35 - Infectious Diseases: Protozoal Helminthic
Editors: McPhee, Stephen J.; Papadakis, Maxine A.; Tierney, Lawrence M.
Title: Current Medical Diagnosis & Treatment, 46th Edition
Copyright 2007 McGraw-Hill
> Table of Contents > 38 - Disorders Due to Physical Agents
function show_scrollbar() {}
38
Disorders Due to Physical Agents
Richard Cohen MD, MPH
Brent R.W. Moelleken MD, FACS
Cold & Heat
Cold tolerance varies considerably among individuals. Factors that increase the likelihood of injury from exposure to cold include poor general physical conditioning, nonacclimatization, advanced age, altered mental status, systemic illness, poor tissue oxygenation, wet or insufficient clothing, previous cold weather injury, smoking, and the use of alcohol or other sedative drugs. High wind velocity ( windchill factor ) increases the severity of cold injury at low temperatures.
Cold Urticaria
Some persons have a familial or acquired hypersensitivity to cold, and urticaria may develop upon even limited exposure to a cold (eg, wind, freezer compartments). The urticaria usually occurs only on exposed areas, but in markedly sensitive individuals the response can be generalized and fatal. Immersion in cold water may result in severe systemic reactions from histamine release, including shock. Familial cold urticaria is an autosomal dominant inflammatory disorder, manifested as a burning sensation of the skin occurring about 30 minutes after exposure to cold. Acquired cold urticaria may be associated with medication (eg, griseofulvin) or with infection. Cold urticaria may occur secondarily to cryoglobulinemia or as a complication of syphilis. Most cases of acquired cold urticaria are idiopathic. For diagnosis, an ice cube is usually applied to the skin of the forearm for 4 5 minutes, then removed, and the area is observed for 10 minutes. As the skin rewarms, an urticarial wheal appears at the site and may be accompanied by itching. Peltier effect-based temperature testing has also been recommended for diagnostic testing. It involves the use of thermoelectric elements to quantitatively measure skin temperature thresholds. Cyproheptadine, 16 32 mg/d orally in divided doses, is the drug of choice. Desloratadine, 5 mg/d orally, has been shown to reduce or prevent symptoms.
Raynaud's Phenomenon
See Chapter 12.
Accidental Systemic Hypothermia
![]() Essentials of Diagnosis
Essentials of Diagnosis
A reduction of core body temperature below 35 C.
An esophageal or rectal probe that measures as low as 25 C is required; oral temperatures are inaccurate.
General Considerations
Systemic hypothermia may result from exposure (atmospheric or immersion) to prolonged or extreme cold. The condition may arise in healthy individuals in the course of occupational or recreational exposure or in victims of accidents.
Systemic hypothermia may follow exposure to cool but not cold temperatures when there is altered homeostasis due to debility or disease. In colder climates, elderly and inactive individuals living in inadequately heated housing are particularly susceptible. Patients with cardiovascular or cerebrovascular disease, mental retardation, acute alcoholism, malnutrition, myxedema, hypopituitarism, or use of sedating or tranquilizing drugs are more vulnerable to accidental hypothermia. Prolonged postoperative hypothermia or administration of large amounts of refrigerated stored blood (without rewarming) can cause systemic hypothermia.
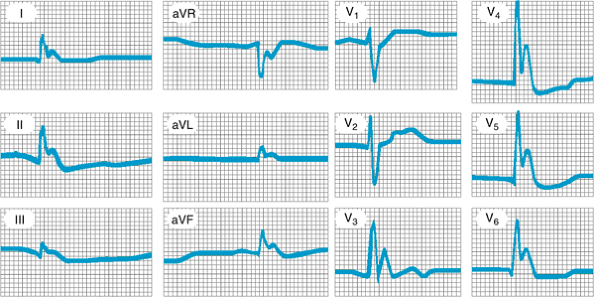 |
Figure 38-1. Hypothermia. The ventricular rate is 50/min. Atrial activity is not seen. The QRS complexes are narrow and are deformed at their terminal portions by a slurred wave occurring prior to the inscription of the ST-T waves; this is the J wave of Osborn. The QT interval is prolonged. (Courtesy of R Brindis. Reproduced, with permission from Goldschlager N, Goldman MJ: Principles of Clinical Electrocardiography, 13th ed. McGraw-Hill, 1989. ) |
Systemic hypothermia causes reduced physiologic function with decreased oxygen consumption and slowed myocardial repolarization, peripheral nerve conduction, gastrointestinal motility, and respirations as well as hemoconcentration and pancreatitis. Defenses against cold exposure are superficial blood vessel constriction and increased metabolic heat production.
Clinical Findings
Early manifestations of hypothermia include weakness, drowsiness, lethargy, irritability, confusion, shivering, and impaired coordination. A lowered body temperature may be the sole finding; the skin may appear
P.1622
blue or puffy. With extended exposure, apathy, impaired judgment, and ataxia appear.
The internal (core) body temperature in accidental hypothermia may range from 25 C to 35 C. At core temperatures below 35 C, the patient may become delirious, drowsy, or comatose and may stop breathing. The pulse and blood pressure may be unobtainable, leading clinicians to believe the patient is dead. Metabolic acidosis, hyperkalemia, pneumonia, pancreatitis, ventricular fibrillation, hypoglycemia or hyperglycemia, coagulopathy, and renal failure may occur. Atrial and ventricular dysrhythmias, prolongation of the PR, QRS, and QT intervals, and the J wave of Osborn (positive deflection in the terminal portion of the QRS complex, most notable in leads II, V5, and V6) are directly related to the lowering of core temperature; cardiac arrhythmias may occur, especially during the rewarming process (Figure 38-1). Death in systemic hypothermia usually results from cardiac asystole or ventricular fibrillation.
Treatment
Aggressiveness of therapy should increase with the severity of hypothermia; a combination of external and core rewarming methods may be necessary. Patients with mild hypothermia (rectal temperature > 33 C) who have been otherwise healthy usually respond well to a warm bed or to rapid passive rewarming with a warm bath or warm packs and blankets. A conservative approach is usually used in treating elderly or debilitated patients, using an electric blanket kept at 37 C. Gentle handling and movement of the patient are essential to avoid triggering arrhythmias.
Patients with moderate or severe hypothermia (core temperatures of < 33 C) do not have the thermoregulatory shivering mechanism and require active rewarming with supportive care. Cardiovascular support, acid-base balance, arterial oxygenation, and adequate intravascular volume should be established prior to rewarming to minimize the risk of organ infarction and afterdrop (recurrent hypothermia). If CPR is initiated, it should be continued if the patient has not regained vital signs until the patient has been rewarmed to at least 32 C. The need for oxygen therapy, endotracheal intubation, controlled ventilation, warmed intravenous fluids, and treatment of metabolic acidosis should be dictated by clinical and laboratory monitoring during the rapid rewarming process. Essential laboratory tests include ECG, chest radiograph, complete blood count, prothrombin time, partial thromboplastin time, electrolytes, blood urea nitrogen (BUN), serum creatinine, liver function tests, amylase, glucose, pH, blood gases, urinalysis, and urine volume. Cardiac rhythm should be monitored, and cardiac, central vascular, or chest trauma or stimulation (catheter, cannulas, etc) should be avoided unless essential because of the risk of inducing ventricular fibrillation. However, patients who are comatose or in respiratory failure should be tracheally intubated. The patient should be evaluated for trauma and peripheral cold injury (eg, frostbite). Antibiotics are not routinely given and should be used only if indicated (neonate, elderly, or immunocompromised patient). Core temperature (esophageal preferred over rectal) should be monitored frequently during and after initial rewarming because of reports of recurrent hypothermia.
P.1623
A. Active External Rewarming Methods
Heated blankets, forced hot air, radiant heat, or warm baths may be used for active external rewarming. Rewarming by a warm bath is best done in a tub of moving water at 40 42 C, with a rate of rewarming of about 1 2 C/h. It is easier, however, to monitor the patient and to perform diagnostic and therapeutic procedures when heated blankets are used for active rewarming. Active external warming methods may predispose patients to ventricular fibrillation and hypovolemic shock and should be accompanied by core rewarming in moderate to severe hypothermia. Forced air rewarming (38 43 C) is recommended for clinic or field use when extracorporeal blood rewarming is not available; heated blankets are recommended for transport.
B. Active Internal (Core) Rewarming Methods
Internal rewarming is essential for patients with severe hypothermia; extracorporeal blood rewarming (cardiopulmonary, arteriovenous (femorofemoral), or venovenous bypass) is the treatment of choice, especially in the presence of cardiac arrest. Thoracic lavage and hemodialysis have also been recommended. Repeated peritoneal dialysis may be used with 2 L of warm (43 C) potassium-free dialysate solution exchanged at intervals of 10 12 minutes until the core temperature is raised to about 35 C. Parenteral fluids (D5 normal saline) should be warmed to 43 C prior to administration. Heated, humidified air warmed to 42 C through a face mask or endotracheal tube may be administered. Warm colonic and gastrointestinal irrigations are of less value.
Prognosis
With proper early care, more than 75% of otherwise healthy patients may survive moderate or severe systemic hypothermia. Prognosis is directly related to the severity of metabolic acidosis; with low pH ( 6.6), elevated PaCO2 ( 8.0 mm Hg), and/or elevated potassium ( 4.0 mEq/L), the prognosis is poor. The risk of aspiration pneumonia is great in comatose patients. The prognosis is grave if there are underlying predisposing causes or if treatment is delayed.
