8. The Nervous System
Authors: Corwin, Elizabeth J.
Title: Handbook of Pathophysiology, 3rd Edition
Copyright 2008 Lippincott Williams & Wilkins
> Table of Contents > Unit III - Integrated Control and Dysfunction > Chapter 8 - The Nervous System
Chapter 8
The Nervous System
The nervous and endocrine systems are the means by which different parts of the body communicate. The nervous system can be separated into the central nervous system, consisting of the nerve pathways of the brain and spinal cord, and the peripheral nervous system, consisting of nerves that innervate the rest of the body. The coordination of our central and peripheral nervous systems allows us to move, talk, think, and respond.
Physiologic Concepts
The Neuron
The neuron, also called a nerve cell, is the functional unit of the nervous system and is a highly specialized cell. Neural maturation occurs before or soon after birth. Once mature, the neuron does not undergo cellular reproduction and cannot be replaced. Each neuron functions to receive incoming stimuli from, and to send outgoing stimuli to, other nerves, muscles, or glands. Neurons pass and receive signals through changes in the flow of electrically charged ions back and forth across their cell membranes.
Parts of the Neuron
Most neurons have four parts: the dendrite, an afferent end that receives incoming signals; the cell body, a central area containing the nucleus; the axon, a long extension on which the signal passes; and the axon terminals, which branch off of the axon and deliver the signal to other cells. A typical neuron is shown in Figure 8-1.
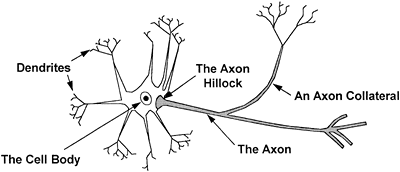 |
Figure 8-1. The neuron. |
P.186
Dendrite
A dendrite is a neural extension from the cell body. The dendrite is the part of the neuron that receives stimulation from other nerves. Each neuron may have many dendritic branches. Excitation of a neuron typically begins at the dendrite. The dendrite passes its excitation on to the adjacent segment, the cell body.
Cell Body
The cell body contains the typical organelles of a human cell. The nucleus, which contains the genetic information of the neuron, orchestrates the production of the proteins, enzymes, and neurotransmitters required by the nerve for its proper function. The cell body delivers these substances as needed to the rest of the neuron. Although neural excitation typically begins with excitation of the dendrites, a cell body sometimes may be stimulated directly by incoming stimuli from other neurons and by chemical and electrical stimuli. The cell body delivers the electrical signal to the next segment, the axon.
Axon
Projecting from the cell body is the axon, the beginning of which is called the initial segment or trigger zone. The axon is a long fiber on which passes the electrical signal initiated in the dendrites and cell body. The axon transmits the original signal to another neuron or to a muscle or gland. Branching off the main stem of the axon may be multiple collateral fibers. Collateral fibers convey information to many other interconnected nerve cells, increasing the influence of the neuron throughout the nervous system. Down the length of the axon, contractile proteins and microtubules transport substances produced in the cell body.
The axon is also called a nerve fiber; many nerve fibers traveling together in a bundle are called a nerve.
In some nerves, the axons are covered by an insulating, lipid sheath, called myelin. Myelin is produced when support cells wrap their plasma membranes around an axon. In the peripheral nervous system, the
P.187
support cells are the Schwann cells. In the central nervous system, myelin is produced by a specialized type of cell, the oligodendrocytes. Myelin increases the velocity with which an electrical signal is transmitted down an axon, as described later.
Axon Terminals
At the end of each main axon stem and collateral, the branching becomes extensive. These final divisions of the axon are called axon terminals. It is through axon terminals that an electrical signal is passed to the dendrites or the cell body of a second neuron. In the peripheral nervous system, the signal also may pass to a muscle or glandular cell.
Categories of Neurons
Neurons that carry information from the periphery to the central nervous system are called sensory or afferent neurons. These neurons are the only type of nerve cell that do not have dendrites, but possess receptors on their distal ends that sense physical or chemical stimuli. Neurons that carry information out of the central nervous system to various target organs (muscle cells, other nerves, or glands) are called motor or efferent neurons. A third group of neurons passes messages between afferent and efferent neurons. These neurons are called interneurons. Almost 99% of all neurons in the body are interneurons, and all interneurons are in the central nervous system.
The Synapse
A synapse is the point of junction between two neurons. Neurons communicate with each other by releasing chemicals into the small cleft (synaptic cleft) separating one from the other. The chemical released from a particular neuron is called a neurotransmitter. Usually, a neurotransmitter is released from the axon terminal of one neuron, diffuses across the synaptic cleft, and binds to a receptor on the dendrite or cell body of the other neuron. However, a synapse can occur between two dendrites, between dendrites and a different cell body, or between an axon and an axon terminal. The cell that releases the neurotransmitter is called the presynaptic neuron. The neuron that completes the synapse is called the postsynaptic neuron. One postsynaptic neuron may receive input from thousands of presynaptic neurons. The postsynaptic neuron integrates and responds to the many signals influencing it. A synapse with two presynaptic neurons is shown diagrammatically in Figure 8-2.
Neurotransmitters and Neuromodulators
Many neurotransmitters are used in the nervous system. Most neurotransmitters are synthesized in the cell body and transported down the axon to the axon terminal. Because neurotransmitters are released from presynaptic neurons, synaptic transmission usually occurs in one direction: from
P.188
the presynaptic to the postsynaptic neuron. Neurotransmitters act rapidly to affect the postsynaptic neuron. To respond to a particular neurotransmitter, the postsynaptic cell must have specific receptors for it on its cell membrane.
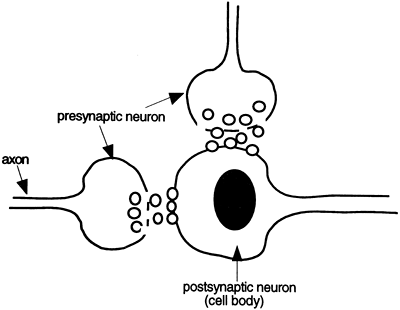 |
Figure 8-2. At a synapse, a presynaptic neuron releases chemicals that diffuse across the synaptic cleft and bind to a postsynaptic cell. |
Most neurons release one neurotransmitter, although some neurons may also release a cotransmitter. Frequently, cotransmitters, called neuromodulators, are a slightly different type of chemical than the neurotransmitter. Neuromodulators typically take longer to act than neurotransmitters, and may function to increase or decrease DNA transcription and protein synthesis. Neuromodulators often affect the response of a post-synaptic cell to a neurotransmitter, and are associated with long-term functions such as learning, mood, and development.
Examples of neurotransmitters and neuromodulators include the following: monoamines norepinephrine, serotonin, dopamine, and histamine; amino acids gamma-aminobutyric acid (GABA), glycine, glutamate, and aspartate; acetylcholine; and the neuropeptides, including the endorphins, enkephalins, substance P, vasoactive intestinal peptide (VIP), and adenosine triphosphate (ATP). Even some gases may serve as neurotransmitters, including nitric oxide and carbon dioxide. Gases do not bind to postsynaptic receptors, but diffuse into the postsynaptic cell to exert an action.
A few neurotransmitters (e.g., acetylcholine and norepinephrine) can either excite or inhibit a postsynaptic cell. Often, however, a neurotransmitter has the same effect (excitatory or inhibitory) on all cells to which it binds. Examples of inhibitory neurotransmitters include GABA, glycine, nitric oxide, and usually dopamine. Glutamine is an example of an excitatory neurotransmitter. The neurotransmitters mentioned earlier may function in the central nervous system or the peripheral nervous system.
P.189
The Membrane Potential
The separation of electrical charge across any structure sets up an electrical potential. Nerve cells, like all cells, have a separation of electrical charge across their cell membranes such that the inside of the cell is negatively polarized (charged) compared to the outside. The separation of charge across a cell is called the membrane potential.
The membrane potential results from a balance between the concentration and electrical gradients that exist across the cell membrane and drive the movement of ions. These gradients unequally distribute electrically charged ions inside and outside the cell, setting up a membrane potential.
The Concentration Gradient Across the Cell Membrane
A concentration gradient exists across all cell membranes because the sodium-potassium pump transports three positively charged sodium ions out of the cell for every two positively charged potassium ions it pumps in. The separation of ions is shown in Figure 8-3. This sets up a concentration gradient with potassium in higher concentration inside the cell than outside, while sodium is in higher concentration outside the cell than inside (Chapter 1). Because potassium and sodium can readily move across the membrane, both tend to diffuse down their concentration gradients potassium diffuses out of the cell, and sodium diffuses into the cell. The cell membrane is over 50 times more permeable to potassium than to sodium. Therefore, more positive charge moves out of the cell than comes in, making the inside of the cell negative.
The Electrical Gradient Across the Cell Membrane
Opposing the concentration gradient is an electrical gradient set up by potassium and sodium diffusion, and by the accumulation of negatively
P.190
charged proteins inside the cell. Because the inside of the cell is negatively charged, potassium, sodium, and other positively charged ions are drawn inside the cell, whereas negatively charged ions such as chloride tend to leave the cell. This sets up an electrical gradient across each cell.
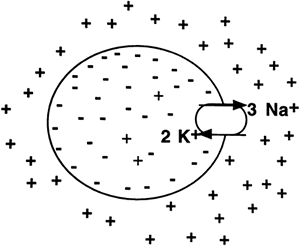 |
Figure 8-3. Excess positive charge on the outside of the membrane compared to the inside contributes to the membrane potential. |
Net Result of Concentration and Electrical Gradients
The final balance reached between the electrical gradient and the concentration gradient across a resting cell is called the resting membrane potential. For any given cell, the resting membrane potential may range from 5 to 100 millivolts (mV), with the inside negative relative to the outside.
Changes in the Membrane Potential of Nerve and Muscle Cells
Cell membranes of neurons and muscles are unique because their permeability to sodium, potassium, chloride, and sometimes calcium can be changed by electrical or chemical stimulation. This allows the membrane potential of neurons and muscle cells to vary from the resting potential.
For neurons at rest, the membrane potential is approximately -70 mV (inside negative). If the inside of the cell becomes less negative, the cell is said to have become depolarized. If the cell becomes more negative inside, the cell is said to have become hyperpolarized. When the cell returns to its resting membrane potential, it is said to be repolarized. Changes in the cell membrane potential of a nerve cell may cause a local change in electrical current, called a graded potential, or may cause a large, propagated change in electrical current, called an action potential. It is through graded potentials and action potentials that the nervous system sends and receives signals.
Graded Potentials
A graded potential is an electrical potential that can vary in amplitude and duration. There are many examples of graded potentials in neurophysiology, including the synaptic potential, the receptor potential, and the muscle end-plate potential (discussed in Chapter 10). Graded potentials are usually produced at a small site on the neuron (synapse, receptor, muscle end-plate) and die out as their charge spreads. Graded potentials are produced by chemical or electrical stimuli, and may be excitatory (depolarizing) or inhibitory (hyperpolarizing). If they are highly excitatory, they may cause an adjacent area of the neuron to depolarize and fire an action potential.
Synaptic Potentials
When a neurotransmitter is released from a presynaptic neuron and binds to a postsynaptic neuron, it will electrically excite (depolarize) or inhibit (hyperpolarize) the postsynaptic cell. If the neurotransmitter depolarizes the postsynaptic cell (makes it more positive inside), the synaptic signal is
P.191
called an excitatory presynaptic potential (EPSP). EPSPs occur if the neurotransmitter opens channels that allow the passage of positive ions, such as sodium or potassium, into the postsynaptic cell. If the binding of the neurotransmitter to the postsynaptic neuron hyperpolarizes the postsynaptic cell (makes it more negative inside), the synaptic signal is called an inhibitory postsynaptic potential (IPSP). IPSPs occur if the transmitter opens channels that allow the passage of negative ions, usually chloride, inside the postsynaptic cell. Of the thousands of incoming signals on a postsynaptic neuron, some will be excitatory and others will be inhibitory. The electrical potential generated in the postsynaptic membrane varies in size, depending on the summation between the IPSPs and EPSPs received, and the amount of neurotransmitter released from each presynaptic cell.
If at the postsynaptic cell, the summation of all EPSPs and IPSPs results in significant excitation of the postsynaptic dendrite or cell body, the electrical excitation will be passed on to the postsynaptic cell. If the summation of EPSPs and IPSPs is inhibitory, the postsynaptic cell will not create an action potential.
Muscle End-Plate Potential
The resting membrane potential of a muscle cell is approximately -90 mV. Stimulation by a motor neuron always causes depolarization at the site where the motor neuron synapses on the muscle cell, called the motor end plate. This depolarization is called an end-plate potential (EPP). The EPP is a graded potential that spreads locally through the muscle fiber and usually causes contraction of the muscle. One motor neuron typically innervates many muscle fibers. One motor neuron and the fibers it innervates are called a motor unit.
Receptor Potential
A receptor potential is the electrical potential produced at the distal end of an afferent neuron after electrical or chemical stimulation.
Specialized cells in sensory organs produce receptor potentials that activate neurons in response to touch, sight, sound, smell, or taste.
A receptor potential is a graded potential; it varies in amplitude and duration and spreads through local current flow. When the receptor potential reaches the cell body, if it is large enough to cause the cell body to depolarize to threshold, the neuron will reach threshold and fire an action potential.
The Action Potential
An action potential is a rapid change in the membrane potential of a neuron or muscle cell. An action potential occurs when depolarization is great enough to cause the cell's voltage-sensitive sodium gates, present throughout the membrane, to burst open. Once the gates open, sodium ions rush inside the cell. The incoming rush of sodium ions causes the charge
P.192
inside the cell to rapidly become more positive, reaching approximately +30 mV in a nerve cell. As the cell becomes more positive, the sodium gates begin to snap shut. At this time, the potassium gates, also affected by the change in membrane potential, open, allowing potassium ions to rush out of the cell. The exit of potassium ions causes the cell to again become negatively charged on the inside. In muscle cells, the action potential also opens calcium gates.
The action potential is an active, transient state of dramatic cell depolarization. Action potentials are different from graded potentials in that they do not vary in amplitude or duration. Instead, action potentials are considered all or none : if the electrical or chemical stimulus, or the EPSP, is great enough to open enough voltage-dependent sodium channels to sufficiently depolarize the membrane, the action potential will occur. If the stimulus is insufficient to cause a certain level of depolarization, the action potential will not occur. The level of depolarization at which a neuron fires an action potential is called the threshold potential. In muscles, it takes one EPP to cause the muscle cell to reach threshold and contract. In nerves, it may take many EPSPs to cause the nerve to reach threshold.
