2 - The Out-of-Hospital Management of Acute Heart Failure
Editors: Peacock, W. Frank
Title: Short Stay Management of Heart Failure, 1st Edition
Copyright 2006 Lippincott Williams & Wilkins
> Table of Contents > 2 - The Out-of-Hospital Management of Acute Heart Failure
2
The Out-of-Hospital Management of Acute Heart Failure
Marvin A. Wayne
Vincent N. Mosesso Jr.
Introduction
Acute heart failure (AHF) is one of only two cardiovascular diseases with an increasing prevalence; the other is atrial fibrillation. Five million Americans have the disease, and more than 500,000 are newly diagnosed each year. AHF is a major disease of our aging population1 because most hospitalizations for AHF are of patients older than 65 years.2 Heart failure (HF) results in more than 2 million hospitalizations annually and accounts for about 3% of the national health care budget.1 Further, it is Medicare's largest single disease expenditure. Approximately 300,000 deaths are annually related to HF.3
Heart failure, and its acute presentation, AHF, is a complex disease that includes at least four clinical syndromes: exacerbation of chronic HF, hypertensive crisis, acute pulmonary edema (APE), and cardiogenic shock.4 Of these four syndromes, APE and cardiogenic shock are the two most serious. Although the major clinical manifestations in both are a combination of decreased peripheral perfusion and pulmonary congestion, they differ in pathophysiology and hemodynamic changes. Accordingly, these syndromes require different therapeutic approaches.
APE, the most common clinical manifestation of AHF,5 is a life-threatening respiratory emergency usually occurring in the out-of-hospital setting. The overall prehospital mortality rate for APE in a retrospective Italian study has been reported to be 8%.6 Although similar data for the United States are not readily available, a favorable outcome for AHF is dependent on rapid assessment and treatment initiated in the out-of-hospital setting.
Pathogenesis of Ape
APE can be of a cardiogenic or noncardiogenic etiology. In the former, pulmonary edema results from increased microvascular hydrostatic pressure, whereas in the latter edema arises from increased pulmonary capillary
P.6
permeability. The end result is identical in both cases: an excessive accumulation of extravascular lung fluid.7 The primary cause of cardiogenic APE is cardiac dysfunction (Table 2-1). Noncardiogenic APE may be caused by several diverse events or diseases, which include systemic or pulmonary infection, trauma, septic shock, toxic inhalation, or aspiration of gastric contents.8 Because both types of APE have the same clinical manifestations dyspnea, diaphoresis, decreased lung compliance, anxiety, and increased shunt fraction distinguishing between the two can be extremely difficult.7
TABLE 2-1 Precipitating Causes of Acute Cardiogenic Pulmonary Edema (APE) | ||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||
APE is often difficult to distinguish clinically from an exacerbation of chronic obstructive pulmonary disease (COPD) or other acute pulmonary disorders. The misdiagnosis of AHF in the out-of-hospital setting has been documented to be 23% in one study9 and as high as 32% in another.10 The need for the correct identification of precipitating events, and the rapid initiation of appropriate treatment, is critical to achieve a positive outcome. Inappropriate therapy, as a result of misdiagnosis, may result in harm to the patient. Hoffman and Reynolds9 reported that adverse effects were more common in misdiagnosed patients. Untoward effects included (a) respiratory depression in patients receiving morphine, (b) hypotension and bradycardia in patients receiving both morphine and nitroglycerin, and (c) arrhythmia associated with hypokalemia in patients receiving furosemide.
