2 - Common Symptoms
Editors: McPhee, Stephen J.; Papadakis, Maxine A.; Tierney, Lawrence M.
Title: Current Medical Diagnosis & Treatment, 46th Edition
Copyright 2007 McGraw-Hill
> Table of Contents > 5 - Care at the End of Life
function show_scrollbar() {}
5
Care at the End of Life
Michael W. Rabow MD
Steven Z. Pantilat MD
The End of Life
The Definition of the End of Life
In the United States, approximately 2.4 million people die each year. While death itself remains a mystery and while caring for the dying traditionally has not been well researched or adequately taught as part of medical training, caring for patients at the end of life is an important responsibility and a rewarding opportunity for clinicians. Clinicians battling to prolong life must recognize when life is ending in order to continue caring properly for their patients. Unfortunately, end-of-life practices do not always meet the standards set by professional organizations and most clinical practice guidelines do not include significant attention to end-of-life care. End-of-life care refers to focusing care for those approaching death on the goals of relieving distressing symptoms and promoting quality of life rather than attempting to cure underlying disease. From the medical perspective, the end of life may be defined as that time when death whether due to terminal illness, acute or chronic illness, or age itself is expected within weeks to months and can no longer be reasonably forestalled by medical intervention. Yet, the palliative approach to caring for patients facing the end of life outlined in this chapter applies equally well to any patient with a serious, chronic illness at any point in their illness trajectory and should be provided simultaneously with all other appropriate medical treatments.
Prognosis at the End of Life
Clinicians play an important role in helping patients understand that their lives are ending. This information influences patients' treatment decisions and may change how they spend their remaining time. While certain diseases such as cancer are more amenable to prognostic estimates regarding the time course to death, the other common causes of mortality in the United States including heart disease, stroke, chronic lung disease, and dementia have more variable trajectories and difficult to predict prognoses (Figure 5-1). Even for patients with cancer, clinician estimates of prognosis are often inaccurate and generally overly optimistic. Nonetheless, clinical experience, epidemiologic data, guidelines from professional organizations,* and computer modeling and prediction tools may be used to help patients identify the end period of their lives. Clinicians can also ask themselves Would I be surprised if this patient died in the next year? to determine whether a discussion of prognosis and provision of end-of-life care would be appropriate. If the answer is no, then the clinician should initiate a discussion. Recognizing that patients may have different levels of comfort with prognostic information, clinicians can introduce the topic by simply saying, I have information about the likely time course of your illness. Would you like to talk about it?
Expectations about the End of Life
Patients' experiences of the end of life are influenced by their expectations about how they will die and the meaning of death. Many people fear how they will die more than death itself. Patients report fear of dying in pain or of suffocation, of loss of control, indignity, isolation, and being a burden to their families. All of these anxieties may be alleviated with good supportive care provided by an attentive group of caretakers.
Since medical advances can often forestall the end of life, death has become medicalized. No longer seen clearly as a natural and profound personal and spiritual event basic to the human condition, death is often regarded by clinicians, patients, and families as a failure of medical science. This attitude can create or heighten a sense of guilt about the failure to prevent dying. Both the general public and clinicians are complicit in denying death, treating dying persons as patients and death as an enemy to be battled furiously in hospitals rather than as an inevitable outcome to be experienced as a part of life at home. As a result, approximately 75 80% of people in the United States die in hospitals or long-term care facilities.
The clinician may continue to pursue cure of potentially reversible disease for some patients. For all, however, offering comfort and helping the patient prepare for death are foremost considerations. Patients at the end of life and their families identify a number of elements
P.69
as important to quality end-of-life care: adequate pain and symptom management, avoiding inappropriate prolongation of dying, preserving dignity, preparing for death, achieving a sense of control, relieving the burden on others, and strengthening relationships with loved ones. Clinicians can help patients pursue their care goals in a process called advance care planning.
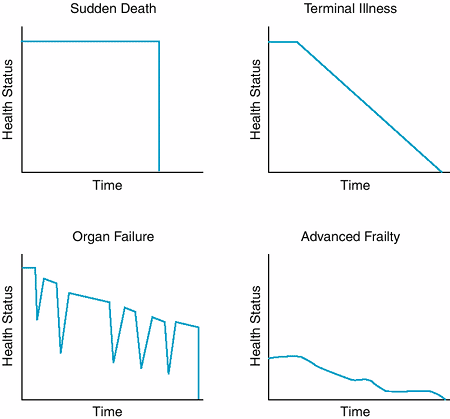 |
Figure 5-1. Theoretical trajectories of dying (Adapted, with permission, from Lunney JR et al: Profiles of older Medicare decedents. J Am Geriatr Soc 2002;50:1108. ) |
Communication & Care of the Patient
Caring for patients at the end of life requires the same skills clinicians use in other tasks of medical care: diagnosing treatable conditions, providing patient education, facilitating decision-making, and expressing understanding and caring. Communication skills are vitally important. In particular, clinicians must become experts at delivering bad news and then dealing with its consequences (Table 5-1). Higher-quality communication is associated with greater satisfaction and awareness of patient wishes. Three further obligations are central to the clinician's role at this time. First, he or she must work to identify, understand, and relieve suffering, which may include physical, psychological, social, or spiritual distress. Disease and disability at the end of life can threaten a person's sense of integrity or intactness and thereby cause suffering. In assisting with redirection and growth, providing support, assessing meaning, and fostering transcendence, clinicians can help ameliorate their patients' suffering and help the patient live fully during this stage of life. Second, clinicians can serve as facilitators or catalysts for hope. While a particular outcome may be extremely unlikely (such as cure of advanced cancer following exhaustive conventional and experimental treatments), hope may be defined as the patient's belief in what is still possible. Although expecting a miraculous cure may be simplistic and even harmful, hope for relief of pain, for reconciliation with loved ones, for discovery of meaning, and for spiritual transformation is realistic at the end of life. With questions such as What is still possible now for you? What do you wish for before you die? What good might come of this? clinicians can help patients uncover hope, explore meaningful and realistic goals, and develop strategies to realize them.
Third, dying patients' feelings of isolation and fear demand that clinicians assert that care will continue to be provided throughout the final stage of life. The promise of nonabandonment is perhaps the central principle of end-of-life care and is a clinician's pledge to an individual patient to serve as a caring partner, a resource for creative problem-solving and relief of suffering,
P.70
a guide during uncertain times, and a witness to the patient's experiences no matter what happens. Clinicians can say to a patient, I will care for you whatever happens. Dying patients need their clinicians to offer their presence not necessarily the ability to solve all problems but rather a commitment to recognize and receive the patients' difficulties and experiences with respect and empathy. At its best, the patient-clinician relationship can be a covenant of compassion and a recognition of common humanity.
Table 5-1. Suggestions for the delivery of bad news. | |
|---|---|
|
Caring for The Family
In caring for patients at the end of life, clinicians must appreciate the central role played by family, friends, and romantic partners and often must deal with strong emotions of fear, anger, shame, sadness, and guilt experienced by those individuals. While significant others may support and comfort a patient at the end of life, the threatened loss of a loved one may also create or reveal dysfunctional or painful family dynamics. Furthermore, clinicians must be attuned to the potential impact of illness on the patient's family: substantial physical caregiving responsibilities and financial burdens as well as increased rates of anxiety, depression, chronic illness, and even mortality. Family caregivers, typically women, commonly provide the bulk of care for patients at the end of life, yet their work is often not acknowledged or compensated.