Spread of an Action Potential
When a nerve fiber reaches threshold and fires an action potential, the action potential is propagated at an equal velocity and voltage along the entire length of the axon, to the axon terminals. Propagation of the action potential occurs because neighboring sites on the axon are affected by the change in current generated by the original action potential. The change in current produced by an action potential will be great enough to cause depolarization at a neighboring site on the neuron, and the action potential will be repeated. As the action potential passes down the axon, the part of the axon that has just fired will be refractory for a period of time until the membrane potential returns to the resting level. Propagation of an action potential compared to the local spread of a graded potential is shown in Figure 8-4.
The speed at which an action potential passes along a nerve fiber depends on the diameter of the fiber and whether the fiber is covered by myelin. Because large fibers present less resistance to the flow of current than small fibers, large fibers transmit action potentials faster than small fibers. Fibers coated with myelin pass action potentials faster than uncoated fibers because the myelin acts as insulation to prevent the current from leaking out across the membrane. This allows the action potential to spread by jumps down the axon in a process called saltatory conduction, rather than step by step. As shown in Figure 8-5, the areas where myelin is absent on the axon, called the nodes of Ranvier, contain a large density of sodium channels that open in response to the spread of current and quickly depolarize to threshold, propagating the signal with great speed. Without myelin covering, the current must depolarize each adjacent area of the axon, a process that slows neural transmission considerably.
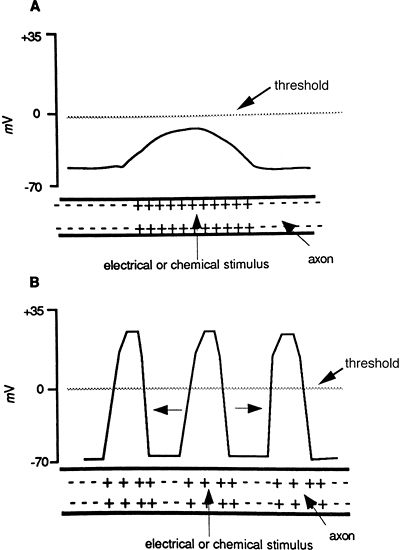 |
Figure 8-4. A graded potential (A) does not reach threshold and passes only a short distance on the membrane and dies out. An action potential (B) fires when depolarization reaches threshold and is repeated along the entire length of the axon. A neuron is capable of propagating an action potential in both directions, although in vivo an action potential starts at one end and travels in one direction. |
P.193
Synaptic Transmission of an Action Potential
Once an action potential reaches the axon terminal, it causes the opening of voltage-gated calcium channels. Although what happens next is not completely understood, it appears that when calcium ions enter into the presynaptic terminal, the ions bind to sites, called release sites, on the inside of the surface of the presynaptic membrane. Binding at the release sites causes them to open, allowing that nerve's neurotransmitter to be discharged into the synaptic cleft. The neurotransmitter is packaged inside vesicles in the
P.194
presynaptic terminal, each containing a few to several thousand molecules of neurotransmitter. The more calcium that enters the pre-synaptic terminal, the greater the number of vesicles that open to release their neurotransmitter. Discharge of a single presynaptic terminal is not generally enough to depolarize to threshold a postsynaptic cell multiple EPSPs from multiple presynaptic neurons are usually required. If there are enough ESPs, the postsynaptic neuron will depolarize and fire its own action potential. If there are not enough EPSPs, the summation of incoming signals on the postsynaptic neuron will not be enough to cause depolarization to threshold, and the signal will not pass on to the next neuron in the chain.
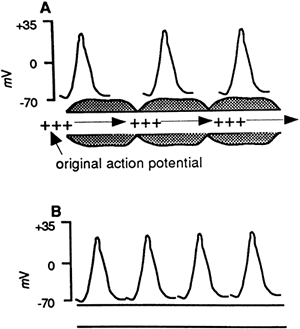 |
Figure 8-5. Propagation in myelinated fiber (A) compared to an unmyelinated fiber (B). Action potentials pass by rapid saltatory conduction in myelinated fibers. |
The Central Nervous System
The brain is a large mass of neural tissue located in the cranium (skull). The brain is composed of neurons and the supporting neuroglial cells. The brain is where reflexes are integrated to maintain the internal environment. It is also the source of several hormones and the site of integration of all sensory information. The brain receives approximately 15% of the cardiac output. Brain cells require glucose for energy metabolism and production of ATP. Figure 8-6 shows the central nervous system divided into the forebrain, midbrain, hindbrain, and spinal cord. The midbrain and hindbrain make up the brainstem. The cerebellum is described separately.
The Forebrain
The forebrain includes the diencephalon, which is located in the core of the brain, and the left and right cerebral hemispheres. The outer shell of
P.195
the cerebral hemispheres is called the cerebral cortex. The cerebral hemispheres are connected together across a longitudinal fissure by axon bundles, one of which is the corpus callosum. The diencephalon includes the epithalamus, thalamus, subthalamus and hypothalamus.
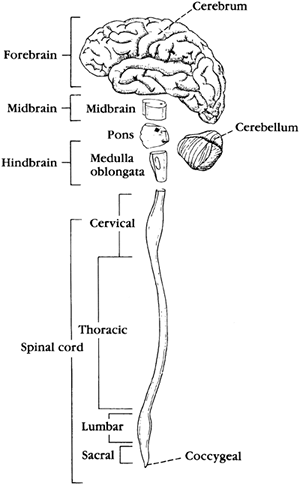 |
Figure 8-6. The central nervous system. |
The Cerebral Cortex
The cerebral cortex is organized horizontally by function and vertically into layers. The vertical layers are clearly delineated and repeated throughout the cortex. The cerebral cortex is the most advanced part of the brain and is responsible for making sense of the environment and initiating thought and goal-oriented behavior. The cortex is called gray matter because of the preponderance of neural cell bodies as opposed to neuronal axons, which tend to appear white. Different sections of the
P.196
cerebral cortex, called lobes, perform different functions. Some parts of the cerebral cortex function as primary sensory areas and directly receive incoming sensory stimuli. These areas are bordered by secondary sensory areas that help interpret sensory stimuli. Other association areas receive information from primary and secondary sensory areas, and from other sites in the cortical and subcortical brain. Association areas allow for complex movements, interpretation and production of language, and appropriate response to friends, enemies, and strangers. The lobes of the cortex are shown in Figure 8-7.
The frontal lobe
The frontal lobe includes the part of the cerebral cortex forward from the central sulcus (fissure or furrow) and above the lateral sulcus. It contains the motor and premotor areas. Broca's area is in the left frontal lobe and controls the production (or articulation) of speech. Many association areas in the frontal lobe receive information from throughout the brain and incorporate that information into thoughts, plans, and behavior. The frontal lobe is responsible for goal-oriented behavior, moral decision making, and complex thought. The frontal lobe also modifies (limits) emotional surges produced in the limbic system and the vegetative reflexes of the brainstem.
Cell bodies in the primary motor area of the frontal lobe send axon projections to the spinal cord, most of which travel in pathways belonging
P.197
to what is described as the corticospinal tract. In the corticospinal tract, motor neurons switch sides: motor information from the left side of the cerebral cortex passes down the right side of the spinal cord and controls motor movements of the right side of the body, and vice versa. Other axons from the motor area travel in extrapyramidal pathways. These fibers control fine motor movement and run outside the corticospinal tract to the spinal cord.
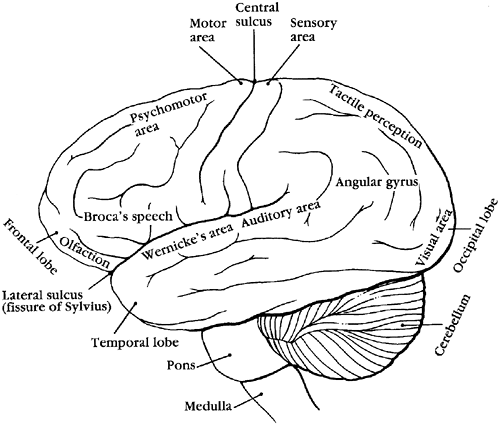 |
Figure 8-7. The brain. |
The parietal lobe
The parietal lobe is the area of the cortex located behind the central sulcus, above the lateral fissure, and extending back to the parieto-occipital fissure. The parietal lobe receives sensory input for touch and pain. Cells of the parietal lobe act as secondary association areas to interpret incoming stimuli. The parietal lobe passes sensory information to many other areas of the brain, including the neighboring motor and visual association areas.
The occipital lobe
The occipital lobe is the posterior lobe of the cerebral cortex. It lies posterior to the parietal lobe and above the parieto-occipital fissure, separating it from the cerebellum. This lobe contains the visual cortex and the visual association areas. The occipital lobe receives information that originated as signals in the retina of the eye.
The temporal lobe
The temporal lobe includes the part of the cerebral cortex extending down from the lateral fissure and back to the parieto-occipital fissure. The temporal lobe is the primary association area for auditory information and includes Wernicke's area, where language is interpreted. It is also involved in the interpretation of smell and is important for the formation and storage of memory. The hippocampus is part of the temporal lobe.
The Diencephalon
The diencephalon structures lie deep between the cerebral hemispheres. The diencephalon includes the thalamus, the hypothalamus, and the basal ganglia.
The thalamus
The thalamus receives all incoming sensory information (except smell) and in turn relays the information through numerous afferent tracts to the rest of the cerebral cortex. Descending fibers from the cerebral cortex also travel down to the thalamus. Function of the cerebral cortex depends on thalamic relay. The thalamus is also part of the reticular activating system, an extensive group of neurons essential in arousal of the individual. The thalamus receives pain information and relays it to the cerebral cortex.
The hypothalamus
The hypothalamus makes up the base of the diencephalon. It is an important endocrine and neural organ responsible for maintaining homeostasis (constancy of the internal environment). The hypothalamus integrates
P.198
and directs information concerning temperature, hunger, autonomic nervous system activity, and emotional status. It also regulates the levels of several hormones, including the pituitary hormones (Chapter 9).
The basal ganglia
The basal ganglia are islands of gray matter lying deep in the diencephalon on either side of the thalamus and upper midbrain that process and influence information in the extrapyramidal nerve tracts. The basal ganglia are important for controlling highly skilled movements that require patterns and quickness of response without intentional thought. The precision of a baseball player and the grace of a ballerina require significant basal ganglia control.
The basal ganglia are composed of several structures that can be anatomically or physiologically separated, including the caudate nucleus, the putamen, and the globus pallidus. The basal ganglia are intimately associated with the substantia nigra and the subthalamic and red nuclei. Virtually all projections to and from the basal ganglia go through the thalamus. Lesions of the basal ganglia cause repetitive movements, grimaces, and tremors, as seen with Huntington's disease (chorea) and Parkinson's disease.
The Limbic System
The limbic system is a diffuse grouping of neurons from different areas of the brain. Neurons in the limbic system include fibers from all lobes of the forebrain and extensive connections from the hypothalamus and thalamus. Midbrain and hindbrain areas also send projections that contribute to the limbic system. The hippocampus is considered part of the limbic system and plays an important role in coding and consolidating memories. The amygdala, also considered part of the limbic system, is involved in the production of emotions, aggression, and sexual behavior. Learning and behavior are also influenced by several limbic system structures and connections.
The Brainstem
The brainstem, or stalk of the brain, is made of up of the pons, medulla oblongata, and mesencephalon (midbrain). In the brainstem are cells that control cardiovascular and respiratory system functions. Neurons pass through the brainstem and carry motor information to and from the cerebral cortex, controlling equilibrium. Ten of the twelve cranial nerves, controlling motor and sensory function of the eyes, face, tongue and neck, leave from the brainstem. The secretory and motor functions of the gastrointestinal tract and the sensory functions of hearing and taste also are controlled by the cranial nerves.
Reticular Formation
Running through the brainstem is a network of many small, branched neurons, called the reticular formation. These neurons include ascending
P.199
and descending tracts, some of which cluster to form centers that control swallowing, vomiting, and respiratory and cardiovascular reflexes. The reticular formation is also essential for wakefulness and is necessary to focus attention. Functioning of the reticular formation is essential for life.
Wakefulness
Various neurons in the reticular formation send information to higher brain areas to maintain wakefulness and arousal. These neurons and their projections are part of a functional rather than anatomic group of cells, called the reticular activating system (RAS). The RAS maintains wakefulness, attention, and concentration. The RAS is stimulated by all sensory input, including painful stimuli.
Sleep
The process of sleep is also under the control of the reticular formation. Like wakefulness, sleep is an active process. It occurs when certain centers in the brainstem send inhibitory signals to neurons throughout the RAS. These inhibitory signals appear to result from release of the neurotransmitter serotonin by the reticular formation cells. Serotonin inhibits RAS firing, temporarily ending conscious behavior. Serotonin levels in the brain eventually decrease, and the person wakes up. Sleep and wakefulness normally follow a cyclic pattern unless that pattern is blocked, changed, or interrupted.
The Cerebellum
The cerebellum sits in the hindbrain posterior to the brainstem. The cerebellum helps maintain balance and is responsible for the smooth skeletal muscle responses that give grace and direction to voluntary movements. It controls fast, repetitive movements required for activities such as typing, piano playing, and bike riding.
The Spinal Canal
The spinal canal or vertebral column is a long, thin column extending from the base of the skull to the sacrum (tailbone). Running down the center of the spinal canal is the spinal cord. It is filled with cerebrospinal fluid (CSF) and surrounded by the bony vertebral column, which extends beyond the terminus of the spinal tract and offers protection to the delicate nerves inside. The spinal cord consists of interneurons whose axons travel up and down in organized tracts. Incoming to the ascending tracts are axon terminals that carry sensory information from peripheral afferent neurons. Many axon terminals synapse in the cord on an interneuron. If the summation of the various incoming IPSPs and EPSPs results in the interneuron reaching threshold, the interneuron will fire an action potential and pass the information further into the central nervous system. The sensory neuron may also stimulate a spinal reflex. This is accomplished
P.200
when a sensory neuron synapses in the spinal canal directly on the dendrites or cell body of a motor neuron (monosynaptic reflex), or when it synapses on an interneuron that secondarily activates a motor neuron (polysynaptic reflex).
Also present in the spinal cord are descending interneurons that innervate dendrites and cell bodies of efferent nerves. Efferent nerves leave the spine in tracts and innervate muscle or endocrine cells.