Initiating events or conditions, including myocardial ischemia, hypertensive crisis, fluid excess, medication noncompliance, diet, and overexertion, may trigger AHF. Each may set in motion a vicious cycle of events that results in cardiogenic APE. The key components of this cycle, outlined in Figure 2-1, all involve left ventricular (LV) dysfunction.11 A marked increase in systemic vascular resistance in conjunction with impaired myocardial
P.7
contractility, from either systolic or diastolic dysfunction, results in pulmonary edema. This increase in vascular resistance leads to an increase in LV diastolic pressure resulting in increased pulmonary venous pressure. This increases hydrostatic pressure, which then forces fluid to leak out of the pulmonary capillaries into the pulmonary interstitial space and alveoli, producing edema. As edema worsens, so does oxygen diffusion, and thus oxygen saturation drops and further compromises cardiac contractility, creating a positive feedback circuit.12
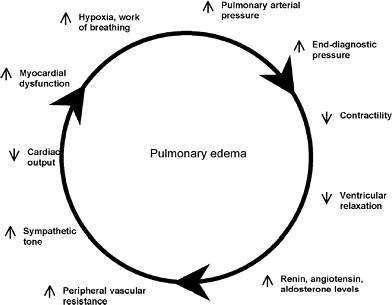 |
FIGURE 2-1 Processes involved in pulmonary edema. Cycle may begin at any point but once begun is self-perpetuating. (From Sacchetti AD, Harris RH. Acute cardiogenic pulmonary edema. What's the latest in emergency treatment? Postgrad Med 1998;103:145 147, with permission.) |
Field Assessment
Assessment begins with a rapid, focused history and physical examination of the patient. This includes patient history, recent illness, prescribed medications, medication compliance, and diet. Together this constitutes an important first step in the field diagnosis of AHF (Table 2-2). Critical elements of the physical examination include accurate determination of vital signs. AHF/APE is often associated with marked elevation in systolic blood pressure. Prehospital providers, even in the absence of peripheral edema, should strongly consider cardiogenic pulmonary edema in patients presenting with acute respiratory distress, hypoxemia, tachypnea, rales or wheezing, and marked hypertension. Such patients often have histories of poorly controlled hypertension and/or prior cardiac disease. Blood pressure of greater than 180/120 mm Hg is common in this setting and is a good sign of reversibility. In these patients, a rapid reduction in blood pressure often produces prompt relief of respiratory distress. Marked
P.8
hypertension associated with acute respiratory distress and wheezing, particularly in elderly patients without a history of asthma or pulmonary infection, is strongly suggestive of APE. Such a presumptive diagnosis may be supported by the presence of cardiovascular medications and the absence of respiratory medications, such as metered-dose inhalers. Even when these facts are present, out-of-hospital personnel should always consider alternate etiologies such as pulmonary embolism, pneumonia, asthma, and drug overdose before diagnosing patients as having APE. Although rhythm strips and standard 12-lead electrocardiograms (ECGs) are useful in identifying arrhythmia and/or acute coronary syndrome, they are insensitive and nonspecific for diagnosing AHF.13,14 Furthermore, these ECGs and rhythm strip tracings may be prone to misinterpretation.15 Recently, a new type of ECG has been developed. This system uses an
P.9
acoustic sensor at leads V3 and V4 to diagnose the presence of S3 and/or S4 heart sounds (Audicor Inovice Medical Systems, Portland, OR). The presence of an S3 in patients older than 45 years is highly specific for the presence of HF and may aid in the diagnosis of AHF.15,16
TABLE 2-2 Diagnosis of Congestive Heart Failure | |||||||||
|---|---|---|---|---|---|---|---|---|---|
|
In addition to these diagnostic tests, a number of other diagnostic aids have been developed to improve accuracy in the evaluation and diagnosis of AHF. Although not currently used in the prehospital environment, a rapid bedside assay of blood levels of B-type natriuretic peptide, a neurohormone secreted mainly by the cardiac ventricles in response to volume expansion and pressure overload, is useful in establishing or excluding the diagnosis of AHF in patients with acute dyspnea in the emergency department (ED).16,17,18,19,20 and 21 Application of such testing in the out-of-hospital environment may be a logical extension and further aid in diagnosis. Currently, noninvasive cardiac output (NICO) devices, such as impedance cardiography,22 have also been suggested as diagnostic tools, but their complexities and cost have to date precluded their out-of-hospital use.
TABLE 2-3 Management of Acute Congestive Heart Failure: Overview | ||
|---|---|---|
|
Management of Ape
Fluid accumulation in the lungs associated with APE, until recently, was attributed to excess accumulation of total body fluid. Accordingly, treatment of APE was aimed at removing excess fluid by promoting massive diuresis. However, this explanation did not reconcile with APE cases without an increase in total body water. The current explanation is that APE results from fluid redistribution within the body whereby a part of the intravascular volume is redistributed to the lungs as a consequence of increased intravascular pressure.12 Primary objectives for the treatment of AHF and associated APE are to reduce pulmonary capillary pressure, to redistribute pulmonary fluid, and to improve forward flow.11,12 These goals may be achieved by reducing LV preload and afterload, providing ventilatory and inotropic supports, and identifying and treating the underlying etiology of the syndrome (Table 2-3). It should be recognized that these treatment measures are intended for APE patients who are normotensive or
P.10
hypertensive and not those who are hypotensive. The latter comprises cardiogenic shock arising secondary to severe LV systolic dysfunction; these patients may need inotropic or mechanical cardiac support, and the treatment of these critically ill patients is beyond the scope of this review.
Reduction of LV Preload
The initial effort to reduce the pulmonary congestion in patients presenting with APE should be to reduce the pressure and volume of blood flow to the pulmonary vasculature. This may be accomplished by dilating the venous capacitance system. This will result in decreased blood return to the right ventricle (preload), hence reducing blood flow to the pulmonary vascular bed. The net result is a reduction in LV preload, which then allows the LV output to more closely match inflow from the pulmonary system.11 Pharmacologic therapy to reduce LV preload includes the use of nitrates primarily and, to a more limited extent, morphine and loop diuretics such as furosemide.
