4. Sigma factor controls binding to DNA
9.4 Sigma factor controls binding to DNA |
| Key terms defined in this section |
| Sigma factor is the subunit of bacterial RNA polymerase needed for initiation; is the major influence on selection of binding sites (promoters). |
The holoenzyme (α2ββ′σ) can be separated into two components, the core enzyme (α2ββ′) and the sigma factor (the σ polypeptide). Only the holoenzyme can initiate transcription. Sigma factor ensures that bacterial RNA polymerase binds in a stable manner to DNA only at promoters. The sigma "factor" is released when the RNA chain reaches 8 V9 bases, leaving the core enzyme to undertake elongation. Core enzyme has the ability to synthesize RNA on a DNA template, but cannot initiate transcription at the proper sites (496).
The core enzyme has a general affinity for DNA, in which electrostatic attraction between the basic protein and the acidic nucleic acid plays a major role Any (random) sequence of DNA that is bound by core polymerase in this general binding reaction is described as a loose binding site. No change occurs in the DNA, which remains duplex. The complex at such a site is stable, with a half-life for dissociation of the enzyme from DNA ~60 minutes. Core enzyme does not distinguish between promoters and other sequences of DNA.
Sigma factor introduces a major change in the affinity of RNA polymerase for DNA. The holoenzyme has a drastically reduced ability to recognize loose binding sites Xthat is, to bind to any general sequence of DNA. The association constant for the reaction is reduced by a factor of ~104, and the half-life of the complex is <1 second. So sigma factor destabilizes the general binding ability very considerably.
But sigma factor also confers the ability to recognize specific binding sites. The holoenzyme binds to promoters very tightly, with an association constant increased from that of core enzyme by (on average) 1000 times and with a half-life of several hours.
The specificity of holoenzyme for promoters compared to other sequences is ~107, but the association constant can be quoted only as an average, because there is wide variation in the rate at which the holoenzyme binds to different promoter sequences. This is an important factor in determining the efficiency of an individual promoter in initiating transcription. The binding constants extend from ~1012 to ~106, reflecting promoter strengths that support initiation frequencies of ~1/sec (rRNA genes) to ~1/30 min (the lacI promoter).
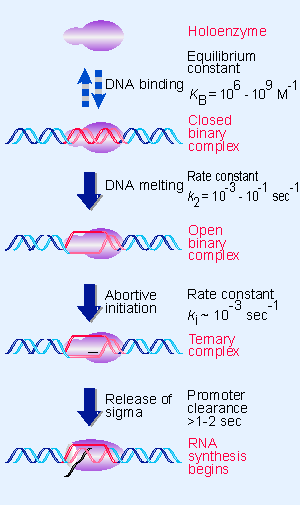 |
Figure 9.9 RNA polymerase passes through several steps prior to elongation. A closed binary complex is converted to an open form and then into a ternary complex. |
We are now in a position to describe the stages of transcription in terms of the interactions between different forms of RNA polymerase and the DNA template. The initiation reaction can be described by the parameters that are summarized in Figure 9.9:
- The holoenzyme Ppromoter reaction starts by forming a closed binary complex. "Closed" means that the DNA remains duplex. Because the formation of the closed binary complex is reversible, it is usually described by an equilibrium constant (KB). There is a wide range in values of the equilibrium constant for forming the closed complex.
- The closed complex is converted into an open complex by "melting" of a short region of DNA within the sequence bound by the enzyme. The series of events leading to formation of an open complex is called tight binding. For strong promoters, conversion into an open binary complex is irreversible, so this reaction is described by a rate constant (k2). This reaction is fast. Sigma factor is involved in the melting reaction (see 9.7 Substitution of sigma factors may control initiation).
- The next step is to incorporate the first two nucleotides; then a phosphodiester bond forms between them. This generates a ternary complex that contains RNA as well as DNA and enzyme. Formation of the ternary complex is described by the rate constant ki; this is even faster than the rate constant k2. Further nucleotides can be added without any enzyme movement to generate an RNA chain of up to 9 bases. After each base is added, there is a certain probability that the enzyme will release the chain. This comprises an abortive initiation, after which the enzyme begins again with the first base. A cycle of abortive initiations usually occurs to generate a series of short (2 V9 base) oligonucleotides.
- When initiation succeeds, the enzyme releases sigma and makes the transition to the elongation ternary complex of core polymerase PDNA Pnascent RNA. The critical parameter here is how long it takes for the polymerase to leave the promoter so another polymerase can initiate. This parameter is the promoter clearance time; its minimum value of 1 V2 sec establishes the maximum frequency of initiation as <1 event per second. The enzyme then moves along the template, and the RNA chain extends beyond 10 bases.
