28 - Lipid Abnormalities
Editors: McPhee, Stephen J.; Papadakis, Maxine A.; Tierney, Lawrence M.
Title: Current Medical Diagnosis & Treatment, 46th Edition
Copyright 2007 McGraw-Hill
> Table of Contents > 31 - HIV Infection
function show_scrollbar() {}
31
HIV Infection
Andrew R. Zolopa MD
Mitchell H. Katz MD
![]() Essentials of Diagnosis
Essentials of Diagnosis
Risk factors: sexual contact with an infected person, parenteral exposure to infected blood by transfusion or needle sharing, perinatal exposure.
Prominent systemic complaints such as sweats, diarrhea, weight loss, and wasting.
Opportunistic infections due to diminished cellular immunity often life-threatening.
Aggressive cancers, particularly Kaposi's sarcoma and extranodal lymphoma.
Neurologic manifestations, including dementia, aseptic meningitis, and neuropathy.
General Considerations
When AIDS was first recognized in the United States in 1981, cases were identified by finding severe opportunistic infections such as Pneumocystis pneumonia that indicated profound defects in cellular immunity in the absence of other causes of immunodeficiency. When the syndrome was found to be caused by HIV, it became obvious that severe opportunistic infections and unusual neoplasms were at one end of a spectrum of disease, while healthy seropositive individuals were at the other end.
The Centers for Disease Control and Prevention (CDC) AIDS case definition (Table 31-1) includes opportunistic infections and malignancies that rarely occur in the absence of severe immunodeficiency (eg, Pneumocystis pneumonia, central nervous system lymphoma). It also classifies persons as having AIDS if they have positive HIV serology and certain infections and malignancies that can occur in immunocompetent hosts but that are more common among persons infected with HIV (pulmonary tuberculosis, invasive cervical cancer). Several nonspecific conditions, including dementia and wasting (documented weight loss) in the presence of a positive HIV serology are considered AIDS. The definition includes criteria for both definitive and presumptive diagnoses of certain infections and malignancies. Finally, persons with positive HIV serology who have ever had a CD4 lymphocyte count below 200 cells/mcL or a CD4 lymphocyte percentage below 14% are considered to have AIDS. Inclusion of persons with low CD4 counts as AIDS cases reflects the recognition that immunodeficiency is the defining characteristic of AIDS. The choice of a cutoff point at 200 cells/mcL is supported by several cohort studies showing that over 80% of persons with counts below this level will develop AIDS within 3 years in the absence of effective antiretroviral therapy. The 1993 definition was also expanded to include persons with positive HIV serology and pulmonary tuberculosis, recurrent pneumonia, and invasive cervical cancer. Dramatic increases in the efficacy of antiretroviral treatments especially those regimens that include protease inhibitors or nonnucleoside reverse transcriptase inhibitors have improved the prognosis of persons with HIV/AIDS. One consequence is that fewer persons with HIV ever develop an infection or malignancy or have a low enough CD4 count to classify them as having AIDS, which means that the CDC definition has become a less useful measure of the impact of HIV/AIDS in the United States. Conversely, persons in whom AIDS had been diagnosed based on a serious opportunistic infection, malignancy, or immunodeficiency may now be markedly healthier, with high CD4 counts, due to the use of highly active antiretroviral therapy (HAART). Therefore, the Social Security Administration as well as most social service agencies focus on functional assessment for determining eligibility for benefits rather than the simple presence or absence of an AIDS-defined illness.
Clinicians with limited experience in HIV/AIDS should refer HIV-infected patients to specialists with experience, given the increasing number and complexity of treatment regimens available. Extra efforts should be made to obtain specialty consultation for those patients not responding to their current regimens; those intolerant of standard antiviral drugs; those in need of systemic chemotherapy; and those with complicated opportunistic infections, particularly when invasive procedures or experimental therapies are needed. Resources are available to help clinicians care for HIV-infected persons. Clinicians should call their state medical associations for a list of local resources.
Table 31-1. CDC AIDS case definition for surveillance of adults and adolescents. | |
|---|---|
|
Epidemiology
The modes of transmission of HIV are similar to those of hepatitis B, in particular with respect to sexual, parenteral, and vertical transmission. Although certain
P.1347
sexual practices (eg, receptive anal intercourse) are significantly riskier than other sexual practices (eg, oral sex), it is difficult to quantify per-contact risks. The reason is that studies of sexual transmission of HIV show that most people at risk for HIV infection engage in a variety of sexual practices and have sex with multiple persons, only some of whom may actually be HIV infected. Thus, it is difficult to determine which practice with which person actually resulted in HIV transmission.
Nonetheless, the best available estimates indicate that the risk of HIV transmission with receptive anal intercourse is between 1:100 and 1:30, with insertive anal intercourse 1:1000, with receptive vaginal intercourse 1:1000, with insertive vaginal intercourse 1:10,000, and with receptive fellatio with ejaculation 1:1000. The per-contact
P.1348
risk of HIV transmission with other behaviors, including receptive fellatio without ejaculation, insertive fellatio, and cunnilingus, is not known.
All per-contact risk estimates assume that the source is HIV infected. If the HIV status of the source is unknown, the risk of transmission is the risk of transmission multiplied by the probability that the source is HIV infected. This would vary by risk practices, age, and geographic area. A number of cofactors are known to increase the risk of HIV transmission during a given encounter, including the presence of ulcerative or inflammatory sexually transmitted diseases, trauma, menses, and lack of male circumcision.
The risk of acquiring HIV infection from a needlestick with infected blood is approximately 1:300. Factors known to increase the risk of transmission include depth of penetration, hollow bore needles, visible blood on the needle, and advanced stage of disease in the source. The risk of HIV transmission from a mucosal splash with infected blood is unknown but is assumed to be significantly lower.
The risk of acquiring HIV infection from illicit drug use with sharing of needles from an HIV-infected source is estimated to be 1:150. Use of clean needles markedly decreases the chance of HIV transmission but does not eliminate it if other drug paraphernalia are shared (eg, cookers).
When blood transfusion from an HIV-infected donor occurs, the risk of transmission is 95%. Fortunately, since 1985, blood donor screening using the HIV enzyme-linked immunosorbent assay (ELISA) has been universally practiced in the United States. Also, persons who have recently engaged in unsafe behaviors (eg, sex with a person at risk for HIV, injection drug use) are not allowed to donate. This eliminates donations from persons who are HIV infected but have not yet developed antibodies (ie, persons in the window period). In recent years, HIV antigen and viral load testing have been added to the screening of blood to further lower the chance of HIV transmission. With these precautions, the chance of HIV transmission with receipt of blood transfusion is about 1:1,000,000.
In the absence of perinatal HIV prophylaxis, between 13% and 40% of children born to HIV-infected mothers contract HIV infection. The risk is higher with vaginal than with cesarean delivery, higher among mothers with high viral loads, and higher among those who breast-feed their children. The risk can be decreased by administering antiretroviral treatment to the mother during pregnancy and to the infant immediately after birth (see below).
HIV has not been shown to be transmitted by respiratory droplet spread, by vectors such as mosquitoes, or by casual nonsexual contact.
Current estimates are that about 950,000 Americans are infected with HIV. At the end of 2004, there were approximately 415,193 persons in the United States living with AIDS. Fifty-one percent of those are gay or bisexual men, 16% are heterosexual male injection drug users, and 9% are heterosexual male noninjection drug users. Women account for 22% of living persons, of whom 64% were infected through heterosexual contact with an infected partner and 34% were infected through injection drug use.
The rapid increase of AIDS cases among women is of great concern. In 1985, women represented only 7% of new AIDS cases; in 2004, women represented 27% of new cases. African Americans have been disproportionately hard hit by the epidemic. The estimated rate in 2004 of new AIDS cases in the United States per 100,000 population was 56.4 among African Americans, 18.0 among Latinos, 7.9 among American Indians and Alaska Natives, 5.9 among whites, and 3.7 among Asian and Pacific Islanders.
In general, the progression of HIV-related illness is similar in men and women. However, there are some important differences. Women seek medical attention later than men. They are at risk for gynecologic complications of HIV, including recurrent candidal vaginitis, pelvic inflammatory disease, and cervical dysplasia. Violence directed against women, pregnancy, and frequent occurrence of drug use and poverty all complicate the treatment of HIV-infected women. Although safer sex campaigns dramatically decreased the rates of seroconversions among gay men living in metropolitan areas in the United States by the mid-1980s, there is concern that relapse to unsafe sexual practices will result in an increase in the number of new seroconversions. Several studies have reported recent increases in the rates of unsafe sexual behaviors and sexually transmitted diseases among gay men in several large cities in the United States and in western Europe. The higher rates of unsafe sex appear to be related to decreased concern about acquiring HIV due to the availability of HAART. Decreased interest in following safe sex recommendations and increasing use of crystal methamphetamine among certain risk groups also appears to be playing a role in the increased unsafe sex rates.
Worldwide there are nearly 40 million persons infected with HIV. In Central and East Africa in some urban areas, as many as one-third of sexually active adults are infected. HIV infection began to spread in Asia in the late 1980s. The most common mode of transmission is bidirectional heterosexual spread. The reason for the greater risk for transmission with heterosexual intercourse in Africa and Asia than in the United States may relate to cofactors such as general health status, the presence of genital ulcers, relative lack of male circumcision, the number of sexual partners, and different HIV serotypes.
Centers for Disease Control and Prevention: HIV/AIDS Surveillance Reports. Available at http://www.cdc.gov/hiv/topics/surveillance/resources/reports/index.htm.
Katz MH et al: Impact of highly active antiretroviral treatment on HIV seroincidence among men who have sex with men: San Francisco. Am J Public Health 2002;93:388.
Kleinman SH et al: The risks of transfusion-transmitted infection. Baillieres Best Pract Clin Haematol 2000;13:631.
P.1349
Mofenson LM: U.S. Public Health Service Task Force recommendations for use of antiretroviral drugs in pregnant HIV-1 infected women for maternal health and interventions to reduce perinatal HIV-1 transmission in the United States. MMWR Recomm Rep 2002;51(RR-18):1.
Taha TE et al: Nevirapine and zidovudine at birth to reduce perinatal transmission of HIV in an African setting: a randomized controlled trial. JAMA 2004;292:202.
Vittinghoff E: Per-contact risk of human immunodeficiency virus transmission between male sexual partners. Am J Epidemiol 1999;150:306.
Etiology
HIV, like other retroviruses, depends on a unique enzyme, reverse transcriptase (RNA-dependent DNA polymerase), to replicate within host cells. The other major pathogenic human retrovirus, human T cell lymphotropic/leukemia virus (HTLV)-I, is associated with lymphoma, while HIV is not known to be directly oncogenic. The HIV genomes contain genes for three basic structural proteins and at least five other regulatory proteins; gag codes for group antigen proteins, pol codes for polymerase, and env codes for the external envelope protein. The greatest variability in strains of HIV occurs in the viral envelope. Since neutralizing activity is found in antibodies directed against the envelope, this variability presents problems for vaccine development.
In addition to the classic AIDS virus (HIV-1), a group of related viruses, HIV-2, has been isolated in West African patients. HIV-2 has the same genetic organization as HIV-1, but there are significant differences in the envelope glycoproteins. Some infected individuals exhibit AIDS-like illnesses, but most West Africans infected with HIV-2 are currently asymptomatic. HIV-2 has been found in several people in the United States. Thus, this variant may be less pathogenic or have a longer period of latency preceding disease. Cases have been documented in which AIDS-like illnesses have occurred in the absence of HIV infection or other known infectious causes of immunodeficiency.
Pathogenesis
The hallmark of symptomatic HIV infection is immunodeficiency caused by continuing viral replication. The virus can infect all cells expressing the T4 (CD4) antigen, which HIV uses to attach to the cell. Chemokine receptors (CCR5 and CXCR4) are important for virus entry, and individuals with CCR5 deletions are less likely to become infected, and, once infected, the disease is more likely to progress slowly. Once it enters a cell, HIV can replicate and cause cell fusion or death. A latent state is also established, with integration of the HIV genome into the cell's genome. The cell principally infected is the CD4 (helper-inducer) lymphocyte, which directs many other cells in the immune network. With increasing duration of infection, the number of CD4 lymphocytes falls. Some of the immunologic defects, however, are explained not by quantitative abnormalities of lymphocyte subsets but by qualitative defects in CD4 responsiveness induced by HIV.
Other cells in the immune network that are infected by HIV include B lymphocytes and macrophages. The defect in B cells is partly due to disordered CD4 lymphocyte function. These direct and indirect effects can lead to generalized hypergammaglobulinemia and can also depress B cell responses to new antigen challenges. Because of these defects, the immunodeficiency of HIV is mixed. Elements of humoral and cellular immunodeficiency are present, especially in children. Macrophages act as a reservoir for HIV and serve to disseminate it to other organ systems (eg, the central nervous system).
Apart from the immunologic effects of HIV, the virus can also directly cause a variety of neurologic effects. Neuropathology largely results from the release of cytokines and other neurotoxins by infected macrophages. Perturbations of excitatory neurotransmitters and calcium flux may contribute to neurologic dysfunction. Direct HIV infection of renal tubular cells and gastrointestinal epithelium may contribute to these organ system manifestations of infection.
Pathophysiology
Clinically, the syndromes caused by HIV infection are usually explicable by one of three known mechanisms: immunodeficiency, autoimmunity, and allergic and hypersensitivity reactions.
A. Immunodeficiency
Immunodeficiency is a direct result of the effects of HIV upon immune cells. A spectrum of infections and neoplasms is seen, as in other congenital or acquired immunodeficiency states. Two remarkable features of HIV immunodeficiency are the low incidence of certain infections such as listeriosis and aspergillosis and the frequent occurrence of certain neoplasms such as lymphoma or Kaposi's sarcoma. This latter complication has been seen primarily in gay or bisexual men, and its incidence has steadily declined through the first 15 years of the epidemic. A herpesvirus (KSHV or HHV-8) is the cause of Kaposi's sarcoma.
B. Autoimmunity/Allergic & Hypersensitivity Reactions
Autoimmunity can occur as a result of disordered cellular immune function or B lymphocyte dysfunction. Examples of both lymphocytic infiltration of organs (eg, lymphocytic interstitial pneumonitis) and autoantibody production (eg, immunologic thrombocytopenia) occur. These phenomena may be the only clinically apparent disease or may coexist with obvious immunodeficiency. Moreover, HIV-infected individuals appear to have higher rates of allergic reactions to unknown allergens as seen with eosinophilic pustular folliculitis ( itchy red bump syndrome ) as well as increased rates of hypersensitivity reactions to medications (for example, the fever and sunburn-like rash seen with trimethoprim-sulfamethoxazole reactions).
