19 - Pain - An introduction
Editors: Goldman, Ann; Hain, Richard; Liben, Stephen
Title: Oxford Textbook of Palliative Care for Children, 1st Edition
Copyright 2006 Oxford University Press, 2006 (Chapter 34: Danai Papadatou)
> Table of Contents > Section 3 - Symptom care > 17 - Symptoms in life-threatening illness: Overview and assessment
function show_scrollbar() {}
17
Symptoms in life-threatening illness: Overview and assessment
Gerri Frager
J. John Collins
Introduction
The aim of this chapter is to enhance and expand each reader's fund of knowledge with respect to the symptoms faced by children with life-limiting illnesses, particularly those with advanced and end-of-life illness. The chapter's components include the prevalence of symptoms in children with life-limiting illness, symptom assessment and measurement, the barriers to adequate symptom management, and practical resources for families and clinicians. This chapter will primarily be an overview of physical symptoms and their assessment, although it is well appreciated that physical symptoms do not exist in isolation, and that physical, psychological, and spiritual factors all impact on one another.
The prevalence of symptoms of children with life-limiting illnesses
The 1995 World Health Organization Report provided the dire statistics that 12.2 million children less than 5 years of age, die each year. Many of these children have life-limiting illnesses for whom palliative care could be beneficial [1]. A US-based study over a period of two decades examined the pattern of childhood and young adult deaths among 15,000 individuals, aged 0 24 years with complex chronic conditions. It was estimated that 5000 of these infants, children, adolescents, and young adults were within their last 6 months of life [2].
What children experience as a result of their life-limiting illness is largely dependent upon where they live in the world. The causes of mortality differ vastly if the child is born into a developing or a developed country, as evident from worldwide childhood mortality patterns. Even with the same illness, how children live and die with that illness may make it seem like an entirely different entity.
For example the treatment options available to children with HIV/AIDS varies according to geographical location. HIV/AIDS is known to cause pain and other systems from multiple causes, including the primary virus itself, opportunistic infections, and where treatment is provided, from the treatment of either the human immunodeficiency virus with anti-retroviral agents, or the treatment of the secondary infections [3]. Even in developed countries, where the knowledge, skills, and pharmaceutical agents are available for symptom relief, multi-factorial barriers continue to impede their access and implementation. Pain significant enough to be reported as having an impact on their life was reported by 59% of children with HIV infection in a US study [4]. The major symptom burden of HIV/AIDS in children is borne by the developing countries, where the numbers of children affected is tragically high, and where access to treatment of the HIV infection, attendant opportunistic infections, and analgesics may be poor (Table 17.1).
What frequently occurs in clinical practice is often not easily shared through the published literature. Parents and health care professionals develop ways of finding out how the child is feeling, tailored to the child. They may do this through trialing different language, facilitating their communication through the use of stuffed toys, through modeling, and non-verbal strategies and innovative verbal means. Comprehensive and consistent symptomatic relief can be a challenge because of the relatively small numbers of children within each diagnostic category.
The past decade has witnessed an exponential growth of interest in the field of pediatric palliative care. The interest and
P.232
Table 17.1 Estimates of life-limiting conditions of childhood (in developed countries, based on UK surveys) | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||
work has been primarily concentrated on pain in the oncology patient and pain assessment in the neonate. However, relative to the adult population, there remains a paucity of information about symptoms in the life-limiting illnesses of childhood. What is known comes largely from the oncology population, even though comprising only a third of childhood deaths. Where studies have been done, they tend to be focused on dying with an illness, inadequately addressing the child's experience of living with their illness.
Cancer-related symptomatology
The majority of studies have been based on caregiver report, whether that be a parent or health professional rather than from the perspective of the child experiencing the symptom. The pattern of symptoms, based on the self-report of US children aged 10 18 treated for cancer, was studied. Inpatients reported more symptoms than outpatients, with a mean of 12.7 4.9 compared with 6.5 5.7, respectively. Recent administration of chemotherapy was associated with significant symptomatology. Children with solid tumors were more symptomatic than children with a diagnosis of leukemia, lymphoma or cancer of a central nervous system. Lack of energy, pain, drowsiness, nausea, cough, anorexia, and psychological symptoms all had a prevalence rate greater than 35%. Review of the physically-based symptoms showed that pain, nausea, and anorexia were also associated with a high degree of reported distress [5].
A similar survey was conducted in children 7 12 years of age, also treated for cancer, who provided self-report of the symptoms they experienced over the 2 days preceding the survey. Although symptoms from the psychological realm were surveyed, the most prevalent physically based experiences were: pain, sleeping difficulty, itch, nausea, tiredness, lack of appetite, with the first 4 symptoms more often associated with high distress [6].
In a review of the records of 157 US children with cancer who were referred for a neurology opinion [7], the symptoms that prompted referral were analysed. Leukemias and lymphomas were the most common cancer (58%). The 42% of cancers represented by solid tumors included neuroblastoma, Ewing's sarcoma, and rhabdomyosarcoma. Headache and seizures were the most common symptoms for referral, in contrast with back pain and altered mental status which were the main symptoms in adults. This reinforces the notion that population-based studies specific to life-limiting conditions in children are both generally lacking and necessary.
More than a decade has passed since studies confirmed the clinical appreciation that procedure-related pain was a major focus of distress for children undergoing cancer treatment [8]. Despite significant advances in procedural pain management, a recent review from Sweden describes the persistent predominance of pain related to cancer-related procedure in the pediatric population [9]. This differs from the adult cancer population, where disease-related pain still predominates.
Pain relief for the vast majority of children with cancer should be achievable with what would be considered standard or conventional doses of analgesics. It is unusual that extraordinary doses or analgesia via unusual routes, such as the subarachnoid route or the provision of sedation to ensure comfort, are required [10]. However, this fact has unfortunately not directly translated into the action of providing standard or conventional opioid therapy. Barriers to appropriate analgesic therapy persist among patients, families and health professionals.
Symptoms at the end of a child's life
Relative to the adult population, few surveys of the symptoms of children with life-limiting illnesses have been performed. Of these, most have consisted of small numbers of participants, are often based at one site, and often have been carried
P.233
Table 17.2 Compilation of retrospective reviews of children's end-of-life symptomology | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
out over several years. What is known about symptoms in dying children is frequently inferred from descriptive chart reviews that identify patients by age, diagnosis, and may include medication use. Symptom assessment has not often been part of the study, and where it has been, is frequently based on the report of someone other than the child, most commonly the parent or a health care professional (Table 17.2).
A chart review study based in Japan, over a 6 year period, examined the signs and symptoms occurring at the end-of-life in 28 children dying from cancer. The demographics of the children studied match the distribution of childhood cancers in the United Kingdom and North America with leukemia/lymphoma predominating at 39%, brain tumors making up 25%, and 35% of the group comprised of other solid tumors. All children experienced anorexia, 82.1% had dyspnea, and pain was documented in 75%. Fatigue was substantial at 71.4%, nausea/vomiting in 57.1%, and other gastrointestinal symptoms notable for constipation at 46.4% and diarrhea at 21.4% [11]. This symptom profile closely matches that of a US-based study that reviewed symptoms in children in their last month of life. The predominant symptoms assessed by parental report, were fatigue, pain, and shortness of breath [12].
A retrospective chart review from Canada gathered information about 77 infants and children whose deaths occurred at least 7 days since admission to hospital. The majority (83%), who ranged in age from 8 days to 16.8 years, died in the intensive care unit with most (78%) intubated. Perhaps a surprising statistic, this is not unusual for many children who die in a tertiary care setting within a developed country. Documentation of non pain symptoms was sporadic. Even for children where pain had been noted to be a significant problem, complete pain assessments were lacking. Although a small number
P.234
of children had symptoms evaluated, the pattern again reflects that of other pediatric end-of-life studies. Pain, anxiety/agitation, decreased level of consciousness, nausea/vomiting, dyspnea, constipation, and feeding difficulties were all featured [13].
Parents of children cared for at their end-of-life in several pediatric intensive care units retrospectively completed an anonymous questionnaire. Pain was only one component assessed and 55% of parents reported that their child was comfortable. Non-pain symptoms were not addressed [14].
