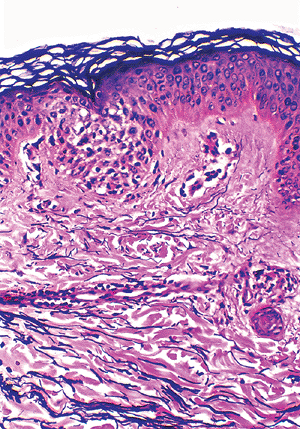 |
Figure 1.31 Distribution of elastic fibers. Elastin fibers are thin and branching in the papillary dermis and thick and fragmented in the reticular dermis. |
The reticular dermis is thicker than the papillary dermis and is composed of multiple layers of well-organized thick bundles of collagens, predominantly type I collagen, mostly arranged parallel to the surface (117,118,119). These layers are built from overlapping of individual fibers of uniform size. The plates are oriented randomly in different directions (120). There are also thick elastic fibers with fragmented appearance detected by special elastic tissue stains (Figure 1.31). Some ground substance and the vessels of the deep plexuses are also present in the reticular dermis.
The resident cells in the dermis mainly include dermal dendritic cells, fibroblasts, and mast cells (1). Dermal dendritic cells are a group of cells with immunophenotypic and functional heterogeneity located in the dermis and possessing a dendritic morphology (121). There are multiple subsets of dendritic cells. At least three types of dermal dendritic cells are recognized as distinct cell types with unique immunophenotype in vivo (122,123).
Factor XIIIa+ dermal dendrocytes are in the perivascular distribution in the papillary dermis and around sweat glands (1,122). Dermal dendrocytes, also known as dendrophages (126), express some markers of mononuclear macrophages (124) and have phagocytic function (125).
CD34+ dendritic cells are present in the mid- and deep dermis around adnexae (1,122).
The dermis harbors a true dendritic cell population, also in a perivascular distribution. These are Langerhans' cell like dendritic cells involved in dermal antigen presentation, expressing HLA-DR and CD1a except for lack of Birbeck granules (123,127,128).
Fibroblasts are the dynamic and fundamental cells of the dermis, synthesizing all types of fibers and ground substances (1,119). They appear as spindle-shaped or stellate cells, which are not reliably differentiated from other dermal spindle-shaped cells and dendritic cells in H&E-stained sections. Ultrastructurally, they contain prominent, well-developed rough endoplasmic reticulum.
Mast cells are derived from bone marrow CD34+ progenitor cells and are sparsely distributed in the perivascular and periadnexal dermis. They are recognized by a darkly stained ovoid nucleus and granular cytoplasm, which is highlighted by Giemsa and toluidine blue stains. Mast cells are positive with tryptase and c-kit (CD117) immunohistochemical stains (129,130,131). Mastocytosis is characterized by abnormal growth and accumulation of mast cells in various organs with heterogenous manifestation. Urticaria pigmentosa is the most common cutaneous manifestation of mastocytosis (132,133).
Macrophages are also seen in the normal dermis; they become visible when pigments or other ingested material is present in the cytoplasm of the cells.
Besides fibrous tissue and cellular components, the dermis also contains amorphous ground substance filling the spaces between fibers and dermal cells. It mainly consists of glycosaminoglycans or acid mucopolysaccharides (the nonsulfated acid mucopolysaccharides [predominantly hyaluronic acid] and, to a lesser degree, sulfated acid mucopolysaccharide [largely chondroitin sulfate)]) (1,5). The ground substance is present in small amounts and is seen as empty spaces between collagen bundles in routine H&E-stained sections; it also is hardly identified with Alcian blue and toluidine blue special stains. In pathologic conditions such as lupus erythematosus, granuloma annulare, and dermal mucinosis, the excessive quantity of ground substance produced can be seen without the aid of special stains as strings of bluish material.
Subcutaneous Tissue
Subcutaneous tissue, also called subcutis or hypodermis, is crucial in thermal regulation, insulation, provision of energy, and protection from mechanical injuries. It is composed of mature adipose tissue arranged into lobules. The mature adipocytes within the lobules are round cells rich in cytoplasmic lipids, which compress the nucleus to the side of the cell membrane. The adipocytes express S-100 protein and vimentin in immunohistochemical stains (123). These lobules of mature adipocytes are separated by the thin bands of dermal connective tissue that constitute the interlobular septa (Figure 1.32). Thus, inflammatory changes involving the subcutaneous tissue can be divided into septal panniculitis (e.g., erythema nodosum) and lobular panniculitis (e.g., panniculitis associated with pancreatitis) (24).
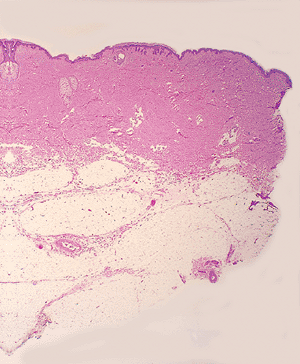 |
Figure 1.32 Septa and lobules of subcutaneous fat. |
P.19
Blood Vessels, Lymphatics, Nerves, and Muscle
The large arteries that supply the skin are located in the subcutaneous tissue, usually within the interlobular septa, and are accompanied by large veins. Smaller arteries, venules, and capillaries constitute the main vasculature seen in the dermis and within the lobules of the subcutaneous fat.
A network of these smaller vessels is located in the papillary dermis (superficial plexus) and in the deep reticular dermis (deep plexus). Superficial vascular plexuses separate the papillary dermis from the reticular dermis, whereas the deep vascular plexuses define the boundary between the reticular dermis and subcutaneous tissue. The division of superficial and deep plexuses is important in the classification and recognition of many inflammatory diseases of the skin in which characteristic infiltrates are located around the superficial, deep, or superficial and deep plexuses. Blood endothelial cells express von Willebrand factor (factor VIII-related antigen), vimentin, CD34, and CD31 antigens (1,123).
Vasculitis is the inflammatory process that involves the blood vessels. It is important to remember that strict criteria are applied for the diagnosis of cutaneous vasculitis, and they include: (a) the presence of inflammatory cell infiltrate within the vessel wall, and (b) the presence of vascular injury, in a spectrum from edema and extravasations of red blood cells, leukocytoclasis, thrombi within the lumina of these blood vessels to fibrinoid necrosis and/or destruction of the blood vessel wall (Figure 1.33). The presence of fibrinoid necrosis of the vessel wall is essential for diagnosis of true vasculitis. Perivascular inflammation alone is not a sign of vasculitis.
Mainly in the acral skin, special arteriovenous anastomosing structures known as glomera are present in the reticular dermis. Each glomus is composed of an arterial segment (the Sucquet-Hoyer canals) connected directly with venous segments. Each Sucquet-Hoyer canal is surrounded by four to six layers of glomus cells, which are considered as vascular smooth muscle cells serving as a spincter. Glomus is involved in thermal regulation (1,5).