Hypothermia of the Extremities
Exposure of the extremities to cold produces immediate localized vasoconstriction followed by generalized vasoconstriction. When the skin temperature falls to 25 C, tissue metabolism is slowed, but the demand for oxygen is greater than the slowed circulation can supply, and the area becomes cyanotic. At 15 C, tissue metabolism is markedly decreased and the dissociation of oxyhemoglobin is reduced; this gives a deceptive pink, well-oxygenated appearance to the skin. Tissue damage occurs at this temperature. Tissue death may be caused by ischemia and thromboses in the smaller vessels or by actual freezing. Freezing (frostbite) does not occur until the skin temperature drops to 4 to 10 C or even lower, depending on such factors as wind, mobility, venous stasis, malnutrition, and occlusive arterial disease. Neuropathic sequelae such as pain, numbness, tingling, hyperhidrosis, cold sensitivity of the extremities, and nerve conduction abnormalities may persist for many years after the cold injury.
Prevention
Keep warm, keep moving, and keep dry. Individuals should wear warm, dry clothing, preferably several layers, with a windproof outer garment. Wet clothing, socks, and shoes should be replaced with dry ones. Extra socks, mittens, and insoles should always be carried in a pack in cold or icy areas. Cramped positions, constricting clothing, and prolonged dependency of the feet are to be avoided. Arms, legs, fingers, and toes should be exercised to maintain circulation. Wet and muddy ground and exposure to wind should be avoided. Tobacco and alcohol should be avoided when the danger of frostbite is present.
Chilblain (Erythema Pernio)
Chilblains or erythema pernio are red, painful, burning, itching skin lesions, usually on the extremities, caused by exposure to cold without actual freezing of the tissues. They may be associated with edema or blistering and are aggravated by warmth. With continued exposure, ulcerative or hemorrhagic lesions may appear and progress to scarring, fibrosis, and atrophy. Chilblain lupus erythematosus, while clinically similar to ordinary chilblain, can be differentiated by an association with other lupus manifestations or by biopsy.
Treatment consists of elevating the affected part slightly and allowing it to warm gradually at room temperature. Do not rub or massage injured tissues or apply ice or heat. Protect the area from trauma and secondary infection. Prazosin, 1 mg orally daily, has been recommended for treatment and prevention of recurrence. Nifedipine (extended release 30 60 mg every 24 hours) has been recommended for pain.
Frostbite
Frostbite is injury due to freezing and formation of ice crystals within tissues. In mild cases, only the skin and subcutaneous tissues are involved; the symptoms are numbness, prickling, and itching. With increasing severity, deep frostbite involves deeper structures, and there may be paresthesia and stiffness. Thawing causes tenderness and burning pain. The skin is white or yellow, loses its elasticity, and becomes immobile. Edema, blisters, necrosis, and gangrene may appear. MRI with magnetic resonance angiography and triple-phase bone scanning have been used to assess the degree
P.1624
of involvement in severe frostbite and to distinguish viable from nonviable tissue.
Treatment
A. Immediate Treatment
Evaluate and treat the patient for associated systemic hypothermia.
1. Rewarming
Superficial frostbite (frostnip) of extremities in the field can be treated by firm steady pressure with the warm hand (without rubbing), by placing fingers in the armpits, and, in the case of the toes or heels, by removing footwear, drying feet, rewarming, and covering with adequate dry socks or other protective footwear.
For deep frostbite, rapid thawing at temperatures slightly above body heat may significantly decrease tissue necrosis. If there is any possibility of refreezing, the frostbitten part should not be thawed, even if this might mean prolonged walking on frozen feet. Refreezing results in increased tissue necrosis. Rewarming is best accomplished by immersing the frozen extremity for several minutes in a moving water bath heated to 40 42 C until the distal tip of the part being thawed flushes. Water in this temperature range feels warm but not hot to the normal hand. Dry heat (eg, stove or open fire) is more difficult to regulate and is not recommended. After thawing has occurred and the part has returned to normal temperature (usually in about 30 minutes), discontinue external heat. Victims and rescue workers should be cautioned not to attempt rewarming by exercise or thawing of frozen tissues by rubbing with snow or ice water.
2. Protection of the part
Pressure or friction is avoided and physical therapy contraindicated in the early stage. The patient is kept at bed rest with the affected parts elevated and uncovered at room temperature. Casts, dressings, or bandages are not applied. A combination of ibuprofen, 200 mg four times daily, and aloe vera has been used to prevent dermal ischemia. Although white blisters may be debrided, hemorrhagic blisters should be left intact.
3. Anti-infective measures
Consider tetanus prophylaxis; frostbite increases susceptibility. Protect skin blebs from physical contact. Local infections may be treated with mild soaks of soapy water or povidone-iodine. Whirlpool therapy at 37 40 C twice daily for 15 20 minutes for a period of 3 or more weeks helps cleanse the skin and debrides superficial sloughing tissue. Antibiotics may be required for deep infections.
4. Other therapy
Monitor fluid and electrolyte balance; administer tetanus prophylaxis if indicated. Other treatments used with varying results include platelet aggregation inhibition, hemodilution, peripheral vasodilation, fibrinolysis, and hyperbaric oxygen.
B. Follow-Up Care
Gentle, progressive physical therapy to promote circulation should be instituted as tolerated.
C. Surgery
Early regional sympathetic blockade can reduce symptoms; sympathectomy (within 36 72 hours) is controversial. In general, other operative procedures are to be avoided. Amputation should not be considered until it is definitely established that the tissues are dead. Tissue necrosis (even with black eschar formation) may be quite superficial, and the underlying skin may sometimes heal spontaneously even after a period of months.
Prognosis
Recovery from frostbite is most often complete, but there may be increased susceptibility to discomfort in the involved extremity upon reexposure to cold.
Immersion Syndrome (Immersion Foot or Trench Foot)
Immersion foot (or hand) is caused by prolonged immersion in cool or cold water or mud, usually less than 10 C. The affected parts are first cold and anesthetic (prehyperemic stage). They become hot with intense burning and shooting pains during the hyperemic stage and pale or cyanotic with diminished pulsations during the vasospastic period (posthyperemic stage); blistering, swelling, redness, heat, ecchymoses, hemorrhage, necrosis, peripheral nerve injury, or gangrene and secondary complications such as lymphangitis, cellulitis, and thrombophlebitis can occur later.
Treatment is best instituted during the stage of reactive hyperemia. Treatment consists of air drying, protecting the extremities from trauma and secondary infection, and gradual rewarming by exposure to air at room temperature (not ice or heat) without massaging or moistening the skin or immersing it in water. Bed rest is required until all ulcers have healed. Affected parts are elevated to aid in removal of edema fluid, and pressure sites (eg, heels) are protected with pillows. Later treatment is as for Buerger's disease (see Chapter 12).
Kempainen RR et al: The evaluation and management of accidental hypothermia. Respir Care 2004;49:192.
Petrone P et al: Surgical management and strategies in the treatment of hypothermia and cold injury. Emerg Med Clin North Am 2003;21:1165.
Simon TD et al: Pernio in pediatrics. Pediatrics 2005;116:e472.
Ulrich AS et al: Hypothermia and localized cold injuries. Emerg Med Clin North Am 2004;22:281.
Disorders Due to Heat
![]() Essentials of Diagnosis
Essentials of Diagnosis
The four disorders that comprise the spectrum of illness due to heat exposure are heat syncope, heat cramps, heat exhaustion, and heat stroke.
Heat disorders are caused by dehydration or electrolyte imbalance, or both.
P.1625
General Considerations
Conduction (convection) the direct transfer of heat from the skin to the surrounding air occurs with diminishing efficiency as ambient temperature rises. The passive transfer of heat from a warmer to a cooler object by radiation accounts for 65% of body heat loss under normal conditions. Radiant heat loss decreases as the temperature of the surrounding environment increases up to 37.2 C, the point at which heat transfer reverses direction. At normal temperatures, evaporation accounts for approximately 20% of the body's heat loss, but at high temperatures it becomes the major mechanism for dissipation of heat; with vigorous exertion, sweat loss can be as much as 2.5 L/h. This mechanism diminishes as humidity rises.
Health conditions that inhibit sweat production or evaporation and increase susceptibility to heat disorders include obesity, skin disorders (miliaria), reduced cutaneous blood flow, dehydration, malnutrition, hypotension, and reduced cardiac output. Medications that impair sweating include anticholinergics, antihistamines, phenothiazines, tricyclic antidepressants, monoamine oxidase inhibitors, and diuretics; reduced cutaneous blood flow results from use of vasoconstrictors and -adrenergic blocking agents; and dehydration results from use of alcohol. Illicit drugs eg, phencyclidine, LSD, amphetamines, and cocaine can cause increased muscle activity and thus generate increased body heat. Drug withdrawal syndromes or prolonged seizures may have the same effect. The risk of heat disorder increases with age, impaired cognition, concurrent illness, body mass index, reduced physical fitness, duration of exertion, and insufficient acclimatization.
Prevention
Medical evaluation and monitoring should be used to identify individuals at increased risk for heat disorders. The public should be made aware of the early symptoms and signs of heat disorders. It is not recommended to make salt tablets available for use without medical supervision; close monitoring of fluid and electrolyte intake and early intervention are recommended in situations necessitating exertion or activity in hot environments. Athletic events should be organized and managed with attention to thermoregulation: the wet bulb globe temperature (WBGT) index should be monitored, fluid consumption should be encouraged, and medical support should be immediately accessible. Competition is not recommended when the WBGT exceeds 26 28 C. Workers should not begin work in hot temperatures without proper acclimatization and should be encouraged to drink water or balanced electrolyte fluids frequently. Guidance regarding heat hazard can be found in the National Weather Service's Heat Index that rates weather conditions based on humidity and temperature measurements (http://www.weather.gov/om/heat/index.shtml). The US Army provides guidance regarding activity levels according to WBGT levels (http://www.usariem.army.mil/heatill/appendc.htm).
Acclimatization is achieved by scheduled regulated exposure to hot environments and by gradually increasing the duration of exposure and the work load until the body adjusts by producing sweat of lower salt content in greater amounts at lower ambient temperatures. Acclimatization is accompanied by increased plasma volume, cardiac output, and cardiac stroke volume and a slower heart rate.