The symptoms of children close to death were surveyed utilising the report of a proxy care-giver [15]. Respiratory failure and encephalopathy were noted as the most frequent physiologic disturbances, although they were not linked to how the child might feel as a consequence. The symptom profile in the last week of life demonstrated a significant constellation of 11.1 5.6 symptoms per child with six symptoms occurring in at least 50% of the children. The most frequently occurring symptoms included lack of energy, drowsiness, skin changes, irritability, pain, and extremity swelling. Lack of energy was associated with the highest level of distress for 30.7% of the children. Although not notably prevalent, nervousness, worry, and numbness/tingling of distal extremities were the most distressing symptoms. The level of patient comfort as reflected by the health professionals' notes indicated that the majority of children were always comfortable to usually comfortable in the last week (64%), day (76.6%), and hour (93.4%) of life. The pattern of many pediatric deaths in the developed world was again reflected in this study with the majority (66.7%) of the hospitalized children dying in the intensive care unit [15].
Over a 5 year period, a Finnish study evaluated the pattern of end-of-life for children dying from cancer. Symptoms were largely inferred by the medications administered with the most detail for pain, as 89% of the children had received pain medication. Pain relief was reported by retrospective parental interview as inadequate in 19% of the children treated with analgesics [16, 17].
Of the children cared for by a palliative home care program at a tertiary care hospital in Canada, a chart review documented that pain severe enough to require opioid therapy occurred in numbers approaching 90%. Parents reported that intractable pain, seizures, and dyspnea were the most distressing complications of their child's end-of-life phase [18].
A review of the deaths occurring over a 7 year period in the inpatient wards at an Australian children's hospital was carried out through chart review. Where the 18 charts did not document the symptoms experienced, inferences were made from the medication record. The diagnoses of cancer, cystic fibrosis, cerebral palsy, and neurodegenerative illnesses each accounted for the deaths in nearly equal proportions other than for one child who died of herpetic encephalitis [19].
The palliative care service at a tertiary care pediatric hospital in the US described some of the characteristics of 154 infants, children, and young adults referred to the service over a 4-year period [20]. The majority of referrals were patients with cancer diagnoses with the balance being made up from a diverse array of childhood illnesses. Analgesic agents corresponding to Step 2 and 3 of the World Health Organization Ladder (WHO) [21] were provided for more than 80% of the patients. In addition to pain, another prominent symptom requiring intervention was sleep disturbance. 44% of the patients with cancer received chemotherapy concurrent with palliative care interventions. This dual model of care is representative of how palliative care in children is often best integrated with care directed to cure and/or to amelioration of the primary disease rather than either goal in isolation [22]. Of the 140 patients who died during the four years of the study period, 60% of them died at home.
The end-of-life care of patients more than 5 years of age and dying from cystic fibrosis was summarized through a chart review at a US tertiary care facility, where they had received their care [23]. Twenty-five percent of these patients had been receiving opioids for more than 3 months prior to their death for relief of chronic headache and/or chest pain. At end-of-life, the percentage of patients treated with opioids for dyspnea and/or chest pain increased to 86%.
Arthritis, headache, and chest pain are known sources of pain in patients living with cystic fibrosis. Osteoporosis may be a cause of pain, associated with protracted coughing with the risk of rib fractures. Headaches would be expected based on the physiology of hypercarbia and/or hypoxia, and/or sinus infection. The arthropathy seen in children with cystic fibrosis can be related to the hypertrophic pulmonary osteoarthropathy (HPOA), antibiotics, infections, or an associated kyphosis [24]. Of children dying of cystic fibrosis and reporting pain, 86% reported serious pain with the most frequent locations being chest, headache, limb, abdomen, and back [25]. The increase in pain in their last three years of life may serve as a warning flag consistent with other signs of advanced, progressive disease [25].
Symptoms in children with neurodegenerative illnesses
While there have been missed opportunities to ask children how they feel in many life-limiting illnesses, many children with neurodegnerative illnesses have been even further marginalized. This population of children most often requires someone's assistance to observe and interpret how they feel. In a study at
P.235
the first free-standing children's hospice in the United Kingdom, the demographics for 127 children was significant for 41% of children having neurodegenerative illness with virtually all children having no speech or impaired speech. The median age from their first admission to death was 2 years [26].
Another study over an 11 year period, at the same hospice, reviewed the care of 30 children admitted in their last month of life. Half of the children were non-communicative, with half of that subgroup having neurodegenerative illnesses. Pain, breathlessness, and oral symptoms, such as difficulty with secretions, were the most common symptoms recorded by the children's caregivers in the last month of life and continued to be noted as frequent symptoms in the last week of life. The reports of excess secretions and cough increased in the last week of life [27].
Symptom assessment
The assessment, diagnosis, and measurement of symptoms is fundamental to the care of any child with a life-limiting illness. Palliative therapeutics should generally be implemented concurrently while investigating, as appropriate, the underlying causative mechanisms, since therapies directed at the primary cause may ultimately have a more effective outcome for symptom management. Symptom assessment is complemented by symptom measurement. Measurement refers to the application of some metric to a specific element of a symptom. There is an increasing interest in the measurement of other domains of symptom experience beyond intensity, frequency and distress, for example.
A historical approach to the assessment and management of symptoms
Consider the child with cystic fibrosis or primary cardiac disease presenting with shortness of breath. The traditional orientation to symptoms has been one focused on the symptom as an important indicator of the disease process, a signal of the underlying pathophysiology and as a marker for the extent of disease. Clinical, research, and educational efforts relating to symptoms have been almost wholly directed to curing or ameliorating the condition responsible for the symptom. Exploring how the child interprets the symptom and addressing any accompanying distress have traditionally not carried as much importance as determining and documenting the symptom's presence, and treating its root cause. A standard of care to aspire to is one where clinical, research, and educational pursuits concurrently address the child's experience of the symptom and to provide relief with the same enthusiasm and urgency as those efforts directed to the primary disease. Children have said or had their families, as witnesses to the child's distress, say that there can be things worse than dying, or they didn't know the cure could be worse than the disease. Symptom measurement may help facilitate an understanding of a child's subjective experience.
The rationale for a focus on symptoms
When faced with the diagnosis of a life-limiting and chronic illness, the parents, health professionals, and the child, as developmentally appropriate, want to know what to expect [28, 29, 30]. What does the future hold? How will the child feel throughout the course of the illness? How can they best prepare for what is to come? A proactive approach, discussing, and planning for what symptoms may occur, confers some measure of control for all involved in the child's care, at a time of tremendous vulnerability. Being able to plan for the what-ifs, to discuss the likelihood of seizures or breathlessness provides reassurance through a discussion and plan. This helps the parent to continue to parent, to support the child through the difficult times, which can be a substantial comfort for both the child and for the parent. Although pain, breathlessness, or nausea may be a feature of the illness, it is reassuring to know that these and other symptoms, are able to be controlled with a variety of agents.
Several end-of-life studies have involved parents monitoring their child's symptoms [31, 32] Parents reported a greater willingness to consider end-of-life care decisions when physical changes were observed. Symptoms have also been the basis for parents'choice of where their child receives end-of-life care [33, 34, 35, 36]. In a feasibility study, parents of children who had died within the previous 2 years, were asked to identify the most troubling symptoms that their child had during the final week of life, final 24 hours of life, and what actions helped or did not help to manage the symptoms (Personal Communication Hinds, P.S. St Jude's Children's Research Hospital, Tennessee) [37]. A total of 34 symptoms were identified by all of the parents; with all parents reporting 2 to 6 symptoms. The most commonly reported were pain, changes in breathing, changes in responsiveness, and urinary output. All families indicated a worsening of the troubling symptoms in the final 24 h of their child's life. Helpful behaviors of professionals included being available to the family, providing information about the symptoms and features of dying, and assuring the parents that they had been good parents (Personal Communication Hinds, P.S. St Jude's Children's Research Hospital, Tennessee) [37]. In addition, a retrospective study examined the perspectives of 25 families of 26 ill children who had been cared for at the first children's hospice
P.236
in the United Kingdom. While families reported feeling well supported for symptom management, they worried about breathlessness, seizures, and pain [38].
Understanding the link between symptoms and specific clinical conditions benefits the child through efforts directed to preventing the symptoms, lessening them, or helping a child and his/her family deal with them. Knowledge about these symptoms can also help health care professionals teach children how to identify and describe a symptom. Such information can further contribute to the prevention, amelioration, or resolution of symptoms.