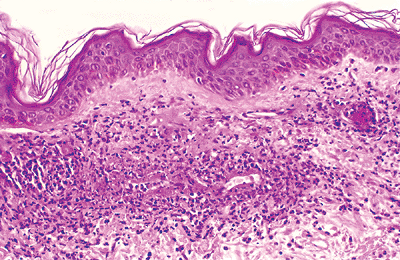 |
Figure 1.33 Vasculitis. Case of leukocytoclastic vasculitis showing damage to the capillary wall. |
The lymphatics of the skin (134) accompany the venules and are also located in the deep and superficial plexuses. Unless valves are seen within these vessels, their recognition in routine sections is impossible. Under normal conditions, they are surrounded by a cuff of elastic fibers. In contrast to blood endothelial cells, lymphatic endothelium do not react with antibodies to von Willebrand factor (factor VIII-related antigen) and CD34 (123).
Large nerve bundles are seen in the subcutaneous fat and in the deep reticular dermis; however, small nerve fibers are present throughout the skin, reaching the papillary dermis. As mentioned earlier, recent immunohistochemical studies have demonstrated that the epidermis contains free nerve axons in association with Langerhans cells (55).
In sections of the palm and sole, some sensory nerves form nerve ending organs. Meissner corpuscles are seen in the papillary dermis, which is composed of several parallel layers of Schwann cells containing an axon; they function as rapid mechanical receptors for the sense of touch. In weight-bearing areas, the Vater-Pacini corpuscles consist of concentrically arranged Schwann cells with an axon and are located in the deep dermis and subcutaneous fat. They serve as receptors for sense of deep pressure and vibration (Figure 1.34).
Smooth muscle is represented in the skin by the arrector pili muscles, which arise in the connective tissue of the dermis and insert into hair follicles below the sebaceous glands. Melanocytes of congenital nevus are often seen within the arrector pili muscle. Smooth muscle is also seen in the skin of external genitalia (tunica dartos) and in the areolae.
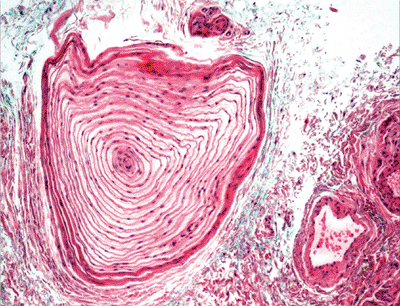 |
Figure 1.34 Vater-Pacini corpuscle. |
P.20
Strands of striated muscle are found in the skin of the neck, face, and particularly the eyelids as muscle of expression.
Histologic Differences of Skin with Age
Newborns and Children
The epidermis of newborns and children is usually of the same thickness as in adults, with the exception of the acral skin. There is a greater density of melanocytes and Langerhans cells.
The dermis is more cellular than in the adult with a higher concentration of ground substance. The number of eccrine glands is higher at birth, while apocrine glands are not well developed until after puberty (135). The sebaceous glands are developed in children, but sebaceous secretion begins at puberty under the influence of androgen stimulation (136).
The adipocytes of the subcutaneous tissue in newborns and children are thin-walled and larger than the adult adipocytes. In addition to white fat as seen in adults, infants possess brown fat, which initially comprises up to 5% of body weight then diminishes with age to virtually disappear by adulthood. Brown adipocytes are rich in mitochondria and contain multiple lipid droplets of varying size in the cytoplasm with centrally located nuclei. Brown fat contains an abundance of blood-filled capillaries and is of particular importance in neonates because it has the ability to produce heat (thermogenesis) by degrading fat molecules into fatty acids (137,138).
Elderly
In the elderly, the histologic differences are mainly due to atrophy and to reduction of most cutaneous elements (139,140,141). The cells of the epidermis are arranged haphazardly because of aberrant proliferation of the basal cell layer, which may predispose to the development of neoplasms (142). There is a marked decrease in the number of melanocytes and in the number of melanosomes, leading to reduced pigmentation (140,143) and, consequently, more exposure to the damaging effects of ultraviolet light. The Langerhans cells also decrease in number and function with advanced age (140,143), which increases the damaging effects of contactants and partially contributes to age-associated deterioration of immune function (144).
In the elderly, the dermis is thinned, relatively acellular, and avascular. The dermal collagen, elastin, and ground substance are altered and reduced (139,142). Elastic fibers show structural and biochemical alterations that change the elasticity of the skin. Collagen bundles are thicker but stiffer. The net effect is that age-associated alterations make the dermis less stretchable, less resilient, and prone to wrinkling (145). Fibroblasts, dendritic cells, and mast cells are also reduced in number.
Because of the reduction in the cutaneous vascular supply, there is a decrease in inflammatory response, absorption, and cutaneous clearance (146).
Both eccrine and apocrine glands are also reduced, with diminished secretions in the elderly. Sebaceous glands increase in size and manifest clinically as sebaceous hyperplasia, but paradoxically their secretory output is lessened by decreased activity (139,147).
With age, the number and rate of growth of hair follicles decreases, vellus hair will develop into terminal hairs in unusual
P.21
sites, such as the ear, nose, and nostrils, resulting in possible cosmetic problems. There is also a decreased functioning of Meissner's and Vater-Pacini corpuscles (148). Finally, there is diminished subcutaneous tissue especially in the face, shins, hands, and feet, but it increases in other areas, particularly the abdomen in men and the thighs in women (139).
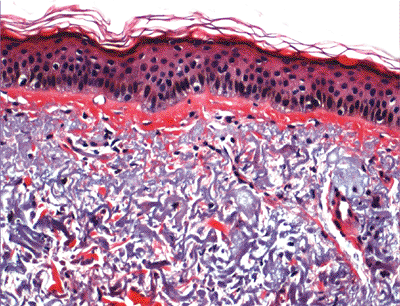 |
Figure 1.35 Solar elastosis in the dermis. |
The pathologic hallmark of extrinsic aging is solar elastosis (Figure 1.35), whereas wrinkling is due to the intrinsic factors mentioned previously (149).
Histologic Variations According to Anatomic Sites
Regional variations of the normal histomorphology are important to recognize so as to avoid misinterpretation of variation as abnormality.
The normal scalp and other densely hair-containing regions show hair follicles extending through the dermis into the subcutaneous fat (Figure 1.36). This is usually not seen in areas with less concentration of hair. Abundant vellus hair is seen in sections taken from the skin of the ear. The skin of the face shows characteristically many pilosebaceous units (Figure 1.37), and large sebaceous glands are seen on the nose.
The squamous layer of the eyelid epidermis is thin and composed of two to three layers of cells and basoloid epithelial buds. Modified apocrine glands (Moll's glands) and vellus hairs are seen in the dermis.
Sections taken from the skin of the trunk, especially the back, show a normally thickened reticular dermis when compared to other sites (Figure 1.38). Unawareness of this normal variation may lead to the erroneous diagnosis of processes producing thick collagen, such as scleroderma. The skin around the umbilicus also shows thick and fibrotic dermis (Figure 1.39).