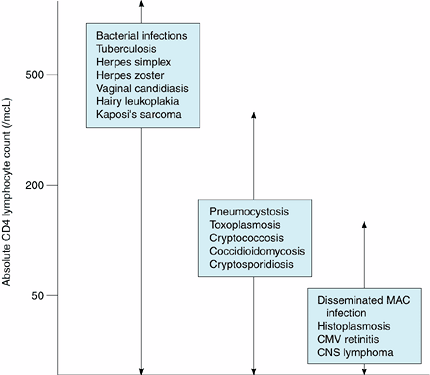 |
Figure 31-1. Relationship of CD4 count to development of opportunistic infections. MAC = Mycobacterium avium complex; CMV = cytomegalovirus; CNS = central nervous system. |
P.1350
Clinical Findings
The complications of HIV-related infections and neoplasms affect virtually every organ. The general approach to the HIV-infected person with symptoms is to evaluate the organ systems involved, aiming to diagnose treatable conditions rapidly. As can be seen in Figure 31-1, the CD4 lymphocyte count provides very important prognostic information. Certain infections may occur at any CD4 count, while others rarely occur unless the CD4 lymphocyte count has dropped below a certain level. For example, a patient with a CD4 count of 600 cells/mcL, cough, and fever may have a bacterial pneumonia but would be very unlikely to have Pneumocystis pneumonia.
A. Symptoms and Signs
Many individuals with HIV infection remain asymptomatic for years even without antiretroviral therapy, with a mean time of approximately 10 years between exposure and development of AIDS. When symptoms occur, they may be remarkably protean and nonspecific. Since virtually all the findings may be seen with other diseases, a combination of complaints is more suggestive of HIV infection than any one symptom.
Physical examination may be entirely normal. Abnormal findings range from completely nonspecific to highly specific for HIV infection. Those that are specific for HIV infection include hairy leukoplakia of the tongue, disseminated Kaposi's sarcoma, and cutaneous bacillary angiomatosis. Generalized lymphadenopathy is common early in infection.
1. Systemic complaints
Fever, night sweats, and weight loss are common symptoms in HIV-infected patients and may occur without a complicating opportunistic infection. Patients with persistent fever and no localizing symptoms should nonetheless be carefully examined, and evaluated with a chest radiograph (Pneumocystis pneumonia can present without respiratory symptoms), bacterial blood cultures if the fever is greater than 38.5 C, serum cryptococcal antigen, and mycobacterial cultures of the blood. Sinus CT scans or sinus radiographs should be considered to evaluate occult sinusitis. If these studies are normal, patients should be observed closely. Antipyretics are useful to prevent dehydration.
Weight loss is a particularly distressing complication of long-standing HIV infection. Patients typically have disproportionate loss of muscle mass, with maintenance or less substantial loss of fat stores. The mechanism of HIV-related weight loss is not completely understood but appears to be multifactorial.
AIDS patients frequently suffer from anorexia, nausea, and vomiting, all of which contribute to weight loss by decreasing caloric intake. In some cases, these symptoms are secondary to a specific infection,
P.1351
such as viral hepatitis. In other cases, however, evaluation of the symptoms yields no specific pathogen, and it is assumed to be due to a primary effect of HIV. Malabsorption also plays a role in decreased caloric intake. Patients may suffer diarrhea from infections with bacterial, viral, or parasitic agents.
Exacerbating the decrease in caloric intake, many AIDS patients have an increased metabolic rate. This increased rate has been shown to exist even among asymptomatic HIV-infected persons, but it accelerates with disease progression and secondary infection. AIDS patients with secondary infections also have decreased protein synthesis, which makes maintaining muscle mass difficult.
Several strategies have been developed to slow AIDS wasting. Effective fever control decreases the metabolic rate and may slow the pace of weight loss, as does treating the underlying opportunistic infection. Food supplementation with high-calorie drinks may enable patients with not much appetite to maintain their intake. Selected patients with otherwise good functional status and weight loss due to unrelenting nausea, vomiting, or diarrhea may benefit from total parenteral nutrition (TPN). It should be noted, however, that TPN is more likely to increase fat stores than to reverse the muscle wasting process.
Two pharmacologic approaches for increasing appetite and weight gain are the progestational agent megestrol acetate (80 mg four times a day) and the antiemetic agent dronabinol (2.5 5 mg three times a day). Side effects from megestrol acetate are rare, but thromboembolic phenomena, edema, nausea, vomiting, and rash have been reported. Euphoria, dizziness, paranoia, and somnolence and even nausea and vomiting have been reported in 3 10% of patients using dronabinol. Dronabinol contains only one of the active ingredients in smoked marijuana, and many patients report better relief of nausea and improvement of appetite with smoking marijuana. Several states allow physicians to recommend the use of smoked marijuana to their patients. However, it is still illegal in the United States to sell marijuana. Thus, a physician's recommendation may at best decrease the chance that patients will be prosecuted for use of marijuana. Unfortunately, neither megestrol acetate nor dronabinol increases lean body mass.
Two regimens that have resulted in increases in lean body mass are growth hormone and anabolic steroids. Growth hormone at a dose of 0.1 mg/kg/d (up to 6 mg) subcutaneously for 12 weeks has resulted in modest increases in lean body mass. Treatment with growth hormone can cost as much as $10,000 per month. Anabolic steroids also increase lean body mass among HIV-infected patients. They seem to work best for patients who are able to do weight training. The most commonly used regimens are testosterone enanthate or testosterone cypionate (100 200 mg intramuscularly every 2 4 weeks). Testosterone transdermal system (apply 5 mg system each evening) and testosterone gel (1%; apply a 5-g packet [50 mg testosterone] to clean, dry skin daily) are also available. The anabolic steroid oxandrolone (20 mg orally in two divided doses) has also been found to increase lean body mass.
Nausea leading to weight loss is sometimes due to esophageal candidiasis. Patients with oral candidiasis and nausea should be empirically treated with an oral antifungal agent. Patients with weight loss due to nausea of unclear origin may benefit from use of antiemetics prior to meals (prochlorperazine, 10 mg three times daily; metoclopramide, 10 mg three times daily; or ondansetron, 8 mg three times daily). Dronabinol (5 mg three times daily) can also be used to increase appetite. Depression and adrenal insufficiency are two potentially treatable causes of weight loss.
2. Sinopulmonary disease
a. Pneumocystis pneumonia
(See also discussions in Chapter 36.) Pneumocystis jiroveci pneumonia is the most common opportunistic infection associated with AIDS. Pneumocystis pneumonia may be difficult to diagnose because the symptoms fever, cough, and shortness of breath are nonspecific. Furthermore, the severity of symptoms ranges from fever and no respiratory symptoms through mild cough or dyspnea to frank respiratory distress.
Hypoxemia may be severe, with a PO2 less than 60 mm Hg. The cornerstone of diagnosis is the chest radiograph. Diffuse or perihilar infiltrates are most characteristic, but only two-thirds of patients with Pneumocystis pneumonia have this finding. Normal chest radiographs are seen in 5 10% of patients with Pneumocystis pneumonia, while the remainder have atypical infiltrates. Apical infiltrates are commonly seen among patients with Pneumocystis pneumonia who have been receiving aerosolized pentamidine prophylaxis. Large pleural effusions are uncommon with Pneumocystis pneumonia; their presence suggests bacterial pneumonia, other infections such as tuberculosis, or pleural Kaposi's sarcoma.
Definitive diagnosis can be obtained in 50 80% of cases by Wright-Giemsa stain or direct fluorescence antibody (DFA) test of induced sputum. Sputum induction is performed by having patients inhale an aerosolized solution of 3% saline produced by an ultrasonic nebulizer. Patients should not eat for at least 8 hours and should not use toothpaste or mouthwash prior to the procedure since they can interfere with test interpretation. The next step for patients with negative sputum examinations still suspected of having Pneumocystis pneumonia should be bronchoalveolar lavage. This technique establishes the diagnosis in over 95% of cases.
In patients with symptoms suggestive of Pneumocystis pneumonia but with negative or atypical chest radiographs and negative sputum examinations, other diagnostic tests may provide additional information in deciding whether to proceed to bronchoalveolar lavage. Elevation of serum lactate dehydrogenase occurs in 95% of cases of Pneumocystis pneumonia, but the
P.1352
specificity of this finding is at best 75%. Either a normal diffusing capacity of carbon monoxide (DLCO) or a high-resolution CT scan of the chest that demonstrates no interstitial lung disease makes the diagnosis of Pneumocystis pneumonia very unlikely. In addition, a CD4 count above 250 cells/mcL within 2 months prior to evaluation of respiratory symptoms makes a diagnosis of Pneumocystis pneumonia unlikely; only 1 5% of cases occur above this CD4 count level (Figure 31-1). This is true even if the patient previously had a CD4 count lower than 200 cells/mcL but has had an increase with antiretroviral therapy. Pneumothoraces can be seen in HIV-infected patients with a history of Pneumocystis pneumonia, especially if they have received aerosolized pentamidine treatment.
b. Other infectious pulmonary diseases
Other infectious causes of pulmonary disease in AIDS patients include bacterial, mycobacterial, and viral pneumonias. Community-acquired pneumonia is the most common cause of pulmonary disease in HIV-infected persons. An increased incidence of pneumococcal pneumonia with septicemia and Haemophilus influenzae pneumonia has been reported. Pseudomonas aeruginosa is an important respiratory pathogen in advanced disease. The incidence of infection with Mycobacterium tuberculosis has markedly increased in metropolitan areas because of HIV infection as well as homelessness. Tuberculosis occurs in an estimated 4% of persons in the United States who have AIDS. Apical infiltrates and disseminated disease occur more commonly than among immunocompetent hosts. Although a purified protein derivative (PPD) test should be performed on all HIV-infected persons in whom a diagnosis of tuberculosis is being considered, the lower the CD4 cell count, the greater the likelihood of anergy. Because anergy skin test panels do not accurately classify those patients who are infected with tuberculosis but unreactive to the PPD, they are not recommended. Treatment of HIV-infected persons with active tuberculosis is similar to treatment of HIV-uninfected tubercular individuals (see Figure 31-1). However, rifampin should not be given to patients receiving indinavir, nelfinavir, amprenavir, lopinavir, or delavirdine. In these cases, rifabutin may be substituted, but it may require dosing modifications depending on the antiretroviral regimen. Multidrug-resistant tuberculosis is a major problem in several metropolitan areas. Noncompliance with prescribed antituberculous drugs is a major risk factor. Several of the reported outbreaks appear to implicate nosocomial spread. The emergence of drug resistance makes it essential that antibiotic sensitivities be performed on all positive cultures. Drug therapy should be individualized. Patients with multidrug-resistant M tuberculosis infection should receive at least three drugs to which their organism is sensitive. Atypical mycobacteria can cause pulmonary disease in AIDS patients with or without preexisting lung disease and responds variably to treatment. Making a distinction between M tuberculosis and atypical mycobacteria requires culture of sputum specimens. If culture of the sputum produces acid-fast bacilli, definitive identification may take several weeks using traditional techniques. DNA probes allow for presumptive identification usually within days of a positive culture. While awaiting definitive diagnosis, clinicians should err on the side of treating patients as if they have M tuberculosis infection. In cases in which the risk of atypical mycobacteria is very high (eg, a person without risk for tuberculosis exposure with a CD4 count under 50 cells/mcL see Figure 31-1), clinicians may wait for definitive diagnosis if the person is smear-negative for acid-fast bacilli, clinically stable, and not living in a communal setting. Isolation of cytomegalovirus (CMV) from bronchoalveolar lavage fluid occurs commonly in AIDS patients but does not establish a definitive diagnosis. Diagnosis of CMV pneumonia requires biopsy; response to treatment is poor. Histoplasmosis, coccidioidomycosis, and cryptococcal disease should also be considered in the differential diagnosis of unexplained pulmonary infiltrates.
c. Noninfectious pulmonary diseases
Noninfectious causes of lung disease include Kaposi's sarcoma, non-Hodgkin's lymphoma, and interstitial pneumonitis. In patients with known Kaposi's sarcoma, pulmonary involvement complicates the course in approximately one-third of cases. However, pulmonary involvement is rarely the presenting manifestation of Kaposi's sarcoma. Non-Hodgkin's lymphoma may involve the lung as the sole site of disease but more commonly involves other organs as well, especially the brain, liver, and gastrointestinal tract. Both of these processes may show nodular or diffuse parenchymal involvement, pleural effusions, and mediastinal adenopathy on chest radiographs.
Nonspecific interstitial pneumonitis may mimic Pneumocystis pneumonia. Lymphocytic interstitial pneumonitis seen in lung biopsies has a variable clinical course. Typically, these patients present with several months of mild cough and dyspnea; chest radiographs show interstitial infiltrates. Many patients with this entity undergo transbronchial biopsies in an attempt to diagnose Pneumocystis pneumonia. Instead, the tissue shows interstitial inflammation ranging from an intense lymphocytic infiltration (consistent with lymphoid interstitial pneumonitis) to a mild mononuclear inflammation. Corticosteroids may be helpful in some cases refractory to antiretroviral therapy.
d. Sinusitis
Chronic sinusitis can be a frustrating problem for HIV-infected patients even in those on adequate antiretroviral therapy. Symptoms include sinus congestion and discharge, headache, and fever. Some patients may have radiographic evidence of sinus disease on sinus CT scan or sinus x-ray in the absence of significant symptoms. Nonsmoking patients with purulent drainage should be treated with amoxicillin (500 mg orally three times a day). Patients who smoke should be treated with amoxicillin-potassium clavulanate (500 mg orally three times a day) to cover
P.1353
H influenzae. Prolonged treatment (3 6 weeks) with an antibiotic and guaifenesin (600 mg orally twice daily) to decrease sinus congestion may be required. For patients not responding to amoxicillin-potassium clavulanate, levofloxacin should be tried (400 mg orally daily). Some patients may require referral to an otolaryngologist for sinus drainage.
3. Central nervous system disease
Central nervous system disease in HIV-infected patients can be divided into intracerebral space-occupying lesions, encephalopathy, meningitis, and spinal cord processes. Many of these complications have declined markedly in prevalence in the era of HAART.
a. Toxoplasmosis
Toxoplasmosis is the most common space-occupying lesion in HIV-infected patients. Headache, focal neurologic deficits, seizures, or altered mental status may be presenting symptoms. The diagnosis is usually made presumptively based on the characteristic appearance of cerebral imaging studies in an individual known to be seropositive for Toxoplasma. Typically, toxoplasmosis appears as multiple contrast-enhancing lesions on CT scan. Lesions tend to be peripheral, with a predilection for the basal ganglia.
Single lesions are atypical of toxoplasmosis. When a single lesion has been detected by CT scanning, MRI scanning may reveal multiple lesions because of its greater sensitivity. If a patient has a single lesion on MRI and is neurologically stable, clinicians may pursue a 2-week empiric trial of toxoplasmosis therapy. A repeat scan should be performed at 2 weeks. If the lesion has not diminished in size, biopsy of the lesion should be performed. Since many HIV-infected patients will have detectable titers, a positive Toxoplasma serologic test does not confirm the diagnosis. Conversely, less than 3% of patients with toxoplasmosis have negative titers. Therefore, negative Toxoplasma titers in an HIV-infected patient with a space-occupying lesion should be a cause for aggressively pursuing an alternative diagnosis.
b. Central nervous system lymphoma
Primary non-Hodgkin's lymphoma is the second most common space-occupying lesion in HIV-infected patients. Symptoms are similar to those with toxoplasmosis. While imaging techniques cannot distinguish these two diseases with certainty, lymphoma more often is solitary. Other less common lesions should be suspected if there is preceding bacteremia, positive tuberculin test, fungemia, or injection drug use. These include bacterial abscesses, cryptococcomas, tuberculomas, and Nocardia lesions.