Clinicians can help families confront the imminent loss of a loved one (Table 5-2) and often must negotiate amid complex and changing family needs. Identifying a spokesperson for the family, conducting family meetings, allowing all to be heard, and providing time for consensus may help the clinician work effectively with the family.
Clinician Self-care
Many clinicians find caring for patients at the end of life to be one of the most rewarding aspects of practice. However, working with the dying requires tolerance of great uncertainty, ambiguity, and existential challenges. Clinicians must recognize and respect their own limitations and attend to their own needs in order to avoid being overburdened, overly distressed, or emotionally depleted. Open recognition of their own feelings enables clinicians to process their emotions and take steps to care for themselves: conferring and consulting with colleagues, retreating, relaxing and recuperating, obtaining informal or professional support, or even under extraordinary circumstances transferring the care of a patient to another clinician when it is no longer possible for the original clinician to meet the patient's needs. Moreover, care of patients at the end of life is not solely the responsibility of physicians. Ideally, physicians, nurses, social workers, chaplains, pharmacists, and other clinicians should coordinate their efforts as part of an interdisciplinary team to care for patients and support one another.
Table 5-2. Clinician behaviors helpful to families of dying patients. | ||
|---|---|---|
|
Footnotes
*For example, the National Hospice Organization.
For example, the Acute Physiology and Chronic Health Evaluation (APACHE) system, the Study to Understand Prognoses and Preferences for Outcomes and Risks of Treatment (SUPPORT) model, or the Palliative Performance Scale.
Lamont EB et al: Complexities in prognostication in advanced cancer: To help them live their lives the way they want to. JAMA 2003;290:98.
Lunney JR et al: Patterns of functional decline at the end of life. JAMA 2003;289:2387.
Mast KR et al: End-of-life content in treatment guidelines for life-limiting diseases. J Palliat Med 2004;7:754.
Mularski RA et al: Quality of dying in the ICU: ratings by family members. Chest 2005;128:28.
Rabow MW et al: Supporting family caregivers at the end of life: they don't know what they don't know. JAMA 2004; 291:483.
Stevens LM et al: JAMA patient page. Palliative care. JAMA 2005; 293:1410.
The Setting & Structure of Care
Ethical & Legal Background
Clinicians may be limited in caring for persons at the end of life not only by their emotional responses but by a sense of moral obligation as well. While the ethical, legal, and professional controversies over assisted suicide are beyond the scope of this chapter, clinicians should be aware of the right to die movement as an expression, at least in part, of patient dissatisfaction with how people are cared for at the end of life. In the United States, assisted suicide is illegal in every state but Oregon and legal there only with careful restrictions. While individual clinicians must decide for themselves within the evolving legal context what their personal limits may be in caring for patients who request aid in dying (eg, assisted suicide), all clinicians can reclaim their long-privileged and universally accepted role of caring for the dying. They can do so by dedicating themselves not to abandon their patients and by providing appropriate attention to symptom
P.71
management, sensitivity to psychological and social stresses, and unconditional presence and openness to spiritual challenges at the end of life. Research has demonstrated that palliative care interventions can cause some patients who have requested assisted suicide to withdraw their request.
Clinicians' care of patients at the end of life is guided by the same ethical and legal principles that inform other types of medical care. Foremost are truth-telling, nonmaleficence, beneficence, autonomy, confidentiality, proportionality, and distributive justice. These principles must guide clinicians in helping patients make difficult decisions about care, including decisions about palliative sedation and the withdrawal and withholding of life-prolonging interventions.
Important ethical principles may be in conflict. For example, while a patient may desire a particular medical intervention, the clinician may decline to undertake the intervention if it is of no therapeutic benefit (ie, futile) or violates the clinician's own moral code. Clinicians must use caution in invoking futility, since what constitutes futility is often a matter of controversy. Studies confirm that most disagreements can be resolved through repeated discussions between clinicians and families. Although clinicians and family members often feel differently about withholding versus withdrawing support, there is consensus among ethicists, supported by legal precedent, of their ethical equivalence. Patients and their surrogates have the same right to stop unwanted medical treatments once begun as they do to refuse those treatments in the first place, including nutrition and hydration. The ethical principle of double effect argues that the potential to hasten imminent death is acceptable if it comes as the unintended consequence of a primary intention to provide comfort and relieve suffering. For example, sufficient doses of morphine should be provided to control pain even if there is the potential unintended secondary effect of depressing respiration. The ethical argument for the practice of palliative sedation is based on this principle. In practice, one can almost always find an effective pain regimen without rendering a patient unconscious.
Decision-Making, Advance Care Planning, & Advance Directives
Well-informed, competent adults have a right to refuse medical intervention even if this is likely to result in death. Many are willing to sacrifice some quantity of life in exchange for protecting a certain quality of life. In order to further patient autonomy, clinicians are obligated to inform patients about the risks, benefits, alternatives, and expected outcomes of end-of-life medical interventions such as cardiopulmonary resuscitation (CPR), intubation and mechanical ventilation, vasopressor medication, hospitalization and ICU care, and nutrition and hydration. Advance directives are oral or written statements made by patients when they are competent that are intended to guide care should they become incompetent. Advance directives allow patients to project their autonomy into the future and are an important part of advance care planning a process whereby patients consider various paths their illness may take and contingencies for responding to these possible developments based on their care goals and values. While oral statements about these matters are ethically binding, they are not legally binding in all states. State-specific advance directive forms are available from a number of sources, including the Web site http://www.caringinfo.org.
In addition to documenting patient preferences for care, the Durable Power of Attorney for Health Care (DPOA-HC) allows the patient to designate a surrogate decision-maker. The DPOA-HC is important since it is often difficult to anticipate what decisions will need to be made. The responsibility of the surrogate is to provide substituted judgment to decide as the patient would, not as the surrogate wants. In the absence of a designated surrogate, clinicians usually turn to family members or next of kin. Unfortunately, surveys demonstrate that clinicians and families often are no better than chance at predicting patient wishes, so it is imperative to have these discussions with all patients.
Clinicians should educate all patients ideally, well before the end of life about the opportunity to formulate an advance directive. Most patients have already thought about end-of-life issues, want to discuss them with their clinician, want the clinician to bring up the subject, and feel better for having had the discussion. Despite regulations requiring health care institutions to inform patients of their rights to formulate an advance directive, only about 10% of people in the United States (including clinicians themselves) actually have completed them, and studies have shown that clinicians are often unaware of or actually ignore their patients' advance directives.
DNAR Orders
As part of advance care planning, clinicians can encourage patients to express their preferences for the use of CPR. Most patients and many clinicians are uninformed or misinformed about the nature and success of CPR. Only about 15% of all patients who undergo CPR in the hospital survive to hospital discharge. Moreover, among certain populations especially those with serious systemic noncardiac disease, metastatic cancer, and sepsis the likelihood of survival to hospital discharge following CPR is virtually nil.
Patients may ask their clinician to write an order that CPR not be attempted on them. Although this order initially was referred to as a DNR ( do not resuscitate ) order, many clinicians prefer the term DNAR ( do not attempt resuscitation ) to emphasize the low likelihood of success.