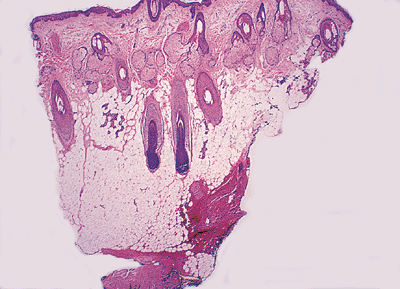 |
Figure 1.36 Section of scalp showing hair follicles extending into the subcutaneous tissue. |
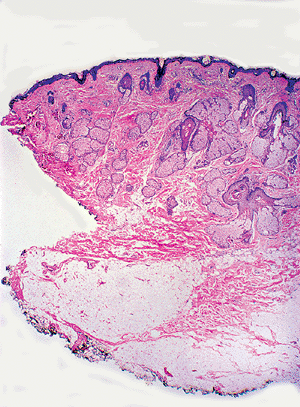 |
Figure 1.37 Skin of face with pilosebaceous units. |
The palms and soles contain stratum lucidum and show a thick, compact cornified layer with loss of the characteristic basket-weave pattern (Figure 1.40). In addition, there are numerous eccrine units, nerve end organs, and glomus structures seen in the dermis. There are no pilosebaceous units. Sections of the skin of the lower leg may show thicker blood vessels in the papillary dermis as a result of gravity and stasis (Figure 1.41). Smooth muscle fibers are seen in the dermis of the skin of external genitalia and areola of the nipple. Cutaneous-mucosal junctions may lack granular and cornified layers, and cells of the squamous layers are larger, with higher glycogen content.
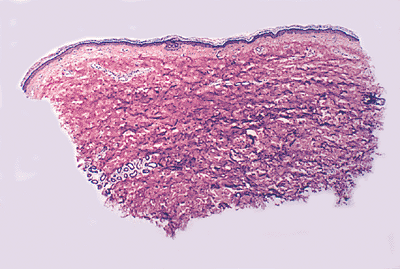 |
Figure 1.38 Section of skin of the back showing the normal reticular dermis. |
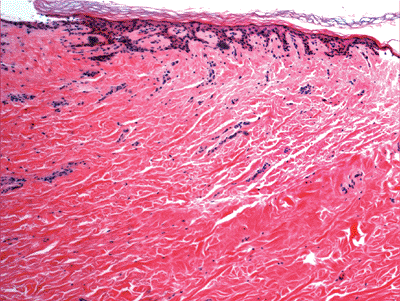 |
Figure 1.39 Umbillicus with dermal fibrosis. |
P.22
Pathologic Changes Found in Biopsies and Interpreted as Normal Skin
Biopsies taken from clinically abnormal skin lesions may be interpreted histologically as normal because of the presence of subtle changes. The following are some examples.
Dermatophytosis is seen in the cornified layer (Figure 1.42) of an otherwise normal skin.
A thick or absent granular layer may indicate an abnormal process of keratinization like psoriasis or an ichthyosiform dermatosis.Vitiligo (Figure 1.43, 1.44) may give the histologic impression of normal skin unless one searches for melanocytes.
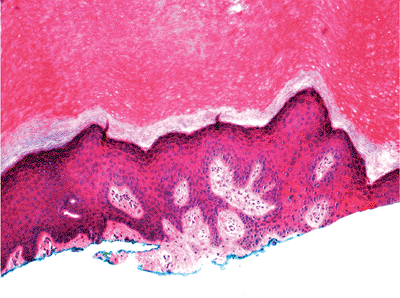
Figure 1.40 Histologic section of the palm with compact cornified layers and stratum lucidum.
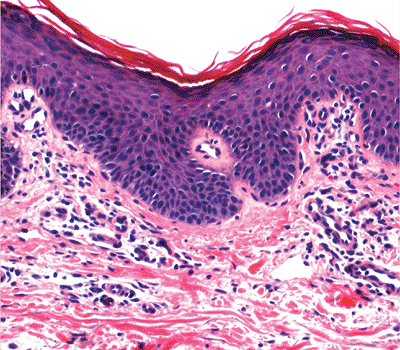
Figure 1.41 Skin of the leg showing a proliferation of small thickened blood vessels secondary to stasis.
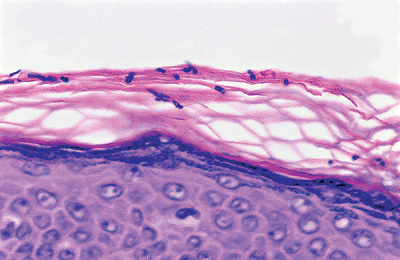
Figure 1.42 Superficial dermatophytosis (PAS stains).
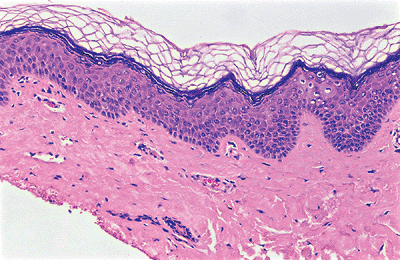
Figure 1.43 Vitiligo. Note the absence of basal melanocytes.
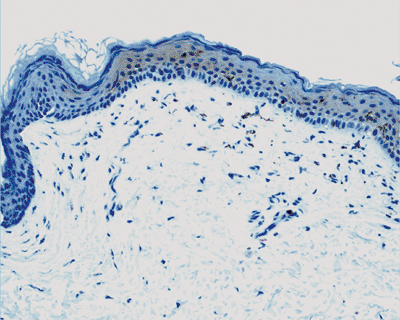
Figure 1.44 Vitiligo. S-100 protein stains show the absence of basal melanocytes.
Macular and lichen amyloidosis (Figure 1.45) may be overlooked because the pink globules of amyloid seen in the papillary dermis can be mistaken for normal dermis.
Urticaria (Figure 1.46) produces only edema, which in routine sections is seen as separation of the collagen bundles in the dermis. Similar changes are seen in the case of dermal mucinosis, in which deposition of mucinous material may be inconspicuous in routine sections. Special stains for mucin will be helpful.
In telangiectasia macularis eruptive perstans, a subtype of cutaneous mastocytosis, the changes may be quite subtle and are composed of dilated blood vessels in the upper dermis with a scant infiltrate of mast cells. The infiltrate must be confirmed with appropriate stains for mast cells.
Trichotillomania is a hair pulling habit resulting in areas of alopecia. Although histologic changes can be numerous (24), at times hair follicles devoid of hair are the only changes seen, which give an impression of normal skin in the biopsy material.
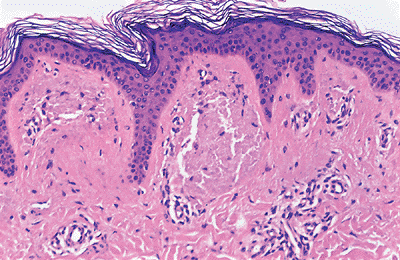
Figure 1.45 Lichen amyloidosis composed of pink globules in the papillary dermis.
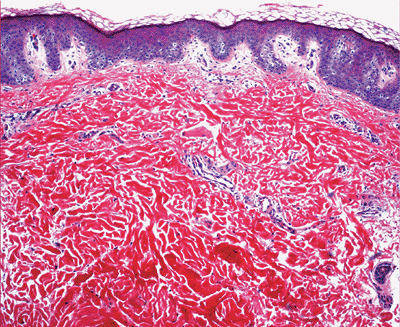
Figure 1.46 Urticaria shows only dermal edema.
Some degenerative disease of skin, such as anetoderma, can represent only as partial loss of elastic fibers in the dermis, which will be demonstrated by special stains of elastic tissue.