Nitrates
Nitroglycerin and related drugs at low dosages are primarily venodilators effective in decreasing pulmonary artery pressure. Intracellularly, they react with and convert sulfhydryl groups to S-nitrosothiols and nitric oxide. These reactive groups then activate the enzyme guanylate cyclase, which catalyzes the formation of cyclic guanosine monophosphate (cGMP). This nucleotide induces the reentry of calcium back into the sarcoplasmic reticulum of vascular smooth muscle thereby causing its relaxation.23
Nitroglycerin is currently the vasodilator agent of choice for the reduction of LV preload in the field. It is fast acting, efficient, and easy to administer.11,12 Nitroglycerin's effectiveness in reducing mortality in patients with APE in the prehospital setting has been demonstrated by Bertini (1997).6 In this study, even hypotensive patients (systolic blood pressure <100 mm Hg) were found to respond positively to nitroglycerin. Likewise, Hoffman and Reynolds9 compared a number of prehospital management protocols for APE and concluded that nitroglycerin was beneficial, whereas morphine and furosemide had no additive effect when combined with nitroglycerin and were occasionally deleterious. The beneficial hypotensive effect of nitroglycerin must be closely monitored so that the reduction in blood pressure does not deleteriously reduce LV preload and the ability to produce adequate cardiac output. Thus, a potential disadvantage of nitroglycerin is that it can lead to excessive hypotension,8 particularly in patients without adequate preload [e.g., hypovolemia and inferior wall myocardial infarction (MI) with significant right ventricular (RV) involvement].
Morphine
Although morphine has been used for decades to treat acute MI, unstable angina, and AHF, few clinical trials have demonstrated its effectiveness in acute congestive heart failure (CHF). Its popularity in treating pulmonary edema arose because of its vasodilatory and antianxiety
P.11
effects, although morphine's vasodilatory effects are transient and are the result of histamine release.11 Recently, concerns have been raised over the use of morphine in treating acute CHF in the ED. A retrospective study of the ED management of APE and intensive care unit (ICU) admissions showed that morphine administered in the ED was associated with significant increases in ICU admissions and the need for endotracheal intubations (ETIs) when compared with treatment with sublingual captopril.24 Additionally, a prospective study of morphine use in prehospital APE treatment showed that the drug was minimally effective as single therapy or in combination with nitrates.9 Furthermore, the effects of morphine in depressing respiration and the central nervous system may be deleterious in misdiagnosed patients.9
Furosemide
Furosemide has been a mainstay of treatment for APE since the 1960s, although its effectiveness has been examined in only a few studies. Its primary mechanism of action involves the inhibition of sodium reabsorption in the ascending limb of Henle's loop in the renal medulla. This results in an increased excretion of salt and water in urine. The net effect of this action is a lowering of plasma volume, a decrease in LV preload, and a decrease in pulmonary congestion. These effects are beneficial in patients presenting with pulmonary volume overload.25 In addition to its diuretic effects, furosemide also induces neurohumoral changes. These include both vasodilation (by promoting renal prostaglandin E2 and atrial natriuretic peptide secretion) and vasoconstricting effects.26 The latter, via the feedback loop, can result in peripheral elevation of mean arterial pressure, LV pressure, heart rate, and systemic vascular resistance through enhancement of the renin-angiotensin system (RAS). Stroke volume index and pulmonary capillary wedge pressure initially decrease but subsequently increase after the RAS enhancement (usually within 15 minutes). The latter effects are not beneficial in the treatment of AHF particularly in the absence of volume overload.11,12 Furthermore, misdiagnosis of AHF and subsequent inducement of inappropriate diuresis can lead to increased morbidity and mortality in patients with other conditions such as pneumonia, sepsis, or COPD.9,25
Combined Drug Therapies with Nitroglycerin, Furosemide, and Morphine
Nitrates are frequently combined with loop diuretics in treating pulmonary edema. A complex, randomized, prospective clinical study from Israel investigated the efficacy and safety of these drugs in treating patients presenting with severe pulmonary edema in the prehospital setting.27 This study concluded that intravenous (IV) nitrates administered as repeated high-dose boluses (3 mg every 5 minutes) after a low dose (40 mg) of furosemide was associated with lower ETI and MI rates than the administration of low-dose nitrates (1 mg per hour, increased by 1 mg per hour every 10 minutes) and high-dose furosemide (80 mg every 15 minutes). A prospective observational study on the use of sublingual nitroglycerin in the prehospital setting in cases of presumed MI or CHF analyzed
P.12
treatment-related adverse events in 300 patients. Only four patients experienced adverse events, most of which were bradycardic-hypotensive reactions, and all recovered subsequently.28
A retrospective case review evaluated outcomes of 57 patients presumed to have prehospital APE who were treated in the field with combinations of nitroglycerin, furosemide, and/or morphine.9 Although only a small study, any combination treatment including nitroglycerin was associated with both subjective and objective (respiratory and heart rates, blood pressure, respiratory distress, mental status) improvement. Combination treatment with furosemide and morphine without nitroglycerin, on the other hand, resulted in a substantial number of patients not responding to treatment and some actually deteriorating. Ultimately, 23 of 57 (47%) patients in this study were found not to have pulmonary edema.