Patients deciding about CPR preferences should also be informed about the sequelae of surviving CPR. It may result in fractured ribs, lacerated internal organs, and neurologic disability, and there is a high
P.72
likelihood of a need for other aggressive interventions, such as ICU care, if CPR is successful.
For some patients at the end of life, decisions about CPR may be not about whether they will live but about how they will die. Clinicians should correct the misconception that withholding CPR in appropriate circumstances is tantamount to not doing everything or just letting someone die. Typically, CPR does not improve the quality of a dying patient's life or alter the patient's underlying prognosis. While respecting the patient's right ultimately to make the decision and keeping in mind their own biases and prejudices clinicians should offer explicit recommendations about DNAR orders and protect dying patients and their families from feelings of guilt and from the sorrow associated with vain hopes. Finally, clinicians should encourage patients and their families to make proactive decisions about what is wanted in end-of-life care rather than focusing only on what is not to be done.
Hospice & Other Palliative Care Institutions
While most patients die in hospitals and while the number of hospital-based palliative care services is growing, good care of the dying may not be the central goal of most hospitals. Hospice is an approach to end-of-life care where the most urgent objective is to address the physical and emotional needs of the dying. Hospice care focuses on the patient and family rather than the disease and on providing comfort and pain relief rather than on treating illness or prolonging life. Hospices provide intensive caring with the goal of helping people live well until they die.
Hospice embodies the palliative care philosophy that emphasizes individualized attention, human contact, and an interdisciplinary team approach. Hospice care can include arranging for respite for family caregivers and providing legal, financial, and other services. While some hospice care is provided in hospitals and institutional residences, about 80% of patients receiving hospice care remain at home where they can be cared for by the family and visiting hospice staff. Primary care clinicians are strongly encouraged to continue caring for their patients during the time they are receiving hospice care.
Hospice care is highly rated by families and has been shown to increase patient satisfaction, to reduce costs (depending on when patients are referred to hospice care), and even to decrease family caregiver mortality. More than 25% of all patients who die in the United States receive hospice care, and about 50% of these people have end-stage cancer. Hospice care tends to be utilized late in the course of the end of life. The median length of stay in hospices in the United States is just 26 days, with 34% of patients dying within 7 days after beginning hospice care.
Most hospice organizations require clinicians to estimate the patient's probability of survival to be less than 6 months, since this is a criterion for eligibility to receive Medicare or other insurance coverage. Regrettably, the hospice benefit can be difficult to provide to people who are homeless or isolated or who have terminal prognoses that are difficult to quantify.
While the initiation of hospice care is often described as a transition from aggressive care to comfort care, hospice care also provides aggressive care, though not directed at achieving a cure or prolonging life. It is more appropriate to consider hospice care as one among many health care resources available to patients at the end of life. For the dying, it may be appropriate to treat pneumonia with morphine and antipyretics rather than with antibiotics. Helping patients decide when to avail themselves of the resources of hospice or inpatient palliative care is an important function even for clinicians providing the most aggressive and intensive curative medical interventions. While most people prefer to die at home, inpatient palliative care may be more appropriate for patients with complex, severe symptoms that are difficult to manage at home.
While the numbers of Americans dying in nursing homes is increasing, many long-term care facilities do not have staff trained in palliative care or provide formal palliative care services. Increasingly, however, long-term care facilities are recognizing their need to become experts in end-of-life care and a number of quality improvement programs show promise.
Cultural Issues
The individual's experience of dying occurs in the context of a complex interaction of personal, philosophic, and cultural influences. Various religious, ethnic, gender, class, and cultural traditions inform patients' styles of communication, comfort in discussing particular topics, expectations about dying and medical interventions, and attitudes about the appropriate disposition of dead bodies. There are differences in knowledge and beliefs regarding advance directives, autopsy, organ donation, hospice care, and withdrawal of support among patients of different ethnic groups. Clinicians must appreciate recent evidence suggesting that palliative care is susceptible to some of the same racial biases previously documented in other medical disciplines. While each patient must be considered an individual, understanding a person's cultural assumptions and beliefs and respecting ethnic traditions are important responsibilities of the clinician caring for a patient at the end of life, especially when the cultures of origin of the clinician and patient differ. A clinician may ask a patient, What do I need to know about you and your beliefs that will help me take care of you?
Christakis NA et al: The health impact of health care on families: a matched cohort study of hospice use by decedents and mortality outcomes in surviving, widowed spouses. Soc Sci Med 2003;57:465.
Hanson LC et al: Meeting palliative care needs in post-acute care settings: to help them live until they die. JAMA 2006; 295:681.
P.73
Lo B et al: Palliative sedation in dying patients: we turn to it when everything else hasn't worked. JAMA 2005;29:1810.
Morrison RS et al: The growth of palliative care programs in United States hospitals. J Palliat Med 2005;8:1127.
Teno JM et al: Family perspectives on end-of-life care at the last place of care. JAMA 2004;291:88.
Tulsky JA: Beyond advance directives: importance of communication skills at the end of life. JAMA 2005;294:359.
Weissman DE: Decision making at a time of crisis near the end of life. JAMA 2004;292:1738.
Management of Pain & Other Common Symptoms
For patients at the end of life, maximizing the quality of life rather than postponing death is often the first priority of care. In this context, symptoms that cause disability and suffering must be considered medical emergencies and managed aggressively by frequent elicitation, continuous reassessment, and individualized treatment. While patients at the end of life may experience a host of distressing symptoms, pain, dyspnea, and delirium are reported to be among the most feared and burdensome. The palliative care of pain and a selected number of other common symptoms is described below. Throughout, the principles of good end-of-life care dictate that comfort is the main focus of palliative care and that properly informed patients or their surrogates may decide to pursue aggressive symptom relief even if, as a known but unintended consequence, the treatments hasten demise or preclude curative attempts. There is even a growing awareness that scrupulous symptom control for patients with end-stage illness may prolong life.
Pain at the End of Life
Definition & Prevalence
Pain is a common problem for patients at the end of life up to 75% of patients dying of cancer experience pain and it is what many people say they fear most about dying. Pain is a common complaint among patients with noncancer diagnoses as well. Pain is undertreated at the end of life. Up to 50% of severely ill hospitalized patients spent half of their time during the last 3 days of life in moderate to severe pain. The Joint Commission on Accreditation of Healthcare Organizations (JCAHO) includes pain management standards for all patient care organizations it accredits.
The experience of pain includes the patient's emotional reaction to it and is influenced by many factors, including the patient's prior experiences with pain, the meaning of the pain, emotional stresses, and the influence of family and culture. Pain is a subjective phenomenon, and clinicians cannot reliably detect its existence or quantify its severity without asking the patient directly. A useful means of assessing pain and evaluating the effectiveness of analgesia is to ask the patient to rate the degree of pain along a numerical or visual pain scale (Table 5-3).
Barriers to Good Care
Poor management of pain at the end of life has been documented in many settings. Some clinicians refer pain management to others when they believe that a patient's pain is not due to the disease for which they are treating the patient. Even oncologists often misperceive the origin of their patients' pain and inappropriately ignore complaints of pain.