Because techniques for stereotactic brain biopsy have improved, this procedure plays an increasing role in diagnosing cerebral lesions. Biopsy should be strongly considered if lesions are solitary or do not respond to toxoplasmosis treatment, especially if they are easily accessible. Diagnosis of lymphoma is important because many patients benefit from treatment (radiation therapy). In the future, it may be possible to avoid brain biopsy by utilizing polymerase chain reaction (PCR) assay of cerebrospinal fluid for Epstein-Barr virus DNA, which is present in 90% of cases.
c. AIDS dementia complex
The diagnosis of AIDS dementia complex (HIV-associated cognitive-motor complex) is one of exclusion based on a brain imaging study and on spinal fluid analysis that excludes other pathogens. Neuropsychiatric testing is helpful in distinguishing patients with dementia from those with depression. Patients with AIDS dementia complex typically have difficulty with cognitive tasks and exhibit diminished motor speed. Patients may first notice a deterioration in their handwriting. The manifestations of dementia may wax and wane, with persons exhibiting periods of lucidity and confusion over the course of a day. Many patients improve with effective antiretroviral treatment. Metabolic abnormalities may also cause changes in mental status: hypoglycemia, hyponatremia, hypoxia, and drug overdose are important considerations in this population. Other less common infectious causes of encephalopathy include progressive multifocal leukoencephalopathy (discussed below), CMV, syphilis, and herpes simplex encephalitis.
d. Cryptococcal meningitis
Cryptococcal meningitis typically presents with fever and headache. Less than 20% of patients have meningismus. Diagnosis is based on a positive latex agglutination test that detects cryptococcal antigen (or CRAG ) or positive culture of spinal fluid for Cryptococcus. Seventy to 90% of patients with cryptococcal meningitis have a positive serum CRAG. Thus, a negative serum CRAG test makes a diagnosis of cryptococcal meningitis unlikely and can be useful in the initial evaluation of a patient with headache, fever, and normal mental status. HIV meningitis, characterized by lymphocytic pleocytosis of the spinal fluid with negative culture, is common early in HIV infection.
e. HIV myelopathy
Spinal cord function may also be impaired in HIV-infected individuals. HIV myelopathy presents with leg weakness and incontinence. Spastic paraparesis and sensory ataxia are seen on neurologic examination. Myelopathy is usually a late manifestation of HIV disease, and most patients will have concomitant HIV encephalopathy. Pathologic evaluation of the spinal cord reveals vacuolation of white matter. Because HIV myelopathy is a diagnosis of exclusion, symptoms suggestive of myelopathy should be evaluated by lumbar puncture to rule out CMV polyradiculopathy (described below) and an MRI or CT scan to exclude epidural lymphoma.
f. Progressive multifocal leukoencephalopathy (PML)
PML is a viral infection of the white matter of the brain seen in patients with very advanced HIV infection. It typically results in focal neurologic deficits such as aphasia, hemiparesis, and cortical blindness. Imaging studies are strongly suggestive of the diagnosis if they show nonenhancing white matter lesions without mass effect. Extensive lesions may be difficult to differentiate
P.1354
from the changes caused by HIV. Several patients have stabilized or improved after the institution of combination antiretroviral therapy or cidofovir.
4. Peripheral nervous system
Peripheral nervous system syndromes include inflammatory polyneuropathies, sensory neuropathies, and mononeuropathies.
An inflammatory demyelinating polyneuropathy similar to Guillain-Barr syndrome occurs in HIV-infected patients, usually prior to frank immunodeficiency. The syndrome in many cases improves with plasmapheresis, supporting an autoimmune basis of the disease. CMV can cause an ascending polyradiculopathy characterized by lower extremity weakness and a neutrophilic pleocytosis on spinal fluid analysis with a negative bacterial culture. Transverse myelitis can be seen with herpes zoster or CMV.
Peripheral neuropathy is common among HIV-infected persons. Patients typically complain of numbness, tingling, and pain in the lower extremities. Symptoms are disproportionate to findings on gross sensory and motor evaluation. Beyond HIV infection itself, the most common cause is prior antiretroviral therapy with stavudine or didanosine. Patients who report these symptoms should be switched to an alternative agent if possible. Caution should be used when administering these agents to patients with a history of peripheral neuropathy. Unfortunately, drug-induced neuropathy is not always reversed when the offending agent is discontinued. Patients with advanced disease may also develop peripheral neuropathy even if they have never taken antiretroviral therapy. Evaluation should rule out other causes of sensory neuropathy such as alcoholism, thyroid disease, vitamin B12 deficiency, and syphilis.
Treatment of peripheral neuropathy is aimed at symptomatic relief. Patients should be initially treated with gabapentin (start at 300 mg at bedtime and increase to 300 900 mg orally three times a day). Although many clinicians initiate a trial of amitriptyline (10 25 mg orally at bedtime), responses to this agent are uncommon.
5. Rheumatologic manifestations
Arthritis, involving single or multiple joints, with or without effusion, has been commonly noted in HIV-infected patients. Involvement of large joints is most common. Although the cause of HIV-related arthritis is unknown, most patients will respond to nonsteroidal anti-inflammatory agents. Patients with a sizable effusion, especially if the joint is warm or erythematous, should have the joint tapped, followed by culture of the fluid to rule out suppurative arthritis as well as fungal and mycobacterial disease.
Several rheumatologic syndromes, including reactive arthritis (Reiter's syndrome), psoriatic arthritis, sicca syndrome, and systemic lupus erythematosus, have been reported in HIV-infected patients (see Chapter 20). However, it is unclear if the prevalence is greater than in the general population. Cases of avascular necrosis of the femoral heads have been reported sporadically, generally in the setting of advanced disease with long-standing infection and in patients receiving long-term antiretroviral therapy. The etiology is not clear but is probably multifactoral in nature.
6. Myopathy
Myopathies are increasingly noted in HIV-infected patients. Proximal muscle weakness is typical, and patients may have varying degrees of muscle tenderness. The most important clinical distinction is between myopathy due to the primary effect of HIV and that due to zidovudine. Patients with symptomatic myopathy, especially with creatine kinase levels greater than 1000 units/L, should have their dose of zidovudine decreased or stopped and should be considered for alternative antiviral therapy. A muscle biopsy can distinguish HIV myopathy from zidovudine myopathy and should be considered in patients for whom continuation of zidovudine is essential.
7. Retinitis
Complaints of visual changes must be evaluated immediately in HIV-infected patients. CMV retinitis, characterized by perivascular hemorrhages and white fluffy exudates, is the most common retinal infection in AIDS patients and can be rapidly progressive. In contrast, cotton wool spots, which are also common in HIV-infected people, are benign, remit spontaneously, and appear as small indistinct white spots without exudation or hemorrhage. This distinction may be difficult at times for the nonspecialist, and patients with visual changes should be seen by an ophthalmologist. Other rare retinal processes include other herpesvirus infections or toxoplasmosis.
8. Oral lesions
The presence of oral candidiasis or hairy leukoplakia is significant for several reasons. First, these lesions are highly suggestive of HIV infection in patients who have no other obvious cause of immunodeficiency. Second, several studies have indicated that patients with candidiasis have a high rate of progression to AIDS even with statistical adjustment for CD4 count.
Hairy leukoplakia is caused by the Epstein-Barr virus. The lesion is not usually troubling to patients and sometimes regresses spontaneously. Hairy leukoplakia is commonly seen as a white lesion on the lateral aspect of the tongue. It may be flat or slightly raised, is usually corrugated, and has vertical parallel lines with fine or thick ( hairy ) projections. Oral candidiasis can be bothersome to patients, many of whom report an unpleasant taste or mouth dryness. There are two major types of oral candidiasis: pseudomembranous (removable white plaques) and erythematous (red friable plaques). Treatment is with topical agents such as clotrimazole 10-mg troches (one troche four or five times a day). Patients with candidiasis who do not respond to topical antifungals can be treated with fluconazole (50 100 mg orally once a day for 3 7 days). Chronic suppression of oral candidiasis with fluconazole has been associated with development of candidiasis resistant to all available azoles and thus should be avoided except in frequently recurring cases.
P.1355
Angular cheilitis fissures at the sides of the mouth is usually due to Candida as well and can be treated topically with ketoconazole cream (2%) twice a day.
Gingival disease is common in HIV-infected patients and is thought to be due to an overgrowth of microorganisms. It usually responds to professional dental cleaning and chlorhexidine rinses. A particularly aggressive gingivitis or periodontitis will develop in some HIV-infected patients; these patients should be given antibiotics that cover anaerobic oral flora (eg, metronidazole, 250 mg four times a day for 4 or 5 days) and referred to oral surgeons with experience with these entities.
Aphthous ulcers are painful and may interfere with eating. They can be treated with fluocinonide (0.05% ointment mixed 1:1 with plain Orabase and applied six times a day to the ulcer). For lesions that are difficult to reach, patients should use dexamethasone swishes (0.5 mg in 5 mL elixir three times a day). The pain of the ulcers can be relieved with use of an anesthetic spray (10% lidocaine). For patients with refractory ulcers, thalidomide, starting at a dose of 50 mg orally daily and increasing to 100 200 mg daily, has proved useful. It should be administered only to patients at zero risk of procreation. The most common side effects are sedation and peripheral neuropathy. Other lesions seen in the mouths of HIV-infected patients include Kaposi's sarcoma (usually on the hard palate) and warts.
9. Gastrointestinal manifestations
a. Candidal esophagitis
(See also discussion in Chapter 14.) Esophageal candidiasis is a common AIDS infection. In a patient with characteristic symptoms, empiric antifungal treatment is begun with fluconazole (200 mg daily for 10 14 days). Further evaluation to identify other causes of esophagitis (herpes simplex, CMV) is reserved for patients who do not improve with treatment.
b. Hepatic disease
Autopsy studies have demonstrated that the liver is a frequent site of infections and neoplasms in HIV-infected patients. However, many of these infections are not clinically symptomatic. Clinicians may note elevations of alkaline phosphatase and aminotransferases on routine chemistry panels. Mycobacterial disease, CMV, hepatitis B virus, hepatitis C virus, and lymphoma cause liver disease and can present with varying degrees of nausea, vomiting, right upper quadrant abdominal pain, and jaundice. Sulfonamides, imidazole drugs, antituberculous medications, pentamidine, clarithromycin, and didanosine have also been associated with hepatitis. HIV-infected patients with chronic hepatitis may have more rapid progression of liver disease because of the concomitant immunodeficiency or hepatotoxicity of antiretroviral therapy. Percutaneous liver biopsy may be helpful in diagnosing liver disease, but some common causes of liver disease (eg, Mycobacterium avium complex, lymphoma) can be determined by less invasive measures (eg, blood culture, biopsy of a more accessible site). With patients living longer as a result of advances in antiretroviral therapy, advanced liver disease and hepatic failure due to chronic active hepatitis B and or C are increasing causes of morbidity and mortality. Treatment of HIV-infected persons with hepatitis B and C with peginterferon has been shown to be efficacious, although less so than in HIV-uninfected persons. HIV-infected persons are also more likely to have difficulty tolerating treatment with peginterferon than uninfected persons. Liver transplants have been performed successfully in HIV-infected patients. This strategy is most likely to be successful in persons who have CD4 counts above 100 cells/mcL and nondetectable viral loads.
c. Biliary disease
Cholecystitis presents with manifestations similar to those seen in immunocompetent hosts but is more likely to be acalculous. Sclerosing cholangitis and papillary stenosis have also been reported in HIV-infected patients. Typically, the syndrome presents with severe nausea, vomiting, and right upper quadrant pain. Liver function tests generally show alkaline phosphatase elevations disproportionate to elevation of the aminotransferases. Although dilated ducts can be seen on ultrasound, the diagnosis is made by endoscopic retrograde cholangiopancreatography, which reveals intraluminal irregularities of the proximal intrahepatic ducts with pruning of the terminal ductal branches. Stenosis of the distal common bile duct at the papilla is commonly seen with this syndrome. CMV, Cryptosporidium, and microsporidia are thought to play inciting roles in this syndrome.
d. Enterocolitis
Enterocolitis is a common problem in HIV-infected individuals. Organisms known to cause enterocolitis include bacteria (Campylobacter, Salmonella, Shigella), viruses (CMV, adenovirus), and protozoans (Cryptosporidium, Entamoeba histolytica, Giardia, Isospora, microsporidia). HIV itself may cause enterocolitis. Several of the organisms causing enterocolitis in HIV-infected individuals also cause diarrhea in immunocompetent hosts. However, HIV-infected patients tend to have more severe and more chronic symptoms, including high fevers and severe abdominal pain that can mimic acute abdominal catastrophes. Bacteremia and concomitant biliary involvement are also more common with enterocolitis in HIV-infected patients. Relapses of enterocolitis following adequate therapy have been reported with both Salmonella and Shigella infections.
Because of the wide range of agents known to cause enterocolitis, a stool culture and multiple stool examinations for ova and parasites (including modified acid-fast staining for Cryptosporidium) should be performed. Those patients who have Cryptosporidium in one stool with improvement in symptoms in less than 1 month should not be considered to have AIDS, as Cryptosporidium is a cause of self-limited diarrhea in HIV-negative persons. More commonly, HIV-infected patients with Cryptosporidium have persistent enterocolitis with profuse watery diarrhea.
P.1356
To date, no consistently effective treatments have been developed for Cryptosporidium infection. The most effective treatment of cryptosporidiosis is to improve immune function through the use of effective antiretroviral treatment. The diarrhea can be treated symptomatically with diphenoxylate with atropine (one or two tablets orally three or four times a day). Those who do not respond may be given paregoric with bismuth (5 10 mL orally three or four times a day). Octreotide in escalating doses (starting at 0.05 mg subcutaneously every 8 hours for 48 hours) has been found to ameliorate symptoms in approximately 40% of patients with cryptosporidia or idiopathic HIV-associated diarrhea.
Patients with a negative stool examination and persistent symptoms should be evaluated with colonoscopy and biopsy. Patients whose symptoms last longer than 1 month with no identified cause of diarrhea are considered to have a presumptive diagnosis of AIDS enteropathy. Patients may respond to institution of effective antiretroviral treatment. Upper endoscopy with small bowel biopsy is not recommended as a routine part of the evaluation.
e. Other disorders
Two other important gastrointestinal abnormalities in HIV-infected patients are gastropathy and malabsorption. It has been documented that some HIV-infected patients do not produce normal levels of stomach acid and therefore are unable to absorb drugs that require an acid medium. This decreased acid production may explain, in part, the susceptibility of HIV-infected patients to Campylobacter, Salmonella, and Shigella, all of which are sensitive to acid concentration. There is no evidence that Helicobacter pylori is more common in HIV-infected persons.
A malabsorption syndrome occurs commonly in HIV-infected patients. It can be due to infection of the small bowel with M avium complex, Cryptosporidium, or microsporidia.