A larger retrospective case series evaluated outcomes in 493 patients receiving prehospital nitroglycerin, furosemide, and/or morphine versus no treatment for CHF. Mortality was significantly reduced in the more critical patients receiving any prehospital drug treatment compared with no treatment (5% vs. 33%, p <0.01) as well as in the entire treated patient population (6.7% vs. 15.4%, p <0.01).29
Reduction of LV Afterload
A variety of pharmacologic agents, including nitroglycerin at higher doses, angiotensin-converting enzyme (ACE) inhibitors, nitroprusside, dobutamine, and dopamine, may be useful in the reduction of LV afterload.
Nitrates At Higher Doses
High-dose nitrates can reduce both preload and afterload and potentially increase cardiac output.30 Because many CHF patients present with very elevated arterial and venous pressures, frequent doses of nitrates may be required to control blood pressure and afterload. Some patients develop tolerance to nitroglycerin, but this is not of concern in the prehospital environment. Another concern with high-dose nitrates is that certain patients are very sensitive to even normal doses and may experience marked hypotension. These are typically patients with tenuous preload status (e.g., preexisting hypovolemia or significant RV infarction in the setting of inferior wall MI). It is therefore critical to monitor blood pressure during high-dose nitrate therapy.
Ace Inhibitors
ACE inhibitors play a primary role in chronic CHF therapy and have multiple therapeutic advantages for treating APE. These include reducing both preload and afterload, increasing splanchnic flow, decreasing LV diastolic dysfunction, reducing sodium retention, and reducing sympathetic stimulation. Captopril (Capoten) is an ACE inhibitor that has been studied in the prehospital setting.11 When a standard tablet is administered sublingually, it rapidly dissolves and has an onset of action of less than 10 minutes. Clinical effects are seen within 15 minutes, with peak effects within 30 minutes.31,32 A retrospective study of 181 patients with APE treated in the ED examined the relationship between pharmacologic
P.13
treatments and rates of ICU admissions.24 Patients in this study were treated with captopril (26%), nitroglycerin (81%), morphine (49%), and/or loop diuretics (73%). Patients receiving captopril had decreased rates of ICU admissions and ETIs as well as shorter ICU stays. A prospective, placebo-controlled, randomized study evaluated the addition of sublingual captopril to the standard treatment regimen (oxygen, nitrates, morphine, and furosemide) in patients brought to the ED with APE.32 Using a clinical APE distress score for assessment, the addition of captopril was found to significantly reduce distress scores over the first 40 minutes compared with placebo. This study indicated that certain features of ACE inhibitors make them attractive for field use, including ease of sublingual administration, fast onset of action, and low cost.11,31,33 However, captopril use may be associated with potential concerns,34 which include occasional hypotension and a variable duration of effect in comparison to nitrates.
In addition to the sublingual route, ACE inhibitors may also be administered intravenously. Enalaprilat maleate (Vasotec IV) is the only IV ACE inhibitor currently available in the United States,33 and its efficacy and safety in the treatment of pulmonary edema and CHF have been demonstrated in two small randomized trials.35,36
Nitroprusside
APE patients presenting with severe hypertension and those refractory to nitrate and ACE inhibitor treatments may be candidates for treatment with nitroprusside sodium.25 However, the need to continuously monitor blood pressure, with a carefully titrated continuous infusion, and the requirement of glass containers shielded from light preclude its utility in the field environment.24
Ventilatory Support
Patients with acute CHF may be treated with a spectrum of ventilatory support modalities based on the patient's clinical condition and comorbid factors. Initial treatment includes oxygen therapy to maintain oxygen saturation of at least 92% to 93% and the use of inhaled bronchodilators when bronchospasm is evident.25 True bronchospasm may be triggered by interstitial edema, especially in patients with underlying reactive airway disease. A new single isomer 2 agonist for inhalation, levalbuterol HCl (Xopenex, Sepracor Pharmaceutical, Marlborough, MA), may cause less tachycardia than racemic albuterol, of particular concern in patients with HF or cardiac ischemia.