Dorsal and Ventral Roots
Groups of afferent nerves entering at each level of the cord on the dorsal (toward the back) side are called dorsal roots. Efferent nerves leave each level of the cord in groups on the ventral (toward the front) side. These are called ventral roots. Dorsal and ventral roots at a given level of the spinal cord join together outside the cord to form 1 of 31 pairs of spinal nerves.
Gray and White Matter
The spinal cord can be separated into gray and white matter. Gray matter occupies the center of the tract and is filled with interneurons, cell bodies, dendrites of efferent neurons, axons of afferent neurons, and various support cells. The white matter, consisting mostly of myelinated ascending and descending tracts, surrounds the gray matter.
The Meninges
The meninges are thin membranes surrounding the brain and spinal cord. There are three meninges: the dura mater ( thick mother ) on the outside, the arachnoid (spider-like) as a middle layer, and the pia mater ( little mother ) lying immediately above the brain. Spaces between the layers are filled with CSF.
The space above the dura mater is called the epidural, and the space below the dura mater but above the arachnoid is called the subdural. The epidural and subdural spaces contain many small blood vessels. Damage to these vessels leads to blood accumulating in the epidural or subdural spaces. CSF circulates in the subarachnoid space (beneath the arachnoid, above the pia mater).
Cerebrospinal Fluid and the Ventricles
Cerebrospinal fluid is a clear fluid surrounding the brain and spinal cord. The CSF circulates in the subarachnoid space, and offers the brain protection against physical jarring. There is some exchange of nutrients and waste products between the CSF and the neural tissue. Although CSF is formed from plasma that flows through the brain, its concentration of electrolytes and glucose differs from that of plasma.
Cerebrospinal fluid is formed as a result of filtration, diffusion, and active transport across special capillaries into the ventricles (cavities) of the brain,
P.201
especially the lateral ventricle. The capillary network responsible for CSF formation is called the choroid plexus. Once in the ventricles, CSF flows toward the brainstem. Through small holes in the brainstem, CSF circulates to the surface of the brain and spinal cord. At the surface of the brain, CSF enters the venous system and returns to the heart. Thus, CSF is continually recirculated through and over the central nervous system. If the ventricle conduction pathways for CSF become blocked, fluid can accumulate, which results in a buildup of pressure inside and on the surface of the brain.
The Blood-Brain Barrier
The blood-brain barrier refers to the unique structure of the brain vascular system that prevents the passage of materials from the blood to the cerebrospinal fluid in the brain. The blood-brain barrier results from tightly fused endothelial cells present in the brain capillaries and from cells lining the ventricles that limit diffusion and filtration. Special transport functions regulate the fluid that crosses out of the general circulation to bathe brain cells. The blood-brain barrier protects delicate brain cells from exposure to potentially harmful substances. Many drugs and chemicals cannot cross the blood-brain barrier.
Brain Blood Flow and Brain Metabolism
The brain receives approximately 15% of the cardiac output. This high rate of blood flow is required to meet the brain's continually high demands for glucose and oxygen.
The brain is unique in that it normally uses only glucose as a source for oxidative phosphorylation and the production of adenosine triphosphate (ATP). Unlike other cells, brain cells do not store glucose as glycogen; therefore, the brain must continually receive oxygen and glucose through brain blood flow. Oxygen deprivation for as little as 5 minutes, or glucose deprivation for 15 minutes, can cause significant brain damage. Brain function depends so much on blood flow that it is possible to identify which parts of the brain are performing which tasks by measuring brain blood flow during specific brain activities.
Studies have shown that when performing a burst of mental work, the brain initially produces ATP by anaerobic glycolysis, rather than oxidative phosphorylation. Anaerobic glycolysis depends on glucose but does not require oxygen. The brain does this even if oxygen is readily available. The result is a rapid utilization and depletion of glucose, with a corresponding increase in oxygen levels. Within a short period, the brain begins oxidative phosphorylation.
Intracranial Pressure
The pressure inside the cranium is called intracranial pressure (ICP). ICP is determined by the volume of blood in the brain, the volume of CSF, and
P.202
the volume of brain tissue. Normally, ICP ranges from 5 to 15 millimeters of mercury (mmHg).
The Peripheral Nervous System
The peripheral nervous system consists of nerves traveling between the brain or spinal cord and the rest of the body. There are 12 nerve pairs traveling to and from the brain and 31 pairs traveling to and from the spinal cord. The peripheral nervous system can be separated into afferent and efferent divisions. In all spinal nerves and most cranial nerves, afferent and efferent fibers travel together in opposite directions. Some cranial nerves carry only afferent information. Afferent neurons convey information to the central nervous system from all sensory organs, pressure and volume receptors, temperature receptors, stretch receptors, and pain receptors. Efferent neurons deliver neural stimulation to muscles and glands. Efferent neurons belong to either the autonomic or the somatic nervous system.
The Autonomic Nervous System
Autonomic nerve fibers leave the spinal cord and innervate smooth and cardiac muscle and the endocrine and exocrine glands. Autonomic nerves fibers are considered involuntary because there is little conscious control over their function. The two divisions of the autonomic nervous system, the sympathetic and parasympathetic divisions, are shown in Figure 8-8. Sympathetic and parasympathetic nerves innervate many of the same organs but typically cause opposite responses. The cell bodies of these neurons lie in the brain or spinal cord. In both divisions of the autonomic system, two nerve fibers participate in the efferent pathway.
The Sympathetic Nervous System
The first fibers of the sympathetic nerves, called the preganglionic fibers, leave from the thoracic or lumbar regions of the spine. Soon after leaving the spine, a preganglionic fiber joins other preganglionic fibers to form an autonomic ganglion. At this point, the preganglionic fiber synapses on the second nerve fiber of the system, the postganglionic fiber, and releases acetylcholine, which causes the postganglionic fiber to fire an action potential. From the autonomic ganglia, the postganglionic fiber travels to its target organ, the muscle or gland. The sympathetic postganglionic fiber usually releases the neurotransmitter norepinephrine. Target organ receptors for norepinephrine are called adrenergic receptors.
The Parasympathetic Nervous System
The fibers of the parasympathetic nervous system leave the brain in the cranial nerves or leave the spinal cord from the sacral area. The preganglionic fiber of the parasympathetic system is typically long and travels to an autonomic ganglion located near the target organ. Preganglionic
P.203
parasympathetic nerves release acetylcholine that then stimulates the postganglionic fiber. The parasympathetic postganglionic fiber then travels a short distance to its target tissue, a muscle or a gland. This nerve also releases acetylcholine. Preganglionic acetylcholine receptors for sympathetic and parasympathetic fibers are called nicotinic receptors. Postganglionic acetylcholine receptors are called muscarinic receptors. These names relate to the experimental stimulation of the receptors by nicotine and muscarine (a mushroom poison).
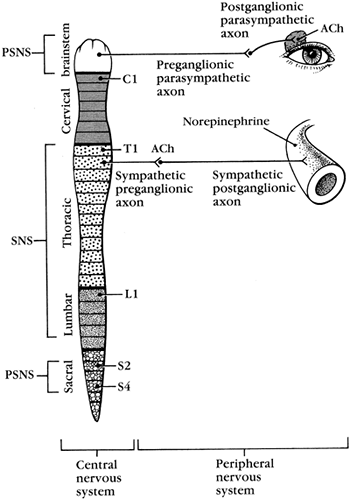 |
Figure 8-8. Sympathetic and parasympathetic systems. |
Functions of the Sympathetic and Parasympathetic Nerves
The sympathetic nervous system innervates the heart, causing an increase in heart rate and strength of contraction. Sympathetic nerves innervate all large and small arteries and veins, causing constriction of all vessels except the arterioles supplying skeletal muscle. Sympathetic nerves innervate the smooth muscle of the gut, causing decreased motility, and the smooth
P.204
muscle of the respiratory tract, causing bronchial relaxation and decreased bronchial secretions. Sympathetic stimulation affects the liver, stimulates secretions of the sweat glands, and is responsible for ejaculation during male orgasm.
Parasympathetic fibers innervate the heart, slowing the heart rate, and the gut, causing increased motility. Parasympathetic nerves innervate bronchial smooth muscle, causing airway constriction, and the genitourinary tract, causing erection in the male.
The Somatic Nervous System
Somatic nerves of the peripheral nervous system consist of efferent motor neurons that leave the brain or spinal cord and synapse directly on skeletal muscle cells. Motor neurons are large myelinated nerves that release acetylcholine at the neuromuscular junction. Acetylcholine binds to receptors on a specialized area of the muscle cell, called the end plate. Binding of acetylcholine causes the muscle cell to reach threshold, resulting in an action potential and the opening of calcium channels (gates) in the membrane. This leads to an increase in intracellular calcium and contraction of the skeletal muscle fiber. There are no inhibitory motor neurons.
Support Cells
Cells in the central and peripheral nervous systems function to support nerve cells by providing nutrients, forming myelin, and clearing away cell debris and other material from the neuronal area. In the central nervous system, these cells are called neuroglia and are more numerous than nerve cells. Examples of neuroglia include the oligodendroglia cells, which make myelin; microglia cells, which support immune function in the central nervous system; and astrocytes, which carry nutrients from capillaries to nerve cells and remove excess neurotransmitters and ions that accumulate around neurons, thus ensuring optimal osmotic and ionic concentrations. Recently, neuroglial cells have received increased attention, with a gathering body of research indicating that they, especially the astrocytes, actually communicate with neighboring neurons. Via these interactions, neuroglial cells appear to influence the formation of synapses and help determine which neural connections become stronger and which become weaker over time. This function has important implications for learning and memory. In the peripheral nervous system, Schwann cells provide a source of nutrients for the neurons and produce myelin.
How the Brain Works
Advances in technology, especially positron emission tomography (PET) and magnetic resonance imaging (MRI), have offered insights into how the brain allows us to think, remember, interpret stimuli, and understand and use language.
P.205
It appears that the mind breaks down complex functions into discrete components of a specific job, and delegates these components to specific neuronal networks. The outcomes from the different networks are then reassembled in a pattern that allows the brain to perform the activity or give meaning to a stimulus.
Communication Pathways
Perhaps best studied are the many steps involved in participating in a conversation. To begin with, spoken sounds are carried from auditory receptors in the ears through the thalamus to the primary auditory area of the cortex. At the same time, nonverbal visual clues are sent from the retina through the thalamus to the primary visual area. From the auditory cortex, signals travel to an area in the left temporal lobe, called Wernicke's area, where meaning is assigned to the words; and to associative areas where the impact of the words is perceived by the listener. To respond verbally, an area of the frontal lobe, called Broca's area, is activated, and an appropriate response is formulated. The reply is then spoken by activating the primary motor area of the brain, then passing the motor signals through the thalamus and down the spinal cord, resulting in activation of motor neurons to the face and throat.
The processing of various tasks by the brain is adaptable; as one becomes proficient at a task, the patterns of processing can change, becoming faster and often more efficient. One can learn to speak before thinking and listen without paying attention.
Memory
Memory is the internal recording of a prior event. The formation of memory is a multistep process that involves (1) focusing attention on a selected event, name, or number, to the exclusion of background events, (2) rehearsing the information, and (3) consolidating the information into chemical storage in the brain.
Focusing attention on one event or piece of information allows that information to enter short-term memory storage. This is an active state wherein the new event is compared with previous experiences. Short-term memory is considered to be the working memory; it is of limited capacity and if the information is not continually rehearsed or attended to, it will be lost when a new input arrives to distract attention. However, if the information is rehearsed, it will stay in short-term storage until it can be consolidated into long-term memory storage. Long-term memory is theoretically unlimited and permanent. Long-term memory depends on several excitatory neurotransmitters, including acetylcholine, dopamine, norepinephrine, and glutamate, and on hormones released during stressful events, including adrenocorticotropic hormone (ACTH), vasopressin, and epinephrine. Inhibitory transmitters, including GABA, can reduce the likelihood of consolidating memory from short-term storage to long-term.
P.206
When a short-term memory is consolidated into a long-term memory, it is done so by breaking the information to be remembered into separate units that are then processed in specific areas of the brain. For example, a visual experience is broken down into discrete attributes of color, shape, and size, and these attributes are stored separately. There are also two general types of long-term memory. Declarative memory is conscious memory for facts and events. This type of memory requires a well-functioning medial temporal lobe, which includes the hippocampus, and structures in the diencephalon. How these various parts of the brain interact during memory coding and retrieval is not known. The declarative memory is stored in the cerebral cortex, but again, how this occurs is not understood. Nondeclarative memory is involved with skill learning, repetition, and classical conditioning. Nondeclarative memory involves unconscious recollection and requires an intact cerebral cortex, basal ganglia, and cerebellum. With most types of dementia, declarative memory is lost before nondeclarative memory. Strokes (brain attacks) may interfere with nondeclarative and declarative memory.
Pathophysiologic Concepts
Alterations in Consciousness
Consciousness is the full awareness of self, location, and time in any environment. To be fully conscious, an intact reticular activating system is required, as is the functioning of higher brain centers in the cerebral cortex. Connections through the thalamus must also be intact.
Alterations in consciousness typically begin with disruption in diencephalon functioning, characterized by dullness, confusion, lethargy, and finally stupor as the person becomes difficult to arouse. Continued decreases in consciousness appear with midbrain dysfunction and are characterized by deepening of the stupor state. Finally, dysfunction of the medulla and pons may occur, resulting in coma. This progressive decrease in consciousness is described as rostral-caudal progression.
Alterations in Pupil Responses
The ability of our eyes to dilate or constrict, rapidly and equally, depends on an intact brainstem. Cerebral hypoxia and many drugs change pupil size and reactivity. Therefore, pupil size and reactivity offer valuable information concerning brain integrity and function.
Important pupil changes seen with brain damage are pinpoint pupils seen with opiate (heroin) overdose and bilaterally fixed and dilated pupils usually seen with severe hypoxia. Fixed pupils are typically seen with barbiturate overdose. Brainstem injury presents with pupils fixed bilaterally in the midposition.
P.207
Alterations in Eye Movements
In a fully conscious person, the steady gaze of the eyes at rest results from an intact cerebral cortex exerting control over the brainstem. With brain injury that involves loss of cortical function, the eyes typically rove and move together toward or away from the side of the brain injured, depending on the type of injury. Loss of higher brain centers results in reflexive eye movements, called doll's head movements. A doll's head movement is that which occurs when the eyes stare forward, always following the position of the head. Normally, when an individual's head is passively turned to one side, the eyes move to face the previous, forward direction.
