2.7 Self-Assembly, Self-Recognition, and Self-Organization
|
2.7 Self-Assembly, Self-Recognition, and Self-Organization
The potentially most far-reaching aspect of molecular recognition lies in its application in self-assembly and/or self-organization. Self-assembly refers to the more or less automatic putting together of parts, based almost purely on molecular recognition. Self-organization refers to self-assembly with the generation of organized entities and may result in networks and feedback systems on different levels.
Self-organization may display positive cooperativity, implying that a given step in the assembly sets the stage for and facilitates the following one. It is self-assembly driven to completion. Positive cooperativity is a basic characteristic of molecular amplification devices, because once initiated, the subsequent steps of the assembly are facilitated. It represents a nonlinear process and confers features of an error filter; only the correct input will, in principle, lead to creating the cooperative structure. In addition, the dynamic features of supramolecular binding make these systems able to undergo annealing and self-repair defects. The ability to "explore" the available energy surface more freely than is possible with covalent bonding allows such systems to exchange incorrect components for correct ones, thus allowing a form of selfmaintenance and error correction. They can also display adaptation by being able to react to environmental changes and to adjust to novel conditions.
Self-Assembled Entities—Micelles and LB Films
Micelles, which are either globular or cylindrical constructions, are formed when a soluble surfactant dissolved in a solvent (usually aqueous) reaches a certain concentration (critical micelle concentration). Such surfactants are usually in the form of hydrocarbon chains with a hydrophilic "head" and a hydrophobic "tail". At the critical concentration, the molecules begin to associate, with the chains packing together (like a sea urchin) and the hydrophilic heads facing the water. Reactant molecules with nonpolar groups may have a much greater affinity for the interior of the micelle than for the polar aqueous phase or the interfacial regions. Consequently, some reactants are concentrated in the micelles, where the high local concentration may favor reactions between them.
In polymerization processes, the micelles can stabilize free-radical initiators by isolating single radicals and preventing the chain termination that results from the reaction of two radicals—at least until another radical enters or is formed within the micelle. This results in higher molecular weight products—so-called emulsion polymerization. In solution, the termination reactions are faster and the average molecular weight is much lower.
Spherical micelles are predominantly formed by surfactants with bulky or charged head groups slightly above the critical micelle concentration. With increasing concentration, there is competition among the micelles for water molecules forming their solvation shell, resulting in cylindrical structures (Gates 1992: 129).
Langmuir-Blodgett Films
In the formation of self-assembled layers of Langmuir-Blodgett (L-B) films, a surfactant that is insoluble in water (a fatty acid molecule with a hydrophilic part [carboxyl group] and a hydrophobic part [a long hydrocarbon chain]) is spread in an organic solvent on the water surface. After evaporation of the solvent, the molecules can be pushed together with a movable barrier until they are densely packed, forming a compact monolayer one molecule thick. This monolayer can then be transferred to the surface of a solid support—for instance, a glass slide. The carboxyl groups of the fatty acid molecules bind to the surface of the glass slide and the ends of the hydrocarbon chains are now facing the air. When dipping in the slide again, it is covered by a second layer of fatty acid in reverse orientation, the molecular "tails" of the two layers facing each other. This can be repeated many times. This technique (the Langmuir-Blodgett technique) has been used as a way to produce supramolecular arrangements with increasing complexity (Kuhn and Forsterling 2000).
Examples of Self-Organization Processes
Molecular organization and self-assembly into layers, membranes, vesicles, construction of multilayer films, and so on make it possible to build up specific supramolecular architectures. Performing the polymerization of the molecular components is a major step in increasing control over the structural properties of such assemblies. Outfitting these polymolecular entities with recognition units and reactive functional groups may lead to systems performing molecular recognition or supramolecular catalysis on external or internal surfaces of organic materials (molecular layers, membranes, vesicles, polymers, etc.) or on inorganic materials (zeolites, clays, sol-gel preparations, etc.). Control of the distance between different reaction sites is already possible in a crude way by incorporating the wished-for reaction site in an L-B film (either at the head or the tail of a fatty acid) and then building up, layer by layer. Undoubtedly, as research progresses in this area, finer and finer control will be obtained over both the ability to incorporate different reaction sites in a "network" molecule, and the ability to judiciously control their level of polymerization.
The further interest in self-organization, aside from the possibility of creating devices out of self-assembling components, is the possibility of being able to trigger supramolecular assemblies by optical, electrical, or chemical stimuli that result in a large-scale change of the organization, thus amounting to signal amplification and expression on the macroscopic level. Such a case of signal amplification is given below.