Knowing the pattern of symptoms for a given life-limiting condition serves to drive interventions directed to relief of the symptom. Where existing therapies are either inadequate or absent, documentation of the symptom prevalence and associated distress can help advance research initiatives. For children who eventually die of their illness, the knowledge that their discomfort was proactively assessed and compassionately managed can, at best, ensure comfort for the bereaved survivors, or otherwise, at least not compound their distress.
Knowing what symptoms are likely to occur at the end-of-life and their time-course, may guide what supportive interventions might be recommended to pursue or help select those best not pursued. For example, a child very ill with end-stage cystic fibrosis has a massive gastrointestinal bleed and a decreased level of consciousness. Actively investigating the bleed and providing multiple transfusions may not necessarily be in this child's best interest, to subsequently have the child rally long enough to become significantly dyspneic as the lung function worsens. Such considerations are generally based on clinical judgement, best managed by discussions with colleagues, the family and child.
Symptoms and quality of life
It does not necessarily hold true that a child's quality of life is directly related to the degree of symptom burden. Quality of Life (QOL) is a complex, multifactorial, and a dynamic process. The words, meaning, and concepts used by quality of life assessment tools for adults require modifications unique to the illness, age and development of the child. In addition, the assessment is frequently delegated by default to someone other than the person whose QOL is being measured. This difficult task often falls to the parent, health professional or other proxy, although studies have demonstrated lack of concordance between self-assessment and proxy-assessments [39, 40].
Despite the difficulties, there is a growing interest to supplement traditional outcomes in pediatric cancer clinical trials such as survival, relapse-free survival, event-free survival, disease-free survival or organ toxicity with those that reflect the impact of treatment on patients and families [41, 42, 43,]. Despite this interest, quality of life, as an endpoint, has yet to make a significant impact on the design and the reporting of Phase III chemotherapeutic trials in pediatric oncology [44]. This is due, in part, to lack of validated measures. Although the measurement of symptoms is one aspect of QOL assessment [45], two of the three validated QOL measures developed for children with cancer, the Play Performance Scale for Children [46] and the Pediatric Quality of Life Scale [47] do not assess symptoms. The Pediatric Cancer Quality of Life Inventory-32 (PCQL-32) is a standardized patient self-report and parent proxy-report assessment instrument designed to systematically assess pediatric cancer patients' health-related quality of life outcomes [48, 49]. It assesses five domains: disease and treatment-related symptoms/problems, and physical, psychological, social, and cognitive functioning. It focuses on pain and nausea in its assessment of physical symptoms.
There is, therefore, a need for the development and use of a comprehensive tool for the measurement of symptoms in the context of quality of life assessment of children with cancer and non-cancer diagnoses.
Other than the child: A bigger view on distress
There may be instances when the parent, family member, friend, or health professional may be the ones most distressed by what they perceive as the child's experience in the course of care. The child's perceptions should be the priority for the symptoms addressed but this is not to the exclusion of other's concern and distress. For example, the noisy pattern of breathing in a child who is unresponsive and unable to swallow or otherwise clear their oropharyngeal and bronchial secretions is a difficult sound to be present for. The distress of those present though this time can be significant despite repeated offered explanations that the child is not disturbed by this. The imperative to provide comfort extends to those accompanying the child at end-of-life as well as the child. Simple measures, including the use of an anti-cholinergic agent, confer a benefit beyond only the recipient of the medication.
When to assess symptoms and when to treat?
Ensuring comfort is a medical imperative throughout the child's entire illness, not one reserved for the period of time identified as solely palliative or end-of-life . Symptoms are not sequestered into a block of time identified as the palliative or end-of-life phase of a life-limiting illness. Rather, symptoms may predominate at diagnosis and any time throughout the entire course of the illness. A US-based study of pediatric oncology patients noted the greatest suffering in children who died of treatment-related complications [12]. Ensuring the relief of pain and other symptoms is the right thing to do
P.237
regardless of when the symptom presents in the course of the illness.
Clinicians are poor predictors of when a child may be approaching end-of-life. One study of clinicians and parents caring for children living with and dying from cancer reported that the understanding that the child had no realistic chance for cure was acquired at vastly different times along the illness trajectory. This ranged from a mean of 206 days before the child's death for physicians to roughly half that for parents [50]. The important message for clinicians is not to wait until the child is clearly deteriorating and has no realistic chance for cure, or until all involved come to such an understanding, before having discussions relating to decision-making and initiating appropriate interventions directed to comfort. Instead, such discussions and incorporation of the principles of palliative care, including the provision of comfort, need to occur throughout the child's entire illness course.
Physical symptoms may be related to different and multiple factors, including the underlying illness, varied treatment-related side-effects, or to causes unrelated to either the primary disease or its treatment. Symptoms, such as fatigue, anorexia, and agitation, tend to cross disease groupings and may have multi-factorial causation.
Symptoms may make a significant contribution to morbidity. Despite a high prevalence, many symptoms may be relatively easily treated with conventional management. This is important information, as there may be an assumption that one must live with certain common aspects of the illness. This misperception may prevent care-givers from initiating relatively simple measures to ensure that the child can feel well and be as active as they can while living with the illness. Even uncommon symptoms can have import because of the degree of associated distress. Breathlessness, pain, or fatigue confer a profound impact on the child's activities and overall QOL. The meaning and value linked with the symptom may carry broader implications, as in the familial anxiety engendered by anorexia and asthenia.
General principles of symptom assessment
A clinical history, detailing the associated aspects of a symptom, including temporal factors, exacerbating features, relieving measures, concurrent sensations etc. serves to clarify information leading to the best strategies for symptom relief. An assessment of the symptom's severity, and how bothersome it is to the child is measured either through self-report and/or observation. Knowing the child's usual intensity and pattern of activity, play, and reported or inferred enjoyment, is crucial to understanding the impact of the symptom on the child's quality of life, even when ill. Physical examination and investigations tailored as appropriate to the child's condition, illness trajectory, and goals of care, complete the picture.
Common symptoms of children with life-limiting illness
Pain
The adequate, proficient and timely assessment, measurement and management of pain in the dying child is of critical importance. Pain is one of the most feared symptoms in the context of life-limiting illness. Not only is it important from an humanitarian viewpoint, but also because the memory of unrelieved pain and other symptoms in dying children may be retained in the memory of parents many years later. It will be impossible for children and their families to negotiate the domains of psychological and spiritual care if pain and other physical symptomatology has not been adequately treated. As few controlled studies of symptom management have been performed in childhood, many of the analgesic and other therapeutic approaches used in children have been developed through experience based on best practice in adults.
Dyspnea
Dyspnea is the subjective sensation of shortness of breath and is a highly distressing symptom for children. The treatment for dyspneic relief is distinct from pain, where for the latter, quality of life is generally improved and function maximized with conventional doses of conventional analgesics. Relief of the sensation of breathlessness in advanced illness may be dependent on providing comfort through sedation.
Nausea
Nausea and vomiting are not uncommon in children receiving palliative care. Nausea and vomiting occur when the vomiting centre in the brain is activated by any of the following: cerebral cortex (e.g. anxiety), vestibular apparatus, chemoreceptor trigger zone (CTZ), vagus nerve, or by direct action on the vomiting centre. Identifying the clear etiologic mechanism can be helpful as the list of potential causes is great and therapies differ, depending on the putative mechanism. One of the most common disease-related cancer treatment effects is nausea and/or vomiting. Vomiting is easily quantified by frequency measurements and is obviously not reliant on self-report. Nausea is very difficult to assess in children, particularly those unable to self-report.
Fatigue
Fatigue is a noteworthy symptom because of its prevalence, impacting on what the child feels able to do, and the paucity of
P.238
therapeutic interventions that provide relief. Parents (n= 103) of children who died of cancer reported fatigue to be the most common and troubling symptom during the final month of their child's life [12]. The etiology of fatigue in children dying of cancer may be due to a combination of factors including: anaemia, poor nutrition, insomnia, metabolic derangement, the increased work of breathing in patients with dyspnoea, side effects of medication, and a range of psychological factors.
In the assessment of fatigue in a child, and the matrix of its potential causes, it is important to establish if this symptom is distressing to the child and/or his family. If so, potential remediable causes should be considered. Therapies directed at the primary cause should be instituted only if these therapies are not a substantial burden to the patient and/or his family.