With injury to the brainstem, loss of ocular movement occurs, and the eyes become fixed in a direct forward position. A skewed deviation, with one eye looking up and one down, suggests a compressive injury to the brainstem. Normal involuntary cyclic movements of the eyeball (nystagmus responses) in response to ice water delivered into the ear are lost with cortical and brainstem dysfunction.
Alterations in Breathing Pattern
Brainstem Damage
The respiratory center in the lower brainstem controls respiration based on hydrogen ion concentration in the surrounding CSF. Damage to the brainstem causes irregular and unpredictable patterns of breathing. Opiate overdose damages the respiratory center and results in a gradual decline in the breathing rate until respiration ceases.
Cerebral Damage
A higher brain center normally maintains the rhythmic, regular breathing patterns seen in healthy individuals. This control center is lost with cerebral damage, and the individual begins to breathe in a pattern dependent on brainstem carbon dioxide and the hydrogen ion it produces. This type of carbon dioxide dependent breathing is called post-hyperventilation apnea. In this pattern, respirations cease (apnea) until carbon dioxide builds up to a certain threshold, which causes the individual to hyperventilate (increase his or her respiratory rate) until the carbon dioxide is removed. At this point respirations cease again.
Cheyne-Stokes respiration also involves breathing based on carbon dioxide levels. In this case the respiratory center is over-responsive to carbon dioxide, which results in a breathing pattern of smooth increases in rate and depth (crescendo breathing) that progresses until a certain carbon dioxide level is reached. The rate and depth of respirations then decrease smoothly until apnea occurs (decrescendo breathing). Cheyne-Stokes respiration is, like post-ventilation apnea, seen with damage to the cerebral hemispheres, and is often associated with metabolically induced coma.
P.208
Alterations in Motor Responses and Movement
Abnormal motor responses include inappropriate or absent movements in response to painful stimuli. Brainstem reflexes such as sucking and grasping responses will occur if higher brain centers have been damaged. Flexion and rigidity of limbs also are motor responses indicative of brain damage. Muscle conditions that indicate abnormal brain function include hyperkinesia (excessive muscle movements), hypokinesia (decreased muscle movements), paresis (muscle weakness), and paralysis (loss of motor function). Specific loss of cerebral cortex functioning, but no loss of brainstem function, results in a particular body posture called flexor posturing. Flexor posturing is characterized by flexion of the upper extremities at the elbows and external rotation and extension of the lower extremities. This posture may be unilateral or bilateral. Extensor posturing occurs with severe injury to higher brain centers and the brainstem and is characterized by rigid extension of the limbs and neck.
Dysphasia
Dysphasia is impairment of language comprehension or production. Aphasia is total loss of language comprehension or production. Dysphasia usually results from cerebral hypoxia, which is often associated with a stroke but can result from trauma or infection. Brain damage leading to dysphasia usually involves the left cerebral hemisphere.
Broca's Dysphasia
Broca's dysphasia results from damage to Broca's area in the frontal lobe. Persons with Broca's dysphasia will understand language, but their ability to meaningfully express words in speech or writing will be impaired. This is called expressive dysphasia.
Wernicke's Dysphasia
Wernicke's dysphasia results from damage to Wernicke's area in the left temporal lobe. With Wernicke's dysphasia, verbal expression of language is intact, but meaningful understanding of spoken or written words is impaired. This is called receptive dysphasia.
Agnosia
Agnosia is the failure to recognize an object because of the inability to make sense of incoming sensory stimuli. Agnosia may be visual, auditory, tactile, or related to taste or smell. Agnosia develops from damage to a particular primary or associative sensory area in the cerebral cortex.
P.209
Persistent Vegetative State
A persistent vegetative state results from the loss of functioning of the cerebral hemisphere. It is a state in which a person is without cognitive function, is unaware of self, place, and time, and is unresponsive to the external environment. This state can occur after several different types of brain injuries and, as its title suggests, can persist for years. Although consciousness is lost, brainstem and cerebellum function remain intact; therefore, respiration, cardiovascular control, maintenance of body temperature, and certain brainstem reflexes such as yawning, grasping, and sucking will continue. Eyes may be open or shut, and a sleep-wake cycle will be followed, but there is no conscious perception of events or deliberate action. This diagnosis requires that the condition exists for at least one month.
A coma is somewhat similar, except there is no opening of the eyes, and no sleep-wake cycle. Comas and persistent vegetative states have legal and ethical implications for the families of those affected and for society.
Brain Death
Brain death is irreversible loss of cerebral hemisphere, brainstem, and cerebellum function. Consciousness is lost, as is maintenance of respiration, cardiovascular, and temperature control function. There is no sleep-wake cycle, no pain response, and no reflexes. The electroencephalogram (EEG) is flat in an individual with brain death.
Establishing brain death has several legal implications. A patient cannot be legally discontinued from life support without prior living will instructions unless brain death is established. Organ donation is allowed only when brain death is established. Unfortunately, a donated organ is more likely to be healthy when taken from an individual before brain death occurs.
Dementia
Dementia is a loss of intellectual functioning without a loss of arousal or vegetative functioning. Memory, general knowledge, abstract thought, judgment, and interpretation of written and oral communication may be affected. Dementia may be caused by infection, drugs, trauma, or tumors. Biochemical disturbances and metabolic imbalance may also cause dementia. Some dementia is reversible if the initiating insult can be relieved. Other types of dementia, such as that caused by Alzheimer's disease, are progressive and irreversible.
Increased Intracranial Pressure
Intracranial pressure may increase with increases in cranial blood, CSF, or tissue. A significant increase in intracranial pressure is called intracranial
P.210
hypertension. Intracranial hypertension causes delicate neurons and capillaries in the brain to become compressed, leading to hypoxia, neuronal injury and death, inflammation and swelling, and ultimately progressive deterioration of brain function. If intracranial pressure reaches systemic mean arterial pressure, blood flow to the brain will stop and the individual will die.
Causes of Increased Intracranial Pressure
Shifts in intracranial pressure are common, and occur with stimuli such as straining at stool, coughing, and sneezing. More significant increases in intracranial pressure can occur with conditions that increase blood flow to the brain, or that block blood flow out of the brain. Anything that significantly increases CSF production or blocks CSF outflow can increase intracranial pressure. Any increase in tissue mass (e.g., that associated with a growing brain tumor) can increase intracranial pressure.
Edema and Swelling of the Interstitial Space
Important sources of increased intracranial pressure are any stimuli that lead to edema and swelling of the interstitial fluid compartment. Infection and inflammation are associated with interstitial swelling and edema resulting from the release of vasoactive mediators of inflammation that stimulate increased capillary blood flow and increased capillary permeability (Chapter 5). Bacterial toxins also cause significant cellular destruction and initiate capillary destruction, again causing interstitial swelling. Therefore, infection and inflammation significantly increase intracranial pressure.
Severe hypertension may increase intracranial pressure by causing filtration of plasma into the interstitial space, leading to edema and swelling. Severe trauma to the head, a burst aneurysm, or a hemorrhagic stroke cause bleeding in the brain, which increases intracranial pressure by acting as a source of expanding tissue (blood) and by causing inflammation with swelling and edema.
The Stages of Intracranial Hypertension
As volume in the brain increases, the brain directs response mechanisms designed to minimize increases in pressure and reduce the extent of brain damage. The response of the brain to increased intracranial pressure is called compensation. However, if the volume in the brain continues to increase, compensation will eventually lose its effectiveness. The brain goes through four stages in response to increased intracranial pressure.
Stage 1
An increase in one of the three volumes in the brain (blood, CSF, or tissue) is normally compensated for by a decrease in one or both of the other volumes. If successful, compensation will allow intracranial pressure to
P.211
remain within the normal range even with a significant increase in one of the brain volumes. If there is increased volume in one compartment, but normal intracranial pressure because of compensation, the brain is said to be in stage 1 of intracranial hypertension. Usually, this stage involves decreased CSF production or increased CSF reabsorption, followed by increased arterial constriction to decrease blood flow into the brain. Persons in stage 1 may demonstrate only subtle behavioral changes, primarily drowsiness and slight confusion.
Stage 2
If the volume continues to increase despite early compensatory mechanisms, intracranial pressure begins to increase significantly and the individual is said to be in stage 2. This stage would occur with the progression of a tumor or continual bleeding from a severed artery or vein. During stage 2, the brain responds by constricting cerebral arteries in an attempt to reduce pressure by reducing blood flow. Reducing blood flow, however, leads to cerebral hypoxia and hypercapnia (increased carbon dioxide levels) and deterioration of brain function. Clinical signs include decreased level of consciousness, alterations in breathing pattern, and pupillary changes.
Stage 3
In response to worsening hypoxia and hypercapnia, the cerebral arteries undergo reflex dilation, with the goal of increasing brain oxygen delivery. As blood volume increases, however, intracranial pressure increases further, thereby worsening the situation. This cycle of increasing hypoxia leading to increasing pressure, thereby worsening the hypoxia, is called decompensation. With the onset of decompensation, the individual is said to enter stage 3 of intracranial hypertension.
In stage 3, the volume-pressure curve develops so that additional small changes in intracranial volume produce large changes in pressure. Fast-rising pressure compresses the arterioles and capillaries, worsening the hypoxia and the hypercapnia, and damaging the neural cells. The result is a pronounced decrease in consciousness, altered respiratory pattern, and loss of pupillary reflexes. As the brain senses worsening hypoxia and hypercapnia, it responds with reflexes geared toward increasing systemic mean arterial pressure in an attempt to increase its own oxygenation. A dramatic increase in systemic blood pressure only serves to further increase intracranial pressure, accelerating the destruction of the brain cells. Cerebral blood flow slows, and consciousness and reflexes are usually lost.
Stage 4
As the swelling and pressure in one compartment of the brain become very high, herniation (bulging) into another compartment occurs. Herniation increases pressure in the other compartment, and eventually the whole brain becomes involved. When intracranial pressure reaches mean systolic
P.212
pressure, cerebral perfusion stops. The volume-pressure curve demonstrating the stages of increased intracranial pressure is shown in Figure 8-9.
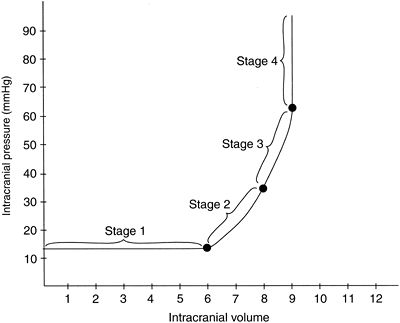 |
Figure 8-9. Intracranial volume versus pressure. |
Treatment of Intracranial Hypertension
Treatment of intracranial hypertension begins with effective monitoring of intracranial pressure (ICP). Treatment also includes osmotic diuretics (mannitol) to reduce blood volume and steroids to decrease inflammation. It is essential that patients suspected of suffering from increased intracranial pressure have accurately measured cerebral perfusion pressure. Hyperventilation is contraindicated under most conditions because it worsens cerebral ischemia.
Tests of Neurologic Functioning
There are several methods to measure neuronal and brain electrical activity and observe for malformations, injuries, or tumors. Some of these techniques are presented briefly.
Electromyography
Electromyography (EMG) measures peripheral nerve function by recording the electrical activity of a motor nerve muscle cell unit. EMG is used to diagnose, describe, and monitor neuromuscular pathology in patients who are suspected of suffering a disorder in neural transmission or muscle cell function.
P.213
Electroencephalography
Electroencephalography (EEG) measures electrical activity occurring in the brain through electrodes placed on the scalp. This technique offers a fast, real-time picture of brain activity. EEG is capable of picking up unusual brain wave signals indicative of brain damage or seizure activity. It is limited by an inability to accurately identify which area of the brain is generating the electrical signal, especially when the areas of desired evaluation are located deep in the brain.
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) captures what is happening in the brain physiologically before, during, and after an individual performs a task. MRI relies on the principle that each atom in the body will act like a little compass needle and line up in a predictable direction when exposed to a magnetic field. Signals unique to each atom are emitted and images can be formed from this information using specific computer programs. Organs are reproduced in more anatomic detail than by radiograph alone.
Magnetic resonance imaging has made a dramatic impact on the study of brain function and pathophysiology. It allows an investigator to non-invasively study oxygen concentration in the brain as an individual performs a task. Because the brain quickly shifts to anaerobic glycolysis with an activity spurt, oxygen levels increase in venous blood leaving an area performing a task. By looking for areas with high oxygen levels, researchers can identify blood flow patterns and active areas of the brain. With MRI, tissue structure and integrity can be imaged clearly. Automated software programs exist that allow clinicians and researchers to associate MRI data with specific anatomical regions, and to compare data originating from one region of the brain with data from other regions, including regions both participating and not participating in the activity.
Advantages of MRI include the absence of ionizing radiation and the high sensitivity the technique offers. It is the imaging technique of choice for most neurologic conditions. A limitation of MRI is the length of time required to scan the brain. Although ultrafast MRI is available, the ultrafast techniques are not as sensitive.
Computed Tomography
Computed tomography (CT) scanning involves computer analysis of multiple radiologic images. In a CT scan, an x-ray beam is rotated around the patient, and passes successively through tissue from multiple directions. The pictures are then recreated by the computer to give a realistic three-dimensional representation of brain structure. Contrast media may be injected before the x-rays to improve fine detail of structures. CT scanning is readily available in most emergency rooms, and is used for rapid evaluation of neurologic emergencies. It is excellent for visualization of bone and is able to detect acute hemorrhage. It is also the technique
P.214
of choice when patients cannot undergo MRI due to the presence of foreign objects in the eye, pacemakers, or metal prostheses. Limitations of a CT scan involve multiple x-ray exposure and less detailed pictures than MRI.
Positron Emission Tomography
Positron emission tomography (PET) involves intravenous injection or inhalation of a positron-emitting isotope, followed by sequential radiographs of the skull that monitor the decay of the isotope in tissues that take up the label. This procedure allows the investigator to study the distribution of a particular substance in the brain. It also allows the investigator to anatomically map the brain and determine blood flow patterns. By observing the flow of blood or the uptake of the isotope in different areas of the brain as an individual performs a task, one can identify which areas of the brain are most responsible for that task. Radiolabeled water is often injected when determining cerebral blood flow. Radiolabeled fluorodeoxyglucose is used to measure cerebral glucose metabolism, and carbon-11 is used to identify biochemical changes in the brain indicative of multiple sclerosis and Parkinson's disease.