Specific Syndromes Due to Heat Exposure
1. Heat Syncope
Sudden unconsciousness can result from volume depletion and cutaneous vasodilation with consequent systemic and cerebral hypotension. Systolic blood pressure is usually less than 100 mm Hg, and there is typically a history of vigorous physical activity for 2 hours or more just preceding the episode. The skin is typically cool and moist, and the pulse is weak.
Treatment consists of rest and recumbency in a cool place and rehydration by mouth (or intravenously if necessary).
2. Heat Cramps
Fluid and electrolyte depletion can result in slow, painful skeletal muscle contractions ( cramps ) and severe muscle spasms lasting 1 3 minutes, usually of the muscles most heavily used. Cramping results from dilutional hyponatremia as sweat losses are replaced with water alone. The skin is moist and cool, and the muscles are tender. There may be muscle twitching. The victim is alert, with stable vital signs, but may be agitated and complaining of pain. The body temperature may be normal or slightly increased. Involved muscle groups are hard and lumpy. There is almost always a history of vigorous activity just preceding the onset of symptoms. Laboratory evaluation may show low serum sodium, hemoconcentration, and elevated urea and creatinine.
The patient should be moved to a cool environment and given oral saline solution (4 tsp of salt per gallon of water) to replace both salt and water. Because of their slower absorption, salt tablets are not recommended. The victim may have to rest for 1 3 days with continued dietary salt supplementation before returning to work or resuming strenuous activity in the heat.
3. Heat Exhaustion
Heat exhaustion results from prolonged strenuous activity with inadequate water or salt intake in a hot environment and is characterized by dehydration, sodium
P.1626
depletion, or isotonic fluid loss with accompanying cardiovascular changes.
The diagnosis is based on prolonged symptoms and a rectal temperature over 37.8 C, increased pulse (> 150% of the patient's normal) and moist skin. Symptoms associated with heat syncope and heat cramps may be present. Nausea, vomiting, malaise, and myalgia may occur. The patient may be quite thirsty and weak, with central nervous system symptoms such as headache, dizziness, fatigue, and, in cases due chiefly to water depletion, anxiety, paresthesias, impaired judgment, hysteria, and occasionally psychosis. Hyperventilation secondary to heat exhaustion can cause respiratory alkalosis; lactic acidosis may also occur due to poor tissue perfusion. Heat exhaustion may progress to heat stroke if sweating ceases.
Treatment consists of patient location in a shaded, cool environment, providing adequate hydration (1 2 L over 2 4 hours), salt replenishment orally, if possible and active cooling (fans, ice packs, etc) if necessary. Physiologic saline or isotonic glucose solution should be administered intravenously when oral administration is not appropriate. Intravenous 3% (hypertonic) saline may be necessary if sodium depletion is severe. At least 24 hours of rest is recommended.
4. Heat Stroke
Heat stroke is a life-threatening medical emergency resulting from failure of the thermoregulatory mechanism. Heat stroke is imminent when the core (rectal) temperature approaches 41 C. It presents in one of two forms: Classic heat stroke occurs in patients with compromised homeostatic mechanisms; exertional heat stroke occurs in healthy persons undergoing strenuous exertion in a thermally stressful environment. Morbidity or even death can result from cerebral, cardiovascular, hepatic, or renal damage.
The hallmarks of heat stroke are cerebral dysfunction with impaired consciousness, high fever, and absence of sweating. Persons at greatest risk are the very young, the elderly (age > 65), chronically infirm, and patients receiving medications (eg, anticholinergics, antihistamines, phenothiazines) that interfere with heat-dissipating mechanisms.
Exertional heat stroke and exertion-related disorders such as rhabdomyolysis are appearing more frequently as complications of participation by unconditioned amateurs in strenuous athletic activities such as marathon and triathlon competition.
Clinical Findings
A. Symptoms and Signs
Heat stroke may present with dizziness, weakness, emotional lability, nausea and vomiting, diarrhea, confusion, delirium, blurred vision, convulsions, collapse, and unconsciousness. The skin is hot and initially covered with perspiration; later it dries. The pulse is strong initially. Tachycardia and hyperventilation (with subsequent respiratory alkalosis) occur. Blood pressure may be slightly elevated at first, but hypotension develops later. The core temperature is usually over 40 C. Exertional heat stroke may present with sudden collapse and loss of consciousness followed by irrational behavior. Anhidrosis may not be present. Twenty-five percent of heat stroke victims have prodromal symptoms for minutes to hours that may include dizziness, weakness, nausea, confusion, disorientation, drowsiness, and irrational behavior. Multiorgan dysfunction or injury is a common and serious complication.
B. Laboratory Findings
Laboratory evaluation may reveal dehydration; leukocytosis; elevated BUN; hyperuricemia; hemoconcentration; acid-base abnormalities (eg, lactic acidosis, respiratory alkalosis); and decreased serum potassium, sodium, calcium, and phosphorus. Urine is concentrated, with elevated protein, tubular casts, and myoglobinuria. Thrombocytopenia, fibrinolysis, and disseminated intravascular coagulopathy (DIC) may occur; coagulopathy is likely. Rhabdomyolysis and myocardial, hepatic, or renal damage may be identified by elevated serum creatine kinase and aminotransferase levels and BUN and by the presence of anuria, proteinuria, and hematuria. Electrocardiographic findings may include ST-T changes consistent with myocardial ischemia. PCO2 may be less than 20 mm Hg.
Treatment
Treatment is aimed at reducing the core temperature rapidly (within 1 hour) while supporting organ system function. Immersion in an ice-water bath (1 5 C) is the most effective cooling method but may not be practical due to its physical requirements and patient access limitations. Evaporative cooling is a rapid and effective alternative and is easily performed in most emergency settings. The patient's clothing should be removed and the entire body sprayed with water (20 C) while ambient or slightly warmed (45 C) air is passed across the patient's body with large fans or other means at high velocity (100 ft/min). The patient should be in the lateral recumbent position or supported in a hands-and-knees position to expose as much skin surface as possible to the air. Also effective are use of cold wet sheets accompanied by fanning, immersion in chilled water, and localized ice or ice slush application. Skin massage is recommended to prevent cutaneous vasoconstriction. Intravascular heat exchange catheter systems as well as hemodialysis using cold dialysate (30 35 C) have been successful in reducing core temperature. Other cooling alternatives include hand and forearm immersion in cold water, ice packs (groin, axillas, neck), and iced gastric lavage, although these are much less effective than evaporative cooling.
Treatment should be continued until the rectal temperature drops to 39 C. The temperature remains
P.1627
stable in most cases, but it should continue to be monitored for 24 hours. Antipyretics (aspirin, acetaminophen) have no effect on environmentally induced hyperthermia and are contraindicated.
Hypovolemic and cardiogenic shock must be carefully distinguished, as either or both may occur. Central venous or pulmonary artery wedge pressure should be monitored. Five percent dextrose in half-normal or normal saline should be administered for fluid replacement.
The patient should also be observed for renal failure due to rhabdomyolysis, hypokalemia, cardiac arrhythmias, DIC, and hepatic failure. Creatine phosphokinase (CPK) >1000 units/L, metabolic acidosis, and elevated liver enzymes are predictive of multiorgan dysfunction, the usual cause of heat stroke-related death. Multiorgan dysfunction and inflammation may continue after temperature is normalized. Hypokalemia frequently accompanies heat stroke but may not appear until rehydration. Maintenance of extracellular hydration and electrolyte balance should reduce the risk of renal failure due to rhabdomyolysis. Fluid administration to ensure a high urinary output (> 50 mL/h), mannitol administration (0.25 mg/kg), and alkalinizing the urine (intravenous bicarbonate administration, 250 mL of 4%) are recommended. Fluid output should be monitored through the use of an indwelling urinary catheter.
Because sensitivity to high environmental temperature may persist for prolonged periods following an episode of heat stroke, immediate reexposure should be avoided.
Glazer JL: Management of heatstroke and heat exhaustion. Am Fam Physician 2005;71:2133.
Lugo-Amador NM et al: Heat-related illness. Emerg Med Clin North Am 2004;22:315.
Burns
The incidence and severity of burn injuries has been declining, with both deaths and acute hospitalizations attributable to burns down about 50%. Over three-fourths of burns involve less than 10% of total body surface area. Aggressive, early excision (24 72 hours postburn) of deeply burned tissues and skin grafting, early enteral feeding, and improved infection control have contributed to significantly lower mortality rates and shorter hospitalizations. Nonetheless, an estimated 1.25 million burn injuries and 51,000 acute hospitalizations of burn victims occur each year in the United States. Severe burns cause problems in the initial phase from hemodynamic compromise, related injuries such as smoke inhalation or fractures, and associated microbacterial superinfection, sepsis and multiorgan failure. Later, secondary scarring and constrictive wounds occur.
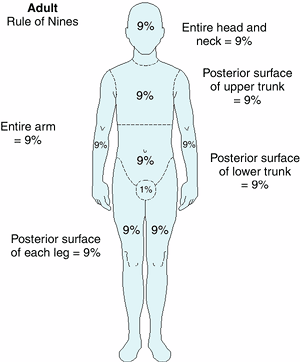 |
Figure 38-2. Estimation of body surface area in burns. |
Significant quality of life and social functionality can be expected even for severely burned patients.
Classification
Burns are classified by extent, depth, patient age, and associated illness or injury. Accurate estimation of burn size and depth will determine the adequacy of resuscitation.
Extent
The rule of nines (Figure 38-2) is useful for rapidly assessing the extent of a burn. More detailed charts based on age are available when the patient reaches the burn unit. Therefore, it is important to view the entire patient after cleaning soot to make an accurate assessment, both initially and on subsequent examinations. Only second- and third-degree burns are included in calculating the total burn surface area, since first-degree burns usually do not represent significant injury in terms of prognosis or fluid and electrolyte management. However, first- or second-degree burns may convert to deeper burns, especially if treatment is delayed or bacterial colonization or superinfection occurs.