In severe respiratory failure (ineffective respiratory effort, hypoxemia, hypercarbia), assisted ventilation is needed. Traditionally this has been accomplished in tandem with ETI. However, ETI is a challenge to accomplish effectively in noncomatose, nonparalyzed patients with the limited resources and personnel usually available in the field setting. Further, ETI is associated with various infectious (e.g., nosocomial pneumonia, sinusitis) and noninfectious (e.g., barotrauma; oral, nasal, or laryngeal trauma; respiratory
P.14
muscle weakness; prolonged weaning) complications.37,38,39 and 40 To avoid these complications and lengthy ICU stays, noninvasive ventilatory support is being increasingly used. ETI remains necessary when altered mental status requires airway protection or when other patient characteristics prevent the successful application of noninvasive positive pressure ventilation (NIPPV).
Noninvasive Positive Pressure Ventilation
Noninvasive positive pressure ventilation (NIPPV), traditionally used for COPD and asthma,41,42,43,44 and 45 is now considered an effective adjunct treatment of AHF/APE.46,47,48,49 and 50 The therapeutic effect of noninvasive pressure support lies in its ability to increase intra-alveolar pressure. This shifts the flow of fluid back into the pulmonary capillaries and thereby reduces pulmonary congestion. NIPPV decreases the work of breathing and thereby decreases myocardial demand. Two different methods of providing NIPPV are used: continuous positive airway pressure (CPAP), which provides a constant level of positive pressure applied throughout inspiration and exhalation, and bilevel positive airway pressure (BiPAP), which allows provision of higher pressure during inspiration than expiration.51
Only a few prehospital studies involving CPAP have been conducted. A Swedish study compared outcomes after treatment for AHF during two time periods: one period of standard therapy consisting of nitroglycerin, furosemide, or both (first period) and one period of intensified treatment with nitroglycerin, furosemide, and CPAP (second period).5 The use of drug therapy and CPAP in patients increased dramatically from the first period to the second period: nitroglycerin from 4% to 68%, furosemide from 13% to 84%, and CPAP from 4% to 91%. Although a greater percentage of patients in the second group had fulminant pulmonary edema (FPE) on ambulance arrival (60% vs. 78%, p < 0.001), at admission to the hospital there was a significant reduction in the percentage with FPE during the second period (93% vs. 76.1%, p < 0.001). There was also a significantly lower mean level of serum creatine kinase MB in patients in the second group, implying less myocardial damage. Although there was an improvement in symptoms during transport and less myocardial damage, mortality remained high with no significant difference between treatment groups.
A small prospective case-series analysis on the prehospital use of CPAP by trained paramedics in 19 patients with cardiogenic pulmonary edema showed that none of the patients required field intubation and that hemoglobin oxygen saturation increased from a mean of 83.3% to 95.4% after CPAP administration via a face mask.10 Two patients intolerant of CPAP required ETI on ED arrival and an additional five patients required ETI within 24 hours. There were no adverse events related to CPAP therapy. These results demonstrate that field use of CPAP is feasible but that certain obstacles need to be overcome. In particular, the authors noted that the paramedic's lack of experience with this therapy led to problems with achieving good mask fit and titrating pressure levels.
P.15
BiPAP has been investigated as an alternative to CPAP in a number of conditions but has shown a significant advantage over CPAP only in patients whose respiratory failure is due to COPD exacerbation.52 A number of individual studies reported some success with BiPAP53,54 and some problems, including increased rates of MI,55,56 associated with its use in treating acute CHF.
In an out-of-hospital study of patients with presumed CHF, emergency medical services (EMS) personnel considered the use of BiPAP to be safe and judged this method to improve dyspnea and respiratory distress in their patients.57 Although oxygen saturation was significantly greater for the BiPAP plus conventional treatment group, compared with the conventional treatment group, treatment times, length of hospital stay, intubation rate, and death rates were not significantly different between the groups.
Of the two types of noninvasive ventilatory support, there is good supporting evidence for the effectiveness of CPAP. New technology appears to make field implementation of CPAP simpler and more efficacious and overcomes some of the earlier concerns about its use. Greater experience of field providers should also lead to better outcomes because this therapy is not only patient dependent but operator dependent as well. In the case of BiPAP, the risk-benefit ratio is conflicting in the literature. In addition, the existing technology for BiPAP is suboptimal for out-of-hospital use. Further field trials investigating the implementation of these modalities and testing technologic refinements are needed.