Single-photon emission computed tomography (SPECT) also involves the injection of a radionuclide to provide information on metabolic processes and blood flow, but involves decay to only a single photon. SPECT is typically less expensive than PET but offers poorer resolution of structure and metabolic activity.
Both PET and SPECT have been used to detect changes in neuropharmacological parameters such as receptor numbers and neurotransmitter levels. These techniques allow researchers to determine the effects of substance abuse in the brain. For example, an acute increase in dopamine release, a decrease in serotonin transporter number, and increased occupancy of opiate receptors all have been documented to occur in response to various mood-altering substances.
Typically, blood flow patterns are determined at rest and during a specific task. Limitations to PET and SPECT involve the invasiveness inherent in injecting a radionuclide, although the ones chosen typically emit low levels of radiation and decay rapidly. Another limitation of these techniques is that neurons typically react faster than blood flow pattern can change, so some brain activity will be missed.
Ultrasonography
Ultrasound techniques use reflected sound to measure blood flow velocity, which is important for assessment of cerebral blood flow when evaluating ischemic cerebrovascular disease. Ultrasound is used during surgical procedures on the brain to study the development of vascular spasm or blockage in real time. It is limited by the high degree of user proficiency required for its performance.
P.215
Conditions of Disease or Injury
Seizure Disorder
A seizure is the sudden, uncontrolled discharge of brain neurons, which produces changes in brain function. Seizures result when certain cerebral neurons exist in a hyperexcitable or easily depolarized state. These neurons appear to have a less-negative-than-normal membrane potential at rest, or are missing important inhibitory connections. As a result, this group of neurons, called an epileptogenic focus, is always close to the threshold potential required for firing an action potential. Neurons of the epileptogenic focus respond to levels of stimuli that do not produce disorderly discharge in other neurons.
Once an epileptogenic focus begins to fire action potentials, the resulting current can spread to neighboring cells, causing them to discharge as well. It may spread to both sides of the brain and throughout cortical, subcortical, and brainstem areas. If the seizure begins diffusely throughout the cerebral cortex and includes both sides of the cortex, it is called a generalized seizure, and consciousness is always lost. If the seizure arises from a discrete focus and is limited to one side of the brain, it is called a partial seizure, and consciousness is usually not lost. Partial seizures may progress and become generalized. The time of unconsciousness after any generalized seizure is called the postictal phase.
As a seizure continues, inhibitory neurons in the brain fire and cause the neuronal discharge to slow, then stop. If one seizure is followed by a second or third seizure before the individual regains consciousness, status epilepticus is said to occur.
Seizure Syndromes
Generalized seizures include tonic-clonic seizures, characterized by sudden onset of rigid, intense contractions of arm and leg muscles (tonic seizure), followed by rhythmic contraction and relaxation of the muscles (clonic seizure). This is the most common type of generalized seizure and has been formally termed a grand mal seizure. Other generalized seizures may be purely tonic, purely clonic, or atonic. Absence seizures, frequently seen in children, are characterized by staring and sudden cessation of activity. Generalized seizures may occur idiopathically (for no known reason) or after brain trauma, infection, tumor, or bleeding.
Partial or focal seizures include simple partial seizures, during which consciousness is not impaired, and complex partial seizures, in which consciousness is impaired. Partial seizures may occur idiopathically or after brain damage.
Consequences of a Seizure
During a seizure, cerebral oxygen demand increases more than 200%. If this oxygen demand cannot be met, brain hypoxia and brain damage may
P.216
occur. Seizures that continue for extended periods, or the occurrence of status epilepticus, greatly increase the chance of brain damage.
Other consequences of a seizure, and especially of repeated seizures, include social isolation and reduced employment. Even the mildest forms of childhood epilepsy may be associated with lifelong social effects, including a reduced likelihood of marriage, childbearing, and academic achievement, even with normal intellectual functioning.
Causes of Seizure Disorders
Seizures can occur in anyone who experiences severe hypoxemia (decreased oxygen in the blood), hypoglycemia (decreased glucose in the blood), acidemia (increased acid in the blood), alkalemia (decreased acid in the blood), dehydration, water intoxication, or high fever. Drug withdrawal, drug abuse, and toxemia in pregnancy also may cause seizures. Some people seem to have a lower seizure threshold and therefore are more prone to seizures than others, suggesting a genetic tendency toward seizures. Seizures caused by metabolic disturbances are reversible if the initiating stimulus is removed. Syncope (fainting) is often misdiagnosed as a seizure because some muscle movements may be similar. Unconsciousness and muscle jerking related to fainting rarely last longer than 5 to 10 seconds, and fainting is not associated with postictal symptoms such as fatigue.
Epilepsy
Epilepsy refers to a seizure that occurs without a reversible, metabolic cause. Epilepsy may be a primary or secondary condition. Primary epilepsy develops spontaneously, usually in childhood, and has a genetic predisposition. Mapping of several genes associated with primary epilepsy is underway. Secondary epilepsy occurs as a result of hypoxemia, head injury, infection, stroke, or central nervous system tumor. Adult-onset epilepsy is usually caused by one of these incidents.
Clinical Manifestations
Partial seizures may be associated with:
Facial movements or grimaces
Jerking beginning in one part of the body, which may spread
Sensory experiences of sights, smells, or sounds
Tingling
An alteration in level of consciousness
Generalized seizures may be associated with:
Unconsciousness, usually accompanied by a fall, except with childhood absence seizures
Uncontrolled jerking of arms and legs
A short period of apnea (breathing cessation)
Salivation and frothing at the mouth
Tongue biting
Incontinence
A postictal stage of stupor or coma, followed by confusion, headache, and fatigue
A prodroma may occur with any seizure type. A prodoma is a certain feeling or symptom that may precede a seizure by hours or days.
An aura may occur with any seizure type. An aura is a certain sensory sensation that frequently or always immediately precedes a seizure.
P.217
Diagnostic Tools
A detailed medical history is required for an accurate diagnosis of a seizure.
Basic laboratory evaluation must be performed to rule out metabolic causes or drug-induced seizures.
Lumbar puncture is performed to rule out meningitis or encephalitis if suspected.
MRI is the imaging modality of choice to identify brain lesions such as tumor, abscess, or vascular malformation as the cause of the seizure.
A CT scan may be used for patients with emerging neurologic symptoms who need immediate diagnostic information.
An EEG may allow diagnosis of the type and location of the occurring seizure. Multiple EEG recordings increase the diagnostic potential.
Complications
Hypoxic brain damage and mental retardation may follow repeated seizures.
Depression and anxiety may develop. As described earlier, long-term social isolation may occur.
Treatment
Identification of the type of seizure is essential for optimal treatment.
Reverse the cause of the seizure disorder if possible.
Medications are available that may decrease the number of seizures an individual experiences. The goal of seizure treatment is zero seizure occurrence with a minimum of treatment-induced side effects. The medication selected must be appropriate for the seizure type.
Resective surgery to excise the epileptogenic focus is becoming more common and is indicated in patients for whom antiepileptic drugs do not completely control seizures. Surgery may also be used to remove connections between the cerebral hemispheres, limiting seizure occurrence (called corpus callostomy).
Vagus nerve stimulation involves an electrical device implanted in the infraclavicular area that provides a certain pattern of vagal stimulation
P.218
to patients with seizures refractory to treatment. This treatment is a relatively new alternative to drug therapy. Vagal nerve stimulators have been shown to be effective in reducing the frequency of seizures in some patients.Counseling for the patient and family is advised.
![]() ediatric Consideration
ediatric Consideration
Seizures in infants and young children brought on by a rapid increase in body temperature, also known as febrile convulsions, are common. Children who have febrile seizures do not appear to experience any long-term intellectual, academic, or behavioral effects.
Head Injury
Head injuries may be open (penetrating through the dura mater) or closed (blunt trauma, without penetration through the dura). Open head injuries allow environmental pathogens direct access to the brain. Damage will occur in either type of injury if blood vessels, glial cells, and neurons are destroyed or torn. Brain damage may develop after severe injury if bleeding and inflammation cause increased intracranial pressure.
Causes of Head Injuries
Causes of head injuries include automobile accidents, fights, falls, and sporting injuries. Open head injuries are often caused by bullet or knife wounds.
![]() ediatric Consideration
ediatric Consideration
Falls are a major cause of head injuries in children. Head injuries in toddlers often are related to falls down the stairs or at playgrounds. An infant or young child who receives a head injury should be evaluated for non-accidental head injury, often referred to as shaken-baby syndrome. This type of injury occurs from violently shaking an infant or small child, and usually involves striking the head of the child against a hard surface. This type of injury is characterized by subdural or subarachnoid hemorrhage.
![]() eriatric Consideration
eriatric Consideration
Falls are also a major cause of head injuries in the elderly. Falls may be related to poor vision, slippery rugs or tubs, and poor muscle strength in the elderly population. An elderly person who receives a head injury also may be the victim of elder abuse.
P.219
Types of Head Injuries
Several different types of head injuries are possible. Some involve an immediate loss of consciousness; others show delayed effects. Some head injuries result in obvious bleeding into the brain; others show no obvious signs of structural damage, but symptoms may develop.
Concussion
A concussion is a closed head injury usually characterized by loss of consciousness. Concussion results in a brief period of apnea. A concussion can be mild, moderate, or severe, depending on the length of time the person is unconscious. A longer period of unconsciousness is predictive of a worse outcome. However, even mild concussions may be associated with subtle behavioral or cognitive changes, even if no obvious brain pathology exists. The condition, called postconcussive syndrome, may last for more than a year.
Epidural Hematoma
An epidural hematoma is the accumulation of blood above the dura mater. An epidural hematoma occurs acutely and is usually caused by a life-threatening arterial bleed.
Subdural Hematoma
A subdural hematoma is the accumulation of blood under the dura mater, but above the arachnoid membrane. Subdural hematomas are usually caused by a venous tear, although occasionally a subdural arterial bleed may occur. A subdural hematoma may develop rapidly, in which case it is called an acute subdural hematoma, or may result from a slow bleed, in which case it is called a subacute subdural hematoma. Chronic alcohol abusers and the elderly may experience a slowly developing hematoma over a period of months after a mild head injury, and may not show any obvious symptoms from the hematoma until it is large. This is called a chronic subdural hematoma. A chronic subdural hematoma is possible because the elderly and chronic alcohol abusers have reduced brain tissue, which allows the cranium to accommodate an expanding hematoma without a significant increase in pressure.
Subarachnoid Hemorrhage
A subarachnoid hemorrhage is the accumulation of blood under the arachnoid membrane, but above the pia mater. This space normally contains only CSF. A subarachnoid hemorrhage usually results from a burst intracranial aneurysm, severe hypertension, an arteriovenous malformation, or a head injury. Blood accumulating on top of or under the meninges causes increased pressure on the underlying brain tissue.
Clinical Manifestations
With a concussion, consciousness is often lost.
Respiratory patterns may become progressively abnormal.
Pupillary responses may be absent or progressively deteriorate.
Headache may occur immediately or develop with increasing intracranial pressure.
Vomiting may occur as a result of increased intracranial pressure.
Behavioral, cognitive, and physical changes in speech and motor movements may occur immediately or develop slowly. Amnesia related to the event is common.
P.220
Diagnostic Tools
A skull radiograph may locate fractures or a developing bleed or blood clot. A CT scan or an MRI may pinpoint the site and extent of injury. A CT scan is usually the diagnostic tool of choice in the emergency room, although results of CT may be misleadingly normal. MRI is a more sensitive and accurate tool, capable of diagnosing diffuse axonal injury, but is costly and less accessible at most facilities.
Complications
Bleeding inside the brain, called an intracerebral hematoma, may accompany a severe closed head injury or, more commonly, an open head injury. With bleeding in the brain, intracranial pressure increases, and neuronal and vascular cells are compressed. This is a type of secondary brain injury. With a hematoma, consciousness may be lost immediately, or may decrease later as the hematoma expands and the interstitial edema worsens.
Subtle behavioral changes and cognitive deficits may exist and linger.
Treatment
Mild and moderate concussions are usually treated with observation and bed rest.
Surgical ligation of a bleeding vessel and evacuation of a hematoma may be required.
Surgical debridement (removal of foreign material and dead cells) may be required, especially for an open head injury.
Decompression through the drilling of holes into the brain, called burr holes, may be required.
Mechanical ventilation may be required.
Antibiotics are required for open head injury to prevent infection.
Methods to decrease intracranial pressure may include the administration of diuretics and anti-inflammatory drugs.
Spinal Injury
Spinal injury usually involves a fracture or other injury to the vertebral bones. The spinal cord, running through the vertebral column, may be sliced, pulled, twisted, or compressed. Damage to the vertebral column or cord may occur at any level. Damage to the cord may involve the entire
P.221
cord or be restricted to one half. Damage to the spine may result in temporary dysfunction or permanent damage if the cord is transected (cut).
Causes of Spinal Injury
The most common causes of spinal injury are automobile and motorcycle accidents, falls, sports injuries, and wounds from guns and knives.
Results of Spinal Injury
Microscopic Hemorrhages
Small hemorrhages develop with all vertebral or spinal cord injuries. These small bleeds, accompanied by inflammatory reactions that lead to swelling and edema, cause increased pressure in and surrounding the cord. Increased pressure compresses the nerves and decreases the vascular supply, which causes hypoxia and dramatically increases the extent of cord injury. Scar tissue can develop, causing the nerves in the area to become blocked or tangled irreversibly. Nerve growth factors are produced that may cause tangling of neurons and a worsening prognosis.
Loss of Sensation, Motor Control, and Reflexes
With severe spinal injury, sensation, motor control, and reflexes at and below the level of cord injury are lost. The loss of all reflexes is called spinal shock. Swelling and edema surrounding the cord may extend two vertebral segments above the site of injury. Therefore, sensory and motor loss and spinal shock may develop starting from two segments above the injury. Spinal shock typically goes away, but the permanent loss of sensation and motor control will continue if the cord has been transected or if severe swelling and hypoxia have occurred.
Spinal Shock
Spinal shock involves immediate loss of all reflexes from two segments above and below the site of cord injury. Lost reflexes include those controlling posture, bladder and bowel function, blood pressure, and maintenance of body temperature. Spinal shock appears to occur from the sudden loss of all of the tonic discharge normally carried in neurons descending from the brain, which acts to maintain the function of the reflexes. Spinal shock typically lasts 7 to 21 days, but may last longer. As spinal shock regresses, hyper-reflexia may occur, characterized by muscle spasticity and reflex bladder and bowel emptying.