Depth
Judgment of depth of injury is difficult. The first-degree burn may be red or gray but will demonstrate excellent capillary refill. First-degree burns are not blistered initially. If the wound is blistered, this represents a partial-thickness injury to the dermis, or a second-degree burn. As the degree of burn is progressively deeper, there is a progressive loss of adnexal structures. Hairs can be easily extracted or are absent, sweat glands become less visible, and the skin appears smoother.
P.1628
Deep second- and third-degree burns are treated in a similar fashion, since neither will heal appropriately without early debridement and grafting; the resultant skin is thin and scarred.
Survival after Burn Injury
The survival from major burn injury has dramatically increased in the last 20 years. Consistently, the three major risk factors for mortality were age greater than 60 years, burn area greater than 40% of total body surface area, and inhalation injury. A good rule of thumb for predicting mortality after severe burn injury is still the Baux Score (age + percent burn, eg, age 50 years + 20% burn = 70% mortality), although this is obviously quite variable depending on associated medical factors. Burn size, age, history of electrical injury, history of concomitant trauma (especially penetrating) and female sex as well as duration of stay in the ICU and the presence of mechanical ventilation presage a poorer outcome. Likewise, a base deficit during resuscitation of greater than 6 mmol/L predicts a far higher incidence of multiple organ dysfunction and death.
Associated Injuries or Illnesses
An injury commonly associated with burns is smoke inhalation (see Chapter 9). Suspicion of inhalation injury is aroused when the nasal hairs are singed, the mechanism of burn involves closed spaces, the sputum is carbonaceous, or the carboxyhemoglobin level exceeds 5% in nonsmokers. This suspicion should lead the clinician to institute early intubation before airway edema supervenes. The products of combustion, not heat, are responsible for lower airway injury. Electrical injury that causes burns may also produce cardiac arrhythmias that require immediate attention. Pancreatitis occurs in severe burns. Development of hyperamylasemia or hyperlipasemia may signal the development of pancreatic inflammation and subsequent pseudocyst or abscess formation. Prior alcohol exposure may exacerbate the pulmonary components of burn injury.
Toxic epidermal necrolysis (TEN) occasionally occurs following sulfonamide or phenytoin administration (see Chapter 6). If TEN is severe, patients are best transferred to a burn unit and treated as having severe burn injury. Corticosteroid therapy should be avoided.
Systemic Reactions to Burn Injury
The actual burn injury is only the incipient event in a cascade of deleterious local tissue and systemic inflammatory reactions leading to multiorgan system failure in the severely burned patient. Locally, substance P, serotonin, prostaglandins E2 and F2 , histamine, platelet-activating factor, nitric oxide, bradykinin, and leukotrienes B4 and D4 play a role in the increased local capillary permeability and initiation of the systemic inflammatory cascade. Systemically, levels of interleukin-2, -4, -6, and interferon-gamma (IFN- ) are elevated in proportion to the severity of the burn injury, perhaps as part of a generalized systemic release of inflammatory mediators and generalized macrophage dysfunction. There is often an altered CD4/CD8 (T helper/T suppressor) cell ratio. In addition, in the first 24 hours following a significant burn, there is production of tumor necrosis factor and IFN- , which in turn stimulate production of the enzyme nitric oxide synthetase in hepatocytes. After 24 hours, lipopolysaccharide plays a dominant role in its production. Nitric oxide has been proposed as a mediator of the acute inflammation. Transforming growth factor beta and IL-10 and a markedly elevated level of IL-6 serve as harbingers of a declining clinical course in severely burned patients.
Initial Management
Airway
The practitioner or emergency medical technician should proceed as with any other trauma using standard Advanced Trauma Life Support (ATLS) guidelines. The priorities are first to establish an airway, recognizing the frequent necessity to intubate a patient who may appear to be breathing normally but who has sustained an inhalation injury; next, to evaluate the cervical spine and head injuries; and finally, to stabilize fractures. Fluid resuscitation by the Parkland formula (see below) may be instituted simultaneously with initial resuscitation. Endotracheal intubation or tracheotomy should be considered for major burn cases regardless of the area of the body involved, because as fluid resuscitation proceeds generalized edema develops, including edema of the soft tissues of the upper airway and perhaps the lungs as well. Chest radiographs are typically normal initially but may develop an acute respiratory distress syndrome picture in 24 48 hours with severe inhalation injury. Supplemental oxygen should be administered. Inhalation injuries should be monitored with serial blood gas determination and bronchoscopy. The use of corticosteroids is contraindicated because of the potential for immunosuppression.
Vascular Access
All clothing and jewelry should be removed and an expedient physical examination performed to assess the extent of burn and associated injuries. Simultaneously with the above procedures, venous access must be sought, since the victim of a major burn may develop hypovolemic shock. A percutaneous large-bore (14- or 16-gauge) intravenous line through nonburned skin is preferred. Subclavian lines are avoided in the emergency setting because of the risk of pneumothorax and subclavian vein laceration when such a line is placed in a volume-depleted patient. Femoral lines provide good temporary access during resuscitation. All lines without exception placed in the emergency department should be changed within 24 hours because of the high risk of nonsterile placement. Distal saphenous cutdown is occasionally necessary. An arterial line is useful for monitoring mean arterial pressure and drawing blood.
P.1629
Fluid Resuscitation
Crystalloids
Generalized capillary leak results from burn injury over more than 25% of total body surface area. This often necessitates replacement of a large volume of fluid.
There are many guidelines for fluid resuscitation. The Parkland formula relies on the use of lactated Ringer's injection. The fluid requirement in the first 24 hours is estimated as 4 mL/kg body weight per percent of body surface area burned. Half the calculated fluid is given in the first 8-hour period. The remaining fluid, divided into two equal parts, is delivered over the next 16 hours. An extremely large volume of fluid may be required. For example, an injury over 40% of the total body surface area in a 70-kg victim may require 11.2 L in the first 24 hours [4 mL 40(%) 70 (kg) = 11,200 mL]. The first 8-hour period is measured from the hour of injury. These guidelines may be inadequate, since crystalloid solutions alone may be insufficient to restore cardiac preload during the period of burn shock.
Deep electrical burns and inhalation injury increase the fluid requirement. Adequacy of resuscitation is determined by clinical parameters, including urinary output and specific gravity, blood pressure, and central venous catheter or, if necessary, Swan-Ganz catheter readings.
Colloids
Overly aggressive crystalloid administration must be avoided in patients with pulmonary injury, since significant pulmonary edema can develop in patients with normal pulmonary capillary wedge and central venous pressures. In addition, routine colloid administration, previously commonplace, is no longer warranted in routine burn resuscitation in view of its deleterious effect on glomerular filtration and its association with pulmonary edema.
Monitoring Fluid Resuscitation
A Foley catheter is essential for monitoring urinary output. Diuretics have no role in this phase of patient management unless fluid overload has occurred or mannitol diuresis is performed in the case of rhabdomyolysis.
Abdominal Compartment Syndrome
Abdominal compartment syndrome is emerging as a complication in severely burned patients. Increased intra-abdominal pressures can cause excessively high peak inspiratory pressures and increase fluid requirements considerably. Elevated intra-abdominal pressure can make pulmonary artery wedge pressure and urinary output unreliable indices of preload or intravascular volume, causing improper management of the patient's fluid status. Only 40% of patients with this severe form of increased intra-abdominal pressure survive. Bladder pressures over 30 mm Hg establish the diagnosis in at-risk patients. Surgical abdominal decompression may be indicated, and may improve ventilation and oxygen delivery.
Escharotomy
As edema fluid accumulates, ischemia may develop under any constricting eschar of an extremity, neck, or trunk if the full-thickness burn is circumferential. Escharotomy incisions through the anesthetic eschar can save life and limb and can be performed in the emergency department or operating room. Recent studies have shown efficacy of enzymatic debridement of wounds requiring escharotomy.
Fasciotomy in Electrical Burns or Crush Injuries
When high-voltage electrical injury occurs, extensive deep tissue necrosis should be suspected. Deep tissue necrosis leads to profound tissue swelling. Because deep tissue compartments in the arms and legs are contained by unyielding fascia, these compartments must be opened by surgical fasciotomy to prevent further soft tissue, vascular, and nerve death.
Electrical burn injuries remain the most devastating and underrecognized burn injuries, causing amputations (often because of unrecognized compartment syndromes) and acute renal failure, resulting in part from rhabdomyolysis. Creatine kinase levels are usually elevated in patients with severe electrical burns (and constitute a negative prognostic indicator). Severe (often underrecognized) muscle necrosis due to electrical burns may occur in the absence of significant skin burns.
The Burn Wound
In general, when burn wounds are seen in the emergency department, the causative agents have been eliminated. However, acute burns caused by hydrofluoric acid should be treated by water rinsing followed by application of topical calcium. Treatment of the burn wound is based on several principles. (1) Protection from desiccation and further injury of those burned areas that will spontaneously reepithelialize in 7 10 days by application of topical antibiotic such as silver sulfadiazine or mafenide acetate. Silver sulfadiazine is the most popular topical agent. It is painless, easy to apply, and effective against most strains of Pseudomonas. (2) Regular and thorough cleansing of burned areas is a critically important intervention in burn units. Early excision and grafting of burned areas as soon as 24 hours after burn injury or when the patient will hemodynamically tolerate the excision and grafting procedure. The tumescent technique (injection under the burn scar of dilute epinephrine and lidocaine prior to surgery) may reduce blood loss, though lidocaine toxicity must be considered.