10. Endocrinologic manifestations
Hypogonadism is probably the most common endocrinologic abnormality in HIV-infected men. The adrenal gland is also a commonly afflicted endocrine gland in patients with AIDS. Abnormalities demonstrated on autopsy include infection (especially with CMV and M avium complex), infiltration with Kaposi's sarcoma, and injury from hemorrhage and presumed autoimmunity. The prevalence of clinically significant adrenal insufficiency is low. Patients with suggestive symptoms should undergo a cosyntropin stimulation test.
Although frank deficiency of cortisol is rare, an isolated defect in mineralocorticoid metabolism may lead to salt-wasting and hyperkalemia. Such patients should be treated with fludrocortisone (0.1 0.2 mg daily).
AIDS patients appear to have abnormalities of thyroid function tests different from those of patients with other chronic diseases. AIDS patients have been shown to have high levels of triiodothyronine (T3), thyroxine (T4), and thyroid-binding globulin and low levels of reverse triiodothyronine (rT3). The causes and clinical significance of these abnormalities are unknown.
11. Skin manifestations
HIV-infected patients commonly develop skin manifestations that can be grouped into viral, bacterial, fungal, neoplastic, and nonspecific dermatitides.
Herpes simplex infections occur more frequently, tend to be more severe, and are more likely to disseminate in AIDS patients than in immunocompetent persons. Because of the risk of progressive local disease, all herpes simplex attacks should be treated with acyclovir (400 mg orally three times a day until healed, usually 7 days), famciclovir (500 mg orally twice daily until healed), or valacyclovir (500 mg orally twice daily until healed). To avoid the complications of attacks, many clinicians recommend suppressive therapy for HIV-infected patients with a history of recurrent herpes. Options for suppressive therapy include acyclovir (400 mg orally twice daily), famciclovir (250 mg orally twice daily), and valacyclovir (500 mg orally daily).
Herpes zoster is a common manifestation of HIV infection. As with herpes simplex infections, patients with zoster should be treated with acyclovir to prevent dissemination (800 mg orally four or five times per day for 7 days). Alternatively, famciclovir (500 mg orally three times a day) or valacyclovir (500 mg three times a day) may be used. Vesicular lesions should be cultured if there is any question about their origin, since herpes simplex responds to much lower doses of acyclovir. Disseminated zoster and cases with ocular involvement should be treated with intravenous (10 mg/kg every 8 hours for 7 10 days) rather than oral acyclovir.
Molluscum contagiosum caused by a pox virus is seen in HIV-infected patients, as in other immunocompromised patients. The characteristic umbilicated fleshy papular lesions have a propensity for spreading widely over the patient's face and neck and should be treated with topical liquid nitrogen.
Staphylococcus is the most common bacterial cause of skin disease in HIV-infected patients; it usually presents as folliculitis, superficial abscesses (furuncles), or bullous impetigo. Because dissemination with sepsis has been reported, attempts should be made to treat these lesions aggressively. Folliculitis is initially treated with topical clindamycin or mupirocin, and patients may benefit from regular washing with an antibacterial soap such as chlorhexidine. Intranasal mupirocin has been used successfully for staphylococcal decolonization in other settings. In HIV-infected patients with recurrent staphylococcal infections, weekly intranasal mupirocin should be considered in addition to topical care and systemic antibiotics. Abscesses often require incision and drainage. Patients may need antistaphylococcal antibiotics as well. Due to high frequency of methicillin-resistant Staphylococcus aureus (MRSA) skin infections in HIV-infected populations,
P.1357
lesions should be cultured prior to initiating empiric antistaphylococcal therapy. Although there is limited experience treating MRSA with oral antibiotics, current recommendations for empiric treatment are trimethoprim-sulfamethoxazole (one double-strength tablet orally twice daily) or doxycycline (100 mg orally twice daily) with close follow-up.
Bacillary angiomatosis is a well-described entity in HIV-infected patients. It is caused by two closely related organisms: Bartonella henselae and Bartonella quintana. The epidemiology of these infections suggests zoonotic transmission from fleas of infected domestic cats. The most common manifestation is raised, reddish, highly vascular skin lesions that can mimic the lesions of Kaposi's sarcoma. Fever is a common manifestation of this infection; involvement of bone, lymph nodes, and liver has also been reported. The infection responds to doxycycline, 100 mg orally twice daily, or erythromycin, 250 mg orally four times daily. Therapy is continued for at least 14 days, and patients who are seriously ill with visceral involvement may require months of therapy.
The majority of fungal rashes afflicting AIDS patients are due to dermatophytes and Candida. These are particularly common in the inguinal region but may occur anywhere on the body. Fungal rashes generally respond well to topical clotrimazole (1% twice a day) or ketoconazole (2% twice a day).
Seborrheic dermatitis is more common in HIV-infected patients. Scrapings of seborrhea have revealed Malassezia furfur (Pityrosporum ovale), implying that the seborrhea is caused by this fungus. Consistent with the isolation of this fungus is the clinical finding that seborrhea responds well to topical clotrimazole (1% cream) as well as hydrocortisone (1% cream).
Xerosis presents in HIV-infected patients with severe pruritus. The patient may have no rash, or nonspecific excoriations from scratching. Treatment is with emollients (eg, absorption base cream) and antipruritic lotions (eg, camphor 9.5% and menthol 0.5%).
Psoriasis can be very severe in HIV-infected patients. Phototherapy and etretinate (0.25 9.75 mg/kg/d orally in divided doses) may be used for recalcitrant cases in consultation with a dermatologist. Because of the underlying immunodeficiency, methotrexate should be avoided.
12. HIV-related malignancies
Four cancers are currently included in the CDC classification of AIDS: Kaposi's sarcoma, non-Hodgkin's lymphoma, primary lymphoma of the brain, and invasive cervical carcinoma. Epidemiologic studies have shown that between 1973 and 1987 among single men in San Francisco, the risk of Kaposi's sarcoma increased more than 5000-fold and the risk of non-Hodgkin's lymphoma more than tenfold. The increase in incidence of malignancies is probably a function of impaired cell-mediated immunity.
Kaposi's sarcoma lesions may appear anywhere; careful examination of the eyelids, conjunctiva, pinnae, palate, and toe webs is mandatory to locate potentially occult lesions. In light-skinned individuals, Kaposi's lesions usually appear as purplish, nonblanching lesions that can be papular or nodular. In dark-skinned individuals, the lesions may appear more brown. In the mouth, lesions are most often palatal papules, though exophytic lesions of the tongue and gingivae may also be seen. Kaposi's lesions may be confused with other vascular lesions such as angiomas and pyogenic granulomas. Visceral disease (eg, gastrointestinal, pulmonary) will develop in about 40% of patients with dermatologic Kaposi's sarcoma. Rapidly progressive dermatologic or visceral disease is best treated with systemic chemotherapy. Liposomally encapsulated doxorubicin given intravenously every 3 weeks has a response rate of approximately 70%. -Interferon (10 million units subcutaneously three times a week) also has activity against Kaposi's sarcoma. However, symptoms such as malaise and anorexia limit the utility of this therapy. Patients with milder forms of Kaposi's sarcoma do not require specific treatment as the lesions usually improve and can completely resolve with antiretroviral therapy. However, it should be noted that the lesions may flare when antiretroviral therapy is first initiated probably as a result of an immune reconstitution process (see Inflammatory reactions below).
Non-Hodgkin's lymphoma in HIV-infected persons tends to be very aggressive. The malignancies are usually of B cell origin and characterized as diffuse large-cell tumors. Over 70% of the malignancies are extranodal.
The prognosis of patients with systemic non-Hodgkin's lymphoma depends primarily on the degree of immunodeficiency at the time of diagnosis. Patients with high CD4 counts do markedly better than those diagnosed at a late stage of illness. Patients with primary central nervous system lymphoma are treated with radiation. Response to treatment is good, but prior to the availability of HAART, most patients died within a few months after diagnosis due to their underlying disease. Systemic disease is treated with chemotherapy. Common regimens are CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone) and modified M-BACOD (methotrexate, bleomycin, doxorubicin, cyclophosphamide, vincristine, and dexamethasone). Granulocyte colony-stimulating factor (G-CSF; filgrastim) is used to maintain white blood counts with this latter regimen. Intrathecal chemotherapy is administered to prevent or treat meningeal involvement.
Although Hodgkin's disease is not included as part of the CDC definition of AIDS, studies have found that HIV infection is associated with a fivefold increase in the incidence of Hodgkin's disease. HIV-infected persons with Hodgkin's disease are more likely to have mixed cellularity and lymphocyte depletion subtypes of Hodgkin's disease and to seek medical attention at an advanced stage of disease.
Anal dysplasia and squamous cell carcinoma have been noted in HIV-infected homosexual men.
P.1358
These lesions have been strongly correlated with previous infection by human papillomavirus (HPV). Although many of the infected men report a history of anal warts or have visible warts, a significant percentage have silent papillomavirus infection. Cytologic (using Papanicolaou smears) and papillomavirus DNA studies can easily be performed on specimens obtained by anal swab. The growing frequency of these problems and the risk of progression from dysplasia to cancer in immunocompromised patients suggest that annual anal swabs for cytologic examination should be done in all HIV-infected persons who have engaged in receptive anal intercourse. An anal Papanicolaou smear is performed by rotating a moistened Dacron swab about 2 cm into the anal canal. The swab is immediately inserted into a cytology bottle.
HPV also appears to play a causative role in cervical dysplasia and neoplasia. The incidence and clinical course of cervical disease in HIV-infected women are discussed below.
13. Gynecologic manifestations
Vaginal candidiasis, cervical dysplasia and neoplasia, and pelvic inflammatory disease are more common in HIV-infected women than in uninfected women. These manifestations also tend to be more severe when they occur in association with HIV infection. Therefore, HIV-infected women need frequent gynecologic care. Vaginal candidiasis may be treated with topical agents (see Chapter 36). However, HIV-infected women with recurrent or severe vaginal candidiasis may need systemic therapy.
The incidence of cervical dysplasia in HIV-infected women is 40%. Because of this finding, HIV-infected women should have Papanicolaou smears every 6 months (as opposed to the Agency for Healthcare Research and Quality [AHRQ] Guideline recommendation for every 12 months). Some clinicians recommend routine colposcopy or cervicography because cervical intraepithelial neoplasia has occurred in women with negative Papanicolaou smears. Cone biopsy is indicated in cases of serious cervical dysplasia.
Cervical neoplasia appears to be more aggressive among HIV-infected women. Most HIV-infected women with cervical cancer die of that disease rather than of AIDS. Because of its frequency and severity, cervical neoplasia was added to the CDC definition of AIDS in 1993.
While pelvic inflammatory disease appears to be more common in HIV-infected women, the bacteriology of this condition appears to be the same as among HIV-uninfected women. At present, HIV-infected women with pelvic inflammatory disease should be treated with the same regimens as uninfected women (see Chapter 17). However, inpatient therapy is generally recommended.
14. Inflammatory reactions (immune reconstitution syndromes or IRIS )
With initiation of HAART, some patients experience inflammatory reactions that appear to be associated with immune reconstitution as indicated by a rapid increase in CD4 count. These inflammatory reactions may present with generalized signs of fevers, sweats, and malaise with or without more localized manifestations that usually represent unusual presentations of opportunistic infections. For example, vitreitis has developed in patients with CMV retinitis after they have been treated with HAART. M avium can present as focal lymphadenitis or granulomatous masses in patients receiving HAART. Tuberculosis may paradoxically worsen with new or evolving pulmonary infiltrates and lymphadenopathy. PML and cryptococcal meningitis may also behave atypically. Clinicians should be alert to these syndromes, which are most often seen in patients who have initiated antiretroviral therapy in the setting of advanced disease and who show rapid increases in CD4 counts with treatment. The diagnosis of IRIS is one of exclusion and can be made only after recurrence or new opportunistic infection has been ruled out as the cause of the clinical deterioration. Management of IRIS is conservative and supportive with use of corticosteroids only for severe reactions. Most authorities recommend that antiretroviral therapy be continued unless the reaction is life-threatening.
B. Laboratory Findings
Specific tests for HIV include antibody and antigen detection (Table 31-2). Conventional HIV antibody testing is done by ELISA. Positive specimens are then confirmed by a different method (eg, Western blot). The sensitivity of screening serologic tests is greater than 99.5%. The specificity of positive results by two different techniques approaches 100% even in low-risk populations. False-positive screening tests may occur as normal biologic variants or in association with recent influenza vaccination or other disease states, such as connective tissue disease. These are usually detected by negative confirmatory tests. Molecular biology techniques (PCR) show a small incidence of individuals (< 1%) who are infected with HIV for up to 36 months without generating an antibody response. However, antibodies that are detectable by screening serologic tests will develop in 95% of persons within 6 weeks after infection.
Rapid HIV antibody tests are now available. They provide results within 10 20 minutes and can be performed in physician offices, including by personnel without laboratory training and without a Clinical Laboratory Improvement Amendment (CLIA) approved laboratory. Persons who test positive on a rapid test should be told that they may be HIV-infected or their test may be falsely reactive. Standard testing (ELISA with Western blot confirmation) should be performed to distinguish these two possibilities. Rapid testing is particularly helpful in settings where a result is needed immediately (eg, a woman in labor who has not recently been tested for HIV) or when the patient is unlikely to return for a result.
Table 31-2. Laboratory findings with HIV infection. | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Nonspecific laboratory findings with HIV infection may include anemia, leukopenia (particularly lymphopenia), and thrombocytopenia in any combination,
P.1359
elevation of the erythrocyte sedimentation rate, polyclonal hypergammaglobulinemia, and hypocholesterolemia. Cutaneous anergy is common.
Several laboratory markers are available to provide prognostic information and guide therapy decisions (Table 31-2). The most widely used marker is the absolute CD4 lymphocyte count. As counts decrease, the risk of serious opportunistic infection over the subsequent 3 5 years increases (Figure 31-1).
There are many limitations to using the CD4 count, including diurnal variation, depression with intercurrent illness, and intralaboratory and interlaboratory variability. Therefore, the trend is more important than a single determination. The frequency of performance of counts depends on the patient's health status. Patients whose CD4 counts are substantially above the threshold for initiation of antiviral therapy (350 cells/mcL) should have counts performed every 6 months. Those who have counts near or below 350 cells/mcL should have counts performed every 3 months. This is necessary for evaluating the efficacy of antiviral therapy and for initiating P jiroveci prophylactic therapy when the CD4 count drops below 200 cells/mcL. Some studies suggest that the percentage of CD4 lymphocytes is a more reliable indicator of prognosis than the absolute counts because the percentage does not depend on calculating a manual differential. While the CD4 count measures immune dysfunction, it does not provide a measure of how actively HIV is replicating in the body. HIV viral load tests (discussed below) assess the level of viral replication and provide useful prognostic information that is independent of the information provided by CD4 counts.
Chung RT et al: Peginterferon alfa-2a plus ribavirin versus interferon alfa-2a plus ribavirin for chronic hepatitis C in HIV-coinfected persons. N Engl J Med 2004;351:451.
DeSimone JA et al: Inflammatory reactions in HIV-1-infected persons after initiation of highly active antiretroviral therapy. Ann Intern Med 2000;133:447.
Ellis E et al: Eosinophilic pustular folliculitis: a comprehensive review of treatment options. Am J Clin Dermatol 2004;5:189.