Seizures
Seizures in children with life-limiting illnesses may be anticipated and managed expectantly, as a component of a particular condition, such as in many of the neurodegenerative illnesses. Seizures may also newly present in a child with a known seizure history who previously had achieved control, as the child becomes more ill. They can also present denovo in an ill child related to biochemical abnormalities without other risk factors. Seizures may be distressing to the child or more evidently distressing for those who witness the child having a seizure.
Case Mitchell was an 11 year old with a brain tumor whose pattern of seizure would start in the fingers of one hand and then march in Jacksonian fashion up the motor strip represented anatomically by the homunculus. His arm, shoulder, then upper torso would sequentially become involved. When the seizure activity reached his abdomen, he was able to be reassured first by his parents and then calm himself, knowing that the seizure would soon be over. He was distressed by seeing his arm shake and his parents learned to comfort him by covering his arm as soon as Mitchell voiced awareness of the impending seizure. These measures were used in combination with titration of his anticonvulsants until better pharmacologic control was achieved.
Hematologic symptoms/signs
Several of the signs manifested by many of the pediatric cancers relate to the effect on the bone marrow either because of direct marrow involvement by the cancer or the treatment's suppression of the marrow's production of white cells, red cells, and platelets. The potential for associated symptoms of infection, fatigue, breathlessness, or bleeding may be problematic for the child and/or the family and those who care for them. Interventions to address the neutropenia, anemia, or thrombocytopenia require discussion, decision-making, and periodic review.
The use of platelet transfusions at home for children with end-stage cancer was reviewed over a 5 year period by a UK children's hospital. 35 transfusions were provided to 12 children for the following indications: platelet counts less than 25X109 per mm3 and bleeding for longer than an hour, or severe, painful bruising unresponsive to tranexamic acid. The average time between repeat transfusions was 4 days with the majority of transfusions noted to be successful for hemostasis [51].
Repeated transfusions, particularly in areas without the capacity for transfusions at home, requiring the child to be in hospital or hooked-up for intermittent and extended periods, may not be how the child and/or family want to spend the remainder of the child's life. Even in the setting of platelet transfusions continuing to be provided at the end-of-life, it is prudent to have a contingency plan to provide rapid sedation for the child, to minimize the distress in the uncommon event that a massive bleed occurs.
Confusion or delirium
Although lacking the prevalence data, clinicians' impressions are that this is an uncommon but significantly troubling symptom. To have the child as they are known and loved, essentially lost' before being lost through death , compounds the distress of the child's family, friends, and care givers. There may be treatable causes of the confusion, such as hypoglycemia or medication influences, both of which can be resolved by a relatively simple intervention. The decision to change medications, such as opioid rotation, in the setting of confusion, is dependent to a large extent, upon the child's proximity to death. It may be reasonable to provide sedation until death rather than making changes in the child's regimen when death is proximate.
The assessment of symptoms at different stages of child development
An infant's symptoms an infant's story
Proactive management of potential symptoms at end-of-life
Case Emily was a newborn with a congenital muscular disorder (Figure 17.1). She had severe hypotonia, ineffective respiratory effort, some facial and spontaneous eye movements, right club-foot, and bilaterally dislocated hips. Emily had no spontaneous movement of her limbs, which had flexion contractures and markedly decreased muscle bulk.
P.239
At 15 days of age, in preparation for withdrawal of ventilatory support, Emily was pre-medicated with an anti-cholinergic as she was unable to manage her secretions. Emily also received an opioid and anxiolytic to ensure she did not experience distress related to not being able to adequately ventilate.
Emily was unable to express how she felt. Being unable to adequately ventilate is recognized as a likely distressing entity and knowing that she would be unable to cough or swallow her secretions, she was medicated prior to extubation. This approach is sometimes necessary in infants where what is known to be a potentially painful or distressing entity should be preemptively and proactively managed rather than waiting for care givers to observe, interpret, and act upon signs of distress [52].
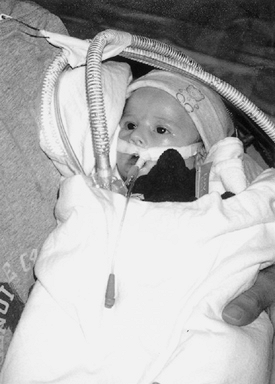 |
Fig.17.1 Emily, a new born with a congenital muscular disorder. |
An adolescent's symptoms an adolescent's story
Whose symptom and whose associated distress?
Case Gina was a savvy, independent 16 year-old, who enjoyed cheerleading and a wide circle of friends (Figure 17.2). She experienced many difficulties during treatment of Ewing's sarcoma. A feisty individual, she toughed it out through an extensive limb salvage procedure, chemotherapy, and radiotherapy, staying hopeful and celebrating the positive aspects of her life.
Gina's course was complicated by obstructive uropathy, necessitating a nephrostomy tube and external bag. Metastases to her lower spine resulted in urinary retention for which she self-catheterized for the the kidney not drained by the nephrostomy tube. Gina had significant neuropathic pain managed with a combination of opioids and multiple adjuvants, from which she had significant adverse reactions, requiring many changes to her analgesic regimen.
Despite achieving good analgesia and managing her nephrostomy and self-catheterizations, Gina became despondent because of fecal incontinence secondary to involvement of the cauda equina from spinal cord metastases. She expressed that she felt life was not worth living because of the incontinence.
With various techniques, Gina was able to achieve fecal continence, enabling her to relax in a hot tub, a goal Gina had for her trip to California with her boyfriend, father, and stepmother [52].
 |
Fig.17.2 Gina a 16 year-old who experienced many difficulties during treatment of Ewing's sarcoma. |
P.240
The importance of Gina's story is that it is the patient's distress with any given symptom that is the priority. The assumption might have been that having her pain well controlled was the priority of care. Such interventions had no import for Gina's quality of life if she could not also achieve fecal continence.
Symptom measurement in children
Pain (See Also Chapter 20, Pain: Assessment)
Self-report scales
In the view of field experts, the best instruments for measuring the severity of pain are visual analogue scales using one of the preferred facial expression scales [53]. In the very young child (aged 3 to 4), the self-report should be simple, with a maximum of three to five options. In persistent, recurrent, or chronic pain, it is appropriate to consider multidimensional assessment, and because few self-report scales of this nature exist, semi structured interviews may be required.
Cognitively intact children aged 7 years and older can rate the severity of their pain or other symptoms with a modified Likert scale scored from 0 to 10, as used in the adult population. Self-report measures for the cognitively intact, younger verbal child of approximately 4 years of age generally use some form of pictorial or photographic representation of faces in various degrees of distress, as detailed in the chapter on pain assessment [54]. To use such a scale, the child needs to understand the concepts of proportionality, that pain or the symptom for which the scale is adapted, is experienced along a continuum, and be able to apply these concepts to visual representation, as in the Faces Pain Scale [55]. Modifications to the scales have been validated for use in pediatric pain assessment, through testing the strength of the tool's ability to measure what is intended. As the experience of pain is a subjective one, the process of validating a measurement tool, such as the visual analogue scale, is a complex one requiring input from multiple, often indirect sources, including behavioral observation. Of interest, is the description of Piaget's and Inhelder's early research from the 70's. This reference described that young children, yet unable to count, were able to understand the concept of quantity along a linear continuum by relating which pile of beads would result in the longer necklace [56, 57]. Measuring other dimensions associated or causally related to having pain have been explored to a less extent in children, as described in a later section on multidimensional symptom assessment scales. Naming the affective state or emotional quality of pain and its severity, such as feeling sad or the pain being unpleasant , is a more abstract task than the rating of pain's severity and is linked to a more advanced developmental understanding, proving difficult for children less than 8 years of age. [56] For clinical purposes, it is less critical what pain intensity scale is chosen. The guiding principle is that pain, especially persistent pain, should be assessed and monitored.
Modifications to these scales for non pain symptoms, although having clinical utility, have not generally been validated for this context.
