13 - Normal Eye and Ocular Adnexa
Editors: Mills, Stacey E.
Title: Histology for Pathologists, 3rd Edition
Copyright 2007 Lippincott Williams & Wilkins
> Table of Contents > VI - Thorax and Serous Membranes > 18 - Lungs
function show_scrollbar() {}
18
Lungs
Thomas V. Colby
Kevin O. Leslie
Samuel A. Yousem
Normal Structure and Histology
The following review is based on several standard references on the topic (1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16).
General
The lungs are paired intrathoracic organs that are divided into lobes (three on the right right upper lobe, right middle lobe, right lower lobe; two on the left left upper lobe, left lower lobe). The lingula is a rudimentary appendage arising from the left upper lobe and is analogous to the middle lobe on the right. The lobes are further divided into bronchopulmonary segments (Table 18.1).
The segmental anatomy of the lung is important for radiologists, bronchoscopists, and pathologists in defining the location of lesions. The lobes are divided by fissures and have their own pleural investments. The segments are not separated by fissures and do not normally have separate pleural investments, although they are recognizable on the basis of their supplying bronchi (segmental bronchi).
The primordial lungs arise as ventral buds of the foregut extending into the primitive thoracic mesenchyme. Bronchial cartilages, smooth muscle, and other connective tissues are derived from the mesenchyme that surrounds these dichotomatously branching buds. The phases of airway and lung parenchymal development are summarized in Table 18.2.
As the lung progresses through these phases of development, there is a complex series of epithelial-mesenchymal reactions modified by physiologic mechanical forces and humoral factors. These events are overseen by master genes (such as homeobox genes), nuclear transcription factors, hormones, and other soluble mediators such as growth
P.474
factors (17,18). Some of these mediators and their relationship to lung development are shown in Table 18.3.
Table 18.1 Bronchopulmonary Segments* | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
Table 18.2 Phases of Lung Development | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Table 18.3 Lung Development and Regulatory Factors* | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
Airways
The airways serve as conduits for air traveling to and from alveoli; for evacuation of material along the mucociliary escalator; and for immunologic, protective, air-moisturizing, and warming functions.
The airways arise by unequal dichotomous branching of the bronchial buds. In a normal individual, there are approximately 20 generations (range: approximately 10 30 divisions, depending on the location in and distance from the trachea) extending from the trachea to the respiratory bronchioles. In histologic sections from normal lungs, the diameter of an airway is approximately the same as its accompanying artery (and vice versa) (19). Disparities in size (of either airway or artery) suggest a pathologic condition. Airways are defined as follows:
Bronchi are cartilaginous airways and are usually more than 1 mm in diameter (Figure 18.1). They are conducting airways, and the cartilage plates in their walls prevent their collapse. The posterior wall is smooth muscle. The cartilage may calcify.
Figure 18.1 Bronchus. Alcian blue staining of the bronchus highlights the presence of goblet cells in the mucosa (slightly increased in this case), as well as the bronchial submucosalglands. Beneath the epithelial basement membrane, there is a vascularized layer of connective tissue with wisps of smooth muscle above the submucosal glands.
Bronchioles are airways that are usually less than 1 mm in diameter; they lack cartilage (Figure 18.2).
Nonrespiratory bronchioles represent all bronchioles proximal to respiratory bronchioles.
Terminal bronchioles are nonrespiratory bronchioles just proximal to respiratory bronchioles.
Respiratory bronchioles are airways that have alveoli budding from their walls.
P.475
In the large bronchi, the surface epithelium rests on a basement membrane, below which there is an elastin-rich layer of connective tissue; together these elements comprise the bronchial mucosa. Beneath the bronchial mucosa lies the submucosa, in which submucosal glands, cartilage, nerves, ganglia, and branches of the bronchial artery may be found. There is no clear histologic boundary between mucosa and submucosa. Outside the submucosa, there is a peribronchial sheath of loose connective tissue, which is continuous with that of the accompanying artery. The bronchial epithelium is a pseudostratified columnar epithelium composed primarily of ciliated columnar cells with interspersed mucous (goblet) cells. Lesser numbers of neuroendocrine cells, basal cells, brush cells, and migratory inflammatory cells are also normally present (see below).
P.476
The height of the pseudostratified columnar epithelium decreases progressively toward the periphery of the lung.
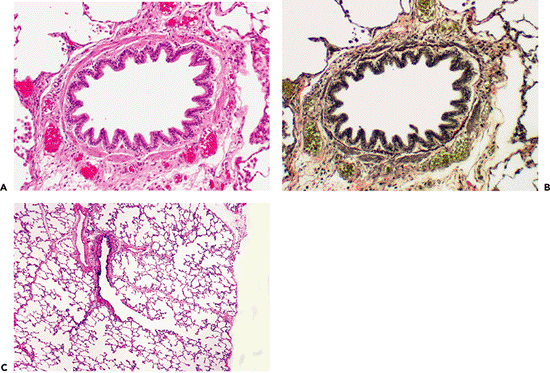 |
Figure 18.2 Bronchioles. Hematoxylin and eosin (A) and elastic tissue stains (B) illustrate normal bronchioles with a thin layer of connective tissue just beneath the epithelium overlying the elastica (B) and the smooth muscle investiture. The mucosa is low columnar, and there is no thickening of the subepithelial region. The smooth muscle is circumferential and is surrounded by an adventitial layer. C. A terminal bronchiole is continuous with the respiratory bronchiole, which extends into the alveolar ducts and ultimately the alveoli. |
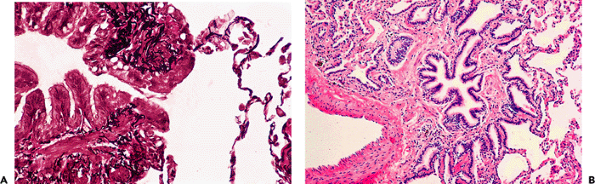 |
Figure 18.3 Lambert's canals. A. These canals represent communications between nonrespiratory bronchioles and adjacent alveoli and are only rarely observed in histologic sections. B. It is these canals that are thought to be the origin of peribronchiolar metaplasia, which is seen as a repair phenomenon involving peribronchiolar alveoli after bronchiolar injury. |
The walls of bronchioles are normally much thinner than those of the bronchi. The surface epithelium rests on a basement membrane, which overlies a thin layer of loose elastin-rich connective tissue. This is surrounded by a muscle layer (muscularis), which is in turn invested by a peribronchiolar connective tissue sheath continuous with that of the adjacent artery. The distinction of mucosa from submucosa in bronchioles is arbitrary, and sometimes the tissue beneath the basement membrane is referred to as submucosal or submembranous connective tissue.
The airway basement membrane region consists of three layers: lamina lucida, lamina densa, and lamina reticularis. The last of these consists of very fine fibrillary collagen and is only found in adults. It is not technically part of the basement membrane, and it is this zone that is thickened in asthma and other inflammatory airway conditions. The basement membrane stains with digested PAS stains and immunostains for type IV collagen and laminin.
Direct communications between nonrespiratory bronchioles and alveoli have been identified and termed Lambert's canals (20). These are thought to be involved in collateral ventilation. Lambert's canals are rarely visible in histologic sections (20). Lambert's canals may be conspicuous in scarred airways surrounded by metaplastic bronchiolar epithelium, which is seen to be continuous with the bronchiolar surface epithelium through Lambert's canals. This has been termed Lambertosis, bronchiolarization, and peribronchiolar metaplasia (Figure 18.3). Sometimes the marked peribronchiolar metaplasia is the most prominent pathologic finding in a surgical biopsy. Peribronchiolar metaplasia is a not uncommon incidental finding in a variety of diffuse lung diseases. It may also be the sole or predominant change and the cause of the diffuse lung disease as described in some recent studies (21,22,23).
Cell types lining the airways include basal cells, Kulchitsky cells, ciliated cells, Clara cells, goblet cells, intermediate cells, and brush cells. Ultrastructural abnormalities in the ciliated cells of the respiratory tract are known to be associated with pathologic conditions (e.g., primary ciliary dyskinesia). Goblet cells and ciliated cells decrease in number as the terminal bronchioles are approached; there is a concomitant increase in Clara cells, and the mucosa becomes less columnar and more cuboidal in appearance. Clara cells have secretory functions (e.g., surfactant-like material) and also act as progenitor cells after bronchiolar injury. Clara cell differentiation in tumors of the lung may be appreciated by the presence of apical periodic acid-Schiff (PAS)-positive, diastase-resistant granules, as well as by their electron microscopic apical-dense granules. Kulchitsky cells contain dense core granules and are part of the diffuse neuroendocrine system. Aggregates of neuroepithelial cells are called neuroepithelial bodies; they tend to occur at airway bifurcations. The finding of goblet cells in bronchioles usually signifies chronic airway injury. The cell types in the airways and parenchyma are summarized in Table 18.4 and illustrated in Figures 18.4, 18.5, and 18.6.
Smooth muscle of the airways plays an important functional role in regulating airflow and is arranged in a complex spiral pattern that becomes progressively less prominent in the distal conducting airways. This muscle receives nutrition from the bronchial arteries. At the level of the alveolar ducts, bundles of airway smooth muscle can be seen, interrupted by alveolar openings; in cross section, these may appear as isolated round or oblong aggregates.
Submucosal salivary-type glands containing both serous and mucous cells are found in the larger bronchi. In older individuals, oncocytic metaplasia can be seen in these glands (Figure 18.7). Within the walls of the large airways, ganglia, nerves, and bronchial arteries are found.
P.477
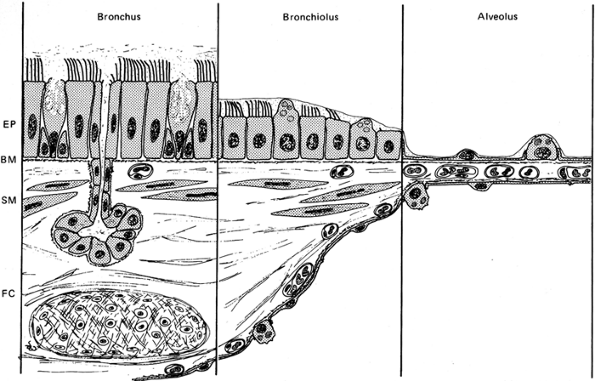 |
Figure 18.4 Respiratory tract epithelia. There is a progression of pseudostratified columnar epithelium in the large airways, to a more cuboidal epithelium in the small airways, to squamous type epithelial cells (type I pneumocytes) in the alveoli. The epithelium in the large airways is designed for maintaining and moving the mucous stream, whereas the squamous pneumocytes in the airspaces facilitate gas transfer. (Reprinted with permission from: Weibel ER, Taylor CR. Design and structure of the human lung. In: Fishman AP, ed. Pulmonary Diseases and Disorders. Vol 1. 2nd ed. New York: McGraw-Hill; 1988:14. ) |
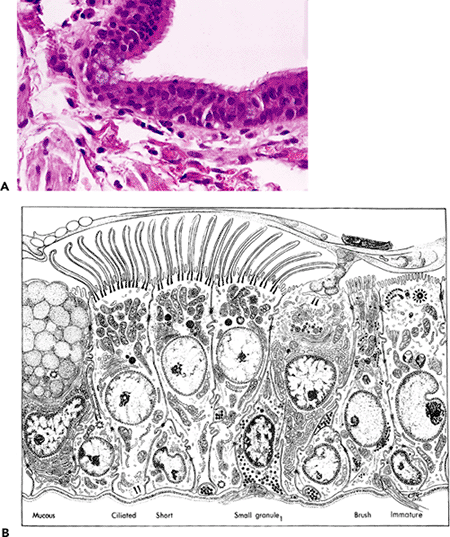 |
Figure 18.5 Bronchial epithelium. A. Normal bronchial epithelium is pseudostratified and columnar, with numerous ciliated cells and scattered basophilic and flocculent-appearing goblet cells. B. This ultrastructural schematic of bronchial epithelium shows the various cell types present. (Figure 18.5B reprinted with permission from: Sorokin SP. The respiratory system. In: Weiss L, ed. Cell and Tissue Biology: A Textbook of Histology. 6th ed. Baltimore: Williams & Wilkins; 1988:769. ) |
P.478
Table 18.4 Major Cell Types of the Lower Respiratory Tract* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
P.479
P.480
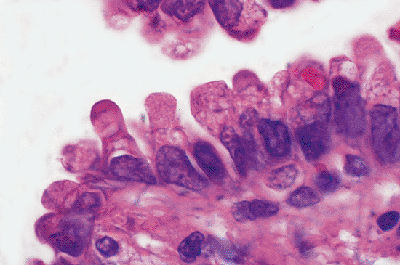 |
Figure 18.6 Clara cells. Although Clara cells sometimes can be identified in bronchioles, these are seen to best advantage in neoplasms such as this nonmucinous bronchioloalveolar carcinoma. The apical snouting and increased cytoplasmic density are apparent. |
Lobule and Acinus
The lung lobule is grossly visible and represents the smallest anatomic subunit of the lung that is bounded by connective tissue (interlobular) septa, which may appear to invaginate from the visceral pleura (Figure 18.8). Lobules are 1 to 2 cm in diameter and are visible to the naked eye on both the pleural surface of the lung and on cut surfaces of the parenchyma by their septal demarcations. Lobules are accentuated in fibrotic conditions in which the septa contract (e.g., honeycombing, healed infectious pneumonia, chronic pleural inflammation). Pulmonary lobules are identifiable with high-resolution computed tomography (CT) scanning of the lung. The term lobule as used here has also been referred to as the secondary lobule of Miller (12). The use of the term secondary lobule is discouraged because it implies that there is a primary lobule, and the latter term is not in current use.