Grinspoon S et al: Effects of testosterone and progressive resistance training in eugonadal men with AIDS wasting. A randomized, controlled trial. Ann Intern Med 2000;133:348.
Keenan PA et al: Rapid HIV testing. Wait time reduced from days to minutes. Postgrad Med 2005;117:47.
Kovacs JA et al: New insights into the transmission, diagnosis, and drug treatment of Pneumocystis carinii pneumonia. JAMA 2001;286:2450.
Little RF et al: HIV-associated non-Hodgkin lymphoma: incidence, presentation, and prognosis. JAMA 2001;285:1880.
Marcellin P et al: Peginterferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HbeAg-negative chronic hepatitis B. N Engl J Med 2004;351:1206.
Norris S: Outcomes of liver transplantation in HIV-infected individuals: the impact of HCV and HBV infection. Liver Transpl 2004;10:1271.
Sacktor N: The epidemiology of human immunodeficiency virus-associated neurological disease in the era of highly active antiretroviral therapy. J Neurovirol 2002;8(Suppl 2):115.
Stevens DL et al: Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clin Infect Dis 2005;41:1373.
P.1360
Wyen C: Progressive multifocal leukoencephalopathy in patients on highly active antiretroviral therapy: survival and risk factors of death. J Acquir Immune Defic Syndr 2004;37:1263.
Differential Diagnosis
HIV infection may mimic a variety of other medical illnesses. Specific differential diagnosis depends on the mode of presentation. In patients presenting with constitutional symptoms such as weight loss and fevers, differential considerations include cancer, chronic infections such as tuberculosis and endocarditis, and endocrinologic diseases such as hyperthyroidism. When pulmonary processes dominate the presentation, acute and chronic lung infections must be considered as well as other causes of diffuse interstitial pulmonary infiltrates. When neurologic disease is the mode of presentation, conditions that cause mental status changes or neuropathy eg, alcoholism, liver disease, renal dysfunction, thyroid disease, and vitamin deficiency should be considered. If a patient presents with headache and a cerebrospinal fluid pleocytosis, other causes of chronic meningitis enter the differential. When diarrhea is a prominent complaint, infectious enterocolitis, antibiotic-associated colitis, inflammatory bowel disease, and malabsorptive symptoms must be considered.
Prevention
A. Primary Prevention
Until vaccination is a reality, prevention of HIV infection will depend on effective precautions regarding sexual practices and injection drug use, use of perinatal HIV prophylaxis, screening of blood products, and infection control practices in the health care setting. Primary care clinicians should routinely obtain a sexual history and provide risk factor assessment of their patients and, when appropriate, screening for HIV infection with pretest and posttest counseling. Pretest counseling should include review of risk factors for HIV infection, discussion of safe sex and safe needle use, and the meaning of a positive test. Posttest counseling should include a review of the importance of safe sex and needle use practices. For persons who test positive, information on available medical and mental health services should be provided as well as guidance for contacting sexual or needle-sharing partners. It is the duty of clinicians to counsel HIV-negative patients on how to avoid exposure to HIV. Patients should be counseled not to exchange bodily fluids unless they are in a long-term mutually monogamous relationship with someone who has tested HIV antibody-negative and has not engaged in unsafe sex, injection drug use, or other HIV risk behaviors for at least 6 months prior to or at any time since the negative test.
Only latex condoms should be used, along with a water-soluble lubricant. Although nonoxynol-9, a spermicide, kills HIV, it is contraindicated because in some patients it may cause genital ulcers that could facilitate HIV transmission. Patients should be counseled that condoms are not 100% effective. They should be made familiar with the use of condoms, including, specifically, the advice that condoms must be used every time, that space should be left at the tip of the condom as a receptacle for semen, that intercourse with a condom should not be attempted if the penis is only partially erect, that men should hold on to the base of the condom when withdrawing the penis to prevent slippage, and that condoms should not be reused. Although anal intercourse remains the sexual practice at highest risk of transmitting HIV, seroconversions have been documented with vaginal and oral intercourse as well. Therefore, condoms should be used when engaging in these activities. Women as well as men should understand how to use condoms so as to be sure that their partners are using them correctly.
Persons using injection drugs should be cautioned never to exchange needles or other drug paraphernalia. When sterile needles are not available, bleach does appear to inactivate HIV and should be used to clean needles.
Current efforts to screen blood and blood products have lowered the risk of HIV transmission with transfusion of a unit of blood to 1:1,000,000.
In the hospital, concerns about nosocomial infection have led to the recommendation for universal body fluid precautions. This involves the rigorous use of gloves when handling any body fluid and the addition of gown, mask, and goggles for procedures that may result in splash or droplet spread as well as the use of specially designed needles with sheath devices to decrease the risk of needle sticks. Reports of transmission of drug-resistant tuberculosis in health care settings also have had infection control implications. All patients with cough in outpatient settings should be encouraged to wear masks. Hospitalized HIV-infected patients with cough should be placed in respiratory isolation until tuberculosis can be excluded by chest radiograph and sputum smear examination.
Primate model data have suggested that development of a protective vaccine may be possible, but clinical trials in humans using gp120 or its precursor gp160 have shown development of neutralizing antibodies to laboratory but not field isolates of HIV and may not be protective of infection. Many scientists have abandoned the quest for a fully protective HIV vaccine and are focusing on developing a vaccine that would reduce the chances of HIV transmission given a particular exposure.
B. Secondary Prevention
In the era prior to the development of highly effective antiretroviral treatment, cohort studies of individuals with documented dates of seroconversion demonstrate that AIDS develops within 10 years in approximately 50% of untreated seropositive persons. Recent improvements in treatment would be expected to substantially improve this prognosis. Too few persons have been treated with these regimens prior to the development of AIDS to provide
P.1361
good data on progression of disease with these new regimens. Nevertheless, decreases in the incidence of AIDS reflecting successful treatment of HIV and successful HIV prevention efforts have been reported in the United States and western Europe.
Table 31-3. Health care maintenance of HIV-infected individuals. | ||
|---|---|---|
|
There is substantial evidence that prophylactic regimens can prevent opportunistic infections and improve survival. Prophylaxis and early intervention prevent several infectious diseases, including tuberculosis and syphilis, which are transmissible to others. Recommendations are listed in Table 31-3.
Because of the increased occurrence of tuberculosis among HIV-infected patients, all such individuals should undergo PPD testing. Although anergy is common among AIDS patients, the likelihood of a false-negative result is much lower when the test is done early in infection. Those with positive tests (defined for HIV-infected patients as > 5 mm of induration) need a chest radiograph. Patients with an infiltrate in any location, especially if accompanied by mediastinal adenopathy, should have sputum sent for acid-fast staining. Patients with a positive PPD but negative evaluations for active disease should receive isoniazid (300 mg daily) with pyridoxine (50 mg daily) for 9 months to a year. An alternative regimen of rifampin plus pyrazinamide for 2 months is no longer recommended because of the high associated incidence of hepatotoxicity. Recent analysis suggests also that HIV-infected individuals at high risk for tuberculosis should receive a course of isoniazid prophylaxis regardless of the PPD status. This may include homeless individuals and injection drug users.
Because of recent increases in the number of cases of syphilis among men who have sex with men, including those who are HIV infected, all such men should be screened for syphilis by rapid plasma reagin (RPR) or Venereal Disease Research Laboratories (VDRL) test every 6 months. Increases of syphilis cases among HIV-infected persons are of particular concern because these individuals are at increased risk for reactivation of syphilis and progression to tertiary syphilis despite standard treatment. Because the only widely available tests for syphilis are serologic and because HIV-infected individuals are known to have disordered antibody production, there is concern about the interpretation of these titers. This concern has been fueled by a report of an HIV-infected patient with secondary syphilis and negative syphilis serologic testing. Furthermore, HIV-infected individuals may lose fluorescent treponemal antibody absorption (FTA-ABS) reactivity after treatment for syphilis, particularly if they have low CD4 counts. Thus, in this population, a nonreactive treponemal test does not rule out a past history of syphilis. In addition, persistence of treponemes in the spinal fluid after one dose of benzathine penicillin has been demonstrated in HIV-infected patients with primary and secondary syphilis. Therefore, the CDC has recommended an aggressive diagnostic approach to HIV-infected patients with reactive RPR or VDRL tests of greater than 1 year or unknown duration. All such patients should have a lumbar puncture with cerebrospinal fluid cell count and cerebrospinal fluid VDRL. Those with a normal cerebrospinal fluid evaluation are treated as having late latent syphilis (benzathine penicillin G, 2.4 million units intramuscularly weekly for 3 weeks) with follow-up titers. Those with a pleocytosis or a positive cerebrospinal fluid-VDRL test are treated as having neurosyphilis (aqueous penicillin G, 2 4 million units intravenously every 4 hours, or procaine penicillin G, 2.4 million units intramuscularly daily, with probenecid, 500 mg four times daily, for 10 days). Some clinicians take a less aggressive approach to patients who have low titers (less than 1:8), a history of having been treated for syphilis, and a normal neurologic examination. Close follow-up of titers is mandatory if such a course is taken. For a more detailed discussion of this topic, see Chapter 34.
The efficacy of pneumococcal vaccine is debated, but since it is safe, HIV-infected individuals should receive it. Patients without evidence of hepatitis B surface antigen or surface antibody should receive hepatitis B vaccination. Live vaccines, such as yellow fever vaccine, should be avoided. Measles vaccination, while a live virus vaccine, appears relatively safe when administered to HIV-infected individuals and should be given if the patient has never had measles or been adequately vaccinated.
A randomized study found that multivitamin supplementation decreased HIV disease progression and mortality in HIV-infected women in Africa. However, supplementation is unlikely to be as effective in well-nourished populations.
P.1362
HIV-infected individuals should be counseled with regard to safe sex. Because of the risk of transmission, they should be warned to use condoms with sexual intercourse, including oral intercourse. Partners of HIV-infected women should use latex barriers such as dental dams (available at dental supply stores) to prevent direct oral contact with vaginal secretions. Substance abuse treatment should be recommended for persons who are using recreational drugs. They should be warned to avoid consuming raw meat or eggs to avoid infections with Toxoplasma, Campylobacter, and Salmonella. HIV-infected patients should wash their hands thoroughly after cleaning cat litter or should forgo this household chore to avoid possible exposure to toxoplasmosis. To reduce the likelihood of infection with Bartonella species, patients should avoid activities that might result in cat scratches or bites. Although the data are not conclusive, many clinicians recommend that HIV-infected persons especially those with low CD4 counts drink bottled water instead of tap water to prevent cryptosporidia infection.
Because of the emotional impact of HIV infection and subsequent illness, many patients will benefit from supportive counseling.
C. HIV Risk for Health Care Professionals
Epidemiologic studies show that needle sticks occur commonly among health care professionals, especially among surgeons performing invasive procedures, inexperienced hospital house staff, and medical students. Efforts to reduce needle sticks should focus on avoiding recapping needles and use of safety needles whenever doing invasive procedures under controlled circumstances. The risk of HIV transmission from a needle stick with blood from an HIV-infected patient is about 1:300. The risk is higher with deep punctures, large inocula, and source patients with high viral loads. The risk from mucous membrane contact is too low to quantitate.
Health care professionals who sustain needle sticks should be counseled and offered HIV testing as soon as possible. HIV testing is done to establish a negative baseline for worker's compensation claims in case there is a subsequent conversion. Follow-up testing is usually performed at 6 weeks, 3 months, and 6 months.
A case-control study by the CDC indicates that administration of zidovudine following a needle stick decreases the rate of HIV seroconversion by 79%. Therefore, providers should be offered therapy with Combivir (zidovudine 300 mg plus lamivudine 150 mg orally twice daily). Providers who have exposures to persons who are likely to have antiretroviral drug resistance (eg, persons receiving therapy who have detectable viral loads) should have their therapy individualized, using at least two drugs to which the source is unlikely to be resistant. Some clinicians recommend triple combination regimens, including a protease inhibitor for all occupational exposures, because of uncertainty about drug resistance. Others save these more aggressive regimens for the higher-risk exposures listed above. Because reports have noted hepatotoxicity due to nevirapine in this setting, this agent should be avoided. Therapy should be started as soon as possible after exposure and continued for 4 weeks. Unfortunately, there have been documented cases of seroconversion following potential parenteral exposure to HIV despite prompt use of zidovudine prophylaxis. Counseling of the provider should include safe sex guidelines.
D. Postexposure Prophylaxis for Sexual and Drug Use Exposures to HIV
Following publication of a case-control study indicating that antiretroviral therapy decreased the odds of seroconversion among health care workers who had occupational exposure, some experts have recommended offering antiretroviral therapy following potential exposure to HIV through sexual activity or drug use. Although there are no efficacy data to support this practice, there are similarities between the immune response following transcutaneous and transmucosal exposures. The goal of postexposure prophylaxis is to reduce or prevent local viral replication prior to dissemination such that the infection can be aborted.
The choice of antiretroviral agents and the duration of treatment are the same as those for exposures that occur through the occupational route (see above). In contrast to those with occupational exposures, some individuals may present very late after exposure. Because the likelihood of success declines with length of time from HIV exposure, it is not recommended that treatment be offered after 72 hours. In addition, because the psychosocial issues involved with postexposure prophylaxis for sexual and drug use exposures are complex, it should be offered only in the context of prevention counseling. Counseling should focus on how to prevent future exposures.
E. Preventing Perinatal Transmission of HIV
A multicenter trial showed that when zidovudine is administered to women during pregnancy, labor, and delivery and to their newborns, the rate of HIV transmission is decreased by two-thirds. An observational trial demonstrated that zidovudine treatment is almost as effective when begun during labor or when administered only to the infant, as long as treatment is begun within 48 hours after birth. Nonetheless, treatment begun by at least the second trimester is still recommended. Many women are currently being offered combination antiretroviral treatment to further lower the risk of transmission. The availability of treatment makes it essential that all women who are pregnant or considering pregnancy be offered HIV counseling and testing. Many obstetricians recommend combination antiretroviral treatment, especially if zidovudine resistance is suspected. HIV-infected women receiving antiretroviral therapy in whom pregnancy is recognized during the first trimester should be counseled about the benefits and potential risks to the fetus of treatment during the first trimester. Because healthy mothers make healthy babies, continuation of therapy should be strongly
P.1363
considered. Because about half of fetal infections in non-breast-feeding women occur shortly before or during the birth process, antiretroviral therapy should be administered whenever a woman initiates perinatal care even if she did not begin therapy in the second trimester. Breast-feeding is thought to increase the rate of transmission by 10 20% and should be avoided.
Aragon TJ et al: Endemic cryptosporidiosis and exposure to municipal tap water in persons with acquired immunodeficiency syndrome (AIDS): a case-control study. BMC Public Health 2003;3:2.
Fawzi WW et al: A randomized trial of multivitamin supplements and HIV disease progression and mortality. N Engl J Med 2004;351:23.
Hammer SM: Management of newly diagnosed HIV infection. N Engl J Med 2005;353:1702.
Johnson WD et al: HIV prevention research for men who have sex with men: a systematic review and meta-analysis. J Acquir Immune Defic Syndr 2002;30(Suppl 1):S118.