Autonomic Hyperreflexia
Autonomic hyperreflexia is characterized by the reflex activation of sympathetic nerves below the level of the cord lesion, which leads to a dangerous increase in blood pressure. This condition can occur anytime after the cessation of spinal shock. Autonomic hyperreflexia occurs when a painful sensory stimulus is relayed to the spinal cord and initiates a spinal reflex involving the activation of the sympathetic nervous system.
P.222
With sympathetic activation, constriction of the blood vessels occurs and systemic blood pressure increases.
In individuals with an intact cord, such an increase in blood pressure would immediately be sensed by baroreceptors that monitor blood pressure (Chapter 13). In response to normal baroreceptor activation, the cardiovascular center in the brain would increase parasympathetic stimulation to the heart, thereby slowing the heart rate. In addition, the sympathetic nervous response would be blocked and dilation of the blood vessels would occur. The parasympathetic and sympathetic changes would serve to rapidly return blood pressure to normal. In an individual with a cord lesion, although the baroreceptors are activated and cause parasympathetic activation to slow heart rate and induce vasodilation above the site of injury, sympathetic reflex vasoconstriction below that level will continue.
With an occurrence of autonomic hyperreflexia, blood pressure can increase more than 200 mmHg systolic, leading to stroke or myocardial infarct. Stimuli that typically cause autonomic hyperreflexia include a distended bladder or bowel and the stimulation of surface pain receptors. Autonomic hyperreflexia is more likely to happen when the lesion is high on the spinal cord.
Paralysis
Paralysis is the loss of sensory and voluntary motor function. With spinal cord transection, paralysis is permanent. Paralysis of the upper and lower extremities occurs with transection of the cord at level C6 or higher and is called quadriplegia. Paralysis of the lower half of the body occurs with transection of the cord below C6 and is called paraplegia. If only one half of the cord is transected, hemiparalysis may occur. Permanent paralysis may occur even when the cord is not transected, as a result of the destruction of the nerves following cord hemorrhage and swelling. In addition, demyelination of the axons in the cord can lead to clinically complete lesions, even though the spinal cord may not be transected. Demyelination of the axons most likely occurs as part of the inflammatory response to cord injury.
Clinical Manifestations
Loss of sensation, motor control, and reflexes below the level of injury, and up to two levels above, will occur. Body temperature will reflect ambient temperature, and blood pressure will be reduced.
The pulse rate is often normal, with low blood pressure.
Diagnostic Tools
Physical examination coupled with CT and MRI will document vertebral and spinal injury and edema.
Complications
If damage and swelling around the cord is in the cervical spine (down to approximately C5), respirations may cease because of compression of
P.223
the phrenic nerve, which exits between C3 and C5 and controls the movement of the diaphragm.Autonomic hyper-reflexia is characterized by high blood pressure with bradycardia (low heart rate), and sweating and flushing of the skin on the face and upper torso.
In the past, individuals suffering from a C2 or higher transection invariably died as a result of respiratory arrest. Although this is still true for many, recent advances in treatment modalities and better emergency rescue service responses have resulted in the survival of many individuals with high cord transection.
A severe spinal cord injury affects virtually all systems of the body to some degree. Commonly, urinary tract and kidney infections, skin breakdown and the development of pressure ulcers, and muscle atrophy occur. Depression, marital and family stress, loss of income, and large medical expenses are some of the psychosocial complications.
Treatment
Immobilization to prevent cord severing or additional damage after any head or neck injury is essential, even if a cord injury is not obvious.
Early surgical intervention to relieve pressure on the cord caused by broken vertebrae or collapsed disks may reduce long-term disability.
Immediate (within the first hour) large-dose administration of steroids has been shown to reduce cord swelling and inflammation and limit the extent of permanent damage. Strategies to stimulate axon regeneration, or to return impulse conduction along preserved but demyelinated axons, will likely lead to improved outcomes of patients with spinal cord injuries.
Surgical fixation of the vertebral column hastens and supports healing.
Physical therapy, including speech therapy if the lesion interferes with speech and respiratory movements, is begun soon after the patient's condition stabilizes.
Education on avoidance and recognition of autonomic hyper-reflexia can reduce the risk of stroke or myocardial infarct.
Treatment of autonomic hyperreflexia includes antihypertensive medications and the removal of the initiating stimulus.
For patients with permanent damage, education and counseling about long-term expectations and complications of skin, reproductive, and urinary systems are essential. Including family members in education and counseling sessions is essential.
![]() eriatric Consideration
eriatric Consideration
A 25-year longitudinal study on the effect of aging on patients with spinal injuries found that the quality of life following a spinal cord injury is not necessarily compromised, a finding that clearly attests to the resiliency of the human spirit.
P.224
Cerebral Vascular Accident
A cerebral vascular accident (CVA), often called a stroke or a brain attack, is a brain injury related to an obstruction in brain blood flow. Especially at risk of suffering a CVA are elderly individuals with hypertension, diabetes, hypercholesterolemia, or heart disease. With a CVA, cerebral hypoxia leading to neuronal cell death and injury occurs. Inflammation, characterized by proinflammatory cytokine release, the production of oxygen free radicals, and swelling and edema of the interstitial space, occurs with cell damage and contributes to the worsening situation. Likewise, acidosis develops subsequent to hypoxia and further injures the brain via activation of acid-sensing neuronal ion channels. Ultimately, brain damage occurs after a CVA, typically peaking 24 to 72 hours after neuronal cell death.
There are two general classifications of CVAs: ischemic and hemorrhagic. Ischemic CVAs develop from a prolonged blockage in arterial blood flow to a part of the brain. Hemorrhagic CVAs occur as a result of bleeding into the brain. See page C4 for illustrations.
Ischemic Stroke
The arterial blockage causing an ischemic stroke may occur as a result of a thrombus (a blood clot in the cerebral artery) or an embolus (a blood clot that has traveled to the brain from elsewhere in the body).
Thrombotic Stroke
A thrombotic stroke occurs from occlusion of blood flow, usually resulting from severe atherosclerosis. Frequently, an individual will experience one or more transient ischemic attacks (TIAs) before a true thrombotic stroke occurs. A TIA is a brief, reversible disruption in brain function resulting from cerebral hypoxia. It is likely that a TIA occurs when an atherosclerotic vessel undergoes a spasm, or when the oxygen demand of the brain increases and this demand cannot be met because of advanced atherosclerosis. By definition, a TIA lasts fewer than 24 hours. Frequent TIAs suggest the likelihood of a true thrombotic stroke.
A thrombotic stroke typically develops over a period of 24 hours. During the period in which a stroke is progressing, the individual is said to be suffering from a stroke in evolution. At the end of that period, the individual is said to have suffered a completed stroke.
Embolic Stroke
An embolic stroke develops after arterial occlusion by an embolus formed outside the brain. Common sources of emboli leading to stroke include the heart after a myocardial infarct or atrial fibrillation, and emboli breaking off the common carotid arteries or the aorta.
Hemorrhagic Stroke
A hemorrhagic stroke occurs when a blood vessel in the brain is broken, leading to ischemia (reduced flow) and hypoxia downstream. Causes of
P.225
hemorrhagic stroke include hypertension, a burst aneurysm, or an arteriovenous malformation (abnormal connection). Hemorrhage into the brain significantly increases intracranial pressure, worsening the resulting brain injury.
Clinical Manifestations
Symptoms of a TIA may include temporary numbness of the face or limbs, slurring of words, confusion, dizziness, and changes or blackouts in vision. If any of these occurs, an individual should immediately seek medical assistance.
With a CVA, the area of the brain that becomes ischemic determines the presenting clinical manifestations. Mentation, emotions, speech, vision, or movement can be affected. Many changes are irreversible, but some are reversible.
A hemorrhagic stroke is frequently accompanied by a severe headache and loss of consciousness.
Diagnostic Tools
Rapid diagnosis of a CVA is essential to minimize damage. CT scan is the method of choice for assessment of an acute presentation of a CVA. CT is highly sensitive to hemorrhage, an important consideration because there are vital differences in the treatment of ischemic versus hemorrhagic strokes. CT scans are also readily accessible, even in small or rural hospitals.
Most MRI devices, although even more sensitive than CT at identifying early brain damage from a CVA, are slower than CT and thus are used less often in this emergency situation. However, after the initial CT scan, MRI is recommended to determine the exact location of damage and to monitor the lesion.
Complications
An individual suffering a major CVA to the part of the brain controlling respiration or cardiovascular response may die. Hypoxic destruction of expressive or receptive areas of the brain may lead to communication difficulties. Hypoxia of motor areas in the brain may lead to paresis. Emotional changes may occur with damage to the cortex, including the limbic system.
An intracerebral hematoma may result from a burst aneurysm or a hemorrhagic stroke, causing secondary brain injury as intracranial pressure increases.
Treatment
In patients in whom the CVA can be identified as ischemic in nature, thrombolytic agents, such as tissue plasminogen activator (TPA), can be administered. TPA should be given as early as possible (at least within
P.226
the first 3 hours of the attack) to be most effective in preventing long-term damage. However, it would be dangerous to treat a hemorrhagic stroke with a thrombolytic because this would increase bleeding and worsen outcome.A hemorrhagic stroke is treated with emphasis on stopping the bleeding and preventing another occurrence. Surgery may be required.
Drug therapies that inhibit acid-sensing ion channels are being developed to limit stroke-induced damage.
All stroke patients are treated with bed rest and a reduction of external stimuli to reduce cerebral oxygen demands. Measures to reduce intracranial edema and pressure may be instituted.
Physical, speech, and occupational therapy are often required.
Central Nervous System Infection
A central nervous system infection may involve the brain tissue (encephalitis), or the meninges (meningitis). With encephalitis and meningitis, inflammatory and immune responses cause increased swelling and edema in or around the brain, increasing intracranial pressure. Encephalitis also is associated with neuron death caused by the infecting microorganism.
Encephalitis
Encephalitis is usually a viral infection of the brain. It is often carried by a mosquito vector or related to infection with herpes simplex 1 or cytomegalovirus. Nerve cell degeneration is widespread and edema and swelling are severe.
Meningitis
Meningitis is the most common serious infection of the CNS. It is usually caused by bacteria or a virus, although fungi, protozoa, and toxins are also causes. Meningitis frequently occurs from the spread of an infection elsewhere in the body, for example, the sinuses, ears, or upper respiratory tract. A posterior basilar skull fracture with a ruptured eardrum may also cause meningitis. With bacterial meningitis, released toxins destroy meningeal cells and stimulate immune and inflammatory reactions. Secondary encephalitis may occur. Even when treated, up to 40% of meningitis cases are fatal and up to 30% of survivors have neurological complications.
In the past, most cases of meningitis were in children younger than the age of 5, and most often the causative agent was Haemophilus influenzae. Since 1990, a vaccine against H. influenzae has become available and is administered to most children in the United States and other countries as a series of injections, beginning in the second month of life. As a result of
P.227
this important intervention, the incidence of meningitis in children aged 1 month to 2 years has decreased 87%. Because of the dramatic decline in H. influenzae-type meningitis in this population, cases of bacterial meningitis overall in the United States have dropped 55%.
Meningitis now occurs most commonly in adults ages 19 to 59. In this age group, the most common cause of bacterial meningitis is Streptococcus pneumoniae (pneumococcal meningitis). The next greatest incidence is in children ages 2 to 18, and the cause is most often Neisseria meningitides (meningococcal meningitis). In the neonate, the cause is most often group B streptococcus; in infants aged 1 to 23 months the causes are split almost equally between S. pneumoniae and N. meningitidis. While college students in general are no more likely to develop meningitis than other young adults of that age group, subgroups of college students are at increased risk. In particular, college freshman living in dormitories have a 6-fold greater risk of developing meningococcal meningitis than those not living in a dormitory. While most colleges now require vaccination against meningococcal meningitis, the vaccination is not effective against all strains.
Clinical Manifestations
Symptoms of increased intracranial pressure may develop with meningitis and encephalitis, including headache, decreased consciousness, and vomiting. Papilledema (swelling of the area around the optic nerve) may occur in severe cases. Typically, the symptoms are worse with encephalitis.
Fever from infection is common in meningitis and encephalitis.
Photophobia (painful response to light) from irritation of the cranial nerves frequently accompanies meningitis and encephalitis.
Inability to flex the chin to the chest without pain (nuchal rigidity) occurs in meningitis and encephalitis as a result of irritation of the spinal nerves.
Encephalitis typically presents with dramatic signs of delirium and a progressive decrease in consciousness. Seizures and abnormal movements may occur.
Diagnostic Tools
In patients with suspected acute bacterial meningitis for whom there is no clinical contraindication, the CSF is collected through lumbar puncture and examined for white blood cells and microorganism sensitivity. Elevated protein, elevated neutrophils, and low glucose in the CSF indicate meningitis.
Laboratory studies for viral meningitis are not indicated (e.g., normal glucose, elevated lymphocytes).
Rapid diagnosis of CNS infection is essential; this is especially true of meningitis. CT scan and MRI may be used to evaluate the degree of
P.228
swelling and sites of necrosis. CT is very rapid and is most useful in emergency situations.Corticosteroid (dexamethasone) therapy to reduce inflammation appears to be beneficial for the adjuvant treatment of most adults with suspected pneumococcal meningitis.
Complications
Individuals may suffer permanent disability, brain damage, or death from encephalitis or, less commonly, meningitis.
Seizures may develop.
Treatment
A broad spectrum antibiotic is administered after CSF collection and is changed if necessary after culture results.
An antiviral drug will be administered for encephalitis.
Measures to reduce intracranial pressure will be initiated, especially for encephalitis.
Some types of meningitis will require the patient to be isolated in the hospital.
Alzheimer Disease
Alzheimer disease is a progressive dementia characterized by the widespread death of brain neurons, especially in an area of the brain called the nucleus basalis. Nerves from here normally project throughout the cerebral hemispheres to areas of the brain responsible for memory and cognition. These nerves release acetylcholine, which has been shown to be essential in building short-term memory at the biochemical level. The enzyme responsible for the production of acetylcholine, choline acetyltransferase, is reduced up to 90% in the brains of individuals who have died of Alzheimer disease compared to those who have died of other causes. Therefore, lack of acetylcholine can account for at least some of the forgetfulness and loss of cognitive function seen in individuals with Alzheimer disease. Other neurotransmitters also appear to be absent in individuals with the disease.