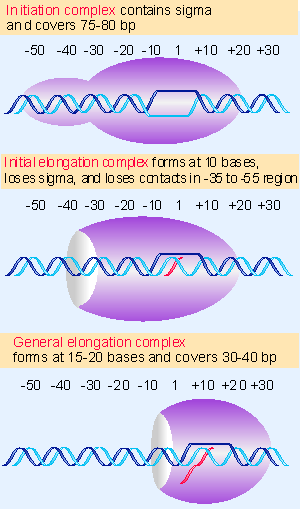 |
Figure 9.10 RNA polymerase initially contacts the region from -55 to +20. When sigma dissociates,the core enzyme contracts to -30; when the enzyme moves a few base pairs, it becomes more compactly organized into the general elongation complex. |
When RNA polymerase binds to DNA, the elongated dimension of the protein extends along the DNA, but some interesting changes in shape occur during transcription. Transitions in shape and size identify three forms of the complex, as illustrated in Figure 9.10 (501):
- When RNA polymerase holoenzyme initially binds to DNA, it covers some 75 V80 bp, extending from V55 to +20. (The long dimension of RNA polymerase (160 Å) could cover ~50 bp of DNA in extended form, which implies that binding of a longer stretch of DNA must involve some bending of the nucleic acid.)
- The release of sigma results in a change in the shape of the RNA polymerase at the transition from initiation to elongation. The loss of sigma is associated with the loss of contacts in the V55 to V35 region, leaving only ~60 bp of DNA covered by the enzyme. This corresponds with the concept that the more upstream part of the promoter is involved in initial recognition by RNA polymerase, but is not required for the later stages of initiation (see later).
- When the RNA chain extends to 15 V20 bases, the enzyme makes a further transition, to form the complex that undertakes elongation; now it covers 30 V40 bp (depending on the stage in the elongation cycle). It is not known whether the change at the start of elongation involves a significant alteration in the shape of the enzyme or whether the DNA is released into a less compact form.
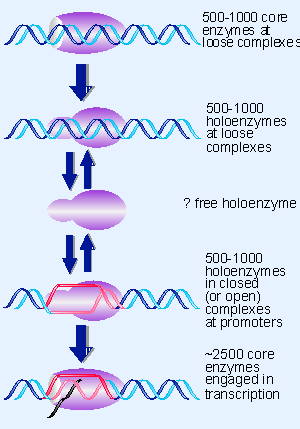 |
Figure 9.11 Core enzyme and holoenzyme are distributed on DNA, and very little RNA polymerase is free. |
How is RNA polymerase distributed in the cell? A (somewhat speculative) picture of the enzyme’s situation is depicted in Figure 9.11:
- Excess core enzyme exists largely as closed loose complexes, because the enzyme enters into them rapidly and leaves them slowly. There is very little, if any, free core enzyme.
- There is enough sigma factor for about one third of the polymerases to exist as holoenzymes, and they are distributed between loose complexes at nonspecific sites and binary complexes (mostly closed) at promoters.
- About half of the RNA polymerases consist of core enzymes engaged in transcription.
- How much holoenzyme is free? We do not know, but we suspect that the amount is very small.
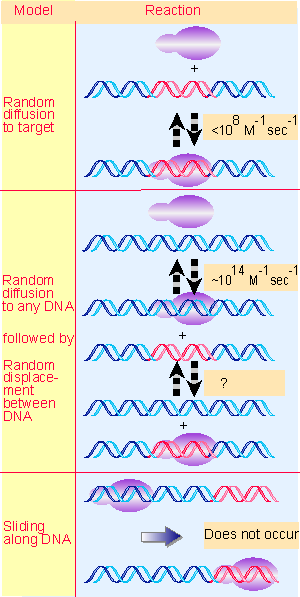 |
Figure 9.12 How does RNA polymerase find target promoters so rapidly on DNA? |
RNA polymerase must find promoters within the context of the genome. Suppose that a promoter is a stretch of ~60 bp; how is it distinguished from the 4 106 bp that comprise the E. coli genome? Figure 9.12 illustrates the principles of three models.
The simplest model is to suppose that RNA polymerase moves by random diffusion. Holoenzyme very rapidly associates with, and dissociates from, loose binding sites. So it could continue to make and break a series of closed complexes until (by chance) it encounters a promoter. Then its recognition of the specific sequence would allow tight binding to occur by formation of an open complex.
For RNA polymerase to move from one binding site on DNA to another, it must dissociate from the first site, find the second site, and then associate with it. Movement from one site to another is limited by the speed of diffusion through the medium. Diffusion sets an upper limit for the rate constant for associating with a 60 bp target of <108 M V1 sec V1. But the actual forward rate constant for some promoters in vitro appears to be ~108 M V1 sec V1, at or above the diffusion limit. If this value applies in vivo, the time required for random cycles of successive association and dissociation at loose binding sites is too great to account for the way RNA polymerase finds its promoter.
RNA polymerase must therefore use some other means to seek its binding sites. The process could be speeded up if the initial target for RNA polymerase is the whole genome, not just a specific promoter sequence. By increasing the target size, the rate constant for diffusion to DNA is correspondingly increased, and is no longer limiting.