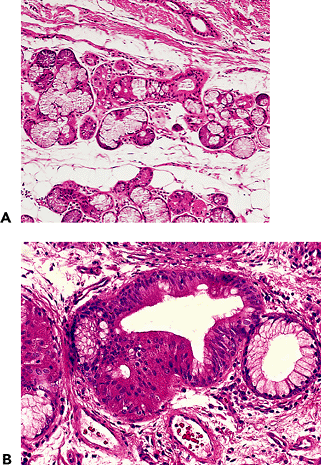 |
Figure 18.7 Bronchial submucosal glands. A. The bronchial submucosal glands are normally located in the submucosa above the bronchial cartilage and contain mixed seromucous glands with a duct leading to the bronchial mucosa (also Figure 18.1). B. Oncocytic metaplasia involving bronchial submucosal glands is relatively common. |
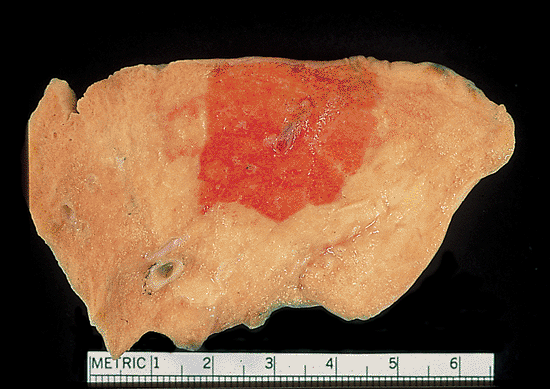 |
Figure 18.8 Pulmonary lobule. This cut section of normal lung tissue (from an explant that could not be used for technical reasons) shows focal hemorrhage highlighting a pulmonary lobule. The hemorrhage stops abruptly at the interlobular septa and, centrally, a bronchovascular bundle can be appreciated. The lobule is approximately 2.0 cm in diameter. |
The functional unit of the lung is the acinus, where gas transfer takes place (Figure 18.9). The precise definition of the acinus has varied. Some define it as the lung tissue supplied by a single terminal bronchiole (12,15). According to this definition, each pulmonary lobule comprises some 3 to 10 acini. The acinus has also been defined as a respiratory bronchiole and its supplied alveolar ducts and sacs (10,13); using this definition, each lobule comprises some 20 to 30 acini and the acini are 1 to 2 mm in diameter.
Squamous (type I pneumocytes) and cuboidal (type II, or granular, pneumocytes) epithelial cells line the alveoli (Table 18.4). Gas exchange takes place across the cytoplasm of type I cells. Type II cells are the progenitors for type I cells, produce surfactant, and proliferate after injury to restore alveolar epithelial integrity. Type II cell hyperplasia represents a nonspecific marker of alveolar injury and repair (Fig 18.10). Macrophages are a normal finding in the lung, scattered over the surfaces of the alveoli and percolating into the interstitium; a number of subpopulations of pulmonary macrophages are definable, based on their anatomic locations and their role in host defense clearance functions (31). Pulmonary macrophages are greatly increased in cigarette smokers; increased Langerhans cells are also found in the
P.481
bronchiolar epithelium of smokers, but their recognition requires special stains (e.g., CD1a) and quantitative studies.
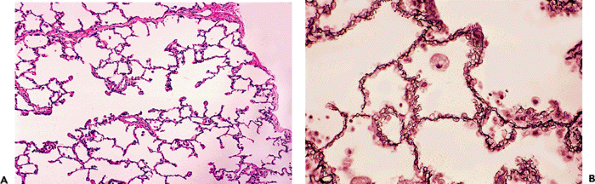 |
Figure 18.9 Distal lung parenchyma. The acinus is the functional unit of the lung where a gas transfer takes place. A. An alveolar duct extends from the left to right and communicates directly with alveolar spaces; a small interlobular septum (top) and the pleura (right) are present. (From the case illustrated in Figure 18.2C.) B. Reticulin stain highlights the vasculature of the alveolar septum, showing pulmonary capillaries winding around the access of the alveolar wall to maximize gas transfer surface area. A foamy macrophage is present (upper center), a normal finding in lung parenchyma. |
The epithelial and capillary basement membranes in the alveolar septum are irregularly fused. Gas transfer takes place across the alveolar-capillary membrane, which includes the attenuated cytoplasm of the type I cell, the endothelial cell cytoplasm, and their fused basement membranes. Pores of Kohn represent direct communications between adjacent alveoli via a pore in the alveolar wall. They are thought to be involved in collateral ventilation. Pores of Kohn are rarely visible with the light microscope.
The lung is invested with a rich framework of connective tissue coursing throughout the interstitium. It is well developed and easily visible along bronchovascular sheaths and in the septa that delimit lobules. This framework is continuous from the hilum to the pleura and encompasses the interstitial compartment of the lung down to the level of the alveolar wall and perivascular areas. Within the alveolar walls, collagen, elastic fibers, mesenchymal cells, and a few inflammatory cells can be identified ultrastructurally. This alveolar interstitial space is normally inconspicuous by light microscopy in adults. In children (up to approximately age 4), some interstitial widening and increased cellularity are a normal histologic finding.
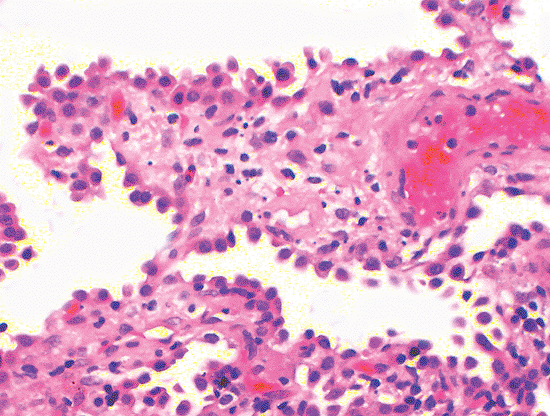 |
Figure 18.10 Alveolar cell hyperplasia. A reactive proliferation of type II cells (alveolar cell hyperplasia) is common after injury. This case illustrates how this appears as a single row of cells protruding from the alveolar surface. Cytologic atypia is lacking. The interstitium shows edema, inflammatory cells, and some fibrinous exudate consistent with recent lung injury. |
Given the concentration of nuclei of the various cell types comprising the alveolar wall, it may be difficult to decide when some degree of cellularity is pathologic. Although one may attempt to count nuclei in a single alveolar wall, it is more practical to assess inflammatory cell infiltrates in the perivenular regions and to compare various foci in the biopsy since interstitial infiltrates are rarely perfectly uniform in distribution.
Vasculature
The lungs have vascular systems: bronchial (systemic) arteries and veins and pulmonary arteries and veins. The alveolar microvasculature is designed for oxygenation of venous blood derived from the pulmonary arteries. Large (elastic) pulmonary arteries in infants are similar to the aorta in structure; the elastic fiber lamellae become more irregular, fragmented, and less compact in adulthood. Elastic tissue remains relatively prominent in the pulmonary arterial tree to approximately the point where bronchi become bronchioles. At this juncture, the pulmonary arteries become primarily muscular arteries. The muscular pulmonary arteries and arterioles have an internal and external elastic
P.482
membrane (Fig. 18.11A); pulmonary veins have only a lamellated single (outer) elastica (Fig. 18.11B). Small intra-acinar pulmonary veins merge into larger veins in the interlobular septa. The bronchial arteries in the walls of bronchi are part of the systemic circulation and have pressures similar to systemic arterial pressures. The pulmonary circulation is a low pressure system with a normal mean pressure of approximately 10 mmHg (it is somewhat higher at higher elevations).
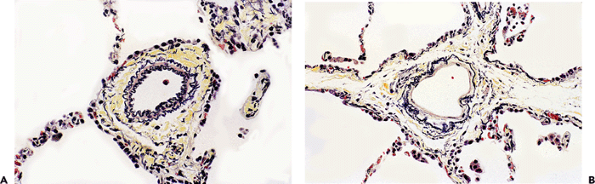 |
Figure 18.11 Pulmonary vasculature (elastic stains). Normal pulmonary arteries contain two elastic lamina (A), whereas veins have a single elastic lamina (B). The location of a vein within a septum (B) is also very helpful in identifying it as a vein. |
It may be difficult to separate small pulmonary arterioles from venules, especially since a single elastic lamina forms as arterioles get smaller. In pathologic states, the veins may develop muscular hypertrophy and increased mural thickness (arterialization), making the distinction of arteries from veins difficult. The location of the vessel, particularly if it is in a septum or accompanied by an airway, is extremely helpful (and sometimes the only way) when separating pulmonary veins from pulmonary arteries. In mild pulmonary hypertension, the pulmonary arteries may appear histologically normal; in general, pressure measurements taken at catheterization are more reliable than histologic subtleties in determining the pressure and degree of pulmonary hypertension. Only plexiform lesions and dilatation lesions connote unequivocal pulmonary hypertension. Conversely, the presence of some degree of diffuse interstitial fibrosis or localized pulmonary scars is commonly associated with mural thickening of pulmonary vessels in the absence of pulmonary hypertension.
Lymphatics and Lymphoid Tissue
The lung is invested with a rich supply of lymphatics and lymphoid tissue. Lymphatic drainage is toward the hilum of the lung. Lymph fluid from the lower lobes tends to drain to infratracheal lymph nodes, with lymph fluid from the remaining portions of the lung draining to tracheobronchial lymph nodes. On the left side, the lymph fluid drains into the thoracic duct; and, on the right side, it drains into the right bronchomediastinal trunk. Both of these ultimately drain into the left and right subclavian veins, respectively. Lymphatic channels are found along bronchovascular structures and pulmonary veins and in the septa and pleura. Valves may be apparent in some sections. Lymphatics do not extend into alveolar walls. The lymphatics vessels are inconspicuous except in pathologic states, such as pulmonary edema or lymphangitic carcinoma. Lymphoreticular infiltrates and some pneumoconioses tend to be distributed along the lymphatic routes, but the lymphatic vessels themselves may not be prominent.
Lymphoid tissue may be seen as small collections of lymphocytes along the lymphatic routes, especially branch points of the bronchovascular bundles; lymphoid tissue is generally absent or inconspicuous except in pathologic states. Lymphoid tissue along the airways is part of the diffuse mucosa-associated lymphoid tissue (MALT) and in the lung is referred to as bronchus-associated lymphoid tissue (BALT). Submucosal lymphoid tissue in the intermediate and small airways is associated with flattening and attenuation of the overlying respiratory mucosa (lymphoepithelium), which has increased HLA class II antigens. At these sites, B lymphocyte emperipolesis is common and is thought to reflect active antigen processing by the BALT. Bronchus-associated lymphoid tissue displays the same reactions observed in lymphoid tissue at other sites; for example, reactive hyperplasia and immunoblastic proliferation, which can be confused with lymphoreticular malignancies.
Immunophenotypically the lymphoid tissue of BALT has an immunoarchitecture with four identifiable compartments, including B-cell rich follicles, B-cell follicular mantle and marginal zones, and T-cell rich interfollicular regions. A follicular dendritic cell network is present. Polyclonal plasma cells are identified in the perifollicular tissue. The finer details of the immunoarchitecture and the
P.483
immunophenotypic characterization of these cells are beyond the scope of this review. The follicles tend to be polarized into a darker side and a paler side, with the latter oriented toward the epithelial surface; BALT also shares features of MALT as seen at other sites. In normal adult lungs, BALT is rarely present, and its presence correlates with some form of chronic antigenic stimulation (32,33,34). According to Tschernig et al. (34), BALT is not present at birth but is found normally with increasing age, probably as a result of exposure to environmental antigens. After the individual has been exposed to most of the common antigens, the BALT regresses and dendritic cells in the airways assume the role of antigen uptake and presentation. Its reappearance in adults follows chronic antigen stimuli, such as chronic infection.
As currently defined, BALT refers only to lymphoid tissue along the airways (35) and not to the lymphoid tissue that may be seen in the pleura and septa as part of diffuse lymphoid hyperplasia in the lung. Hyperplasia of BALT is frequently accompanied by lymphoid hyperplasia at these other sites.
Intrapulmonary peribronchial lymph nodes are a normal finding, but peripheral intraparenchymal lymph nodes are less common; however, in smokers and others with high dust exposure, they are increasingly recognized (and biopsied) with current imaging techniques (36). Intrapulmonary lymph nodes are usually septal or subpleural in location (see below). Anthracosis and small amounts of silica and silicates in these nodes is common and nonspecific.
Pleura
The visceral pleura is composed of connective tissue, elastic tissue, and an outer mesothelial layer (Figure 18.12). The elastic tissue is often not a single layer, and several layers may be apparent. In pathologic conditions the elastic tissue may greatly increase. Elastic tissue stains are useful in assessing whether a given pathologic process, such as carcinoma, has transgressed the visceral pleura (37), but interpretation is difficult when the pleura is fibrotic and the elastic tissue is increased. Pleural assessment is important in separating T1 carcinomas from T2 carcinomas; the latter invade the visceral pleura (21). Lymphatic vessels that are continuous with those in the interlobular septa are also identifiable in the pleura. When the pleura is fibrotic and in foci of pleural adhesions, vessels may be thick and sclerotic or even pseudoangiomatous in appearance. Fatty metaplasia is often striking in foci of pleural and subpleural scarring. The visceral pleura often remains viable over pulmonary infarcts because it has a separate vascular supply. Submesothelial fibroblasts may undergo mesothelial metaplasia (and become cytokeratin-positive) in inflammatory conditions of the pleura.