Alzheimer disease typically develops after the age of 65, causing senile dementia. However, it may occur earlier and result in presenile dementia. There is a genetic tendency to develop the disease, especially early onset disease. In fact, family history is second only to age as a risk factor for developing Alzheimer disease. The disease is the most common cause of dementia today.
Pathology
The pronounced autopsy findings of patients with Alzheimer disease include widespread development of neuronal tangles, the axons of which
P.229
coalesce into plaques, called senile plaques. The senile plaques include remnants of the dying nerve terminals, aluminum deposits, and abnormal protein fragments. The protein fragments always include pieces of a protein known as the amyloid beta peptide (Abeta). Abeta is a peptide fragment from a larger, membrane-spanning protein called amyloid precursor protein (APP). In addition, major morphological changes include pronounced atrophy with narrowing of the gyri and widening of the sulci. These features are most pronounced in the frontal and temporal lobes.
Development Theories
One theory concerning the development of Alzheimer disease involves the accumulation of Abeta in certain areas of the brain. Typically, the degradation of APP involves cleavage of the Abeta fragment as well, with the result that Abeta is lost from the area. In patients with Alzheimer's disease, APP appears to be incorrectly cleaved, such that the Abeta fragment remains intact and accumulates in the surrounding plaques. It has been suggested that the abnormal processing of the larger APP allows pieces of Abeta to protrude out of nerve cell membranes, somehow initiating the tangles and killing the cells. Support for this theory comes from the finding that the gene coding for APP lies on chromosome 21, which when present in triplicate (rather than as a pair) causes Down syndrome. Virtually all individuals with Down syndrome who live into their 40s will develop Alzheimer disease. However, at least two other chromosomes have also been linked to Alzheimer disease in different groups of patients, suggesting that there may be more than one genetic cause of the disease.
A second theory as to the cause of Alzheimer disease involves the discovery that the risk of developing the disease increases with inheritance of a certain gene coding for a specific type of cholesterol-shuttling protein, called apolipoprotein E (APO-E4). Inheritance of the gene for APO-E4, as opposed to one of the other varieties of this protein, APO-E2 or APO-E3, may somehow destabilize the nerve cell membrane, leading to tangling and neuronal cell death. Homozygotes for APO-E4 are at increased risk of developing the disease compared to heterozygotes.
Another area of research concerning Alzheimer disease involves glutamate, an excitatory neurotransmitter present throughout the brain. Glutamate plays an important role in learning and memory; however, some studies suggest that excess glutamate or abnormal cell sensitivity to glutamate may result in too much calcium entering nerve cells, causing neuronal cell death. The hippocampus, an area of the brain involved in memory, has been found to be especially sensitive to glutamate.
Clinical Manifestations
The diagnosis of Alzheimer disease is usually a clinical one, based on history, physical, and biochemical and radiologic examinations. A clinical diagnosis of Alzheimer is typically highly sensitive in diagnosing positive
P.230
cases, but may misdiagnose falsely, especially among the very old. Clinical manifestations include the following:
Insidious, slowly progressing forgetfulness, decreased judgment, behavioral and personality changes developing over a period of up to 10 years.
Short-term memory loss and problems with math concepts are common.
Diagnostic Tools
During a comprehensive physical examination, clinicians and attending family members often will recognize that patients suspected of dementia cannot provide reliable information on medical history. While not diagnostic, this is an important red flag.
There is no definitive means of diagnosing Alzheimer disease during an individual's lifetime other than eliminating metabolic or vascular causes of the mental deterioration. However, increasingly sensitive MRI, PET, and SPECT scans can provide clinical support for the diagnosis. Newer techniques are likely to become available for more accurate identification of neuronal tangles and senile plaques.
Although Alzheimer's disease is the most common cause of dementia, approximately one-third of suspected dementia cases are caused by reversible disorders, including metabolic imbalance, drug effects, CVA, vitamin deficiencies, and depression. These causes must be ruled out using CT or MRI, a complete blood count, and metabolic studies.
Treatment
Patient and family education regarding memory aids, diet, and safety issues may slow the progression of symptoms.
Medications (Cognex) for the slowing or reversal of early Alzheimer symptoms are available and may delay the progression of symptoms in some patients.
Cholinesterase inhibitors (donepezil, rivastigmine and galantamine) that prolong the effective half-life of acetylcholine are the recommended drug therapy for mild to moderate dementia.
Memantine, an N-methyl-D-aspartate (NMDA)-receptor antagonist, is approved for treatment of moderate to severe Alzheimer disease. Memantine works by moderately blocking glutamine receptors in the brain, allowing for some but not excessive glutamate stimulation.
Some studies suggest that vitamin E supplementation may slow the progression of death, institutionalization, and severe dementia in some patients.
Long-term benefits of all medications are undetermined.
Parkinson Disease
Parkinson disease is a progressive brain disorder characterized by the loss of neurons in an area of the midbrain known as the substantia nigra. These
P.231
neurons use dopamine as a neurotransmitter and project their axons to the thalamus and the caudate and putamen areas of the basal ganglia. Parkinson disease results when about 80% of the cells that make up the substantia nigra are lost; there is also a reduction in dopamine receptors in the basal ganglia. Onset of the disease typically occurs in the sixth or seventh decade of life. It is the second most common neurodegenerative disease in adults. Although there may be a small genetic influence on the development of Parkinson disease, it seems mostly limited to early onset disease (before age 50).
Dopamine acts as an inhibitory neurotransmitter; thus, reduced dopamine stimulation in the substantia nigra and the basal ganglia leads to an imbalance between this inhibitory neurotransmitter and the excitatory neurotransmitter acetylcholine. Without dopamine, neurons are overstimulated by acetylcholine, resulting in excess muscle tone characterized by tremor and rigidity. A fixed facial tone projects a lack of emotional responsiveness, although there is often no emotional or cognitive deficit in patients with Parkinson disease.
Cause
The cause of Parkinson disease is unknown. As noted previously, recent evidence suggests that there is at least some genetic tendency toward developing the disease. Viruses and toxins have been implicated in some studies.
Other Conditions Whose Symptoms Resemble Parkinson
Symptoms of Parkinson disease can develop in persons without the disease who suffer from certain types of brain trauma, infection, or tumors. Likewise, individuals who suffer from schizophrenia and require therapy with certain psychotropic drugs such as the phenothiazines may develop symptoms of Parkinson disease. Some symptoms of secondary Parkinson can be relieved by treatment of the injury or infection or by removal of the tumor or drug, but others may remain.
Clinical Manifestations
A tremor at rest.
Drooling and dysphagia (difficulty swallowing).
A shuffling gait, muscle rigidity, and stiffness.
Akinesia, which is described as a lack of movement, including that involved with facial expressions and other voluntary movement, characterizes the disease.
Loss of postural reflexes leading to loss of equilibrium and a tendency to stoop cause the typical bent-over posture seen in patients with Parkinson.
Diagnostic Tools
In patients with Parkinson-like symptoms, diagnosis is often made based on history and physical examination. A positive response to levodopa is strongly indicative of Parkinson.
P.232
Complications
Many patients with Parkinson disease develop dementia.
Treatment
Dopaminergic (L-dopa) or anticholinergic drugs may be administered to reduce symptoms.
Transplanting cells from the basal ganglia or adrenal medulla (where dopamine is also produced) of fetuses into the brains of patients with Parkinson disease has been successful in some studies.
Huntington Disease
Huntington disease (chorea) is a rare, degenerative disease of the basal ganglia and cerebral cortex. It is passed genetically as an autosomal-dominant disorder, apparently caused by expansion of a repeating codon located on chromosome 4. Onset of the disease typically occurs in the fourth or fifth decade of life.
With degeneration of the basal ganglia and cerebral cortex, several different neurotransmitters are lost. Many complications of the disease result from the loss of the inhibitory neurotransmitter GABA. There also appear to be gross abnormalities in energy production by the neuronal cell mitochondria.
Characteristic movements seen in patients with Huntington disease include extreme involuntary jerking, called chorea. These abnormal movements can occur all over the body and lead to physical exhaustion. Persons with Huntington disease undergo progressive loss of mental functioning, leading to dementia. Death usually results within 10 years.
Clinical Manifestations
Choreic (jerking) movements.
Personality changes, depression, and slowly progressing dementia.
Diagnostic Tools
Identification of the gene responsible for Huntington disease allows the diagnosis of the trait prenatally or before the onset of symptoms in an adult.
Occasionally, MRI is used to image the brain. Atrophy is apparent in late disease. PET scan may be used to demonstrate hypometabolism of specific areas of the brain.
Treatment
There is no treatment for Huntington disease. Because genetic identification of asymptomatic individuals who will likely develop the disease is possible, counseling is essential for those who choose to know their status and those who do not so choose.
P.233
Multiple Sclerosis
Multiple sclerosis is an autoimmune disease characterized by both a cell-mediated immune response and a humoral immune response with activated T cells and antibodies, both produced against self-antigens. These autoimmune activities lead to the destruction of neurons in the central nervous system. The peripheral nervous system is unaffected. It is unclear whether the immune attack is aimed at the myelin sheath directly, resulting in slowing of neural transmission down the axon, or is against the axon itself, causing severing of the axon and subsequent destruction of the myelin. In either case, the transmission of neural impulses in the brain and spinal cord is slowed, leading to dramatic alterations in movements and reflexes and, in some cases, changes in mental status such as deficits in memory, sustained attention, and rate of processing information. Mental status changes have been suggested to develop due to cholinergic deficits that may accompany increased cerebral glucose utilization. Continued inflammatory responses contribute to the disease by producing swelling and edema, which further injure the neurons and cause the development of scar tissue plaques on the myelin.
Categories of Multiple Sclerosis
There are four categories of multiple sclerosis, called syndromes, based on the original nerve tracts affected. These syndromes are the corticospinal syndrome, the brainstem syndrome, the cerebellar syndrome, and the cerebral syndrome. Initial symptoms usually fit into one of these syndromes. As the disease progresses, different tracts are affected and the symptoms become more widespread.
Besides separate neural tracts involved, multiple sclerosis can also take different forms based on the rate of progression of the disease. In the first type, relapsing-remitting form, the course of the disease is characterized by exacerbations of symptoms, followed by partial or full remission back to the preceding state. The second type, called primary progressive form, is characterized by a quick downhill course, without remission, from the beginning of the disease. The third type, secondary progressive form, begins as the relapsing-remitting type, and then changes to a fast progression without remission.
Causes of Multiple Sclerosis
The cause of multiple sclerosis is unknown, but there appears to be a genetic tendency toward developing this and other autoimmune diseases. Some evidence suggests that a childhood viral infection, perhaps measles or a type of herpes infection, may initiate the immune response. It has been suggested that a breakdown in the blood-brain barrier during the time of the viral infection may have allowed a B-cell lymphocyte, developed against the virus, to gain entrance to and colonize the brain. An IgG clone (IgG from one B-cell line) is often present in the CSF of an individual
P.234
with multiple sclerosis. These clones increase in number with the many exacerbations of the disease.
In support of this theory are observations that viral infections and multiple sclerosis occur more frequently in individuals who live at northern latitudes. An individual's risk of developing multiple sclerosis appears to be related to the latitude in which the individual lived for approximately the first 15 years of life, with those from northern latitudes most at risk.
Clinical Manifestations
Episodes of motor, visual, or sensory disturbances that partially resolve and then recur.
Bladder dysfunction may occur with some types of multiple sclerosis.
Some individuals may develop cognitive or emotional disorders.
Symptoms are frequently precipitated by stress. Stressors may include the birth of an infant, illness, fever, fatigue, or high temperatures.
Diagnostic Tools
Clonal IgG bands in the CSF are found using electrophoresis techniques in approximately 90% of patients.
Elevations in other types of CSF and plasma IgG are frequently present.
MRI, and to a lesser degree CT scans, may allow for visualization of central nervous system plaques. MRI may identify disease activity even in the absence of acute clinical findings, and is capable of differentiating between old and new lesions.
Techniques to measure muscle cell discharge will demonstrate delayed muscle excitation in some types of the disease.
Complications
Severe neurologic deficits including loss of sight, increasing fatigue, and intellectual deterioration may develop over the course of the disease.
Depression, loss of social support, family and spousal stress, and financial problems are common.
Treatment
Aggressive immunosuppressant therapy at the start of the disease and with any exacerbation may limit the autoimmune destruction of the neuron or myelin.
Antiviral drugs may slow the progress of the disease.
Education on bladder training, sexual functioning, and avoidance of complications associated with reduced mobility may increase lifestyle satisfaction and overall health.
Education regarding the need to avoid chronic fatigue and high temperatures may reduce symptoms.
Innovative drug therapies are being tried that aim to foster antigenic self-tolerance by providing myelin protein for ingestion. These therapies
P.235
are based on the hypothesis that an individual may tolerate (not attack in an immune response) a substance that enters the body through the GI tract.
Amyotrophic Lateral Sclerosis
Amyotrophic lateral sclerosis (ALS) is a rapidly progressing degenerative disease of the upper and lower motor neurons resulting in near total paralysis. The loss of motor neurons does not include cranial nerves III, IV, and VI. Therefore, some facial movements, including blinking, are maintained. ALS is also known as Lou Gehrig's disease and usually occurs in the fourth or fifth decade of life. The disease is usually fatal within 5 years, although some individuals may live much longer. Degeneration of the motor neurons occurs without any obvious inflammation. Although the myelin is not a primary site of degeneration, loss of the nerve axon causes the subsequent loss of myelin, and scarring occurs. No known cause of ALS has been identified, although viral infection, metabolic disturbances, and trauma have been suggested. In addition, there is a familial form of ALS, which affects approximately 10% of ALS patients. Recent evidence suggests that a genetic link may be present in substantially more cases as well.
Clinical Manifestations
Initial weakness develops in one muscle group, which progresses to weakness and paralysis in all skeletal muscles except the extraocular muscles.
Intellectual and sensory functioning remains normal until death.
Complications
ALS may be difficult to diagnose. Once confirmed, depression may occur. In many cases, severe family hardship develops as the disease progresses quickly.
Diagnostic Tools
Muscle biopsy demonstrates lower neuron degeneration and confirms a strong clinical diagnosis.
Treatment
Psychological support is essential for the individual and family, as is education on maintaining eye communication.
Myasthenia Gravis
Myasthenia gravis is a peripheral nervous system disorder characterized by autoantibody production against the receptors for acetylcholine present on the motor end-plate region of skeletal muscles. The IgG
P.236
autoantibodies competitively bind to the acetylcholine receptors, prevent acetylcholine from binding to the receptors, and therefore prevent muscle contraction. Eventually, receptors at the neuromuscular junction are destroyed.