Summary
This review focuses on the importance of understanding that the pathogenesis of APE is related to intravascular fluid redistribution rather than to primary volume overload (Figure 2-1). Management of suspected APE begins with correct assessment and management of underlying causes of elevated ventricular filling pressures and continues with improving oxygenation with the application of ventilatory support, reduction of LV preload and afterload with nitroglycerin and inotropic support when necessary. Care must be taken with drugs such as furosemide and morphine because patient outcomes can be adversely affected when administered for conditions mimicking APE. EMS personnel must assiduously seek hallmarks of acute CHF as opposed to other medical conditions associated with acute dyspnea. Sophisticated, rapid tools such as quantitative B-type natriuretic peptide (BNP) assays and noninvasive hemodynamic devices may one day provide greater diagnostic accuracy in the field. A guide based on clinical evidence for treating prehospital patients suffering from acute CHF is suggested (Table 2-4).
Nitrates are recommended as first-line therapy for APE in the field with symptom resolution as the primary treatment goal. Nitrates provide both subjective and objective improvement (with high-dose, repeat sublingual or spray administration). Blood pressure is an important gauge of
P.16
P.17
effective nitrate dosing. Endpoints should be primarily guided by the patient's level of dyspnea and oxygen saturation and avoidance of hypotension. In patients with systolic blood pressures less than 90 to 100 mm Hg, nitroglycerin should not be routinely administered.
TABLE 2-4 Classification for Treatment | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||
The use of loop diuretics such as furosemide requires careful consideration. Although furosemide is beneficial in transiently decreasing the pulmonary capillary wedge pressure, it can also increase the systemic vascular resistance. The primary concern involves the increased morbidity and mortality associated with prehospital administration of diuretics in conditions that mimic APE and in patients with APE who do not have an excess of total body water. This may occur in patients with new onset HF, such as in association with an acute MI. The use of diuretics in combination with nitrates has also been proved to have no early clinical benefit. In the absence of peripheral edema or other evidence of excess total body water (such as documented acute weight gain), routine diuretic administration should be avoided.
Evidence for the use of ACE inhibitors is not currently sufficient to recommend their use in the prehospital care of APE. Although they have an essential role in chronic HF, the lack of supporting data and potential disadvantages including hypotension, adverse interaction with aspirin, decrease in glomerular filtration rate, and longer duration of action preclude endorsing their use in APE at this time.
Bronchodilator use is appropriate when wheezing is the result of bronchospasm but should not preclude delivery of other specific therapy for CHF, such as nitroglycerin or CPAP. It is not clear whether CPAP or BiPAP provides a consistent outcome advantage. Initial field studies have been promising, but issues such as the need for specialized training in mask adjustment and pressure titration, high-volume oxygen consumption requirement for operation, or suboptimal portability due to the need for an electrical power source present logistical obstacles. New technology may make the field administration of CPAP simpler and more efficacious.
Conclusion
APE is a common and often life-threatening condition encountered by prehospital emergency medical personnel. Patients with this condition must receive rapid, accurate assessment and aggressive treatment. High-dose nitrates represent the out-of-hospital treatment of choice, whereas diuretics and morphine should be reserved for select patient groups. More data are needed on the efficacy and safety of ACE inhibitors to justify their use in the field. CPAP has been shown to be effective, but more experience and refinement of delivery systems for the prehospital environment are needed. Logistical delivery issues also exist for BiPAP, and there is currently less convincing evidence of its safety in this setting. Emerging diagnostic assays and tools offer promise of fast and accurate diagnosis of CHF. Finally, transport of APE patients should be matched with the cardiovascular care resources of receiving facilities to optimize chances of survival.
P.18
Acknowledgment
Portions of this chapter are reprinted with the permission of the National Association of EMS Physicians from Mosesso VN Jr, Dunford J, Blackwell T, Griswell JK. Prehospital therapy for acute congestive heart failure: state of the art. Prehosp Emerg Care 2003;7:13 23.
References
1. Nohria A, Lewis E, Stevenson LW. Medical management of advanced heart failure. JAMA 2002;287:628 640.
2. Croft JB, Giles WH, Pollard RA, et al. Heart failure survival among older adults in the United States: a poor prognosis for an emerging epidemic in the Medicare population. Arch Intern Med 1999;159:505 510.
3. Hunt SA, Baker DW, Chin MH, et al. ACC/AHA guidelines for the evaluation and management of chronic heart failure in the adult: executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to revise the 1995 Guidelines for the Evaluation and Management of Heart Failure). J Am Coll Cardiol 2001;38:2101 2113.
4. Cotter G, Moshkovitz Y, Milovanov O, et al. Acute heart failure: a novel approach to its pathogenesis and treatment. Eur J Heart Failure 2002;4:227 234.
5. Gardtman M, Waagstein L, Karlsson T, Herlitz J. Has an intensified treatment in the ambulance of patients with acute severe left heart failure improved the outcome? Eur J Emerg Med 2000;7:15 24.