The so-called connective tissue nevus representing a hamartoma with an overproduction of collagen bundles and increased, normal or decreased elastic tissue in the dermis is another condition that can be erroneously interpreted as normal skin.
P.23
Other conditions that might be missed as normal skin include caf -au-lait spots, cutis laxa (elastolysis), myxoedema, scleromyxedema, and more. Therefore, the clinical information combined with careful histological examination, sometimes special stains, or immunohistochemical studies of the biopsy material is crucial to avoid misinterpretation of skin disorders as normal tissue.
Specimen Handling
Once the biopsy is done, the specimen should be placed in formalin fixative immediately for the purpose of routine histological examination. Specimens needed for direct immunofluorescent study ideally should be placed in Michel's medium or, alternatively, put in saline-moistened gauze if it is going to be processed within 24 hours. Specimens required for flow cytometry, molecular studies, and electron microscopy are sent fresh in saline-soaked gauze; they should be processed as soon as possible. If the specimens are excisional biopsies or larger surgical material,
P.24
proper sharing of the specimen is done, always with consideration that histology has priority if no prior diagnosis exists for that particular patient.
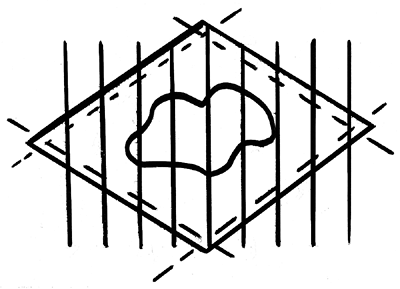 |
Figure 1.47 The entire neoplasm in the center of the lesion is examined by bread-loafing the specimen; the deep margin is also evaluated. The lateral margins are included in each section submitted for histologic evaluation, or they can be submitted separately by cutting them along the depicted interrupted lines. |
Punch and shave biopsies are described grossly and either embedded intact or sectioned, depending on the size of the biopsy. Then the specimen is embedded on edge (vertical). Five levels are usually obtained for histologic examination.
Excisional biopsies and surgical specimens obtained for neoplasm are described grossly, and the entire deep and lateral margins of the specimen are inked prior to sectioning. The margins are evaluated by cutting along all margins or, most commonly, by entirely bread loafing the specimen (Figure 1.47). The entire neoplasm is also evaluated using the bread-loafing technique.
Artifacts
Poor histologic preparation as a result of artifacts will hamper the evaluation of slides by the pathologists. These artifacts can be the result of a variety of factors.
Fixation problems such as poor or no fixation of the specimen prior to cutting, old solutions being used, insufficient fixation time, or inadequate volume of fixative (ideally, the specimen must be properly fixed in a solution 15 to 20 times the volume of the specimen) (150).
Improper monitoring of the multiple steps involved in the preparation of a slide, such as cutting, temperature of the water bath, freshness of the staining solutions employed, and other factors.
Artifacts produced at the time of excision, such as cautery (Figure 1.48) and excessive squeezing of the specimen.
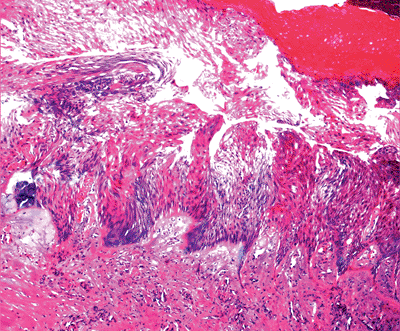
Figure 1.48 Cautery effect with vertical elongation of keratinocytes; such a sample is difficult to evaluate.
Specimens stored at low temperatures, giving freezing artifacts (Figure 1.49).
Artifacts characteristically seen in certain pathologic processes, such as tissue holes in basal cell carcinoma (Figure 1.50) and the lack of epidermis in sections from toxic epidermolytic necrolysis.
Staining Methods
The majority of the skin lesions can be diagnosed with well-prepared H&E-stained sections. However, they will not provide an adequate answer in all cases. A comprehensive review of special stains is beyond the scope of this chapter because every case is different and may require a specific approach. The following are the most common stains used in our laboratory in the study of cutaneous tissue.
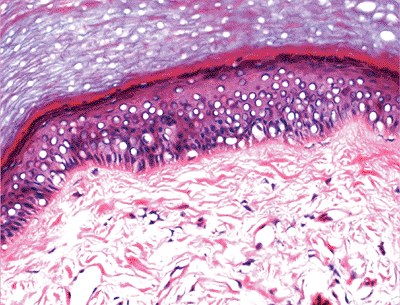 |
Figure 1.49 Freezing artifacts of a specimen with vacuolar changes in the epidermis. |
 |
Figure 1.50 Tissue defects in a basal cell carcinoma, which appear in the spaces after multiple sections were performed. |
P.25
Histochemical Stains
PAS: to study the thickness of the basement membrane, for glycogen (diastase liable) and fungal organisms (diastase resistant)
Gomori's methanamine silver: for fungal organisms and cutaneous Pneumocystis carinii
Ziehl-Nielsen and Fite stains: for acid-fast organisms
Gram's stains: for bacteria
Steiner and Warthin-Starry stains: in cases of bacillary angiomatosis and for spirochetes
Giemsa: for mast cells and protozoan organisms, such as Leishmania
Mucicarmine: for mucin
Alcian blue: for acid mucopolysaccharides (pH 2.5) and sulfated mucopolysaccharides (pH 0.5)
Congo red: for amyloid
Elastic van Gieson (EVG): for elastic fibers
Fontana-Masson: for melanin
Von Kossa: for calcium
Immunofluorescence
Immunofluorescence plays an important place in the diagnosis and evaluation of skin disorders such as lupus erythematosus and autoimmune blistering diseases. Either direct immunofluorescence using tissue or indirect immunofluorescence using serum of patients is available for diagnosis of skin diseases. Direct immunofluorescent studies are performed on cryostat sections of skin specimen using fluorescein isothiocyanate conjugated antisera to examine for the presence of immunoglobulins A, G, and M, as well as fibrinogen and complement.
Immunohistochemical Stains
Immunohistochemical stains are used widely now in combinations of H&E-stained sections for diagnosis of difficult cases, such as poorly differentiated malignant tumors, spindle cell neoplasms, and lymphoproliferative malignancies. Most of the times, these stains are used in panels and not as single preparations. The most commonly used in our laboratory are:
Epithelial markers: Cytokeratin CAM 5.2 (Figure 1.51) (low-molecular weight cytokeratin), a combination of cytokeratins AE1/AE3 (low- and intermediate-molecular weight keratin, carcinoembryonic antigen (CEA), and epithelial membrane antigen (EMA): They are used in the differential diagnosis of epithelial tumors, adnexal tumors, and Paget's disease.
Melanocytic markers: S-100 protein, Melan A/MART-1, and HMB-45.
Mesenchymal markers:
Vimentin: dendritic cells, macrophages;
Factor XIIIa: fibroblast-like mesenchymal cells, dermal dendrocytes;
Factor VIII-associated antigen, CD31 and CD34: endothelial cells; CD34 in combination with Factor XIIIa are useful markers in the diagnosis of dermatofibrosarcoma protuberans (151);
HHF35 (muscle specific actin), smooth muscle actin and desmin: muscle differentiation;
S-100 protein: nerve, fat, and cartilage.