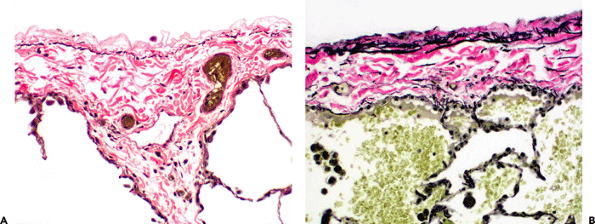 |
Figure 18.12 Pleural elastic tissue (elastic stains). The pleura contains an elastic tissue membrane, which may appear as a single thin layer of elastic tissue (A) or as a richer network with occasional elastic fibers distributed through the thickness of the visceral pleura (B.). The field illustrated in (A) shows a normal lymphatic at the juncture between an interlobular septum and the visceral pleura. |
Special Stains and the Evaluation of Lung Histology
While most diagnostic work in lung histology and pathology can be performed with routine hematoxylin-eosin (H&E) staining, there are a number of special stains that aid interpretation and or highlight findings. Elastic tissue staining is helpful in evaluating the pulmonary vasculature, airways, and pleura. Sometimes arteries and veins can be distinguished from each other with elastic tissue staining. Elastic tissue staining highlights damage to the intima and media that might not be apparent on H&E staining. Elastic
P.484
tissue stains are also useful in highlighting abnormalities of bronchioles and may confirm the bronchiolar or alveolar duct origin of small scars in cases of complete obliteration of the small airways. Elastic tissue staining is helpful in highlighting the pleural elastica and its relationship to tumors.
Trichrome staining (or other stains highlighting connective tissues) may also be useful in highlighting normal and pathologic findings in the lung, but they do not provide quite as much information as elastic tissue stains. Trichrome staining may be useful in assessing the distribution and extent of fibrosis in fibrosing diseases. Reticulin stains and stains for collagen type IV may be used to highlight the reticulin network of the lung. This staining may be useful in research studies and in highlighting normal histology but is not generally useful in diagnostic surgical pathology of the lung.
Immunostains for epithelial markers (such as EMA and cytokeratin stains) may be beneficial in atelectatic or fibrotic lung when it is difficult to appreciate the architectural features on H&E staining. Comparing these stains with stains for endothelial cells (such as CD31 or CD34) may provide an additional aid in assessing lung architecture and structural relationships. Use of CD31 may be somewhat confusing since alveolar macrophages frequently show prominent staining. Lymphoid tissue in the lung is assessed with the panoply of lymphoid markers used at other sites. Both CD3 and CD20 are often useful to check the proportion and distribution of T and B cells, respectively. In general, most inflammatory conditions have a preponderance of T cells as the diffuse component of the infiltrates, with scattered B-cell follicles. In lymphoproliferative conditions, cytokeratin staining highlights lymphoepithelial lesions. Both S-100 and CD1a are useful in identifying Langerhans cells; the latter stain is much more specific.
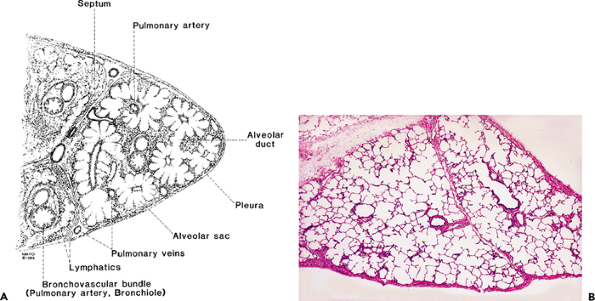 |
Figure 18.13 Wedge lung biopsy. A. This stylized diagram is used to depict anatomic landmarks. Structures depicted in (A) can be appreciated in an actual wedge biopsy specimen (B). |
Mesothelial cells lining the pleura typically stain with calretinin, cytokeratin 5/6, and WT-1 (nuclear positivity).
Pattern Recognition Based on Normal Anatomic Landmarks
It is useful to define pathologic conditions in the lung in relation to normal anatomic landmarks (Figure 18.13). This can usually be done with diffuse diseases and often with localized processes. Such an exercise in diffuse lung disease is extremely useful in correlation with gross findings, as well as with high-resolution computed tomography (HRCT) scanning (38). Histologic patterns and their corresponding HRCT patterns can be recognized and are shown in Table 18.5.
It is apparent that there is close correlation (but not a 1:1 relationship) between histology and HRCT. With some communication between the pathologist and the radiologist, there is a significant mutual appreciation for the similarity of the abnormalities seen. In this regard, the pathologist may be significantly aided by HRCT findings, which may suggest distributions that might not be apparent in biopsy material because of such things as the size of the biopsy (e.g., transbronchial biopsy) or sampling issues (nonrepresentative biopsies).
P.485
Table 18.5* Corresponding Histologic and Radiologic Patterns | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
Site-Related Changes Commonly Seen in Surgical Pathology Material
Site specific changes that may be primary lesions or incidental findings in surgical material are shown in Table 18.6.
Biopsies from lobar tips, particularly from the lingula or right middle lobe, may show incidental inflammatory and fibrotic changes (39), including interstitial fibrosis, epithelial metaplasia, and even focal honeycombing all of which may not be representative of a diffuse process. Myointimal proliferation is common in the arteries and veins of these biopsies. The airspaces may contain aggregates of macrophages and neutrophils. Because of their accessibility, these sites are often biopsied. Incidental changes in lobar tips are usually obvious as such, since the more proximal lung tissue is either not affected or significantly less affected. Thus, in evaluation of lobar tip wedge biopsies, the findings of greatest significance are often in the more proximal portions of the specimen. Nonspecific inflammatory changes in lobar tips become a problem in small wedge biopsies, particularly those that are 2 cm or less in greatest dimension. Since lobar tips are so readily accessible to the surgeon, it is difficult to discourage surgeons from biopsying them. If possible, such biopsies should be at least 3.0 cm in greatest dimension. The possibility of middle lobe syndrome (which may affect the lingula, the right middle lobe, or both) (40,41) should be considered for persistent infiltrates in these sites.
Table 18.6 Site-Specific Changes in Lung Tissue | |
|---|---|
|
Apical caps (Figure 18.14) were once thought to be the result of healed tuberculosis, but they are common in patients who have never had tuberculosis; they are now thought to be an ischemic alteration related to their apical position (42,43). Apical caps are best known in the apex of the upper lobe, but they are also encountered in the upper regions of the lower lobe (44). Apical caps are regions of fibrosis in the pleura and subpleural lung parenchyma that
P.486
are rich in thickened elastic fibers; the background structure of collapsed alveolar walls and interposed eosinophilic collagen can often be discerned in the tissue; elastic tissue stains enhance these features. Ossification and nests of metaplastic pneumocytes may be present. The changes are sufficiently distinctive that apical caps can often be suspected histologically, even when one does not know the site of origin; even so, any ancient lung scar may sometimes show a similar elastotic appearance. Apical caps in older individuals are sometimes biopsied or resected to exclude a carcinoma or a visceral pleural tumor (44).
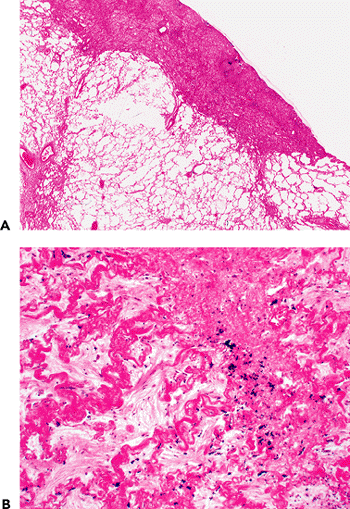 |
Figure 18.14 Apical cap. A. Apical caps are found at the apex of the upper lobe, as well as the apex of the lower lobe. They represent regions of pleural and subpleural thickening by elastotic fibrous tissue that may be grayish or eosinophilic in appearance. B. Higher power sometimes shows a rich elastic tissue network that appears to highlight alveolar wall structure; some anthracotic pigmented is also noted. |
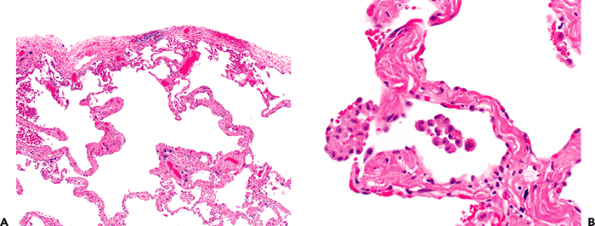 |
Figure 18.15 Subpleural emphysematous change in smoking. A. There is simplification of airspaces. B. Some of the alveolar walls show mild fibrosis with hyaline appearing collagen. Occasional clusters of pigmented alveolar macrophages are also noted. |
Centriacinar (centrilobular) emphysema is a pathologic abnormality that is more common and more severe in the upper lobes (45); it is found predominantly in cigarette smokers and is a common finding in lobes resected for bronchogenic carcinoma. Emphysematous changes are frequently accompanied by some degree of fibrosis, particularly when bullous change is present. The fibrosis tends to appear as strands of dense, hypocellular, brightly eosinophilic collagenous septa traversing the emphysematous spaces. Anthracosis is also a common finding in smokers and urban dwellers. Focal pleural and subpleural fibrosis (Figure 18.15) is extremely common, especially as an incidental microscopic finding in lobar resections from smokers (46); abnormal airspaces formed by irregular fibrous septa are accompanied by smooth muscle hyperplasia, mucostasis, and bronchiolar metaplasia.
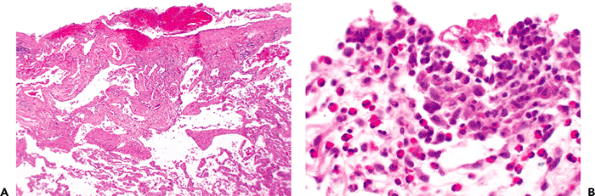 |
Figure 18.16 Pneumothorax. A. So-called subpleural blebs are thought to predispose to pneumothorax. These are regions of pleural and subpleural scarring with abnormal airspaces somewhat reminiscent to smoking-related changes (also Figure 18.15). B. Pneumothorax is often associated with a pleural reaction (top), which has been labeled eosinophilic pleuritis because of the association of eosinophil infiltrate and mesothelial and macrophage reaction on the pleural surface. |
Subpleural blebs (Figure 18.16) are often the only pathologic change found in patients with recurrent pneumothoraces and who don't have diffuse lung disease (47). These are typically evident in the upper lobes. By convention, blebs are defined as being less than 1.0 cm in diameter and some are derived from air dissection into the visceral pleura; bullae are 1.0 cm or greater in diameter (48).
P.487
Their rupture introduces air into the pleural space, inciting a mesothelial proliferation with numerous macrophages, giant cells, and eosinophils (referred to as eosinophilic pleuritis), and this reaction may accompany any condition that is associated with pneumothorax (49). Collections of interstitial air in the lung tissue, with or without giant cell and/or eosinophilic reaction, may be an accompanying finding. Focal subpleural scarring can usually be distinguished from chronic fibrosing interstitial pneumonias by its restriction to the subpleural region, lack of active fibroblastic proliferation, and the relative abrupt transition to normal alveolar walls. Clinical history and radiographic findings may also be supportive. When an interstitial lung disease such as pulmonary Langerhans cell histiocytosis (eosinophilic granuloma) or lymphangioleiomyomatosis has resulted in pneumothorax, both the primary process and the secondary eosinophilic pleuritis may be recognizable on the biopsy.
Adhesions between the pleura and chest wall are composed of fibrovascular tissue with pockets of bland mesothelial cells and are identical to their peritoneal counterparts. The adhesions identified in surgical specimens and even autopsy tissue often have no obvious cause and represent an incidental finding. Dense, hyalinized fibrotic pleural (visceral or parietal) plaques (50,51,52) are the consequence of a variety of prior inflammatory events. Their presence bilaterally often is an incidental finding; and, if there is no obvious inflammatory cause, prior asbestos exposure is likely (53).
Artifacts Seen in Lung Biopsy and Resection Material
Artifacts related to lung biopsy of prior procedures are described in Table 18.7.
Knowledge of the clinical course of events prior to lung biopsy usually allows the pathologist to avoid misinterpreting changes of prior instrumentation. Previous bronchial biopsies may cause hemorrhage, airway inflammation, ulceration, and a granulation tissue reaction. Strips of epithelium may be dislodged by mechanical trauma and embedded in inspissated mucus; residual basal cells may be all that is left adhering to the basement membrane. Bronchoalveolar lavage can produce vacuolation of alveolar pneumocytes and macrophages. Previous needle biopsies may induce necrosis and hemorrhage in the parenchyma, followed by organization and reactive epithelial atypia (Figure 18.17). Patients who have been on positive-pressure ventilation (Figure 18.18) may have disproportional bronchiolar and alveolar duct distension, especially when high inspiratory pressures are used in the adult respiratory distress syndrome (ARDS). Some associated acute inflammatory exudate in the lumen is common, often with relative absence of associated inflammation in the airway wall or surrounding alveolar spaces.