Many clinicians have limited training and clinical experience with pain management and thus are understandably reluctant to attempt to manage severe pain. Lack of knowledge about the proper selection and dosing of analgesic medications carries with it attendant and typically exaggerated fears about the side effects of pain medications, including the possibility of respiratory depression with an overdose of opioids. Most clinicians, however, can develop good pain management skills, and nearly all pain, even at the end of life, can be managed without hastening death through respiratory depression.
Fears of the physiologic effects of opioids are often coupled with concerns on the part of clinician, patient, or family that patients will become addicted to opioid pain medications. While physiologic tolerance (requiring increasing dosage to achieve the same analgesic effect) and dependence (requiring continued dosing to prevent symptoms of medication withdrawal) are expected with opioid use, at the end of life the use of opioids for relief of pain and dyspnea is not associated with a risk of psychological addiction (misuse of a substance for purposes other than one for which it was prescribed and despite negative consequences in health, employment, or legal and social spheres). Even patients who demonstrate some of the behaviors sometimes associated with addiction (demand for specific medications and doses, anger and irritability, poor cooperation or disturbed interpersonal reactions) may in fact not be addicted. The term pseudo-addiction has been used when patients exhibit behaviors associated with addiction but only because their pain is inadequately treated. Once they achieve pain relief, these behaviors cease. In all cases, clinicians must be willing to use appropriate doses of opioids in order to relieve distressing symptoms for patients at the end of life.
Finally, some clinicians fear legal repercussions from prescribing the high doses of opioids sometimes necessary to control pain at the end of life. Some states have enacted special licensing and documentation requirements for opioid prescribing. However, governmental and professional medical groups, regulators, and the US Supreme Court are making it clear that appropriate treatment of pain is the right of the patient and a fundamental responsibility of the clinician. In fact, clinicians have been successfully sued for undertreatment of pain. Although clinicians may feel trapped between consequences
P.74
of overprescribing or underprescribing opioids, there remains a wide range of practice in which clinicians can appropriately treat pain. Referral to pain management experts is appropriate whenever pain cannot be controlled expeditiously by the primary clinician.
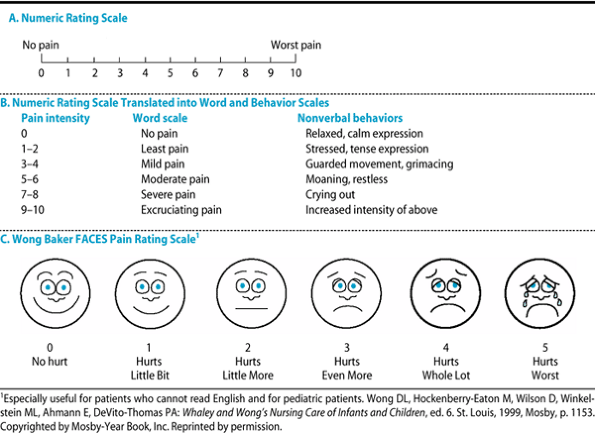 |
Table 5-3. Pain assessment scales. |
Principles of Pain Management
General guidelines for management of pain are recommended for the treatment of all patients with pain (Table 5-4). Because pain is so common at the end of life, all patients should be asked about its presence. Clinicians should ask about the nature, severity, timing, location, quality, and aggravating and relieving factors of the pain. Distinguishing between somatic, visceral, and neuropathic pain is essential to proper tailoring of pain treatments. The goal of pain management is properly decided by the patient. Some patients may wish to be completely free of pain even at the cost of significant sedation, while others will wish to control pain to a level that still allows maximal functioning.
Chronic severe pain should be treated continuously. For ongoing pain, one can give a long-acting analgesic around the clock plus a short-acting drug as needed for breakthrough pain. At the end of life, the oral route of administration is preferred because it is easier to administer at home, is not painful, and imposes no risk from needle exposure. Rectal, transdermal, and subcutaneous administration are also frequently used, as is intravenous or intrathecal administration when necessary. Patient-controlled analgesia (PCA) of intravenous medications is appreciated by patients, may lead to less medication use, and has been adapted for use with oral administration.
When possible, the cause of pain should be diagnosed and treated, assuming that the burden of these efforts does not increase the patient's suffering. Removing the underlying cause of pain can preempt the need for ongoing treatment with analgesic medications along with their side effects. Regardless of decisions about seeking and treating the underlying cause of pain, however, prompt symptomatic relief of pain should be offered to every patient.
Pharmacologic Pain Management Strategies
Typically, pain can be well controlled with analgesic medications both opioid and nonopioid. Evidence-based summaries and guidelines are available from the Agency for Healthcare Research and Quality (http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat1.chapter.86205) and other organizations (http://www.nccn.org/professionals/physician_gls/PDF/pain.pdf).
P.75
For mild to moderate pain, acetaminophen, aspirin, and nonsteroidal anti-inflammatory drugs (NSAIDs) may be sufficient. For moderate to severe pain, analgesics that include those agents combined with opioids may be helpful. Severe pain typically requires full opioid agonists.
Table 5-4. Recommended clinical approach to pain management. | ||
|---|---|---|
|
A. Acetaminophen and NSAIDs
Appropriate doses of acetaminophen may be just as effective as an analgesic and antipyretic as NSAIDs but without anti-inflammatory effects and without the risk of gastrointestinal bleeding or ulceration. Acetaminophen can be given at a dosage of 500 1000 mg orally every 6 hours, although it can be taken every 4 hours as long as the risk of hepatotoxicity is kept in mind. Hepatotoxicity is a concern at doses greater than 4 g/d long-term, and doses for older patients and those with liver disease generally should not exceed 2 g/d.
Aspirin (325 650 mg orally every 4 hours) is an effective analgesic, antipyretic, and anti-inflammatory medication. Gastrointestinal irritation and bleeding, bleeding from other sources, allergy, and an association with Reye's syndrome in children and teenagers limit its use.
Commonly used NSAIDs and their dosages are listed in Table 5-5. Like aspirin, the other NSAIDs are antipyretic, analgesic, and anti-inflammatory. NSAIDs inhibit prostaglandin synthesis, inhibiting platelet aggregation and consequently increasing the risk of gastrointestinal bleeding by 1.5 times normal. The risks of bleeding and nephrotoxicity from NSAIDs are both increased in elders. Gastrointestinal bleeding and ulceration may be decreased with the concurrent use of proton pump inhibitors (eg, omeprazole, 20 40 mg orally daily) or with the class of NSAIDs that inhibit only cyclooxygenase (COX)-2. Serious concerns about the safety of COX-2 inhibitors have led to the withdrawal of both rofecoxib and valdecoxib by their manufacturers; the Food and Drug Administration is conducting an ongoing review of this drug class. The only currently available COX-2 inhibitor is celecoxib (100 mg/d to 200 mg twice daily orally), which clinicians should use with caution in patients with cardiac disease because of its possible association with an increased risk of myocardial infarction. The NSAIDs, including COX-2 inhibitors, can lead to exacerbations of congestive heart failure and should be used with caution in patients with that disorder.