If two (or more) complementary units A or A′ are grafted onto a backbone (also called a template), mixing the two can lead to the self-assembly of a linear or crosslinked, main-chain supramolecular "copolymer" species.
As an example of this, condensation of the complementary groups 2,6 diaminopyridine P and uracil U with long chain derivatives of L-, D-, or meso (M) tartaric acid yields substances LP2, LU2, MP2, MU2, and so on; each contains two identical units capable of undergoing association via triple hydrogen bonding (Fouquey, Lehn, and Levelut 1990). Whereas the individual (pure) species are solids, the mixtures (LP2 + LU2), (DP2 + LU2) and (MP2 + MU2) display liquid crystalline behavior. The overall process thus may be described as the self-assembly of a supramolecular liquid-crystalline polymer based on molecular recognition. Varying the concentration allowed one to track, via electron microscopy, the progressive assembly of supramolecular-polymolecular entities from small up to very large ones: small nuclei → filaments → treelike structures → fibers. The species formed presented helicity induced by the chirality of the original components and successively transferred to the larger scale entities. A mixture of all four of the components yielded long superhelices of opposite handedness that coexisted in the same sample—in other words, spontaneous resolution through chiral selection in a molecular recognitiondirected self-assembly. Here, molecular chirality is transduced into supramolecular helicity, which is then repeated in the structure of the material at nanometric and micrometric scales, amounting to a size amplification of chirality.
It should be pointed out that the separation of material into mesoscopic entities of one or the other chirality implies a sorting out of molecular components. Due to the supermolecular nature of the bonds, this signal can be reversed and the system dissolved back into its components simply by heating.
Another example of complex self-assembly is the formation of double-stranded helices—termed helicates—around metallic ions of specific coordination geometries. Certain molecular strands containing two to five 2,2′ bipyridine groups will spontaneously assemble in the presence of Cu(I) ions. This double helix structure results from the tetrahedral-like coordination imposed by each Cu(bipy)−2+ site and from the design of the ligands, which disfavors binding to only a single strand. Using a slightly different molecular chain and metal ions, which favor an octahedral symmetry (nickel [II]), one can produce similar triple helical complexes, as shown in figure 2.14 (Kraemer, Lehn, and Marquis-Rigault 1993).
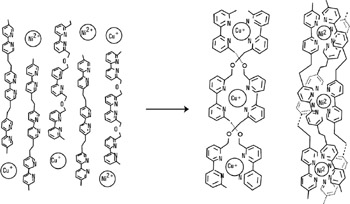
Figure 2.14: Self-recognition in the self-assembly of double and triple helices from a mixture of two different oligobipyridine strands and of Cu(I) and Ni(II)ions (ClO4− counter-ions not shown) (Kr mer, Lehn, and Marquis-Rigault. 1993 Proc. Natl. Acad. Sci. USA.)
This can been seen as a sort of molecular program being carried out, with the information carried both in the strands and in the ions. Molecular programming involves the incorporation of instructions into molecular components for the generation of the desired supramolecular architecture. Depending on the design of the interaction patterns between the components, more or less strict programming of the output entity will be achieved. The program is molecular, the information being carried in the covalent framework—while its operation through noncovalent recognition "algorithms"—is supramolecular. The processing of molecular information via molecular recognition events implies the passage from the molecular to the supramolecular level. In the example outlined above, in addition to the nature and the coordination geometry of the metal ions involved, three main features that bear structural information determining the nature and shape of the helical species formed may be distinguished: (1) the precise structure of the binding site—it specifies the ability to coordinate metal ions with a given geometry as well as the number of strands bound; (2) the spacer separating the binding sites—it must favor interover intrastrand binding, and it influences the tightness of the helix (the pitch) through the rotation of one metal center with respect to the next; and (3) most important, the configuration of the coordination centers determines whether the structure is indeed of a helical nature; helicity implies that all metal centers have the same screw sense. The program containing all these information features is molecular and linear. Its operation is supramolecular and takes place through the reading of the information according to the coordination algorithm of the metal ion.
The full power of such applied molecular recognition comes to fruition in such systems. When all of the components (both types of bipy [bipyridine] strands and both types of metal ions) are mixed together, only the "pure" double helices containing Cu(I) and the triple helicate with Ni(II) were formed, with no crossover (Kr mer 1993). This is an example of a parallel operation of two "programmed" molecular systems leading to the clean self-assembly of two well-defined helical complexes from a mixture of their four components.
|