Table 18.7 Artifacts Seen in Lung Biopsies and Resections | |
|---|---|
|
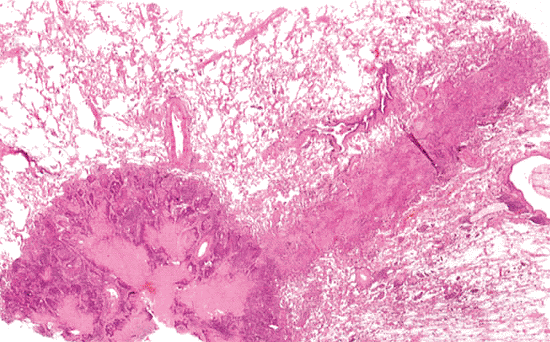 |
Figure 18.17 Needle tract to a small peripheral adenocarcinoma. The biopsy had been performed a week earlier, and the needle tract shows evidence of hemorrhage, necrosis, organization, and epithelial regeneration. |
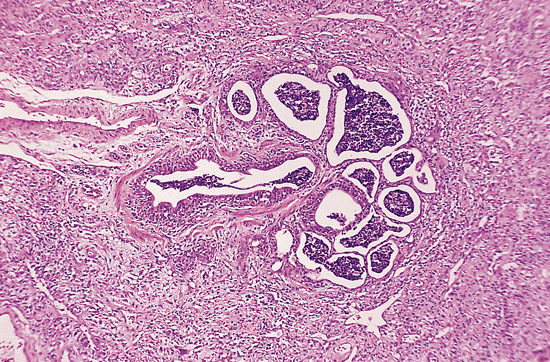 |
Figure 18.18 Ventilator-associated changes. Common changes in the setting of positive-pressure ventilator therapy include distension of bronchioles, flattening of their epithelium, and an acute inflammatory exudate in the lumen with minimal change in the surrounding alveolar wall. |
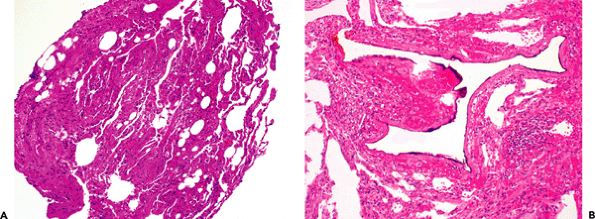 |
Figure 18.19 Bubble artifact. A. This fragment from a transbronchial biopsy shows compression and atelectasis (left) with some recognizable alveolar walls (right). The rounded spaces represent bubble artifact, which is common in such compressed biopsies. B. Sponge artifact is illustrated. Note the marked irregularity of the space corresponding to the irregular surface of sponges used in cassettes. |
P.488
Compression of the lung tissue, particularly in transbronchial biopsies, may produce rounded spaces in alveoli that resemble fat vacuoles and can easily be mistaken for exogenous lipoid pneumonia (Figure 18.19). This artifactual change, often called bubble artifact (54,55), can be recognized because there are no macrophages or giant cells with small intracytoplasmic lipid vacuoles (all the vacuoles are extracellular), and there is usually little fibrosis, which is a constant feature of chronic exogenous lipoid pneumonia.
Compression of airways produces crinkling and telescoping of the epithelium, similar to that seen in endometrial biopsies. Sometimes entire strips of mucosa are displaced into the bronchiolar or alveolar spaces.
Compression-induced nuclear smearing artifact can be produced in any cellular tumor and even in reactive lymphoid tissue in the bronchial mucosa or biopsies of hilar or mediastinal nodes. The resulting changes may suggest small cell carcinoma. Reactive lymphoid follicles, lymphomas, and carcinoid tumors may be extremely difficult to distinguish from small cell carcinoma when this phenomenon is present. Recognition of these diagnostic pitfalls, examining multiple levels (especially at the periphery), and enlisting the aid of concomitant cytology specimens and immunohistochemistry allows resolution of most cases. In rare instances, rebiopsy may be necessary.
Atelectasis may be encountered in all types of lung biopsies since lung tissue is soft and readily compressible (54,55). It is particularly common in video-assisted thoracic surgical wedge biopsies because this procedure requires that the lung be collapsed and samples may become further compressed during retrieval through a small hole in the chest wall (56). Atelectasis may also be encountered in biopsies inflated by the pathologist when not all lobules uniformly inflate. This phenomenon may be misinterpreted as interstitial pneumonia or interstitial fibrosis because the apposition of alveolar walls produces apparent scarring and hypercellularity (Figure 18.20). With experience, this change can be recognized on routine H&E sections; connective tissue stains show an absence of scarring and a normal background of supporting fibrous tissue along vessels and in septa. In atelectatic lung, the vessels and septa may appear to have more collagen than normal and to be thickened because they are contracted and shortened. Careful assessment of the nuclei in atelectatic lung shows that most
P.489
are endothelial or epithelial in origin, rather than inflammatory. Leukocyte common antigen immunostaining and immunohistochemical stains for other lymphoid markers may be useful in this setting. It is also unusual in open biopsies (and larger specimens) for the entire specimen to be uniformly atelectatic; and, therefore, a low-power survey of the entire pattern generally helps one appreciate atelectasis merging with more normal lung. In fact, when significant fibrosis is present (usually associated with foci of honeycombing), atelectasis is rarely a problem because the more rigid fibrotic lung tissue tends to retain its configuration.
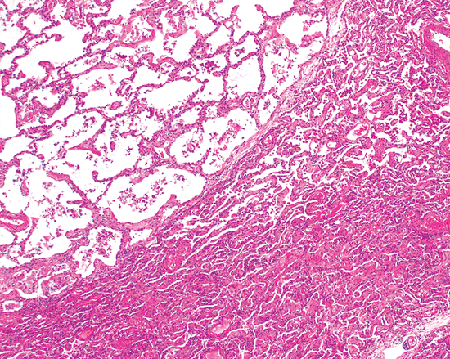 |
Figure 18.20 Atelectasis. Careful examination shows that recognizable alveolar walls can be traced into the region of atelectasis (lower right) and that the process stops somewhat abruptly at an interlobular septum that courses diagonally across the field (lower left to upper right). |
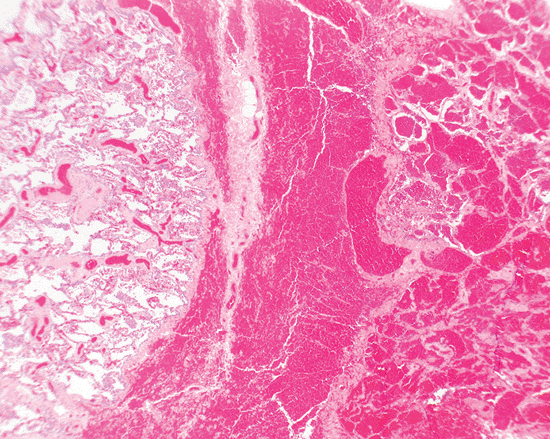 |
Figure 18.21 Traumatic hemorrhage. The right side of the field shows extensive hemorrhage into alveolar spaces, whereas the tissue on the left shows a complete absence of hemorrhage. There is also dissection of red blood cells into the interlobular septal region (center). Such a well-demarcated focus of hemorrhage and dissection of red blood cells into connective tissue would be very unusual in a diffuse alveolar hemorrhage syndrome. The findings are typical of traumatic hemorrhage. |
Fresh intra-alveolar hemorrhage due to the trauma of surgery is extremely common in biopsy material and should not be overinterpreted as pathologic (Figure 18.21). In fact, hemorrhage related to the trauma of the procedure is the most common cause of fresh blood in alveolar spaces. One can approach this problem from three points of view: the statistical, the histologic, and the clinical. Statistically, the vast majority of cases of fresh alveolar hemorrhage are traumatic since pathologic alveolar hemorrhage is relatively uncommon; thus, in any given case, acute hemorrhage is unlikely to be significant. When pathologic hemorrhage is present, there is usually (but not always) an associated fibrinous exudate or hyalin membranes, obvious distention of alveoli with blood, and evidence of prior hemorrhage manifested by hemosiderin-filled macrophages in the interstitium or airspaces. This last finding is not a reliable histologic criterion in patients who have been smokers and those with venous obstruction or chronic passive congestion.
The most common cause of macrophages staining positively with iron stains is respiratory bronchiolitis in smokers (Figure 18.22). The hemosiderin in smoker's macrophages is finely granular, in contrast to the coarse, dark blue staining in chronic hemorrhage as highlighted by the Prussian blue histochemical stain for iron. Nevertheless, it is surprising how many darkly staining Prussian blue positive cells can be seen in smokers.
Finally, the clinician can usually confirm whether an alveolar hemorrhage syndrome or alveolar hemorrhage due to some other cause (e.g., cardiac) is in the realm of possibility in any given patient.
Prolonged surgical manipulation of the lung, and even multiple transbronchial biopsies, can lead to margination of neutrophils in capillaries (especially those in the pleura), which mimics capillaritis (56). Capillaritis is usually associated with some evidence of an alveolar hemorrhage syndrome or other clinical or histologic features of a vasculitic syndrome (such as vascular necrosis, karyorrhexis, and fibrin thrombi). It is distributed throughout a biopsy and is not limited to those regions manipulated during surgery. Clamping of the biopsy specimen prior to removal can result in lymphatic obstruction, dilatation, and septal edema.
Lung pathologists are divided on the issue of inflation fixation of biopsy specimens. This can be easily accomplished with a syringe filled with formalin and a fine gauge needle. Careful inflation of a lung biopsy specimen (57) may be helpful diagnostically and is aesthetically pleasing since the lung architecture is more easily appreciated (and particularly amenable to photography). A heavy hand can create overdistension of the alveoli and an emphysematous appearance. If this occurs, clinical correlation may be required to assess whether emphysema is actually a clinical consideration. Patchy atelectatic (uninflated) portions of lung tissue are common in biopsies that have been nonuniformly inflated. One problem that may be encountered in inflated biopsies is that cells and fluid may be washed out of the airspaces. This is especially true of smoker's (respiratory) bronchiolitis, which may be quite subtle in inflated specimens. A practical compromise to the issue of whether or not to inflate wedge biopsies was proposed by Dr. Lewis Woolner at the Mayo Clinic several decades ago (personal communication). One takes a wedge biopsy (even specimens from which tissue has been taken for studies such as frozen section) and simply places it in a closed container of formalin and shakes it up for a few seconds. This simple procedure allows the tissue to inflate somewhat on its own and the resultant histologic sections are remarkably good.
Video-assisted thoracic surgical (VATS) lung biopsies (thoracoscopic biopsies) have largely replaced traditional open lung biopsy as the diagnostic procedure for obtaining wedge biopsies of lung tissue for histologic evaluation. Although there are minor disadvantages in comparison to traditional open lung biopsy, diagnostic accuracy does not appear to be compromised (56,58,59,60). Allowing for the
P.490
fact that bimanual palpation is not as feasible with VATS biopsies, and that the tissue is forcibly pulled through a small hole in the chest wall, it is remarkable that sampling error and specimen artifacts are only a minor problem. The specimen size approaches that achieved with traditional open lung biopsy, and the minor degrees of hemorrhage, atelectasis/overinflation, and neutrophil margination represent artifactual changes that do not compromise diagnostic accuracy (56,58,59,60).
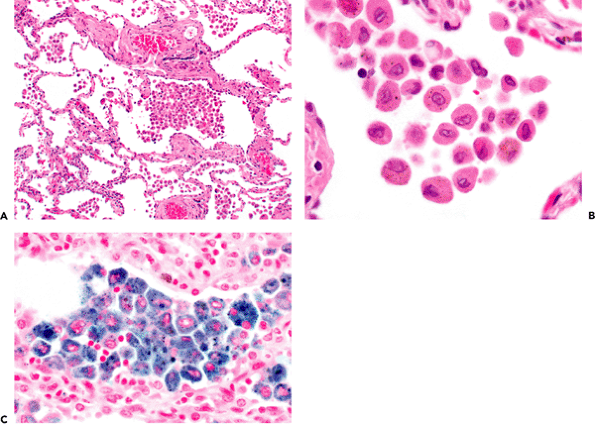 |
Figure 18.22 Respiratory bronchiolitis. A respiratory bronchiole (A) shows an accumulation of pigmented macrophages in the lung with slight associated interstitial widening. The macrophages have a slight tan-brown appearance, contain flecks of dark material (B), and are positive with Prussian blue staining (C) with a fine granularity. |
Plastic sponges are often put in cassettes to ensure that small specimens are not lost during processing. Sponge artifact, with triangular holes in the tissue, is a well-known artifact in any tissue that is processed this way, particularly if it is placed on the sponges prior to complete formalin fixation (55).
Incidental Findings in Lung Biopsy and Resection Tissue
There are a number of incidental findings in lung tissue that may not be related to the primary process that prompted the biopsy or resection (Table 18.8). Sometimes these incidental findings are of clinical and pathologic significance, and other times they are of no significance. Correlation of the individual finding(s) with the clinical and radiologic presentation helps to determine their significance.
Findings related to smoking and emphysema are extremely common, especially in lungs resected for bronchogenic carcinoma (46). They may be divided into three broad groups: large airway changes, small airway lesions, and abnormalities of the alveolar parenchyma.
In the large airways, one sees goblet cell hyperplasia, squamous metaplasia (with or without dysplasia), basement membrane thickening, hypertrophy and hyperplasia of bronchial glands with dilated ducts and mucostasis, and, often, a mild submucosal chronic inflammatory infiltrate (45,48).
The changes in the small airways may be quite dramatic and may mimic or even produce an interstitial lung disease (46,61,62). Smoking-induced respiratory bronchiolitis (Figure 18.22) includes goblet cell metaplasia, a mild inflammatory infiltrate in the airway walls, metaplasia of type II cells in the surrounding alveoli, mild peribronchiolar
P.491
fibrosis, and prominent accumulations of pigmented macrophages in adjacent airspace and the bronchiolar lumen. The pigmented macrophages (smokers' macrophages) contain phagocytosed debris from inhaled cigarette smoke and have prominent secondary lysosomes in their cytoplasm. This results in a dirty granular-tan or brown appearance to the cytoplasm. They contain PAS-positive (lysosomes) and finely granular Prussian blue positive (hemosiderin) material, as well as tiny, irregular flecks of brown-black material.
Table 18.8 Incidental Findings in Lung Tissue | |
|---|---|
|
The smoking-related changes in the most distal pulmonary parenchyma usually manifest as centriacinar emphysema with airspace enlargement and loss of alveolar walls (45,48). Histologic quantification of emphysema on biopsy material is difficult, although one can often determine whether it is present and subjectively quantify it. Bullous emphysema (with abnormal airspaces greater than 1 cm in diameter) is more common in the upper lobes and is usually associated with some fibrosis in the septa of the bullae and adjacent alveolar walls. The fibrosis is relatively acellular, noninflammatory, eosinophilic, and poorly vascularized. Metaplasia or ulceration of the epithelium and interstitial dissection of air occur in bullae and may induce a giant cell response analogous to that seen in persistent interstitial emphysema.