B. Opioid Medications
For many patients at the end of life, opioids are the mainstay of pain management. Opioids are appropriate for severe pain due to any cause. Opioid medications are listed in Table 5-6. Full opioid agonists such as morphine, hydromorphone, oxycodone, methadone, fentanyl, hydrocodone, and codeine are used most commonly. Hydrocodone and codeine are typically combined with acetaminophen or an NSAID. Short-acting formulations of oral morphine sulfate (starting dosage 4 mg orally every 3 4 hours), hydromorphone (1 mg orally every 3 4 hours), or oxycodone (5 mg orally every 3 4 hours) are useful for acute pain and as rescue treatment for patients experiencing pain that breaks through long-acting medications. For chronic stable pain, one should give sustained-release morphine (one to three times a day) or oxycodone (two or three times a day), methadone (three or four times a day), or transdermal fentanyl (starting dose 25 mcg every 3 days, with 24 40 hours required to achieve full analgesia).
Meperidine is not useful for chronic pain because it has a short half-life and a toxic metabolite that can cause irritability and seizures. Partial agonists such as buprenorphine are limited by a dose-related ceiling effect. Mixed agonist-antagonists such as pentazocine and butorphanol tartrate also have a ceiling effect and are contraindicated in patients already receiving full agonist opioids since they may reverse the pain control achieved by the full agonist and cause a withdrawal effect.
A useful technique for opioid management of chronic pain at the end of life is equianalgesic dosing (Table 5-6). The dosages of any full opioid agonists used to control pain can be translated into an equivalent dose of any other opioid. In this way, 24-hour opioid requirements and dosing regimens established initially using shorter-acting opioid medications can be translated into equivalent dosages of longer-acting medications or formulations. Cross-tolerance is often incomplete, however, so less than the full calculated equianalgesic dosage is generally administered initially when switching between opioid formulations. In addition, equianalgesic dosing for methadone is somewhat more complex and varies by dose.
While some clinicians and patients inexperienced with the management of severe chronic pain may feel more comfortable with combined nonopioid-opioid
P.76
P.77
P.78
P.79
P.80
agents, full agonist opioids are typically a better choice in patients with severe pain because the dose of opioid is not limited by the toxicities of the acetaminophen or NSAID component of combination preparations. There is no maximal allowable or effective dose for full opioid agonists. The dose should be increased to whatever is necessary to relieve pain, remembering that certain types of pain may respond better to agents other than opioids.
Table 5-5. Acetaminophen, COX-2 inhibitors, and useful nonsteroidal anti-inflammatory drugs. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 5-6. Useful opioid agonist analgesics. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
While physiologic tolerance is possible with opioids, failure of a previously effective opioid dose to adequately relieve pain is usually due to an increase in the underlying pain. In this case, for moderate unrelieved pain, the dosage of opioid can be increased by 25 50%. For severe unrelieved pain, a dosage increase of 50 100% may be appropriate. The frequency of dosing should be adjusted so that pain control is continuous. In addition, long-term dosing may be adjusted by adding the amount of short-acting opioid necessary for breakthrough pain over the preceding 24 hours to the long-acting medication dose. In establishing or reestablishing adequate dosing, frequent reassessment of the patient's pain and medication side effects is necessary.
As opioids are titrated upwards, increasing difficulty with the side effects of can be expected. Constipation is common and should be anticipated and prevented in all patients. The prophylaxis and management of opioid-induced constipation are outlined below.
Sedation can be expected with opioids, although tolerance to this effect typically develops within 24 72 hours at a stable dose. Sedation typically appears well before significant respiratory depression. If treatment for sedation is desired, dextroamphetamine (2.5 7.5 mg orally at 8 AM and noon) or methylphenidate (2.5 10 mg orally at 8 AM and noon) may be helpful. Some patients even use caffeinated beverages to help manage minor opioid sedation.
Although sedation is more common, patients may experience euphoria when first taking opioids or when the dosage is increased. However, tolerance to this effect often develops after a few days at a stable dose. At very high doses of opioids, multifocal myoclonus may develop. This symptom may resolve after lowering the dose or switching opioids. While waiting for the level of the offending medication to fall, low doses of lorazepam or dantrolene may be helpful for treating myoclonus.
Nausea due to opioids may occur with initiation of therapy and resolve after a few days. If it is severe or persistent, it can be treated with prochlorperazine, 10 mg orally or intravenously every 8 hours or 25 mg rectally every 6 hours. The most common cause of nausea in patients taking opioids is constipation.
Although clinicians may worry about respiratory depression with opioids, that effect is uncommon when a low dose is given initially and titrated upward slowly. Even patients with pulmonary disease can tolerate low-dose opioids, although they should be monitored carefully. Clinicians should not allow concerns about respiratory depression to prevent them from treating pain adequately.
True allergy (with urticaria) to opioids is rare. More commonly, patients will describe an intolerance due to side effects such as nausea, pruritus, or urinary retention in response to a particular opioid. If such symptoms develop, they can usually be relieved by lowering the dose or switching to another opioid.
C. Neuropathic Pain
It is essential when taking a patient's history to listen for such descriptions as burning, shooting, pins and needles, or electricity, and for pain associated with numbness. Such a history suggests neuropathic pain, which is treated with some medications not typically used for other types of pain. Opioids are effective for neuropathic pain as are tricyclic antidepressants [TCAs], gabapentin, tramadol, and the lidocaine patch. These five medications have been found to be effective in multiple randomized trials and are considered first-line agents in the pharmacologic management of neuropathic pain (Table 5-7). The TCAs are a good first choice and usually have an effect within days and at lower doses than are needed for an antidepressant effect. Desipramine, 10 150 mg/d orally, and nortriptyline, 10 150 mg/d orally, are good first choices as they cause less orthostatic hypotension and have fewer anticholinergic effects than amitriptyline. One can start with a low dosage (10 mg orally daily) and titrate upward every 4 or 5 days.
The anticonvulsant gabapentin can be used in the same dosages that are used to prevent seizures. Gabapentin can cause sedation, dizziness, ataxia, and gastrointestinal side effects and therefore should be started at low dosages of 100 300 mg orally three times a day. The dose of gabapentin can be titrated upward by 300 mg/d every 4 or 5 days to a dosage of 3600 mg/d or higher. Gabapentin is relatively safe in accidental overdosage and may be preferred over TCAs for a patient with a history of congestive heart failure or arrhythmia or if there is a risk of suicide. Gabapentin and morphine in combination are more effective at lower doses of each than as single agents. Carbamazepine can also be effective for neuropathic pain, but because it can cause bone marrow suppression, a complete blood count is obtained periodically in patients taking this drug.
The 5% lidocaine patch is effective in postherpetic neuralgia and may be effective in other types of neuropathic pain also. A new patch is applied to the painful region daily for up to 12 hours. Tramadol should be started at 50 mg orally daily and can be titrated up to 100 mg orally four times daily. Successful management of neuropathic pain often requires the use of more than one effective medication. Duloxetine (60 mg/d), a norepinephrine and serotonin reuptake inhibitor, is approved for the treatment of diabetic neuropathy.
D. Adjuvant Pain Medications and Treatments
If pain cannot be controlled without uncomfortable medication side effects, clinicians should consider using lower doses of multiple medications rather than larger doses of one or two medications as is common for neuropathic pain. For bone pain, the anti-inflammatory effect
P.81
of NSAIDs can be particularly helpful. Radiation therapy and bisphosphonates may also relieve bone pain. For some patients, such as those with pain from pancreatic cancer, a nerve block in this case, block of the celiac plexus can provide relief. Intrathecal pumps may be useful for patients with severe pain responsive to opioids but who require such large doses that systemic side effects such as sedation and constipation become limiting. Neurolysis, rhizotomy, or ablative surgery and neurosurgery may be tried in selected patients. Cannabinoids have not been proven to work as analgesics. Chemotherapeutic agents are sometimes used for symptom management with palliative intent.