Myasthenia gravis may first cause weakness of the muscles controlling eye movements (ocular myasthenia gravis) or may affect the entire body (generalized myasthenia gravis). Progression of the disease is variable and may be slowly progressing, with or without remissions, or rapidly progressing, leading to death by respiratory paralysis and failure.
The cause of myasthenia gravis is unknown but appears to be associated with a familial tendency toward developing autoimmune disease. The thymus gland is frequently hyperplastic and appears to function as it did in early childhood, suggesting it may be initiating or perpetuating the immune response.
Clinical Manifestations
Weakness of the muscles of the eyes, causing ptosis (drooping of the eyelids).
Weakness of face, neck, and throat muscles, causing difficulty eating and swallowing.
Continued spread of muscle weakness. Initially there is easy fatigue with recovery of strength after rest. Eventually there is no recovery of strength after rest.
Diagnostic Tools
Normally acetylcholine is broken down at the neuromuscular junction by the enzyme acetylcholinesterase. A clinical diagnosis of myasthenia gravis can be confirmed on the basis of the return of muscle strength after intravenous administration of a medication that prevents the activity of acetylcholinesterase, thereby prolonging the half-life of acetylcholine. This medication, edrophonium chloride (Tensilon), allows acetylcholine to have a better chance of binding to its receptors, allowing voluntary muscle contraction. The effect of Tensilon lasts several minutes, after which muscle weakness reappears.
Electromyography (EMG) measurements of skeletal muscle action potentials show reduced amplitude on motor neuron stimulation.
Complications
Myasthenic crisis, characterized by severe worsening of skeletal muscle function culminating in respiratory distress and death as the diaphragm and intercostal muscles become paralyzed, may occur after a stressful experience such as an illness, emotional upset, surgery, or during pregnancy.
Cholinergic crisis is a toxic response occasionally seen with the use of too much anticholinesterase drug. A hypercholinergic state can develop
P.237
that is characterized by increased intestinal motility, pupillary constriction, and bradycardia. The individual may develop nausea, vomiting, sweating, and diarrhea. Respiratory distress may occur.
Treatment
Frequent rest periods during the day conserve strength.
Anticholinesterase medications are provided to prolong the half-life of acetylcholine at the neuromuscular junction. The medications must be taken on schedule each day to prevent muscle fatigue and collapse.
Anti-inflammatory medications are used to limit the autoimmune attack.
A myasthenic crisis may be treated with additional medication, and respiratory support if necessary.
A cholinergic crisis is treated with atropine (acetylcholine blocker) and respiratory support, until symptoms resolve. Anticholinesterase therapy is withheld until toxic levels of the drug are reversed.
Myasthenia crisis and cholinergic crisis present similarly but are treated differently. Tensilon administration is used to differentiate between the two disorders.
Plasmapheresis (blood dialysis with the removal of IgG antibodies) and thymectomy (surgical removal of the thymus) are sometimes performed, with variable long-term results.
Guillain-Barr Syndrome
Guillain-Barr syndrome is a peripheral nervous system disease characterized by the sudden onset of muscle paralysis or paresis. Guillain-Barr results from an autoimmune attack against the myelin surrounding the peripheral nerves. With destruction of the myelin, the axons can be damaged. Symptoms of Guillain-Barr disappear as the autoimmune attack ceases and the axons regenerate. If destruction of the cell body occurred during the attack, some degree of disability may remain. Although the cause of Guillain-Barr is unknown, the disease usually occurs 1 to 4 weeks after a viral infection or immunization.
The muscles of the lower extremities are usually affected first, with paralysis advancing up the body. Respiratory muscles may be affected, leading to respiratory collapse. Cardiovascular function may be impaired because of interruption of autonomic nerve function.
Clinical Manifestations
Ascending muscle weakness or paralysis.
Diagnostic Tools
Nerve conduction tests will demonstrate neuronal dysfunction.
Elevated protein in the CSF is common.
P.238
Complications
Respiratory or cardiovascular collapse may cause death.
Weakness of some muscles may persist.
Treatment
Ventilator support may be required if the respiratory muscles are affected.
Anti-inflammatory medications may limit the autoimmune attack.
Spina Bifida
Spina bifida is a congenital neural tube defect characterized by a failure of the vertebral arches to close. This results in a cyst-like protrusion of the meninges alone (meningocele) or of the meninges and the spinal cord (myelomeningocele) out of the vertebral column. In the case of a meningocele, neural tissue is unexposed, and thus neural deficits are absent or minor. With a myelomeningocele, the spinal cord, in a cyst-like protrusion with its nerves, suffers injury, inflammation, and scarring. The result is some loss of neural function, often including paralysis. Another type of spina bifida is one in which minor irregularities in the vertebral arches exist that are not obvious at birth. This is called spina bifida occulta (hidden).
A meningocele can occur in any area of the spine; cranial or upper cervical meningoceles are frequently associated with hydrocephalus. A myelomeningocele typically occurs in the lumbar or lumbosacral area.
Causes of Spina Bifida
Although the cause of spina bifida is unknown, a genetic predisposition may exist. Increased risk of the disorder occurs with maternal folic acid deficiency. Folic acid deficiency is common in women; therefore, it is strongly recommended that all women anticipating pregnancy begin taking folic acid vitamin supplements at least 3 months before conception.
Clinical Manifestations
Spina bifida occulta may be asymptomatic or associated with:
Hair growth along the spine
Midline dimple, usually in the lumbosacral area
Gait or foot abnormalities
Poor bladder control
A meningocele may be asymptomatic or associated with:
A sac-like protrusion of meninges and CSF from the back
Clubbed foot
Gait disturbance
Bladder incontinence
P.239
A myelomeningocele is associated with:
Protrusion of meninges, CSF, and spinal cord
Neurologic deficits at and below the site of exposure
Diagnostic Tools
Elevated levels of a fetal protein, called alpha-fetoprotein, in maternal serum may indicate fetal spina bifida.
Ultrasound may diagnose the condition in utero.
Complications
Hydrocephalus may occur with a meningocele or myelomeningocele.
Treatment
No treatment may be required for spina bifida occulta or meningocele.
Surgical repair of the myelomeningocele, and sometimes the meningocele, is required.
If surgical repair is performed, placement of a shunt to allow for CSF drainage is necessary to prevent hydrocephalus and a subsequent increase in intracranial pressure.
Planned cesarean section before the initiation of labor can be important in reducing the neurologic damage seen in an infant with a spinal cord defect.
Hydrocephalus
Hydrocephalus ( water on the brain ) is characterized by an accumulation of CSF anywhere in the ventricles of the brain. Hydrocephalus may result from overproduction of CSF, obstruction of the flow of CSF within the ventricular system, or a decrease in the absorption of CSF out of the ventricles. Hydrocephalus can be apparent with a sonogram before birth, or it may develop in adulthood. In adults, hydrocephalus may develop suddenly after a head injury or slowly in response to a growing tumor.
Types of Hydrocephalus
There are two types of hydrocephalus: non-communicating and communicating.
Non-communicating Hydrocephalus
Non-communicating hydrocephalus occurs as a result of obstruction of CSF flow within the ventricular system. This type of hydrocephalus may occur with a tumor or as a result of a congenital irregularity in the ventricular pathways.
Communicating Hydrocephalus
Communicating hydrocephalus occurs as a result of a blockage in the absorption of CSF. Causes of this type of hydrocephalus include a buildup
P.240
of tissue (usually a neoplasm) or blood in the subarachnoid space. Head injuries may cause communicating hydrocephalus.
Effect of Hydrocephalus
Intracranial pressure increases with hydrocephalus; this can directly injure underlying nervous tissue and compromise cerebral blood flow and the neuronal supply of oxygen and glucose. Compensation for increased ICP may occur with slowly developing hydrocephalus. With an acute brain injury, rapidly developing hydrocephalus dramatically increases intracranial pressure and compensation is usually ineffective.
Clinical Manifestations
Newborns with hydrocephalus may have an enlarged head and a high-pitched cry.
Acutely developing hydrocephalus causes a rapid increase in intracranial pressure and may present with a severe headache, decreased consciousness, papilledema, and vomiting.
Slowly progressing hydrocephalus may present with irritability and changes in cognition and behavior.
Diagnostic Tools
Ultrasound may allow diagnosis in utero.
After birth, diagnosis is made by clinical inspection, measurements of head circumference, and observation of cranial suture lines.
Complications
Mental retardation may result.
Treatment
Placement of a shunt to drain CSF in utero or after birth may be performed.
Treatment of the underlying cause is required.
Cerebral Palsy
Cerebral palsy is brain damage that occurs in an infant before, during, or soon after birth. It results in some degree of motor dysfunction. Cerebral palsy is nonprogressive and is caused by cerebral hypoxia or increased intracranial pressure after physical trauma to the brain. Increased intracranial pressure may directly damage neuronal cells or may cause hypoxia by compressing the blood vessels. Frequently, hemorrhage is the cause of increased intracranial pressure.
Clinical Manifestations
Cerebral palsy may result in a motor deficiency in any or all limbs and usually involves muscle spasticity.
Vision disturbances, mental impairment, and seizures may occur.
P.241
Diagnostic Tools
Typically the infant is diagnosed based on clinical signs at birth or in early infancy.
Complications
Developmental and social delays are common and may lead to family and marital stress.
Treatment
Treatment depends on the extent of the physical impairment, mental status, and the occurrence of seizures. Surgery may be required to relieve contractions.
All treatment regimens must include physical therapy.
Counseling is important for the family and the child.
Selected Bibliography
Amato, M.P. (2005). Donepezil for memory impairment in multiple sclerosis. Lancet Neurology 4, 72 73.
Benveniste, M., & Dingledine, R. (2005). Limiting stroke-induced damage by targeting an acid channel. New England Journal of Medicine 352, 85 86.
Bertram. L., & Tanzi, R.E. (2005). The genetic epidemiology of neurodegenerative disease. Journal of Clinical Investigation 115, 1449 57.
Boechxstaens, G.E., & Pelckmans, P.A. (1997). Nitric oxide and the non-adrenergic noncholinergic neurotransmission. Comparative Biochemistry and Physiology. Part A, Physiology 118, 925 932.
Chen, Y.Y., DeVivo, M.J., & Jackson, A.B. (2005). Pressure ulcer prevalence in people with spinal cord injury: Age-period-duration effects. Archives of Physical Medicine and Rehabilitation 86, 1208 1213.
Craig, M.C., Maki, P.M., & Murphy, D.G.M. (2005). The Women's Health Initiative Memory 35, Study: findings and implications for treatment. Lancet Neurology 4(Suppl.), 190 194.
Duhaime, A., Christian, C.W., Rorke, L.B., & Zimmerman, R.A. (1998). Nonaccidental head injury in infants the shaken-baby syndrome. New England Journal of Medicine 338, 1822 1829.
Estep, M. (2005). Meningococcal meningitis in critical care: an overview, new treatments/preventions, and a case study. Critical Care Nursing Quarterly 28, 111 21
Fields, R.D. (2004). The other half of the brain. Scientific American 290, 55 61.
Gabrieli, J.D. (1998). Cognitive neuroscience of human memory. Annual Review of Psychology 49, 87 115.
Gilman, S. (1998). Imaging the brain: First of two parts. New England Journal of Medicine 338, 812 820.
Gilman, S. (1998). Imaging the brain: Second of two parts. New England Journal of Medicine 338, 889 896.
Gruen, P., & Liv, C. (1998). Current trends in the management of head injury. Emergency Medicine Clinics of North America 16, 63 83.
Klein, A., & Hirsch, J. (2005). Mindboggle: A scatterbrained approach to automate brain labeling. Neuroimage 24, 261 280.
Krause, J.S. & Broderick, L. (2005). A 25-year longitudinal study of the natural course of aging after spinal cord injury. Spinal Cord 43, 349 356.
Krupp, L.B., Christodoulou, C., Melville, R.N., Scherl, W.F., McAllister, W.S., & Elkin, L.E. (2004). Donepezil improved memory in multiple sclerosis in a randomized clinical trial. Neurology 63, 1579 1585.
Lingford-Hughes, A. (2005). Human brain imaging and substance abuse. Current Opinion in Pharmacology 5, 42 46.
P.242
Marks, W.J., & Garcia, P.A. (1998). Management of seizures and epilepsy. American Family Physician 57, 1589 1600.
Mayeux, R., Saunders, A. M., Shea, S., et al. (1998). Utility of the apolipoprotein E genotype in the diagnosis of Alzheimer's disease. New England Journal of Medicine 338, 506 511.
Miller, G. (2005). How are memories stored and retrieved? Science 309(5731), 92.
Peskind, E.R., Tangalos, E.G., & Grossberg, G.T. (2005). A case-based approach to Alzheimer's disease. Clinical Advisor, June, 34 46.
Rossom, R., Adityanjee, & Dysken M. (2004). Efficacy and tolerability of memantine in the treatment of dementia. American Journal of Geriatric Pharmacotherapy 2, 303 12.
Sillanpaa, M., Jalava, M., Kaleva, O., & Shinnar, S. (1998). Long-term prognosis of seizures with onset in children. New England Journal of Medicine 338, 1715 1722.
Skirton, H. (2005). Huntington disease: a nursing perspective. Medsurg Nursing 14, 167 72.
Trapp, B.D., Peterson, J., Ransohoff, R.M., Rudick, R., Mork, S., & Bo, L. (1998). Axonal transection in the lesions of multiple sclerosis. New England Journal of Medicine 338, 278 285.
Tseng, B.P., Kitazawa, M., & LaFerla, F.M. (2004). Amyloid beta-peptide: the inside story. Current Alzheimer Research 1, 231 239.
Umeda, S., Akine, Y., Kato, M., Muramatsu, T., Mimura, M., Kandatsu, S., et al. (2005). Functional network in the prefrontal cortex during episodic memory retrieval. Neuroimage 26, 932 940.
Verity, C.M., Greenwood, R., & Golding, J. (1998). Long-term intellectual and behavioral outcomes of children with febrile seizures. New England Journal of Medicine 338, 1723 1728.
Waxman, S.G. (1998). Demyelinating diseases: New pathological insights, new therapeutic targets. New England Journal of Medicine 338, 323 325.
Werler, M.M., Shapiro, S., & Mitchell, A.A. (1993). Periconceptional folic acid exposure and risk of occurrent neural tube defects. Journal of the American Medical Association 269, 1257 1261.
EAN: 2147483647
Pages: 26