Asthmatics are predisposed to a number of lung conditions that may lead to lung biopsy, although the asthmatic changes themselves may not be the dominant lesion. These airway changes include goblet cell metaplasia in the airway epithelium, thickening of the basement membrane and submembranous region, smooth muscle hypertrophy and hyperplasia, lymphoid hyperplasia, and a variable infiltrate of eosinophils, lymphocytes, and a few neutrophils, and fibrous tissue in the wall (45,48). Mucostasis (including Curschmann's spirals) may be an accompanying feature; but, when mucostasis is extensive and is associated with sloughing of epithelial fragments into the airways (creola bodies), one should suspect that the asthma itself is the main lesion. Such an appearance is typical of status asthmaticus. Some distal atelectasis with a few airspace macrophages and eosinophils may also be seen. In patients with quiescent asthma or past history of asthma, the airways may be entirely normal or show only minor inflammatory or fibrotic changes.
Metaplastic bone, including bone marrow and calcification, is an aging change that is occasionally seen in bronchial cartilages (63). Metaplastic bone may also be seen in regions of scarring (dystrophic ossification), particularly in apical caps. Small bony nodules may also be seen with no apparent associated pathologic changes. In some older individuals with chronic bronchitis, the bronchial submucosa may have a gray elastotic appearance, particularly in bronchoscopic biopsies.
Carcinoid tumorlets (64,65,66,67), now referred to simply as tumorlets, and minute pulmonary meningothelial-like nodules (67,68,69) are nodular proliferations that are quite common. They may be mistaken for each other, other lesions, or even metastases. Tumorlets (Figure 18.23) represent well-circumscribed proliferations of neuroendocrine cells that usually occur around and within the walls of small airways, particularly in scarred or bronchiectatic airways, and in a small number of patients with airflow destruction. They lack mitotic figures and necrosis; and, while there is a superficial resemblance to small cell carcinoma, they are actually more similar to spindle cell carcinoid tumors. In frozen sections,
P.492
tumorlets may be mistaken for other lesions, particularly metastases. This confusion is compounded by the fact that sometimes tumorlets have cellular clusters at the periphery that retract from the surrounding stroma, simulating lymphatic or airspace invasion.
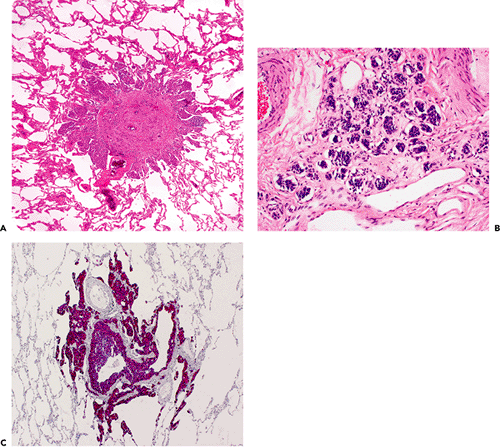 |
Figure 18.23 Tumorlet. A. Tumorlets are typically discrete nodules that are bronchiolocentric. The bronchiole is not apparent in A because it has been overrun by the proliferation, but the adjacent pulmonary artery is identifiable. B. Cytologically, tumorlets have a neuroendocrine appearance with nests of cells with granular chromatin and absence of necrosis and significant mitotic activity. The nests are often embedded in a fibrous stroma, and some of them may appear to float freely in airspaces or spaces that resemble lymphatics. C. Neuroendocrine markers, such as chromogranin, are strongly positive. |
Tumorlets are often multiple; some may become large enough to be recognized radiographically and to be removed to exclude carcinoma. Exactly where one draws the line between a carcinoid tumorlet and a carcinoid tumor, particularly in cases with multiple lesions, is quite arbitrary, although a cutoff point of 0.5 cm diameter or larger for carcinoid tumor is reasonable (41).
Diffuse intrapulmonary neuroendocrine cell hyperplasia (DIPNECH) is closely related to tumorlets, and the two often coexist (67). Arbitrarily, DIPNECH is defined as a proliferation of neuroendocrine cells limited to the bronchiolar epithelium. According to the World Health Organization (WHO), DIPNECH is defined as a generalized proliferation of scattered single cells, small nodules (neuroendocrine bodies), or linear proliferations of pulmonary neuroendocrine cells (PNCs) that may be confined to the bronchiole and bronchiolar epithelium, include local extraluminal proliferation in the form of tumorlets, or extend to the development of carcinoid tumors (67). When this proliferation expands and goes beyond the bounds of bronchioles, the designation of tumorlet is appropriate. Tumorlets are most frequently encountered as isolated or occasionally multiple nodules. When DIPNECH is encountered, usually multiple airways
P.493
are involved, and the patients may have evidence of airflow obstruction (67). Similarly, the presence of multiple tumorlets (and multiple carcinoid tumors) should prompt one to search for evidence of DIPNECH and to consider the possibility of clinical evidence of airflow obstruction (70).
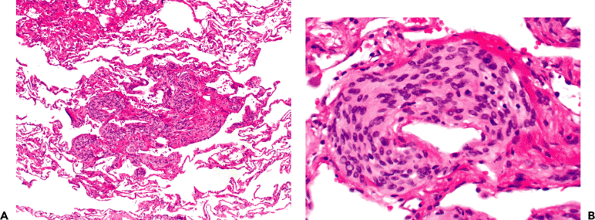 |
Figure 18.24 Minute pulmonary meningothelial-like nodule. These small parenchymal nodules are sometimes associated with pulmonary veins. A. They are associated with syncytial appearing cells in a collagenous stroma. B. Cytologically, they bear a distinct resemblance to meningothelial cells. |
Minute pulmonary meningothelial-like nodules (formerly called minute pulmonary chemodectomas) (Figure 18.24) were originally thought to represent an intrapulmonary proliferation of perivenular chemoreceptor cells (67,68,69). However, the accumulated evidence suggests that the cellular constituents are more closely related to meningothelial cells (67,68,69). A study by Ionescu et al. (71) showed that meningothelial-like nodules were almost uniformly positive for vimentin, about one-third stain for EMA, and they are negative with cytokeratin and synaptophysin by immunohistochemistry. In a genotypic study, some loss of heterozygosity was demonstrated by the same authors, and this was more common and affected more loci in cases of multiple meningothelial-like nodules in comparison to cases with solitary meningothelial-like nodules (71). The authors concluded that isolated meningothelial-like nodules were probably reactive, that cases of multiple meningothelial-like nodules might represent a transition from a reactive to a neoplastic proliferation, and that meningothelial-like nodules were different from meningiomas based on the major molecular events and their formation and progression (71). Minute pulmonary meningothelial-like nodules are composed of interstitial clusters of fusiform cells with pale eosinophilic cytoplasm, forming small stellate nodules (occasionally grossly appreciable) near small veins. Their location and characteristic bland cytology resembling meningothelial cells are very distinctive.
Atypical adenomatous hyperplasia (AAH) is a proliferative epithelial process occurring as small nodular lesions in the lung parenchyma (Figure 18.25). These were first recognized in resection specimens for carcinoma and named bronchioloalveolar cell adenomas (72). Miller found these lesions in 23 (10.74%) of 247 consecutive resection specimens for carcinoma (72). They are most easily recognized grossly in cases that are inflated with Bouin's fixative. The WHO defines AAH as a localized proliferation of mild to moderately atypical cells lining involved alveoli and, sometimes, respiratory bronchioles, resulting in focal lesions in peripheral alveolated lung, usually less than 5 mm in diameter and generally in the absence of underlying interstitial inflammation and fibrosis (67). The lesions are considered a precursor to some nonmucinous bronchioloalveolar carcinomas and mixed-type adenocarcinomas; and the evidence supporting this is epidemiologic, morphologic, morphometric, cytofluorometric, and genetic. Atypical adenomatous hyperplasia generally represents an incidental histologic finding (rare cases have been identified radiologically) and is found in 2 to 4% of routine autopsies of noncancer bearing patients and up to 35% of lobectomies for adenocarcinoma of the lung. In general, we advocate a conservative approach to these lesions, since: (a) there are many causes of type II cell proliferation, and many of them are reactive; (b) there are no studies documenting the pathologic progression of AAH to bronchioloalvoelar carcinoma (BAC) or studies that document how long such a progression takes); and (c) no studies exist showing what percent of lesions of AAH progress to carcinoma.
Focal scars (see following), healed granulomatous disease, and organized infarcts are among other incidental nodular lesions occasionally encountered in the lung. Early infarcts have a wedge shape with hemorrhagic necrosis, and the overlying pleura is viable with a fibrinous pleuritis. They often also have a rim of granulation tissue. Older infarcts are often rounded and have a rim of fibrous tissue; they can even be mistaken for healed granulomas. Necrotic tumor nodules sometimes mimic infarcts. Squamous metaplasia is
P.494
common in the airspaces adjacent to organizing infarcts and around bronchioles in organizing diffuse alveolar damage; it may be sufficiently exuberant to be mistaken for a neoplastic process. A single anthracosilicotic nodule is an occasional finding that may be accepted as incidental and insignificant if there is no clinical or radiologic evidence of pneumoconiosis. When silicotic nodules are multiple, the occupational history and the possibility of pneumoconiosis should be explored.
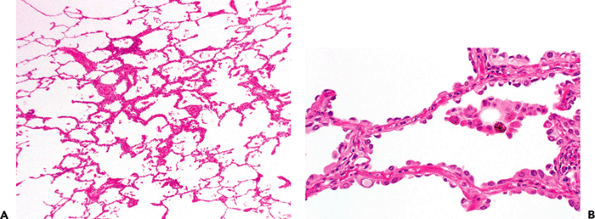 |
Figure 18.25 Atypical adenomatous hyperplasia (AAH). A. In this well-inflated specimen, a 2 to 3 mm diameter lesion is apparent as slight alveolar septal thickening and a proliferation of knob-shaped cells along alveolar walls. B. Cytologically, the cells lining the alveoli have mild to moderate atypia but lack the crowding and marked atypia associated with bronchioloalveolar carcinoma. Some nuclear inclusions characteristic of type II cells are also apparent (lower left). |
Although extensive parenchymal scarring is usually a pathological process, focal scars a few millimeters in diameter are a common incidental finding in biopsy material. One distinctive form of scar that is frequently observed (and may be identifiable as 2 3 mm centrilobular nodules in CT scans) consists of scars centered on alveolar ducts in the periphery of the lung, particularly common in (ex-) smokers (Figure 18.26). They are round or somewhat stellate in character and have numerous fascicles of smooth muscle and thus sometimes have been confused with primary muscle proliferations such as lymphangioleiomyomatosis. There are a number of histologic changes that accompany pulmonary scarring, regardless of cause. These include vascular intimal and medial thickening, sometimes to the point of luminal occlusion (endarteritis obliterans); smooth muscle and myofibroblastic hyperplasia in the interstitium; metaplasia and hyperplasia of type II cells or bronchiolar-type epithelium (peribronchiolar metaplasia); accumulations of intra-alveolar macrophages and mucostasis; carcinoid tumorlets (particularly along scarred small airways); dystrophic calcification or ossification; microscopic pericicatricial emphysema; and metaplastic adipose tissue in the pleural and peribronchial regions. The proliferation of type II cells associated with scars may be
P.495
confused with bronchioloalveolar carcinoma. Bronchioloalveolar carcinoma generally has uniform dense cellularity and cellular crowding with abrupt transition to normal alveolar walls, significant cytologic atypia, and lack of ciliated cells. Although AAH tends to have relatively little interstitial widening and fibrosis, AAH enters into the differential diagnosis in this situation.
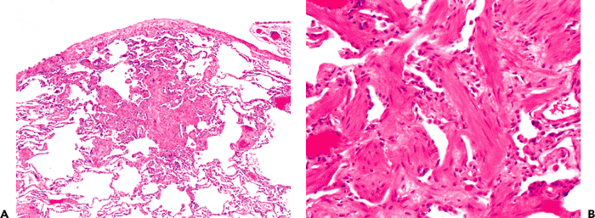 |
Figure 18.26 Incidental parenchymal scar. These are typically subpleural and appear to center on alveolar ducts (A) and commonly have fascicles of normal appearing smooth muscle (B). |
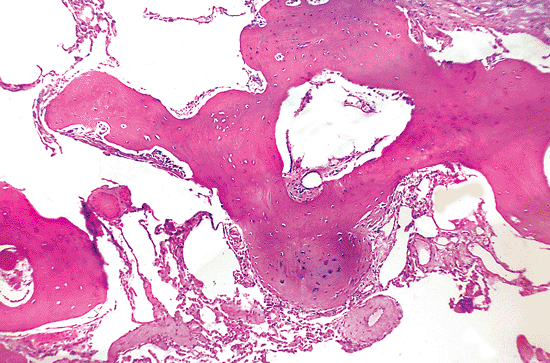 |
Figure 18.27 Ossification. An incidental focus of pulmonary ossification is noted; tissue is from a patient with usual interstitial pneumonia. |
In rare instances, mature bone is observed in normal alveoli; it may represent the residue of an organized airspace exudate from chronic passive congestion of mitral stenosis or an ancient organized pneumonia (73). Extensive dystrophic ossification (and rarely calcification) occasionally accompanies lesions with diffuse pulmonary fibrosis (Figure 18.27) (74). Focal dystrophic ossification is a common finding in focal lung scar, regardless of cause.
Intrapulmonary lymph nodes (Figure 18.28) are not uncommonly encountered in wedge biopsies. They vary from loosely organized microscopic foci of lymphoid tissue to fully developed lymph nodes identified grossly or radiologically.