Lymphoid markers: all available CD markers for the characterization of lymphoid proliferations and other hematologic conditions are used.
Histiocytic markers: CD68 (KP-1) and lysozymes
Langerhans cells and Langerhans cell granulomatosis: CD1a and S-100 protein.
Neuroendocrine cells: neurospecific enolase (NSE), chromogranin and synaptophysin. These markers are also expressed in Merkel cell carcinoma; however, cytoplasmic stains for cytokeratin CK20 is most characteristic.
Cell proliferation marker: Ki-67 is an excellent marker for determining the so-called growth fraction of a given
P.26
cell population (152). Higher staining intensity in malignant lesions might be correlated with significant proliferative capabilities in tumor; Ki-67 labeling may have some value in the differentiation between benign and malignant lesions (153,154).
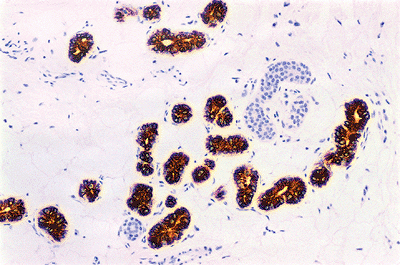 |
Figure 1.51 CAM 5.2 immunostaining. The secretory glands but not the ducts are stained. |
References
1. Kanitakis J. Anatomy, histology and immunohistochemistry of normal human skin. Eur J Dermatol 2002;12:390 399.
2. Montagna W, Parakkal PF. The Structure and Function of the Skin. 3rd ed. New York: Academic Press; 1974.
3. Montagna W, Freedberg IM, eds. Cutaneous biology 1950 1975. J Invest Dermatol 1976;67:1 230.
4. Mckee P. Pathology of the Skin. 2nd ed. London: Mosby Wolfe; 1996.
5. Murphy GF. Histology of the skin. In: Elder DE, Elenitsas R, Johnson, BL Jr, Murphy GF, eds. Lever's Histopathology of the Skin. 9th ed. Philadelphia: Lippincott Williams & Wilkins; 2005.
6. Breathnach AS. Embryology of human skin. A review of ultrastructural studies. J Invest Dermatol 1971;57:133 143.
7. Holbrook KA, Odland GF. The fine structure of developing human epidermis: light, scanning and transmission electron microscopy of the periderm. J Invest Dermatol 1975;65:16 38.
8. Hentula M, Peltonen J, Peltonen S. Expression profiles of cell-cell and cell-matrix junction proteins in developing human epidermis. Arch Dermatol Res 2001;293:259 267.
9. Smith LT, Sakai LY, Burgeson RE, Holbrook KA. Ontogeny of structural component at the dermal-epidermal junction in human embryonic and fetal skin: the appearance of anchoring fibrils and type VII collagen. J Invest Dermatol 1988;90:480 485.
10. Sagebiel RW, Rorsman H. Ultrastructural identification of melanocytes in early human embryos [abstract]. J Invest Dermatol 1970;54:96.
11. Foster CA, Holbrook KA, Farr AG. Ontogeny of Langerhans cells in human embryonic and fetal skin: expression of HLA-DR and OKT-6 determinants. J Invest Dermatol 1986;86:240 243.
12. Foster CA, Holbrook KA. Ontogeny of Langerhans cells in human embryonic and fetal skin: cell densities and phenotypic expression relative to epidermal growth. Am J Anat 1989;184:157 164.
13. Foster CA, Holbrook KA, Farr AG. Ontogeny of Langerhans cells in human embryonic and fetal skin: expression of HLA-DR and OKT-6 determinants. J Invest Dermatol 1986;86:240 243.
14. Winkelmann RK, Breathnach AS. The Merkel cell. J Invest Dermatol 1973;60:2 15.
15. Gould VE, Moll R, Moll I, Lee I, Franke WW. Neuroendocrine (Merkel) cells of the skin: hyperplasias, dysplasias and neoplasias. Lab Invest 1985;52:334 353.
16. Moll R, Moll I, Franke WW. Identification of Merkel cells in human skin by specific cytokeratin antibodies: changes of cell density and distribution in fetal and adult plantar epidermis. Differentiation 1984;28:136 154.
17. Ochiai T, Suzuki H. Fine structural and morphometric studies of the Merkel cell during fetal and postnatal development. J Invest Dermatol 1981;77:437 443.
18. Moll I, Lane AT, Franke WW, Moll R. Intraepidermal formation of Merkel cells in xenografts of human fetal skin. J Invest Dermatol 1990;94:359 364.
19. Boot PM, Rowden G, Walsh N. The distribution of Merkel cells in human fetal and adult skin. Am J Dermatopathol 1992;14:391 396.
20. Breathnach AS. Development and differentiation of dermal cells in man. J Invest Dermatol 1978;71:2 8.
21. Smith LT, Holbrook KA, Madri JA. Collagen types I, III, and V in human embryonic and fetal skin. Am J Anat 1986;175:507 521.
22. Mehregan AH, Hashimoto K, Mehregan DA, Mehregan DR. Normal structure of the skin. In: Mehregan AH, Hashimoto K, Mehregan DA, Mehregan DR, eds. Pinkus' Guide to Dermatohistopathology. 6th ed. Norwalk, CT; Appleton & Lange; 1995.
23. Lavker RM, Sun TT, Oshima H, et al. Hair follicle stem cells. J Investig Dermatol Symp Proc 2003;8:28 38.
24. Ackerman AB. Skin, structure and function. In: Ackerman AB, ed. Histologic Diagnosis of Inflammatory Skin Diseases. Philadelphia: Lea & Febiger; 1978.
25. Downig DT, Stewart ME, Strauss JJ. Biology of sebaceous glands. In: Fitzpatrick TB, Eisen AZ, Wolff K, Freedberg IM, Austen KF, eds. Dermatology in General Medicine. Vol 1. 3rd ed. New York: McGraw-Hill; 1987:185 190.
26. Muller M, Jasmin JR, Monteil RA, Loubiere R. Embryology of the hair follicle. Early Hum Dev 1991;26:159 166.
27. Hashimoto K, Gross BG, Lever WF. The ultrastructure of the skin of human embryos. I. The intraepidermal eccrine sweat duct. J Invest Dermatol 1965;45:139 151.
28. Montagna W. Embryology and anatomy of the cutaneous adnexa. J Cutan Pathol 1984;11:350 351.
29. Salmon JK, Armstrong CA, Ansel JC. The skin as an immune organ. West J Med 1994;160:146 152.
30. Girolomoni G, Caux C, Lebecque S, Dezutter-Dambuyant C, Ricciardi-Castagnoli P. Langerhans cells: still a fundamental paradigm for studying the immunobiology of dendritic cells. Trends Immunol 2002;23:6 8.
31. Smack DP, Korge BP, James WD. Keratin and keratinization. J Am Acad Dermatol 1994;30:85 102.
32. Bos JD, Kapsenberg ML. The skin immune system: progress in cutaneous biology. Immunol Today 1993;14:75 78.
33. Kerr JFR, Winterford CM, Harmon BV. Apoptosis. Its significance in cancer and cancer therapy. Cancer 1994;73:2013 2026.