6. Bertini G, Giglioli C, Biggeri A, et al. Intravenous nitrates in the prehospital management of acute pulmonary edema. Ann Emerg Med 1997;30:493 499.
7. Guntupalli KK. Acute pulmonary edema. Cardiol Clin 1984;2:183 200.
8. Goldman. Cecil textbook of medicine, 21st ed. Philadelphia: WB Saunders, 2000.
9. Hoffman JR, Reynolds S. Comparison of nitroglycerin, morphine and furosemide in treatment of presumed prehospital pulmonary edema. Chest 1987; 92:586 593.
10. Kosowsky JM, Stephanides SL, Branson RD, Sayre MR. Prehospital use of continuous positive airway pressure (CPAP) for presumed pulmonary edema: a preliminary case series. Prehosp Emerg Care 2001;5:190 196.
11. Sacchetti AD, Harris RH. Acute cardiogenic pulmonary edema. What's the latest in emergency treatment? Postgrad Med 1998;103:145 147.
12. Cotter G, Kaluski E, Moshkovitz Y, et al. Pulmonary edema: new insight on pathogenesis and treatment. Curr Opin Cardiol 2001;16:159 163.
13. Mulrow C, Lucey C, Farnett L. Discriminating causes of dyspnea through the clinical examination. J Gen Intern Med 1993;8:383 392.
14. Schmitt B, Kushner M, Wiener S. The diagnostic usefulness of history of the patient with dyspnea. J Gen Intern Med 1986;1:386 393.
15. Little B, Ho KJ, Scott L. Electrocardiogram and rhythm strip interpretation by final year medical students. Ulster Med J 2001;70:108 110.
16. Marcus GM, Gerger IL, McKeown BH, et al. Association between phonocardiographic third and fourth heart sounds and objective measures of left ventricular function. JAMA 2005;293:2238 2244.
17. Peacock WF, Collins SP, Linsdsell CJ, et al. Gender and heart sounds in decompensated heart failure. Acad Emerg Med 2005;12(5):55(abst).
18. Maisel AS, Krishnaswamy P, Nowak RM, et al. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med 2002;347:161 167.
P.19
19. Maisel A. B-type natriuretic peptide in the diagnosis and management of congestive heart failure. Cardiol Clin 2001;19:557 571.
20. Dao Q, Krishnaswamy P, Kazanegra R, et al. Utility of B-type natriuretic peptide in the diagnosis of congestive heart failure in an urgent-care setting. J Am Coll Cardiol 2001;37:379 385.
21. Morrison LK, Harrison A, Krishnaswamy P, et al. Utility of a rapid B-natriuretic peptide assay in differentiating congestive heart failure from lung disease in patients presenting with dyspnea. J Am Coll Cardiol 2002;39:202 209.
22. Tabbibizar R, Maisel A. The impact of B-type natriuretic peptide levels on the diagnoses and management of congestive heart failure. Curr Opin Cardiol 2002;17:340 345.
23. Lee SC, Stevens TL, Sandberg SM, et al. The potential of brain natriuretic peptide as a biomarker for New York Heart Association class during the outpatient treatment of heart failure. J Card Fail 2002; 8:149 154.
24. Teboul A, Gaffinel A, Meune C, et al. Management of acute dyspnea: use and feasibility of brain natriuretic peptide (BNP) assay in the prehospital setting. Resuscitation 2004;61:91 96.
25. Marx J, Hockberger R, Walls R. Rosen's emergency medicine: concepts and clinical practice, 5th ed. St. Louis: Mosby, 2002.
26. Ventura HO, Pranulis MF, Young C, Smart FW. Impedance cardiography: a bridge between research and clinical practice in the treatment of heart failure. Congest Heart Fail 2000;6:94 102.
27. Kukovetz WR, Holzmann S. Mechanisms of nitrate-induced vasodilation and tolerance. Eur J Clin Pharmacol 1990;38:9.
28. Sacchetti A, Ramoska E, Moakes ME, et al. Effect of ED management on ICU use in acute pulmonary edema. Am J Emerg Med 1999;7:571 574.
29. Packer M. Neurohormonal interactions and adaptations in congestive heart failure. Circulation 1988;77:721.
30. Cotter G, Metzkor E, Kaluski E, et al. Randomised trial of high-dose isosorbide dinitrate plus low-dose furosemide versus high-dose furosemide plus low-dose isosorbide dinitrate in severe pulmonary oedema. Lancet 1998;351:389 393.
31. Wuerz R, Swope G, Meador S, et al. Safety of prehospital nitroglycerin. Ann Emerg Med 1994;23:31 36.
32. Northridge D. Frusemide or nitrates for acute heart failure? Lancet 1996;347:667 668.
33. Leeman M, Deguate JP. Invasive hemodynamic evaluation of sublingual captopril and nifedipine in patients with arterial hypertension after abdominal aortic surgery. Crit Care Med 1995;23:847.