Katz MH et al: Management of occupational and nonoccupational postexposure HIV prophylaxis. Curr HIV/AIDS Rep 2004;1:159.
Smith DK: Antiretroviral postexposure prophylaxis after sexual, injection-drug use, or other nonoccupational exposure to HIV in the United States: recommendations from the U.S. Department of Health and Human Services. MMWR Recomm Rep 2005;54(RR-2):1.
Treatment
Treatment for HIV infection can be divided into four categories: therapy for opportunistic infections and malignancies, antiretroviral treatment, hematopoietic stimulating factors, and prophylaxis of opportunistic infections.
Experimental treatment regimens for HIV infection are constantly changing. Clinicians may obtain up-to-date information on experimental treatments by calling the AIDS Clinical Trials Information Service (ACTIS), 800-TRIALS-A (English and Spanish), and the National AIDS Hot Line, 800 342-AIDS (English), 800 344-SIDA (Spanish), and 800-AIDS-TTY (hearing-impaired).
A. Therapy for Opportunistic Infections and Malignancies
Treatment of common HIV infections and malignancies is detailed in Table 31-4. In the era prior to the use of HAART, patients required lifelong treatment for many infections, including CMV retinitis, toxoplasmosis, and cryptococcal meningitis. However, among patients who have a good response to HAART, maintenance therapy for some opportunistic infections can be terminated. For example, in consultation with an ophthalmologist, maintenance treatment for CMV infection can be discontinued in persons with durable suppression of viral load on HAART who have a CD4 count > 100 150 cells/mcL. Similar results have been observed in patients with M avium complex bacteremia.
Table 31-4. Treatment of AIDS-related opportunistic infections and malignancies.1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The emergence of resistance is more common for some infections of HIV-infected people (eg, acyclovir-resistant herpes simplex) than among immunocompetent individuals. In addition, HIV-infected patients have an increased incidence of side effects to standard drugs such as trimethoprim-sulfamethoxazole.
Treating patients with repeated episodes of the same opportunistic infection can pose difficult therapeutic challenges. For example, patients with second or third episodes of Pneumocystis pneumonia may have developed allergic reactions to standard treatments with a prior episode. Fortunately, there are several alternatives available for the treatment of Pneumocystis infection. Trimethoprim with dapsone and primaquine with clindamycin are two combinations that often are tolerated in patients with a prior allergic reaction to trimethoprim-sulfamethoxazole and intravenous pentamidine. On the positive side, patients in whom second episodes of Pneumocystis pneumonia develop while taking prophylaxis tend to have milder courses.
Well-established alternative regimens now also exist for most AIDS-related opportunistic infections: amphotericin B or fluconazole for cryptococcal meningitis; ganciclovir, cidofovir, or foscarnet for CMV infection; and sulfadiazine or clindamycin with pyrimethamine for toxoplasmosis.
Although conceptually it would seem that corticosteroid therapy should be avoided in HIV-infected patients, corticosteroid use has been shown to improve the course of patients with moderate to severe pneumocystosis (oxygen saturation < 90%, PO2 < 65 mm Hg) when administered within 72 hours after diagnosis. The mechanism of action is presumed to be a decrease in alveolar inflammation.
B. Antiretroviral Therapy
The availability of agents that in combination suppress HIV replication (Table 31-5) has had a profound impact on the natural history of HIV infection. Patients who achieve excellent suppression of HIV generally have stabilization or improvement of their clinical course that results from partial immunologic reconstitution and a subsequent decrease in complications of immunosuppression. The best time to initiate antiretroviral treatment remains controversial. It is best to weigh the benefits of viral suppression against the side effects of the drugs for each patient. In general, treatment for asymptomatic HIV disease should be initiated when the CD4 cell count drops below 350 cells/mcL or symptomatic HIV disease. Patients with rapidly dropping CD4 counts or very high viral loads (> 100,000/mcL) should be considered for earlier treatment. For those patients who might have difficulty adhering to treatment or who are at higher risk for toxicity (eg, underlying liver disease), waiting until the CD4 count nears 200 cells/mcL may be a better strategy. Although emerging data suggest that every effort should be made to initiate antiretroviral therapy at CD4 counts below 350 cells/mcL, patients who started treatment at higher CD4 levels than
P.1364
P.1365
are currently recommended for initiation of treatment are usually maintained on their regimens if they are doing well and are not suffering side effects from the treatment. When medication is stopped in this population, most patients drop back to their pretreatment nadir CD4 count in a matter of months. Some patients whose counts rise dramatically and who are fully suppressed (ie, plasma viral load < 50 copies/mcL) may be successfully transitioned from a high potency regimen to a lower potency regimen with fewer side effects; however, this induction-maintenance strategy is still being evaluated in clinical trials.
Once a decision to initiate therapy has been made, several important principles should guide therapy. First, because drug resistance to antiretroviral agents develops in HIV-infected patients, a major goal of therapy should be total suppression of viral replication as measured by the serum viral load. Therapy that achieves a plasma viral load of < 50 or < 75 copies/mcL (depending on the test used) has been shown to correlate with antiviral effect in other compartments. To achieve this and maintain virologic control over time, aggressive combination therapy is essential, and partially suppressive combinations such as dual nucleoside therapy should be avoided. Similarly, if toxicity develops, it is preferable to either interrupt the entire regimen or change the offending drug rather than reduce individual doses. The current standard is to use at least three agents simultaneously. Because the number of drugs and potential combinations is finite and the development of drug resistance may severely compromise the efficacy of treatment, patients must be
P.1366
P.1367
P.1368
able to adhere to these regimens. Patients who may miss many doses of treatment should be counseled to defer starting therapy until they are able to adhere to the regimen. Adherence can be promoted through the use of medication boxes with compartments (eg, Medisets), supportive counseling, or daily supervision of therapy. Therefore, decisions to withhold treatment should not be based on a patient's circumstances (eg, active drug use or housing status) alone. Often, a trial intervention such as offering Pneumocystis pneumonia prophylaxis may be helpful in determining the likelihood of adherence to a more complex antiretroviral regimen.
Table 31-5. Antiretroviral therapy. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Monitoring of antiretroviral therapy has two goals. Laboratory evaluation for toxicity depends on the specific drugs in the combination but generally should be done approximately every 3 months once a patient is on a stable regimen. The second aspect of monitoring is to regularly measure objective markers of efficacy. The CD4 cell count and HIV viral load should be repeated 1 2 months after the initiation or change of antiretroviral regimen and every 3 4 months thereafter in clinically stable patients. Reasons for changing antiretroviral regimens include intolerable adverse reactions, rising or persistently detectable viral loads, the clinical progression of disease, and continued immunologic deterioration as reflected by a declining CD4 cell count. Although a rebound in HIV viral load is cited as an indication to change therapy, exact parameters have not been established, and many patients appear to have continued clinical benefit in the face of rising viral load measurements. When therapy is modified, clinicians should attempt to start at least two agents to which an individual has not been exposed or to which there is minimal or no resistance at the same time. Conversely, there is a risk to stopping a single medication because of the possibility of its causing a side effect if the remaining regimen contains too few medications to prevent rapid development of resistance.
Drug resistance testing is recommended for patients who are experiencing treatment failure (persistent or rising viral load despite adherence to an efficacious regimen).
Although the ideal combination of drugs has not yet been defined for all possible clinical situations, possible choices can be better understood after a review of the available agents. These drugs can be grouped into four major categories: nucleoside and nucleotide reverse transcriptase inhibitors (NRTI), protease inhibitors (PI), nonnucleoside reverse transcriptase inhibitors (NNRTI), and entry inhibitors.
1. Nucleoside and nucleotide reverse transcriptase inhibitors
a. Zidovudine
Zidovudine (azidothymidine; AZT) was the first approved antiviral drug for HIV infection and remains an important agent. It is administered at a dose of 300 mg orally twice daily. A combination of zidovudine 300 mg and lamivudine 150 mg (Combivir) allows more convenient dosing of medication for individuals taking both of these agents. Side effects seen with zidovudine are listed in Table 31-5. Approximately 40% of patients experience subjective side effects that usually remit within 6 weeks. The common dose-limiting side effects are anemia and neutropenia, which can be exacerbated by other drugs such as ganciclovir that cause bone marrow suppression. A minority of patients have the more rapid onset of red cell aplasia. Bone marrow toxicity is generally reversible with cessation of the drug.
In monitoring patients receiving zidovudine, complete blood counts with platelet and differential counts should be done monthly for the first 2 months of therapy and then every 3 months. Creatine phosphokinase (CPK) levels should be checked every 6 months.
b. Didanosine
The most convenient formulation of didanosine (ddI) is the enteric-coated capsule. For adults weighing at least 60 kg, the dose is one 400-mg enteric-coated capsule orally daily; for those 30 59 kg, the dose is one 250-mg enteric-coated capsule orally daily. Didanosine should be taken on an empty stomach; if taken with indinavir, the two medications should be taken at least 1 hour apart. Didanosine levels are increased when it is administered with tenofovir, potentially resulting in increased side effects. Therefore, this combination should be used very cautiously.
Unlike zidovudine, didanosine does not cause anemia but may cause neutropenia. It has also been associated with pancreatitis. The incidence of pancreatitis with didanosine is 5 10% of fatal pancreatitis, less than 0.4%. Patients with a history of pancreatitis, as well as those taking other medications associated with pancreatitis (including trimethoprim-sulfamethoxazole and intravenous pentamidine) are at higher risk for this complication. Patients should be warned to use alcohol judiciously while taking didanosine. Clinicians should also teach patients to watch for the symptoms of pancreatitis and to stop treatment if abdominal pain, nausea, or vomiting develops while taking didanosine until it can be determined if they have pancreatitis. Other common side effects with didanosine include a dose-related, reversible, painful peripheral neuropathy, which occurs in about 15% of patients, and dry mouth. Fulminant hepatic failure and electrolyte abnormalities, including hypokalemia, hypocalcemia, and hypomagnesemia, have been reported in patients taking didanosine.
c. Stavudine
Stavudine (d4T) has shown good activity as an antiretroviral drug. The dose is 40 mg orally twice daily for individuals weighing 60 kg or more. However, because of its side effects including lipoatrophy, lipodystrophy, peripheral neuropathy and, rarely, lactic acidosis and hepatitis, this drug should no longer be used except when there is no alternative. Patients taking stavudine should be routinely changed to abacavir or tenofovir, both of which are less likely to cause lipoatrophy and lipodystrophy.
d. Lamivudine
Lamivudine (3TC) is a safe and well-tolerated agent. The dosage is 150 mg orally twice daily or 300 mg orally once a day. There are no significant side effects with this agent, and it has activity also
P.1369
against hepatitis B. The dose should be reduced with renal insufficiency.
e. Emtricitabine
Emtricitabine is a nucleoside analog that is dosed at 200 mg orally. It was developed primarily as a once a day alternative to lamivudine. However, lamivudine can be dosed daily, eliminating the special indication for emtricitabine. As is true of lamivudine, emtricitabine has activity against hepatitis B and its dosage should be reduced in patients with renal insufficiency.
f. Abacavir
A daily dose of 300 mg orally twice daily results in potent antiretroviral activity. Abacavir retains activity against some HIV strains that have become resistant to other nucleoside drugs. Abacavir is formulated with zidovudine and lamivudine in a single pill (Trizivir, one tablet orally twice daily). Although it is a triple-drug regimen, it is not as efficacious as combining two nucleoside/nucleotide analogs with a PI or an NNRTI and should be used only for patients who cannot comply with other regimens. Abacavir is also available as a fixed dose combination pill with lamivudine for use as a once daily pill (Epzicom). The main toxicity of abacavir is a hypersensitivity syndrome in about 5% of patients, characterized by a flu-like syndrome with rash and fever that worsens with successive doses; individuals in whom this syndrome develops should not be rechallenged with this agent as reactions with rechallenge can be fatal.
g. Zalcitabine
Zalcitabine (ddC) is thought to be one of the least effective antiretroviral agents and is therefore not commonly used. Its advantages are that it is inexpensive, easy to administer, and has no known hematologic side effects. The usual dosage of zalcitabine is approximately 0.005 0.01 mg/kg orally every 8 hours. This drug is formulated in 0.375-mg and 0.75-mg tablets.
Zalcitabine, like didanosine, may cause peripheral neuropathy. It is also associated with aphthous ulcers, rash, and rare cases of pancreatitis.
h. Tenofovir
Tenofovir is the only licensed nucleotide analog. It is given as a single daily oral dose of 300 mg and is generally well tolerated. As is true of lamivudine, tenofovir is active against hepatitis B. Because HIV variants that are resistant to other nucleoside analogs are often sensitive to tenofovir, it is often included in second- and third-line treatment regimens. It is also available in a fixed dose combination pill with emtricitibine (Truvada) for daily dosing. A once a day single fixed dose combination pill that contains tenofovir, emtricitabine, and efavirenz should be available in 2006.
2. Protease inhibitors
Nine PIs indinavir, nelfinavir, ritonavir, saquinavir, amprenavir, fosamprenavir, lopinavir (in combination with ritonavir), atazanavir, and tipranavir are currently available. PIs have been shown to potently suppress HIV replication in vitro and in vivo and are administered as part of a combination regimen.
All the PIs to differing degrees are metabolized by the cytochrome P-450 system, and each can inhibit and induce various P-450 isoenzymes. Therefore, drug interactions are common and difficult to predict. Clinicians should consult the product inserts before prescribing PIs with other medications. Drugs such as rifampin that are known to induce the P-450 system should be avoided.
The fact that the PIs are dependent on metabolism through the cytochrome P-450 system has led to some innovative dosing strategies. In particular, ritonavir has been used to boost the drug levels of saquinavir, lopinavir, indinavir, atazanivir, tipranavir, and amprenavir, allowing use of lower doses and simpler dosing schedules of these PIs.
PIs and NRTIs have been linked to a constellation of metabolic abnormalities, including elevated cholesterol levels, elevated triglyceride levels, insulin resistance, diabetes mellitus, and changes in body fat composition (eg, buffalo hump, abdominal obesity). The lipid abnormalities and body habitus changes are referred to as lipodystrophy. Its prevalence is known to be increased among persons treated with certain NRTIs (especially stavudine) and most of the PIs, but it has been seen also in HIV-infected persons who have never been treated with these agents.