Behavioral observation measures
The measurement of symptoms in children who are unable to self-report, because of their age, impaired cognition, or difficulty communicating with voice or by pointing requires methods reliant on behavioural observations. Unobtrusive observation of the child and/or a caregiver's description of their observations are incorporated in the assessment. Watching how the infant moves, appears with diaper changes, or positions itself in sleep and interacts while playing are all opportunities for assessing how the way they are feeling may be impacting on their behavior. Measurement tools to assist in gathering this information have been developed for children experiencing or likely to experience pain in certain circumstances, such as the post operative period, in the care of premature and other ill neonates, and in the context of procedures, such as veni punctures and bone marrow aspirates.
There is a paucity of measurement tools for pain that may be chronic or other non pain symptoms. The Gauvain-Piquard rating scale is designed for the assessment of chronic pain in oncology patients 2 6 years-of-age. The modified scale consists of 15 observed items, 9 of which are specific to pain assessment, 6 are related to what is referred to as psychomotor retardation', and 4 assess anxiety. There is a maximum possible score of 60 as each item is rated from 0 to 4, with a score greater than 12 indicative of pain [58].
The child with cognitive impairment
The reliance on functional measures as indicators of pain or other distressing symptoms, such as observing the child's behavior when sleeping or playing assumes greater relevance in the child unable to self-report. Biologic markers, such as heart rate and blood pressure or diaphoresis attenuate with time, may be influenced by medications or the child's illness, and are therefore unreliable indicators of pain, particularly when pain may be more longstanding.
The non-verbal child whose medical condition causes them to have facial grimacing, dystonic posturing, and/or spasticity accompanied by increased tone makes observational assessment of distress even more difficult. There have
P.241
been efforts to assess pain in this patient population, many of whom have life-limiting illnesses in whom death in childhood would be anticipated. The input of a primary caregiver, someone who knows the child well, and who can help interpret their behavior is extremely helpful. The kind of items indicative of distress are fairly intuitive but it is helpful to have a checklist against which to compare and rate one's observations. The reader is referred to the chapter on pain assessment in this volume and primary references for additional detail [56, 59, 60].
Nausea and vomiting
Instruments for the assessment of nausea and vomiting have been studied in the context of childhood cancer. A rating scale for nausea and vomiting utilising verbal descriptors was used in a series of assessment studies in children with cancer aged 5 18 [61, 62, 63, 64]. Children younger than 10 years had faces included above numbers on the scale. There was 80% agreement between parent and child rating when they were assessed independently.
A comparison of child and parental ratings of children's nausea and emesis symptoms was assessed among 33 children (aged 1.7 17.5 years, median 4.7 years) with acute lymphoblastic leukemia receiving identical chemotherapy [65]. The measures utilised nausea and vomiting vignettes designed to assess the frequency and severity of nausea and emesis symptoms as reported by children and their parents based on the previous chemotherapy experience of the child. The vignettes, based on the work of Zeltzer [66], consisted of 12 questions separately assessing nausea and emesis at three time intervals: prior to, during, and after chemotherapy. A 5-point Likert-type rating scale ranging from not at all to all the time for the frequency items and from not bad to real bad on the severity items were employed. A composite nausea/vomiting score was determined by calculating the mean of the 12 frequency and severity items. Children younger than 5 years were not asked to complete this measure because of their difficulty in understanding the instructions. This study demonstrated a significant correlation between child and parent ratings of nausea. Significant inter-rater correlations for nausea frequency and severity but not for emesis frequency or severity was found.
Fatigue
Recent work developing valid measures of child or adolescent cancer-related fatigue should help establish prevalence and incidence data about fatigue in children with varied life-limiting illnesses [67]. A new Children's Fatigue Scale (CFS), was tested in 7 12-year-old oncology patients (n = 149), just over half of whom were within 6 months of diagnosis. The most frequently endorsed items included not being able to play, being tired in the morning, sleeping more at night, not being able to run, and laying around. Children and adolescents have noted distressing fatigue at the time of their cancer diagnosis, throughout treatment, and for several years following successful treatment [6, 68, 69]. Associated factors in cancer-related fatigue include altered sleep, sadness or depression, anemia, hospitalization, inadequate nutrition, and lack of enjoyment for social encounters [6, 67, 70].
Dyspnea
The many tools assessing apparent breathlessness have not been found to correlate with the patient's report of dyspnea, which is based on the self-report of a subjective sensation. Rather, these tools, such as the physiologic measures of respiratory rate, carbon dioxide or oxygen saturation or their functional counterparts of spirometry, or inspiratory nasal pressure, the walk, step or shuttle test which evaluate exercise capacity, do not correlate with the patient's sensory experience of breathing or their difficulty with it. There have been several tools developed and modified for subjective report by adults, such as the Borg Scale, also referred to as an RPE Scale (Report of Perceived Exertion). The original scale was used to calculate heart rate in athletes and had numbers ranging from 6 to 20 accompanied by verbal descriptors, such as maximal exertion [71]. Such language is poorly understood by most children and has little clinical utility for this patient population. Even the ten descriptors arranged along a vertical line in the modified Borg include moderate or slight and would similarly challenge the comprehension of many children. These scales need to be markedly modified by asking qualitative questions oriented to the sensation of ease or discomfort in breathing by asking modifiers like: How much is your breathing bothering you, do you find breathing is hard for you?, or from the functional perspective, Are there things you would like to do but can't because you have trouble catching your breath; is your breathing keeping you from running or playing?, etc.
For quantitative self-report, pictorial scales were developed for self-rating the sensation of dyspnea, based on focus groups of children living with asthma and cystic fibrosis. These four, 7-item scales were subsequently trialed in children with respiratory illnesses. Also evaluated were ratings of pain, fear, and sleep. Referred to as the Dalhousie Dyspnea Scale , this visual analogue scale has been shown to have reliability for self-rating the intensity of one's breathlessness for children 6 years of age and older [72].
P.242
Multidimensional symptom assessment tools for children
The Memorial Symptom Assessment Scale (MSAS) 10 18 is a 30 item patient-rated instrument adapted from a previously validated adult version to provide multidimensional information about the symptoms experienced by children with cancer aged 10 18 [5]. The scale was created to enable children to rate their own symptoms in terms of severity, frequency and distress. The analyses supported the reliability and validity of the MSAS 10 18 subscale scores as measures of physical, psychological, and global symptom distress, respectively. The majority of patients could easily complete the scale in a mean of 11 min.
A revised MSAS was created as an instrument for the self-assessment of symptoms by children with cancer aged 7 12 years [6]. Validity was evaluated by comparison with the medical record, parental report, and concurrent assessment on visual analogue scales for selected symptoms. The data provide evidence of the reliability and validity of MSAS (7 12) and demonstrated that children as young as 7 years with cancer could report clinically relevant and consistent information about their symptom experience. The completion rate for MSAS 7 12 was high and the majority of children completed the instrument in a short period of time and with little difficulty. The instrument appeared to be age appropriate and may be helpful to older children unable to independently complete MSAS 10 18.
 |
Fig.17.3 Daniel a 5 year-old with metastatic neuroblastoma. |
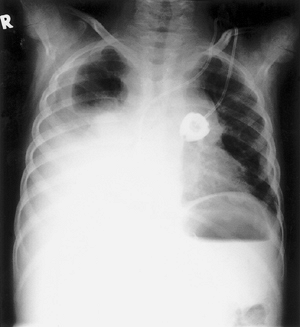 |
Fig.17.4 Daniel's chest X-ray. |
A child's symptoms a child's story
Breathlessness by whose assessment?
Case Daniel was a 5 year-old with metastatic neuroblastoma, having relapsed following treatment with chemotherapy, radiation, surgery, and a bone marrow transplant (Figure 17.3). He had been able to describe his pain as an owie and tell his parents when he was hurting ever since his diagnosis, when he was 3 years old. With modifications by Daniel's mother, he was able to use the Faces Pain Scale to self-report pain.
Daniel would describe his nausea as feeling like I have to spew and tummy cramps . His nausea and vomiting were managed with regular antiemetics including a pro-kinetic agent. Following occasional emesis, Daniel would continue to want small amounts of food and his family would have the foods he enjoyed readily available.
When Daniel developed a pleural effusion compounded by anemia with a hemoglobin of 62, he was reported as looking distressed with apparent breathlessness. However, he was unable to identify if he was breathless or had trouble with his breathing , nor could he say if his breathing was bothering him (Figure 17.4). His parents noted that he was self-restricting his activity and arranging himself on the couch to achieve a position of comfort. He would lie with the pleural effusion side dependent eventhough he had previously always preferred to lie in the opposite direction to watch the activities in the family room.