P.1630
Systemic infection remains a leading cause of morbidity among patients with major burn injuries, with nearly all severely burned patients having one or more septicemic episodes during the hospital course. Coagulase-negative staphylococci (63%) and Staphylococcus aureus (20%) are commonly cultured from burn wounds. Methicillin-resistant S aureus and Pseudomonas aeruginosa are also commonly cultured but usually later in the hospital course. Methicillin resistance in gram-positive organisms is becoming common, but vancomycin-resistant staphylococci are fortunately still rare in burn patients. Increasingly, sterile multiple organ failure is regarded as a leading cause of death among severely burned patients.
Wound Closure
The goal of therapy after fluid resuscitation is rapid and stable closure of the wound. Wounds that will not heal spontaneously in 7 10 days (ie, deep second-degree or third-degree burns) are best treated by excision and autograft; otherwise, granulation and infection may develop, and the quality of the skin in regenerated deep partial thickness burns is marginal because of the very thin dermis that emerges.
Biosynthetic skin replacements are now routinely used when autologous skin grafts are in short supply. Cultured allogeneic keratinocyte grafts, autologous/allogeneic composites, acellular biological matrices, and cellular matrices, including biological substances as fibrin sealant, various types of collagen, and hyaluronic acid, have provided alternatives. With severe burns, skin substitution with cultured grafts can be lifesaving. Research continues on the development of the completely engineered skin substitute, with especially promising results in the area of fetal cell constructs.
Patient Support
Burn patients require extensive support. An attempt must be made to maintain normal core body temperature (by maintaining environmental temperature at or above 30 C) in patients with burns over more than 20% of total body surface area, since the hypermetabolic state of burns is exacerbated by subnormal temperatures. Respiratory injury, sepsis, and multiorgan failure are common. Enteral feedings may be started once the ileus of the resuscitation period has resolved, usually the day after the injury. There is often a markedly increased metabolic rate after burn injury, due in large part to whole body synthesis and increased fatty acid substrate cycles. If the patient does not tolerate low-residue tube feedings, total parenteral nutrition should be started without delay through a central venous catheter. As much as 4000 6000 kcal/d in high-carbohydrate, high-fat diet may be required in the postburn period. The metabolic demands are immense. A useful guide is to provide 25 kcal/kg body weight plus 40 kcal per percent of burn surface area. Early aggressive enteral nutrition reduces infections, noninfectious complications, length of hospital stay, impaired healing, and mortality. Anticatabolic treatments such as -blockade, growth hormone, insulin, oxandrolone, and synthetic testosterone have been advocated. Occasionally, acute respiratory distress syndrome or respiratory failure unresponsive to maximal ventilatory support may develop in burn patients. In addition, the incidence of venous thromboembolism in the lower extremities is high among burn patients. Duplex ultrasonography is the best method for identifying venous thromboembolism. Early pulmonary dysfunction after severe burn injury is widely recognized. It is now apparent in children that late and permanent pulmonary dysfunction with later obstructive and restrictive lung disease can result from major burn injury. Growth hormone (0.05 mg/kg/d from hospital discharge to 1 year postdischarge) may reduce the long-term deleterious effects on hepatic acute phase and constitutive proteins.
Ophthalmologic consultation is indicated early when ocular or corneal injury is suspected.
Prevention of long-term scars remains a formidable problem in seriously burned patients. The V-beam laser (or long-pulsed dye laser) is emerging as adjunctive treatment to the usual regimen of corticosteroid injections, silicone patches, compression, and scar revision. Permanent sequelae can be avoided by prevention of infection, early aggressive rehabilitation, compressive garments, and early psychological support. Prefabricated flaps offer promise to severely burned patients. Facial transplantation has become a controversial but emerging treatment for those patients with devastating facial burns.
Complex reconstructions of severely burned structures such as the neck continue to benefit from advances in microsurgical plastic surgery techniques. After severe hand burns, function may be aided by expedient splinting or axial pin fixation to prevent flexion contractures and facilitate prompt skin grafting and physical therapy. Aggressive microsurgical reconstructions of hand injuries facilitate early motion and improved function of burn injuries to the hand.
Patients who have suffered severe burn injuries can recover to a general health status slightly lower than that of the general population but still have very significant vocational and psychological problems.
Atiyeh BS et al: State of the art in burn treatment. World J Surg 2005;29:131.
Herndon DN et al: Support of the metabolic response to burn injury. Lancet 2004;363:1895.
Hohlfeld J et al: Tissue engineered fetal skin constructs for paediatric burns: Lancet 2005;366:840.
Horch RE et al: Tissue engineering of cultured skin substitutes: J Cell Mol Med 2005;9:592.
Kono T et al: Treatment of hypertrophic scars using a long-pulsed dye laser with cryogen-spray cooling: Ann Plast Surg 2005;54:487.
Krieger Y et al: Escharotomy using an enzymatic debridement agent for treating experimental burn-induced compartment syndrome in an animal model. J Trauma 2005;58:1259.
P.1631
Lee JO et al: Nutrition support strategies for severely burned patients. Nutr Clin Pract 2005;20:325.
Nagasao T et al: Preliminary repair of eyelids for the treatment of opaque corneas caused by burns. Scand J Plast Reconstr Surg Hand Surg 2005;39:227.
Oda J et al: Effects of escharotomy as abdominal decompression on cardiopulmonary function and visceral perfusion in abdominal compartment syndrome with burn patients. J Trauma 2005;59:369.
Ulkur E et al: Comparison of silver-coated dressing (Acticoat), chlorhexidine acetate 0.5% (Bactigrass), and silver sulfadiazine 1% (Silvadine) for topical antibacterial effect in Pseudomonas aeruginosa-contaminated, full-thickness burn wounds in rats. J Burn Care Rehabil 2005;26:430.
Electric Shock
![]() Essentials of Diagnosis
Essentials of Diagnosis
Injury is determined by quantity, duration, and type of current; area of exposure/contact; and pathway of current through the body.
General Considerations
Current passing through skeletal muscle can cause muscle necrosis and contractions severe enough to result in bone fracture. Current traversing peripheral nerves can cause acute or delayed neuropathy. Delayed effects can include damage to the eye, spinal cord, peripheral nerves, bone, kidneys, and gastrointestinal tract. If the current passes through the heart or brainstem, death may be immediate due to ventricular fibrillation, asystole, or apnea.
With alternating currents (AC) of 25 300 Hz, low voltages (< 220 Hz) tend to produce ventricular fibrillation; high voltages (> 1000 Hz) cause respiratory failure; intermediate voltages (220 1000 Hz) cause both. More than 100 mA of domestic house AC of 110 volts at 60 Hz is, accordingly, dangerous to the heart, since it can cause ventricular fibrillation. Direct current (DC) contact is more likely to cause asystole.
Lightning injuries differ from high-voltage electric shock injuries; lightning usually involves higher voltage, briefer duration of contact, asystole, nervous system injury, and multisystem pathologic involvement.
Electrical burns are of three distinct types: flash (arcing) burns, flame (clothing) burns, and the direct heating effect of tissues by the electric current. The latter lesions are usually sharply demarcated, round or oval, painless yellow-brown areas (Joule burn) with inflammatory reaction. Significant subcutaneous damage can be accompanied by little skin injury, particularly with larger skin surface area electrical contact.
Clinical Findings
Electric shock may produce loss of consciousness. With recovery there may be muscular pain, fatigue, headache, and nervous irritability. The physical signs vary according to the action of the current. Ventricular fibrillation or respiratory failure or both can occur; the patient may be unconscious, pulseless, hypotensive, cold and cyanotic, and without respirations.
Treatment
A. Emergency Measures
The victim must be separated from the electric current prior to initiation of CPR or other treatment; the rescuer must be protected. Turn off the power, sever the wire with a dry wooden-handled ax, make a proper ground to divert the current, or separate the victim using nonconductive implements such as dry clothing.
B. Hospital Measures
Lightning or unstable electric shock victims should be hospitalized when revived and observed for shock, arrhythmia, thrombosis, infarction, sudden cardiac dilation, hemorrhage, and myoglobinuria. A urinalysis, urine myoglobin, serum creatine kinase and creatine kinase-MB, and an ECG should be obtained immediately. Victims should also be evaluated for hidden injury (eg, ophthalmic, otologic, muscular, compartment syndromes), organ injury (myocardium, liver, kidney, pancreas), blunt trauma, dehydration, skin burns, hypertension, posttraumatic stress, acid-base disturbances, and neurologic damage. Indications for hospitalization include significant arrhythmia or electrocardiographic changes, large burn, loss of consciousness, pulmonary or cardiac symptoms, or evidence of significant deep tissue or organ damage. Extra caution is indicated when the electroshock current has followed a transthoracic route (hand to hand or hand to foot) and in patients with a cardiac history.
To counteract fluid losses and myoglobinuria due to electric shock (not lightning) burns, aggressive hydration with Ringer's lactate should seek to achieve a urinary output of 50 100 mL/h.
Prognosis
Complications may occur in almost any part of the body but most commonly include sepsis; gangrene requiring limb amputation; or neurologic, cardiac, cognitive, or psychiatric dysfunction. Psychiatric support may be necessary following lightning or severe electroshock exposures.
Edlich RF et al: Modern concepts of treatment and prevention of lightning injuries. J Long Term Eff Medical Implants 2005;15:185.
O'Keefe Gatewood M et al: Lightning injuries. Emerg Med Clin North Am 2004;22:369.
Selvaggi G et al: Rehabilitation of burn injured patients following lightning and electrical trauma. NeuroRehabilitation 2005;20:35.
P.1632
Ionizing Radiation Reactions
The extent of damage due to radiation exposure depends on the quantity of radiation delivered to the body, the dose rate, the organs exposed, the type of radiation (x-rays, neutrons, gamma rays, alpha or beta particles), the duration of exposure, and the energy transfer from the radioactive wave or particle to the exposed tissue. The Chernobyl experience suggests that the best biologic indicators of dose from an acute exposure are the duration of the asymptomatic latent period (particularly for nausea or emesis), the severity of early symptoms, the rate of decline of the lymphocyte count, and the number and distribution of dicentric chromosomes in peripheral lymphocytes.