Micronodular pneumocyte hyperplasia (MNPH) is defined by the WHO (1999) as a multifocal micronodular proliferation of type II cells with mild thickening of the interstitium (Figure 18.29) (67,75). This condition is rare and generally an incidental finding in a biopsy taken for another lesion (usually lymphangioleiomyomatosis). Rarely, MNPH is the sole lesion present, and the lesions may be sufficiently large to be identifiable on CT scans as multiple small nodules. The lesions are typically less than 5 mm in size. They can usually be distinguished from AAH and other causes of alveolar cell hyperplasia by their distinct rounded nodular character at scanning microscopy, the large plump eosinophilic type II cells lacking significant atypia, the slight interstitial collagen deposition within the lesions, and the presence of airspace histiocytes in the regions of the nodules.
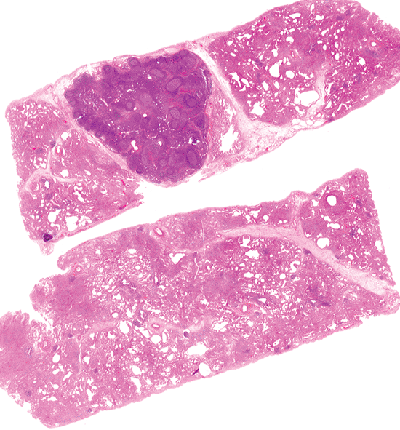 |
Figure 18.28 Intrapulmonary lymph node. There is a relatively large lymph node that appears to be within or adjacent to an interlobular septum in a wedge biopsy from peripheral lung. Reactive follicles are apparent even at scanning power microscopy. |
A number of intra-alveolar and intracellular structures are seen in the lung. Small numbers of intra-alveolar macrophages are a normal finding, and an increase in their numbers is nonspecific and a common reaction in smokers (43). Focal desquamative interstitial pneumonia-like reactions are seen in many pathologic conditions, especially in smokers and in conditions with fibrosis and architectural disorganization (46,76). When hemosiderin is present, causes for alveolar hemorrhage (both primary and secondary) should be excluded.
Corpora amylacea (77,78) are eosinophilic, rounded, slightly lamellated proteinaceous bodies (Figure 18.30) that stain positively with PAS stains and faintly with Congo red stain; they are more common in the lungs of older individuals. Sometimes there is a blue-gray, calcified, or polarizable crystalline particulate body in the center and a macrophage or giant cell response around them. The exact nature and cause of corpora are unclear, but they are of no clinical significance.
Blue bodies (Figure 18.31) are intra-alveolar, lamellated, basophilic, calcified structures found in airspaces associated with alveolar macrophages and giant cells (78). They are a nonspecific finding in a number of diffuse lung diseases related to accumulation of macrophages and are of no diagnostic significance. They are thought to be related to macrophage catabolism; they are composed primarily of calcium carbonate.
P.496
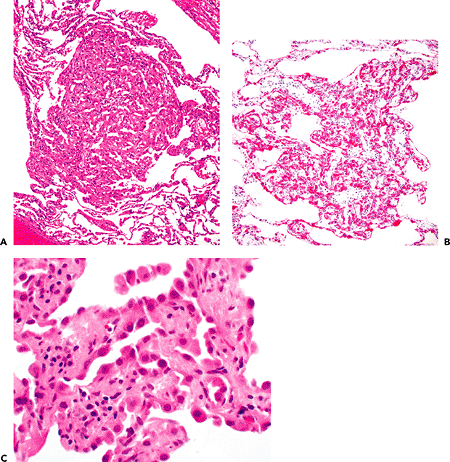 |
Figure 18.29 Micronodular pneumocyte hyperplasia (MNPH). A. There is a small central nodular region composed of plump type II cells. B. The size of the type II cells relative to the surrounding type I cells is apparent in cytokeratin staining. C. Cytologically, the type II cells have a fairly benign appearance. |
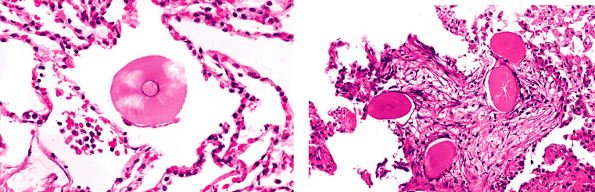 |
Figure 18.30 Pulmonary corpora amylacea. A. These show a central bluish nidus and a giant cell or macrophage reaction around them. Others may have radiating proteinaceous arrays and show cracking in histologic sections. They are often Congo red positive. B. Multiple corpora amylacea are seen as an incidental finding associated with organizing pneumonia. |
P.497
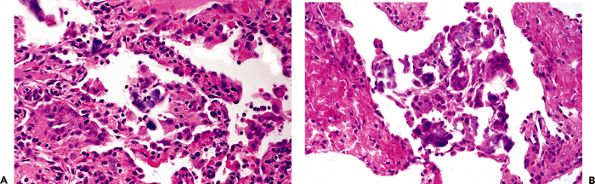 |
Figure 18.31 Blue bodies. A. and B. Blue bodies are gray or blue, intra-alveolar, calcified, lamellated structures often associated with clusters of macrophages, including giant cells. |
Schaumann bodies (Figure 18.32) are similar lamellated, calcified bodies seen in giant cells and associated with granulomas (78,79,80). They are also of no diagnostic significance and may be found in granulomas from diverse causes. Schaumann bodies are endogenously derived and may be mistaken for exogenous material since they may be partially birefringent, due to concomitant presence of oxalate crystals (see following).
Asteroid bodies (Figure 18.33) represent a starlike array of crystallized intracellular protein and are seen in giant cells of many granulomatous conditions; other than being aesthetically pleasing, they are nonspecific.
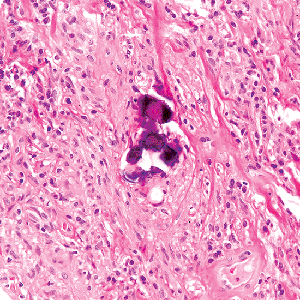 |
Figure 18.32 Schaumann body. There is a bluish calcified structure associated with surrounding granulomatous inflammation. A lamellated appearance similar to a psammoma body is focally apparent. The pale zones in this case represent loosened oxalate crystals that would be birefringent (see Figure 18.34). |
Calcium oxalate crystals are lucent, birefringent, platelike crystals, often in giant cells, that may be mistaken for exogenous material (78,79) (Figure 18.34). Accumulation of oxalate crystals around aspergillomas and in patients with forms of invasive aspergillosis is also common.
Material similar to Mallory's hyalin (Figure 18.35) may be found in the reactive type II cells of a number of interstitial diseases. It is distinctive, but nonspecific. It may stain for keratin and ubiquitin (81).
Ferruginous bodies, many of which are asbestos bodies, are indicative of significant inhalational exposure to the ferruginated material, but their presence does not necessarily correspond to clinically significant lung disease. Recent studies with electron-probe analysis have helped elucidate the variety of materials that may become iron encrusted, of which asbestos fibers comprise only a portion (82).
In virtually any urban adult, one may find short, needlelike, birefringent material (usually silica or silicates) in association with anthracotic pigment (Figure 18.36), either along lymphatic routes in the lung or in regional lymph nodes. Silicates are more brightly birefringent than silica.
P.498
The amount of birefringent material may be quite impressive in individuals (particularly smokers) who have no significant occupational exposure. This material should be distinguished from formalin pigment, as well as from surgical glove talc or starch; the latter are limited to handled surfaces and often demonstrate a Maltese-cross configuration with polarization. The diagnosis of silicosis (and silicatosis) is not based solely on the presence of birefringent material; it requires clinicopathologic correlations: appropriate chest radiograph findings, and parenchymal silicotic nodules or masses of histiocytes (within which early fibrotic nodules may be seen to form) along lymphatic routes. Precise characterization of any material identified requires special techniques such as electron-probe analysis.
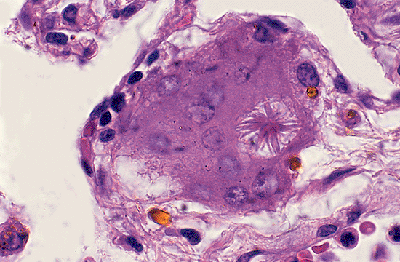 |
Figure 18.33 Asteroid body. An asteroid body in a giant cell is illustrated from a case of asbestosis. |
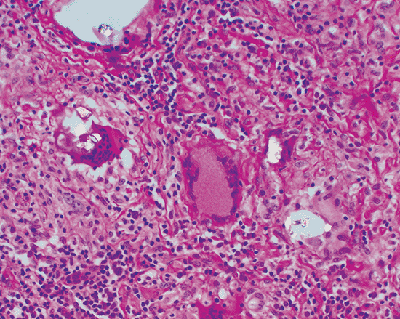 |
Figure 18.34 Calcium oxalate crystals. Calcium oxalate crystals are commonly associated with Schaumann bodies and granulomatous inflammation and show bright birefringence, as noted in this partially polarized photomicrograph. The association with giant cells is characteristic. |
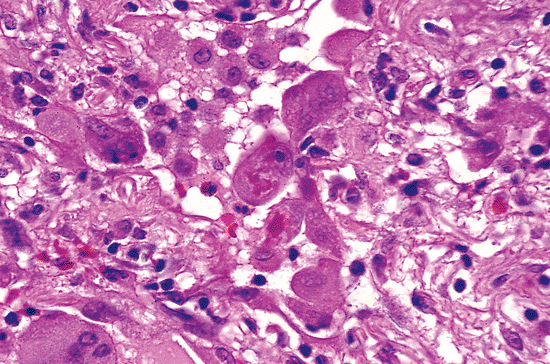 |
Figure 18.35 Hyalin. Material resembling Mallory's hyalin may be seen in reactive type II cells in a number of acute and chronic conditions. |
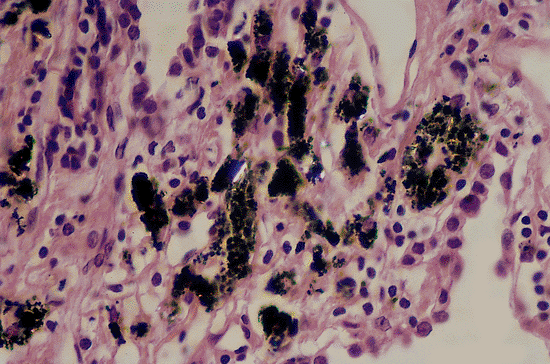 |
Figure 18.36 Silica and silicates in normal lung. Birefringent silica and silicate particles are a common nonspecific finding in and around the anthracotic pigment that is so commonly seen in urban adults and smokers. |
An occasional nonnecrotizing epithelioid granuloma may be found in the lungs of patients who have no evidence of granulomatous disease; they are analogous to the occasional granuloma seen at many sites in the body. Likewise, cholesterol granulomas or single giant cells containing cholesterol clefts may also be an occasional incidental finding (Figure 18.37). In some instances, the presence of cholesterol granulomas has been linked to prior alveolar hemorrhage, mucostasis, or pulmonary hypertension (83), but usually no significance can be ascribed to them. Lipogranulomas are an occasional nonspecific finding, said to be more common in diabetics (84).
An interesting finding is the presence of scattered megakaryocytes in alveolar walls (Figure 18.38), predominantly within alveolar capillaries. Large numbers can be seen, particularly during sepsis. They are of no diagnostic significance but should not be overinterpreted as malignant or virally infected cells. Along with the bone marrow and spleen, the lung acts as a major reservoir for megakaryocytes.
Bone marrow emboli, common in autopsy material, are also seen in biopsy specimens (Figure 18.39). Rarely correlated with any clinically significant process, they may be a consequence of bony trauma or excision of ribs. In some cases, however, such as thoracoscopic biopsies, bony
P.499
trauma cannot be implicated, and the marrow emboli are an unexplained incidental finding.
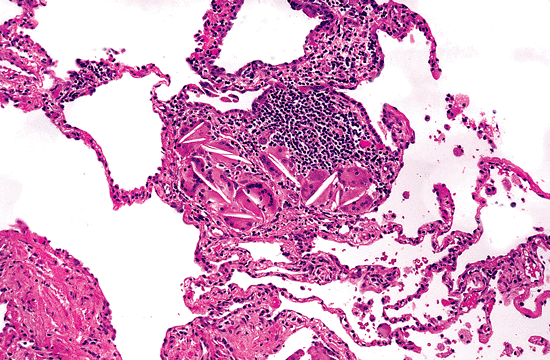 |
Figure 18.37 Cholesterol granulomas. Granulomas and clusters of giant cells containing cholesterol clefts are a frequent nonspecific finding in interstitial lung disease. |
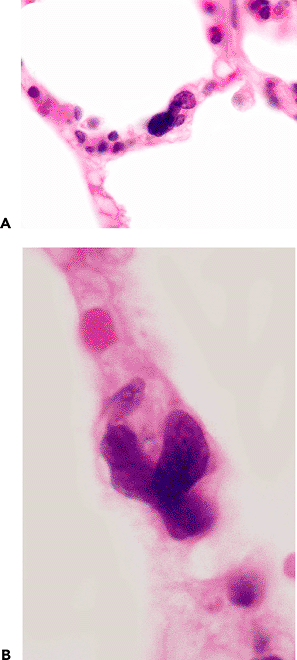 |
Figure 18.38 Megakaryocytes. A. and B. Megakaryocytes are a common finding in normal lung, often present within alveolar capillaries. |
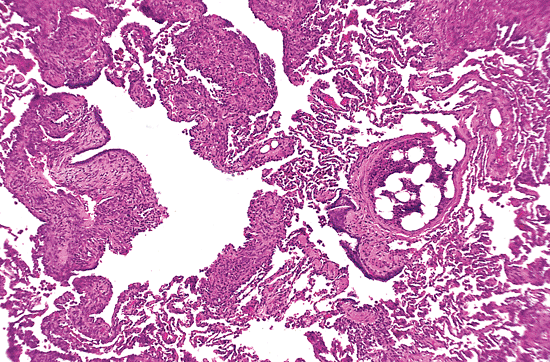 |
Figure 18.39 Bone marrow embolus. An incidental bone marrow embolus (right center) identified in a pulmonary artery in a biopsy from a patient with lymphangioleiomyomatosis (left). |
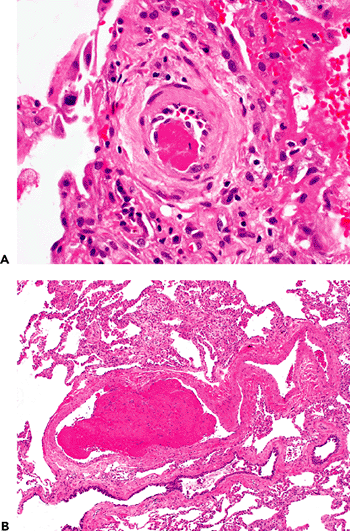 |
Figure 18.40 Thromboemboli in acute lung injury. Small (A) or even somewhat large (B) fibrin thrombi are commonly present in biopsies showing extensive acute lung injury (A, B). Hyalin membranes are apparent adjacent to the artery in A, and organization is apparent at the top of the field in B. |
Recent intravascular thrombi (probably formed in situ) are a relatively common accompanying finding in any severe acute inflammatory lung disease (Figure 18.40), and they should not be considered evidence of pulmonary emboli without corroborating clinical information.