Of the different manifestations of lipodystrophy, the dyslipidemias that occur are of particular concern because of the likelihood that increased levels of cholesterol and triglycerides will result in increased prevalence of heart disease. All patients taking PIs or NRTIs should have a fasting cholesterol, low-density lipoprotein cholesterol, and triglyceride level performed every 3 6 months. Until there is longer follow-up of HIV-infected persons with elevated cholesterol levels, treatment decisions should be the same as those for uninfected persons (see Chapter 28). HIV-infected persons who do not respond to dietary interventions should be started on pravastatin (20 mg daily orally) or atorvastatin (10 mg daily orally). Lovastatin and simvastatin should be avoided because of their interactions with PIs. Patients with persistently elevated fasting serum triglyceride levels of 500 mg/dL or more who do not respond to dietary intervention should be treated with gemfibrozil (600 mg twice daily prior to the morning and evening meals).
a. Indinavir
The standard dose of indinavir is 800 mg orally three times a day. Twice-daily dosing of indinavir is feasible in combination with ritonavir. It is recommended that indinavir be taken without food (if not boosted with ritonavir) but with water 1 hour before or 2 hours after a meal. Nausea and headache are common complaints with this drug. Indinavir crystals are present in the urine in approximately 40% of patients; this results in clinically apparent nephrolithiasis in about 15% of patients receiving indinavir. Lower urinary tract symptoms and renal failure have been increasingly reported. Patients taking this drug should be instructed to drink at least 48 ounces of fluid a day to ensure adequate hydration in an attempt to avoid these complications, but if stones occur repeatedly, an
P.1370
alternative PI may be needed. Mild indirect hyperbilirubinemia is also commonly observed in patients taking indinavir but is not an indication for discontinuation of the drug.
b. Nelfinavir
This agent appears to have slightly less potent antiviral activity than either indinavir or ritonavir but has shown short-term benefit in combination regimens. The dose of nelfinavir is 750 mg orally three times daily or 1250 mg orally twice daily. Diarrhea is a side effect in 25% of patients taking nelfinavir, and this symptom may be controlled in the majority of patients with over-the-counter antidiarrheal agents.
c. Ritonavir
Use of this potent PI has been limited by its inhibition of the cytochrome P-450 pathway causing a large number of drug-drug interactions and by its frequent side effects of fatigue, nausea, and paresthesias when the full dose of 600 mg orally twice daily is given. One of the major benefits of ritonavir is that it increases the bioavailability of other PIs (eg, saquinavir, lopinavir) and it is widely used in lower dose (eg, 100 mg daily to 100 mg twice daily) as a booster of other PIs.
d. Saquinavir
Saquinavir has two different formulations: a hard-gel capsule (Invirase) and a soft-gel capsule (Fortovase). The two are not bioequivalent and cannot be used interchangeably. The hard-gel capsule can be used only when boosted by ritonavir (1000 mg of hard-gel saquinavir with 100 mg of ritonavir orally twice daily). The soft-gel capsule (Fortovase) can be used as the sole PI (ie, without ritonavir boosting) at a dosage of 1200 mg three times daily with food. However, because of the large pill burden and because of the necessity for three-times-daily dosing with food, soft-gel saquinavir is not commonly used. The most common side effects with saquinavir are diarrhea, nausea, dyspepsia, and abdominal pain.
e. Amprenavir
Amprenavir has efficacy and side effects similar to those of other PIs. Common side effects are nausea, vomiting, diarrhea, rash, and perioral paresthesia. The dose is 1200 mg orally twice daily. This requires that the patient take 16 capsules a day. When used in combination with ritonavir, the recommended doses are amprenavir 1200 mg with ritonavir 200 mg daily or amprenavir 600 mg with ritonavir 100 mg twice daily.
f. Fosamprenavir
Fosamprenavir is a prodrug of amprenavir. Its major advantage over using amprenavir is a much lower pill burden. It can be dosed at 1400 mg orally twice daily (four capsules a day) or at 1400 mg orally daily (two capsules) with ritonavir 200 mg orally daily (two capsules). Side effects are similar to those with amprenavir.
g. Lopinavir/r
Lopinavir/r is lopinavir formulated with a low dose of ritonavir to maximize the bioavailability of lopinavir. It has been shown to be more effective than nelfinavir when used in combination with stavudine and lamivudine. The usual dose is 400 mg lopinavir with 100 mg of nelfinavir (two tablets) orally twice daily with food. When given along with efavirenz or nevirapine, a higher dose (600 mg/150 mg three tablets) is usually prescribed. The most common side effect is diarrhea, and lipid abnormalities are frequent.
h. Atazanavir
Atazanavir has two major advantages compared to other PIs. It needs to be dosed only once daily (two 200-mg capsules with food) and has only minimal impact on cholesterol and triglyceride levels. The most common side effect is mild hyperbilirubinemia that resolves with discontinuation of the drug. Because ritonavir increases the serum concentrations of atazanavir, when these drugs are used together, atazanavir is dosed at 300 mg orally daily and ritonavir is dosed at 100 mg orally daily. Both tenofovir and efavirenz lower the serum concentration of atazanavir. Therefore, when either of these two drugs is used with atazanavir, it should be boosted by administering ritonavir. Proton pump inhibitors are contraindicated in patients taking atazanavir because atazanavir requires an acidic pH to remain in solution.
i. Tipranavir
Tipranavir is the first approved nonpeptidic PI. Because of its unique structure, it is active against some strains of the virus that are resistant to other available PIs. It is dosed with ritonavir (two 250 mg capsules of tipranavir with two 100 mg capsules of ritonavir orally twice daily with food). The most common side effects are nausea, vomiting, diarrhea, fatigue, and headache. Tipranavir/ritonavir has been also associated with liver damage and should be used very cautiously in patients with underlying liver disease. Because it is a sulfa-containing drug, its use should be closely monitored in patients with sulfa allergy.
3. Nonnucleoside reverse transcriptase inhibitors
NNRTIs inhibit reverse transcriptase at a site different from that of the nucleoside and nucleotide agents described above. All three have shown antiviral activity as measured by HIV viral load and CD4 responses. The major advantage of the NNRTIs is that two of them (nevirapine and efavirenz) have potencies comparable to that of PIs with lower pill burden. In particular, they do not appear to cause lipodystrophy; patients with cholesterol and triglyceride elevations who are switched from a PI to an NNRTI may have improvement in their symptoms. The resistance patterns of the NNRTIs are distinct from those of the PIs, so their use still leaves open the option for future PI use.
The NNRTIs can be used with PIs in patients who are difficult to suppress on simpler regimens or when it is difficult to identify at least two agents to which the patient is not resistant. Because these agents may cause alterations in the clearance of PIs, dose modifications may be necessary when these two classes of medications are administered concomitantly. Resistance to one drug in this class uniformly predicts resistance to other drugs in the class, though second-generation drugs are being developed. There is no therapeutic reason for using more than one NNRTI at the same time.
P.1371
a. Nevirapine
The target dose of nevirapine is 200 mg orally twice daily, but it is initiated at a dose of 200 mg once a day to decrease the incidence of rash, which is as high as 40% when full doses are begun immediately. If rash develops while the patient is taking 200 mg a day, liver enzymes should be checked and the dose should not be increased until the rash resolves. Patients with mild rash and no evidence of hepatotoxicity can continue to be treated with nevirapine. The hepatotoxicity of the drug can be fatal, particularly in women and those patients with high CD4 counts (> 250 mcL), which limits its role as a first-line agent.
b. Efavirenz
The major advantage of efavirenz is that it can be given once daily in a single dose (600 mg orally). The side effects are neurologic, with patients reporting symptoms ranging from lack of concentration and strange dreams to delusions and mania. Fortunately, the neurologic side effects of efavirenz subside over time, usually within a month. Due to teratogenicity, efavirenz should be avoided in women who wish to conceive or are already pregnant. A once-daily fixed dose combination of efavirenz, tenofovir, and emtricitabine in a single pill should be available in 2006.
c. Delavirdine
Of the three available NNRTIs, delavirdine is least used largely as a result of its less convenient dosing and pill burden compared to the other available NNRTIs. Unlike nevirapine and efavirenz, delavirdine inhibits P-450 cytochromes rather than inducing these enzymes. This means that delavirdine can act like ritonavir and boost other antiretrovirals, although delavirdine is not as potent as ritonavir in this capacity. The dosage is 400 mg orally three times a day, which makes it less convenient for patients than the other two medications in this class. As with nevirapine, the major side effect is rash, but the incidence of rash is lower with delavirdine.
4. Novel agents
a. Entry inhibitors
Peptide T-20 (enfuvirtide) is the first drug in the new class of fusion inhibitors, which block the entry of HIV into cells. The addition of enfuvirtide to an optimized antiretroviral regimen improved CD4 counts and lowered viral loads in heavily pretreated patients with multidrug-resistant HIV. The dose is 90 mg by subcutaneous injection twice daily.
b. CCR5 antagonists
There are currently 2 CCR5 inhibitors in phase II/III clinical trials in humans. Although the exact role and level of activity of this new class of drugs have not been fully defined, it is clear that there is good antiviral activity and when combined with other active drugs can result in excellent virologic responses.
c. Integrase inhibitors
HIV integrase inhibitors are currently in phase II clinical trials and early reports suggest excellent antiviral activity. Because these drugs target a different enzyme of the HIV virus, there is great optimism that they will be helpful for patients who are resistant to other classes of medications and that they might help prevent resistance by use with available medications that target the reverse transcriptase and protease enzymes.
d. Maturation inhibitors
Recently, a drug has been tested in humans that appears to interfere with the maturation of HIV virions. This represents a promising new potential target for HIV drug development.
5. Constructing regimens
There is now little debate about the necessity for combining drugs to achieve long-term suppression of HIV and its associated clinical benefit. Only combinations of three or more drugs have been able to decrease HIV viral load by 2 3 logs and allow suppression of HIV RNA to below the threshold of detection for longer than 10 years in some individuals. However, there have been few studies directly comparing one triple combination therapy regimen to another. Therefore, clinicians and patients face difficult decisions in selecting therapy.
Current evidence supports the use of Truvada (tenofovir and emtricitabine) as the nucleoside/nucleotide backbone combined with efavirenz as the initial regimen. This regimen has been shown to be more effective and better tolerated than Combivir (zidovudine and lamivudine). It has the advantages of once daily dosing and low pill burden (two pills per day). This recommendation is likely to grow stronger with the availability of a single pill that contains efavirenz, tenofovir, and emtricitabine and that can be dosed once daily. Because 8 10% of newly infected persons in some urban areas of the United States have NNRTI resistance, resistance testing should be performed prior to initiating efavirenz in this population.
There are increasing data to suggest the virologic and clinical inferiority of regimens that include only nucleoside and nucleotide analogs without nonnucleoside agents or PIs. Thus, triple nucleoside/nucleotide regimens should be avoided when other options exist. Studies of quad-nucs in which four nucleosides or nucleotides are used in combination are currently ongoing but cannot be recommended at this time.
In the absence of head-to-head comparisons of different regimens in different situations, several general principles should guide the choice of combinations. The most important determinant of treatment efficacy is adherence to the regimen. Therefore, it is vitally important that the regimen chosen be one to which the patient can easily adhere. In general, patients are more compliant with medication regimens that are once or twice a day only, do not require special timing with regard to meals, can be taken at the same time as other medications, do not require refrigeration or special preparation, and do not have bothersome side effects. Second, it is desirable to prescribe combinations that have demonstrated clinical benefit; because these data do not exist for many combinations, it is reassuring to know that the combination under consideration has shown beneficial effects on HIV viral load levels in
P.1372
short-term studies. To the extent possible, agents to which the patient has not been exposed are preferable to drugs for which resistance mutations may have already occurred. Toxicities should ideally be nonoverlapping. An individual's relative contraindications to a given drug or drugs should be considered. The regimen should not include agents that are either virologically antagonistic or incompatible in terms of drug-drug interactions. Compatible dosing schedules prescribing medications that can be taken at the same time improve adherence to treatment. Finally, highly complex therapeutic regimens should be reserved for individuals who are capable of adhering to the rigorous demands of taking multiple medications and having this therapy closely monitored. Conversely, simplified regimens that deliver the lowest number of pills given at the longest possible dosing intervals are desirable for patients who have difficulty taking multiple medications.
 |
Figure 31-2. Approach to antiretroviral therapy. |
Possible ways of incorporating nonnucleoside agents and PIs into combinations are displayed in Figure 31-2.
A number of points about the nucleoside/nucleotide backbone of regimens have become clearer. The combination of stavudine plus didanosine should be avoided as there is increased risk of toxicities, in particular in pregnant women because of the increased risk of lactic acidosis. Also notable is the fact that the addition of lamivudine to didanosine does not appear to result in the same level of viral suppression as when lamivudine is combined with zidovudine or stavudine. Moreover, the nucleoside pair of zidovudine and stavudine should be avoided because of increased toxicity and the potential for antagonism that results from intracellular competition for phosphorylation. Finally, the combination of didanosine with tenofovir can cause declines in CD4 counts despite excellent virologic response and appropriate dose adjustment of didanosine; the reason for this is unknown.
In choosing which agents to include in an initial antiretroviral regimen, ease of administration, minimization of side effects, drug resistance pattern, and future treatment options should all be considered. For some patients who have been heavily treated with antiretroviral agents, it may be difficult to design second-line regimens when they are showing evidence of progression of disease on their existing regimen. In designing second-line regimens (sometimes called salvage regimens a term that some patients may find offensive), the goal is to identify drugs with at least some degree of antiviral activity. This can be quite complicated because of the problem of cross-resistance between drugs within a class. For example, the resistance patterns of lopinavir/ritonavir and indinavir are overlapping, and patients with virus resistant to these agents are unlikely to respond to nelfinavir or saquinavir even though they have never received treatment
P.1373
with these agents. Similarly, the resistance patterns of nevirapine and efavirenz are overlapping. Ideally, at least two drugs to which the patient's virus is susceptible should be added at the same time.
In addition to taking a careful history of what antiretroviral agents a patient has taken and for how long, genotypic and phenotypic resistance testing can provide useful information in designing second-line regimens (see below).
Whatever regimen is chosen, patients should be coached in ways to improve adherence. This should include taking medicines on an established schedule (eg, first thing in the morning), keeping a medication diary, and keeping medications in a variety of places the patient is likely to be (eg, car, workplace). For certain populations (eg, unstably housed individuals), specially tailored programs that include drug dispensing are needed.
For some patients, it is impossible to construct a tolerable regimen that fully suppresses HIV. In such cases, clinicians and patients should consider their goals. Patients maintained on effective antiretroviral agents often benefit from these regimens (eg, higher CD4 counts, fewer opportunistic infections) even if their virus is detectable. In some cases, patients may request a drug holiday during which they are taken off all medications. Patients often immediately feel better because of the absence of drug side effects. Unfortunately, structured treatment interruptions generally result in viral rebound and CD4 decline, and patients who interrupt their treatment fare poorly compared with patients who continue their regimens without interruption.
6. The challenge of drug resistance
HIV-1 drug resistance limits the ability to fully control HIV replication and is a leading cause for antiretroviral regimen failure. Resistance has been documented for all currently available antiretrovirals including the new class of fusion inhibitors. The problem of drug resistance is widespread in HIV-infected patients undergoing treatment in countries where antiretroviral therapy is widely available. A recent prevalence study of a representative sample of patients being treated in U.S. clinics revealed that nearly 80% of patients with detectable viremia had at least some degree of drug resistance. Patients who have been on various antiretroviral regimens and who now have resistant HIV-1 represent a major challenge for the treating clinician. However, the issue of resistant virus does not just concern the treatment-experienced patient. Resistance is now also documented in patients who are antiretroviral treatment naive, but who have been infected with a drug resistant strain primary resistance. Cohort studies of antiretroviral treatment-naive patients entering care in North America and Western Europe show that roughly 10 12% of recently infected individuals have been infected with a drug-resistant strain of HIV-1.