34. Polakowska RR, Piacentini M, Bartlett R, Goldsmith LA, Haake AR. Apoptosis in human skin development: morphogenesis, periderm, and stem cells. Dev Dyn 1994;199:176 188.
35. Weedon D, Strutton G. Apoptosis as the mechanism of the involution of hair follicles in catagen transformation. Acta Derm Venereol (Stockh) 1981;61:335 339.
36. McCall CA, Cohen JJ. Programmed cell death in terminally differentiating keratinocytes: role of endogenous endonuclease. J Invest Dermatol 1991;97:111 114.
37. Tamada Y, Takama H, Kitamura T, et al. Identification of programmed cell death in normal human skin tissues by using specific labeling of fragmented DNA. Br J Dermatol 1994;131:521 524.
38. Stenn KS, Lawrence L, Veis D, Korsmeyer S, Seiberg M. Expression of the bcl-2 protooncogene in the cycling adult mouse hair follicle. J Invest Dermatol 1994;103:107 111.
39. Seiberg M, Marthinuss J, Stenn KS. Changes in expression of apoptosis-associated genes in skin mark early catagen. J Invest Dermatol 1995;104;78 82.
40. Weedon D, Strutton G. The recognition of early stages of catagen. Am J Dermatopathol 1984;6:553 555.
41. Eckert RL, Efimova T, Dashti SR, et al. Keratinocyte survival, differentiation, and death: many roads lead to mitogen-activated protein kinase. J Investig Dermatol Symp Proc 2002;7:36 40.
42. Masellis M. Deep burns of the knee: joint capsule reconstruction with dermis graft. Annals of Burns and Fire Disasters 1997;10:3 11.
43. Scrivener Y, Cribier B. Morphology of sweat glands. Morphologie 2002;86:5 17.
44. Sato K, Kang WH, Saga K, Sato KT. Biology of sweat glands and their disorders. I. Normal sweat gland function. J Am Acad Dermatol 1989;20:537 563.
45. Sato K, Sato F. Individual variations in structure and function of human eccrine sweat gland. Am J Physiol 1983;245;R203 R208.
46. Uno H. Sympathetic innervation of sweat glands and piloerector muscles of macaques and human beings. J Invest Dermatol 1977;69:112 120.
47. Headington JT. Primary mucinous carcinoma of skin: histochemistry and electron microscopy. Cancer 1977;39:1055 1063.
48. Shah VP, Epstein WL, Riegelman S. Role of sweat in accumulation of orally administered griseofulvin in skin. J Clin Invest 1974;53:1673 1678.
49. Sato K, Kang WH, Saga K, Sato KT. Biology of sweat glands and their disorders. II. Disorders of sweat gland function. J Am Acad Dermatol 1989;20:713 726.
P.27
50. Gesase AP, Satoh Y. Apocrine secretory mechanism: recent findings and unresolved problems. Histol Histopathol 2003;18:597 608.
51. Porter AM. Why do we have apocrine and sebaceous glands? J R Soc Med 2001;94:236 237.
52. Zeng C, Spielman AI, Vowels BR, Leyden JJ, Biemann K, Preti G. A human axillary odorant is carried by apolipoprotein D. Proc Natl Acad Sci USA 1996;93:6626 6630.
53. Wenzel FG, Horn TD. Nonneoplastic disorders of the eccrine glands. J Am Acad Dermatol 1998;38:1 17.
54. Cheshire WP, Freeman R. Disorders of sweating. Semin Neurol 2003;23:399 406.
55. Hosoi J, Murphy GF, Egan CL, et al. Regulation of Langerhans cell function by nerves containing calcitonin gene-related peptide. Nature 1993;363:159 163.
56. Ishiko A, Matsunaga Y, Masunaga T, Aiso S, Nishikawa T, Shimizu H. Immunomolecular mapping of adherens junction and desmosomal components in normal human epidermis. Exp Dermatol 2003;12:747 754.
57. Holbrook KA, Wolff K. The structure and development of skin. In: Fitzpatrick TB, Eisen AZ, Wolff K, Freedberg IM, Austen KF, eds. Dermatology in General Medicine. 3rd ed. New York: McGraw-Hill; 1987.
58. Wolff K, Schreiner E. Ultrastructural localization of pemphigus autoantibodies within the epidermis. Nature 1971;229:59 61.
59. Stanley JR, Klaus-Kovtun V, Sampaio SAP. Antigenic specificity of fogo selvagem autoantibodies is similar to North American pemphigus foliaceus and distinct from pemphigus vulgaris autoantibodies. J Invest Dermatol 1986;87:197 201.
60. Moll R, Cowin P, Kapprell HP, Franke WW. Desmosomal proteins: new markers for identification and classification of tumors. Lab Invest 1986;54:4 25.
61. Toker C. Clear cells of the nipple epidermis. Cancer 1970;25:601 610.
62. Willman JH, Golitz LE, Fitzpatrick JE. Clear cells of Toker in accessory nipples. J Cutan Pathol 2003;30:256 260.
63. Kumarasinghe SP, Chin GY, Kumarasinghe MP. Clear cell papulosis of the skin: a case report from Singapore. Arch Pathol Lab Med 2004;128:e149 152.
64. Marucci G, Betts CM, Golouh R, Peterse JL, Foschini MP, Eusebi V. Toker cells are probably precursors of Paget cell carcinoma: a morphological and ultrastructural description. Virchows Arch 2002 Aug;441(2):117 123. Epub 2002 Feb 01.
65. Kuo TT, Chan HL, Hsueh S. Clear cell papulosis of the skin. A new entity with histogenetic implications for cutaneous Paget's disease. Am J Surg Pathol 1987;11:827 834.
66. Tschen JA, McGavran MH, Kettler AH. Pagetoid dyskeratosis: a selective keratinocytic response. J Am Acad Dermatol 1988;19:891 894.
67. Kim YC, Mehregan DA, Bang D. Clear cell papulosis: an immunohistochemical study to determine histogenesis. J Cutan Pathol 2002;29:11 14.
68. Lazarus GS, Hatcher VB, Levine N. Lysosomes and the skin. J Invest Dermatol 1975;65:259 271.
69. Elias PM. Epidermal lipids, barrier function, and desquamation. J Invest Dermatol 1983;80(suppl):44s 49s.
70. Zirra AM. The functional significance of the skin's stratum lucidum. Morphol Embryol (Bucur) 1976;22:9 12.
71. Katz SI. The epidermal basement membrane zone structure, ontogeny, and role in disease. J Am Acad Dermatol 1984;11:1025 1037.
72. Foidart JM, Bere EW Jr, Yaar M, et al. Distribution and immunoelectron microscopic localization of laminin, a noncollagenous basement membrane glycoprotein. Lab Invest 1980;42:336 342.
73. Smith JB, Taylor TB, Zone JJ. The site of blister formation in dermatitis herpetiformis is within the lamina lucida. J Am Acad Dermatol 1992;27:209 213.