In patients with chronic hemorrhagic, chronic pulmonary congestion, or metabolic abnormalities, calcification and iron encrustation of the pulmonary elastic tissue may occur and even elicit a giant cell reaction (Figure 18.41). This phenomenon has been inappropriately labeled endogenous pneumoconiosis (85).
Intravascular foreign material is usually birefringent and may have a giant cell reaction. While it usually occurs in intravenous drug abuse (IV talcosis), occasional fragments of
P.500
foreign material are seen in patients without a history of drug abuse, perhaps related to intravenous lines during hospitalization or surgery.
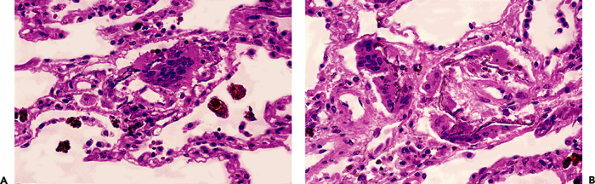 |
Figure 18.41 Iron deposition on elastic tissue (A, B). Chronic hemorrhage, in this case due to severe chronic passive congestion, may result in encrustation of interstitial and vascular elastic fibers by hemosiderin. A giant cell reaction is a frequent accompanying finding (A). |
The finding of interstitial air, either localized or diffuse, is well known to pediatric lung pathologists and may be an incidental finding in adults who have been on ventilators. Interstitial air can occur as an incidental finding in a region of subpleural fibrosis but also be a significant pathologic finding in its own right (86) (Figure 18.42). The abnormal air-filled spaces resemble honeycombing; however, they lack the expected metaplastic epithelial lining and careful inspection shows either a complete lack of a cellular lining or a histiocytic and giant cell reaction lining the spaces. Interstitial air presenting in this way may be encountered in patients with interstitial lung disease, in patients with a history of pneumothorax, and in and around bullae and blebs. As in ventilated children, interstitial air may be encountered in adults on assisted ventilation and sometimes may be completely missed, being interpreted as tissue tearing.
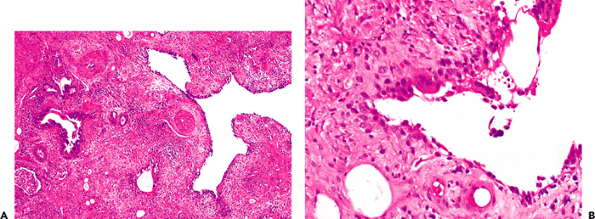 |
Figure 18.42 Incidental interstitial air. Peculiarly shaped airspaces (A) lined by giant cells (B) represent interstitial air, which is an occasional incidental finding in patients with interstitial lung disease, including those who have been on positive-pressure ventilation. The case illustrated in A represents an example of nonspecific interstitial pneumonia. |
Hilar and peribronchial lymph nodes are rarely carefully examined beyond the evaluation for metastatic carcinoma. Nevertheless, they frequently exhibit a number of characteristic changes. Clusters of dust-filled macrophages in the sinuses and paracortical areas may resemble small granulomas (Figure 18.43). Silicotic or anthracosilicotic nodules, old healed infectious granulomas, and sinus histiocytosis are also common. When sarcoidosis is a consideration, one may have difficulty distinguishing the normal histiocytosis of hilar nodes from the granulomas of sarcoidosis. Generally, this problem can be approached by maintaining a high threshold for granulomas, requiring well-formed, rounded granulomatous masses (particularly with accumulations of
P.501
intergranulomatous fibrinoid or hyalinized material), giant cells, and careful clinicopathologic correlation.
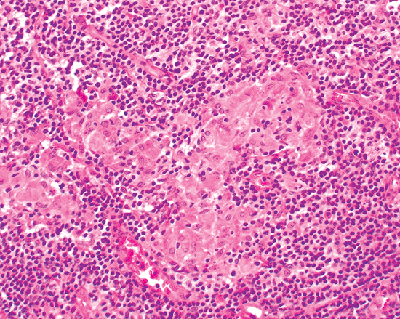 |
Figure 18.43 Histiocyte clusters in hilar nodes. Normal hilar and mediastinal lymph nodes commonly have clusters of histiocytes with variable amounts of dust particles in them. Sometimes they may appear somewhat granulomatous and may be difficult to distinguish from a true granulomatous reaction. |
Although multiple intraparenchymal silicotic nodules should make one suspect the possibility of silicosis, anthracosilicotic nodules in hilar nodes are quite common in the absence of silicosis. The nodules are composed of concentric whorled and layered hyalinized collagen and are usually surrounded by a nonpalisaded rim of dust-filled macrophages that contain birefringent material when examined with polarized light. Central degenerative changes may be evident. Silicotic nodules should be distinguished from old/healed granulomatous disease.
Hamazaki-Wesenberg bodies (87) are small (average: 5 m in length), yellow-brown intracellular or extracellular structures associated with sinus histiocytes in lymph nodes, especially lung hilar nodes (Figure 18.44). The cause of these bodies is unknown, but they resemble lipofuscin. Positive staining with methenamine silver and PAS stains may lead to their confusion with yeast forms, but their H&E appearance, lack of associated necrosis or inflammation, and positive reaction with Fontana-Masson stain should facilitate their recognition and distinction from fungi.
Changes in the pleura in wedge biopsies may be incidental or part of the underlying pathologic process, and their significance needs to be assessed on an individual case basis.
Incidental Findings in Transbronchial Biopsies
The major artifacts or incidental findings in transbronchial biopsies that lead to misinterpretation include atelectasis (misinterpreted as interstitial pneumonia), bubble artifact (misinterpreted as lipoid pneumonia), and portions of the pleura (either entirely missed or misinterpreted as neoplastic or suspicious for being neoplastic) (54,55). Such portions of pleura may even include some pleural fat, a particularly common finding in fibrosing interstitial pneumonias. The finding of pleural tissue in transbronchial biopsies (Figure 18.45) is not uncommon but also is not well recognized among most pathologists. Strips of reactive mesothelial cells in such specimens may be confused with carcinoma.
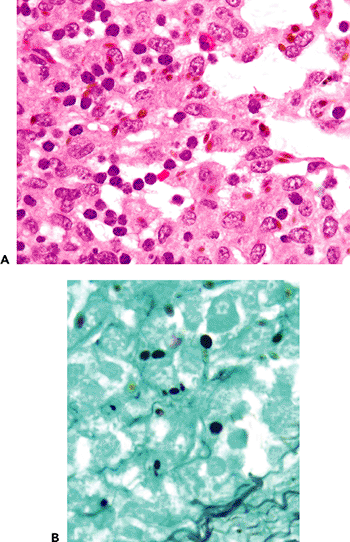 |
Figure 18.44 Hamazaki-Wesenberg bodies. A. Hamazaki-Wesenberg bodies represent yellow-brown oval structures associated with histiocytes, occasionally encountered in hilar and mediastinal lymph nodes. B. Their positive staining with silver stains sometimes leads to confusion with fungi. |
As in wedge biopsies, the most common cause of red blood cells in airspaces in transbronchial biopsies is trauma related to the procedure of the biopsy rather than an alveolar hemorrhage syndrome.
P.502
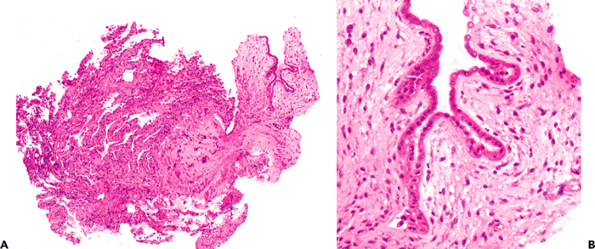 |
Figure 18.45 Visceral pleura in transbronchial biopsies. Transbronchial biopsies directed toward the periphery of the lung may not uncommonly sample portions of the visceral pleura (A, right; and B). These may lead to misdiagnosis, particularly if there is an associated reactive pleuritis. |
Effects of Aging
Some of the effects of aging on the lung are shown in Table 18.9. Calcification and ossification of cartilages in the large airways may be seen. Intimal thickening is an age-related change in pulmonary arteries and veins (Figure 18.46), and apical pulmonary arteries are affected more often (2). Intimal thickening of veins is often hyalin and sclerotic in character, and should be distinguished from the more cellular intimal and muscular proliferation seen in pulmonary veno-occlusive disease and chronic passive congestion. Mural hyalinization of small arterioles is also an aging change (as well as being common in emphysematous lungs). Some degree of alveolar enlargement accompanies aging (88) and is called senile emphysema. Longitudinal elastic tissue fibers are a normal finding immediately beneath the surface epithelium in the large airways. These may take on an elastotic appearance in the lungs of older individuals. Senile amyloid is usually an incidental finding, is perivascular, and increases in incidence with age (89).
Table 18.9 Effects of Aging seen in Lung Biopsies | |
|---|---|
|
The Biopsy that Looks Normal at First Glance
When a lung biopsy for presumed diffuse lung disease initially appears normal, a number of possibilities should be considered (Table 18.10), especially pulmonary edema (Figure 18.47).
Although one can argue that interstitial infiltrates that are so subtle as to be overlooked are probably not clinically significant, their recognition is necessary. This situation can arise, for example, with biopsy material from the less severely affected portions of lungs in patients with interstitial pneumonias, in cases in which the inflammatory infiltrate has
P.503
been suppressed by steroids or immunosuppressive therapy, and in pathologic entities characterized by patchy inflammation. The presence of inflammatory cells in the perivenular regions and alveolar wall with reactive type II cells is usually indicative of an interstitial pneumonia.
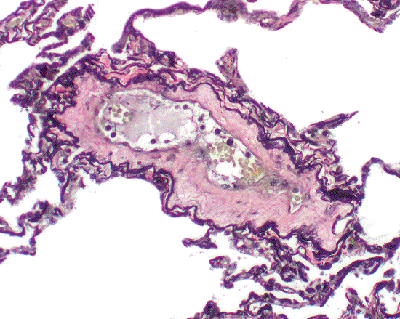 |
Figure 18.46 Aging change in vessels. In elderly individuals, a hyalin thickening of the intima may be encountered as an incidental finding. Clinical correlation is suggested, since this feature may also be encountered in the setting of pulmonary hypertension. |
Table 18.10 Situations in Which a Lung Biopsy May Appear Normal | |
|---|---|
|
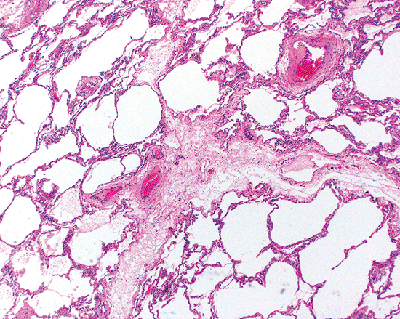 |
Figure 18.47 Pulmonary edema. Pulmonary edema may produce a deceptively normal appearance in a biopsy. Attention to septal widening, as noted in the accompanying text, and faint flocculent material in airspaces may be a clue to the diagnosis. |
References
1. Nagaishi C. Functional Anatomy and Histology of the Lung. Baltimore: University Park Press; 1972.
2. Wagenvoort CA, Wagenvoort N. Pathology of Pulmonary Hypertension. New York: John Wiley; 1977.
3. Kuhn C III. Ultrastructure and cellular function in the distal lung. In: Thurlbeck WM, Abell MR, eds. The Lung. Baltimore: Williams & Wilkins; 1978.
4. Scadding JG, Cumming G, eds. Scientific Foundations of Respiratory Medicine. Philadelphia: WB Saunders; 1981.
5. Gail DB, Lenfant CJ. Cells of the lung: biology and clinical implications. Am Rev Respir Dis 1983;127:366 387.
6. Bienenstock J, Befus AD. Gut-and-bronchus-associated lymphoid tissue. Am J Anat 1984;170:437 445.
7. Langston C, Kida K, Reed M, Thurlbeck WM. Human lung growth in late gestation and in the neonate. Am Rev Respir Dis 1984;129:607 613.
8. Murray JF. The Normal Lung. 2nd ed. Philadelphia: WB Saunders; 1986.
9. Fawcett DW. Bloom and Fawcett: A Textbook of Histology. 12th ed. New York: Chapman and Hall; 1994.
10. Coalson JJ. The adult lung: structure and function. In: Saldana MJ, ed. Pathology of Pulmonary Disease. Philadelphia: JB Lippincott: 1994:3 14.