Daniel did not find supplemental oxygen help him feel better. His apparent breathlessness was managed with a blood trans-fusion, opioids, anxiolytics, and physical measures, such as positioning, and cool air to his face. His parents rearranged the family room, so that Daniel could continue to enjoy watching the family activity in comfort.
P.243
Daniel required modifications to the developmentally appropriate pain scale. His mother tailored the visual analogue scale so that it was both acceptable to him and able to provide useful information about his pain. Fastidious observers of their children, parents are reliable sources of information about how their child may be behaving differently. They can help interpret for the health professional what behaviors may be indicative of physical distress.
Barriers to symptom assessment
General barriers
Children and their families need to be provided with the rationale for the questions detailing the assessment. They may tire of the questions, particularly if they do not see an outcome that seems to be of benefit. Children may believe, If I tell them how I feel they're only going to poke at me and do more tests.
There may be an assumption that symptoms are an expected part of the child's illness, and therefore, symptoms may not be reported. Congruent with this perception may be the child's or family's assumption that because the health professionals have a diagnosis, these clinicians would also know what the child would be experiencing. Children and families may have a view that the clinician would be addressing their symptoms if there was something that could be done about them [72].
Barriers specific to childhood development
The tools developed for use in the adult population are generally not applicable to pediatrics because of several factors. The items typically scored in adult symptom and QOL assessments would not be applicable to a child's life nor understood by a child because of developmental constraints.
Where assessment tools exist for children' symptoms and QOL, they are from the frequently narrow focus of a particular illness not easily generalizable or extrapolated to other illnesses. Life-limiting conditions of childhood are frequently unusual, uncommon, with an unclear prognosis. Both the diversity and the small number of life-limiting childhood conditions relative to the adult population compound the difficulty in acquiring knowledge about the patterns of the accompanying symptoms. The main contribution to the understanding of symptoms in childhood conditions to date has been in cancer and generally focused on pain. In general, the focus on physical symptoms has been on the acute pain measurement typical of the post operative period.
Whether and how children understand what is happening to their bodies and whether they perceive that they have any control over the situation can influence their degree of distress [72]. On occasion, illness can cause regression in the child's usual capacity to understand and interact.
It is essential to the child's care that symptoms are anticipated and expectantly managed. Generally, compared with adults, children have little patience with medications that may make them feel some way they don't like, even if they are benefiting in some other way. Children are not known as masters of delayed gratification. This may be partially related to a relative lack of understanding about the relationship between cause and effect. A child who experiences nausea or itching as an unwanted opioid-related side effect, will be reluctant to continue with the opioid, despite excellent analgesia [73].
Care of the pediatric population includes those who are non verbal by virtue of their age and others with cognitive impairment, both unable to use even those scales specifically tailored for children. A substantial proportion of children living with or dying from a life-limiting illness are represented by these individuals, among them children less than one year of age who comprise the single largest age-related group of pediatric deaths.
Clinicians'obstacles to optimal symptom assessment
Despite the knowledge that medications exist to safely and adequately manage pain and other symptoms, clinicians do not necessarily use them appropriately or in a timely fashion. Despite the fact that opioids can be used safely and long-term, misperceptions and fears persist about their use [74].
Some clinicians continue to inappropriately choose to keep such agents in reserve , when the child really needs them or restrict their use to a time that is clearly end-of-life. Perhaps physicians may find that bearing witness to the child's distress more tolerable if they do not ask about it and the patient and family inadvertently maintain an interaction of they don't ask and we don't tell . This may also be related to clinicians feeling less than wholly competent in aspects related to symptom management. Despite its importance, this particular aspect of care has been poorly addressed in health care professionals' training.
If clinicians feel they are unable to conduct an accurate assessment of a child's pain or other symptom, they may be reluctant to trial an intervention, such as an opioid or an adjuvant for pain relief. The concern for potential adverse effects is generally greatly disproportionate to the reality. Unfortunately, such lack of understanding continues to be a formidable foe of excellent and appropriate care, being notably resilient, obstinate in the face of comprehensive evidence to the contrary.
P.244
Resources for symptoms in the life-limiting conditions of childhood
In addition to various references in the expanding literature relating to pediatric palliative care [75, 76, 77, 78, 79], some of the best current resources about symptoms in life-limiting conditions are through disease-specific internet-based organizations, often parent driven, such as the mucopolysaccharidoses (MPS) group. There may be parallel venues tailored for the child (as appropriate), family, or health professional frequently with national or regional links, support groups, and ask the expert sections.
To help with the symptom profile of various illnesses
|
To help with the assessment and management of symptoms
A listserv accessible worldwide through e-mail/internet for issues relating to pediatric palliative care is accessible by sending an e-mail to paedpalcare@act.org.uk with subscribe as the subject line.
A listserv accessible worldwide through e-mail/internet for issues relating to pediatric pain is accessible by sending an e-mail to LISTSERV@is.dal.ca with the message SUB PEDIATRIC-PAIN and your first then last name.
www.act.org.uk and www.cnpcc.ca are two comprehensive web sites with excellent resources, including policies and educational materials and links for palliative care for children.
www.childendoflifecare.org A web-based resource with information, including audio clips for health care professionals, and families.
www.ich.ucl.ac.uk/cpap An initiative of the Institute of Child's Health and based at Great Ormond Street Hospital for Children, has developed The Children's Pain Assessment Project. Has links to other internet and paper-based resources.
www.ippcweb.org The Initiative for Pediatric Palliative Care A comprehensive website with curricula and resources.
UNIPAC Eight A Hospice/Palliative Medicine Approach to caring for Pediatric Patients. A paper-based, self-study curriculum from the American Academy of Hospice and Palliative Medicine.
ChIPPS Curriculum, from a collaboration between the Children's International Project on Palliative/Hospice Services and the National Hospice and Palliative Care Organization, www.nhpco.org.
To help with support for patients and their families (Information, Resources)
In addition to those listed above www.Bravekids.org www.faculty.fairfield.edu/fleitas/contents.html (also known as Bandaids and Blackboards ). www.virtualhospice.ca Canadian Virtual Hospice is an interactive network for people dealing with life-threatening illness and loss.
Final thoughts and future directions
A recent study reported that 76% of the parents interviewed had noted that pain or discomfort was very important in guiding their decision to forgo life support for their children being cared for in several US-based pediatric intensive care units [28]. It should be contested that symptoms or the lack of their adequate management not be the driving force behind the decision-making in end-of-life care. The solace imagined to be offered by suggesting that, at least their child is no longer suffering provides a shallow refuge and a hollow victory that clinicians must continually strive to reach beyond.
The numbers of children facing life-limiting illnesses will hopefully always be small, and so the barriers to research in this area will likely persist. What would improve research and positively influence care would be more collaborative initiatives, where several centers collectively pool their resources and experience. Potential research initiatives in this content area are vast, as there is much room for new initiatives and enhancement of existing work. Some thoughts include further exploration into the assessment of nonacute pain and non pain symptoms in children with cognitive impairment or non verbal expressive abilities. Commitment to greater integration into practice and clinical utility should be required of researchers developing pain and symptom measurement tools and those who fund them. Linking measurement development to the outcome assessment of therapeutic interventions, such as pharmacologic trials for the relief of dyspnea, is one strategy. Aligned efforts are needed to test the best ways for education and dissemination of the acquired knowledge, and evaluation of the impact on care and comfort of children with life-limiting conditions.
P.245
To improve this aspect of care, at least five things must happen
If what is already known about what can help relieve pain and other symptoms is applied in every location where children experience distressing symptoms, from tertiary care in patient units, intensive care units, at home and in small rural community hospitals, the collective symptom burden could be profoundly reduced.
The focus needs to be grounded first with the child. Efforts put into finding the right words and methods understandable and acceptable to the child should drive research initiatives on pain- and symptom-assessment in pediatrics. Systematic research directed to finding out how the children feel, from the children themselves, will lead to addressing what most bothers them, and helping them to feel better.
The enthusiasm with which health professionals face the challenge of curing the child's illness needs to be shared with addressing how the child's feels living with and dying from their illness.