Table 5-7. Pharmacologic management of neuropathic pain. | |||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||
Corticosteroids such as dexamethasone or prednisone can be helpful for patients with headache due to increased intracranial pressure, pain from spinal cord compression, metastatic bone pain, and neuropathic pain due to invasion or infiltration of nerves by tumor. Because of the side effects of long-term corticosteroid administration, they are most appropriate in patients with end-stage disease.
Nonpharmacologic Treatments
Nonpharmacologic therapies are also valuable in treating pain. Hot or cold packs, massage, and physical therapy can be helpful for musculoskeletal pain. Similarly, biofeedback, acupuncture, chiropractic, meditation, music therapy, cognitive behavioral therapy, guided imagery, cognitive distraction, and framing may be of help in treating pain. Because mood and psychological issues play an important role in the patient's perception of and response to pain, psychotherapy, support groups, prayer, and pastoral counseling can also help in the management of pain. Major depression, which may be instigated by chronic pain or may alter the response to pain, should be treated aggressively.
Dyspnea
Dyspnea is the subjective experience of difficulty in breathing and may be characterized by patients as tightness in the chest, shortness of breath, or a feeling of suffocation. Dyspnea is common among dying patients up to 50% of severely ill patients may experience severe dyspnea.
Treatment of dyspnea is usually first directed at the cause, which may be related to pneumonia, pulmonary embolism, pleural effusion, bronchospasm, tracheal obstruction, neuromuscular disease, restriction of movement of the chest or abdominal walls, cardiac ischemia, congestive heart failure, superior vena cava syndrome, or severe anemia.
At the end of life, dyspnea is often treated nonspecifically with opioids. Immediate-release morphine orally or intravenously treats dyspnea effectively and typically at doses lower than would be necessary for the relief of moderate pain. Nebulized morphine provides no advantage over other routes of administration and the mask used for administration may exacerbate dyspnea. Supplemental oxygen may be useful for the dyspneic patient who is hypoxic and may provide subjective benefit to other dyspneic patients as well. However, a nasal cannula and face mask are sometimes not well tolerated, and fresh air from a window or fan may provide relief. Judicious use of nonpharmacologic relaxation techniques such as
P.82
meditation and guided imagery may be beneficial for some patients. Benzodiazepines may be useful for the anxiety associated with dyspnea but do not appear to act directly to relieve dyspnea.
Nausea & Vomiting
Nausea and vomiting are common and distressing symptoms. As with pain, the management of nausea may be maximized by continuous dosing. An understanding of the four major inputs to the vomiting center may help direct treatment (see Chapter 14).
The chemoreceptor trigger zone may be stimulated by certain drugs (eg, opioids, NSAIDs), metabolic derangements, and chemotherapeutic agents. Vomiting associated with a particular opioid may be relieved by substitution with an equianalgesic dose of another opioid or a sustained-release formulation. In addition to the other dopamine receptor antagonist antiemetics listed in Table 14-2 that block the trigger zone, haloperidol (0.5 5 mg orally every 4 6 hours) is commonly used. Vomiting associated with chemotherapy may respond to agents such as ondansetron, granisetron, and dolasetron.
Vomiting may be due to stimulation of peripheral afferent nerves from the gut. Offering patients small amounts of food only when they are hungry may prevent nausea and vomiting. Nasogastric suction may provide rapid, short-term relief for vomiting associated with constipation, gastroparesis, or gastric outlet obstruction, with the addition of laxatives, prokinetic agents (only in the setting of partial not complete obstruction) metoclopramide (10 20 mg orally or intravenously four times a day), scopolamine (1.5-mg patch every 3 days), and high-dose corticosteroids as more definitive treatment. Treatment with high-dose corticosteroids (eg, dexamethasone, 20 mg orally or intravenously) or cyclizine (5 mg orally every 8 hours) may be useful for nausea and vomiting due to disease of intra-abdominal or pelvic organs.
Increased intracranial pressure may cause vomiting and may be relieved with high-dose corticosteroids or palliative cranial radiation. Vomiting due to disturbance of the vestibular apparatus may be treated with anticholinergic and antihistaminic agents (including diphenhydramine, 25 mg orally or intravenously every 8 hours, or scopolamine, 1.5-mg patch every 3 days).
Benzodiazepines can be effective in preventing the anticipatory nausea associated with chemotherapy but are otherwise not indicated for nausea and vomiting. Because they are sedating, these agents may increase the risk of aspiration in patients who vomit and therefore should rarely be used alone for the relief of nausea.
Finally, many patients find dronabinol (2.5 20 mg orally every 4 6 hours) helpful in the management of nausea and vomiting.
Constipation
Given the frequent use of opioids, poor dietary intake, and physical inactivity, constipation is a common problem among the dying. Clinicians must inquire about any difficulty with hard or infrequent stools. Constipation is an easily preventable and treatable cause of discomfort, distress, and nausea and vomiting (see Chapter 14).
Constipation may be prevented or relieved if patients can increase their activity and their intake of dietary fiber and fluids. Simple considerations such as privacy, undisturbed toilet time, and a bedside commode rather than a bedpan may be important for some patients.
For patients taking opioids, anticipating and preventing constipation is important. A prophylactic bowel regimen of stool softeners (docusate) and stimulants (bisacodyl or senna) should be started when opioid treatment is begun. Lactulose, sorbitol, magnesium citrate, and enemas can be added as needed (see Table 14-4).
Delirium & Agitation
Many terminally ill patients die in a state of delirium a disturbance of consciousness and a change in cognition that develops over a short time and is manifested by misinterpretations, illusions, hallucinations, disturbances in the sleep-wake cycle, psychomotor disturbances (eg, lethargy, restlessness), and mood disturbance (eg, fear, anxiety). Delirium complicated by myoclonus or convulsions at the end of life has been called terminal restlessness.
Careful attention to patient safety and nonpharmacologic strategies to help the patient remain oriented (clocks, calendars, a familiar environment, reassurance and redirection from caregivers) may be sufficient to prevent or manage minor delirium. Some delirious patients may be pleasantly confused, and a decision by the patient's family and the clinician not to treat delirium may be justified.
More commonly, however, delirium at the end of life is distressing to patients and family and requires treatment. Delirium may interfere with the family's ability to feel comforting to the patient and may prevent a patient from being able to recognize and report important symptoms.
While there are many reversible causes of delirium (see Chapter 25), identifying and correcting the underlying cause at the end of life is often simply a question of attention to the choice and dosing of psychoactive medications.
When the cause of delirium cannot be identified, treated, or corrected rapidly enough, delirium may be treated symptomatically with neuroleptics. Haloperidol (1 10 mg orally, subcutaneously, intramuscularly, or intravenously twice or three times a day) is used commonly, but extrapyramidal adverse effects may occur. Newer agents such as risperidone (1 3 mg orally twice a day) also may be helpful in delirium.