11. Wang NS. Anatomy. In: Dail DH, Hammar SP, eds. Pulmonary Pathology. 2nd ed. New York: Springer-Berlag; 1994:21 44.
12. Kuhn C III. Normal anatomy and histology. In: Thurlbeck WM, Churg AM, eds. Pathology of the Lung. 2nd ed. New York: Thieme Medical Publishers; 1995:1 36.
13. Weibel ER, Taylor CR. Functional design of the human lung for gas exchange. In: Fishman AP, ed. Pulmonary Diseases and Disorders. Vol 1. 3rd ed. New York: McGraw-Hill; 1998:21 61.
14. Albertine KH, Williams MC, Hyde DM. Anatomy of the lungs. In: Murray JF, Nadel JA, eds. Textbook of Respiratory Medicine. 3rd ed. Philadelphia: WB Saunders; 2000:3 33.
15. Corrin B. Pathology of the Lungs. London: Churchill Livingstone; 2000.
16. Leslie KO, Wick MR. Lung anatomy. In: Leslie KO, Wick MR, eds. Practical Pulmonary Pathology. Philadelphia: Churchill Livingstone; 2005:1 18.
17. Chinoy MR. Lung growth and development. frontiers. Bioscience 2003;8:392 415.
18. Roth-Kleiner M, Post M. Genetic control of lung development. Biol Neonate 2003;84:83 88.
19. Yaegashi H, Takahashi T. The airway dimension in ordinary human lung. A standardized morphometry of lung sections. Arch Pathol Lab Med 1994;118:969 974.
20. Lambert MW. Accessory bronchiolealveolar communications. J Pathol Bacteriol 1955;70:311 314.21.
21. Yousem SA, Dacic S. Idiopathic bronchiolocentric interstitial pneumonia. Mod Pathol 2002;15:1148 1153.
22. Churg A, Meyers J, Suarez T, et al. Airway-centered interstitial fibrosis: a distinct form of aggressive diffuse lung disease. Am J Surg Pathol 2004;28:62 68.
23. Fukuoka J, Franks TJ, Colby TV, et al. Peribronchiolar metaplasia: a common histologic lesion in diffuse lung disease and a rare cause of interstitial lung disease; clinicopathologic features of 15 cases. Mod Pathol 2003;17:336a.
24. Colby TV, Koss MN, Travis WD. Tumors of the lower respiratory tract. In: Rosai J, ed. Atlas of Tumor Pathology. 3rd series, fascicle 13. Washington DC: Armed Forced Institute of Pathology; 1995:465 471.
25. Rogers AV, Dewar A, Corrin B, Jeffery PK. Identification of serous-like cells in the surface epithelium of human bronchioles. Eur Respir J 1995;6:498 504.
26. Castranova V, Rabovsky J, Tucker JH, Miles PR. The alveolar type II epithelial cell: a multifunctional pneumocyte. Toxicol Appl Pharmacol 1988;93:472 483.
27. Kasper M, Reimann T, Hempel U, et al. Loss of caveolin expression in type I pneumocyte as an indicator of subcellular alterations during lung fibrogenesis. Histochem Cell Biol 1998;109:41 48.
28. Lou YP, Takeyama K, Grattan KM, et al. Platelet-activating factor induces goblet cell hyperplasia and mucin gene expression in airways. Am J Respir Crit Care Med 1998; 157(pt 1):1927 1934.
29. Kreda SM, Gynn MC, Fenstermacher DA, Boucher RC, Gabriel SE. Expression and localization of epithelial aquaporins in the adult human lung. Am J Respir Cell Mol Biol 2001;24:224 234.
30. Ryerse JS, Hoffmann JW, Mahmoud S, Nagel BA, deMello DE. Immunolocalization of CC10 in Clara cells in mouse and human lung. Histochem Cell Biol 2001;115:325 332.
31. Lehnert BE. Pulmonary and thoracic macrophage subpopulations and clearance of particles from the lung. Environ Health Perspect 1992;97:17 46.
32. Gould SJ, Isaacson PG. Bronchus-associated lymphoid tissue (BALT) in human fetal and infant lung. J Pathol 1993;169:229 234.
P.504
33. Richmond J, Pritchard GE, Ashcroft T, Avery A, Corris PA, Walters EH. Bronchus associated lymphoid tissue (BALT) in human lung: its distribution in smokers and non-smokers. Thorax 1993;48:1130 1134.
34. Tschernig T, Kleemann WJ, Pabst R. Bronchus-associated lymphoid tissue (BALT) in the lungs of children who had died from sudden infant death syndrome and other causes. Thorax 1995;50:658 660.
35. Bienenstock J. Bronchus-associated lymphoid tissue. Int Arch Allergy Appl Immunol 1985;76(suppl 1):62 69.
36. Kradin RL, Spirn PW, Mark EJ. Intrapulmonary lymph nodes. Clinical, radiologic, and pathologic features. Chest 1985;87:662 667.
37. Gallagher B, Urbanski SJ. The significance of pleural elastica invasion by lung carcinomas. Hum Pathol 1990;21:512 517.
38. Colby TV, Swensen SJ. Anatomic distribution and histopathologic pattern in diffuse lung disease: correlation with HRCT. J Thorac Imaging 1996;11:1 26.
39. Newman SL, Michael RP, Wang NS. Lingular lung biopsy: is it representative? Am Rev Respir Dis 1985;132:1084 1086.
40. Albo RJ, Grimes OF. The middle lobe syndrome: a clinical study. Dis Chest 1966;50:509 518.
41. Kwon KY, Myers JL, Swensen SJ, Colby TV. Middle lobe syndrome: a clinicopathological study of 21 patients. Hum Pathol 1995;26:302 307.
42. Renner RR, Markarian B, Pernice NJ, Heitzman ER. The apical cap. Radiology 1974;110:569 573.
43. McCloud TC, Isler RJ, Novelline RA, Putman CE, Simeone J, Stark P. The apical cap. AJR 1981;137:299 306.
44. Yousem SA. Pulmonary apical cap: a distinctive but poorly recognized lesion in pulmonary surgical pathology. Am J Surg Pathol 2001;25:679 683.
45. Thurlbeck WM, Wright JL. Thurlbeck's Chronic Airflow Obstruction. 2nd ed. Hamilton, Ontario: BC Decker; 1999.
46. Fraig M, Shreesa U, Savici D, Katzenstein AL. Respiratory bronchiolitis: a clinicopathologic study in current smokers, ex-smokers and never-smokers. Am J Surg Pathol 2002;26:647 653.
47. Lichter I, Gwynne JF. Spontaneous pneumothorax in young subjects: a clinical and pathological study. Thorax 1971;26:409 417.
48. Thurlbeck WM. Chronic Airflow Obstruction in Lung Disease. Philadelphia: WB Saunders; 1976.
49. Askin FB, McCann BG, Kuhn C. Reactive eosinophilic pleuritis: a lesion to be distinguished from pulmonary eosinophilic granuloma. Arch Pathol Lab Med. 1977;101:187 191.
50. Churg A. Asbestos fibers and pleural plaques in a general autopsy population. Am J Pathol 1982;109:88 96.
51. Meurman L. Asbestos bodies and pleural plaques in a Finnish series of autopsy cases. Acta Pathol Microbiol Immunol Scand 1966;181:1 107.
52. Roberts GH. The pathology of parietal pleural plaques. J Clin Pathol 1971;24:348 353.
53. Hillerdal G. Pleural plaques and risk for bronchial carcinoma and mesothelioma. A prospective study. Chest 1994;105:144 150.
54. Katzenstein ALA. Katzenstein and Askin's Surgical Pathology of Non-neoplastic Lung Disease. 3rd ed. Philadelphia: WB Saunders; 1997.
55. Kendall DM, Gal AA. Interpretation of tissue artifacts in transbronchial lung biopsy specimens. Ann Diagn Pathol 2003;7:20 24.
56. Kadokura M, Colby TV, Myers JL, et al. Pathologic comparison of video-assisted thoracic surgical lung biopsy with traditional open lung biopsy. J Thorac Cardiovasc Surg 1995;109:494 498.
57. Churg A. An inflation procedure for open lung biopsies. Am J Surg Pathol 1983;7:69 71.
58. Bensard DD, McIntyre RC Jr, Waring BJ, Simon JS. Comparison of video thoracoscopic lung biopsy to open lung biopsy in the diagnosis of interstitial lung disease. Chest 1993; 103:765 770.
59. Ferson PF, Landreneau RJ, Dowling RD, et al. Comparison of open versus thoracoscopic lung biopsy for diffuse infiltrative pulmonary disease. J Thorac Cardiovasc Surg 1993;106:194 199.
60. Carnochan FM, Walker WS, Cameron EW. Efficacy of video assisted thoracoscopic lung biopsy: an historical comparison with open lung biopsy. Thorax 1994;49:361 363.
61. Niewoehner DE, Kleinerman J, Rice DB. Pathologic changes in the peripheral airways of young cigarette smokers. N Engl J Med 1974;291:755 758.
62. Myers JL, Veal CF Jr, Shin MS, Katzenstein AL. Respiratory bronchiolitis causing interstitial lung disease. A clinicopathologic study of six cases. Am Rev Respir Dis 1987;135:880 884.
63. Ashley DJ. Bony metaplasia in trachea and bronchi. J Pathol 1970;102:186 188.
64. Churg A, Warnock ML. Pulmonary tumorlet. A form of peripheral carcinoid. Cancer 1976;37:1469 1477.
65. Gould VE, Linnoila RI, Memoli VA, Warren WH. Neuroendocrine components of the bronchopulmonary tract: hyperplasias, dysplasias, and neoplasms. Lab Invest 1983;49:519 537.
66. Ranchod M. The histogenesis and development of pulmonary tumorlets. Cancer 1977;39:1135 1145.
67. Travis W, Brambilla E, Harris C, Muller-Hermelink K, eds. World Health Organization Classification of Tumours. Vol 5. Pathology and Genetics: Tumors of the Lung, Pleura, Thymus and Heart. Lyon, France: IARC Press; 2004.
68. Kuhn C III, Askin FB. The fine structure of so-called minute pulmonary chemodectomas. Hum Pathol 1975;6:681 691.
69. Gaffey MJ, Mills SE, Askin FB. Minute pulmonary meningothelial-like nodules. A clinicopathologic study of so-called minute pulmonary chemodectoma. Am J Surg Pathol 1988;12:167 175.
70. Miller RR, Muller NL. Neuroendocrine cell hyperplasia and obliterative bronchiolitis in patients with peripheral carcinoid tumors. Am J Surg Pathol 1995;19:653 658.
71. Ionescu DN, Sasatomi E, Aldeeb D, et al. Pulmonary meningothelial-like nodules: a genotypic comparison with meningiomas. Am J Surg Pathol 2004;28:207 214.
72. Miller RR. Bronchioloalveolar cell adenomas. Am J Surg Pathol 1990;14:904 912.
73. Elkeles A, Glynn LE. Disseminated parenchymatous ossification in the lungs in association with mitral stenosis. J Pathol Bacteriol 1946;58:517 522.
74. Green JD, Harle TS, Greenberg SD, Weg JG, Nevin H, Jenkins DE. Disseminated pulmonary ossification. A case report with demonstration of electron-microscopic features. Am Rev Respir Dis 1970;101:293 298.
75. Muir TE, Leslie KO, Popper H, et al. Micronodular pneumocyte hyperplasia. Am J Surg Pathol 1998;22:465 472.
76. Bedrossian CW, Kuhn C III, Luna MA, Conklin RH, Byrd RB, Kaplan PD. Desquamative interstitial pneumonia-like reaction accompanying pulmonary lesions. Chest 1977;72:166 169.
77. Hollander DH, Hutchins GM. Central spherules in pulmonary corpora amylacea. Arch Pathol Lab Med 1978;102:629 630.
78. Koss MN, Johnson FB, Hochholzer L. Pulmonary blue bodies. Hum Pathol 1981;12:258 266.
79. Visscher D, Churg A, Katzenstein AL. Significance of crystalline inclusions in lung granulomas. Mod Pathol 1988;1:415 419.
80. Schaumann J. On the nature of certain peculiar corpuscles present in the tissue of lymphogranulomatosis benigna. Acta Med Scand 1941;106:239 253.
81. Warnock ML, Press M, Churg A. Further observations on cytoplasmic hyaline in the lung. Hum Pathol 1980;11:59 65.
82. Churg A, Warnock ML. Asbestos and other ferruginous bodies: their formation and clinical significance. Am J Pathol 1981;102:447 456.
83. Glancy DL, Frazier PD, Roberts WC. Pulmonary parenchymal cholesterol-ester granulomas in patients with pulmonary hypertension. Am J Med 1968;45:198 210.
84. Reinila A. Perivascular xanthogranulomatosis in the lungs of diabetic patients. Arch Pathol Lab Med 1976;100:542 543.
85. Walford RL, Kaplan L. Pulmonary fibrosis and giant-cell reaction with altered elastic tissue: endogenous pneumoconiosis. AMA Arch Pathol 1957;63:75 90.
86. Unger JM, England DM, Bogust GA. Interstitial emphysema in adults: recognition and prognostic implications. J Thorac Imaging 1989;4:86 94.
87. Ro JY, Luna MA, Mackay B, Ramos O. Yellow-brown (Hamazaki-Wesenberg) bodies mimicking fungal yeasts. Arch Pathol Lab Med 1987;111:555 559.
88. Gillooly M, Lamb D. Airspace size in lungs of lifelong non-smokers: effect of age and sex. Thorax 1993;48:39 43.
89. Kunze WP. Senile pulmonary amyloidosis. Pathol Res Pract 1979;164:413 422.