Granting for any clinical research should be incumbent on ensuring the study has clinical utility and the investigators describe a feasible strategy for how the results of the proposed study will be disseminated and implemented to enhance care.
Research granting agencies need to develop and strengthen their commitment to funding beyond the research, to ensure the study results advance care.
The human and monetary resources currently designated for pediatric palliative care are inadequate to support the work that needs to be done, the work that would ultimately improve the lives of these children, and those who care for them. All who are involved in the health of children, whether from a personal, clinical, educational, or research focus, can be important activists for the advancement of pediatric palliative care.
Acknowledgements
Grateful thanks are extended to the families of Emily, Mitchell Fraser, Daniel Penman, and Gina Smith, who very generously allowed their children's stories and pictures to be shared.
The kind contribution of Pamela S. Hinds PhD, RN, CS, Director of Nursing Research at St Jude Children's Research Hospital, Tennessee, is very much appreciated through the generous sharing of her work and review of the draft manuscript.
References
1. WHO. The World Health Report 1995: Bridging the gap. Report of the Director General. Geneva: WHO, 1995.
2. Feudtner, C., Hays, R.M., Haynes, G., Geyer, J.R., Neff, J.M., and Koepsell, T.D. Deaths attributed to pediatric complex chronic conditions: National trends and implications for supportive services. Pediatrics 2001;107:99.
3. Oleske, J.M. and Czarniecki, L. Continuum of palliative care: Lessons from caring for children infected with HIV-1. Lancet 1999; 354:1287 90.
4. Hirschfeld, S., Moss, H., Dragisic, K., Smith, W., and Pizzo, P.A. Pain in pediatric immunodeficiency virus infection: Incidence and characteristics in a single-institution pilot study. Pediatrics 1996; 98:449 52.
5. Collins, J.J., Byrnes, M.E., Dunkel, I. et al. The Memorial Symptom Assessment Scale (MSAS): Validation study in children aged 10 18. Pain Symptom Manage 2000;23:363 77.
6. Collins, J.J., Devine, T.B., Dick, G. et al. The measurement of symptoms in young children with cancer: The validation of the Memorial Symptom Assessment Scale in children aged 7 12. J Pain Symptom Manage 2002;23:10 6.
7. Antunes, N.L. and De Angelis, L.M. Neurologic consultations in children with systemic cancer. Pediatr Neurol 1999;20:121 4.
8. McGrath, P.J., Hsu, E., Capelli, M., Luke, B., Goodman, J.T., and Dunn-Geir, J. Pain from paediatric cancer: A survey of an outpatient oncology clinic. J Psychosocial Oncol 1990;8:109 24.
9. Ljungman, G., Gordh, T., Sorensen, S., and Kreuger, A. Pain Variations during cancer treatment in children: A descriptive survey. Pediatr Hematol Oncol 2000;17(3):211 21.
10. Collins, J., Grier, H., Kinney, H., and Berde, C.B. Control of severe pain in children with terminal malignancy. Pediatrics 1995; 126:653 7.
11. Hongo, T., Watanabe, C., and Okada, S. Analysis of the circumstances at the end of life in children with cancer: symptoms, suffering and acceptance. Pediatr Int 2003;45:60 4.
12. Wolfe, J., Grier, H.E., Klar, N. et al. Symptoms and suffering at the end of life in children with cancer. NEJM 2000;342:326 33.
13. McCallum, D.E., Byrne, P., and Bruera, E. How children die in hospital. JPSM 2000;20:417 23.
14. Meyer, E.C., Burns, J.P., Griffith, J.L., and Truog, R.D. Parental perspectives on EOL care in the PICU. Critical Care Med 2002;2:226 231.
15. Drake, R., Frost, J., and Collins, J.J. The symptoms of dying children. J Pain Symptom Manage 2003;226:594 603.
16. Sirki , K., Ahlgren, B., and Hovi, L. Terminal care of the child with cancer at home. Acta Paediatr 1997;86:125 30.
17. Sirki , K., Ahlgren, B., Pouttu, J., and Saarinen-Pihkala, U.M. Pain medication during terminal care of children with cancer. J Pain Symptom Manage 1998;15:220 6.
18. St-Laurent-Gagnon, T. Paediatric palliative care in the home. Paediatr Child Health 1998;3:165 8.
19. Mallinson, J. and Jones, P.D. A 7-year review of deaths on the general paediatric wards at John Hunter Children's Hospital, 1991 97. Child Health 2000;36:252 5.
P.246
20. Belasco, J.B., Danz, P., Drill, A., Schmid, W., and Burkey, E. Supportive care: Palliative care in children, adolescents, and young adults Model of care, interventions, and cost of care: A Retrospective review. Care 2000;16:39 46.
21. WHO. The World Health Organization. Cancer Pain Relief and Palliative Care in Children. In Geneva: (ed.) World Health Organization, 1998.
22. Frager, G. Pediatric Palliative Care: Building the Model, Bridging the Gaps. J Palliat Care 1996;12(3):9 12.
23. Robinson, W.M., Ravilly, S., Berde, C., and Wohl, M.E. End-of-life care in cystic fibrosis. Pediatrics 1997;100:205 9.
24. Massie, R.J., Towns, S.J., Bernard, E., Chaitow, J., Howman-Giles, R., and Van Asperen, P.P. The musculoskeletal complications of cystic fibrosis. Paediatr Child Health 1998;34:467 70.
25. Ravilly, S., Robinson, W.M., Suresh, S., Wohl, M.E., and Berde, C. Chronic pain in cystic fibrosis. Pediatrics 1996;98:741 7.
26. Hunt, A. and Burne, R. Medical and nursing problems of children with neurodegenerative disease. 1995;9:19 26.
27. Hunt, A.M. A survey of signs, symptoms and symptom control in 30 terminally ill children. Dev Med Child Neurol 1990;32: 341 6.
28. Angst, D.B. and Deatrick, J.A. Involvement in health care decisions: parents and children with chronic illness. J Family Nurs 1996;2(2):174 94.
29. James, L. and Johnson, B. The needs of parents of pediatric oncology patients during the palliative care phase. J Pediatr Oncol Nurs 1997;14:83 95.
30. Lewis, C., Knopf, D., Chastain-Lorber, K. et al. Patient, parent, and physician perspectives on pediatric oncology rounds. J Pediatr 1988;112:378 84.
31. Hinds, P., Bradshaw, G., Oakes, L., and Pritchard, M. Children and their rights in life and death situations. In R. Kastenbaum, (ed.) Macmillan Encyclopedia of Death and Dying. New York: Macmillan, 2003, pp. 139 47.
32. Kirschbaum, M.S. Life support decisions for children: What do parents value? Adv Nurs Sci 1996;19(1):51 71.
33. Martinson, I.M. Why don't we let them die at home? RN 1976; 39(1):58 64.
34. Martinson, I.M. and Henry, W.F. Home care for dying children. Hastings Center Report 1980;10(2):5 7.
35. Martinson, I.M., Moldow, D.G., Armstrong, G.D., Henry, W.R., and Nesbit, M.E. Home care for children dying of cancer. Res Nurs Health 1986;9(1):11 16.
36. Davies, B., Deveau, E., deVeber, B. et al. Experiences of mothers in five countries whose child died of cancer. Cancer Nurs 1998;21(5): 301 11.
37. Hinds, P.S. Personal communication.
38. Stein, A., Forrest, G.C., Woolley, H., and Baum, J.D. Life limiting illness and hospice care. Arch Dis Child 1989;64:697 702.
39. Gerharz, E.W., Eiser, C., and Woodhouse, C.R. Current approaches to assessing the quality of life in children and adolescents. BJU Int 2003;91(2):150 4.
40. Eiser, C. and Morse, R. Can parents rate their child's health-related quality of life? Results of a systematic review. Qual Life Res 2001;10(4):347 67.
41. Reaman, G.H. and Haase, G.M. Quality of life research in childhood cancer. Cancer 1996;78(6):1330 2.
42. Spieth, L.E. and Harris, C.V. Assessment of health-related quality of life in children and adolescents: An integrative review. J Pediatr Psychol 1996;21(2):175 93.
43. Bradlyn, A.S., Ritchey, A.K., Harris, C.V. et al. Quality of life research in pediatric oncology. Cancer 1996;78(6):1333 9.