The National Committee on Radiation Protection has established the maximum permissible radiation exposure for occupationally exposed workers over age 18 as 1 mSv (0.1 rem)* per week for the whole body (but not to exceed 50 mSv per year) and 15 mSv per week for the hands. (For purposes of comparison, routine chest radiographs deliver from 1 mSv to 2 mSv.) The FDA has recommended 1 Gy as a threshold for skin-absorbed dose from medical fluoroscopy.
Death after whole body acute lethal radiation exposure is usually due to hematopoietic failure, gastrointestinal mucosal damage, central nervous system damage, widespread vascular injury, or secondary infection. The acute radiation syndrome may be dominated by central nervous system, gastrointestinal, or hematologic manifestations depending on dose and survival. Fatigue, weakness, and anorexia can occur following exposures exceeding 50 cGy. Hematopoietic effects consisting of anemia, platelet loss, and bone marrow suppression can occur 1 3 weeks after exposures exceeding 100 cGy. Exposure to 400 600 cGy may be fatal within 60 days; death is usually due to hemorrhage, anemia, and infection secondary to hematopoietic injury. Whole body exposure levels of 1000 3000 cGy destroy gastrointestinal mucosa; this leads to toxemia and death within 2 weeks. Total body doses above 3000 cGy cause widespread vascular damage, cerebral anoxia, hypotensive shock, and death within 48 hours.
Acute (Immediate) Ionizing Radiation effects on Normal Tissues
Clinical Findings
A. Injury to Skin and Mucous Membranes
Irradiation may cause erythema, epilation, destruction of fingernails, or epidermolysis. Ionizing radiation burns appear similar to thermal burns but usually have a slower onset and course.
B. Injury to Deep Structures
1. Hematopoietic tissues
Injury to the bone marrow may cause diminished production of blood elements. Lymphocytes are most sensitive, polymorphonuclear leukocytes next most sensitive, and erythrocytes least sensitive. Damage to the blood-forming organs may vary from transient depression of one or more blood elements to complete destruction.
2. Cardiovascular system
Pericarditis with effusion or constrictive pericarditis may occur after a period of months or even years. Myocarditis is less common. Smaller vessels (the capillaries and arterioles) are more readily damaged than larger blood vessels.
3. Reproductive effects
In men, small single doses of radiation (200 300 cGy) cause temporary aspermatogenesis, and larger doses (600 800 cGy) may cause permanent sterility. In women, single doses of 200 cGy may cause temporary cessation of menses, and 500 800 cGy may cause permanent castration. Moderate to heavy irradiation of the embryo results in injury to the fetus (eg, mental retardation) or in embryonic death and abortion.
4. Respiratory tract
High or repeated moderate doses of radiation may cause pneumonitis, often delayed for weeks or months.
5. Mouth, pharynx, esophagus, and stomach
Mucositis with edema and painful swallowing of food may occur within hours or days after exposure. Gastric secretion may be inhibited by high doses of radiation.
6. Intestines
Inflammation and ulceration may follow moderately large doses of radiation.
7. Endocrine glands and viscera
Hepatitis and nephritis may be delayed effects of therapeutic radiation. The normal thyroid, pituitary, pancreas, adrenals, and bladder are relatively resistant to low or moderate doses of radiation; parathyroid glands are especially resistant.
8. Nervous system
The brain and spinal cord are much more sensitive to acute exposures than the peripheral nerves.
C. Systemic Reaction (Radiation Sickness)
The basic mechanisms of radiation sickness are not known. Anorexia, nausea, vomiting, weakness, exhaustion, lassitude, and in some cases prostration may occur, singly or in combination. Dehydration, anemia, and infection may follow. Radiation sickness associated
P.1633
with x-ray therapy is most likely to occur when the therapy is given in large dosage to large areas over the abdomen, less often when given over the thorax, and rarely when therapy is given over the extremities.
Prevention
Persons handling radiation sources can minimize exposure to radiation by recognizing the importance of time, distance, and shielding. Areas housing x-ray and nuclear materials must be properly shielded. X-ray equipment should be periodically checked for reliability of output, and proper filters should be used. When feasible, it is advisable to shield the gonads, especially of young persons. Fluoroscopic procedures should be performed as rapidly as possible using an optimal combination of beam characteristics and filtration, and the beam size should be kept to a minimum required by the examination. Special protective clothing may be necessary to protect against contamination with radioisotopes. In the event of accidental contamination, all clothing should be removed and the body vigorously bathed with soap and water. This should be followed by careful instrument (Geiger counter) check to localize the ionizing radiation.
Nuclear Terrorism & Emergency Treatment for Radiation Accident Victims
The proliferation of radiation equipment and nuclear energy plants, terrorism, and the increasing need for transportation of radioactive materials have made necessary hospital plans for managing patients who are accidentally exposed to ionizing radiation or are contaminated with radioisotopes. The plans should provide for effective emergency care and disposition of victims and materials with the least possible risk of spreading radioactive contamination to health care personnel and facilities.
The threat of nuclear terrorism is raising the level of awareness about medical aspects of ionizing radiation exposure. The Gusev and Mettler references (see below) provide detailed guidance regarding decontamination, diagnosis, treatment, and isotope-specific postexposure prophylaxis. The Radiation Assistance Center provides 24-hour access to expert guidance regarding medical and safety aspects of ionizing radiation exposures and incidents (865 576-1005). The Centers for Disease Control and Prevention Radiation Emergency website (http://www.bt.cdc.gov/radiation/index.asp) is also helpful.
Treatment
The success of treatment of local radiation effects depends on the extent, degree, and location of tissue injury. Particulate or radioisotope exposures should be decontaminated in designated confined areas. Serial lymphocyte counts are useful for dose estimation and monitoring of the clinical impact of exposure. For some radioisotopes (eg, iodine), chelation, blocking, or dilution therapy may be indicated. Treatment of systemic reactions is symptomatic and supportive. Blood and platelet transfusions, blood stem cell transplantation, bone marrow transplantation, antibiotics, fluid and electrolyte maintenance, and other supportive measures may be useful. Recombinant hematopoietic colony stimulating factors/cytokines (filgrastim, sargramostim, or pegfilgrastim) have been effective in accelerating hematopoietic recovery.
Delayed effects of Ionizing Radiation
These disorders may occur following excessive ionizing radiation exposure: skin scarring, atrophy, and telangiectasis; cataract, dry eye syndrome, retinopathy; neuropathy, myelopathy, cerebral injury; obliterative endarteritis, coronary artery disease, pericarditis; thyroid disease; pulmonary fibrosis, hepatitis, intestinal stenosis, nephritis, and chromosomal aberrations. Neoplastic disease, including leukemia and cancers of the skin, breast, lung, and thyroid, is increased in persons exposed to radiation at relatively low doses (< 0.2 Gy). High-dose radon exposure is associated with an increased risk of lung cancer. Association of ionizing radiation with several other cancers has been reported but not well quantified (salivary glands, skin, stomach, colon, bladder, ovary, and central nervous system). Prenatal irradiation may increase the risk of childhood cancer.
Microcephaly and other congenital abnormalities may occur in children exposed in utero, especially if the fetus was exposed during early pregnancy. Carcinogenesis from low-dose (50 100 mSv/5 10 rem) exposure to adults probably occurs, but risk is proportional to total dose. Age-related differences in sensitivity to radiation have also resulted in carcinogenesis following childhood exposures (eg, Chernobyl and childhood thyroid cancer).
Footnote
*In radiation terminology, a rad is the unit of absorbed dose and a rem is the unit of any radiation dose to body tissue in terms of its estimated biologic effect. Roentgen (R) refers to the amount of radiation dose delivered to the body. For x-ray or gamma ray radiation, rems, rads, and roentgens are virtually the same. For particulate radiation from radioactive materials, these terms may differ greatly (eg, for neutrons, 1 rad equals 10 rems). In the Syst me International (SI) nomenclature, the rad has been replaced by the gray (Gy), and 1 rad equals 0.01 Gy = 1 cGy. The SI replacement for the rem is the Sievert (Sv), and 1 rem equals 0.01 Sv.
Brenner DJ et al: Cancer risks attributable to low doses of ionizing radiation: Assessing what we really know. Proc Natl Acad Sci U S A 2003;100:13761.
Burnham JW et al: Radiation. Crit Care Clin 2005;21:785.
Koenig KL et al: Medical treatment of radiological casualties: current concepts. Ann Emerg Med 2005;45:643.
Radiation Emergency Assistance Center/Training Site (REACTS): http://www.orau.gov/reacts.
Waselenko JK et al: Strategic National Stockpile Radiation Working Group: Medical management of the acute radiation syndrome: recommendations of the Strategic National Stockpile Radiation Working Group. Ann Intern Med 2004;140:1037.
Drowning
![]() Essentials of Diagnosis
Essentials of Diagnosis
Clinical manifestations of hypoxemia, pulmonary edema, and hypoventilation.
P.1634
General Considerations
The asphyxia of drowning is usually due to aspiration of fluid, but it may result from airway obstruction caused by laryngeal spasm while the victim is gasping under water. The rapid sequence of events after submersion hypoxemia, laryngospasm, fluid aspiration, ineffective circulation, brain injury, and brain death may take place within 5 10 minutes. This sequence may be delayed for longer periods if the victim, especially a child, has been submerged in very cold water or if the victim has ingested significant amounts of barbiturates. About 10% of victims develop laryngospasm after the first gulp and never aspirate water ( dry drowning ). Immersion in cold water can also cause a rapid fall in the victim's core temperature, so that systemic hypothermia and death may occur before actual drowning.