If this idea is correct, a free RNA polymerase binds DNA and then remains in contact with it. How does the enzyme move from a random (loose) binding site on DNA to a promoter? The most likely model is to suppose that the bound sequence is directly displaced by another sequence. Having taken hold of DNA, the enzyme exchanges this sequence with another sequence very rapidly, and continues to exchange sequences until a promoter is found. Then the enzyme forms a stable, open complex, after which initiation occurs. The search process becomes much faster because association and dissociation are virtually simultaneous, and time is not spent commuting between sites. Direct displacement can give a "directed walk," in which the enzyme moves preferentially from a weak site to a stronger site.
Another idea supposes that the enzyme slides along the DNA by a one-dimensional random walk, being halted only when it encounters a promoter. However, there is no evidence that RNA polymerase (or other DNA-binding proteins) can function in this manner.
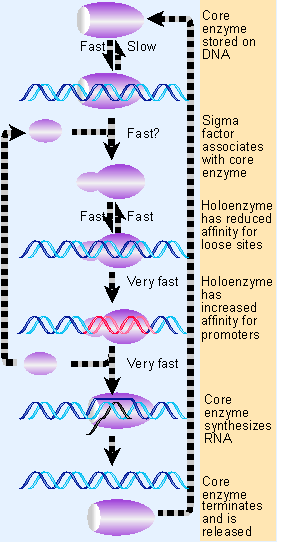 |
Figure 9.13 Sigma factor and core enzyme recycle at different points in transcription. |
RNA polymerase encounters a dilemma in reconciling its needs for initiation with those for elongation. Initiation requires tight binding only to particular sequences (promoters), while elongation requires close association with all sequences that the enzyme encounters during transcription. Figure 9.13 illustrates how the dilemma is solved by the reversible association between sigma factor and core enzyme.
Sigma factor is involved only in initiation. It is released from the core enzyme when abortive initiation is concluded and RNA synthesis has been successfully initiated. Free sigma factor becomes immediately available for use by another core enzyme. We do not know whether sigma is released as a consequence of overcoming abortive initiation, or whether instead it is the release of sigma factor that ends abortive initiation and allows elongation to commence.
Release of sigma leaves the core enzyme on DNA. The core enzyme in the ternary complex is bound very tightly to DNA. It is essentially "locked in" until elongation has been completed. When transcription terminates, the core enzyme is released. It is then "stored" by binding to a loose site on DNA. It must find another sigma factor in order to undertake a further cycle of transcription.
Core enzyme has a high intrinsic affinity for DNA, which is increased by the presence of nascent RNA. But its affinity for loose binding sites is too high to allow the enzyme to distinguish promoters efficiently from other sequences. By reducing the stability of the loose complexes, sigma allows the process to occur much more rapidly; and by stabilizing the association at tight binding sites, the factor drives the reaction irreversibly into the formation of open complexes. To avoid becoming paralyzed by its specific affinity for the promoter, the enzyme releases sigma, and thus reverts to a general affinity for all DNA, irrespective of sequence, that suits it to continue transcription.
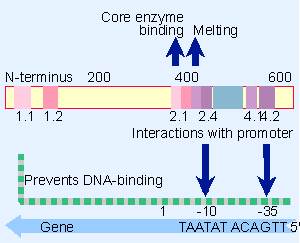 |
Figure 9.19 A map of the E. coli s70 factor identifies conserved regions. Regions 2.1 and 2.2 contact core polymerase, 2.3 is required for melting, and 2.4 and 4.2 contact the -10 and -35 promoter elements. The N-terminal region prevents 2.4 and 4.2 from binding to DNA in the absence of core enzyme. |
What is responsible for the ability of holoenzyme to bind specifically to promoters? Sigma factor has domains that recognize the promoter DNA. As an independent polypeptide, sigma does not bind to DNA, but when holoenzyme forms a tight binding complex, σ contacts the DNA in the region upstream of the startpoint. This difference is due to a change in the conformation of sigma factor when it binds to core enzyme. The N-terminal region of free sigma factor suppresses the activity of the DNA-binding region; when sigma binds to core, this inhibition is released, and it becomes able to bind specifically to promoter sequences (see also Figure 9.19 later). The inability of free sigma factor to recognize promoter sequences may be important: if σ could freely bind to promoters, it might block holoenzyme from initiating transcription. We do not know what role the core subunits play in promoter recognition.
| Research | |
| 496: | Travers, A. A. and Burgess, R. R. (1969). Cyclic reuse of the RNA polymerase sigma factor. Nature 222, 537-540. |
| 501: | Krummel, B. and Chamberlin, M. J. (1989). RNA chain initiation by E. coli RNA polymerase. Structural transitions of the enzyme in early ternary complexes Biochemistry. Biochemistry 28, 7829-7842. |