44. Bradlyn, A.S., Harris, C.V., and Speith, L.E. Quality of life assessment in pediatric oncology: A retrospective review of Phase III reports. Soc Sci Med 1995;41(10):1463 5.
45. Moinpour, C.M., Feigl, P., Metch, B. et al. Quality of life end points in cancer clinical trials: review and recommendations. J National Cancer Ins 1989;81(7):485 95.
46. Lansky, S.B., List, M.A., Lansky, L.L., Ritter-Sterr, C., and Miller, D.R. The measurement of performance in childhood cancer patients. Cancer 1987;60:1651 6.
47. Goodwin, D.A., Boggs, S.R., and Graham-Pole, J. Development and validation of the Pediatric Quality of Life Scale. Psychol Assess 1994;6:321 8.
48. Varni, J.W., Katz, E.R., Seid, M. et al. The Pediatric Cancer Quality of Life Inventory-32 (PCQL-32). Reliability and validity. Cancer, 1998;82(11):84 96.
49. Varni, J.W., Seid, M., Knight, T.S., Uzark, K., and Szer, I.S. The PedsQL 4.0 Generic Core Scales: sensitivity, responsiveness, and impact on clinical decision-making. J Behav Med 2002;25(2):175 93.
50. Wolfe, J., Klar Neil, Grier, H. et al. Understanding of prognosis among parents of children who died of cancer: impact on treatment goals and integration of palliative care. JAMA 2000; 284:2469 75.
51. Brook, L., Vickers, J., and Pizer, B. Home platelet transfusion in pediatric oncology terminal care. Med Pediatr Oncol 2003;40:249 251.
52. McConnell, Y. and Frager, G. (internet) Decision-making in pediatric palliative care. An Educational Module. In The Ian Anderson Continuing Education Program in End-of-Life Care, University of Toronto and The Temmy Latner Centre For Palliative Care, Mount Sinai Hospital, 2003, with permission.
53. Champion, G.D., Goodenough, B., von Baeyer, C.L., and Thomas, W. Measurement of Pain by Self-Report. In G.A. Finley, P.J. McGrath, (ed.) Progress in Pain Research and Management, 1998; Vol. 10: Seattle: IASP Press.
54. Hicks, C.L., von Baeyer, C.L., Spafford, P., van Korlaar, I., and Goodenough, B. The Faces Pain Scale-Revised: Toward a common metric in pediatric pain measurement. Pain 2001;93:173 83.
55. Collins, J.J. Symptom control in life-limiting illness. In D. Doyle, G.W.C., Hanks, N. Cherny, K.C., Calman, (eds.) Oxford Textbook of Palliative Medicine (3rd Edition) Oxford: Oxford University Press, 2004; pp.789 798.
56. Gaffney, A., McGrath, P.J., and Dick B. Measuring pain in children: Developmental and instrument issues. In N. L. Schechter, C. B. Berde, M. Yaster, (ed.) Pain in Infants, Children and Adolescents Vol. 2: Williams & Wilkins, PA: Lippincott, 2003; pp. 128 41.
57. Piaget, J. and Inhelder B. The Child's Construction of Quantities. London: Routledge and Kegan Paul, 1977.
58. Gauvain-Piquard, A., Rodary, C., Rezvani, A., and Serbouti, S. The development of the DEGRR: A scale to assess pain in young children. European J Pain. 1999;3:165 76.
59. Breau, L.M., McGrath, P.J., Camfield, C.S., and Finley, G.A. Psychometric properties of the non-communicating children's pain checklist-revised. Pain 2002;99:349 57.
P.247
60. Stallard, P., Williams, L., Velleman, R., Lenton, S., and McGrath, P.J. Brief report: Behaviors identified by caregivers to detect pain in noncommunicating children. J Pediatr Psychology 2002;27: 209 14.
61. Zeltzer, L., Kellerman, J., Ellenberg, L., and Dash J. Hypnosis for reduction of vomiting associated with chemotherapy and disease in adolescents with cancer. J Adolesc Health Care 1983;4:77 84.
62. Zeltzer, L., LeBaron, S., and Zeltzer, P.M. The effectiveness of behavioral intervention for reducing nausea and vomiting in children and adolescents receiving chemotherapy. J Clin Oncol 1984;2:683 90.
63. Zeltzer, L.K., LeBaron, S., and Zeltzer. P.M. A prospective assessment of chemotherapy related nausea and vomiting in children with cancer. Am J Pediatr Hematol/Oncol 1984;6:5 16.
64. LeBaron, S. and Zeltzer L. Behavioral intervention for reducing chemotherapy-related nausea and vomiting in adolescents with cancer. J Adoles Health Care 1984;5:178 82.
65. Tyc, V.L., Mulhearn, R.K., Fairclough, D. et al. Chemotherapy induced nausea and emesis in pediatric cancer patients: External validity of child and parent ratings. Dev Behav Pediatr 1993;14(4): 236 41.
66. Zeltzer, L.K., LeBaron, S., Richie, D.M., and Reed, D. Can children understand and use a rating scale to quantify somatic symptoms? Assessment of nausea and vomiting as a model. J Consult Clin Psychol 1988;56(4):567 72.
67. Hockenberry, M.J., Hinds, P.S., Barrera, P. et al. Three instruments to assess fatigue in children with cancer: The child, parent and staff perspectives. Pain Symptom Manage, 2003;25: 319 28.
68. Hinds, P., Scholes, S., Gattuso, J., Riggins, M., and Heffner B. Adaptation to illness in adolescents with cancer. J Pediatr Oncol Nurs 1990;7:64 5.
69. Hinds, P.S., Quargnenti, A., Bush, A.J. et al. An evaluation of the impact of a self-care coping intervention on psychological and clinical outcomes in adolescents with newly diagnosed cancer. Eur JOncol Nurs 2000;4:6 17.
70. Hinds, P.S., Hockenberry-Eaton, M., Gilger, E. et al. Comparing patient, parent, and staff descriptions of fatigue in pediatric oncology patients. Cancer Nurs 1999;22:277 89.
71. Borg, G. Borg's Perceived Exertion and Pain Scales. Stockholm: Human Kinetics, 1998.
72. Pianosi, P., McGrath, P.J., and Smith, C. Four pictorial scales to evaluate dyspnea in children. Am J Resp Crit Care Med 157 (Suppl), A782.
73. McGrath, P.J. and Frager, G. Psychological barriers to optimal pain management in infants and children. Clin J Pain, 1996;12:135 41.
74. Frager, G. Pediatric palliative care. In S.K. Joishy, (ed.) Palliative Medicine Secrets, Hanley & Belfus, 1999; pp. 158 73.
75. Liben, S. Pediatric palliative medicine: obstacles to overcome. J Palliat Care 1996;12:24 28.
76. Goldman, A., Frager, G., and Pomietto, M. Pain and Palliative Care. In N.L., Schechter, C.B. Berde, M. Yaster, (ed.) Pain in Infants, Children and Adolescents, Vol. 2: Williams & Wilkins, PA: Lippincott 2003, pp. 539 62.
77. Goldman, A. Life-limiting illnesses and symptom control in children. In D. Doyle, G.W.C. Hanks, and N. MacDonald, (ed.) Oxford Textbook of Palliative Medicine, Oxford: Oxford University Press, 1998; Vol.2:pp. 1033 43.
78. Levetown, M. Treatment of Symptoms Other than Pain in Pediatric Palliative Care. In R.K. Portenoy, E. Bruera, (eds.) Topics in Palliative Care. New York: Oxford University Press, 1998; pp. 51 72.
79. Goldman, A., Burne, Rees, P., and Duggan, C. Different Illnesses and Problems they Cause. In A. Goldman, (ed.) Care of the Dying Child. Oxford: Oxford University Press, 1999; pp. 14 51.
80. Miser, J.S. and Miser, A.W. Pain and symptom control. In A. Armstrong-Dailey, S.Z. Goltzer, (ed.) Hospice Care for Children, New York: Oxford University Press, 1993; pp. 22 59.
EAN: 2147483647
Pages: 47
- Using SQL Data Definition Language (DDL) to Create Data Tables and Other Database Objects
- Using Keys and Constraints to Maintain Database Integrity
- Working with Comparison Predicates and Grouped Queries
- Understanding Transaction Isolation Levels and Concurrent Processing
- Working with SQL Database Data Across the Internet