74. Leblond CP, Inoue S. Structure, composition, and assembly of basement membrane. Am J Anat 1989;185:367 390.
75. Woodley DT, Burgeson RE, Lunstrum G, Bruckner-Tuderman L, Reese MJ, Briggaman RA. Epidermolysis bullosa acquisita antigen is the globular carboxyl terminus of type VII procollagen. J Clin Invest 1988;81:683 687.
76. Shimizu H, McDonald JN, Gunner DB, et al. Epidermolysis bullosa acquisita antigen and the carboxy terminus of type VII collagen have a common immunolocalization to anchoring fibrils and lamina densa of basement membrane. Br J Dermatol 1990;122:577 585.
77. Elder DE, Johnson BL Jr, Elenitsas R, eds. Histology of the skin. In: Elder DE, Johnson BL Jr, Elenitsas R, eds. Lever's Histopathology of the Skin. 9th ed. Baltimore: Lippincott Williams & Wilkins: 2004:35.
78. Scott GA, Cassidy L, Tran H, Rao SK, Marinkovich MP. Melanocytes adhere to and synthesize laminin-5 in vitro. Exp Dermatol 1999;8:212 221.
79. Tarnowski WM. Ultrastructure of the epidermal melanocyte dense plate. J Invest Dermatol 1970;55:265 268.
80. Nordlund JJ, Sober AJ, Hansen TW. Periodic synopsis on pigmentation. J Am Acad Dermatol 1985;12:359 363.
81. Quevedo WC Jr, Fitzpatrick TB, Szabo G, et al. Biology of the melanin pigmentary system. In: Fitzpatrick TB, Eisen AZ, Wolff K, Freedberg IM, Austen KF, eds. Dermatology in General Medicine. Vol I. 3rd ed. New York: McGraw-Hill; 1987.
82. Seiberg M. Keratinocyte-melanocyte interactions during melanosome transfer. Pigment Cell Res 2001;14:236 242.
83. Barral DC, Seabra MC. The melanosome as a model to study organelle motility in mammals. Pigment Cell Res 2004;17:111 118.
84. Murphy GF. Structure, function and reaction patterns. In: Murphy GF, ed. Dermatopathology. Philadelphia: WB Saunders; 1995.
85. Gown AM, Vogel AM, Hoak D, Gough F, McNutt MA. Monoclonal antibodies specific for melanocytic tumors distinguish subpopulations of melanocytes. Am J Pathol 1986;123:195 203.
86. Kanitakis J. Immunohistochemistry of normal skin. In: Kanitakis J, Vassileva S, Woodley D, eds. Diagnostic Immunohistochemistry of the Skin. An Illustrated Text. London: Chapman & Hall Med; 1998:38 51.
87. Birbeck NS, Breathnach AS, Everall JD. An electron microscope study of basal melanocytes and high-level clear cells (Langerhans cells) in vitiligo. J Invest Dermatol 1961;37:51 64.
88. Niebauer G, Krawczyk W, Wilgram GF. The Langerhans cell organelle in Letterer Siwe's disease. Arch Klin Exp Dermatol 1970;239:125 137.
89. Valladeau J, Dezutter-Dambuyant C, Saeland S. Langerin/CD207 sheds light on formation of birbeck granules and their possible function in Langerhans cells. Immunol Res 2003;28:93 107.
90. Mc Dermott R, Ziylan U, Spehner D, et al. Birbeck granules are subdomains of endosomal recycling compartment in human epidermal Langerhans cells, which form where Langerin accumulates. Mol Biol Cell 2002;13:317 335.
91. Halata Z, Grim M, Baumann KI. The Merkel cell: morphology, developmental origin, function. Cas Lek Cesk 2003;142:4 9.
92. Halata Z, Grim M, Bauman KI. Friedrich Sigmund Merkel and his Merkel cell, morphology, development, and physiology: review and new results. Anat Rec A Discov Mol Cell Evol Biol 2003;271:225 239.
93. Tachibana T, Nawa T. Recent progress in studies on Merkel cell biology. Anat Sci Int 2002;77:26 33.
94. Santa Cruz DJ, Bauer EA. Merkel cells in the outer follicular sheath. Ultrastruct Pathol 1982;3:59 63.
95. Camisa C, Weissmann A. Friedrich Sigmund Merkel Part II. The cell. Am J Dermatopathol 1982;4:527 535.
96. Gu J, Polak JM, Van Noorden S, Pearse AG, Marangos PJ, Azzopardi JG. Immunostaining of neuron-specific enolase as a diagnostic tool for Merkel cell tumors. Cancer 1983;52:1039 1043.
97. Leff EL, Brooks JSJ, Trojanowski JQ. Expression of neurofilament and neuron-specific enolase in small cell tumors of skin using immunohistochemistry. Cancer 1985;56:625 631.
98. Rosen ST, Gould VE, Salwen HR, et al. Establishment and characterization of a neuroendocrine skin carcinoma cell line. Lab Invest 1987;56:302 312.
99. van Muijen GNP, Ruiter DJ, Warnaar SO. Intermediate filaments in Merkel cell tumors. Hum Pathol 1985;16:590 595.
100. Wang NP, Zee S, Zarbo RJ, Bacchi CE, Gown AM. Coordinate expression of cytokeratins 7 and 20 defines unique subsets of carcinomas. Appl Immunohistochem 1995;3:99 107.
P.28
101. Moll I, Kuhn C, Moll R. Cytokeratin 20 is a general marker of cutaneous Merkel cells while certain neuronal proteins are absent. J Invest Dermatol 1995;104:910 915.
102. Headington JT. Transverse microscopic anatomy of the human scalp. A basis for a morphometric approach to disorders of the hair follicle. Arch Dermatol 1984;120:449 456.
103. de Viragh PA. The mantle hair of Pinkus . A review on the occasion of its centennial. Dermatology 1995;191:82 87.
104. Alonso LC, Rosenfield RL. Molecular genetic and endocrine mechanisms of hair growth. Horm Res 2003;60:1 13.
105. Nakagawa H, Imokawa G. Characterization of melanogenesis in normal human epidermal melanocytes by chemical and ultrastructural analysis. Pigment Cell Res 1996;9:175 178.
106. Jimbow K, Ishida O, Ito S, Hori Y, Witkop CJ Jr, King RA. Combined chemical and electron microscopic studies of pheomelanosomes in human red hair. J Invest Dermatol 1983;81:506 511.
107. Biasiolo M, Bertazzo A, Costa CV, Allegri G. Correlation between tryptophan and hair pigmentation in human hair. Adv Exp Med Biol 1999;467:653 657.
108. Burchill SA, Ito S, Thody AJ. Effects of melanocyte-stimulating hormone on tyrosinase expression and melanin synthesis in hair follicular melanocytes of the mouse. J Endocrinol 1993;137:189 195.
109. Smith KR Jr. The Haarscheibe. J Invest Dermatol 1977;69:68 74.
110. Baron DA, Briggman JV, Spicer SS. Tubulocisternal endoplasmic reticulum in human eccrine sweat glands. Lab Invest 1984;51:233 243.
111. Sbarbati A, Osculati A, Morroni M, Carboni V, Cinti S. Electron spectroscopic imaging of secretory granules in human eccrine sweat glands. Eur J Histochem 1994;38:327 330.