As an adjuvant to the above neuroleptics, especially in the setting of significant, unrelieved anxiety, benzodiazepines such as lorazepam (0.5 2 mg orally, sublingually,
P.83
subcutaneously, or intravenously every 4 6 hours) may be useful. When delirium is refractory to treatment and remains intolerable, sedation may be required to provide relief and may be achieved rapidly with midazolam (0.5 5 mg/h subcutaneously or intravenously) or barbiturates.
Bruera E et al: Nebulized versus subcutaneous morphine for patients with cancer dyspnea: a preliminary study. J Pain Symptom Manage 2005;29:613.
Dworkin et al: Advances in neuropathic pain: diagnosis, mechanisms, and treatment recommendations. Arch Neurol 2003; 60:1524.
Gilron I et al: Morphine, gabapentin, or their combination for neuropathic pain. N Engl J Med 2005;352:1324.
Hallenbeck J: Palliative care in the final days of life: they were expecting it at any time. JAMA 2005;293:2265.
Pantilat SZ et al: Palliative care for patients with heart failure. JAMA 2004;291:2476.
Other Specific Tasks of Caring
Nutrition & Hydration
Tube feedings do not prevent aspiration pneumonia, and there is debate about whether artificial nutrition prolongs life in the terminally ill. In fact, there has been a growing awareness of the potential medical benefits of forgoing unwanted or artificial nutrition and hydration (including tube feedings, parenteral nutrition, and intravenous hydration) at the end of life.
Eating without hunger and artificial nutrition are associated with a number of potential complications. Force feeding may cause nausea and vomiting in ill patients, and eating will lead to diarrhea in the setting of malabsorption. Nutrition may increase oral and airway secretions and the risk of choking, aspiration, and dyspnea. Nasogastric and gastrostomy tube feeding and parenteral nutrition impose risks of infection, epistaxis, pneumothorax, electrolyte imbalance, and aspiration as well as the need to physically restrain the delirious patient to prevent dislodgment of catheters and tubes.
Withholding nutrition at the end of life causes remarkably little hunger or distress. Ill people often have no hunger with total caloric deprivation, and the associated ketonemia produces a sense of well-being, analgesia, and mild euphoria. However, carbohydrate intake even in small amounts (such as that provided by 5% intravenous dextrose solution) blocks ketone production and may blunt the positive effects of total caloric deprivation.
Withholding hydration may lead to death in a few days to a month. The quality of life for those at the end of life may be adversely affected by supplemental hydration because of its contribution to oral and airway secretions (leading to aspiration or the death rattle ), polyuria, and the development or worsening of ascites, pleural or other effusions, and peripheral and pulmonary edema.
Although it is unclear to what extent withholding hydration at the end of life creates an uncomfortable sensation of thirst, any such sensation is usually relieved by simply moistening the dry mouth. Ice chips, hard candy, swabs, or a solution of equal parts nystatin solution, viscous lidocaine, diphenhydramine, and minted mouthwash are effective.
Individuals at the end of life have a right to refuse nutrition and hydration. However, providing or withholding oral food and water is not simply a medical decision because feeding may have profound social and cultural significance for patients, families, and clinicians themselves. Withholding supplemental enteral or parenteral nutrition and hydration challenges the assumption that offering food is an expression of compassion and love and invokes distressing images of starvation. In fact, individuals at the end of life who choose to forgo nutrition and hydration are unlikely to suffer from hunger or thirst. Family and friends can be encouraged to express their love and caring in ways other than intrusive attempts at forced feeding or hydration.
Withdrawal of Curative efforts
Requests from appropriately informed and competent patients or their surrogates for withdrawal of life-sustaining interventions must be respected. The clinician receiving such requests should recognize and explore the significance of this change in health care goals. Alternatively, clinicians may determine unilaterally that further intervention is medically inappropriate eg, continuing renal dialysis in a patient dying of multiorgan failure. In such cases, the clinician's intention to withdraw a specific intervention should be communicated to the patient and family. If differences of opinion exist about the appropriateness of what is being done, the assistance of an institutional palliative care service or ethics committee should be sought.
Limitation of life support prior to death is an increasingly common practice in intensive care units. The withdrawal of life-sustaining interventions such as mechanical ventilation must be approached carefully to avoid needless patient suffering and distress for those in attendance. Clinicians should educate the patient and family about the expected course of events and the difficulty of determining the precise timing of death after withdrawal of support. Sedative and analgesic agents should be administered to ensure patient comfort even at the risk of respiratory depression or hypotension. Scopolamine (10 mcg/h subcutaneously or intravenously, or a 15-mg patch every 3 days) or atropine (1% ophthalmic solution, 1 or 2 drops sublingually as often as every hour) can be used for controlling airway secretions and the resultant death rattle.
P.84
A guideline for withdrawal of mechanical ventilation is provided in Table 5-8.
Table 5-8. Guidelines for withdrawal of mechanical ventilation. | ||
|---|---|---|
|
Psychological, Social, & Spiritual Issues
Dying is not exclusively or even primarily a biomedical event. It is an intimate personal experience with profound psychological, interpersonal, and existential meanings. For many people at the end of life, the prospect of impending death stimulates a deep and urgent assessment of their identity, the quality of their relationships, and the meaning and purpose of their existence.
Psychological Challenges
In 1969, Elisabeth K bler-Ross identified five psychological stages or patterns of emotions that patients at the end of life may experience: denial and isolation, anger, bargaining, depression, and acceptance. Not every patient will experience all these emotions, and not necessarily in an orderly progression. In addition to these five stages are the perpetual challenges of anxiety and fear of the unknown. Simple information, listening, assurance, and support may help patients with these psychological challenges. In fact, patients and families rank emotional support as one of the most important aspects of good end-of-life care. Psychotherapy and group support may be beneficial as well.
Despite the significant emotional stress of facing death, clinical depression is not normal at the end of life and should be treated. Cognitive and affective signs of depression (such as hopelessness) may help distinguish depression from the low energy and other vegetative signs common with end-stage illness. Although traditional antidepressant treatments such as selective serotonin reuptake inhibitors are effective, more rapidly acting medications such as dextroamphetamine or methylphenidate may be particularly useful when the end of life is near.
Social Challenges
At the end of life, patients should be encouraged to discharge personal, professional, and business obligations. This might include completing important work or personal projects, distributing possessions, writing a will, and making funeral and burial arrangements. The prospect of death often prompts patients to examine the quality of their interpersonal relationships, including the relationship with the clinician. Dying may intensify a patient's need to feel cared for by the doctor, highlighting the clinician's obligation of nonabandonment and the need for clinician empathy and compassion. Concern about estranged relationships or unfinished business with significant others and interest in reconciliation may become paramount at this time. At the end of life, even healthy interpersonal relationships must reach completion (Table 5-9).
Spiritual Challenges
Spirituality is the attempt to understand or accept the underlying meaning of life, one's relationships to oneself and other people, one's place in the universe, and the possibility of a higher power in the universe. Spirituality is distinguished from particular religious practices or beliefs and is generally considered a universal human concern.