The primary effect is hypoxia due to perfusion of poorly ventilated alveoli, intrapulmonary shunting, and decreased compliance. The first requirement of rescue is immediate CPR.
A number of circumstances or primary events may precede near drowning and must be taken into consideration in management: (1) use of alcohol or other drugs (a contributing factor in an estimated 25% of adult drownings), (2) extreme fatigue, (3) intentional hyperventilation, (4) sudden acute illness (eg, seizure, arrhythmia, myocardial infarction), (5) head or spinal cord injury sustained in diving, (6) venomous stings by aquatic animals, and (7) decompression sickness in deep water diving.
Spontaneous return of consciousness often occurs in otherwise healthy individuals when submersion is very brief. Many other patients respond promptly to immediate ventilation. Other patients, with more severe degrees of near drowning, may have frank respiratory failure, pulmonary edema, shock, anoxic encephalopathy, cerebral edema, and cardiac arrest. A few patients may be deceptively asymptomatic during the recovery period only to deteriorate or die as a result of acute respiratory failure within the following 12 24 hours.
Clinical Findings
A. Symptoms and Signs
The patient may be unconscious, semiconscious, or awake but apprehensive, restless, and complaining of headaches or chest pain. Vomiting is common. Examination may reveal cyanosis, trismus, apnea, tachypnea, and wheezing. A pink froth from the mouth and nose indicates pulmonary edema. Cardiovascular manifestations may include tachycardia, arrhythmias, hypotension, cardiac arrest, and circulatory shock. With prolonged or cold water immersion, hypothermia is likely.
B. Laboratory Findings
Urinalysis shows proteinuria, hemoglobinuria, and acetonuria. Leukocytosis is usually present. The PaO2 is usually decreased and the PaCO2 increased or decreased. The blood pH is decreased as a result of metabolic acidosis. Chest x-rays may show pneumonitis or pulmonary edema.
Prevention
Prevention consists of avoidance of alcohol during recreational swimming or boating, close supervision of toddlers, swimming lessons early in life, and use of personal flotation devices when boating. All swimming pools should be fenced.
Treatment
A. First Aid
Immediate measures to combat hypoxemia at the scene of the incident sustained ventilation, oxygenation, and circulatory support are critical to survival with complete recovery. Hypothermia and cervical spine injury should always be suspected.
Standard CPR is initiated if pulse and respirations are absent.
Do not attempt to drain water from the victim's lungs. The Heimlich maneuver (subdiaphragmatic pressure) should be used only if airway obstruction by a foreign body is suspected. The cervical spine should be immobilized if neck injury is possible.
Do not discontinue basic life support for seemingly hopeless patients until core temperature reaches 32 C. Complete recovery has been reported after prolonged resuscitation of hypothermic patients.
B. Hospital Care
Requirements are careful observation of the patient; continuous monitoring of cardiorespiratory function; maintenance of cerebral oxygenation, serial determination of arterial blood gases, pH, renal function (serum creatinine), and electrolytes; and measurement of urinary output. Pulmonary edema may not appear for 24 hours.
1. Ensure optimal ventilation and oxygenation
The danger of hypoxemia exists even in the alert, conscious patient who appears to be breathing normally. Oxygen should be administered immediately at the highest available concentration. Endotracheal intubation and mechanical ventilation are necessary for patients unable to maintain an open airway or normal blood gases and pH. Nasogastric intubation will allow removal of swallowed water and prevention of aspiration. If the victim does not have spontaneous respirations, intubation is required. Oxygen saturation should be maintained at 90% or higher. Continuous positive airway pressure (CPAP) is the most effective means of reversing hypoxia in patients with spontaneous respirations and patent airways. Positive end-expiratory pressure (PEEP) is also effective for treating respiratory insufficiency. Assisted ventilation may be necessary with pulmonary edema, respiratory failure, aspiration, pneumonia, or severe central nervous system injury. Serial
P.1635
physical examinations and chest x-rays should be carried out to detect possible pneumonitis, atelectasis, and pulmonary edema. Bronchospasm due to aspirated material may require use of bronchodilators. Antibiotics should be given only when there is clinical evidence of infection not prophylactically.
2. Cardiovascular support
Central venous pressure (or, preferably, pulmonary artery wedge pressure) may be monitored as a guide to determining whether vascular fluid replacement and pressors or diuretics are needed. If low cardiac output persists after adequate intravascular volume is achieved, pressors should be given. Otherwise, standard therapy for pulmonary edema is administered.
3. Correction of blood pH and electrolyte abnormalities
Metabolic acidosis is present in 70% of near-drowning victims, but it is usually of minor importance and corrected through adequate ventilation and oxygenation. Glycemic control improves outcome.
4. Cerebral injury
Some near-drowning patients may progress to irreversible central nervous system damage despite apparently adequate treatment of hypoxia and shock. Mild hyperventilation to achieve a PaCO2 of approximately 30 mm Hg is recommended to lower intracranial pressure.
5. Hypothermia
Core temperature should be measured and managed as appropriate (see Systemic Hypothermia, above).
Course & Prognosis
Victims of near drowning who have had prolonged hypoxemia should remain under close hospital observation for 2 3 days after all supportive measures have been withdrawn and clinical and laboratory findings have been stable. Residual complications of near drowning may include intellectual impairment, convulsive disorders, and pulmonary or cardiac disease.
Olshaker JS: Submersion. Emerg Med Clin North Am 2004;22:357.
Salomez F et al: Drowning: a review of epidemiology, pathophysiology, treatment and prevention. Resuscitation 2004;63:261.
Other Disorders due to Physical Agents
Decompression Sickness & Dysbaric Illness
Decompression sickness and other disorders related to rapid changes in environmental pressure are hazards for fliers and for divers who are involved in recreational diving (eg, scuba diving), deep-water exploration, rescue or salvage operations, or construction.
At low depths the greatly increased pressure (eg, at 30 meters [100 feet] the pressure is four times greater than at the surface) compresses the respiratory gases into the blood and other tissues. During ascent from depths greater than 9 meters (30 feet), gases dissolved in the blood and other tissues escape as the external pressure decreases. The appearance of symptoms depends on the depth and duration of submersion; the degree of physical exertion; the age, weight, and physical condition of the diver; and the rate of ascent. The size and number of gas bubbles (notably nitrogen) escaping from the tissues depend on the difference between the atmospheric pressure and the partial pressure of the gas dissolved in the tissues. The release of gas bubbles and (particularly) the location of their release determine the symptoms. Predisposing factors include injury, right to left cardiac shunt, obesity, dehydration, alcoholic excess, hypoxia, some medications (eg, narcotics, antihistamines), and cold.
Decompression sickness also occurs among fliers during rapid ascent from sea level to high altitudes when there is no adequate pressurizing protection. Deep-sea and scuba divers may be vulnerable to air embolism if airplane travel is attempted too soon (within a few hours) after diving. Asthma, pneumothorax, reduced pulmonary function, lung cysts, or thoracic trauma may be contraindications to diving.
Clinical Findings
The range of clinical manifestations includes gas bubble formation in the joints ( bends ), cerebral or pulmonary decompression sickness, arterial gas embolism (cerebral, pulmonary), ear and sinus barotrauma, and dysbaric osteonecrosis. Reported sequelae include hemiparesis, neurologic dysfunction, and bone damage.
The onset of acute decompression symptoms occurs within 30 minutes in half of cases and almost invariably within 6 hours. Symptoms, which are highly variable, include pain (largely in the joints), headache, fatigue, numbness, confusion, pruritic rash, visual disturbances, nausea, vomiting, loss of hearing, weakness, paralysis, dizziness, vertigo, dyspnea, paresthesias, aphasia, and coma.
Pulmonary decompression sickness ( chokes ) presents with burning, pleuritic substernal pain, cough, and dyspnea.
Treatment
Early recognition and prompt treatment are extremely important. Continuous administration of 100% oxygen is indicated as a first aid measure whether or not cyanosis is present. Aspirin may be given for pain, but narcotics should be used very cautiously, since they may obscure the patient's response to recompression. Rapid transportation to a treatment facility for recompression, hyperbaric oxygen, hydration treatment of plasma deficits, and supportive measures is necessary not only to relieve symptoms but
P.1636
also to prevent permanent impairment. It has been recommended, however, that decompression symptoms be treated whenever they are seen even up to 2 weeks post injury since it is still possible to reduce morbidity. The clinician should be familiar with the nearest compression center. The National Divers Alert Network at Duke University (DAN; 919 684-8111 or http://www.diversalertnetwork.org) provides assistance in the management of underwater diving accidents.
Bove AA, Davis J: Diving Medicine, 4th ed. WB Saunders, 2003.
DeGorordo A et al: Diving emergencies. Resuscitation 2003;59:171.
Hamilton-Farrell M et al: Barotrauma. Injury 2004;35:359.
Altitude Illness
![]() Essentials of Diagnosis
Essentials of Diagnosis
Five manifestations of altitude illness are: (1) acute mountain sickness, (2) high-altitude pulmonary edema, (3) high-altitude encephalopathy, (4) subacute mountain sickness, and (5) chronic mountain sickness (Monge's disease).
General Considerations
Lack of sufficient time for acclimatization, increased physical activity, and varying degrees of health may be responsible for the acute, subacute, and chronic disturbances that result from (hypobaric) hypoxia at altitudes greater than 2000 meters (6560 feet). Acclimatization to altitudes above 5500 m (18,045 ft) is incomplete or physiologically impossible, although individual differences in tolerance to hypoxia exist. Altitude exacerbated conditions include cardiac and pulmonary dysfunction and disease, sickle cell, high-risk pregnancy, radial keratotomy, and others.