34. Barnett J, Zink KM, Touchon RC. Sublingual captopril in the treatment of acute heart failure. Curr Ther Res 1991;49:274 281.
35. Hamilton RJ, Carter WA, Gallagher EJ. Rapid improvement of acute pulmonary edema with sublingual captopril. Acad Emerg Med 1996;3:205 212.
36. Ahmed A. Interaction between aspirin and angiotensin-converting enzyme inhibitors: should they be used together in older adults with heart failure? J Am Geriatr Soc 2002;50:1293 1296.
37. Annane D, Bellissant E, Pussard E, et al. Placebo-controlled, randomized, double-blind study of intravenous enalaprilat efficacy and safety in acute cardiogenic pulmonary edema. Circulation 1996;94:1316 1324.
38. Podbregar M, Voga G, Horvat M, et al. Bolus versus continuous low dose of enalaprilat in congestive heart failure with acute refractory decompensation. Cardiology 1999;91:41 49.
39. Colucci WS. Nesiritide for the treatment of decompensated heart failure. J Card Fail 2001;7:92 100.
40. Pingleton SK. Complications of acute respiratory failure. Am Rev Respir Dis 1988; 137:1463 1493.
P.20
41. Colice GL, Stukel TA, Dain B. Laryngeal complications of prolonged intubation. Chest 1989;96:877 884.
42. Craven DE, Steger KA. Epidemiology of nosocomial pneumonia: new perspectives on an old disease. Chest 1995;108:1S 16S.
43. Schnapp LM, Chin DP, Szaflarski N, et al. Frequency and importance of barotrauma in 100 patients with acute lung injury. Crit Care Med 1995;23:272 278.
44. Meduri GU, Turner RE, Abou-Shala N. Noninvasive positive pressure ventilation via face mask: first-line intervention in patients with acute hypercapnic and hypoxemic respiratory failure. Chest 1996;109:179 193.
45. Brochard L, Mancebo J, Wysocki M, et al. Noninvasive ventilation for acute exacerbations of chronic obstructive pulmonary disease. N Engl J Med 1995;333:817 822.
46. Bernstein AD, Holt AW, Vedig AE, et al. Treatment of severe cardiogenic pulmonary edema with continuous positive airway pressure delivered by face mask. N Engl J Med 1991;325:1825 1830.
47. Kramer N, Meyer TJ, Meharg J, et al. Randomized, prospective trial of noninvasive positive pressure ventilation in acute respiratory failure. Am J Respir Crit Care Med 1995;151:1799 1806.
48. Meduri GU, Cook TR, Turner RE, et al. Noninvasive positive pressure ventilation in status asthmaticus. Chest 1996;110:767 774.
49. Meduri GU, Conoscenti CC, Menashe P, et al. Noninvasive face mask ventilation in patients with acute respiratory failure. Chest 1989;95:865 870.
50. Pennock BE, Crashaw L, Kaplan PD. Noninvasive nasal mask ventilation for acute respiratory failure: institution of a new therapeutic technology for routine use. Chest 1994;105:441 444.
51. Baratz DM, Westbrook PR, Shah PK, et al. Effect of nasal continuous positive airway pressure on cardiac output and oxygen delivery in patients with congestive heart failure. Chest 1992;102:1397 1401.
52. Takeda S, Nejima J, Takano T, et al. Effect of nasal continuous positive airway pressure on pulmonary edema complicating acute myocardial infarction. Jpn Circ J 1998;62: 553 558.
53. Kelly AM, Georgakas C, Bau S, Rosengarten P. Experience with the use of continuous positive airway pressure (CPAP) therapy in the emergency management of acute severe cardiogenic pulmonary oedema. Aust N Z J Med 1997;27:319 322.
54. Keenan SP, Kernerman PD, Cook DJ, et al. Effect of noninvasive positive pressure ventilation on mortality in patients admitted with acute respiratory failure: a meta-analysis. Crit Care Med 1997;25:1685 1692.
55. Pang D, Keenan SP, Cook DJ, Sibbald WJ. The effect of positive pressure airway support on mortality and the need for intubation in cardiogenic pulmonary edema: a systematic review. Chest 1998;114:1185 1192.
56. Mehta S, Jay GD, Woolard RH, et al. Randomized, prospective trial of bilevel versus continuous positive airway pressure in acute pulmonary edema. Crit Care Med 1997;25:620 628.
57. Masip J, Betbese AJ, Paez J, et al. Non-invasive pressure support ventilation versus conventional oxygen therapy in acute cardiogenic pulmonary oedema: a randomised trial. Lancet 2000;356:2126 2132.
EAN: 2147483647
Pages: 18