Current expert guidelines recommend resistance testing for patients who are recently infected and for pregnant women. Resistance testing is also recommended for patients who are on an antiretroviral regimen and have suboptimal viral suppression (ie, viral loads > 1000 copies/mcL). Both genotypic and phenotypic tests are commercially available and in randomized controlled studies their use has been shown to result in improved short-term virologic outcomes compared to making treatment choices without resistance testing. Furthermore, multiple retrospective studies have conclusively demonstrated that resistance tests provide prognostic information about virologic response to newly initiated therapy that cannot be gleaned from standard clinical information (ie, treatment history, examination, CD4 count, and viral load tests).
Because of the complexity of resistance tests, many clinicians require expert interpretation of results. In the case of genotypic assays, results may show that the mutations that are selected for during antiretroviral therapy are drug-specific or contribute to broad cross-resistance to multiple drugs within a therapeutic class. An example of a drug-specific mutation for the reverse transcriptase inhibitors would be the M184V mutation that is selected for by lamivudine or emtricitabine therapy this mutation causes resistance only to those two drugs. Conversely, the thymidine analog mutations ( TAMs ) of M41L, D67N, K70R, L210W, T215Y/F, and T219Q/K/E are selected for by either zidovudine or stavudine therapy, but cause resistance to all the drugs in the class and often extend to the nucleotide inhibitor tenofovir when three or more of these TAMs are present. Further complicating the interpretation of genotypic tests is the fact that some mutations that cause resistance to one drug can actually make the virus that contains this mutation more sensitive to another drug. The M184V mutation, for example, is associated with increased sensitivity to zidovudine, stavudine and tenofovir. Figures 31-3, 31-4, and 31-5 show the most common mutations associated with drug resistance and cross-resistance patterns for NRTIs, NNRTIs, and PIs. Phenotypic tests also require interpretation in that the distinction between a resistant virus and sensitive one is not fully defined for all available drugs.
 |
Figure 31-3. Most common mutations associated with drug resistance and cross-resistance with nucleotide reverse transcriptase inhibitors. AZT = zidovudine; d4T = stavudine; TDF = tenofovir; ABC = abacavir; ddI = didanosine; 3TC = lamivudine; FTC = emtricitabine. (Reproduced, with permission, from Stanford HIV Drug Resistance Database, http://www.hivdb.stanford.edu.) |
Both methods of resistance testing are limited by the fact that they may measure resistance in only some of the viral strains present in an individual. Resistance results may also be misleading if a patient is not taking antiretroviral medications at the time of testing. Thus, resistance results must be viewed cumulatively ie, if resistance is reported to an agent on one test, it should be presumed to be present thereafter even if subsequent tests do not give the same result.
C. Hematopoietic Stimulating Factors
Epoetin alfa (erythropoietin) is approved for use in HIV-infected patients with anemia, including those with anemia secondary to zidovudine use. It has been shown to decrease the need for blood transfusions. The drug is expensive, and an endogenous erythropoietin level < 500 mU/mL should be demonstrated before starting therapy. The starting dose is 8000 units
P.1374
subcutaneously three times a week. The target hematocrit is 35 40%. The dose may be increased by 12,000 units every 4 6 weeks as needed to a maximum dose of 48,000 units per week. Hypertension is the most common side effect.
Human G-CSF (filgrastim) and granulocyte-macrophage colony-stimulating factor (GM-CSF [sargramostim]) have been shown to increase the neutrophil counts of HIV-infected patients. G-CSF is preferred because of the theoretical concern of GM-CSF-stimulating HIV replication in infected monocytes. In patients receiving cytotoxic chemotherapy for lymphoma or Kaposi's sarcoma, daily subcutaneous doses of G-CSF at approximately 5 mcg/kg (a 300 mcg or 480 mcg vial, depending on weight) are given beginning 5 7 days after chemotherapy until the neutrophil count has rebounded to above 1000/mcL. G-CSF may also have a role in ameliorating neutropenia caused by
P.1375
other drugs such as zidovudine or ganciclovir, when other therapeutic alternatives are not possible. Because the cost of this therapy is approximately $150 per vial, dosage should be closely monitored and minimized, aiming for a neutrophil count of 1000/mcL. When the drug is used for indications other than cytotoxic chemotherapy, one or two doses at 5 mcg/kg per week are usually sufficient.
 |
Figure 31-4. Most common mutations associated with drug resistance and cross-resistance patterns for nonnucleoside reverse transcriptase inhibitors. NVP = nevirapine; DLV = delavirdine; EFV = efavirenz. (Reproduced, with permission, from Stanford HIV Drug Resistance Database, http://www.hivdb.stanford.edu.) |
 |
Figure 31-5. Most common mutations associated with drug resistance and cross-resistance patterns for protease inhibitors. NFV = nelfinavir; SQV = saquinavir; IDV = indinavir; RTV = ritonavir; fAPV = fosamprenavir; LPV = lopinavir; ATV = atazanavir. (Reproduced, with permission, from Stanford HIV Drug Resistance Database, http://www.hivdb.stanford.edu.) |
D. Prophylaxis of Opportunistic Infections
In general, decisions about prophylaxis of opportunistic infections are based on the CD4 count, other evidence of severe immune suppression (eg, oral candidiasis), and a history of having had the infection in the past. In the era prior to HAART, patients started on prophylactic regimens were maintained on them indefinitely. However, studies have shown that in patients with robust improvements in immune function as measured by increases in CD4 counts above the levels that are used to initiate treatment prophylactic regimens can safely be discontinued.
Primary prophylaxis for Pneumocystis pneumonia should be offered to patients with CD4 counts below 200 cells/mcL, a CD4 lymphocyte percentage below 14%, or weight loss or oral candidiasis. Patients with a history of Pneumocystis pneumonia should receive secondary prophylaxis until they have had a durable virologic response to HAART for at least 3 6 months and maintain a CD4 count of > 250 cells/mcL.
Regimens for prophylaxis are trimethoprim-sulfamethoxazole, dapsone, atovaquone, and aerosolized pentamidine (see Table 31-6). Trimethoprim-sulfamethoxazole is inexpensive, widely available, and the most effective agent for prophylaxis. In two studies comparing once-daily double-strength trimethoprim-sulfamethoxazole with aerosolized pentamidine for primary and secondary prophylaxis of Pneumocystis pneumonia, patients randomized to trimethoprim-sulfamethoxazole were significantly less likely to develop Pneumocystis pneumonia. Bacterial infections (eg, pneumonia, sinusitis) are also less likely to develop in patients who receive trimethoprim-sulfamethoxazole. Common side effects with trimethoprim-sulfamethoxazole are primarily fever, rash, and nausea and vomiting. Trimethoprim-sulfamethoxazole is dosed as one double-strength tablet three times a week to once daily. Patients in whom mild rashes develop on this regimen may be treated with diphenhydramine (25 50 mg every 4 hours). However, clinicians and patients must watch carefully for signs of Stevens-Johnson syndrome. Some clinicians are also using desensitization regimens to overcome allergic reactions. Reports suggest that desensitization may be successful in 40% of cases. Dapsone is a second-line prophylactic agent with minimal side effects. As is the case with trimethoprim-sulfamethoxazole, it is inexpensive and widely available. It may be used in patients with an allergic reaction to trimethoprim-sulfamethoxazole. Before prescribing dapsone, clinicians
P.1376
should make certain that the patient is not glucose-6-phosphate dehydrogenase deficient, since such patients are at high risk for developing hemolytic anemia with dapsone therapy. Patients taking dapsone concomitantly with didanosine should take the dapsone at least 2 hours prior to the didanosine since dapsone is not absorbed well in the neutral pH stomach environment created by the didanosine buffering agent. Finally, atovaquone may be an option for individuals intolerant of other systemic therapies, but it appears to be less effective. It is available only as an oral suspension (1500 mg daily) and must be taken with food to promote absorption.
Table 31-6. Pneumocystis jiroveci prophylaxis. | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Finally, aerosolized pentamidine can be used if no systemic agents can be tolerated. Its disadvantages are expense (approximately $100 for one treatment per month) and decreased effectiveness in the apical and peripheral areas of the lung. Cases of extrapulmonary Pneumocystis infections in patients receiving aerosolized pentamidine have also been reported.
Aerosolized pentamidine may also increase the incidence of pneumothorax in patients with a history of Pneumocystis infection, although this complication is rarely seen in the current treatment era.
Patients who develop Pneumocystis infection on a particular prophylactic regimen should be switched to a different one.
Prophylaxis against M avium complex infection should be given to patients whose CD4 counts fall below 75 100 cells/mcL. Clarithromycin (500 mg orally twice daily) and azithromycin (1200 mg orally weekly) have both been shown to decrease the incidence of disseminated disease by approximately 75%, with a low rate of breakthrough of resistant disease. The latter regimen is generally preferred on the basis of ease of compliance and cost. Adding rifabutin increases the toxicity of the regimen but does not significantly increase its efficacy and is therefore not recommended. As sole therapy, rifabutin (300 mg orally daily) is less effective and more toxic than clarithromycin or azithromycin. Clinicians should make certain that patients do not have active M tuberculosis infection by examination of a chest radiograph prior to starting rifabutin because of concern about the development of resistance to rifabutin with cross-resistance to rifampin. Similarly, before initiating prophylaxis, clinicians should establish with a blood culture that the patient does not have disseminated M avium complex infection. Common side effects of both azithromycin and clarithromycin are nausea and diarrhea. Two common side effects with rifabutin are rash and hepatic dysfunction. Rifabutin may induce hepatic enzymes, thereby decreasing the activity of some drugs metabolized by the liver.
Prophylaxis against M avium complex infection may be discontinued among patients whose CD4 counts go above 100 cells/mcL in response to HAART and whose plasma viral load has been optimally suppressed to < 50 75 copies/mL.
Prophylaxis against M tuberculosis infection isoniazid 300 mg daily plus pyridoxine 50 mg orally daily for 9 months to a year should be given to all HIV-infected patients with positive PPD reactions (defined for HIV-infected patients as > 5 mm of induration).
Toxoplasmosis prophylaxis is desirable in patients with positive IgG toxoplasma serology and CD4 counts below 100 cells/mcL. Trimethoprim-sulfamethoxazole (one double-strength tablet daily) offers good protection against toxoplasmosis, as does a combination of pyrimethamine, 25 mg orally once a week, plus dapsone, 50 mg orally daily, plus leucovorin, 25 mg orally once a week.
CMV infection is also common in late HIV disease. Oral ganciclovir (1000 mg orally three times daily with food) is approved for CMV prophylaxis among HIV-infected persons with advanced disease (eg, CD4 counts below 50 cells/mcL). However, because the drug causes neutropenia, it is not widely used. Clinicians should consider performing serum CMV IgG antibody testing. Persons who are CMV IgG-negative are not at risk for development of CMV disease. Importantly, patients
P.1377
who are CMV IgG-negative should receive CMV-negative blood if they require a transfusion. Because over 99% of gay men are positive for CMV IgG, it is appropriate to reserve testing for heterosexuals with HIV.
Cryptococcosis, candidiasis, and endemic fungal diseases are also candidates for prophylaxis. One prophylactic trial showed a decreased incidence of cryptococcal disease with the use of fluconazole, 200 mg orally daily, but the treated group had no benefit in terms of mortality. Fluconazole (200 mg orally once a week) was found to prevent oral and vaginal candidiasis in women with CD4 counts below 300 cells/mcL. In areas of the world where histoplasmosis and coccidioidomycosis are endemic and are frequent complications of HIV infection, prophylactic use of fluconazole or itraconazole may prove to be useful strategies. However, the problem of identifying individuals at highest risk makes the targeting of prophylaxis difficult.
Because individuals with advanced HIV infection are susceptible to a number of opportunistic pathogens, the use of agents with activity against more than one pathogen is preferable. It has been shown, for example, that trimethoprim-sulfamethoxazole confers some protection against toxoplasmosis in individuals receiving this drug for Pneumocystis prophylaxis.
Course & Prognosis
With improvements in therapy, patients are living longer after the diagnosis of AIDS. This has resulted in dramatic decreases in AIDS deaths nationally. In 2004 in the United States there were 15,798 deaths due to AIDS, compared with 50,610 deaths in 1995. It remains to be seen whether the decreases in number of deaths can be sustained over the long term. Maintaining access to quality care and treatment is one key element. Unfortunately, studies continue to show less access to treatment for some underserved groups, especially blacks, the homeless, and injection drug users. Another key element in sustaining lower mortality is developing new treatments for patients who have been heavily treated with existing agents. Despite new therapeutic options, people continue to die of HIV infection. For patients whose disease progresses even though they are receiving appropriate treatment, meticulous palliative care must be provided (see Chapter 5), with attention to pain control, spiritual needs, and family (biologic and chosen) dynamics.
Bamberger JD et al: Helping the urban poor stay with antiretroviral HIV drug therapy. Am J Pub Health 2000;90:699.
Benson CA: Treating opportunistic infections among HIV-infected adults and adolescents: recommendations from CDC, the National Institutes of Health, and the HIV Medicine Association/Infectious Diseases Society of America. MMWR Recomm Rep 2004;53(RR-1):1.
El-Sadr WM et al: Discontinuation of prophylaxis for Mycobacterium avium complex disease in HIV-infected patients who have a response to antiretroviral therapy. N Engl J Med 2000;342:1085.
Gallant JE et al; Study 934 Group: Tenofovir DF, emtricitabine, and efavirenz vs.zidovudine, lamivudine, and efavirenz for HIV. N Engl J Med 2006;354:251.
Gallant JE et al; 903 Study Group: Efficacy and safety of tenofovir DF vs stavudine in combination therapy in antiretroviral-na ve patients: a 3-year randomized trial. JAMA 2004;292:191.
Grinspoon S et al: Cardiovascular risk and body-fat abnormalities in HIV-infected adults. N Engl J Med 2005;352:48.
Hirsch MS et al: Antiretroviral drug resistance testing in adults infected with human immunodeficiency virus type 1: 2003 recommendations of an International AIDS Society-USA panel. Clin Infect Dis 2003;37:113.
Lazzarin A et al: Efficacy of enfuvirtide in patients infected with drug-resistant HIV-1 in Europe and Australia. N Engl J Med 2003;348:2186.
Levine AM et al: Evaluation and management of HIV-infected women. Ann Intern Med 2002;136:228.
Martin DF et al: A controlled trial of valganciclovir as induction therapy for cytomegalovirus retinitis. N Engl J Med 2002;346:1119.
Robbins GK et al: Comparison of sequential three-drug regimens as initial therapy for HIV-1 infection. N Engl J Med 2003;349:2293.
Shafran SD et al: Successful discontinuation of therapy for disseminated Mycobacterium avium complex infection after effective antiretroviral therapy. Ann Intern Med 2002;137:734.
Stringer JR et al: A new name (Pneumocystis jiroveci) for Pneumocystis from humans. Emerg Infect Dis 2002;8:891.
Vincent I et al: Modalities of palliative care in hospitalized patients with advanced AIDS. AIDS Care 2000;12:211.
Yeni PG et al: Treatment for adult HIV infection: 2004 recommendations of the International AIDS Society-USA Panel. JAMA 2004;292:251.
EAN: 2147483647
Pages: 49