Acute Mountain Sickness
The severity of acute mountain sickness (AMS) correlates with altitude and rate of ascent. Initial manifestations include headache (most severe and persistent symptom), lassitude, drowsiness, dizziness, chilliness, nausea and vomiting, facial pallor, dyspnea, and cyanosis. Later, there is facial flushing, irritability, difficulty in concentrating, vertigo, tinnitus, visual disturbances, auditory disturbances, anorexia, insomnia, increased dyspnea and weakness on exertion, increased headaches (due to cerebral edema), palpitations, tachycardia, Cheyne-Stokes breathing, and weight loss. More severe manifestations include pulmonary edema and encephalopathy (see below). Voluntary periodic hyperventilation may relieve symptoms. In most individuals, symptoms clear within 24 48 hours, but in some instances, if the symptoms are sufficiently persistent or severe, the patient must be returned to lower altitudes. Definitive treatment is immediate descent, which is essential if reduced consciousness, ataxia, or pulmonary edema occurs. Administration of oxygen, 1 2 L/min, will often relieve acute symptoms. If immediate descent is not possible, portable hyperbaric chambers can provide symptomatic relief depending on altitude and severity. Acetazolamide, 250 mg orally every 8 12 hours, or dexamethasone, 8 mg orally initially followed by 4 mg every 6 hours, for as long as symptoms persist, is recommended therapy; they may be used together in severe cases.
Preventive measures include slow ascent 300 meters (984 feet) per day adequate rest and sleep the day before travel, reduced food intake, and avoidance of alcohol, tobacco, and unnecessary physical activity during travel. Acetazolamide, 250 mg orally every 8 12 hours, beginning the day before ascent and continuing for 48 72 hours at altitude, may be used as prophylaxis. Dexamethasone, 4 mg orally every 12 hours beginning on the day of ascent, continuing for 3 days at the higher altitude, and then tapering over 5 days, is an alternative.
Acute High-Altitude Pulmonary Edema
This serious complication usually occurs at levels above 3000 meters (9840 feet). Early symptoms may appear within 6 36 hours after arrival at a high-altitude area: incessant dry cough, shortness of breath disproportionate to exertion, headache, decreased exercise performance, fatigue, dyspnea at rest, and chest tightness. Later, wheezing, orthopnea, and hemoptysis may occur. Recognition of the early symptoms may enable the patient to descend before incapacitating pulmonary edema develops, but strenuous exertion should be avoided. An early descent of even 500 or 1000 meters may result in improvement of symptoms. Physical findings include tachycardia, mild fever, tachypnea, cyanosis, prolonged respiration, and rales and rhonchi. The patient may become confused or comatose, and the clinical picture may resemble severe pneumonia. The white count is often slightly elevated, but the erythrocyte sedimentation rate is usually normal. Chest x-ray findings vary from irregular patchy infiltration in one lung to nodular densities bilaterally or with transient prominence of the central pulmonary arteries. Transient nonspecific electrocardiographic changes, occasionally showing right ventricular strain, may occur. Pulmonary arterial blood pressure is elevated, whereas wedge pressure is normal.
Treatment, which must often be given under field conditions, consists of rest in the semi-Fowler position (head raised) and administration of 100% oxygen by mask at a rate of 4 6 L/min for 15 30 minutes. Immediate descent (at least 610 meters [2000 feet]) is essential. Recompression in a portable hyperbaric bag will temporarily reduce symptoms if rapid or immediate descent is not possible. To conserve oxygen, lower
P.1637
flow rates (2 4 L/min) may be used until the victim recovers or can be evacuated to a lower altitude and SaO2 90%. Treatment for acute respiratory distress syndrome (see Chapter 9) may be required for some patients who have a prolonged course of pulmonary edema. Nifedipine, 10 mg initially followed by 30 mg slow-release tablets every 12 hours is recommended for symptomatic relief. Dexamethasone, 4 mg every 6 hours, has been recommended if central nervous system symptoms are present. Acetazolamide, 250 mg every 8 12 hours, should be administered if acute mountain sickness is suspected. If bacterial pneumonia occurs, appropriate antibiotic therapy should be given.
Preventive measures include education of prospective mountaineers regarding the possibility of serious pulmonary edema, optimal physical conditioning before travel, gradual ascent to permit acclimatization, and a period of rest and inactivity for 1 2 days after arrival at high altitudes. Salmeterol, 125 mcg by inhaler every 12 hours beginning 24 hours prior to ascent, has been shown to reduce the incidence of high-altitude pulmonary edema (HAPE) in susceptible patients. Prompt medical attention with rest and high-flow oxygen if respiratory symptoms develop may prevent progression to frank pulmonary edema. Persons with a history of HAPE should be hospitalized for further observation if possible. Pulmonary embolism and high-altitude bronchitis can also occur. Mountaineering parties at levels of 3000 meters (9840 feet) or higher should carry a supply of oxygen and equipment sufficient for several days. Persons with symptomatic cardiac or pulmonary disease should avoid high altitudes.
Acute High-Altitude Encephalopathy
High-altitude encephalopathy appears to be an extension of the central nervous system symptoms of AMS and results from cerebral vasogenic edema. It usually occurs at elevations above 2500 meters (8250 feet) and is more common in unacclimatized individuals. Clinical findings are due largely to hypoxemia and cerebral edema. Hallmarks are altered consciousness and ataxic gait; the patient appears mildly drunk. Severe headaches, confusion, truncal ataxia, urinary retention or incontinence, focal deficits, papilledema, nausea, vomiting, and seizures may also occur and progress to obtundation and coma. High-altitude retinopathy is a separate but related effect of altitude. It can include dilated vessels, retinal hemorrhage, vitreous hemorrhage, and papilledema.
Treatment is immediate descent for at least 610 meters (2000 feet), continuing until symptoms improve. One-hundred percent oxygen (2 4 L/min) should be administered by mask. Dexamethasone, 4 8 mg orally every 6 hours, is recommended thereafter. If immediate descent is impossible, a portable hyperbaric chamber should be used until symptomatic improvement occurs.
Prophylaxis using acetazolimide as recommended for AMS is effective.
Subacute Mountain Sickness
This occurs most frequently in unacclimatized individuals and at altitudes above 4500 meters (14,764 feet). Symptoms dyspnea and cough are probably due to hypoxic pulmonary hypertension and secondary congestive heart failure. There are additional problems of dehydration, skin dryness, and pruritus. The hematocrit may be elevated, and there may be electrocardiographic and chest x-ray evidence of right ventricular hypertrophy. Treatment consists of rest, oxygen administration, diuretics, and return to lower altitudes.
Chronic Mountain Sickness (Monge's Disease)
This uncommon condition, consisting of chronic hypoxia, polycythemia and, in some cases, pulmonary hypertension in residents of high-altitude communities who have lost their acclimatization to such an environment, is difficult to differentiate from chronic pulmonary disease. The disorder is characterized by somnolence, mental depression, hypoxemia, cyanosis, clubbing of fingers, hemoglobin > 22 g/dL, polycythemia (hematocrit often > 75%), signs of right ventricular failure, electrocardiographic evidence of right axis deviation and right atrial and ventricular hypertrophy, and x-ray evidence of right heart enlargement and central pulmonary vessel prominence. There is no x-ray evidence of structural pulmonary disease. Pulmonary function tests usually disclose alveolar hypoventilation and elevated PCO2 but fail to reveal defective oxygen transport. There is a diminished respiratory response to CO2. Almost complete disappearance of all abnormalities eventually occurs when the patient returns to sea level.
Otherwise, phlebotomy, oxygen supplementation, respiratory training, medroxyprogesterone (20 60 mg/d orally for 10 weeks), acetazolamide (250 mg/d orally for 3 weeks) and enalapril (5 mg orally daily) have each been recommended for treatment.
Gallagher SA et al: High-altitude illness. Emerg Med Clin North Am 2004;22:329.
Hackett PH et al: High altitude cerebral edema. High Alt Med Biol 2004;5:136.
Leon-Velarde F et al: Consensus statement on chronic and subacute high altitude diseases. High Alt Med Biol 2005;6:147.
Medical effects of Air Travel & Selection of Patients for Air Travel
The medical safety of air travel depends not only on the nature and severity of the traveler's illness but also on such factors as the duration of flight, pressurization, the availability of supplementary oxygen and other medical supplies, the presence of health care professionals, and other special considerations. Airline policies, charges, and other details regarding on-board oxygen and medical supplies must be checked with each carrier. The medical hazard most likely to be realized is hypoxia.
P.1638
The most common in-flight emergencies are cardiovascular, respiratory, syncopal, neuropsychiatric, metabolic, and substance related. The Air Transport Association of America defines an incapacitated passenger as one who is suffering from a physical or mental disability and who, because of such disability or the effect of the flight on the disability, is incapable of self-care; would endanger the health or safety of such person or other passengers or airline employees; or would cause discomfort or annoyance of other passengers.
Table 38-1. More frequent contraindications to air travel. | ||
|---|---|---|
|
Pretravel clinician evaluation is advisable for patients with hospitalization, surgery, chronic disease, emergency care, or substantive medication change within the past 4 weeks.
Patients subject to motion sickness can be given sedatives or antihistamines (eg, dimenhydrinate, 50 mg orally every 4 6 hours; promethazine, 25 mg orally every 6 hours; or meclizine, 25 50 mg orally every 24 hours) before and during the flight. Small meals of easily digested food before and during the flight may reduce the tendency to nausea and vomiting. Table 38-1 lists the more common contraindications to air travel.
Pregnancy
Pregnant women may be permitted to fly during the first 8 months of pregnancy unless there is a history of complications of pregnancy or premature birth. During the ninth month of pregnancy, air travel is not recommended; if travel is essential, a physician's authorization is required. Infants less than 1 week old should not be flown at high altitudes or for long distances.
DeHart RL: Health issues of air travel. Annu Rev Public Health 2003;24:133.
Low JA et al: Air travel in older people. Age Ageing 2002;31:17.
EAN: 2147483647
Pages: 49