112. Schaumburg-Lever G, Lever WF. Secretion from human apocrine glands: an electron microscopic study. J Invest Dermatol 1975;64:38 41.
113. Sato K, Leidal R, Sato F. Morphology and development of an apoeccrine sweat gland in human axillae. Am J Physiol 1987;252:R166 R180.
114. Kamada A, Saga K, Jimbow K. Apoeccrine sweat duct obstruction as a cause for Fox-Fordyce disease. J Am Acad Dermatol 2003;48:453 455.
115. Saga K. Histochemical and immunohistochemical markers for human eccrine and apocrine sweat glands: an aid for histopathologic differentiation of sweat gland tumors. J Investig Dermatol Symp Proc 2001;6:49 53.
116. Reed RJ, Ackerman AB. Pathology of the adventitial dermis. Anatomic observations and biologic speculations. Hum Pathol 1973;4:207 217.
117. Meigel WN, Gay S, Weber L. Dermal architecture and collagen type distribution. Arch Dermatol Res 1977;259:1 10.
118. Junqueira LC, Montes GS, Martins JE, Joazeiro PP. Dermal collagen distribution. A histochemical and ultrastructural study. Histochemistry 1983;79:397 403.
119. Sorrell JM, Caplan AI. Fibroblast heterogeneity: more than skin deep. J Cell Sci 2004;117(pt 5):667 675.
120. McNeal JE. Scleroderma and the structural basis of skin compliance. Arch Dermatol 1973;107:699 705.
121. Nestle FO, Nickoloff BJ. A fresh morphological and functional look at dermal dendritic cells. J Cutan Pathol 1995;22:385 393.
122. Narvaez D, Kanitakis J, Faure M, Claudy A. Immunohistochemical study of CD34-positive dendritic cells of human dermis. Am J Dermatopathol 1996;18:283 288.
123. Kanitakis J. Immunohistochemistry of normal human skin. Eur J Dermatol 1998;8:539 547.
124. Headington JT. The dermal dendrocyte. Adv Dermatol 1986;1:159 171.
125. Headington JT, Cerio R. Dendritic cells and the dermis: 1990. Am J Dermatopathol 1990;12:217 220.
126. Nickoloff BJ, Griffiths CE. Not all spindled-shaped cells embedded in a collagenous stroma are fibroblasts: recognition of the collagen-associated dendrophage. J Cutan Pathol 1990;17:252 254.
127. Sepulveda-Merrill C, Mayall S, Hamblin AS, Breathnach SM. Antigen-presenting capacity in normal human dermis is mainly subserved by CD1a+ cells. Br J Dermatol 1994;131:15 22.
128. Meunier L, Gonzalez-Ramos A, Cooper KD. Heterogeneous populations of class II MHC+ cells in human dermal cell suspensions. Identification of a small subset responsible for potent dermal antigen-presenting cell activity with features analogous to Langerhans cells. J Immunol 1993;151:4067 4080.
129. Walls AF, Jones DB, Williams JH, Church MK, Holgate ST. Immunohistochemical identification of mast cells in formaldehyde-fixed tissue using monoclonal antibodies specific for tryptase. J Pathol 1990;162:119 126.
130. Arber DA, Tamayo R, Weiss LM. Paraffin section detection of the c-kit gene product (CD117) in human tissues: value in the diagnosis of mast cell disorders. Hum Pathol 1998;29:498 504.
131. Longley BJ, Reguera MJ, Ma Y. Classes of c-KIT activating mutations: proposed mechanisms of action and implications for disease classification and therapy. Leuk Res 2001;25:571 576.
132. Metcalfe DD, Akin C. Mastocytosis: molecular mechanisms and clinical disease heterogeneity. Leuk Res 2001;25:577 582.
133. Valent P, Horny HP, Escribano L, et al. Diagnostic criteria and classification of mastocytosis: a consensus proposal. Leuk Res 2001;25:603 625.
134. Ryan TJ, Mortimer PS, Jones RL. Lymphatics of the skin. Neglected but important. Int J Dermatol 1986;25:411 419.
135. Johnson BL, Honig PJ, Jaworsky C. In: Johnson BL, Honig PJ, Jaworsky C, eds. Pediatric Dermatopathology. Newton, MA: Butterworth-Heinemann; 1994.
136. Pochi PE, Strauss JS, Downing DT. Age-related changes in sebaceous gland activity. J Invest Dermatol 1979;73:108 111.
137. Klaus S. Functional differentiation of white and brown adipocytes. Bioessays 1997;19:215 223.
138. Klaus S. Adipose tissue as a regulator of energy balance. Curr Drug Targets 2004;5:241 250.
139. Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol 1986;15(pt 1):571 585.
140. Kurban RS, Bhawan J. Histologic changes in skin associated with aging. J Dermatol Surg Oncol 1990;16:908 914.
141. Smith L. Histopathologic characteristics and ultrastructure of aging skin. Cutis 1989;43:414 424.
142. Patterson JAK. Structural and physiologic changes in the skin with age. In: Patterson JAK, ed. Aging and Clinical Practice: Skin Disorders, Diagnosis and Treatment. New York: Igaku-Shoin; 1989.
143. Montagna W, Carlisle K. Structural changes in ageing skin. Br J Dermatol 1990;122(suppl 35):61 70.
144. Sauder DN. Effect of age on epidermal immune function. Dermatol Clin 1986;4:447 454.
145. Lavker RM, Zheng PS, Dong G. Morphology of aged skin. Clin Geriatr Med 1989;5:53 67.
146. Balin AK, Pratt LA. Physiological consequences of human skin aging. Cutis 1989;43:431 436.
147. Bolognia JL. Aging skin. Am J Med 1995;98:99S 103S.
148. Cerimele D, Celleno L, Serri F. Physiological changes in ageing skin. Br J Dermatol 1990;122(suppl 35):13 20.
149. Rongioletti F, Rebora A. Fibroelastolytic patterns of intrinsic skin aging: pseudoxanthoma-elasticum-like papillary dermal elastolysis and white fibrous papulosis of the neck. Dermatology 1995;191:19 24.
150. Mondragon G, Nygaard F. Routine and special procedures for processing biopsy specimens of lesions suspected to be malignant melanomas. Am J Dermatopathol 1981;3:265 272.
151. Altman DA, Nickoloff BJ, Fivenson DP. Differential expression of factor XIIIa and CD34 in cutaneous mesenchymal tumors. J Cutan Pathol 1993;20:154 158.
152. Scholzen T, Gerdes J. The Ki-67 protein: from the known and the unknown. J Cell Physiol 2000;182:311 322.
153. Li LX, Crotty KA, McCarthy SW, Palmer AA, Kril JJ. A zonal comparison of MIB1-Ki67 immunoreactivity in benign and malignant melanocytic lesions. Am J Dermatopathol 2000;22:489 495.
154. Abdelsayed RA, Guijarro-Rojas M, Ibrahim NA, Sangueza OP. Immunohistochemical evaluation of basal cell carcinoma and trichepithelioma using Bcl-2, Ki67, PCNA and P53. J Cutan Pathol 2000;27:169 175.