Perhaps because of an inappropriately exclusive attention to the biologic challenge of forestalling death or perhaps from feelings of discomfort or incompetence, clinicians frequently ignore their patients' spiritual concerns or reflexively refer these
P.85
important issues to psychiatrists or other caretakers (nurses, social workers, clergy). However, the existential challenges of dying are central to the well-being of people at the end of life and are the proper concern of clinicians. Within a biologic, psychosocial, and spiritual model of medical care, clinicians may work to provide more than simple physical comfort and control of bothersome symptoms. Clinicians can help dying patients by providing care to the whole person by providing physical comfort and social support and by helping patients discover their own unique meaning in the world and an acceptance of death as a part of life.
Table 5-9. Five statements often necessary for the completion of important interpersonal relationships. | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||
Unlike physical ailments such as infections and fractures, which usually require a clinician's intervention to be treated, the patient's spiritual concerns often require only a clinician's attention, listening, and witness. Clinicians might choose to inquire about the patient's spiritual concerns and ask whether the patient wishes to discuss them. For example, asking, How are you within yourself? communicates that the clinician is interested in the patient's whole experience and provides an opportunity for the patient to share perceptions about his or her inner life. Questions that might constitute an existential review of systems are presented in Table 5-10.
Attending to the spiritual concerns of patients calls for listening to their stories. Story-telling gives patients the opportunity to verbalize what is meaningful to them and to leave something of themselves behind the promise of being remembered. Story-telling may be facilitated by suggesting that the patient share his or her life story with family members, record it on audio or video tape, assemble a photo album, organize a scrapbook, or write an autobiography.
Table 5-10. An existential review of systems. | ||
|---|---|---|
|
While dying may be a period of inevitable loss of physical functioning, the end of life also offers an opportunity for psychological, interpersonal, and spiritual development. Individuals may grow even achieve a heightened sense of well-being or transcendence in the process of dying. Through listening, support, and presence, clinicians may help foster this learning and be a catalyst for this transformation. Rather than thinking of dying simply as the termination of life, clinicians and patients may be guided by a developmental model of dying that recognizes a series of lifelong developmental tasks and landmarks and allows for growth at the end of life.
Chochinov HM: Dignity-conserving care a new model for palliative care: helping people feel valued. JAMA 2002;287: 2253.
Lo B et al: Discussing religious and spiritual issues at the end of life: a practical guide for physicians. JAMA 2002;287:749.
Monroe MH et al: Primary care physician preferences regarding spiritual behavior in medical practice. Arch Intern Med 2003;163:2751.
Murphy LM et al: Percutaneous endoscopic gastrostomy does not prolong survival in patients with dementia. Arch Intern Med 2003;163:1351.
Rubenfeld GD: Principles and practice of withdrawing life-sustaining treatments. Crit Care Clin 2004;20:435.
Tasks after Death
After the death of a patient, the clinician is called upon to perform a number of tasks, both required and recommended. The clinician must plainly and directly inform the family of the death. Providing words of sympathy and reassurance, time for questions and initial grief, and a quiet private room for the family at this time is appropriate and much appreciated.
The Pronouncement & Death Certificate
In the United States, state policies direct clinicians to confirm the death of a patient in a formal process called pronouncement. The clinician must verify the absence of spontaneous respirations and cardiac activity. A note describing these findings and the time of death is entered in the patient's chart. In many states, when a patient whose death is expected dies outside of the hospital (at home or in prisons, for example) nurses may be authorized to report the death over the telephone to a physician who must then sign the death certificate within 24 hours. For traumatic deaths, some states allow emergency medical technicians to
P.86
pronounce a patient dead at the scene based on clearly defined criteria and with physician telephonic or radio supervision.
While the pronouncement may often seem like an awkward and unnecessary formality, clinicians may use this time to reassure the patient's loved ones at the bedside that the patient died peacefully and that all appropriate care had been given. Both clinicians and families may use the ritual of the pronouncement as an opportunity to begin to process emotionally the death of the patient.
Physicians are legally required to accurately report the underlying cause of death on the death certificate. This reporting is important both for patients' families (for insurance purposes and the need for an accurate family medical history) and for the epidemiologic study of disease and public health. Physicians are untrained in and unskilled at correctly completing death certificates. The physician should be specific about the major cause of death (eg, decompensated cirrhosis ) and its contributory cause (eg, hepatitis B and hepatitis C infections and chronic alcoholic hepatitis ) as well as any associated conditions (eg, acute renal failure ) and not simply put down cardiac arrest as the cause of death.
Autopsy & Organ Donation
Discussing the options and obtaining consent for autopsy and organ donation with patients themselves prior to death is usually the best practice. This advances the principle of patient autonomy and lessens the responsibilities of distressed family members during the period immediately following the death. After a patient dies, however, designated organ transplant personnel are more successful than the treating clinicians at obtaining consent for organ donation from surviving family members. Federal regulations now require that a designated representative of an organ procurement organization approach the family about organ donation. Most people in the United States support the donation of organs for transplants. Currently, however, organ transplantation is severely limited by the availability of donor organs. Many potential donors and the families of actual donors experience a sense of reward in contributing, even through death, to the lives of others.
Clinicians must be sensitive to ethnic and cultural differences in attitudes about autopsy and organ donation. Patients or their families should be reminded of their right to limit autopsy or organ donation in any way they choose. Pathologists can perform autopsies without interfering with funeral plans or the appearance of the deceased.
The results of an autopsy may help surviving family members (and clinicians) understand the exact cause of a patient's death and foster a sense of closure. A clinician-family conference to review the results of the autopsy provides a good opportunity for clinicians to assess how well families are grieving and to answer questions. Despite the advantages of conducting postmortem examinations, autopsy rates have fallen drastically to less than 15% today. Families report refusing autopsies out of fear of disfigurement of the body or delay of the funeral or say they were simply not asked. They allow autopsies in order to advance medical knowledge, to identify the exact cause of their loved one's death, and to be reassured that appropriate care was given. Routinely addressing these issues when discussing autopsy may help increase the autopsy rate; the most important mistake is the failure to ask for permission to perform it.
Follow-Up & Grieving
Proper care of patients at the end of life includes following up with surviving family members after the patient has died. Following up enables the clinician to assess how families are grieving, to reassure them about the nature of normal grieving, and to identify complicated grief or depression. Clinicians can recommend support groups and counseling as needed. A card or telephone call from the clinician to the family days to weeks after the patient's death (and perhaps on the anniversary of the death) allows the clinician to express concern for the family and the deceased.
After a patient dies, the clinician too may need to grieve. Although clinicians may be relatively unaffected by the deaths of some patients, other deaths may cause distressing feelings of sadness, loss, and guilt. These emotions should be recognized as the first step toward processing them or preventing them in the future.
For clinicians, grieving the loss of a patient is normal. Each clinician may find personal or communal resources that help with the process of grieving. Shedding tears, the support of colleagues, time for reflection, and traditional or personal mourning rituals all may be effective. Attending the funeral of a patient who has died can be a satisfying personal experience that is almost universally appreciated by families and that may be the final element in caring well for people at the end of life.
Lakkireddy DR et al: Death certificate completion: how well are physicians trained and are cardiovascular causes overstated? Am J Med 2004;117:492.
Marchand L et al: Death pronouncements: using the teachable moment in end-of-life care residency training. J Palliat Med 2004;7:80.
Penson RT et al: When does the responsibility of our care end: bereavement. Oncologist 2002;7:251.
Shear K et al: Treatment of complicated grief: a randomized controlled trial. JAMA 2005;293:2601.
EAN: 2147483647
Pages: 49